Abstract
Wnt/β-catenin signaling is instrumental for the development of mammary gland and the properties of mammary stem cells (MaSCs). The Wnt signaling downstream effectors that engage in regulating MaSCs have not been extensively studied. Here, we report that Neuropilin-1 (Nrp1) expression is induced by Wnt/β-catenin signaling in MaSCs, and its function is critical for the activity of MaSCs. Nrp1 is particularly expressed in MaSCs that are marked by the expression of Protein C Receptor (Procr). Knockdown of Nrp1 by shRNA diminishes MaSCs’ in vitro colony formation and in vivo mammary gland reconstitution ability. Similar results are seen when antagonizing Nrp1 using a dominant negative peptide. In genetic experiments, deletion of Nrp1 results in delay of mammary development. In addition, knockdown of Nrp1 inhibits MMTV-Wnt1 tumor growth in xenograft. Our data demonstrate that Nrp1 is critical for mammary development and tumorigenesis, revealing new insights into MaSC regulation and targeting stem cells in treatment of breast cancer.
Introduction
The mammary gland is an epithelial organ, with a tree-like pattern of ductal networks. The majority of mammary development occurs postnatally. At the onset of puberty at around 3 weeks of age in mice, in response to ovarian hormones, the preexisting rudimentary ductal tree rapidly expands and extends across the fat pad, occupying the whole mammary fat pad by approximately 7 weeks of age1. Highly elongated basal cells and cuboidal luminal cells compose the two main cellular lineages of the nulliparous and non-pregnant mammary gland. The basal cell population (Lin−, CD24+, CD29hi/CD49fhi) is able to generate new mammary glands in transplantation assays, thus representing a mammary stem cells (MaSCs)-enriched population2, 3. More recently, study from our lab reveals a more refined MaSC population that is marked by the expression of Protein C Receptor (Procr). Procr+ MaSCs are composed of about 3–8% of total basal cells depending on the genetic background. Procr+ MaSCs have the highest reconstitution efficiency in transplantation assays compared to total basal cells and other known basal subpopulation4.
Wnt/β-catenin signaling has been implicated in almost all stages of mammary development and is instrumental for MaSC self-renewal and expansion activities (reviewed in refs 5–7). Studies have directly addressed Wnts as niche factors for MaSCs8, 9. In 3D Matrigel cultures, addition of Wnt3A or Wnt4 proteins to MaSC-enriched basal cell culture can maintain stem cell properties and promote MaSC expansion. The retention of stem cell properties is demonstrated by the ability of the cultured cells to efficiently reconstitute mammary glands in transplantation8, 9. In an attempt to identify Wnt targets specifically expressed in MaSCs, microarray analysis of cultured MaSC-enriched basal cells was performed, leading to the discovery of the MaSC specific surface marker Procr4.The microarray analysis also suggests other new Wnt downstream target genes in mammary epithelial cells, which are potentially critical for the activities of MaSCs.
Neuropilin-1 (Nrp1) is a single-pass transmembrane glycoproteins, with a small cytoplasmic domain and multiple extracellular domains10. Nrp1 binds to a variety of ligand families, functioning as co-receptors in a complex with other transmembrane receptors11. The class 3 semaphorins (SEMA3) and vascular endothelial growth factor (VEGF) family are well established ligands for Nrp112, 13. Evidence has revealed that the Nrp1 also interacts with other growth factors11. Nrp1 and it close family member Nrp2 are mostly known for the regulation of cell motility, particularly with respect to neural and vascular development12–17. Nrp1 may play a role in epithelial cells as well. Robust Nrp1 expression has been found in human epithelial tumor cells derived from lung, breast, prostate, pancreatic, and colon carcinomas11. Nrp1 has also been implicated in the migration and survival of breast cancer cells18–20, however its potential role in MaSCs and in normal mammary development remains elusive. In this study, we identified Nrp1 as a novel target of Wnt/β-catenin signaling. We showed that the expression of Nrp1 is enriched in Procr+ MaSCs, and that Nrp1 plays an essential role in MaSC property maintenance and MMTV-Wnt1 mammary tumor growth.
Results
Nrp1 is upregulated by Wnt signaling in Procr+ MaSCs
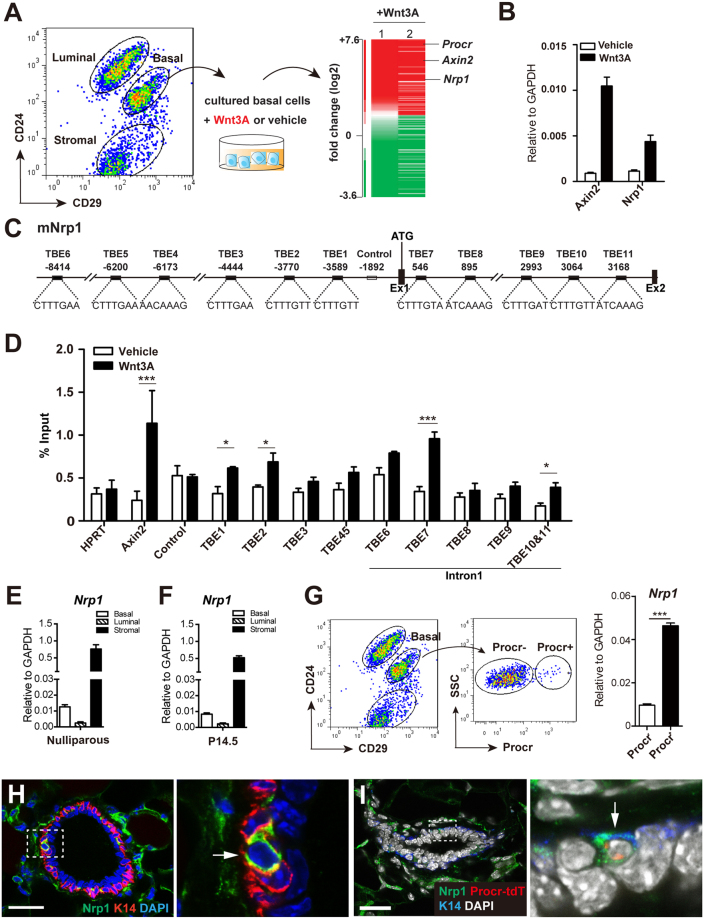
Previous studies established in vitro culture system in which MaSC properties can be maintained using purified Wnt proteins8. In this culture system, mammary basal cells (Lin−, CD24+, CD29hi) were isolated using fluorescence-activated cell sorting (FACS) and cultured in 3D Matrigel in the presence or absence of Wnt3A proteins4. Microarray was performed using the cultured cells to identify downstream effectors of Wnt signaling in regulating MaSCs (Fig. 1A). Among the candidates whose expressions were increased in the presence of Wnt3A, which included Axin2 and Procr, we identified Nrp1 (Fig. 1A). Quantitative PCR (qPCR) confirmed that Nrp1 expression is upregulated by Wnt3A treatment (Fig. 1B). Upregulation of Axin2 in this condition served as a positive control (Fig. 1B).
Figure 1.
Nrp1 is upregulated by Wnt signaling in MaSCs. (A) Mammary basal cells were FACS-sorted from 8-week-old nulliparous mammary gland and cultured in 3D Matrigel in the presence of Wnt3A protein or vehicle. Microarray analysis of the cultured cells indicated that Nrp1 was upregulated with Wnt3A treatment. 1 and 2 represented two independent experiments. (B) qPCR analysis validating the increased expression of Nrp1 in Wnt3A treated cells. Axin2 serves as a positive control. (C) Schematic illustration of the promoter and first intron of mouse Nrp1. TCF-binding elements (TBEs) and negative control region are indicated. Ex, Exon; Int, Intron. (D) ChIP-qPCR analysis with anti-β-catenin antibody. qPCR results were normalized to HPRT. Axin2 promoter region with known effective TEB serves as positive control. (E,F) qPCR analysis of nulliparous mammary gland (E) and pregnant day 14.5 (P14.5) (F) indicating that Nrp1 expression is higher in basal cells compared to luminal cells, and it has the highest expression in stromal cells. (G) qPCR analysis of FACS-isolated Procr+ and Procr− basal cells from nulliparous mammary gland indicating that Nrp1 is mainly expressed in Procr+ basal cells. Data are pooled from three independent experiments, are presented as mean ± SEM. ***P < 0.001. (H) Immunohistochemistry indicating that Nrp1 is expressed in a subpopulation of basal cells (arrow). Basal cells are marked by the expression of Keratin 14 (K14) in red. (I) Immunohistochemistry indicating Nrp1 expression (green) in a tdTomato+ Procr-expressing cell (Procr-tdT). Scale bar, 20 μm.
Sequence analysis revealed six putative TCF-binding elements (TBEs) at the promoter and distal region and five putative TBEs at the first intron of Nrp1 (Fig. 1C). To test whether the β-catenin can directly associated withNrp1, we performed chromatin immunoprecipitation (ChIP) assays. We used β-catenin antibody to immunoprecipitate chromatin DNA in mouse mammary cells and examined specific binding of the following: 9 separate fragments containing the TBE of Nrp1; a fragment without TBE at Nrp1 promoter region which acted as a negative control; and a fragment with one TBE at the Axin2 promoter as a positive control. As shown in Fig. 1D, the β-catenin antibody immunoprecipitated TBE1 and TBE2 at the promoter, and TEB7, TBE 10/11 at the intron, whereas no precipitation was observed in the negative control. These data suggest that Nrp1 is a target of the Wnt/β-catenin pathway in mammary cells.
To investigate the expression pattern of Nrp1 in the mammary gland, we isolated basal (Lin−, CD24+, CD29hi), luminal (Lin−, CD24+, CD29lo) and stromal cells (Lin−, CD24−) from 8-week old nulliparous mice, and analyzed Nrp1 levels by qPCR. We found that in the epithelial compartment, Nrp1 is markedly higher in basal cells than that in luminal cells, while it displays the highest level in stromal cells (Fig. 1E). Similar expression patterns were observed in mammary gland at pregnant day 14.5 (Fig. 1F). The expression of Nrp1 was further examined in basal cells in higher resolution. MaSCs (Lin−, CD24+, CD29hi, Procr+) and the other basal cells (Lin−, CD24+, CD29hi, Procr−) were isolated from mature nulliparous female mice. We found that Nrp1 is expressed at a higher level (4.5 fold increase) in Procr+ MaSCs by qPCR analysis (Fig. 1G). Immunofluorescent staining in mammary gland section confirmed the Nrp1 expression pattern. We observed that a small population of basal cells is Nrp1+ (5.48 ± 1.93%), whereas no Nrp1+ cells are found in luminal cells (n = 1,000 basal or luminal cells) (Fig. 1H). Of note, a large number of Nrp1+ cells were seen in the stromal compartment (Fig. 1H), which is consistent with the qPCR results. To investigate whether Nrp1+ basal cells coincide with Procr+ MaSCs, we utilized Procr-tdTomato knock-in (ProcrCreERT2-IRES-tdTomato) mouse model, in which tdTomato serves as a reporter for Procr expression4. Immunostaining indicated that there is a substantial overlap of Nrp1 and tdTomato expression in basal compartment, in which about 34% of Procr+ basal cells were Nrp1+ (116 out of 348 Procr+ basal cells) (Fig. 1I). This can be explained by the potential heterogeneity of the Procr+ basal cells. In contrast, very few Procr− basal cells were Nrp1+ (11 out of 838 Procr− basal cells, 1.3%). Together, these data suggest that in the epithelial compartment, Nrp1 is highly expressed in Procr+ MaSCs.
Nrp1 is critical for MaSC activities in vitro and in vivo
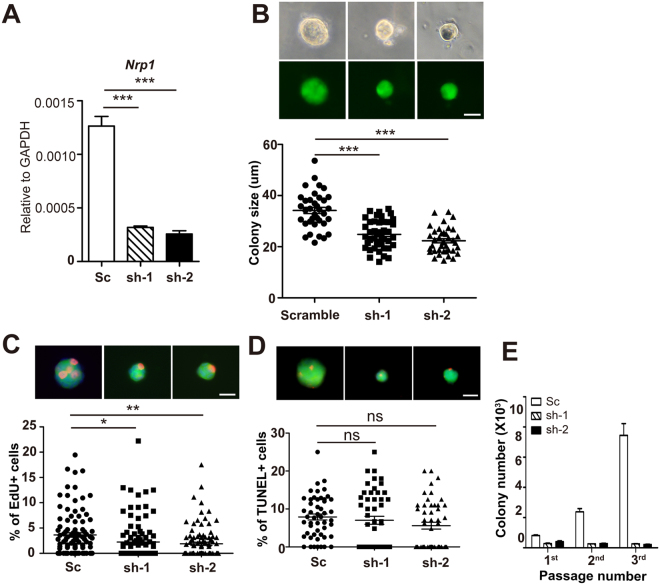
Next, we investigated the role of Nrp1 in MaSCs in vitro. Isolated basal cells from nulliparous mice were infected with lentivirus containing GFP tagged Nrp1 shRNA. Infected cells were placed in 3D Matrigel culture and the formed colonies were visualized by GFP. To minimize potential off-target effect, two independent sh-RNAs were constructed for Nrp1 knockdown. The knockdown efficiency of sh-Nrp1 was confirmed by qPCR analysis (Fig. 2A). We observed that knockdown of Nrp1 results in reduction of colony sizes (Fig. 2B), suggesting a decreased proliferation. Reduced EdU incorporation in Nrp1 knockdown cells corroborated with the idea (Fig. 2C). In addition, TUNEL staining suggested no discernable difference in cell death upon Nrp1 knockdown (Fig. 2D). Furthermore, we found that colony numbers were decreased in serial passages when Nrp1 expression is inhibited (Fig. 2E), suggesting that the numbers of stem cell-like cells were decreased in each passage. These data suggest that Nrp1 is critical for MaSCs in vitro colony formation.
Figure 2.
Nrp1 expression is important for MaSC colony formation in vitro. (A) qPCR analysis validating the Nrp1 knockdown efficiency of two independent shRNAs. Data are pooled from three independent experiments, are presented as mean ± SEM. ***P < 0.001. (B) Representative images of colonies upon infection with scramble or Nrp1 shRNAs (sh-1, sh-2) viruses (top panels), and quantification of colony sizes at cultured day 6 (bottom panels). Each dot represents one colony. Scale bar, 20 μm. Data are presented as mean ± SEM. ***P < 0.001. (C) Representative images of EdU labeled basal colonies (top panels). EdU+ cell percentages (EdU+ cells/DAPI+ cells in each colony) were quantified at day 6 (bottom panel). Each dot represents one colony. Scale bar, 20μm. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01. (D) Representative images of TUNEL staining of basal colonies (top panels). TUNEL+ cell percentages (TUNEL+ cells/DAPI+ cells in each colony) were quantified at day 6 (bottom panels). Each dot represents one colony. Scale bar, 20μm. Data are presented as mean ± SEM. (E) Serial passage of basal colonies after lentiviral infection with scramble or Nrp1 shRNAs. The colony numbers of each passage were quantified.
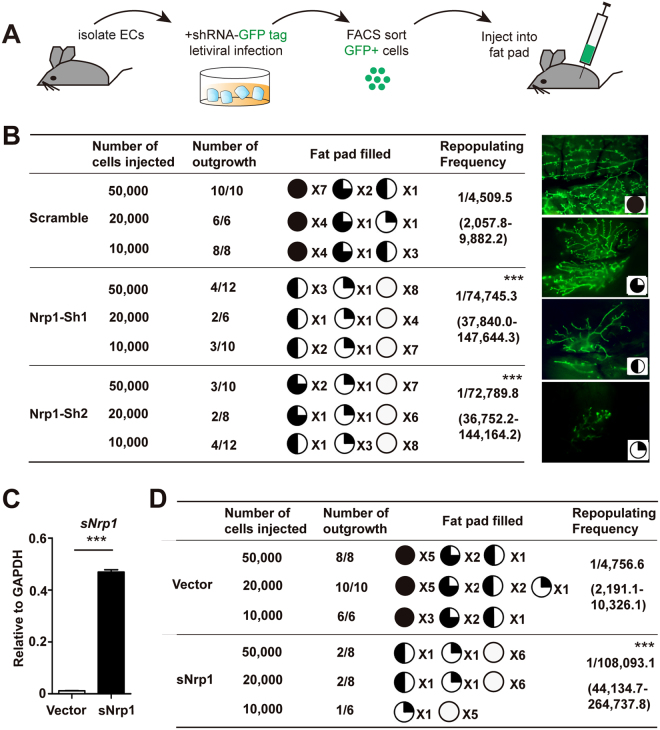
We next investigated the impact of Nrp1 knockdown on MaSCs in transplantation assays. Mammary cells were isolated and virally infected by Nrp1 shRNA with GFP tag, the successfully infected cells were FACS-isolated by GFP expression and transplanted to cleared fat pads of recipients (illustrated in Fig. 3A). We found that knockdown of Nrp1 by either shRNA leads to significantly decreased repopulating efficiency, reduction of fat pad filling (Fig. 3B). To determine whether the ligand binding function of Nrp1 is important for MaSCs, we utilized the extracellular domain of Nrp1, a soluble form of Nrp1 (sNrp1), which inhibits the association of Nrp1 with its ligands21. sNrp1 was overexpressed in MaSCs using GFP tagged lentivirus, infected cells were FACS-isolated and transplanted to cleared fat pads. Enhanced expression of sNrp1 was confirmed by qPCR analysis (Fig. 3C). Consistently, sNrp1 overexpression resulted in reduced repopulation efficiency and fat pad filling (Fig. 3D), suggesting a decrease in MaSC numbers. Together, these data suggest that Nrp1 is critical for the regenerative capacity of MaSCs in vivo.
Figure 3.
Inhibition of Nrp1 attenuates MaSCs’ reconstitution ability in vivo. (A) Schematic illustration of transplantation assay setup. Mammary epithelial cells (ECs) were isolated from 8-week-old nulliparous mice, followed by infection with scramble or Nrp1 shRNAs lentivirus. After 6 days of culture, positively infected cells (GFP+) were FACS isolated and transplanted into the cleared fat pad of 3-week-old nude mice and allowed for the formation of mammary outgrowth. (B) 10,000 or 20,000 or 50,000 of infected cells were injected into each of the cleared fat pad, and the mammary outgrowth were examined at 8 weeks post transplantation. Number of outgrowths and percentage of fat pad filled are shown. Representative images of different percentage of fat pad filled are shown on the right panels. Data are pooled from three independent experiments. (C) qPCR analysis of soluble Nrp1 (sNrp1) expression in infected (GFP+) epithelial cells. (D) 10,000 or 20,000 or 50,000 of infected cells with vector or sNrp1 overexpression were injected into each of the cleared fat pad, and the mammary outgrowths were examined at 8 weeks post transplantation. Data were pooled from three independent experiments. Data are presented as mean ± SEM. ***P < 0.0001.
Nrp1 is important for mammary development
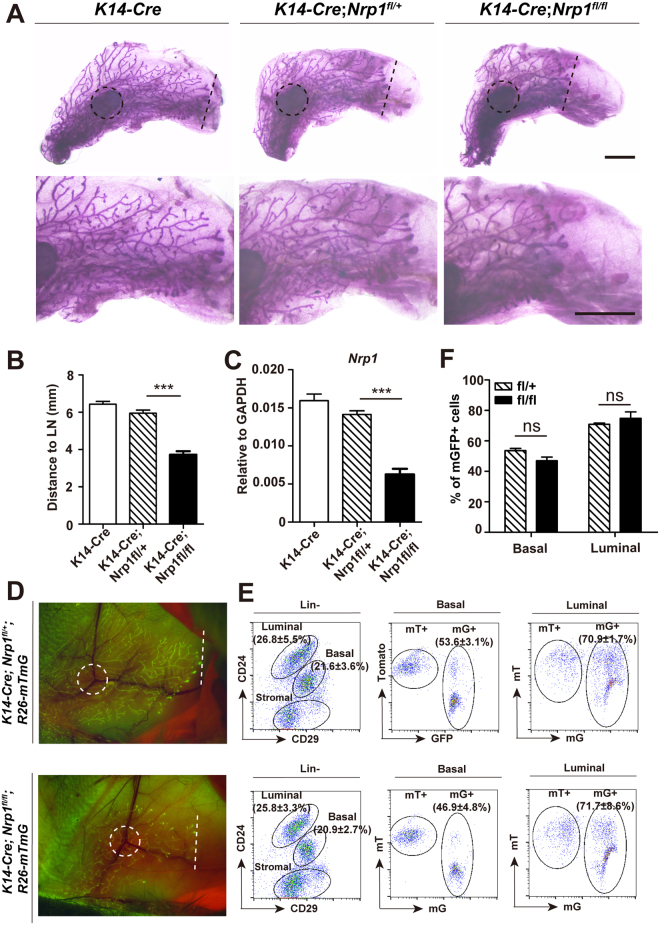
To investigate the function of Nrp1 in development, we utilized the Nrp1 flox allele12, and generated K14-Cre;Nrp1 fl/fl through genetic crosses. K14 is expressed in all mammary epithelial cells during embryonic development, subsequently it is restricted in basal cells after birth. Thus, K14-Cre primarily facilitated deletion of Nrp1 in mammary basal cells. The 4th pair of mammary glands were harvested at 7-week old. We observed a delay in ductal extension in K14-Cre;Nrp1 fl/fl mice, compared to K14-Cre and K14-Cre;Nrp1 fl/+ littermate controls (Fig. 4A,B). qPCR analysis in FACS-isolated basal cells confirmed the decrease of Nrp1 expression (Fig. 4C). These results suggest that Nrp1 is important for mammary development. Next, we investigated whether Nrp1 influences lineage commitment using K14-Cre;Nrp1 fl/fl;R26-mTmG mice. The expressed Cre recombinase in basal cells would result in excision of the stop cassette in the reporter, thus marking the basal cells and their progeny, including luminal cells, with membrane-bound GFP (mG). The mammary glands were harvested at 6-week old. In whole mount imaging, consistently we observed a delayed ductal extension phenotype in K14-Cre;Nrp1 fl/fl;R26-mTmG when compared to the control (Fig. 4D). FACS analysis of the isolated primary cells indicated that 46.9% of basal cells and 71.7% of luminal cells expression mG (Fig. 4E). There were no significant differences of mG+ cell percentage compared to the control (Fig. 4E,F), suggesting that Nrp1 deletion does not affect basal luminal cell fate commitment.
Figure 4.
Loss of Nrp1 in basal cells compromises mammary development. (A) Representative images of whole mount carmine staining for 7-week old K14-Cre, K14-Cre;Nrp1 fl/+ and K14-Cre;Nrp1 fl/fl mice. Dash line, forefront of epithelium extension; dash line circle, lymph node (L.N.). Scale bar, top panels, 2mm; bottom panels, 2mm. (B) Quantification of the mammary duct extension in K14-Cre, K14-Cre;Nrp1 fl/+ and K14-Cre;Nrp1 fl/fl mice. The distance from L.N. to epithelium forefront was measured. n = 3 mice in each group. Data are presented as mean ± SEM. ***P < 0.0001. (C) qPCR analysis validating the Nrp1 knockout efficiency using FACS-isolated basal cells. (D) Whole mount imaging of mammary glands with lineage-traced mGFP at 6-week old. (E,F) FACS analysis indicating the proportion of basal and luminal cells, and the percentages of mG+ cells in basal and luminal compartments (E). Quantification indicating no significant differences of mG+ cell percentage in either basal or luminal compartment when comparing the Nrp1 knockout (fl/fl) and the Ctrl (fl/+) (F). n = 3 mice in each group.
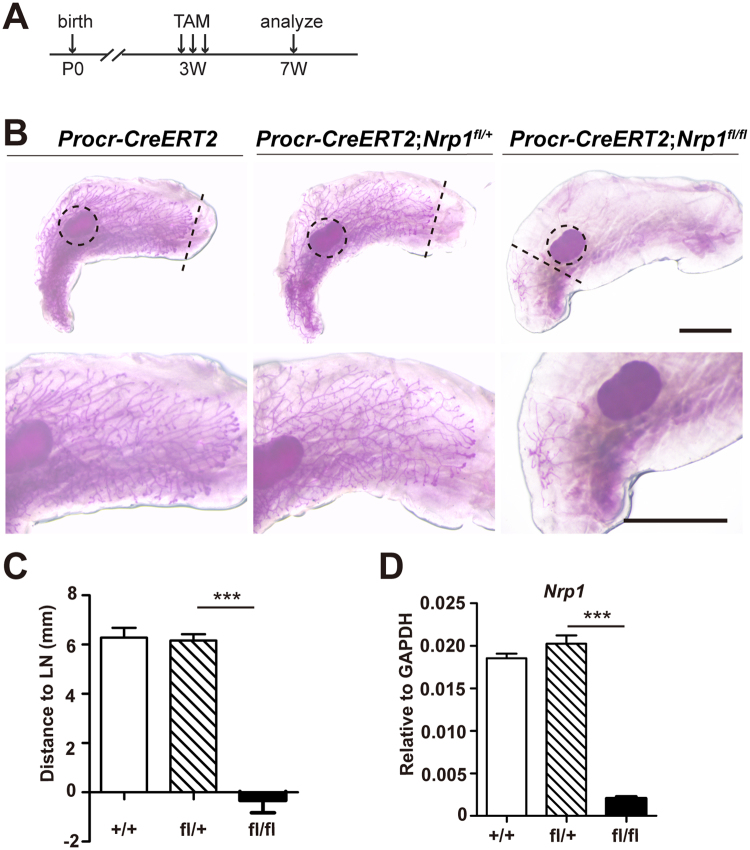
Towards a more specific deletion of Nrp1 in MaSCs, we generated Procr-CreERT2;Nrp1 fl/fl mice. Tamoxifen (TAM) was administered in pubertal mice at 3-week old every other day for 3 times, the phenotype resulted from Nrp1 deletion was evaluated at 4 weeks later (Fig. 5A). At 7-week old, the TAM-treated control mammary glands (Procr-CreERT2 and Procr-CreERT2;Nrp1 fl/+) had almost completed the epithelium extension and occupied the whole fat pad (Fig. 5B). Strikingly, deletion of Nrp1 in Procr-CreERT2;Nrp1 fl/fl mice completely prevented the growth of the epithelium: the forefront of the epithelium halted at a position where the forefront was at the initiation of Nrp1 deletion (at around the lymph node) (Fig. 5B,C). qPCR analysis in FACS-isolated basal cells confirmed the decrease of Nrp1 expression (Fig. 5D). These data reinforced that Nrp1 plays an important functional role in MaSC and its deficiency affects mammary development.
Figure 5.
Loss of Nrp1 in MaSCs severely delays mammary development. (A) Experimental setup for Nrp1 conditional knockout using Procr-CreERT2 line. TAM was administrated at 3-week at every other day for a total of three times, mammary glands were harvest at 7-week. (B) Whole mount carmine staining of 7-week old Procr-CreERT2, Procr-CreERT2;Nrp1 fl/+ and Procr-CreERT2;Nrp1 fl/fl mammary glands. Dash line, epithelial front; dash line circle, LN. Scale bars, 2 mm. (C) Quantification of the mammary duct extension in Procr-CreERT2 (+/+), Procr-CreERT2;Nrp1 fl/+ (fl/+) and Procr-CreERT2;Nrp1 fl/fl (fl/fl) mice. The distance from L.N. to epithelium forefront was measured. n = 4 mice in each group. Data are presented as mean ± SEM. ***P < 0.0001. (D) qPCR analysis validating the Nrp1 knockout efficiency in FACS-isolated basal cells. Data are presented as mean ± SEM. ***P < 0.0001.
Nrp1 knockdown inhibits MMTV-Wnt1 tumor growth
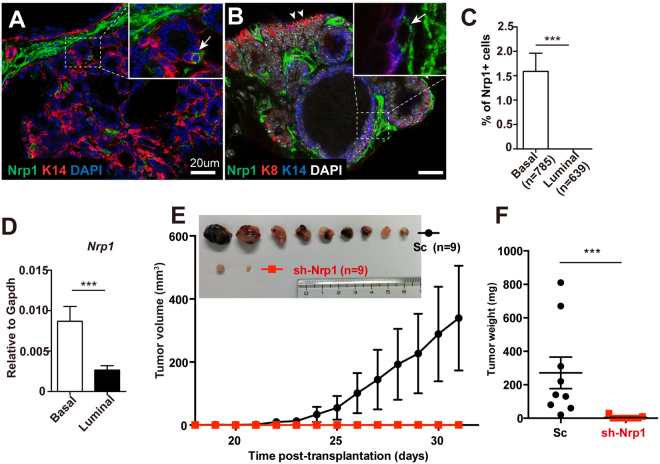
MMTV-Wnt1 mammary tumor is a malignancy that has close association with MaSCs2, 22. Considering the important role of Nrp1 in MaSCs, we examined its impact in MMTV-Wnt1 tumor growth. Immunofluoescent staining in tumor section suggested a sporadic pattern of Nrp1+ cells in MMTV-Wnt1 tumor epithelial compartment (Fig. 6A). Immunostaining of lineage marker suggested that Nrp1+ cells are exclusively expressed in K14+ basal cells (Fig. 6A–C). qPCR analysis of FACS-isolated tumor cells confirmed the Nrp1 expression pattern (Fig. 6D). To investigate the role of Nrp1 in tumorigenesis, isolated MMTV-Wnt1 mammary tumor cells were infected by Nrp1 shRNA. The infected cells that are marked by GFP expression were engrafted to the fat pad of nude recipients. We found that Nrp1 knockdown drastically inhibits tumor formation and attenuates tumor growth, when compared with the scramble control (Fig. 6E,F). These data suggest that Nrp1 plays an important role in MMTV-Wnt1 tumor growth.
Figure 6.
Knockdown of Nrp1 inhibits MMTV-Wnt1 tumor growth. (A–C) Immunostaining indicating the expression of Nrp1 (green) in MMTV-Wnt1 tumor sections. Basal cells were marked by K14 expression (red in A, blue in B), luminal cells were marked by K8 (red in B). Nrp1+K14+ cells were indicated by arrows (A and B). K8+ cells were indicated by arrowheads (B). The percentages of Nrp1+ cells were quantified in basal and luminal populations (C). Scale bar, 20 μm. Data are presented as mean ± SEM. ***P < 0.0001. (D) qPCR analysis of FACS-isolated basal and luminal cells in the tumor indicating Nrp1 expression is higher in basal cells. Data are pool from three independent experiments and presented as mean ± SEM. ***P < 0.0001. (E,F) Primary cells were isolated from MMTV-Wnt1 tumor and infected with scramble (Sc) or Nrp1 shRNA (sh-Nrp1) lentivirus. After 6 days of culture, infected cells were FACS-sorted and injected into fat pad of 8-week-old nude mice. Tumor volume was measured as indicated (E). Inserted image showing the tumors harvested at day 31 post surgery (E). Tumor weights were measured at day 31 post surgery (F). Data are pooled from three independent experiments and presented as mean ± SEM. ***P < 0.0001.
Discussion
Nrp1 have been studied extensively for their roles in neural development and to some extent vascular development, but their potential contribution to epithelial tissues has not been investigated extensively. In this study, we investigated the functional role of Nrp1 in mammary development. We revealed the connection of Wnt/β-catenin signaling and Nrp1 expression in the mammary gland. In the epithelium compartment, Nrp1 was mainly expressed in Procr+ MaSCs. In vitro colony formation and in vivo transplantation experiments demonstrated that Nrp1 is critical for the activity of MaSCs. We provided genetic evidence that deletion of Nrp1 compromises mammary development. Furthermore, inhibition of Nrp1 attenuated MMTV-Wnt1 mammary tumor growth.
The specific Nrp1 expression in Procr+ MaSCs was suggested by FACS analysis with elaborated surface markers and qPCR analysis, and further confirmed by immunofluorescent staining. Nrp1 was highly expressed in mesenchymal cells surrounding the mammary epithelium, while Nrp1 in the epithelial compartment was mainly expressed in Procr+ basal cells. Considering Procr+ MaSCs are localized in ducts, and are excluded from the TEBs in development mammary gland4, it is logical that Nrp1 expression would not be found in TEB epithelium. These analysis in high resolution and conclusions are contrary to a previous report, in which individual TEB or duct segments were isolated under dissecting microscope, followed by microarray and qPCR analysis, which led to a conclusion that Nrp1 is equally expressed in both TEB and duct23. Consider the abundant Nrp1 expression in surrounding mesenchymal cells revealed in current study, we believe that the Nrp1 expression detected from isolated mammary fragments (comprising both epithelium and mesenchyme) are most likely contributed by the Nrp1 in mesenchymal compartment.
Nrp1 has a structural homolog, Nrp2, with 44% amino acid identity. They differ in their regulation and function (reviewed in ref. 11). Nrp2 has also been reported to contribute towards branching morphogenesis of mammary epithelial cells24. Nrp2 is expressed preferentially in the TEBs of developing glands. Deletion of Nrp2 using MMTV-Cre results in defects in branching morphogenesis24. VEGF165, an isoform of VEGF, induces Nrp2-dependent branching morphogenesis as demonstrated in vitro 3D culture model with a mouse mammary cell line24. In this study, we found that Nrp1 is particularly expressed in Procr+ MaSCs, distinct from the TEB enriched expression of Nrp2; Nrp1 is a Wnt-target gene in the mammary epithelium while Nrp2 is not (data not shown). Our data demonstrated the critical role of Nrp1 for the activities of MaSCs and the development of mammary gland, yet the mechanism by which Nrp1 functions in MaSC remains to be elucidated. Mammary epithelial cells express VEGF24, implying a possibility that VEGF is a potential ligand for Nrp1 in MaSCs. Considering that Nrps function as co-receptor for other growth factor, including fibroblast growth factor (FGF) family25, galectin-126, hepatocyte growth factor/scatter factor (HGF/SF)27–29, transforming growth factor-β1 (TGF-β1)30, 31, platelet-derived growth factor (PDGF)32, and integrin receptors33, thus it is possible that Nrp1 also mediates the impact of other ligands in MaSCs.
Excessive expression of both NRPs has been detected in breast cancer cells19, 34, associated with poor prognosis35, 36. Overexpression of NRPs is shown to enhance growth and correlate with invasion in various tumor types, including breast cancer18–20, 36, 37. In this study, our data demonstrated that Nrp1 is expressed in MaSCs of normal mammary tissues. Knockdown of Nrp1 expression in MMTV-Wnt1 inhibited the growth of mammary tumors. Our findings reinforced the close association between MMTV-Wnt1 mammary tumor and MaSCs2, 22, and suggested Nrp1 as a target to inhibit this type of breast cancer.
In conclusion, our results demonstrated the upregulation of Nrp1 by Wnt/β-catenin signaling in the mammary epithelium, exhibited the specific expression of Nrp1 in MaSCs, and revealed its critical functional roles in mammary development and tumorigenesis. Results presented in this study reinforce the connection of Wnt/β-catenin signaling and the function of MaSC, providing new insight into targeting stem cells in breast cancer through antagonizing Nrp1.
Experimental Material and Procedures
Experimental mice
The K14-Cre, Procr-CreERT2-IRES-tdTomato, Nrp1 flox and MMTV-Wnt1 mice were used. Nrp1 flox mice were purchased from Jax (005247). All experimental mice were treated according to the protocols approved by the Animal Care and Use Committee of Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences.
Isolation of primary mammary cells
The fat pads were isolated from 8-week-old nulliparous or other specified-stage female mice. Fat pads were minced and placed in appropriate volume (1 g/10 ml) digestion mix (RPMI 1640 with 25 mM HEPES, 5% fetal bovine serum, 1% penicillin-streptomycin-glutamine, 300 U/ml type 3 collagenase (Worthington) and digested for 2 h in a 100 rpm/min shaker at 37 °C, give a good shake every 15 min. Cells were pelleted and treated with red blood lysis buffer (Sigma) for 5 min, single cell suspension was obtained by subsequential incubation with 0.25% trypsin-EDTA at 37 °C for 5 min and 0.1 mg/ml DNase I (Sigma) for 5 min with gentle pipetting, and then filtrated through 70-μm cell strainer.
Cell labeling and flow Cytometry
For cell labeling, the following antibodies were used: FITC conjugated CD31, CD45, TER119 (BD Pharmingen); CD24-PE-Cy7, CD29-APC (Biolegend), Procr-PE (eBioscience). Antibody incubation was performed on ice for 15 min in PBS with 5% fetal bovine serum. Before the FACS sorting, cells were filtrated through 45-μm cell strainer, and cells were sorted by using a FACSJazz (BD).
Preparation of vectors and lentivirus
To construct shRNA vectors, the annealed oligonucleotides were ligated to pLKO.1-EGFP, which was modified by replacement of the puromycin-resistance gene with EGFP. The target sequences of shRNAs were listed: Scramble shRNA: 5′-TCCTAAGGTTAAGTCGCCCTCG-3′; Nrp1 sh1: 5′-TGGCTGCAAGATAACAGATTA-3′; Nrp1 sh2: 5′-CCAGAGAATCATGATCAACTT-3′. The extracellular domain of mouse Nrp1 (NM_008737.2) were amplified by PCR from E14.5 cDNA, and then cloned into pHIV-EGFP to overexpress sNrp1 proteins. Recombinant lentiviral particles were produced in HEK293 cells by a polyethylenimine, linear (MW 25,000, Polysciences) transfection system. Lentiviral particles were concentrated 38-fold by ultracentrifugation (Beckman Coulter) for 2 hours at 27,000 rpm, 4 °C.
Colony formation assay
FACS sorted cells were infected with lentivirus in a ultralow attachment cluster at a density of 3 × 105 cells/ml overnight, and resuspended at a density of 4 × 105 cells/ml in chilled 100% growth-factor-reduced Matrigel (BD Bioscience), 50 μl/well of 24 wells cluster. The mixture was plated in 37 °C incubator for polymerization before being cultured with growth medium (DMEM/F12 1:1, 50 ng/ml EGF (Sigma) and ITS (1:100; Sigma)) which was changed the other day. Primary colony numbers were counted and the colony sizes were measured after 6 days culture. For passaging colonies, the medium was aspirated, and Matrigel was incubated in 500 μl of dispase (BD Bioscience) for 1 h at 37 °C. Colonies were released from Matrigel and single cells were obtained by incubation in 0.05% Trypsin-EDTA for 5–10 min at 37 °C. Single cells were then resuspended in Matrigel as described above at the same density.
EdU labelling
Half of in vitro culture colony growth medium was aspirated, replaced with growth medium containng 0.2 mg/ml EdU (Life Technologies), followed experiment steps were performed according to the manufacturer’s protocol of Click-it EdU imaging kit with Alexa Fluo 594 (Life Technologies) prior to immunohistochemistry detection of EGFP.
Quantitative RT-PCR
Total RNA was extracted with RNAiso plus (Takara) or mini RNA preparation kit (Qiagen), and the reverse transcription reaction was performed by using PrimeScript RT Master mix kit (Takara). Quantitative RT-PCR was performed on StepOne Plus (Applied Biosystems) with FastStart Universal SYBR Green Master Mix kit (Roche). The RNA levels were analyzed by ΔΔCt method and normalized to Gapdh. qPCR product specificity was determined by melt curve analysis. The primers using in this study were listed:
Gapdh-F: TGTGATGGGTGTGAACCACGAGAA
Gapdh-R: CTGTGGTCATGAGCCCTTCCACAA
Axin2-F: AGCCTAAAGGTCTTATGTGGCTA
Axin2-R: ACCTACGTGATAAGGATTGACT
Nrp1-F: ACAAATGTGGCGGGACCATA
Nrp1-R: CCGGAGCTTGGATTAGCCAT
Procr-F: CTCTCTGGGAAAACTCCTGACA
Procr-R: CAGGGAGCAGCTAACAGTGA
ChIP-qPCR
Comma D β cells (2 × 106) were plated for 48 h and treated with vehicle or Wnt3A protein for 24 h before PFA crosslinking. Briefly, cells were cross-linked for 10 min with final concentration 1% PFA at room temperature. 125 mM final glycin was added to inactivate PFA at 37 °C for 5 min. Cells were scrapped and pelleted, cell pellet was incubated in chilled Mg-NI NP40 buffer for 10 min, then resuspended in lysis buffer with proteinase inhibitors. Genomic DNA was sonicated. Precleared sample (0.1 ml) was saved to assess input DNA. Anti-β-catenin antibody (4 μg, BD Pharmingen) was added to the precleared sample and gently mixed for O/N at 4 °C, followed by rotation with 50 μl protein G-agarose/Salmon sperm DNA (Millipore) for 2 h at 4 °C. Subsequent wash, elution, proteinase K treatment and de-cross-linking procedures were performed. TBEs enrichment was determined by qPCR analysis.
Immunohistochemistry
Frozen sections were prepared by air-drying and fixation for 1 h in pre-chilled 4% PFA. Tissue sections were blocked with PBT contained 10% normal goat or cow serum for 1 h, then incubated with primary antibodies at 4 °C overnight, followed by PBT wash 3 times, incubated with secondary antibodies for 2 h at room temperature, and counterstained with DAPI (Life Technologies) for 2 min, mounted with prolong gold mounting medium. The primary antibodies used in this work were goat anti-mouse Nrp1 (R&D systems, AF566, 1:100), Rat anti-K8 (Developmental Hybridoma Bank, TROMA-I, 1:200) and rabbit anti-mouse Keratin 14 (Covance, PRB-155P, 1:1000). IHC detection was performed with appropriate secondary conjugates (Life Technologies). Confocal images were captured by using Leica DM6000 TCS/ SP8 laser confocal scanning microscope.
Mammary gland whole mount carmine staining
The 4th pair of mammary glands were fixed in pre-chilled 4% PFA and washed with PBS twice. Tissues were incubated with carmine alum overnight, followed by destaining with solutions (50% ethanol, 2% HCl) for 2 h, dehydrated in 70%, 85%, 95%, 100% ethanol for 1 h, and treated with histoclear for 1 h. Images were captured by a dissection microscope (Leica).
Transplantation into mammary fat pad
FACS Sorted cells were resuspended in 50% matrigel, PBS with 20% FBS, and 0.04% Trypan Blue (Sigma) and injected 10 μl volumes into the cleared fat pads of 3-week-old female nude mice. Reconstituted mammary glands were harvested 8 weeks after transplantation. Outgrowths were detected under a dissection microscope (Leica) based on fluorescence or carmine staining. Outgrowths with >10% of the host fat pad filled were scored as positive, and fat pad filled was marked as 4/4 (all) filled, 3/4, 1/2, 1/4 and no growth. The repopulating frequencies were calculated using the “statmod” software package for the R computing environment (http://www.R-project.org). Repopulating frequencies were estimated with a complementary log-log generalized linear model. Two-sided 95% Wald confidence intervals were computed, except in the case of zero outgrowths, when one-sided 95% Clopper-Pearson intervals were used instead.
Statistical analysis
Student’s t-test was performed and the P value was calculated in Graphpad Prism 5 on data represented by histograms, which consisted of results from at least three independent experiments unless specified otherwise. For all experiments with error bars, the standard deviation (SD) was calculated to indicate the variation within each experiment. P < 0.05 (*), P < 0.01 (**) and P < 0.001 (***) were considered as significance.
Acknowledgements
We thank Weimin Jiang for technical assistance. We are grateful to Dr. Qing Cissy Yu for critical reading of the manuscript and all lab members for helpful discussion. This work was supported by grants from National Natural Science Foundation of China (31530045, 31371500 to Y.A.Z.), Ministry of Science and Technology of China (2014CB964800 to Y.A.Z.), and Chinese Academy of Sciences (XDB19000000, XDA12020349 to Y.A.Z.).
Author Contributions
Y.A.Z. conceived the study. L.W., W.T. and X.D. performed the experiments. L.W., W.T. and Y.A.Z. analyzed the data and wrote the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Yu, Q. C., Verheyen, E. M. & Zeng, Y. A. Mammary Development and Breast Cancer: A Wnt Perspective. Cancers (Basel)8, doi:10.3390/cancers8070065 (2016). [DOI] [PMC free article] [PubMed]
- 2.Shackleton M, et al. Generation of a functional mammary gland from a single stem cell. Nature. 2006;439:84–88. doi: 10.1038/nature04372. [DOI] [PubMed] [Google Scholar]
- 3.Stingl J, et al. Purification and unique properties of mammary epithelial stem cells. Nature. 2006;439:993–997. doi: 10.1038/nature04496. [DOI] [PubMed] [Google Scholar]
- 4.Wang D, et al. Identification of multipotent mammary stem cells by protein C receptor expression. Nature. 2015;517:81–84. doi: 10.1038/nature13851. [DOI] [PubMed] [Google Scholar]
- 5.Wend P, Holland JD, Ziebold U, Birchmeier W. Wnt signaling in stem and cancer stem cells. Semin Cell Dev Biol. 2010;21:855–863. doi: 10.1016/j.semcdb.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 6.Incassati A, Chandramouli A, Eelkema R, Cowin P. Key signaling nodes in mammary gland development and cancer: beta-catenin. Breast cancer research: BCR. 2010;12:213. doi: 10.1186/bcr2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Roarty K, Rosen JM. Wnt and mammary stem cells: hormones cannot fly wingless. Current opinion in pharmacology. 2010;10:643–649. doi: 10.1016/j.coph.2010.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zeng YA, Nusse R. Wnt proteins are self-renewal factors for mammary stem cells and promote their long-term expansion in culture. Cell Stem Cell. 2010;6:568–577. doi: 10.1016/j.stem.2010.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cai C, et al. R-spondin1 is a novel hormone mediator for mammary stem cell self-renewal. Genes Dev. 2014;28:2205–2218. doi: 10.1101/gad.245142.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fujisawa H, et al. Roles of a neuronal cell-surface molecule, neuropilin, in nerve fiber fasciculation and guidance. Cell and tissue research. 1997;290:465–470. doi: 10.1007/s004410050954. [DOI] [PubMed] [Google Scholar]
- 11.Wild JR, Staton CA, Chapple K, Corfe BM. Neuropilins: expression and roles in the epithelium. Int J Exp Pathol. 2012;93:81–103. doi: 10.1111/j.1365-2613.2012.00810.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gu C, et al. Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development. Dev Cell. 2003;5:45–57. doi: 10.1016/S1534-5807(03)00169-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Geretti E, Shimizu A, Klagsbrun M. Neuropilin structure governs VEGF and semaphorin binding and regulates angiogenesis. Angiogenesis. 2008;11:31–39. doi: 10.1007/s10456-008-9097-1. [DOI] [PubMed] [Google Scholar]
- 14.Kolodkin AL, et al. Neuropilin is a semaphorin III receptor. Cell. 1997;90:753–762. doi: 10.1016/S0092-8674(00)80535-8. [DOI] [PubMed] [Google Scholar]
- 15.Kitsukawa T, Shimono A, Kawakami A, Kondoh H, Fujisawa H. Overexpression of a membrane protein, neuropilin, in chimeric mice causes anomalies in the cardiovascular system, nervous system and limbs. Development. 1995;121:4309–4318. doi: 10.1242/dev.121.12.4309. [DOI] [PubMed] [Google Scholar]
- 16.Kawasaki T, et al. A requirement for neuropilin-1 in embryonic vessel formation. Development. 1999;126:4895–4902. doi: 10.1242/dev.126.21.4895. [DOI] [PubMed] [Google Scholar]
- 17.Giger RJ, et al. Neuropilin-2 is required in vivo for selective axon guidance responses to secreted semaphorins. Neuron. 2000;25:29–41. doi: 10.1016/S0896-6273(00)80869-7. [DOI] [PubMed] [Google Scholar]
- 18.Bachelder RE, et al. Vascular Endothelial Growth Factor Is an Autocrine Survival Factor for Neuropilin-expressing Breast Carcinoma Cells. Cancer Res. 2001;61:5736–5740. [PubMed] [Google Scholar]
- 19.Bachelder RE, et al. Competing autocrine pathways involving alternative neuropilin-1 ligands regulate chemotaxis of carcinoma cells. Cancer Res. 2003;63:5230–5233. [PubMed] [Google Scholar]
- 20.Barr MP, et al. A peptide corresponding to the neuropilin-1-binding site on VEGF(165) induces apoptosis of neuropilin-1-expressing breast tumour cells. Br J Cancer. 2005;92:328–333. doi: 10.1038/sj.bjc.6602308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gagnon ML, et al. Identification of a natural soluble neuropilin-1 that binds vascular endothelial growth factor: In vivo expression and antitumor activity. Proc Natl Acad Sci USA. 2000;97:2573–2578. doi: 10.1073/pnas.040337597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li Y, et al. Evidence that transgenes encoding components of the Wnt signaling pathway preferentially induce mammary cancers from progenitor cells. Proc Natl Acad Sci USA. 2003;100:15853–15858. doi: 10.1073/pnas.2136825100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morris JS, et al. Involvement of axonal guidance proteins and their signaling partners in the developing mouse mammary gland. J Cell Physiol. 2006;206:16–24. doi: 10.1002/jcp.20427. [DOI] [PubMed] [Google Scholar]
- 24.Goel HL, et al. Neuropilin-2 promotes branching morphogenesis in the mouse mammary gland. Development. 2011;138:2969–2976. doi: 10.1242/dev.051318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.West DC, et al. Interactions of multiple heparin binding growth factors with neuropilin-1 and potentiation of the activity of fibroblast growth factor-2. J Biol Chem. 2005;280:13457–13464. doi: 10.1074/jbc.M410924200. [DOI] [PubMed] [Google Scholar]
- 26.Hsieh SH, et al. Galectin-1, a novel ligand of neuropilin-1, activates VEGFR-2 signaling and modulates the migration of vascular endothelial cells. Oncogene. 2008;27:3746–3753. doi: 10.1038/sj.onc.1211029. [DOI] [PubMed] [Google Scholar]
- 27.Matsushita A, Gotze T, Korc M. Hepatocyte growth factor-mediated cell invasion in pancreatic cancer cells is dependent on neuropilin-1. Cancer Res. 2007;67:10309–10316. doi: 10.1158/0008-5472.CAN-07-3256. [DOI] [PubMed] [Google Scholar]
- 28.Hu B, et al. Neuropilin-1 promotes human glioma progression through potentiating the activity of the HGF/SF autocrine pathway. Oncogene. 2007;26:5577–5586. doi: 10.1038/sj.onc.1210348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sulpice E, et al. Neuropilin-1 and neuropilin-2 act as coreceptors, potentiating proangiogenic activity. Blood. 2008;111:2036–2045. doi: 10.1182/blood-2007-04-084269. [DOI] [PubMed] [Google Scholar]
- 30.Cao S, et al. Neuropilin-1 promotes cirrhosis of the rodent and human liver by enhancing PDGF/TGF-beta signaling in hepatic stellate cells. The Journal of clinical investigation. 2010;120:2379–2394. doi: 10.1172/JCI41203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Glinka Y, Prud’homme GJ. Neuropilin-1 is a receptor for transforming growth factor beta-1, activates its latent form, and promotes regulatory T cell activity. Journal of leukocyte biology. 2008;84:302–310. doi: 10.1189/jlb.0208090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ball SG, Bayley C, Shuttleworth CA, Kielty CM. Neuropilin-1 regulates platelet-derived growth factor receptor signalling in mesenchymal stem cells. The Biochemical journal. 2010;427:29–40. doi: 10.1042/BJ20091512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shintani Y, Takashima S, Kato H, Komamura K, Kitakaze M. Extracellular protein kinase CK2 is a novel associating protein of neuropilin-1. Biochemical and biophysical research communications. 2009;385:618–623. doi: 10.1016/j.bbrc.2009.05.108. [DOI] [PubMed] [Google Scholar]
- 34.Stephenson JM, Banerjee S, Saxena NK, Cherian R, Banerjee SK. Neuropilin-1 is differentially expressed in myoepithelial cells and vascular smooth muscle cells in preneoplastic and neoplastic human breast: A possible marker for the progression of breast cancer. Int J Cancer. 2002;101:409–414. doi: 10.1002/ijc.10611. [DOI] [PubMed] [Google Scholar]
- 35.Yasuoka H, et al. Neuropilin-2 expression in breast cancer: correlation with lymph node metastasis, poor prognosis, and regulation of CXCR4 expression. BMC cancer. 2009;9:220. doi: 10.1186/1471-2407-9-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ghosh S, et al. High levels of vascular endothelial growth factor and its receptors (VEGFR-1, VEGFR-2, neuropilin-1) are associated with worse outcome in breast cancer. Human pathology. 2008;39:1835–1843. doi: 10.1016/j.humpath.2008.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Timoshenko AV, Rastogi S, Lala PK. Migration-promoting role of VEGF-C and VEGF-C binding receptors in human breast cancer cells. Br J Cancer. 2007;97:1090–1098. doi: 10.1038/sj.bjc.6603993. [DOI] [PMC free article] [PubMed] [Google Scholar]