Abstract
Background
Programmed cell death 1 (PD1) inhibitors have recently shown promising anti-cancer effects in a number of solid tumor types. A predictive biomarker to this class of drugs has not been clearly identified; however, overexpression of the PD1 ligand (PD-L1) has shown particular promise in lung adenocarcinoma. In this study, we explore the staining characteristics, prevalence, and clinico-molecular correlates of PD-L1 overexpression in pancreatic ductal adenocarcinoma (PDAC).
Methods
A tissue microarray (TMA) was constructed from cases of resected PDAC. PD-L1 immunohistochemistry (IHC) was performed using the SP142 primary antibody. Immunohistochemical assessment for deficient mismatch repair status (MMRd), CD3 and CD8 were performed. All biomarkers were assessed independently by two anatomical pathologists and consensus achieved on all cases. Survival analysis was performed using three thresholds (> = 1%, >5% and >10%) for tumor cell membrane staining.
Results
Two-hundred fifty-two cases were included in the TMA and evaluable by IHC. Thirty-one (12%), 17 (7%), 12(5%) cases were positive at percentage cut offs of >0, >5, and >10% respectively. Increased PD-L1 expression was associated with inferior prognosis (p = 0.0367). No statistically significant association was identified between PD-L1 status and MMR status or tumor infiltrating lymphocytes.
Conclusions
This data suggests that there is an inverse relationship between PD-L1 expression and disease specific survival times in resected PDAC. Consequently, this association may represent a phenotype where increased PD-L1 expression has an effect on tumor biology and could therefore identify a subgroup where PD1 blockade could have enhanced effectiveness.
Keywords: Pancreatic cancer, Programmed cell death 1 ligand, DNA mismatch repair, Tumor-infiltrating lymphocytes, Biomarkers, Immuno-oncology
Background
Pancreatic ductal adenocarcinoma (PDAC) ranks fourth for overall cancer-related death with over forty-thousand estimated deaths in 2015 in the United States. The five-year survival rate is 26% in resectable disease and drops to 2% if unresectable. Surgical resection is only attempted in 20% of cases [1].
Inhibitors of the programmed cell death 1 (PD1) signalling axis have yielded improved survival benefits for a number of solid tumor types. Large randomized clinical trials have been successful in treating melanoma, non-small cell lung cancer, and renal cell carcinoma [2, 3]. Three phase 1/2 drug trials are ongoing involving treatment of PDACs with immunotherapy (NCT02583477, NCT02305186, NCT02452424). To date, no biomarker has been established to predict benefit from PD1-axis inhibition for this disease [4].
PD-1 is an inhibitory receptor expressed by T cells and other immune cell types. It plays an important role in immune suppression when activated by its ligand (PD-L1). The latter is physiologically expressed by normal tissue and can occasionally be aberrantly expressed by tumor cells as a means for evading immune destruction [4–8]. Blockade of the PD-1/PD-L1 interaction promotes T-cell response against tumor cells [3, 9].
The response to PD-1/PD-L1 inhibition has been mixed in various malignancies such as colorectal, prostatic and pancreatic adenocarcinomas and is exemplified in the results of a study by Brahmer et al. which failed to show an objective response to anti-PD-L1 therapy in 14 patients with pancreatic cancer [3, 10]. In those cases, the use of biomarkers may have been useful in the identification of patients who are more likely to respond to PD1-axis inhibition. Mismatch repair (MMR) status has been shown to be predictive in colorectal carcinoma [11] and PD-L1 expression by immunohistochemistry (IHC) may be useful in lung and bladder carcinomas [12, 13]. However, no cut-off has been uniformly defined for PD-L1 expression that would trigger the use of PD-L1 inhibitors in PDAC. Current clinical trials often use 1% [14] but evidence suggests that higher cut-points may optimize patient stratification for PD-L1 therapies [15]. PD-1 expression in tumor infiltrating immune cells, the direct target of nivolumab, has shown, unlike tumor PD-L1 expression, only borderline association with clinical outcome to PD-1 blockade [16]. Other methods to predict response to immune checkpoint inhibitors have also been investigated, including immune cell infiltration, hypermutation signature, and gene expression linked to chemokine expression [17–19], but are yet to be validated in prospective clinical trials.
In this study, we explore the prevalence of PD-L1 expression in PDAC using IHC and compare this to clinical characteristics, including MMR status and tumor infiltrating lymphocytes and examine if an association with clinical outcome exists.
Methods
Ethical approval and a waiver of consent for research on this retrospective cohort was obtained from the University of British Columbia Clinical Research Ethics Board (H12–03484).
Sample identification and TMA construction
A tissue microarray was constructed using duplicate 0.6 mm cores from the epithelial component of all available, resected, pathologically confirmed pancreatic ductal adenocarcinomas derived from the archives of the Vancouver Coastal Health Region between 1995 and 2014. All patients received primary surgery with a subset receiving adjuvant chemotherapy with a pyrimidine nucleoside analog. Cores for the tissue microarray were obtained from areas of tumor as determined by routine microscopy on hematoxylin and eosin-stained sections. Cases were excluded if they lacked clinical follow-up data or if clinico-pathologic variables were lacking.
Immunohistochemical staining of PD-L1 and mismatch repair markers
Immunohistochemistry was performed on 4-μm-thick formalin-fixed paraffin-embedded sections of tissue microarrays. PD-L1 immunohistochemistry was performed at the Deely Research Centre at the British Columbia Cancer Agency using the Intellipath FLX autostainer (Biocare) platform. Mismatch repair, CD3 and CD8 immunohistochemistry was performed in the clinical laboratory of Vancouver General Hospital using the Ventana Discovery XT and the Ventana Benchmark XT automated system (Ventana Medical Systems, Tucson, AZ).
For PD-L1, slides were incubated with the clone SP142 (Spring Bioscience, Pleasanton, USA) at 1/100 dilution in Da Vinci Green diluent at room temperature for 30 min. Slides were then washed and incubated with Mach2 Rabbit-HRP polymer for 30 min at room temperature and detected with IP DAB chromogen for 5 min. Nuclei were counterstained with a 1/10 dilution of CAT hematoxylin then slides were again washed, air dried and coverslipped with Ecomount. The antibody clone was selected based on its strong concordance to three other PD-L1 clones and RNA in situ hybridization (ISH) in NSCLC [19].
For MMR stains, slides were incubated with MLH1 (mouse monoclonal antibody, 1:50 dilution, cat#: NCL-L- MLH1, clone ID:ES05; Leica Microsystems, New- castle, UK), MSH2 (mouse monoclonal antibody, 1:1000 dilution, cat#: 286 M-16, clone ID:G219–1129; Cellmarque, Rocklin, CA), MSH6 (rabbit monoclonal antibody, 1:200 dilution, cat#: CLAC-0047, clone ID: EP49; Cedarlane Corporation, Burlington, ON, Canada), and PMS2 (rabbit monoclonal antibody, 1:20 dilution, cat#: CLAC-0049, clone ID:EP51; Cedarlane Corporation) for 32 min at room temperature. For the slides to be stained for PMS2 were additionally prepped with the Epitomics DAB prep kit before antibody incubation.
Antibodies were detected using the Ventana DABMap kit, counterstained with hematoxylin and treated with a proprietary bluing agent (Ventana). Positive and negative controls are performed as part of the routine clinical quality assurance; in addition to the external quality control program (Canadian Immunohistochemistry Quality Control (cIQc), a provider of proficiency testing for Canadian clinical laboratories).
Interpretation of Immunohistochemical stains
PD-L1 status was assessed independently by two anatomical pathologists (BSS and DG) and consensus achieved on all cases. Positivity was evaluated by H-Score, a combination of staining intensity and percentage of tumor cell staining. Staining intensity was scored as 0 (negative), 1 (weak), 2 (moderate), or 3 (strong) based on membranous localization and each score multiplied by the percentage of cells (0% - 100%) staining. Therefore, H-scores ranged from 0 to 300. To account for potential intra-tumoral heterogeneity, the mean of both cores was used to generate the score for each case.
Mismatch repair (MMR) was quantified as per Riazy et al. [20]. Briefly, protein expression for MLH1, MSH2, MSH6, and PMS2 was considered intact (normal) if any percentage of definite positive nuclear staining of the malignant cells was detected on either TMA core. In cases where one or more mismatch repair proteins were interpreted as negative staining, examination utilizing immunohistochemistry on whole sections was performed. Each protein was considered lost (abnormal) if there was complete loss of nuclear staining in the tumor cells and if there was a positive internal control (intact nuclear staining of stromal elements such as inflammatory cells and/or endothelial cells) on whole section. Cases showing a complete absence of nuclear staining pattern of both tumor cells and stromal elements were deemed uninterpretable and thus excluded from the study. Cases that demonstrated loss of any MMR marker on the TMA were subjected to confirmatory whole slide section staining and were scored independently by two pathologists (BSS and DFS), who were blinded to clinical characteristics and patient outcomes. Divergent assessments were reconciled by consensus conference. A case was labeled as mismatch repair deficient (MMRd) if any of the four mismatch repair proteins was completely absent on immunohistochemistry. Cases were classified as mismatch repair proficient (MMRp) if all four proteins stained positive to some degree.
Individual tumor infiltrating lymphocytes were counted and typed in the epithelial and stromal compartments using clinically validated IHC stains for CD3 and CD8. Scoring was performed independently by two anatomical pathologists (BSS and MA-K) and consensus achieved on all cases. To account for potential intra-tumoral heterogeneity, the average of both cores were used to generate the final score for CD8+ and CD3+ tumor infiltrating lymphocytes.
Clinico-pathologic variables and outcome
Standard treatment, clinical and pathologic parameters were collected from the British Columbia Cancer Agency which included: age at surgery, sex, adjuvant chemotherapy agents used, lymphovascular invasion, perineural invasion, pathologic primary tumor (pT) stage, and pathologic regional lymph-node status (pN). The primary outcome measure was defined to be disease-specific survival, where survival time was calculated as the difference between the date of last follow-up and the date of surgery, expressed in years. Patients were censored if they were alive at last follow-up or had died from a cause other than their pancreatic malignancy. Deaths attributable to treatment related toxicities or inter-current diseases were considered censored observations for this analysis.
Statistical analysis
To determine if H-Score or the percent of positive cells for PD-L1 expression yield differential prognostic ability, each scoring method was subjected to an omnibus assessment utilizing the Cox-Proportional Hazards Model to determine if the expression of PD-L1 was a significant prognostic marker in the context of the clinico-pathologic variables outlined previously with the exception of pT-Stage due the fact that the vast majority of the cases in this cohort are pT3. The proportionality assumption for each variable was assessed through examination of Cox-Snell residuals and continuous variables were assessed for linearity. The PD-L1 scoring methodology with the smallest P-value was determined to have the strongest prognostic effect. Parametric survival analysis was used in order to further elucidate the gradient dependent effect of PD-L1 expression on disease specific survival (DSS) for the scoring methodology with the greatest prognostic effect. This procedure modelled the disease specific survival data with 5 different distributions which included: weibull, log-normal, exponential, frechet, and log-logistic. The best distribution to be used for parametric survival analysis was determined by selecting the one with the lowest Bayesian Information Criterion from the model fits. This analysis produces a quantile plot with logSurvival Time plotted against PD-L1 expression which illustrates the gradient dependent relationship between disease specific survival and PD-L1 expression. Based on these findings, a series of three cut-points were created starting with an H-Score or percentage positive cells of 1- as these identify the equivalent cases. Subsequent cut-points were set at increments of 5 which correspond to the increments used for the assessment of percent positive cells. The resultant groups were subjected to univariable survival analysis to quantify differences in disease specific survival using the Kaplan-Meier method. A multivariable approach to disease specific survival, using the Cox Proportional Hazards Model, was used to determine if survival differences between PD-L1 expression categories were independent of adjuvant chemotherapy. Assessment for heterogeneity of clinico-pathologic variables was performed with the following statistical approaches: continuous variables were examined using the Wilcoxon Rank-Sum Test, categorical comparisons were computed using Fisher’s exact test. A P-value of <0.05 was considered as statistically significant for all analyses. All analyses were computed with JMP v13.1 (SAS Institute, Cary, NC).
Results
After exclusion criteria were applied, two-hundred fifty-two cases remained (Fig. 1). The demographic information for the cohort were tabulated and are shown in Table 1. The Cox-Proportional Hazards Analysis for PD-L1 H-score and percent positive indicated that the latter was a stronger prognostic indicator with P = 0.0466 compared to H-Score with a P = 0.10 (Table 2).
Fig. 1.
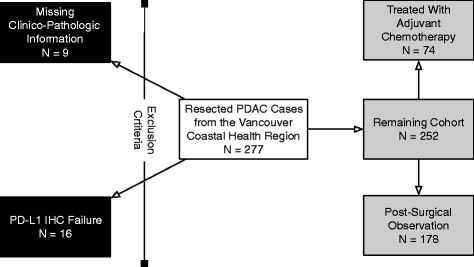
Patient selection diagram illustrating inclusion and exclusion criteria for this study with final numbers for the cohorts who received adjuvant pyrimidine nucleoside analogs or subjected to post-surgical observation only
Table 1.
Demographics of the entire cohort
| Variable | Levels | Values |
|---|---|---|
| Age | Median [IQR] | 66.4 [13.3] |
| Sex | Male | 139 (55.2%) |
| Female | 113 (44.8%) | |
| Adjuvant Chemotherapy | Given | 74 (29.4%) |
| Observation | 178 (70.6%) | |
| Histologic Grade | 1 | 2 (0.8%) |
| 2 | 186 (73.8%) | |
| 3 | 64 (25.4%) | |
| Lymphovascular Invasion | Present | 144 (57.1%) |
| Absent | 108 (42.9%) | |
| Perineurial Invasion | Present | 232 (92.1%) |
| Absent | 20 (7.9%) | |
| pT Stage | 1 | 2 (0.8%) |
| 2 | 11 (4.4%) | |
| 3 | 238 (94.4%) | |
| 4 | 1 (0.4%) | |
| pN Stage | 0 | 64 (25.4%) |
| 1 | 168 (74.6%) | |
| Resection Status | R0 | 190 (75.4%) |
| R1 | 62 (24.6%) | |
| CD3 Epithelial | Median [IQR] | 0 [0] |
| CD3 Stromal | Median [IQR] | 50 [52] |
| CD8 Epithelial | Median [IQR] | 0 [0] |
| CD8 Stromal | Median [IQR] | 11 [36] |
| MMR Status | Proficient | 211 (84.1%%) |
| Deficient | 40 (15.9%) | |
| Follow-up Time (Years) | Median [IQR] | 1.33 [1.59] |
| Events | Disease Specific Deaths | 200 (79.4%) |
| Censorings | 52 (20.6%) |
Table 2.
Multivariable disease specific survival analysis for PD-L1 expression quantified by percent positive & H-score
| Variable | Comparison | Risk ratio | 95%CI | p-value |
|---|---|---|---|---|
| PD-L1 H-Score | ||||
| PD-L1 H-Score | Per unit change | 1.01 | 0.997–1.02 | 0.10 |
| Age at Surgery | Per unit change | 1.01 | 0.991–1.02 | 0.44 |
| Sex | Male v Female | 1.14 | 0.85–1.53 | 0.37 |
| Adjuvant Chemotherapy | Given v Observation | 0.59 | 0.42–0.81 | 0.0011 |
| Histopathologic Grade | 1 v 2 | 0.63 | 0.10–2.10 | 0.50 |
| 1 v 3 | 0.49 | 0.08–1.67 | 0.28 | |
| 2 v 3 | 0.77 | 0.55–1.09 | 0.13 | |
| Lymphovascular Invasion | Present v Absent | 1.27 | 0.93–1.74 | 0.13 |
| Perineural Invasion | Present v Absent | 1.66 | 0.96–3.09 | 0.07 |
| pN-Stage | 1 v 0 | 1.83 | 1.27–2.68 | 0.0010 |
| Resection Status | R0 v R1 | 0.67 | 0.49–0.94 | 0.0202 |
| PD-L1 Percent Positive | ||||
| PD-L1 Percent Positive | Per unit change | 1.03 | 1.0005–1.05 | 0.0466 |
| Age at Surgery | Per unit change | 1.0006 | 0.99–1.02 | 0.42 |
| Sex | Male v Female | 1.16 | 0.86–1.55 | 0.33 |
| Adjuvant Chemotherapy | Given v Observation | 0.58 | 0.42–0.80 | 0.0008 |
| Histopathologic Grade | 1 v 2 | 0.64 | 0.10–2.13 | 0.51 |
| 1 v 3 | 0.50 | 0.08–1.71 | 0.30 | |
| 2 v 3 | 0.78 | 0.56–1.10 | 0.15 | |
| Lymphovascular Invasion | Present v Absent | 1.28 | 0.94–1.76 | 0.12 |
| Perineural Invasion | Present v Absent | 1.68 | 0.98–3.13 | 0.06 |
| pN-Stage | 1 v 0 | 1.85 | 1.28–2.71 | 0.0009 |
| Rescetion Status | R0 v R1 | 0.68 | 0.49–0.95 | 0.0189 |
Subjecting the survival data to the 5 distribution models outlined in the methods and ranking those fits by the Bayesian Information Criterion revealed that the log-logistic distribution fit best and was used as the basis for parametric disease specific survival analysis. Parametric disease specific survival of PD-L1 percent positive and H-Score demonstrated an inverse relationship between increased PD-L1 expression and survival time (Fig. 2). As indicated in the multivariable survival analysis, PD-L1 percent positive had a slightly stronger prognostic association compared to H-Score.
Fig. 2.
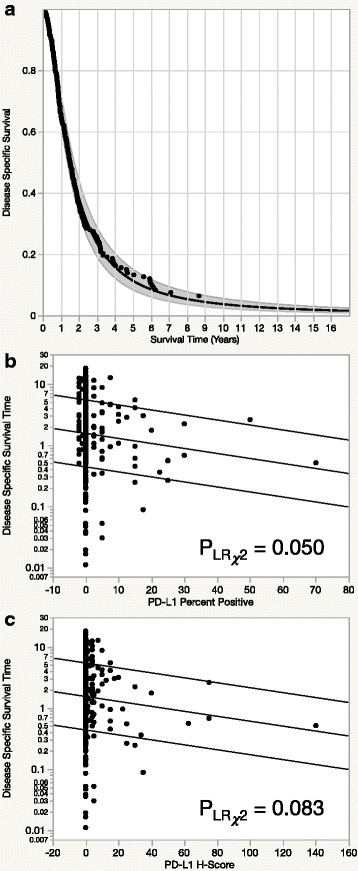
Parametric disease specific survival analysis using the log-logistic distribution to model disease specific survival in the entire cohort (a). Modeling of disease specific survival against PD-L1 expression assessed by percent positive (b) and H-Score (c) survival using a log-logistic distribution demonstrates a substantial association between reduced survival and increased PD-L1 expression. Curves shown are fitted as a function of the regressor representing the 0.9, 0.5, and 0.1 quantiles
Based on the prognostic non-inferiority of PD-L1 percent positive, we elected to pursue this scoring method for the remainder of the study. Cut-points were determined according to our criteria outlined in the methods and resulted in: > = 1% (N = 31; 12.3% of the cohort), >5% (N = 17; 6.7% of the cohort), and >10% (N = 12; 4.8% of the cohort). Univariable survival analysis using these three cut-points showed no disease specific survival differences at the > = 1 cut-point (p = 0.51) or the >5 cut-point (0.52), but the >10 cut-point yielded statistically significant disease specific survival differences of p = 0.027 (Fig. 3). Multivariable DSS analysis of the >10% positive PD-L1 expression cut-point along with the other clinico-pathologic covariates outlined in Table 2, indicates that this subset of twelve cases has a trend toward inferior prognosis with a Risk Ratio and 95%CI 1.90 [0.96–3.42] (P = 0.06). When we sequentially removed statistically insignificant variables from the model (age, histopathologic grade, sex, and lyphovascular Invasion, PD-L1 > 10% became statistically significant Risk Ratio and 95%CI 2.05 [1.03–3.66] (P = 0.0410). The remaining statistically significant variables included pN-Stage (P < 0.0001), adjuvant chemotherapy (P = 0.0002), and perineurial invasion (P = 0.009) and resection status (P = 0.0263).
Fig. 3.
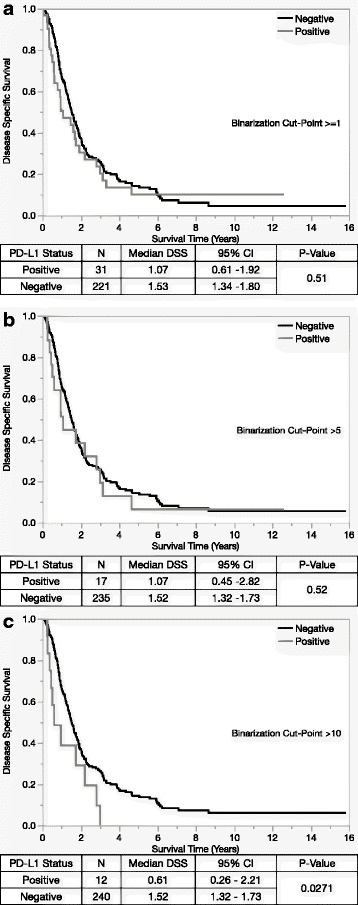
Binarization cut-points for percent positive (a-c) show that only the highest cut-point (>10) yields statistically significant survival differences
Analysis for heterogeneity across clinico-pathologic parameters which included: age, sex, adjuvant chemotherapy use, histopathological grade, lymphovascular invasion, perineural inavasion, pN-Stage, and resection status demonstrated a significant relationship between increased PD-L1 expression and higher grade cases (Table 3). The remaining clinico-pathologic variables, mismatch repair, and the stromal or epithelial compartment specific prevalence of CD3+ or CD8+ tumor infiltrating lymphocytes were not associated with the PD-L1 > 10% positive cells cut-point.
Table 3.
Assessment for heterogeneity using a percent positive binarization cut-point of >10
| Variable | Levels | PD-L1%Positive < 10 | PD-L1%Positive > 10 | P-Value |
|---|---|---|---|---|
| Age | Median [IQR] | 66.5 [13.1] | 65.6 [20.5] | 0.96 |
| Sex | Male | 132 (55.0%) | 7 (58.3%) | 1.0 |
| Female | 108 (45.0%) | 4 (41.8%) | ||
| Adjuvant Chemotherapy | Given | 71 (29.6%) | 3 (25.0%) | 1.0 |
| Observation | 169 (70.4%) | 9 (75.0%) | ||
| Histologic Grade | 1 | 2 (0.8%) | 0 (0.0%) | 0.029 |
| 2 | 181 (75.4%) | 5 (41.7%) | ||
| 3 | 57 (23.8%) | 7 (58.3%) | ||
| Lymphovascular Invasion | Present | 136 (56.7%) | 8 (66.7%) | 0.56 |
| Absent | 104 (43.3%) | 4 (33.3%) | ||
| Perineurial Invasion | Present | 221 (92.1%) | 11 (91.7%) | 1.0 |
| Absent | 19 (7.9%) | 1 (9.3%) | ||
| pT Stage | 1 | 2 (0.8%) | 0 (0.0%) | 0.88 |
| 2 | 10 (4.2%) | 1 (8.3%) | ||
| 3 | 227 (94.6%) | 11 (91.7%) | ||
| 4 | 1 (0.4%) | 0 (0.0%) | ||
| pN Stage | 0 | 62 (25.8%) | 2 (16.7%) | 0.74 |
| 1 | 178 (74.2%) | 10 (83.3%) | ||
| Resection Status | R0 | 184 (76.7%) | 6 (50%) | 0.08 |
| R1 | 56 (23.3%) | 6 (50%) | ||
| CD3 Epithelial | Median [IQR] | 0 [0] | 0 [4] | 0.16 |
| CD3 Stromal | Median [IQR] | 50 [52] | 63 [76] | 0.52 |
| CD8 Epithelial | Median [IQR] | 0 [0] | 0 [0] | 0.51 |
| CD8 Stromal | Median [IQR] | 11 [36] | 14 [39] | 0.72 |
| MMR Status | Proficient | 200 (83.7%) | 11 (91.7%) | 0.70 |
| Deficient | 39 (16.3%) | 1 (8.3%) |
Discussion
In this study, we have found a gradient dependent association between PD-L1 expression and inferior disease specific survival in resected pancreatic ductal adenocarcinoma. This finding was independent of the improved prognosis associated with the application of adjuvant chemotherapy with a pyrimidine nucleoside analog.
We assessed multiple scoring methods, H-Score or percent of positive cells, for the quantification of PD-L1 expression and determined that the estimation of percent positive cells yields a stronger association with inferior survival than H-Score. This suggests that the addition of a subjective intensity assessment to generate an H-score may represent an unnecessary step for the quantification of PD-L1 in this disease. Examination of other clinico-pathologic parameters revealed no statistically significant associations with PD-L1 expression at any cut-point, which indicates that PD-L1 expression does not select for any known prognostic variable other than histo-pathologic grade. Due to the limited power associated with our cohort combined with the small fraction of cases that express PD-L1 at a high level, we were limited in our ability to perform multivariable disease specific survival analyses with numerous variables. Exploratory multivariable disease specific survival modeling suggested that our categorized PD-L1 expression utilizing the cut-point of >10% of positive cells is independently associated with inferior disease specific survival and was only surpassed by the presence of regional lymph node metastasis and perineural invasion in terms of negative prognostic variables.
Recent studies have demonstrated that PD-L1 expression is associated with tumor types known to have higher somatic mutation load, as is the case for melanomas, NSCLC and RCC [21, 22]. Considering that PDAC has a lower mutation burden, it is not surprising that we found only 4 to 12% PD-L1 positive tumors compared to the reported 83% in melanoma, 50% in NSCLC and 80% in RCC [10]. Nonetheless, PDAC is associated with tobacco use and BRCA loss-of-function, and is predicted to, at least occasionally, show an increased mutation burden as a result of these [23]. Consequently, the lower than average rate of PD-L1 expression in PDAC compared to other malignancies may explain poor response to checkpoint inhibitors in clinical trials since PD-L1 was either not accounted for or the positivity thresholds were only set between 1% and 5% [10, 11]. Although our patient cohort was mostly treatment naive, we were able to identify differential outcomes based on higher PD-L1 expression.
The observed increased trend of lymphocyte tumor infiltration (CD3+) in PD-L1 positive patients has been reported in previous studies [24]. Sanmamed et al. showed that tumor infiltrating lymphocytes release IFN-Gamma as part of the host response to the tumor, which induces upregulation, and expression of, PD-L1 by tumor cells [25]. Our results indicate that a cut-point > = 1% yields the strongest association with CD3+ infiltrating T-cells but due to reduced power associated with increasing the PD-L1 cut-point, statistical significance is lost at higher thresholds.
We found no significant association between MMR and PD-L1 status. Our results are somewhat different from what was observed by Le et al. (2016) who reported that, in a series of 30 cases, PD-L1 was only expressed in MMR deficient (MMRd) tumors, most of which being colorectal carcinomas [11]. This inconsistency might be explained by the lower mutational burden seen in PDAC compared to MMRd colon carcinoma, melanoma, NSCLC and RCC [22]. Tumors with low mutational burden tend to be less immunogenic, making them less likely to develop immune silencing mechanism during their evolution.
There are several limitations to our study, one being the lack of consensus for PD-L1 IHC expression cut-off and gold standard, which our study has attempted to explore. Our IHC protocol for PD-L1 previously showed fairly strong concordance when compared to three other PD-L1 clones and RNA in situ hybridization (ISH), Sheffield et al., in NSCLC [26]. Our sample size is limited given the small percentage of PD-L1 expression and may have been underpowered to detect some more subtle associations, especially in the higher PD-L1 cut-points. Finally, since the IHC was performed on a TMA rather than full section, we might have underrepresented the amount of PD-L1 positive PDAC due to sampling error, although this method approximates the biopsy sampling error in encountered in clinical practice.
The prevalence of PD-L1 positivity in PDAC has been examined in numerous other studies with the percentage of tumor cells staining positive ranging from 4% - 49%. Each of these previous studies utilized different cut-points that varied between 1% - 10% making their results nearly impossible to compare [27–29]. Of particular interest, our results are somewhat different from what has been reported by Nomi et al. who demonstrated a found a 39% PD-L1 positivity in pancreatic cancer using a 10% positivity threshold [28]. Their cohort included 51 cases from Japan, which were stained using Anti-Human CD274, clone MIH1. The difference in PD-L1 expression is notable and although the CD274 is not commonly used in the clinical research setting this result may indicate variability associated with ethnicity.
Conclusions
In summary, this is the first study to systematically investigate the association between clinical outcome and biomarker expression across differing scoring methodologies and cut-points for PD-L1 immunohistochemistry in this disease. We have demonstrated a gradient dependent association between PD-L1 expression and inferior survival that is independent of the prognostic advantage conferred by adjuvant chemotherapy. We postulate that the association presented here may indicate that higher PD-L1 protein expression levels represent a phenotype where PD-1 inhibition may be more effective. However, this hypothesis would have to be tested in the context of a randomized clinical trial. With studies in other diseases also indicating that deficient MMR (MMRd) status has been shown to be a predictive biomarker for immunotherapy, it is entirely plausible that PD-L1 immunohistochemistry is an imperfect biomarker for sensitivity to anti-PD-1 therapy. Interestingly, we found no association between MMRd status and PD-L1 expression in this cohort. More data on the role of PD-1-axis inhibition in PDAC is needed, specifically examining the use of predictive biomarkers in the context of patients treated with immunotherapy. Future studies should endeavor to build predictive models based on multi-marker expression that will serve as tools to triage the PDAC patient population to immunotherapy or other treatment regimens.
Acknowledgements
We, the authors, would like to recognize the patients and their families for their direct and indirect contributions toward fighting this disease.
Funding
This work was supported through unrestricted research funds provided by the VGH and UBC Hospital Foundation and the BC Cancer Foundation which were administered through the Pancreas Centre BC. The above funders of this research had no influence upon the design of the study, collection, analysis nor interpretation of the data or writing of the manuscript.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- MMRd
Mismatch Repair Deficient
- MMRp
Mismatch Repair Proficient
- PD1
Programmed Cell Death 1
- PDAC
Pancreatic Ductal Adenocarcinoma
- PD-L1
Programmed Cell Death Ligand 1
Authors’ contributions
BTC: Conceived the study, wrote the first draft of the manuscript and had input on revisions after internal review by co-authors. SEK: Conceived the study, performed all statistical analyses and participated in the writing of the manuscript. MA: Developed the method and provided all scoring for immune infiltrates for epithelial and stromal components of each PDAC case. KM: Developed and optimized the immunohistochemical staining procedure for the PD-L1 antibody and was responsible for applying this procedure to the TMA. Contributed to the methods section of the manuscript. DG: Developed the scoring system used for this study and performed the scoring for the PD-L1 antibody. Contributed to the methods section of the manuscript. BHN: Advised on the design of the study and served as an internal reviewer for the manuscript. DJR: Acquired the clinical follow-up for patients included in this TMA case series. Served as an internal reviewer for the manuscript and served in a co-supervisory capacity for the project. BSS: Performed consensus IHC scoring for PD-L1, CD3, CD8, and MMR immunohistochemistry. Advised on study design and helped to improve the manuscript though critical review. DFS: Advised on the study design and aided with the interpretation of the results. Served as an internal reviewer for the manuscript and served in a co-supervisory capacity for the project. Performed original scoring assessment for MMR status. All authors have given final approval of the manuscript in its current form and agree to take responsibility for the accuracy and integrity of it’s content. Any errors and omissions are our own.
Ethics approval and consent to participate
Ethical approval for research on this retrospective cohort was obtained from the University of British Columbia Clinical Research Ethics Board (H12- 03484). A waiver of consent was provided to account for the large proportion of patients that were deceased at the time of the assembly of this cohort.
Consent for publication
Not Applicable
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Footnotes
Basile Tessier-Cloutier and Steve E Kalloger contributed equally to this work.
Daniel J. Renouf and David F. Schaeffer co-supervised this work.
Contributor Information
Basile Tessier-Cloutier, Email: basile.tessiercloutier@mail.mcgill.ca.
Steve E. Kalloger, Phone: 604-889-0997, Email: skalloger@mac.com
Mohammad Al-Kandari, Email: moh.alkandari@gmail.com.
Katy Milne, Email: kmilne@bccrc.ca.
Dongxia Gao, Email: dongxia.Gao@vch.ca.
Brad H. Nelson, Email: bnelson@bccrc.ca
Daniel J. Renouf, Email: drenouf@bccancer.bc.ca
Brandon S. Sheffield, Email: brandon.s.sheffield@gmail.com
David F. Schaeffer, Email: david.Schaeffer@vch.ca
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.Topalian SL, Sznol M, McDermott DF, Kluger HM, Carvajal RD, Sharfman WH, et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020–1030. doi: 10.1200/JCO.2013.53.0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brahmer JR, Tykodi SS, Chow LQ, Hwu W-JJ, Topalian SL, Hwu P, et al. safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366:2455–2465. doi: 10.1056/NEJMoa1200694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.De Guillebon E, Roussille P, Frouin E, Tougeron D. Anti program death-1/anti program death-ligand 1 in digestive cancers. World J Gastrointest Oncol. 2015;7:95–101. doi: 10.4251/wjgo.v7.i8.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8:793–800. doi: 10.1038/nm0902-1039c. [DOI] [PubMed] [Google Scholar]
- 6.Dong H, Zhu G, Tamada K, Chen L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med. 1999;5:1365–1369. doi: 10.1038/70932. [DOI] [PubMed] [Google Scholar]
- 7.Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000;192:1027–1034. doi: 10.1084/jem.192.7.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Topalian SL, Drake CG, Pardoll DM. Targeting the PD-1/B7-H1(PD-L1) pathway to activate anti-tumor immunity. Curr Opin Immunol. 2012;24:207–212. doi: 10.1016/j.coi.2011.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iwai Y, Ishida M, Tanaka Y, Okazaki T, Honjo T, Minato N. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci U S A. 2002;99:12293–12297. doi: 10.1073/pnas.192461099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Topalian SL, Hodi SF, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–2454. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372:2509–2520. doi: 10.1056/NEJMoa1500596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Herbst RS, Soria J-CC, Kowanetz M, Fine GD, Hamid O, Gordon MS, et al. predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature. 2014;515:563–567. doi: 10.1038/nature14011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Powles T, Eder J, Fine GD, Braiteh FS, Loriot Y, Cruz C, et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature. 2014;515:558–562. doi: 10.1038/nature13904. [DOI] [PubMed] [Google Scholar]
- 14.Herbst RS, Baas P, Kim D-WW, Felip E, Pérez-Gracia JL, Han J-YY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387:1540–1550. doi: 10.1016/S0140-6736(15)01281-7. [DOI] [PubMed] [Google Scholar]
- 15.Meng X, Huang Z, Teng F, Xing L, Yu J. Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat Rev. 2015;41:868–76. [DOI] [PubMed]
- 16.Taube JM, Klein A, Brahmer JR, Xu H, Pan X, Kim JH, et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res. 2014;20:5064–5074. doi: 10.1158/1078-0432.CCR-13-3271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ascierto PA, Capone M, Urba WJ, Bifulco CB, Botti G, Lugli A, et al. The additional facet of immunoscore: immunoprofiling as a possible predictive tool for cancer treatment. J Transl Med. 2013;11:54. doi: 10.1186/1479-5876-11-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Messina JL, Fenstermacher DA, Eschrich S, Qu X, Berglund AE, Lloyd MC, et al. 12-chemokine gene signature identifies lymph node-like structures in melanoma: potential for patient selection for immunotherapy? Sci Rep. 2012;2:765. doi: 10.1038/srep00765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015;348:124–128. doi: 10.1126/science.aaa1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Riazy M, Kalloger SE, Sheffield BS, Peixoto RD, Li-Chang HH, Scudamore CH, et al. Mismatch repair status may predict response to adjuvant chemotherapy in resectable pancreatic ductal adenocarcinoma. Mod Pathol. 2015;28:1383–1389. doi: 10.1038/modpathol.2015.89. [DOI] [PubMed] [Google Scholar]
- 21.Grosso J, Inzunza D, Wu Q, Simon J, Singh P, Zhang X, et al. Programmed death-ligand 1 (PD-L1) expression in various tumor types. Journal for immunotherapy of cancer. 2013;1:P53. doi: 10.1186/2051-1426-1-S1-P53. [DOI] [Google Scholar]
- 22.Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature. 2013;499:214–218. doi: 10.1038/nature12213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SA, Behjati S, Biankin AV, et al. Signatures of mutational processes in human cancer. Nature. 2013;500:415–421. doi: 10.1038/nature12477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Furuta J, Inozume T, Harada K, Shimada S. CD271 on melanoma cell is an IFN-γ-inducible immunosuppressive factor that mediates downregulation of melanoma antigens. J Invest Dermatol. 2014;134:1369–1377. doi: 10.1038/jid.2013.490. [DOI] [PubMed] [Google Scholar]
- 25.Sanmamed MF, Chen L. Inducible expression of B7-H1 (PD-L1) and its selective role in tumor site immune modulation. Cancer J. 2014;20:256–261. doi: 10.1097/PPO.0000000000000061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sheffield BS, Fulton R, Kalloger SE, Milne K, Geller G, Jones M, et al. Investigation of PD-L1 biomarker testing methods for PD-1 Axis inhibition in non-squamous non-small cell lung cancer. J Histochem Cytochem. 2016;64:587–600. doi: 10.1369/0022155416665338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Diana A, Wang L, D’Costa Z, Allen P, Azad A, Silva MA, et al. Prognostic value, localization and correlation of PD-1/PD-L1, CD8 and FOXP3 with the desmoplastic stroma in pancreatic ductal adenocarcinoma. Oncotarget. 2016;7:40992–41004. doi: 10.18632/oncotarget.10038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nomi T, Sho M, Akahori T, Hamada K, Kubo A, Kanehiro H, et al. Clinical significance and therapeutic potential of the programmed death-1 ligand/programmed death-1 pathway in human pancreatic cancer. Clin Cancer Res. 2007;13:2151–2157. doi: 10.1158/1078-0432.CCR-06-2746. [DOI] [PubMed] [Google Scholar]
- 29.Wang L, Ma Q, Chen X, Guo K, Li J, Zhang M. Clinical significance of B7-H1 and B7-1 expressions in pancreatic carcinoma. World J Surg. 2010;34:1059–1065. doi: 10.1007/s00268-010-0448-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.


