Abstract
Rationale:
Neovascular glaucoma (NVG) is one of the most aggressive types of glaucoma, and its abnormal fibrovascular tissue growth on the iris and trabecular meshwork may create difficulties to control the intraocular pressure (IOP) and perform the operation such as trabeculectomy.
Patient concerns:
Ex-PRESS glaucoma shunt has been introduced to serve as one alternative operation for glaucoma, and is thought to have the potential advantage of being less traumatic than traditional trabeculectomy. The purpose of our study is to evaluate the efficacy and safety of primary Ex-PRESS implantation in eyes with NVG.
Diagnoses and interventions:
This was a retrospective study of four patients with NVG in whom primary Ex-PRESS implantation was performed between January 2012 and October 2016.
Outcomes:
After a mean follow-up of 20.8 ± 8.9 months (range, 15–34 months), three of four NVG patients (75.0%) receiving primary Ex-PRESS implantation had postoperative IOP under 21 mmHg without any antiglaucoma medication control at the last follow-up, but shunt reposition had been performed due to failed blebs or recurrent NVG. Besides, the best corrected visual acuity at the last follow-up was stabilized or improved in three of four NVG patients (75.0%), accompanied with the regression of iris neovascularization. Only one patient had self-resolved postoperative hyphema; otherwise, there were no other perioperative complications.
Lessons:
Primary Ex-PRESS implantation might constitute a safe and alternative treatment for patients with NVG. In spite of the possible reoperations, the final outcomes were still shown effective after shunt reposition.
Keywords: Ex-PRESS glaucoma shunt, glaucoma drainage device, neovascular glaucoma
1. Introduction
Glaucoma is the leading cause of irreversible blindness worldwide, and the number of people with glaucoma is predicted to increase from 64.3 million in 2013 to 111.8 million in 2040, especially in Asian and African populations.[1] Neovascular glaucoma (NVG), which results from abnormal fibrovascular tissue growth on the iris and trabecular meshwork, is one of the most aggressive types of glaucoma. The contraction of fibrovascular tissue will cause progressive angle closure and poorly controlled intraocular pressure (IOP), no matter what kind of topical drug is used.[2–5] There are various topical and oral antiglaucoma drugs, and surgery will usually be considered if medication and laser treatment cease to control IOP.[2–4] Trabeculectomy, the most common type of glaucoma filtration surgery, is considered the mainstay of incisional glaucoma surgeries.[4–6] Takihara et al demonstrated that the success rate of trabeculectomy for NVG was 62.6% 1 year after the operation, declining to 51.7% by 5 years of follow-up.[6] However, this technique is still associated with some postoperative complications, including hyphema, vitreous hemorrhage (VH), choroidal detachment, transient bleb leak, and endophthalmitis.[6–9]
In recent years, glaucoma drainage devices have gained popularity in the surgical treatment of NVG because their success is thought to be less dependent on control of intraocular inflammation and the failure of a filtering bleb.[3,4,10–31] Since the late 1990s, the Ex-PRESS glaucoma filtration device (Alcon Laboratories, Fort Worth, TX) has been introduced to serve as an alternative to trabeculectomy.[32–54] It is a nonvalved stainless steel implant that drains aqueous humor from the anterior chamber into the subconjunctival space. Generally, Ex-PRESS implantation is thought to have the potential advantage of being less traumatic than traditional trabeculectomy as it does not require a sclerotomy or peripheral iridotomy.[32–54] Due to potentially reduced intraoperative bleeding and postoperative inflammation, there might be more obvious benefits of performing Ex-PRESS surgery in patients with NVG. However, to the best of our knowledge, no study has exclusively evaluated the effect of primary Ex-PRESS implantation on NVG. Considering the potential treatment benefits, we then reviewed the IOP lowering efficacy and complication profile of the Ex-PRESS Mini Glaucoma Shunt, P200 model, placed under a scleral flap in our patients with NVG.
2. Materials and methods
2.1. Patient selection
This was a retrospective, noncomparative case series study. We reviewed the medical records of patients who had NVG presented with ocular hypertension and iris neovascularization (NVI), and hence received primary Ex-PRESS (P200 model) implantation between January 1, 2012 and October 31, 2016, at the Renai branch of the Taipei City Hospital in Taiwan. Slit lamp examinations were performed to exclude pupillary block, uveitic glaucoma, and other possible diagnoses of ocular hypertension. The indication for glaucoma surgery was the presence of persistent IOP ≥ 22 mm Hg or adverse symptoms with maximum-tolerated antiglaucoma therapy. Four eyes from 4 patients were included in the study. None of the patients had undergone iris laser therapy or an operation for glaucoma before. Informed consent was acquired from each patient, after a full explanation was provided, before the operation.
2.2. Surgical procedures
Under retrobulbar anesthesia, fornix-based peritomy and a 3 × 3 mm2 half-layer scleral flap were prepared in the upper half of sclera. The scleral flap was carefully extended to the blue-gray transition zone between the sclera and cornea. A piece of cotton soaked with 0.4 mg/mL mitomycin-C was then placed under the scleral flap for 4 minutes before irrigation with a balanced salt solution. With a 24-gauge needle, a penetration was made into the anterior chamber under the scleral flap. The Ex-PRESS P200 shunt was then inserted into the anterior chamber via the penetration site. The scleral flap was sutured at the corners and the overlying conjunctiva was sutured as well with 10-0 nylon interruptedly. Finally, the water-tightness of the bleb and the patency of the shunt were examined by inflating the anterior chamber with a balanced salt solution. All procedures were performed by 1 surgeon (GLT) and all patients in the study received at least 15 months of postoperative follow-up. Postoperative treatment consisted of steroid-antibiotic combination eye drops 4 times a day for 1 week.
If a failed bleb was found during follow-up, bleb revision was considered if IOP was poorly controlled by medication. If high IOP was still noted despite bleb revision, the reposition of the Ex-PRESS shunt to another site was considered with the same procedure, but with wider peritomy.
2.3. Data collection
Information on gender, age at the time of surgery, the etiology of NVG, the affected eye, lens status, IOP, Snellen best corrected visual acuity (BCVA), the grading of NVI, antiglaucoma agents used before and after the operation, surgical complications, and the follow-up period were collected. IOP was measured with a pneumotonometer (The Canon TX-F Full Auto Tonometer). NVI grading was conducted according to the grading system proposed by Teich and Walsh,[55] as shown in Table 1.
Table 1.
Grading of NVI (Teich and Walsh, 1981).[55]

2.4. Patient profiles and results
2.4.1. Patient 1
An 85-year-old female presented to our hospital in July 2013, with poorly controlled IOP and grade 2 NVI (Fig. 1A). She had previously suffered from rhegmatogenous retinal detachment (RD) (oculus sinister [OS]), and underwent pars plana vitrectomy (PPV) and silicone oil tamponade at another hospital in October 2011. Elevated IOP (OS) had been treated with anterior chamber tapping and cyclocryotherapy on December 7, 2012, but all in vain. Her IOP (OS) was still around 27.3 mm Hg with 4 kinds of antiglaucoma drugs. Poor BCVA of no light perception was also found in her left eye. Silicone oil removal (OS) was performed on August 1, 2013, but NVG (OS) was found on August 30, 2013. The IOP increased to 31.1 mm Hg, even with 4 kinds of antiglaucoma drugs, and Ex-PRESS implantation (OS) was then arranged on November 23, 2013.
Figure 1.
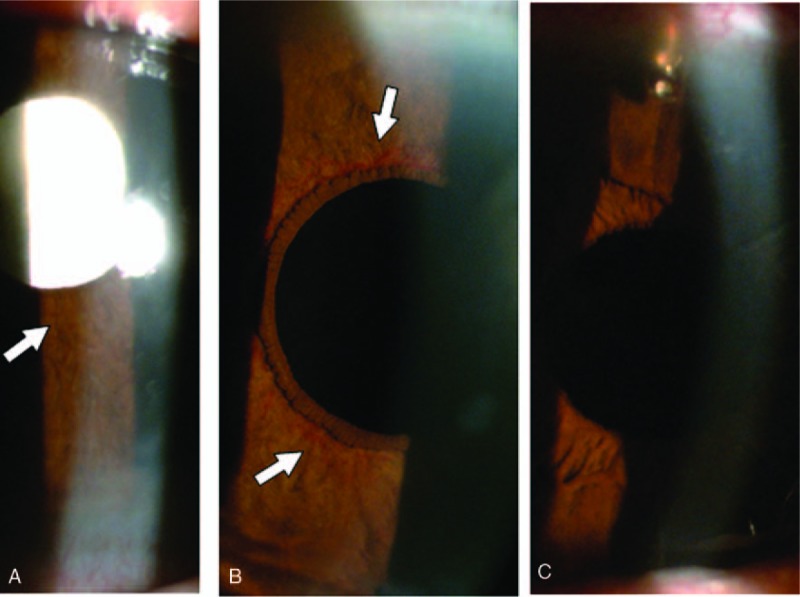
The NVI grading of Patient 1 before and after Ex-PRESS shunt implantation. (A) Grade 2 NVI (OS) was found before Ex-PRESS shunt implantation (see white arrow). (B) NVG recurrence (OS) with grade 2 NVI and poorly controlled IOP had occurred for 1 mo, and a shunt reposition was arranged (see white arrows). (C) NVI regression (OS) was noted 1 wk after shunt reposition. IOP = intraocular pressure, NVG = neovascular glaucoma, NVI = iris neovascularization, OS = oculus sinister.
Low IOP around 6 mm Hg with a formed anterior chamber had been found in the first week after surgery, but the IOP increased to 42.4 mm Hg with a flat bleb 1 more week later. Although IOP could be kept in the normal range with 3 kinds of medication, the patient could not tolerate the side effects. Bleb revision with mitomycin-C soaking was done on December 21, 2013. The IOP had been kept <21 mm Hg without antiglaucoma drugs for only 2 weeks, and a failed bleb with an increased IOP level of 36.4 mm Hg (OS) was found. The antiglaucoma drugs added up to 4 kinds to control IOP under 21 mm Hg, and could only be tapered to 2 kinds of antiglaucoma drugs later on. Her IOP still gradually climbed to 48.7 mm Hg with a recurrence of grade 2 NVI (OS) on March 6, 2015 (Fig. 1B), and the IOP could only be controlled at around 32 mm Hg with maximum-tolerated medical therapy.
Shunt reposition (OS) was performed on April 18, 2015 for the failed bleb and poor IOP control, and IOP of around 6 mm Hg with a formed anterior chamber was found during the first month after the operation. NVI regression was also found 1 week after shunt reposition (Fig. 1C). The IOP remained under 21 mm Hg most of the time after shunt reposition, except for 3 episodes of intermittent IOP fluctuation up to the highest level of 25.2 mm Hg that then came back to normal without medication control. The BCVA (OS) at the last follow-up remained the same, with no light perception, and the IOP (OS) was 21.8 mm Hg. The bleb was formed with no NVI found, and no antiglaucoma drug was given.
2.4.2. Patient 2
A 52-year-old male had end-stage renal disease under hemodialysis and type 2 diabetes mellitus with proliferative diabetic retinopathy (PDR), status post-panretinal photocoagulation (PRP) (oculus uterque [OU]). NVG (oculus dexter [OD]) with grade 2 NVI had emerged since November 2013 (Fig. 2A and B), and his Snellen BCVA was only 0.02. Although the NVI had gradually regressed to grade 0, IOP of up to 35 mm Hg was still observed under maximum-tolerated medical therapy for his right eye. Ex-PRESS implantation was performed on April 4, 2015, and IOP of 7.7 mm Hg with a formed anterior chamber was found the next day after surgery. Without further management, the IOP rose back to around 10 mm Hg 1 week after surgery and remained <21 mm Hg without antiglaucoma agents for 17 months. There was no NVI recurrence after the operation (Fig. 2C). However, a VH occurred on August 30, 2016, and subsided after 2 weeks. The Snellen BCVA (OD) at the last follow-up improved to 0.05, and the IOP in his right eye was 13.9 mm Hg without medication control.
Figure 2.
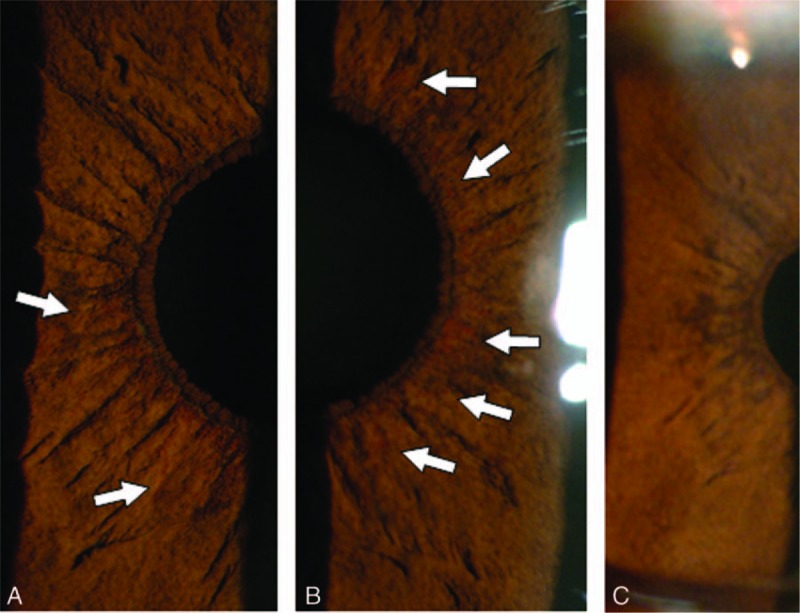
The NVI grading of Patient 2 before and after Ex-PRESS shunt implantation. (A, B) Grade 2 NVI (OD) was noted at its first appearance on November 22, 2013 (see white arrows). (C) No NVI recurrence was found after the operation. NVI = iris neovascularization, OD = oculus dexter.
2.4.3. Patient 3
Patient 3 was a 57-year-old male who also had type 2 diabetes mellitus with PDR and received PRP (OU) in 2012. He had received an intravitreal bevacizumab injection and PPV with focal laser photocoagulation for VH (OS) in 2014. In May 2015, poor control of diabetes was noted with a hemoglobin A1c level of 8.4%, and NVG (OD) emerged with a high IOP level of 51.8 mm Hg (Fig. 3A). The IOP was still high (up to 34.8 mm Hg) with maximum antiglaucoma drugs 2 weeks later, and the Snellen BCVA was only 0.02. Ex-PRESS implantation was then arranged on May 21, 2015. There was no hypotony or hyphema the next day after surgery, and the IOP was kept around 7.4 mm Hg, with a slightly shallow anterior chamber 1 week after surgery. Gradual NVI regression was also found during the follow-up, and full regression to grade 0 was noted 5 weeks after the operation (Fig. 3B and C). Without further treatment, the IOP had fluctuated between 8 and 12 mm Hg for 2 months and then gradually increased to 25 mm Hg in September 2015. Topical beta-blocker was administered, and the IOP decreased to 19.7 mm Hg about 6 weeks later.
Figure 3.
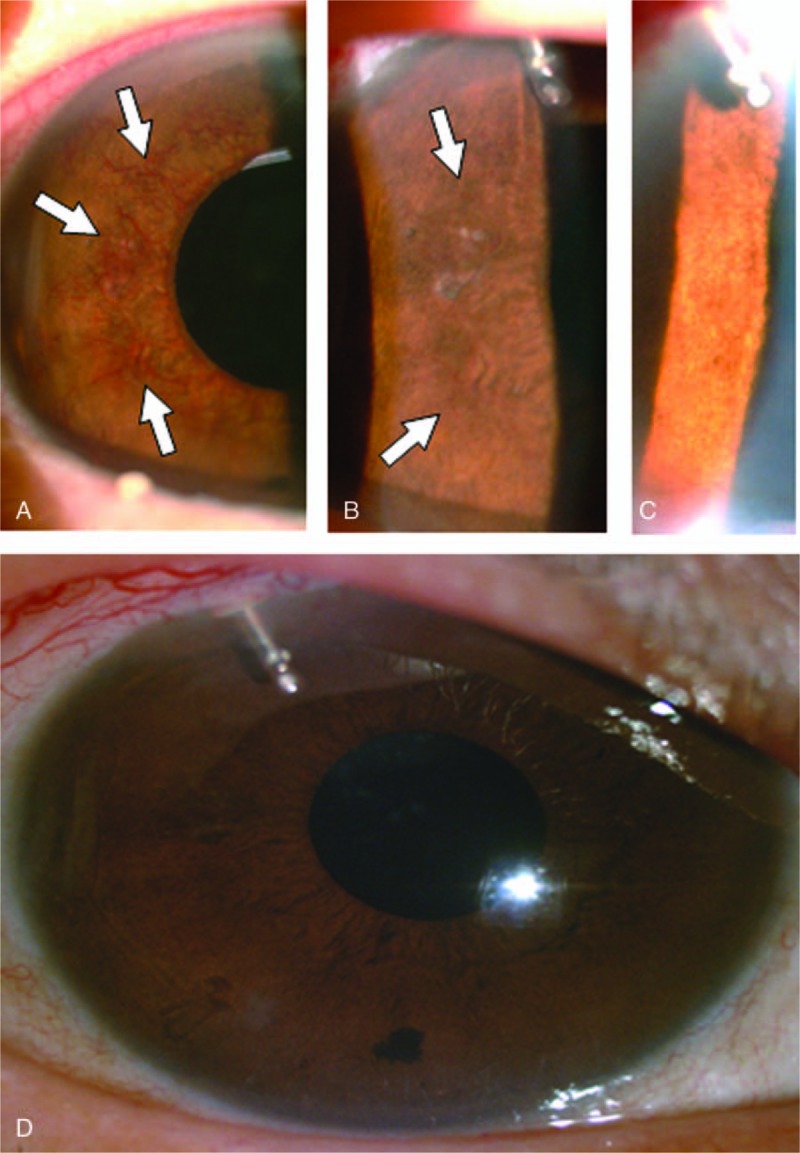
The NVI grading of Patient 3 before and after Ex-PRESS shunt implantation. (A) Grade 4 NVI (OD) was noted before Ex-PRESS shunt implantation (see white arrows). (B) Five days after the operation, the NVI had regressed to grade 3 (see white arrows). (C) Five weeks after the operation, the NVI had fully regressed to grade 0. (D) Although NVI recurred in May, 2016, gradual regression had been found during the 3-mo follow-up. NVI = iris neovascularization, OD = oculus dexter.
In addition, 3 episodes of VH (OD) emerged on June 8, 2015, June 30, 2015, and August 18, 2015. PPV with focal laser photocoagulation was done as needed. Recurrent NVG with a malfunctioning subconjunctival bleb had been noted since November 2015. Despite the administration of 3 kinds of antiglaucoma drugs, high IOP of up to 42.2 mm Hg was still found in January 2016, accompanied with recurrent VH (OD). Bleb revision (OD) was performed on January 18, 2016, and a low IOP level of 7.7 mm Hg with mild choroid detachment was noted the next day after surgery. Without medication control, the IOP had fluctuated between 7.9 and 19.3 mm Hg during the 2 weeks after surgery, and then remained within the normal range for 10 weeks. VH (OD) also resolved gradually. Nevertheless, NVG (OD) recurred with an IOP level of 28.5 mm Hg and a malfunctioning subconjunctival bleb about 4 months after bleb revision. The IOP showed no obvious decrease when treated with alpha-agonist and combination eye drops with beta-blocker and carbonic anhydrase inhibitor (CAI), despite the gradual regression of NVI (Fig. 3D). Shunt reposition (OD) was performed on August 29, 2016, and the IOP decreased to 15.7 mm Hg the next day after surgery.
However, macula-off RD and grade 2 NVI (OD) were found 3 weeks after shunt reposition, and the Snellen BCVA decreased from 0.3 to hand motion. The IOP (OD) was 7 mm Hg, accompanied by choroid detachment. PPV with internal drainage, endolaser photocoagulation, and C3F8 tamponade were then performed on October 17, 2016, and the retina was reattached. After the operation, the extent of grade 2 NVI decreased, and the IOP was 7.5 mm Hg.
2.4.4. Patient 4
A 68-year-old male had the underlying diseases of type 2 diabetes mellitus and chronic renal insufficiency. He came to our hospital on June 17, 2015 for a second opinion on the glaucoma control in his left eye. Upon his first appearance, his IOP level was <21 mm Hg under a combined treatment of eye drops with beta-blocker and CAI, along with oral acetazolamide 250 mg 3 times a day. However, NVI with a mid-dilated pupil and corneal edema (OS) were noted (Fig. 4A), suggesting IOP was still not well controlled. The BCVA in his left eye was only hand motion at 20 cm. After considering his underlying diseases that might not allow for the long-term administration of oral acetazolamide, we then arranged for an Ex-PRESS implantation (OS) on June 22, 2015.
Figure 4.
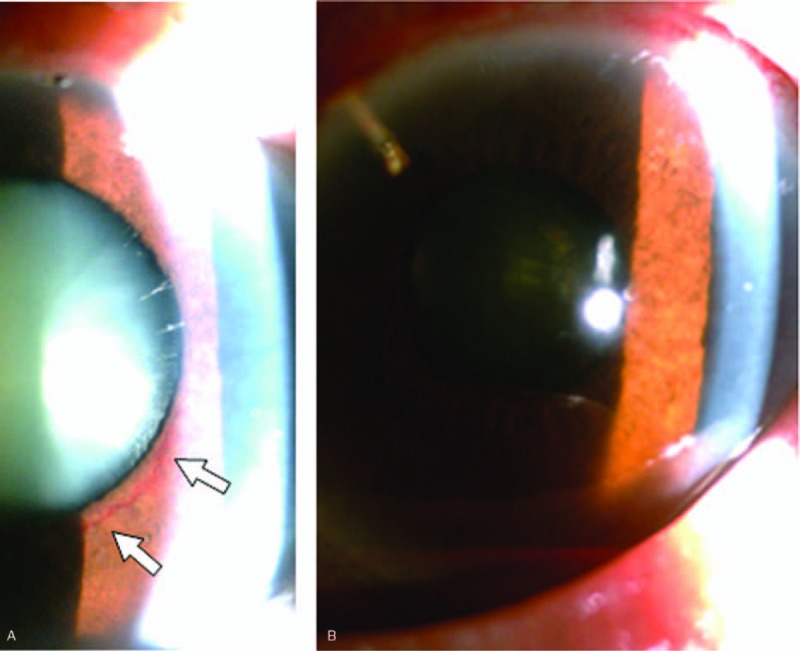
The NVI grading of Patient 4 before and after Ex-PRESS shunt implantation. (A) Grade 2 NVI (OS) was found before Ex-PRESS shunt implantation (see white arrows). (B) The extent of NVI (OS) had been decreasing 1 wk after Ex-PRESS shunt implantation, while some hyphema was not yet fully resolved. NVI = iris neovascularization, OS = oculus sinister.
The IOP was 6.4 mm Hg on the first day after operation, and there was also mild postoperative hyphema. However, a malfunctioning bleb and raised IOP level of 32.3 mm Hg (OS) were noted 1 week later, and we had to use 4 kinds of antiglaucoma drugs, including oral CAI, to control the IOP. The NVI had gradually regressed after Ex-PRESS implantation (Fig. 4B). Bleb revisions were performed twice for a failed bleb in July 2015, and NVG recurrence was found between 2 operations. Mild hyphema with an IOP level of 29.7 mm Hg was noted the next day after the second bleb revision. Although the IOP was maintained at <21 mm Hg with 3 kinds of topical eye drops and oral CAI, the failed bleb with NVI (OS) was still unresolved. Shunt reposition (OS) was hence performed on August 17, 2015, and the IOP in his left eye remained around 8 mm Hg most of the time, until the last follow-up in September 2016, with the lowest level of 5.1 mm Hg. No antiglaucoma drug had been used since shunt reposition. There was no associated complication of low IOP, and the NVI had fully regressed 3 months after shunt reposition. At the last follow-up, the BCVA in his left eye increased to hand motion at 60 cm, and the IOP (OS) was 7.8 mm Hg.
2.5. Summary of patient profiles
2.5.1. Baseline characteristics
The mean age of 4 patients was 65.5 ± 14.6 years. The most common cause of NVG in our study was diabetic retinopathy, which was found in 3 out of 4 eyes, and the other eye was caused by old RD under silicone oil tamponade. All 3 of these diabetic retinopathy patients had already received PRP before NVG appeared. The mean follow-up time was 20.8 ± 8.9 months (range: 15–34 months). The preoperative baseline characteristics of the patients are shown in Table 2.
Table 2.
Preoperative patient characteristics.

2.5.2. IOP results
After primary Ex-PRESS implantation, 2 eyes (Patient 1 and 4) had failed blebs, with ocular hypertension 2 weeks after the operation, and bleb revisions were arranged 1 month after the operation for both patients. Patient 3 also received bleb revision 8 months after the operation for NVG recurrence. However, all 3 patients received shunt reposition afterward due to the recurrence of NVG (Patient 1 and 3 at 16 and 7 months after bleb revision, respectively; Patient 4 at 1 month after bleb revision).
After shunt reposition, the IOP of these 3 patients became <21 mm Hg without medication afterward, except Patient 1, who had intermittent IOP fluctuation up to the highest level of 25.2 mm Hg, with the level then returning to the normal range without medication control. IOP of Patient 1 at the last follow-up was 21.8 mm Hg. However, there was no NVG recurrence or failed bleb. As for Patient 4, the IOP remained around 8 mm Hg without medication control for 13 months, with the lowest level of 5.1 mm Hg recorded about 1 month after shunt reposition.
Patient 3 had low IOP with choroid detachment after bleb revision, but the symptoms improved 1 week later. A similar episode occurred again after the first week following shunt reposition, but macula-off RD was further noted 3 weeks after the surgery.
The postoperative IOP of Patient 2 always remained in the normal range without medication for 17 months. A detailed IOP profile of these 4 patients is shown in Figure 5. At the last follow-up, with the exception of the IOP of Patient 1 that slightly exceeded 21 mm Hg, all patients’ IOP remained <21 mm Hg without antiglaucoma agents, as shown in Table 3.
Figure 5.
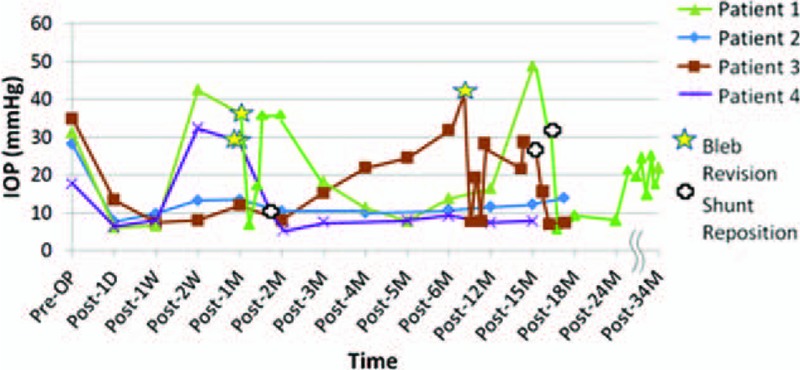
IOP changes of patients from baseline to at least 15 mo of follow-up. Out of 4 NVG patients receiving primary Ex-PRESS implantation, 3 had postoperative IOP <21 mm Hg without any antiglaucoma medication control after a mean follow-up of 20.8 ± 8.9 mo (range, 15–34 mo), although 3 out of 4 patients had received bleb revision and shunt reposition during follow-ups. IOP = intraocular pressure, NVG = neovascular glaucoma.
Table 3.
Postoperative patient characteristics at the last follow-up.

2.5.3. Visual acuity
Two of the 4 patients (Patient 2 and 4) had improved BCVA. Patient 3 also had increased BCVA after the operation, but the BCVA declined to only hand motion after the patient's episode of macula-off RD. The retina was reattached after PPV and long-standing C3F8 tamponade. The BCVA of Patient 1 still had no light perception after the surgery, the same as the condition before surgery. Pale optic discs were noted in Patients 1, 2, and 4. Patient 2 also had dystrophy of the retinal pigment epithelium in the macula. None of the 4 patients received subsequent cataract surgery during their follow-ups. The final BCVA of each patient is shown in Table 3.
2.5.4. Iris neovascularization
Preoperative NVI grading is shown in Table 2. After the operation, NVI regressed in all of the patients in about 1 month; however, NVG recurred with a failed bleb in 3 eyes (75.0%), except of Patient 2. The mean time interval between the operation and NVI recurrence in the 3 eyes was 7.3 months (Patient 1 at 15 months after the operation; Patient 3 at 6 months after the operation; Patient 4 at 1 month after the operation). Reoperations of bleb revision and shunt reposition were arranged for these 3 patients, and NVI gradually regressed again 1 week to 3 months after reoperation in all 3 patients. However, Patient 3 still had a recurrence of grade 2 NVI about 2 weeks after shunt reposition at the last follow-up. The results for all 4 patients are presented in Table 3. Except for Patient 3, no other patient received adjuvant laser therapy. Patient 3 had received 4 times of PPV with endolaser photocoagulation for VH due to PDR and RD, which were performed 4, 9, 16, and 17 weeks after Ex-PRESS implantation. Furthermore, none of these 4 patients received anti-vascular endothelial growth factor (VEGF) agents.
2.5.5. Surgical complications
After Ex-PRESS implantation, 1 patient (Patient 4) had mild postoperative hyphema that spontaneously resolved in 2 weeks; otherwise, there were no intraoperative or postoperative complications in any of the other patients, such as expulsive hemorrhage, hypotony, bleb leak, or endophthalmitis.
As for complications after reoperation, Patient 4 had mild, self-limited hyphema after bleb revision, and a low IOP of 5.1 mm Hg without associated complications 4 days after shunt reposition. Patient 3 also had a low IOP level of 7.7 mm Hg with choroid detachment the day after bleb revision, and the symptoms gradually resolved in 1 week. A similar episode occurred again in Patient 3 after the first week of shunt reposition, and macula-off RD was even found 3 weeks after the surgery. The retina was reattached after PPV with internal drainage and C3F8 tamponade.
3. Discussion
To the best of our knowledge, our study is the first one to exclusively evaluate the effect of primary Ex-PRESS implantation on NVG. There was only 1 related case report published in 2012, but no postoperative condition was discussed about.[56] Trabeculectomy has been thought to be the standard operation for glaucoma.[4–6] The surgical goal is to bypass the trabecular meshwork and allow aqueous humor to flow into the subconjunctival bleb, thereby reducing ocular hypertension. Nevertheless, in patients with NVG, neovascularization on the iris and trabecular meshwork will cause more complications during or after the operation.[3,4,6,9,57–62] In previous studies, the most common complication of trabeculectomy was hyphema. The incidence was around 30.0% and 58%, and significantly decreased to the levels around 10.0% and 33.3% if adjuvant anti-VEGF or peripheral iris cauterization was used.[6,9,25,57,59,61] In contrast with trabeculectomy, Ex-PRESS implantation is less invasive and causes less complications, subsequent inflammation, less IOP variance and faster recovery during the early postoperative period.[34–39,41,42,44,45,47,48,50–54] The biomolecular findings of 1 animal study also proved the similar results, which showed lower levels of transforming growth factor β2 after Ex-PRESS implantation under a scleral flap, compared with filtering sclerostomy in rabbits.[63] In our study, only 1 patient (Patient 4) had postoperative hyphema, and the extent of hyphema was mild enough that it spontaneously resolved in 2 weeks without surgical intervention. Another patient (Patient 3) had a low IOP level of 7.7 mm Hg with choroid detachment after bleb revision, but the IOP returned to the normal level with the application of a topical steroid after 1 week. However, Patient 3 suffered another similar episode without resolution 1 week after shunt reposition. Moreover, macula-off RD (OD) was also found 2 more weeks later, and the retina was attached back after PPV with internal drainage and C3F8 tamponade. No complications were noted after Ex-PRESS implantation in the other 2 patients (Patients 1 and 2).
In 2013, Wang et al systematically reviewed the medical literature on Ex-PRESS implantation, and the efficacy of IOP control was similar to trabeculectomy in treating uncontrolled glaucoma. Besides, significantly lower frequencies of hypotony and hyphema after Ex-PRESS implantation were also found.[52] However, Ex-PRESS implantation for NVG was not exclusively discussed in this and other previous studies. In regard to mitomycin-C trabeculectomy (MMCT) for NVG, several studies have been reported. Elgin and associates demonstrated the results of MMCT combined with direct cauterization of the peripheral iris, which showed that IOP was ≤22 mm Hg in 48 of 72 eyes (66.7%), with a relatively short follow-up period of 6 months.[57] Kiuchi and associates reported on the surgical results of 35 diabetic patients with a mean follow-up period of 25.6 months. The cumulative probability of success was 67.0% after 1 year and 61.8% after 2 to 3 years.[58] A retrospective, comparative case series conducted by Takihara and associates showed the probability of success 120, 240, and 360 days after MMCT of 87.5%, 79.2%, and 65.2% in 33 eyes with NVG.[59] Another study by Takihara reported the surgical results of 101 eyes with a mean follow-up period of 9.3 months. The probability of success 1, 2, and 5 years after MMCT was 62.6%, 58.2%, and 51.7%, respectively.[6]
In recent years, glaucoma drainage devices have been advocated for primary surgical treatment of NVG because their success is thought to be less dependent on control of intraocular inflammation and the failure of a filtering bleb.[3,4,10–31] Ahmed glaucoma valve is one of the most commonly used implants and its postoperative success with or without the control of topical antiglaucoma agents was 62.5% to 83.8% at 1 year, 59.3% to 74.7% at 2 years, and 68.5% at 3 years, respectively.[10,12,14–16,18,19,23,25,27] The 5-year success rates of Ahmed glaucoma valve implantation were varied, ranging from 20.6% to 70.0%.[12,26] In our study, a significant mean IOP reduction was observed in the first postoperative week, but 3 patients had failed blebs and recurrent NVG afterward. However, IOP of 2 of these 3 patients remained <21 mm Hg without medication after shunt reposition. At the last follow-up, IOP control ≤21 mm Hg without antiglaucoma agents was shown in 3 cases out of 4 patients (75.0%) in our study. The postoperative results did not seem to be worse than in the aforementioned studies, although a longer follow-up period and a larger case number might be needed to support this conclusion.
As for the final Snellen BCVA, 2 out of 4 patients (50%) in our study demonstrated improvement, though 1 patient reflected the same level of no light perception, as in the preoperative condition, and another patient (Patient 3) had deterioration due to macula-off RD. Before RD, the BCVA of Patient 3 was actually better than his preoperative BCVA. Compared to other interventions, 40.0% of NVG patients had improved BCVA after MMCT, whereas 46.7% of patients experienced deteriorated BCVA.[59] As for Ahmed valve implantation for NVG, the postoperative BCVA was stabilized or improved in 58.1% to 77.7% of the eyes.[10,16,30] The results of our final BCVA measurements also seemed to be not inferior than that of other studies.
In addition to achieving IOP reduction by medical and surgical means, the most critical treatment for effective long-term outcomes should be the reduction of the ischemic drive, which reduces the production of VEGF to prevent neovascularization.[3,4] Generally, prompt PRP to the ischemic retina and blood sugar control in diabetic patients are the mainstay treatments.[3–5] However, in patients with cloudy media that renders the fundus view inadequate for PRP, PPV with endolaser photocoagulation should be strongly considered.[4] In our study, with the exception of Patient 3, who received PPV with endolaser photocoagulation for VH and RD, other patients did not receive adjuvant laser or anti-VEGF therapy before NVI resolution. The mechanism of NVI resolution after Ex-PRESS implantation remained unclear due to the lack of a related study in the current literature. We suggested the possible explanation that NVI resolution might be caused by increased aqueous outflow through Ex-PRESS, resulting in a decreased VEGF level in the anterior chamber. Further studies, such as a molecular analysis, might be proposed to prove our hypothesis.
In this study, 3 out of 4 patients (75%) underwent a reoperation due to a failed bleb. These 3 patients had tried bleb revision with mitomycin-C soaking at least once, but all in vain. Shunt reposition was then performed, and almost all of these 3 patients had been able to maintain their IOPs at <21 mm Hg without medication control at the last follow-up, except Patient 1, who had a slightly elevated IOP of 21.8 mm Hg. This result suggested that the reoperation rate of Ex-PRESS implantation might be high in NVG patients, but also that the outcomes were still quite strong after shunt reposition.
Increased levels of VEGF expression in the aqueous humor and Tenon tissue of patients who had failed glaucoma surgeries had been found by previous studies, compared to patients for whom the surgery had been successful and patients without glaucoma.[11,64,65] A certain correlation between VEGF expression and the outcome of glaucoma surgery was suggested and the potential usefulness of anti-VEGF therapy was then considered in improving the success rate of glaucoma surgery.[11,64,65] Kim et al systematically reviewed the effect of subconjunctival or intravitreal injection of bevacizumab in combination with trabeculectomy, and increased bleb survival time was found in their related animal study.[5] However, Takihara et al also evaluated the effects of intravitreal bevacizumab before MMCT in 24 human eyes, and found no significant difference in bleb survival times.[59] Because only 1 dose of bevacizumab was administered before trabeculectomy, they suggested that the lack of improvement in the bleb survival rate might be related to the transient nature of bevacizumab. Despite the assumed limited effect, significantly less postoperative hyphema was found in the combination therapy of intravitreal bevacizumab and MMCT, and significantly lower IOPs were also found at 7 and 10 days. Other studies also showed the significantly reduced perioperative complications and favorable short-term outcomes in glaucoma surgeries combined with anti-VEGF injection, but the surgical outcomes for more than 0.5 to 1 year did not show significant improvement.[9,18,22,24–27,59,61] Takihara et al suggested that repeated adjuvant anti-VEGF injections might be considered in further studies to control for neovascularization and improve postoperative outcomes.[59] There was no systemic complication after the patients received intravitreal bevacizumab during follow-up for 360 days.[59] This concept of repeated perioperative anti-VEGF injections may be adapted for Ex-PRESS implantation in the future to increase success rate.
There are several limitations to our study. First, due to its retrospective nature, some recall bias might be considered while obtaining information from chart review, such as NVI grading. Furthermore, the NVI grading was determined by slit lamp examination in our study, instead of more precise methods, like fluoroangiography. The case number and relatively short postoperative follow-up might have impacted our results. However, this is the first study to evaluate Ex-PRESS implantation for NVG, and the results seemed to be no worse than with MMCT or other aqueous drainage devices. Generally, Ex-PRESS implantation is thought to be indicated for patients with open angles and not likely to be successful in the setting of NVG. This study might provide a different perspective, however, on NVG treatment by Ex-PRESS implantation.
In conclusion, our results showed almost all 4 of the NVG patients who received primary Ex-PRESS implantation had postoperative IOPs under 21 mm Hg without medication control at the last follow-up, although 3 of the 4 patients had received shunt reposition due to failed blebs or recurrent NVG. Most patients’ BCVAs showed improvement or no deterioration at the last follow-up, and NVI regression was also found in 3 of the 4 patients. Moreover, the perioperative complications of primary Ex-PRESS implantation were few and all self-limited. In summary, primary Ex-PRESS implantation might constitute a safe and alternative treatment for patients with NVG. Further studies with a longer follow-up period and a larger sample size are still needed to confirm our results.
Footnotes
Abbreviations: BCVA = best corrected visual acuity, CAI = carbonic anhydrase inhibitor, IOP = intraocular pressure, MMCT = mitomycin-C trabeculectomy, NVG = neovascular glaucoma, NVI = iris neovascularization, OD = oculus dexter, OS = oculus sinister, OU = oculus uterque, PDR = proliferative diabetic retinopathy, PPV = pars plana vitrectomy, PRP = panretinal photocoagulation, RD = retinal detachment, VEGF = vascular endothelial growth factor, VH = vitreous hemorrhage.
The authors have no funding and conflicts of interest to disclose.
References
- [1].Tham YC, Li X, Wong TY, et al. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology 2014;121:2081–90. [DOI] [PubMed] [Google Scholar]
- [2].Allingham RRSM. Shields’ Textbook of Glaucoma. Philadelphia, PA: Ovid Technologies Inc; 2011. [Google Scholar]
- [3].Sivak-Callcott JA, O’Day DM, Gass JD, et al. Evidence-based recommendations for the diagnosis and treatment of neovascular glaucoma. Ophthalmology 2001;108:1767–76. [DOI] [PubMed] [Google Scholar]
- [4].Olmos LC, Lee RK. Medical and surgical treatment of neovascular glaucoma. Int Ophthalmol Clin 2011;51:27–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Kim M, Lee C, Payne R, et al. Angiogenesis in glaucoma filtration surgery and neovascular glaucoma: a review. Surv Ophthalmol 2015;60:524–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Takihara Y, Inatani M, Fukushima M, et al. Trabeculectomy with mitomycin C for neovascular glaucoma: prognostic factors for surgical failure. Am J Ophthalmol 2009;147:912–8. [DOI] [PubMed] [Google Scholar]
- [7].Jampel HD, Solus JF, Tracey PA, et al. Outcomes and bleb-related complications of trabeculectomy. Ophthalmology 2012;119:712–22. [DOI] [PubMed] [Google Scholar]
- [8].Saeedi OJ, Jefferys JL, Solus JF, et al. Risk factors for adverse consequences of low intraocular pressure after trabeculectomy. J Glaucoma 2014;23:e60–8. [DOI] [PubMed] [Google Scholar]
- [9].Alkawas AA, Shahien EA, Hussein AM. Management of neovascular glaucoma with panretinal photocoagulation, intravitreal bevacizumab, and subsequent trabeculectomy with mitomycin C. J Glaucoma 2010;19:622–6. [DOI] [PubMed] [Google Scholar]
- [10].Faghihi H, Hajizadeh F, Mohammadi SF, et al. Pars plana Ahmed valve implant and vitrectomy in the management of neovascular glaucoma. Ophthalmic Surg Lasers Imaging 2007;38:292–300. [DOI] [PubMed] [Google Scholar]
- [11].Kim YG, Hong S, Lee CS, et al. Level of vascular endothelial growth factor in aqueous humor and surgical results of Ahmed glaucoma valve implantation in patients with neovascular glaucoma. J Glaucoma 2009;18:443–7. [DOI] [PubMed] [Google Scholar]
- [12].Netland PA. The Ahmed glaucoma valve in neovascular glaucoma (an AOS thesis). Trans Am Ophthalmol Soc 2009;107:325–42. [PMC free article] [PubMed] [Google Scholar]
- [13].Rush R. Ciliary sulcus Ahmed Glaucoma Valve tube placement in neovascular glaucoma. Ophthalmic Surg Lasers Imaging 2009;40:489–92. [DOI] [PubMed] [Google Scholar]
- [14].Yildirim N, Yalvac IS, Sahin A, et al. A comparative study between diode laser cyclophotocoagulation and the Ahmed glaucoma valve implant in neovascular glaucoma: a long-term follow-up. J Glaucoma 2009;18:192–6. [DOI] [PubMed] [Google Scholar]
- [15].Netland PA, Ishida K, Boyle JW. The Ahmed Glaucoma Valve in patients with and without neovascular glaucoma. J Glaucoma 2010;19:581–6. [DOI] [PubMed] [Google Scholar]
- [16].Park UC, Park KH, Kim DM, et al. Ahmed glaucoma valve implantation for neovascular glaucoma after vitrectomy for proliferative diabetic retinopathy. J Glaucoma 2011;20:433–8. [DOI] [PubMed] [Google Scholar]
- [17].Jeong HS, Nam DH, Paik HJ, et al. Pars plana Ahmed implantation combined with 23-gauge vitrectomy for refractory neovascular glaucoma in diabetic retinopathy. Korean J Ophthalmol 2012;26:92–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Ma KT, Yang JY, Kim JH, et al. Surgical results of Ahmed valve implantation with intraoperative bevacizumab injection in patients with neovascular glaucoma. J Glaucoma 2012;21:331–6. [DOI] [PubMed] [Google Scholar]
- [19].Teixeira SH, Doi LM, Freitas Silva AL, et al. Silicone Ahmed glaucoma valve with and without intravitreal triamcinolone acetonide for neovascular glaucoma: randomized clinical trial. J Glaucoma 2012;21:342–8. [DOI] [PubMed] [Google Scholar]
- [20].Cheng Y, Liu XH, Shen X, et al. Ahmed valve implantation for neovascular glaucoma after 23-gauge vitrectomy in eyes with proliferative diabetic retinopathy. Int J Ophthalmol 2013;6:316–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Mahdy RA, Nada WM, Fawzy KM, et al. Efficacy of intravitreal bevacizumab with panretinal photocoagulation followed by Ahmed valve implantation in neovascular glaucoma. J Glaucoma 2013;22:768–72. [DOI] [PubMed] [Google Scholar]
- [22].Sevim MS, Buttanri IB, Kugu S, et al. Effect of intravitreal bevacizumab injection before Ahmed glaucoma valve implantation in neovascular glaucoma. Ophthalmologica 2013;229:94–100. [DOI] [PubMed] [Google Scholar]
- [23].Zhou MW, Wang W, Huang WB, et al. Adjunctive with versus without intravitreal bevacizumab injection before Ahmed glaucoma valve implantation in the treatment of neovascular glaucoma. Chin Med J 2013;126:1412–7. [PubMed] [Google Scholar]
- [24].Kang JY, Nam KY, Lee SJ, et al. The effect of intravitreal bevacizumab injection before Ahmed valve implantation in patients with neovascular glaucoma. Int Ophthalmol 2014;34:793–9. [DOI] [PubMed] [Google Scholar]
- [25].Arcieri ES, Paula JS, Jorge R, et al. Efficacy and safety of intravitreal bevacizumab in eyes with neovascular glaucoma undergoing Ahmed glaucoma valve implantation: 2-year follow-up. Acta Ophthalmol 2015;93:e1–6. [DOI] [PubMed] [Google Scholar]
- [26].Sahyoun M, Azar G, Khoueir Z, et al. Long-term results of Ahmed glaucoma valve in association with intravitreal bevacizumab in neovascular glaucoma. J Glaucoma 2015;24:383–8. [DOI] [PubMed] [Google Scholar]
- [27].Tang M, Fu Y, Wang Y, et al. Efficacy of intravitreal ranibizumab combined with Ahmed glaucoma valve implantation for the treatment of neovascular glaucoma. BMC Ophthalmol 2016;16:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Eid TE, Katz LJ, Spaeth GL, et al. Tube-shunt surgery versus neodymium:YAG cyclophotocoagulation in the management of neovascular glaucoma. Ophthalmology 1997;104:1692–700. [DOI] [PubMed] [Google Scholar]
- [29].Chalam KV, Gandham S, Gupta S, et al. Pars plana modified Baerveldt implant versus neodymium:YAG cyclophotocoagulation in the management of neovascular glaucoma. Ophthalmic Surg Lasers 2002;33:383–93. [PubMed] [Google Scholar]
- [30].Yalvac IS, Eksioglu U, Satana B, et al. Long-term results of Ahmed glaucoma valve and Molteno implant in neovascular glaucoma. Eye 2007;21:65–70. [DOI] [PubMed] [Google Scholar]
- [31].Eid TM, Radwan A, el-Manawy W, et al. Intravitreal bevacizumab and aqueous shunting surgery for neovascular glaucoma: safety and efficacy. Can J Ophthalmol 2009;44:451–6. [DOI] [PubMed] [Google Scholar]
- [32].Dahan E, Carmichael TR. Implantation of a miniature glaucoma device under a scleral flap. J Glaucoma 2005;14:98–102. [DOI] [PubMed] [Google Scholar]
- [33].Coupin A, Li Q, Riss I. Ex-PRESS miniature glaucoma implant inserted under a scleral flap in open-angle glaucoma surgery: a retrospective study. J Fr Ophtalmol 2007;30:18–23. [DOI] [PubMed] [Google Scholar]
- [34].Maris PJ, Jr, Ishida K, Netland PA. Comparison of trabeculectomy with Ex-PRESS miniature glaucoma device implanted under scleral flap. J Glaucoma 2007;16:14–9. [DOI] [PubMed] [Google Scholar]
- [35].Filippopoulos T, Rhee DJ. Novel surgical procedures in glaucoma: advances in penetrating glaucoma surgery. Curr Opin Ophthalmol 2008;19:149–54. [DOI] [PubMed] [Google Scholar]
- [36].Hendrick AM, Kahook MY. Ex-PRESS mini glaucoma shunt: surgical technique and review of clinical experience. Expert Rev Med Dev 2008;5:673–7. [DOI] [PubMed] [Google Scholar]
- [37].De Feo F, Bagnis A, Bricola G, et al. Efficacy and safety of a steel drainage device implanted under a scleral flap. Can J Ophthalmol 2009;44:457–62. [DOI] [PubMed] [Google Scholar]
- [38].de Jong LA. The Ex-PRESS glaucoma shunt versus trabeculectomy in open-angle glaucoma: a prospective randomized study. Adv Ther 2009;26:336–45. [DOI] [PubMed] [Google Scholar]
- [39].Gallego-Pinazo R, Lopez-Sanchez E, Marin-Montiel J. Postoperative outcomes after combined glaucoma surgery. Comparison of ex-press miniature implant with standard trabeculectomy. Arch Soc Esp Oftalmol 2009;84:293–7. [DOI] [PubMed] [Google Scholar]
- [40].Kanner EM, Netland PA, Sarkisian SR, Jr, et al. Ex-PRESS miniature glaucoma device implanted under a scleral flap alone or combined with phacoemulsification cataract surgery. J Glaucoma 2009;18:488–91. [DOI] [PubMed] [Google Scholar]
- [41].Bissig A, Feusier M, Mermoud A, et al. Deep sclerectomy with the Ex-PRESS X-200 implant for the surgical treatment of glaucoma. Int Ophthalmol 2010;30:661–8. [DOI] [PubMed] [Google Scholar]
- [42].Freedman J. What is new after 40 years of glaucoma implants. J Glaucoma 2010;19:504–8. [DOI] [PubMed] [Google Scholar]
- [43].Vetrugno M, Ferreri P, Sborgia C. Ex-PRESS miniature glaucoma device in vitrectomized eyes. Eur J Ophthalmol 2010;20:945–7. [DOI] [PubMed] [Google Scholar]
- [44].de Jong L, Lafuma A, Aguade AS, et al. Five-year extension of a clinical trial comparing the EX-PRESS glaucoma filtration device and trabeculectomy in primary open-angle glaucoma. Clin Ophthalmol 2011;5:527–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Francis BA, Singh K, Lin SC, et al. Novel glaucoma procedures: a report by the American Academy of Ophthalmology. Ophthalmology 2011;118:1466–80. [DOI] [PubMed] [Google Scholar]
- [46].Good TJ, Kahook MY. Assessment of bleb morphologic features and postoperative outcomes after Ex-PRESS drainage device implantation versus trabeculectomy. Am J Ophthalmol 2011;151: 507–13.e501. [DOI] [PubMed] [Google Scholar]
- [47].Lankaranian D, Razeghinejad MR, Prasad A, et al. Intermediate-term results of the Ex-PRESS miniature glaucoma implant under a scleral flap in previously operated eyes. Clin Exp Ophthalmol 2011;39:421–8. [DOI] [PubMed] [Google Scholar]
- [48].Marzette L, Herndon LW. A comparison of the Ex-PRESS mini glaucoma shunt with standard trabeculectomy in the surgical treatment of glaucoma. Ophthalmic Surg Lasers Imaging 2011;42:453–9. [DOI] [PubMed] [Google Scholar]
- [49].Sarkisian SR., Jr The Ex-PRESS miniature glaucoma device and the glaucoma surgery treatment algorithm. Clin Exp Ophthalmol 2011;39:381–2. [DOI] [PubMed] [Google Scholar]
- [50].Dahan E, Ben Simon GJ, Lafuma A. Comparison of trabeculectomy and Ex-PRESS implantation in fellow eyes of the same patient: a prospective, randomised study. Eye 2012;26:703–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Seider MI, Rofagha S, Lin SC, et al. Resident-performed Ex-PRESS shunt implantation versus trabeculectomy. J Glaucoma 2012;21:469–74. [DOI] [PubMed] [Google Scholar]
- [52].Wang W, Zhou M, Huang W, et al. Ex-PRESS implantation versus trabeculectomy in uncontrolled glaucoma: a meta-analysis. PLoS One 2013;8:e63591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Mariotti C, Dahan E, Nicolai M, et al. Long-term outcomes and risk factors for failure with the EX-press glaucoma drainage device. Eye 2014;28:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Netland PA, Sarkisian SR, Jr, Moster MR, et al. Randomized, prospective, comparative trial of EX-PRESS glaucoma filtration device versus trabeculectomy (XVT study). Am J Ophthalmol 2014;157: 433–40.e433. [DOI] [PubMed] [Google Scholar]
- [55].Teich SA, Walsh JB. A grading system for iris neovascularization. Ophthalmology 1981;88:1102–6. [DOI] [PubMed] [Google Scholar]
- [56].Angmo D, Sobti A, Panda A. The surgical treatment of neovascular glaucoma with Ex-PRESS P-50 Miniature Glaucoma Shunt. J Curr Glaucoma Pract 2012;6:13–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Elgin U, Berker N, Batman A, et al. Trabeculectomy with mitomycin C combined with direct cauterization of peripheral iris in the management of neovascular glaucoma. J Glaucoma 2006;15:466–70. [DOI] [PubMed] [Google Scholar]
- [58].Kiuchi Y, Sugimoto R, Nakae K, et al. Trabeculectomy with mitomycin C for treatment of neovascular glaucoma in diabetic patients. Ophthalmologica 2006;220:383–8. [DOI] [PubMed] [Google Scholar]
- [59].Takihara Y, Inatani M, Kawaji T, et al. Combined intravitreal bevacizumab and trabeculectomy with mitomycin C versus trabeculectomy with mitomycin C alone for neovascular glaucoma. J Glaucoma 2011;20:196–201. [DOI] [PubMed] [Google Scholar]
- [60].Al Obeidan SA, Osman EA, Al-Amro SA, et al. Full preoperative panretinal photocoagulation improves the outcome of trabeculectomy with mitomycin C for neovascular glaucoma. Eur J Ophthalmol 2008;18:758–64. [DOI] [PubMed] [Google Scholar]
- [61].Kitnarong N, Chindasub P, Metheetrairut A. Surgical outcome of intravitreal bevacizumab and filtration surgery in neovascular glaucoma. Adv Ther 2008;25:438–43. [DOI] [PubMed] [Google Scholar]
- [62].Kiddee W, Tantisarasart T, Wangsupadilok B. Neovascular glaucoma: a retrospective review of 5-year experience in Songklanagarind Hospital. J Med Assoc Thai [Chotmaihet thangphaet] 2012;95(suppl 4):S36–42. [PubMed] [Google Scholar]
- [63].Sampson EM, Esson DW, Schultz GS, et al. Expression of transforming growth factor-b2 following sclerostomy and ExPRESS R glaucoma drainage implant beneath a scleral flap in a rabbit model. Invest Ophthalmol Vis Sci 2005;46:52–152. [Google Scholar]
- [64].Park H, Kim J, Ahn M, et al. Level of vascular endothelial growth factor in tenon tissue and results of glaucoma surgery. Arch Ophthalmol 2012;130:685–9. [DOI] [PubMed] [Google Scholar]
- [65].Li Z, Van Bergen T, Van de Veire S, et al. Inhibition of vascular endothelial growth factor reduces scar formation after glaucoma filtration surgery. Invest Ophthalmol Vis Sci 2009;50:5217–25. [DOI] [PubMed] [Google Scholar]


