Abstract
Reactive oxygen species (ROS) are essential cellular metabolites widely implicated in many diseases including cancer, inflammation, and cardiovascular and neurodegenerative disorders. Yet, ROS signaling remains poorly understood, and their measurements are a challenge due to high reactivity and instability. Here, we report the development of 13C-thiourea as a probe to detect and measure H2O2 dynamics with high sensitivity and spatiotemporal resolution using hyperpolarized 13C magnetic resonance spectroscopic imaging. In particular, we show 13C-thiourea to be highly polarizable and to possess a long spin–lattice relaxation time (T1), which enables real-time monitoring of ROS-mediated transformation. We also demonstrate that 13C-thiourea reacts readily with H2O2 to give chemically distinguishable products in vitro and validate their detection in vivo in a mouse liver. This study suggests that 13C-thiourea is a promising agent for noninvasive detection of H2O2 in vivo. More broadly, our findings outline a viable clinical application for H2O2 detection in patients with a range of diseases.
Graphical abstract
Oxidative stress is a hallmark of many pathophysiological diseases corresponding to the oxidative damage incurred by aberrant reactive oxygen species (ROS) production. These ROS are essential for cellular signaling.1,2 They include highly reactive oxygen metabolites such as hydrogen peroxide (H2O2), superoxide (O2•–), and hydroxyl radical (−OH). Dysregulation of ROS homeostasis leads to oxidative stress and irreversible molecular damage, which is associated with various disease states such as cancer, inflammation, and cardiovascular and neurodegenerative disorders.3–7
Despite their importance, the precise mechanisms of ROS signaling are poorly understood. The high reactivity and short lifetime of ROS have further complicated efforts for determining their spatiotemporal dynamics in vivo. ROS detection in biological systems therefore requires probes that rapidly react with ROS to produce stable quantifiable products. Indeed, several studies have reported the development of ROS-specific probes for live in vivo optical detection.8,9 These probes capitalized on the enhanced contrast of a caged fluorescence emission that is activated upon ROS-induced transformation. The use of small molecule and nanoparticle probes in these studies has in turn, inspired us to exploit 13C magnetic resonance spectroscopic imaging (MRSI)10 to study ROS flux in vivo.
In contrast to optical techniques, MRSI enables noninvasive acquisition of deep-tissue images in vivo and the investigation of real-time cellular metabolism with high spatial and temporal resolution.11,12 However, MRSI experiments have inherently low sensitivity and are therefore limited to the detection of abundant 1H nuclei from water molecules and lipid chains in vivo. To gain information on intermediary cellular metabolites and their fluxes, we have utilized 13C MRSI by taking advantage of its wide chemical shift differences to encompass signals from both a substrate and its metabolic product(s).10 To overcome the naturally low abundance of a 13C signal, we hyperpolarized the 13C-labeled substrate by using dynamic nuclear polarization (DNP) to afford more than 10 000-fold signal enhancement of the 13C-probe and its subsequent metabolic products.13,14 In combination with a rapid dissolution process, hyperpolarized (HP) 13C-substrates such as [1-13C]-pyruvate,15 [2-13C]-pyruvate,16 [13C]-glucose,17 [13C]-bicarbonate,18 [2-13C]-fruc-tose,19 and [2-13C]-dihydroxyacetone20 have been used as metabolic probes to investigate cellular metabolism with high spectral resolution. Notably, [1-13C]-dehydroascorbate (DHA)21,22 and [1-13C]-benzoylformic acid (BFA)23 have been reported as HP 13C-substrates to probe oxidative stress specific to redox and H2O2 sensing, respectively.
To design new HP 13C-probes for ROS detection in vivo, we focused on small-molecule metabolites exhibiting several key properties. First, the substrate should be highly polarizable either by DNP or other methods.24–26 Second, both the substrate and its metabolic product(s) should possess a long spin–lattice relaxation time (T1), as needed for robust in vivo detection. Third, the HP probe should reveal a distinctive chemical shift for its 13C nuclei upon reaction with ROS in order to allow spectroscopic detection of both the probe and its products. Finally, the probe should be nontoxic (at micromolar to millimolar concentration) and be rapidly transported for ROS-mediated reaction (or react in the extracellular space).
To this end, we explored 13C-thiourea (13C-TU) as an HP probe for ROS detection. Thiourea is a water-soluble ROS scavenger that has been associated with antioxidant proper-ties.27,28 In liver microsomes, flavin-containing monooxygenases (FMO) catalyze an NADPH-dependent S-oxygenation of thiourea in the presence of O2,29 leading to the formation of formamidine sulfenic acids (TUO) that in turn either react reversibly with glutathione (GSH) or undergo further oxidation to form thiourea dioxide (TUO2). Notably, no thiourea transporters have been identified in mammals, even though studies have implicated urea transporters for a shared transport mechanism with thiourea in erythrocytes.30 In addition, saturation kinetics in perfused rat inner medullary collecting duct showed saturation of the urea transporter with a KM = 20 mM for thiourea.31 Moreover, studies on a single intra-peritoneal exposure of thiourea in different animal models have yielded a range of lethal dose (LD50) concentrations of up to 1340 mg/kg body weight in rats.32 Together, these studies suggest that 13C-TU is an attractive HP probe candidate for ROS detection.
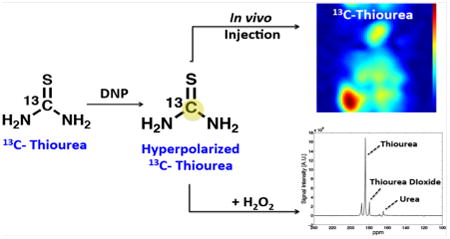
To test our hypothesis, we determined the selectivity of 13C-TU with different ROS that are responsible for many cellular signaling events. These ROS have been implicated in oxidative damage inflicted on fatty acid, DNA, and proteins as well as other cellular components (Figure 1A).33 Briefly, thiourea was exposed to several biologically relevant ROS for 10 min, and the formation of oxidized products was monitored by 13C NMR. In the presence of •OOH (H2O2 or tBuOOH), we detected oxidation of TU (δ184 ppm) to form TUO2 (δ 179.5 ppm), as identified by LC-MS (Figure 1B and S1). When reacted with other ROS such as superoxide (O2•−) and hydroxyl radical (•OH), thiourea remained unchanged. Reaction with hypochlorite (−OCl) and peroxynitrite (ONOO−) yielded urea (δ 165 ppm) as the hydrolyzed byproduct (Figure 1B and C). While in vitro oxidation of thiourea is not limited to peroxide, we were especially interested in using 13C-TU to detect H2O2 in vivo due to its central role in cellular signaling.34,35 Such a measurement is physiologically relevant because FMO-mediated oxygenation of thiourea in vivo is reported to proceed via reaction with H2O2.36 We therefore investigated thiourea oxidation by reacting different concentrations of H2O2 (2-27 μM) with 10 μM 13C-TU and examining 13C-TUO2 formation by 13C NMR (Figure 1D). By plotting the ratio of the integrated 13C-TUO2 product peak to the total signal intensities (13C-TU + 13C-TUO2) against peroxide, we showed a good linear correlation to increasing H2O2 concentration (Figure 1E) and confirmed its sensitivity for detecting low micromolar peroxide concentration in vitro.
Figure 1.
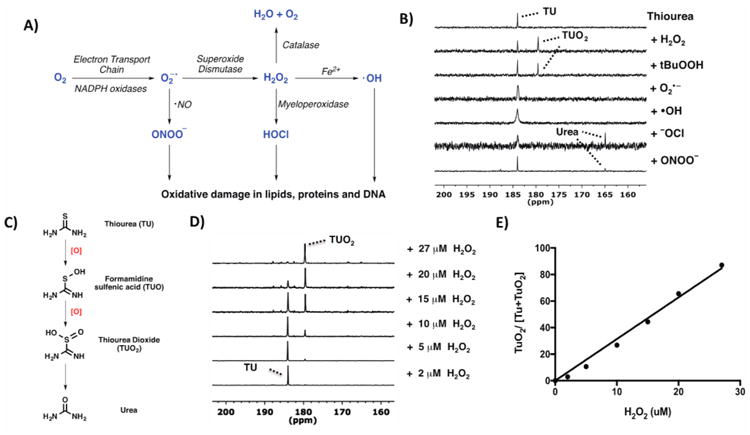
(A) Different kinds of ROS generated via multiple enzymatic steps. (B) Response of thiourea (TU) to various ROS at thermal equilibrium. All ROS (H2O2, tBuOOH, NaONOO, and NaOCl) were added to TU in D2O (except for KO2-treated sample in DMSO) and the spectra recorded with 13C NMR at 125 mHz. Reaction with H2O2 and tBuOOH yielded thiourea dioxide (TUO2) as the oxidized product. (C) TU was oxidized to TUO2 before getting hydrolyzed to urea. (D) 13C NMR spectra of 10 μM 13C-TU after 10 min reaction with various concentrations of H2O2 (2–27 μM). (E) Linear correlation of the ratio of integrated peak intensities of 13C-TUO2 to the total signal intensities (13C-TU + 13C-TUO2) against H2O2 concentration, R2 = 0.994.
Next, we sought to measure the rate of thiourea oxidation by H2O2 using UV/vis spectroscopy. We first performed a UV sweep of thiourea solutions (12.5–200 μM) and determined a maximum absorbance at λmax = 237 nm with an extinction coefficient ε = 0.0113 μM−1 cm−1 (Figure S2). We measured the kinetics of thiourea oxidation by detecting A237 change (depletion of thiourea) when reacted with H2O2. Briefly, two thiourea solutions (0.0625 and 0.125 mM) were prepared in a buffered solution at pH 7.4, incubated with different H2O2 concentrations (0.125–8 mM), and monitored for change in A237 over 50 min (Figure 2A). By plotting the initial rate against peroxide concentration, we found the results to be in close agreement with a previous report,37 that the rate of thiourea oxidation is first-order with respect to each thiourea and H2O2 concentrations (Figure 2B). Kinetics measurements reveal that the reaction between thiourea and H2O2 proceeds rapidly to produce thiourea dioxide with a second-order rate constant (k2) of 0.082 ± 0.002 M−1 s−1 (Figures 2B and S3).
Figure 2.
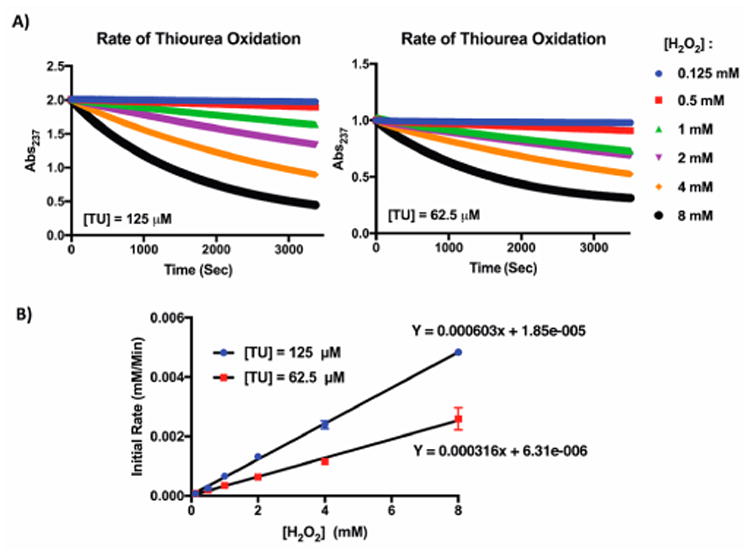
(A) UV/vis measurement of TU oxidation with H2O2. Representative measurement of thiourea consumption with different H2O2 concentrations (0.125–8 mM) as measured by A237 change. (B) Oxidation follows a first order reaction kinetic with respect to TU and H2O2. Second-order rate constant, k2 = 0.082 ± 0.002 M−1 s−1.
We then proceeded to test the ability of 13C-TU to undergo hyperpolarization using a preclinical HyperSense (Oxford Instruments) and a clinical SPINlab (GE Healthcare) polarizer. The polarization of a 13C-labeled substrate exploits the principle of the nuclear Overhauser effect, which transfers nuclear spin polarization from one spin bath to another (e.g., free radical species to the 13C nuclei of thiourea) using microwave irradiation at near absolute zero temperature (1.2 K for HyperSense, 0.8 K for SPINlab). The hyperpolarized sample was then rapidly dissolved, and the corresponding T1 measured with a clinical 3T MRI scanner. Samples polarized with the HyperSense exhibited a polarization level of 3.2 ± 0.1% and T1 = 30.2 ± 0.8 s, while the SPINlab produced a polarization level of 10.4 ± 1.1% and T1 = 53.8 ± 3.7 s (Figure 3a and Figure S4). Notably, higher polarization of the sample from the SPINlab might be attributed to the use of a hand-held electromagnetic carrier, which preserves polarization during sample transfer,38 and the longer T1 measurement was due to the exclusion of gadolinium. Together, polarization by the HyperSense and SPINlab represented a 12 200- and 39 500-fold enhancement, respectively, of the 13C-TU signals relative to thermal equilibrium. We then evaluated 13C-TU for spectroscopic detection of H2O2 in vitro by characterizing its oxidation product(s) with 13C-MRS. Briefly, 4 M solutions of 13C-TU and 15 mM of OX063 (Oxford Instruments) radicals in 60% (v/v) glycerol were hyperpolarized at 1.2 K and irradiated with microwaves (139.88 GHz) using a HyperSense polarizer. After hyperpolarization, the sample was rapidly dissolved in 100 mM Trizma-HCl buffer (pH ∼ 7.6) to a final thiourea concentration of 50 mM and reacted with 50 μM H2O2 for ∼30 s before spectral acquisition with a 3T scanner. Representative spectra from the peroxide-treated 13C-TU showed several distinct peaks. In particular, we identified 13C-TUO2 (179.6 ppm) as the oxidation product of 13C-TU (184 ppm) within 50 s of H2O2 reaction (Figure 3B and C). Additional byproducts of TU oxidation were also detected at 164.5 and 168.7 ppm, which were assigned to 13C-urea and 13C-TU2, respectively.39 Resonance frequencies of TU and the oxidized products are assigned based on the resonance of a 1 M 13C-bicarbonate phantom. Indeed, peroxide-mediated oxidation of TU proceeds via a mono-oxygenated intermediate (TUO), which further oxidizes to TUO2. Additionaly, the TUO intermediates may undergo reversible disulfide (TU2) formation in the presence of excess TU.40 Any resulting TUO2 can then be readily hydrolyzed to form urea under mildly basic conditions.40 Finally, we observed a previously unidentified peak at 188.1 ppm, which likely corresponds to the 13C-TUO intermediate. Indeed, time course analysis of the reaction revealed a nonlinear decay in 13C-TUO signal at t = 35–40 s, which was followed by an increase in the 13C-TUO2 signal at t = 50 s (Figure 3C, Figure S5). In contrast, a linear decay was observed in 13C-TU as a result of the spin–lattice relaxation time of the hyperpolarized signal (Figure S5). These findings suggest that despite the spin-lattice relaxation time, we were able to detect 13C-TUO oxidation to 13C-TUO2 in real time. Together, these results demonstrate that the identification of a transient 13C-TUO intermediate is required for 13C-TUO2 production and that TU can successfully detect H2O2 by 13C-MRS.
Figure 3.
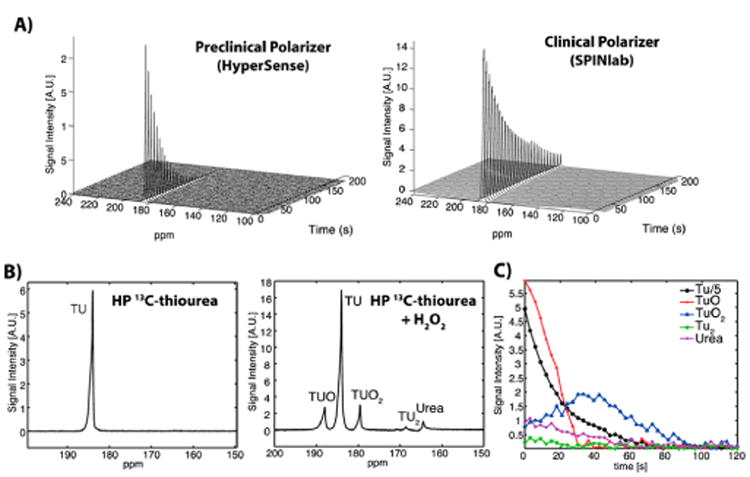
In vitro 13C MR spectra of hyperpolarized TU. (A) 13C-TU was hyperpolarized using a HyperSense (preclinical) or a SPINlab (clinical) polarizer. Polarization and T1 of the hyperpolarized TU samples were measured using a clinical 3T MR scanner and a nonselective pulse-and-acquire MRS sequence (flip-angle = 5.625°, temporal resolution = 3 s) after a 30 s delay from the dissolution due to mixing of 13C-TU with H2O2 and transfer to the magnet. (B) Time-averaged spectra of hyperpolarized 13C-TU showed multiple oxidation products (TUO, TUO2, TU2, and urea) when reacted with 50 μM H2O2. (C) Temporal changes of hyperpolarized TU and products due to T1 decay and the reaction to H2O2 were detected from time courses of hyperpolarized 13C TU and the oxidation products. See SI for experimental details.
To evaluate TU for in vivo H2O2 sensing, we applied HP 13C-TU to detect the endogenous H2O2 level in healthy male rats. Briefly, 4 M solutions of 13C-TU and 15 mM AH111501 (GE Healthcare) radicals were hyperpolarized using a SPINlab polarizer, rapidly dissolved into 50 mM final thiourea concentration and intravenously injected into an anesthetized rat for imaging with a 3T MR scanner. A slice-selective 13C spectroscopic image over an oblique plane encompassing several major organs was acquired with free induction decay chemical shift imaging sequence using a 13C/1H dual-tuned volume coil (FID CSI, field of view = 80 × 80 mm2, matrix size = 16 × 16, flip angle = 10°, acquisition time = 19 s, repetition time = 75 ms), and the corresponding 1H MRI confirmed significant 13C-TU resonance in well-perfused tissues of a rat, particularly in the heart, liver, and kidney (Figure 4A). However, no metabolic products from thiourea oxidation were detected above the noise level, presumably due to native antioxidant responses that kept endogenous H2O2 concentration low (∼10-8 M)41 and thus undetectable by our HP probe. To assess whether thiourea oxidation could be detected in vivo, we coinjected HP 13C-TU with H2O2 into the intraperitoneal cavity of an anesthetized mouse to simulate elevated ROS level under oxidative stress. A small transmit/ receive surface radiofrequency coil was then used for localized data acquisition from the mouse liver, and a 13C-MRS scan of the injection site revealed several oxidized species in the liver (Figure 4B). One major oxidative product detected downfield of 13C-TU (184 ppm) was identified as the mono-oxygenated 13C-TUO intermediate (188.1 ppm). Moreover, several smaller but detectable oxidative products were assigned to 13C-TUO2 (179.5 ppm), 13C-urea (164.5 ppm), and 13C-TU2 (168.1 ppm) based on their in vitro chemical shifts. Both 13C-TUO and 13C-TUO2 exhibited a linear rate of decay in vivo with apparent rate constants (1/T1) of kTUO = 0.0342 s−1 and kTUO2 = 0.0249 s−1, respectively, which were similar to 13C-TU decay (kTU = 0.0308 s−1; Figure 4c). In addition, we examined the extent of thiourea oxidation by calculating the relative TUO2/TUO ratios in vitro and in vivo and found that the relative TUO2 signal is 7.9-fold lower in vivo. These results suggest that 13C-TUO was not actively oxidized to 13C-TUO2 in vivo despite the exogenous peroxide addition and that the lack of in vivo oxidation of 13C-TUO might be attributed to the rapid antioxidant response to clear any excess peroxide.
Figure 4.
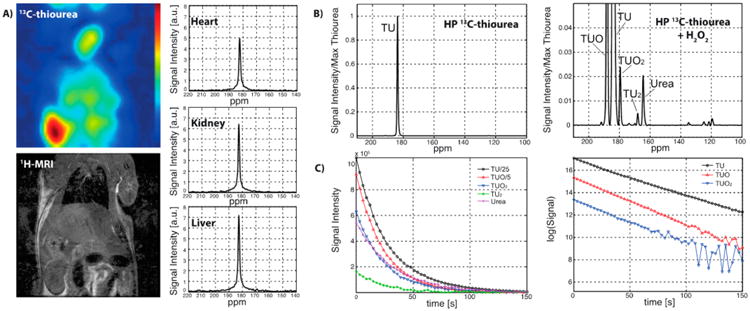
In vivo detection of hyperpolarized 13C-TU. (A) A slice-selective 2D 13C chemical shift imaging (CSI) and a 1H MRI over an oblique plane that contains heart, kidney, and liver of a healthy rat, acquired using a 13C/1H dual-tuned volume coil. The CSI scan started 20 s after a bolus injection (i.v.) of hyperpolarized 13C-TU, estimating the spatial distribution of the TU and measuring its perfusion into each organ (field of view = 80 × 80 mm2, matrix size = 16 × 16, slice thickness = 15 mm). No 13C-oxidized product was observed. (B) 13C-MR spectra of a mouse liver after an injection (i.p.) of hyperpolarized 13C-TU with and without H2O2. A nonselective pulse-and-acquire MRS sequence was used (flip-angle = 10°, temporal resolution = 3 s). The localization was achieved by positioning a small 13C transmit/receive surface coil (diameter = 28 mm) on top of the mouse liver and the intraperitoneal cavity. Oxidized products (TUO and TUO2) and byproducts (TU2 and urea) were detected in vivo immediately when TU and H2O2 were coinjected. No oxidized product was observed with the TU-only injection. (C) All the hyperpolarized signals of 13C-metabolites exhibited monoexponential decays as a result of the spin–lattice relaxations (T1). See SI for experimental details.
In summary, we have introduced a potential new 13C-MRSI probe to detect ROS level in real-time. Indeed, the present studies represent an ongoing effort to measure oxidative stress noninvasively. Our data demonstrate that 13C-TU may be a viable 13C-MRSI agent with important applications for real-time H2O2 detection. Notably, we were successful in detecting thiourea oxidation products in vivo using hyperpolarized 13C-TU and coinjected H2O2. In addition, we found the highest 13C-TU resonance in well-perfused organs, implicating its potential use for extracellular ROS measurements in blood and/or plasma. Future studies with 13C-TU in diseased animal models may provide important insights into the role of oxidative stress in pathophysiology.
Supplementary Material
Acknowledgments
This work is dedicated to the memory of Dr. G. Sommer.
Funding: This work was supported by NIH grants R01 EB019018, DK063158, S10 OD012283, and P41 EB015891; DOD CDMRP PC100427; the Gambhir-RSL grant; and the Stanford BioX program.
Abbreviations
- HP
hyperpolarized
- TU
thiourea
- TUO
formamidine sulfenic acid
- TUO2
thiourea dioxide
- MRS
magnetic resonance spectroscopy
- MRSI
magnetic resonance spectroscopic imaging
Footnotes
Supporting Information: The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acschem-bio.7b00130.
Detailed methods include selectivity assays, NMR spectroscopy, kinetic measurement, polarization procedures, spectral measurement and imaging (PDF)
Notes: The authors declare no competing financial interest.
References
- 1.D'Autréaux B, Toledano MB. ROS as signalling mole- cules: mechanisms that generate specificity in ROS homeostasis. Nat Rev Mol Cell Biol. 2007;8:813–824. doi: 10.1038/nrm2256. [DOI] [PubMed] [Google Scholar]
- 2.Giorgio M, Trinei M, Migliaccio E, Pelicci PG. Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat Rev Mol Cell Biol. 2007;8:722–778. doi: 10.1038/nrm2240. [DOI] [PubMed] [Google Scholar]
- 3.Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie MJ, Kulp AN, Qian D, Lam JS, Alles LE, Wong M, Joshua B, Kaplan MJ, Wapnir I, Dirbas FM, Somlo G, Garberoglio C, Paz B, Shen J, Lau SK, Quake SR, Brown JM, Weissman IL, Clarke MF. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature. 2009;458:780–783. doi: 10.1038/nature07733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weinberg F, Hamanaka R, Wheaton WW, Weinberg S, Joseph J, Lopez M, Kalyanaraman B, Mutlu GM, Budinger GR, Chandel NS. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proc Natl Acad Sci U S A. 2010;107:8788–8793. doi: 10.1073/pnas.1003428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou R, Yazdi AS, Menu P, Tschopp J. A role for mitochondria in NLRP3 inflammasome activation. Nature. 2011;469:221–225. doi: 10.1038/nature09663. [DOI] [PubMed] [Google Scholar]
- 6.Brown DI, Griendling KK. Regulation of signal transduction by reactive oxygen species in the cardiovascular system. Circ Res. 2015;116:531–549. doi: 10.1161/CIRCRESAHA.116.303584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lin MT, Beal MF. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 2006;443:787–795. doi: 10.1038/nature05292. [DOI] [PubMed] [Google Scholar]
- 8.Shuhendler AJ, Pu K, Cui L, Uetrecht JP, Rao J. Real-time imaging of oxidative and nitrosative stress in the liver of live animals for drug-toxicity testing. Nat Biotechnol. 2014;32:373–380. doi: 10.1038/nbt.2838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Van de Bittner GC, Dubikovskaya EA, Bertozzi CR, Chang CJ. In vivo imaging of hydrogen peroxide production in a murine tumor model with a chemoselective bioluminescent reporter. Proc Natl Acad Sci U S A. 2010;107:21316–21321. doi: 10.1073/pnas.1012864107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shulman RG, Rothman DL. 13C NMR of intermediary metabolism: implications for systemic physiology. Annu Rev Physiol. 2001;63:15–48. doi: 10.1146/annurev.physiol.63.1.15. [DOI] [PubMed] [Google Scholar]
- 11.Gadian DG, Radda GK. NMR studies of tissue metabolism. Annu Rev Biochem. 1981;50:69–83. doi: 10.1146/annurev.bi.50.070181.000441. [DOI] [PubMed] [Google Scholar]
- 12.van der Graaf M. In vivo magnetic resonancespectroscopy: basic methodology and clinical applications. Eur Biophys J. 2010;39:527–540. doi: 10.1007/s00249-009-0517-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ardenkjaer-Larsen JH, Fridlund B, Gram A, Hansson G, Hansson L, Lerche MH, Servin R, Thaning M, Golman K. Increase of signal-to-noise ratio of more than 10,000 times in liquid state NMR. Proc Natl Acad Sci U S A. 2003;100:10158–10163. doi: 10.1073/pnas.1733835100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Golman K, Ardenkjaer-Larsen JH, Petersson JS, Mansson S, Leunbach I. Molecular imaging with endogenous substances. Proc Natl Acad Sci U S A. 2003;100:10435–10439. doi: 10.1073/pnas.1733836100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mayer D, Yen YF, Tropp J, Pfefferbaum A, Hurd RE, Spielman DM. Application of subsecond spiral chemical shift imaging to real-time multislice metabolic imaging of the rat in vivo after injection of hyperpolarized 13C1-pyruvate. Magn Reson Med. 2009;62:557–564. doi: 10.1002/mrm.22041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Park JM, Josan S, Grafendorfer T, Yen YF, Hurd RE, Spielman DM, Mayer D. Measuring mitochondrial metabolism in rat brain in vivo using MR Spectroscopy of hyperpolarized [2-C-13]pyruvate. NMR Biomed. 2013;26:1197–1203. doi: 10.1002/nbm.2935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rodrigues TB, Serrao EM, Kennedy BW, Hu DE, Kettunen MI, Brindle KM. Magnetic resonance imaging of tumor glycolysis using hyperpolarized C-labeled glucose. Nat Med. 2013;20:93–97. doi: 10.1038/nm.3416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Korenchan DE, Flavell RR, Baligand C, Sriram R, Neumann K, Sukumar S, VanBrocklin H, Vigneron DB, Wilson DM, Kurhanewicz J. Dynamic nuclear polarization ofbiocompatible 13C-enriched carbonates for in vivo pH imaging. Chem Commun. 2016;52:3030–3033. doi: 10.1039/c5cc09724j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Keshari KR, Wilson DM, Chen AP, Bok R, Larson PE, Hu S, Van Criekinge M, Macdonald JM, Vigneron DB, Kurhanewicz J. Hyperpolarized [2-13C]-Fructose: A Hemik et al DNP Substrate for In Vivo Metabolic Imaging. J Am Chem Soc. 2009;131:17591–17596. doi: 10.1021/ja9049355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Marco-Rius I, von Morze C, Sriram R, Cao P, Chang GY, Milshteyn E, Bok RA, Ohliger MA, Pearce D, Kurhanewicz J, Larson PE, Vigneron DB, Merritt M. Monitoring acute metabolic changes in the liver and kidneys induced by fructose and glucose using hyperpolarized [2-13 C]dihydroxyacetone. Magn Reson Med. 2017;77:65–73. doi: 10.1002/mrm.26525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Keshari KR, Kurhanewicz J, Bok R, Larson PE, Vigneron DB, Wilson DM. Hyperpolarized 13C dehydroascorbate as an endogenous redox sensor for in vivo metabolic imaging. Proc Natl Acad Sci U S A. 2011;108:18606–18611. doi: 10.1073/pnas.1106920108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bohndiek SE, Kettunen MI, Hu DE, Kennedy BWC, Boren J, Gallagher FA, Brindle KM. Hyperpolarized [1-13C]-Ascorbic and Dehydroascorbic Acid: Vitamin C as a Probe for Imaging Redox Status in Vivo. J Am Chem Soc. 2011;133:11795–11801. doi: 10.1021/ja2045925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lippert AR, Keshari KR, Kurhanewicz J, Chang CJ. A hydrogen peroxide-responsive hyperpolarized 13C MRI contrast agent. J Am Chem Soc. 2011;133:3776–3779. doi: 10.1021/ja111589a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schroder L, Lowery TJ, Hilty C, Wemmer DE, Pines A. Molecular imaging using a targeted magnetic resonance hyperpolarized biosensor. Science. 2006;314:446–449. doi: 10.1126/science.1131847. [DOI] [PubMed] [Google Scholar]
- 25.Bowers CR, Weitekamp DP. Parahydrogen and synthesis allow dramatically enhanced nuclear alignment. J Am Chem Soc. 1987;109:5541–5542. [Google Scholar]
- 26.Reineri F, Santelia D, Viale A, Cerutti E, Poggi L, Tichy T, Premkumar SSD, Gobetto R, Aime S. Para-hydrogenated Glucose Derivatives as Potential 13C-Hyperpolarized Probes for Magnetic Resonance Imaging. J Am Chem Soc. 2010;132:7186–7193. doi: 10.1021/ja101399q. [DOI] [PubMed] [Google Scholar]
- 27.Doroshow JH. Role of hydrogen peroxide andhydroxyl radical formation in the killing of Ehrlich tumor cells byanticancer quinones. Proc Natl Acad Sci U S A. 1986;83:4514–4518. doi: 10.1073/pnas.83.12.4514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takeyama K, Dabbagh K, Jeong Shim J, Dao-Pick T, Ueki IF, Nadel JA. Oxidative stress causes mucin synthesisvia transactivation of epidermal growth factor receptor: role ofneutrophils. J Immunol. 2000;164:1546–1552. doi: 10.4049/jimmunol.164.3.1546. [DOI] [PubMed] [Google Scholar]
- 29.Poulsen LL, Hyslop RM, Ziegler DM. S-Oxygenation of N-substituted Thioureas catalyzed by the pig liver microsomal FAD-containing monooxygenase. Arch Biochem Biophys. 1979;198:78–88. doi: 10.1016/0003-9861(79)90397-7. [DOI] [PubMed] [Google Scholar]
- 30.Zhao D, Sonawane ND, Levin MH, Yang B. Comparative transport efficiencies of urea analogues through urea transporter UT-B. Biochim miophys Acta, Biomembr. 2007;1768:1815–1821. doi: 10.1016/j.bbamem.2007.04.010. [DOI] [PubMed] [Google Scholar]
- 31.Chou CL, Sands JM, Nonoguchi H, Knepper MA. Concentration dependence of urea and thiourea transport pathway in rat inner medullary collecting duct. Am J Physiol. 1990;258:486–494. doi: 10.1152/ajprenal.1990.258.3.F486. [DOI] [PubMed] [Google Scholar]
- 32.Ziegler-Skylakakis K. Concise International Chemical Assessment Document 49: Thiourea. World Health Organization; Geneva: 2003. [Google Scholar]
- 33.Halliwell B, Gutteridge JMC. Free Radicals in Biology and Medicine. 5th. Oxford University Press; Oxford; 2015. [Google Scholar]
- 34.Rhee SG. Cell signaling. H2O2, a necessary evil for cell signaling. Science. 2006;312:1882–1883. doi: 10.1126/science.1130481. [DOI] [PubMed] [Google Scholar]
- 35.Paulsen CE, Carroll KS. Orchestrating redox signaling networks through regulatory cysteine switches. ACS Chem Biol. 2010;5:47–62. doi: 10.1021/cb900258z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Siddens LK, Krueger SK, Henderson MC, Williams DE. Mammalian flavin-containing mono-oxygenase (FMO) as a source of hydrogen peroxide. Biochem Pharmacol. 2014;89:141–147. doi: 10.1016/j.bcp.2014.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hoffmann M, Edwards JO. Kinetics andmechanism of the oxidation of thiourea and N, N′-dialkylthioureas byhydrogen peroxide. Inorg Chem. 1977;16:3333–3338. [Google Scholar]
- 38.Shang H, Skloss T, von Morze C, Carvajal L, Van Criekinge M, Milshteyn E, Larson PE, Hurd RE, Vigneron DB. Handheld electromagnet carrier for transfer of hyperpolarizedcarbon-13 samples. Magn Reson Med. 2016;75:917–922. doi: 10.1002/mrm.25657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arifoglu MA, Marmer WN, Dudley RB. Reaction of thiourea with hydrogen peroxide: 13C NMR studies of anoxidative/reductive bleaching process. Text Res J. 1992;62:94–100. [Google Scholar]
- 40.Makarov SV, Horvath AK, Silaghi-Dumitrescu R, Gao Q. Recent Developments in the Chemistry of Thiourea Oxides. Chem - Eur J. 2014;20:14164–14176. doi: 10.1002/chem.201403453. [DOI] [PubMed] [Google Scholar]
- 41.Giorgio M, Trinei M, Migliaccio E, Pelicci PG. Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat Rev Mol Cell Biol. 2007;8:722–728. doi: 10.1038/nrm2240. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


