Abstract
Sperm cryopreservation of live-bearing fishes, such as those of the genus Xiphophorus is only beginning to be studied, although these fishes are valuable models for biomedical research and are commercially raised as ornamental fish for use in aquariums. To explore optimization of techniques for sperm cryopreservation of these fishes, this study measured the volumetric shrinkage response during freezing of sperm cells of Xiphophorus helleri by use of a shape-independent differential scanning calorimeter (DSC) technique. Volumetric shrinkage during freezing of X. helleri sperm cell suspensions was obtained in the presence of extracellular ice at a cooling rate of 20 °C/min in three different media: (1) Hanks’ balanced salt solution (HBSS) without cryoprotective agents (CPAs); (2) HBSS with 14% (v/v) glycerol; and (3) HBSS with 10% (v/v) dimethyl sulfoxide (DMSO). The sperm cell was modeled as a cylinder of 33.3 μm in length and 0.59 μm in diameter with an osmotically inactive cell volume (Vb) of 0.6Vo, where Vo is the isotonic or initial cell volume. By fitting a model of water transport to the experimentally determined volumetric shrinkage data, the best-fit membrane permeability parameters (reference membrane permeability to water, Lpg or Lpg[cpa] and the activation energy, ELp or ELp[cpa]) of the Xiphophorus helleri sperm cell membrane were determined. The best-fit membrane permeability parameters at 20 °C/min in the absence of CPAs were: Lpg = 0.776 × 10−15 m3/Ns (0.0046 μm/min atm), and ELp = 50.1 kJ/mol (11.97 kcal/mol) (R2 = 0.997). The corresponding parameters in the presence of 14% glycerol were Lpg[cpa] = 1.063×10−15 m3/Ns (0.0063 μm/min atm), and ELp[cpa] = 83.81 kJ/mol (20.04 kcal/mol) (R2 = 0.997). The parameters in the presence of 10% DMSO were Lpg[cpa] = 1.4 × 10−15 m3/Ns (0.0083 μm/min atm), and ELp[cpa] = 90.96 kJ/mol (21.75 kcal/mol) (R2 = 0.996). Parameters obtained in this study suggested that the optimal rate of cooling for X. helleri sperm cells in the presence of CPAs ranged from 20 to 35 °C/min and were in close agreement with recently published, empirically determined optimal cooling rates.
Keywords: Differential scanning calorimetry, Water transport, Reference membrane permeability and activation energy
1. Introduction
Live-bearing fishes of the genus Xiphophorus are widely used in diverse areas of contemporary scientific research, including evolution [1,2], sex determination [3–5], endocrinology [6,7], ethology and behavioral ecology [8–11], toxicology [12,13], parasitology [14,15], immunology [16,17], and cancer genetics [18,19]. In addition to their value as experimental models for biomedical research, swordtails and platyfish are also valued as ornamental fish because of vibrant body coloration and a long sword-like tail. At present, more than 22 species and more than 67 pedigreed lines of Xiphophorus are currently maintained only as live animal stocks, perpetuated by a labor-intensive breeding regime (Xiphophorus Genetic Stock Center of Texas State University, San Marcos, TX, USA). As new species are identified, new lines developed, and rare chromosomal conditions delineated, it becomes increasingly important to cryogenically preserve Xiphophorus sperm samples for the long-term.
Despite the study of sperm cryopreservation in some 200 species of freshwater and marine fishes [20], sperm cryopreservation has only recently been explored in live-bearing fishes as a group [21–23]. The focus of the present study was to measure a biophysical response (water transport) in X. helleri sperm cells during freezing using a recent advance in measurement methodology, pioneered by Devireddy and colleagues, using a differential scanning calorimeter (DSC) [24–29]. In the DSC technique, two heat releases from the same cell suspension are measured: (i) during freezing of osmotically active (live) cells in suspension; and (ii) during freezing of osmotically inactive (dead) cells in the same suspension. The difference in heat release measured between the two cooling runs from the same experiment is correlated to water transport. This has been demonstrated by Devireddy and colleagues in a variety of sperm cell suspensions, including murine [24], human [25], equine [26,27], canine [28], boar [29], and recently in an aquatic species, the Pacific oyster Crassostrea gigas [30], and independently verified by Yuan and Diller [31] and Diller [32]. As described elsewhere, knowledge of the water transport (cellular dehydration) during freezing of a cell suspension can be used to predict a priori the optimal rates of freezing [24–29,33,34]. Briefly, the loss of post-thaw viability is either due to intracellular ice formation [35,36] or prolonged exposure to freeze-concentrated extracellular solution [37]. Differences in membrane permeability to water and the probability of IIF result in different optimal cooling rates for different cells [34,38]. Therefore, to optimize a cryopreservation protocol, it is important to measure cell membrane permeability to water.
The aim of this project was to establish the water transport (and consequently the membrane permeability parameters) of X. helleri sperm during freezing using a DSC technique at a cooling rate of 20 °C/min in three different media: Hanks’ balanced salt solution (HBSS) without cryoprotectants; HBSS with 14% glycerol; and HBSS with 10% dimethyl sulfoxide (DMSO). The experimentally determined membrane permeability parameters were used to numerically predict the loss of intracellular water under a variety of cooling rates (5–100 °C/min). The numerical predictions were analyzed to predict the amount of water left in the sperm cell after dehydration in the absence of IIF, and consequently the optimal cooling rates for X. helleri sperm cryopreservation.
2. Materials and methods
2.1. Collection and isolation of sperm cells
A total of 26 male X. helleri were shipped weekly by overnight delivery from the Xiphophorus Genetic Stock Center (XGSC) of Texas State University (San Marcos, TX, USA) to the Aquaculture Research Station of the Louisiana State University Agricultural Center in January through March of 2004. The fish were anesthetized in 0.01% MS-222 (Western Chemical Inc., Ferndale, WA, USA) for 2 min, and the standard lengths and wet weights were measured. Sperm were collected by surgical removal of the testis, adherent tissue was dissected away and testes were placed in tared resealable plastic bags (NASCO whirl-pak, MBCOCT, New Haven, CT, USA) and weighed [22]. Based on previously published results that report the osmolality of blood plasma of X. helleri as 320 mOsm/kg [21–23], HBSS at 300 mOsm/kg was added to the testis before crushing of the testis to release sperm. Sperm concentrations were adjusted to 0.9−1.6×109 cells/mL, while the motility of fresh sperm always ranged from 80 to 90% motile. The concentration of the cells was adjusted such that the product of initial intracellular cell water (~5 μm3), the latent heat of fusion of water (335 mJ/mg), the density of intracellular water (1000 kg/m3) and the number of cells in a DSC sample pan (~ 109 cells/mL) translated to a value that could be measured by a DSC-7 machine (Perkin Elmer Corporation, Norwalk, CT, USA) which was 1–3 mJ/mg of sample (see [39] by Devireddy et al. for a more detailed description for the expected value of the DSC heat release). The samples were transported to the LSU bioengineering laboratory for DSC experiments.
2.2. DSC experiments
The DSC experiments were carried out in the absence and presence of CPAs. Nine separate DSC experiments were conducted in the absence of any CPA, in the presence of two CPAs glycerol (14%, v/v) and dimethyl sulfoxide (10%, v/v). Approximately 10 μL of the sperm suspension was loaded in a DSC standard aluminum sample pan (Perkin Elmer Corporation, Norwalk, CT, USA) with ~ 0.5 mg of Pseudomonas syringae (ATCC, Rockville, MD, USA). The DSC dynamic cooling protocol used to measure the water transport out of the sperm cells was the same as reported in earlier studies on mammalian [24–29] and aquatic [30] sperm cells and will be briefly stated here. It is important to note that previously published data on X. helleri sperm suggested that these cells were “viable” for as long as 2 weeks after collection [22,23]. However, in the interest of consistency all water transport (DSC) experiments were completed within 3–6 h of sperm collection.
2.3. DSC dynamic cooling protocol
Step 1, sperm cell suspensions with or without CPAs initially at room temperature were cooled at 5 °C/min until the extracellular ice nucleated. Step 2, after nucleation, the sample was thawed at a warming rate (5 °C/min) such that phase-change temperature Tph was reached (but not overshot) and ice remained in the extracellular solution. The phase change temperatures were determined to be − 0.6 °C for the HBSS, − 4.1 °C for HBSS with 14% glycerol, and − 3.2 °C for HBSS with 10% DMSO. Step 3, the sample was cooled to − 50 °C at 20 °C/min causing the sperm cells to undergo dehydration. The lower curve in Fig. 1 corresponds to the heat release associated with dehydration and the total area is represented by qinitial. Step 4, the sample was re-equilibrated at Tph by thawing at 20 °C/min. Step 5, to differentiate between the heat released by the media and the intracellular fluid in Step 3, the sample was cooled at 200 °C/min to − 150 °C. This caused all the sperm cells to lyse and become osmotically inactive. Step 6, Step 4 was repeated. Step 7, the sample was cooled to − 50 °C at 20 °C/min to measure the final heat release due to lysed (osmotically inactive) sperm cells mixed with media. The upper curve in Fig. 1 corresponds to this heat release and the total area is represented by qfinal.
Fig. 1.
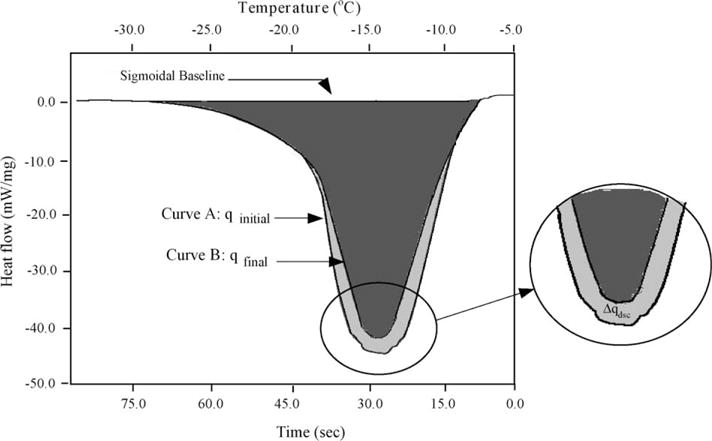
Superimposed heat flow thermograms obtained during the initial (osmotically active cells; Curve A) and final (osmotically inactive cells; Curve B) cooling trials of X. helleri sperm cells at 20 °C/min obtained in the presence of DMSO. The negative values on the y-axis for the heat flow implies an exothermic heat release in the DSC sample. The heat flow (mW/mg) is plotted along the y-axis and the sub-zero temperatures (°C) are plotted along the top x-axis and time (s) is plotted on the bottom x-axis.
Although no independent experiments were conducted as part of this study to assess the viability of the cells prior to the start or during the course of the experiments, that we measured a positive difference in heat release between Step 3 and Step 7 of the DSC cooling protocol described above, confirmed that the cells were osmotically active or intact prior to the start of the DSC experiment. Should the cells be osmotically inactive or lysed prior to the start of the experiment, the DSC cooling protocol will measure no difference in heat release, as described in earlier studies [24–30,39,40]. This assumption is further supported by previously published data [21–23] that suggested that the sperm cells of X. helleri remain viable even 2 weeks after collection.
2.4. Translation of heat release to cell volume data for dynamic cooling
The heat release measurements of interest are Δqdsc and Δq(T)dsc which are the total and fractional difference between the heat releases measured by integration of the heat flows during freezing of osmotically active (live) cells in media qinitial, and during freezing of osmotically inactive (dead) cells in media, qfinal. As defined in a series of earlier studies by Devireddy and colleagues, this difference in heat release has been shown to be related to cell volume changes in several biological systems [24–30,39,40] as:
| (1) |
This equation can be rearranged to measure water transport data from the DSC measured heat releases Δ(T)dsc and Δqdsc as:
| (2) |
The unknowns required in Eq. (2) apart from the DSC heat release readings are Vi (the initial cell volume) and Vf (the final cell volume). The initial volume of the sperm cells in the HBSS solution was assumed to be the isotonic cell volume, Vo (~ 9 μm3) (see Fig. 2 and Table 1) and from this the initial cell volume, Vi, was calculated to be 1.5Vo in HBSS with 14% glycerol and 1.35Vo in HBSS with 10% DMSO. Similarly, the final volume in HBSS was assumed to be the osmotically inactive cell volume, Vb (=0.6Vo or ~ 5.4 μm3) and from this the final volume Vf, was calculated to be 0.69Vi in HBSS with 14% glycerol and 0.67Vi in HBSS with 10% DMSO.
Fig. 2.
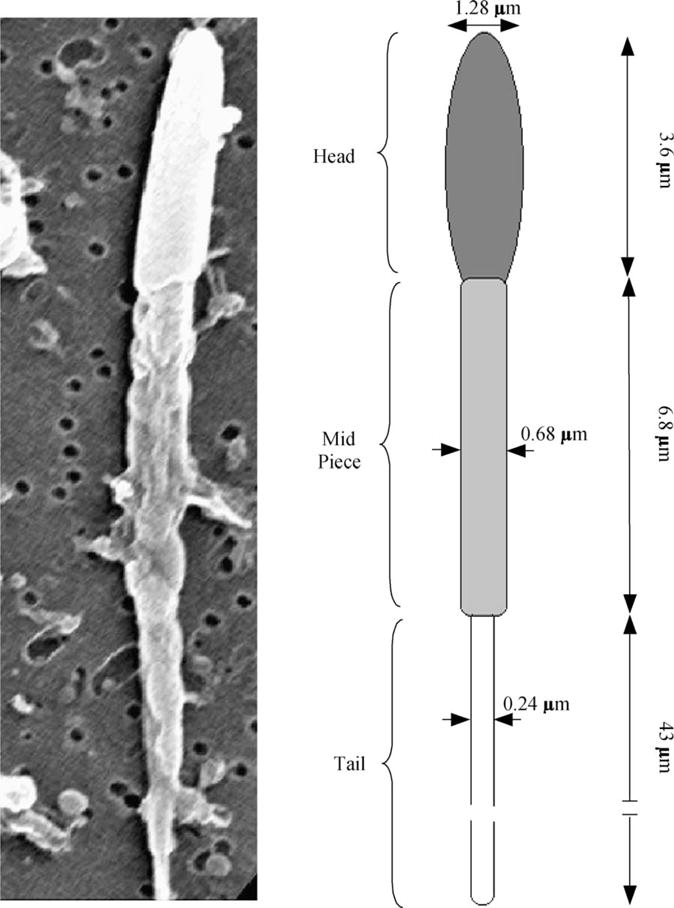
Left, representative scanning electron micrographs (SEM) of sperm from X. helleri showing the relationship of sperm head and tail in alignment with model used to calculate the cell volumes and surface areas shown on the right. Right, the “geometric” model used to calculate the X. helleri sperm cell volumes and surface areas from the SEM images shown on the left.
Table 1.
Measurements, and calculated volumes, surface areas, and ratios of surface area to volume (S:V) for head, midpiece, tail, and combined total of sperm cells from X. helleri (n = 6)
| Sperm cell geometry | Diameter (μm) | Length (μm) | Mean volume (V) (μm3) | Mean surface area (S) (μm2) | S:V (μm−1) |
|---|---|---|---|---|---|
| Heada | 1.28 ± 0.2 | 3.6 ± 0.4 | 4.6 | 14.70 | 3.07 |
| Mid-piecea | 0.68 ± 0.1 | 6.8 ± 0.5 | 2.5 | 14.53 | 5.88 |
| Taila | 0.24 ± 0.05 | 43.0 ± 2.2 | 1.95 | 32.42 | 16.62 |
| Totalb | 9.05 | 61.65 | 6.81 |
The volumes of the head, mid-piece and tail are all assumed to be cylinders.
Sum of the volumes and surface areas of the head, the mid-piece and the tail. Note that as long as the S:V is maintained as close to the original values as possible, any geometry can be assumed for the sperm cell—for simplicity we assumed the X. helleri can be modeled as a thin, long cylinder of length 33.3 μm and a diameter of 0.59 μm (which results in a surface area to volume ratio of 6.78 or within 0.4% of the calculated value). Also see Section 3.5.
To ensure the accuracy and repeatability of the experimental data, a set of calibration and control experiments were performed as detailed previously for this instrument (DSC-7, Perkin Elmer Corporation, Norwalk, CT, USA) [39]. One of these tests was to compare the heat release measured during the final cooling run, qfinal, with the DSC measured heat release from a separate control experiment composed of osmotically inactive (lysed) X. helleri spermatozoa; the magnitude (~ 220 mJ/mg) of the heat releases were found to be within ± 1%. This indicated that, after the fast cooling in the DSC protocol (200 °C/min to − 150 °C), the X. helleri spermatozoa were osmotically inactive.
2.5. Water transport model
A model for water and solute transport in response to chemical potential gradients based on irreversible thermodynamics has been proposed by Kedem and Katchalsky [41]. The K–K model consisted of two differential equations which describe the water and CPA flux across the membrane [41]. If the flux of CPA is negligible in comparison to the flux of water [42,43], the K–K model reduces to a water transport model, as proposed by Mazur [33] and later modified by Levin et al. [44]. The water transport model of Mazur was further modified [45] to incorporate the presence of CPAs on the volumetric shrinkage response of cells during freezing as:
| (3) |
with Lp, the sperm cell membrane permeability to water defined by Levin et al. [44] as:
| (4) |
where, Lpg[cpa] is the reference membrane permeability at a reference temperature, TR (=273.15 K); ELp[cpa] is the apparent activation energy (kJ/mol) or the temperature dependence of the cell membrane permeability; V is the sperm cell volume at temperature T (K); Ac is the effective membrane surface area for water transport, assumed to be constant during the freezing process; Vo the isotonic (initial) sperm cell volume and Vb is the osmotically inactive cell volume. In this study, the X. helleri sperm cell was modeled as a cylinder with length (L) of 33.3 μm and a radius (ro) of 0.295 μm, which translated to Vo of ~ 9 μm3 and Ac of ~ 61.7 μm2 (Table 1 and Fig. 2); R is the universal gas constant; B is the constant cooling rate (K/min); ncpa is the number of moles of salt; vcpa is the molar volume of CPA; vw is the molar volume of water; φs is the disassociation constant for salt; ns is the number of osmoles of salt (=Ci(Vo – Vb), where Ci is the initial cell osmolality); ΔHf is the latent heat of fusion of water, and ρ is the density of water. Note that when ncpa is zero (i.e. no CPA is present), Eqs. (3) and (4) reduce to the water transport model as described by Mazur [33] and Levin et al. [44] and Lp is an Arrhenius function of Lpg and ELp. The two unknown membrane permeability parameters of the model either Lpg[cpa] and ELp[cpa] in the presence of CPA or Lpg and ELp in the absence of CPA, are determined by curve-fitting the water transport model to experimentally obtained volumetric shrinkage data during freezing. The various assumptions made in the development of Mazur’s model of water transport are discussed in detail elsewhere [36,40,43,45] and are beyond the scope of this report.
2.6. Numerical methods
A non-linear least squares curve-fitting technique was implemented using a computer program to calculate the membrane permeability parameters that best fit the volumetric shrinkage data as previously described [46]. The optimal fit of Eq. (3) to the experimental data was obtained by selecting a set of parameters that minimized the residual variance, χ2, and maximized a goodness of fit parameter, R2 [47]. All of the curve-fitting results presented have an R2 value ≥ 0.96, indicating that there was good agreement between the experimental data and the fit calculated using the estimated membrane permeability parameters. To simulate the biophysical response of a sperm cell under a variety of cooling rates, the best-fit parameters were substituted in the water transport equation. The water transport equation was then solved numerically using a custom written FORTRAN code (the code utilizes a 4th order Runge–Kutta method) on a Mac Powerbook G4 (Apple Computer Inc., Cupertino, CA, USA) workstation [30,39].
3. Results
3.1. Dynamic cooling response and membrane permeability parameters
The water transport data and simulation using the best-fit parameters in Eq. (3) were measured at a cooling rate of 20 °C/min in HBSS (Fig. 3A), in HBSS with 14% glycerol (Fig. 3B) and in HBSS with 10% DMSO (Fig. 3C). Water transport cessation was observed in the DSC heat release data as an overlap of the thermograms from the heat release signature obtained using osmotically active (initial) and inactive (final) cells (Fig. 1). The dynamic portion of the cooling curve (where the thermograms were distinct) was found to be between − 0.6 and − 10 °C with HBSS, between − 4.1 and − 20 °C with HBSS and 14% glycerol, and between − 3.2 and − 25 °C with HBSS and 10% DMSO. The membrane permeability parameter values that best fit the 20 °C/min water transport data in the absence and presence of CPAs were calculated (Table 2). The model-simulated equilibrium cooling response was generated by setting the left hand side (LHS) of Eq. (3) is equal to 0 and balancing the intracellular and extracellular unfrozen chemical activity of water on the right hand side (RHS) at a particular subzero temperature (shown as a dashed line in Fig. 3).
Fig. 3.
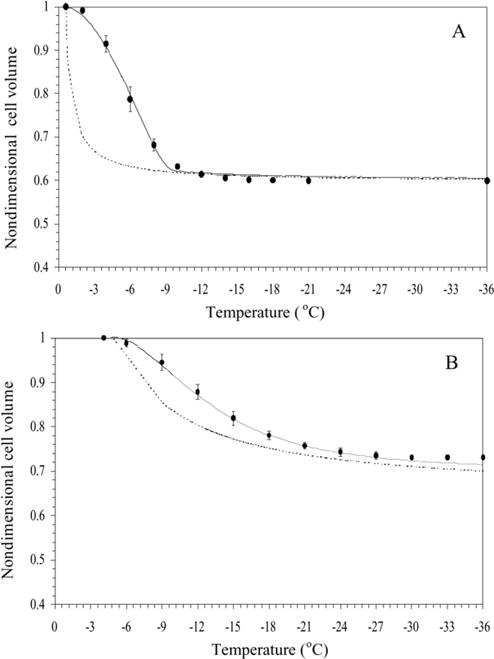
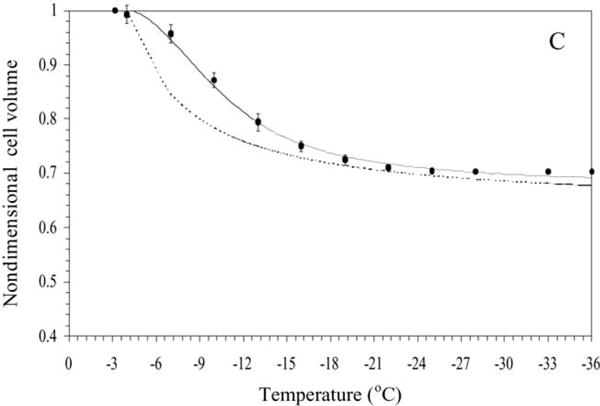
Volumetric response of X. helleri sperm cells as a function of sub-zero temperatures obtained using the DSC technique in the presence of extracellular ice (A), in the presence of extracellular ice and glycerol (B), and in the presence of extracellular ice and DMSO (C). The filled circles represent the experimentally obtained water transport (volumetric shrinkage) at a cooling rate of 20 °C/min. The dynamic cooling response at 20 °C/min is shown as a solid line and was obtained by using the “best fit” membrane permeability parameters (Lpg and ELp or Lpg[cpa] and ELp[cpa]) (Table 1) in the water transport equation (Eqs. (3) and (4)). The model-simulated equilibrium cooling response obtained is shown as a dotted line in all the figures. The non-dimensional cell volume is plotted along the y-axis and the sub-zero temperatures are shown along the x-axis. The error bars represent the standard deviation for the mean values of nine separate DSC experiments (n = 9).
Table 2.
Predicted sub-zero membrane permeability parameters (the reference membrane permeability in the absence and presence of CPAs, Lpg or Lpg[cpa] shown in Column 3 and the activation energy in the absence and presence of CPAs, ELp or ELp[cpa] shown in Column 4; see text for further description) and the optimal rates of freezing for X. helleri sperm cells in the presence of extracellular ice and cryoprotective agents (CPAs)
| Assumed inactive cell volume (Vb) | Freezing media | Lpg or Lpg[cpa] × 1015 m3/Ns (μm/min atm) | ELp or ELp[cpa] kJ/mol (kcal/mol) | Goodness of fit, R2 valueb | Predicted optimal cooling rate °C/min)c |
|---|---|---|---|---|---|
| 0.6Vo | HBSSa | 0.78 (0.0046) | 50.10 (11.97) | 0.997 | 89 |
| HBSS with 14% glycerola | 1.06 (0.0063) | 83.80 (20.04) | 0.997 | 29 | |
| HBSS with 10% DMSOa | 1.40 (0.0083) | 91.00 (21.75) | 0.996 | 32 |
Hanks balanced salt solution (300 mOsm/kg) which is physiologically isotonic for X. helleri sperm.
A value of 1.0 represents a “perfect” fit between the experimentally measured water transport data and the model-simulated water transport response.
As described in the text “the optimal rate of freezing” is defined as the cooling rate which “traps” 5% of the initial water volume inside the cell at −30 °C.
3.2. Membrane permeability parametric space
The contour plots of the goodness of fit parameter, R2 (=0.96) in the Lpg and ELp (or Lpg[cpa] and ELp[cpa]) space that corresponds to the water transport data at 20 °C/min in HBSS (Fig. 4A), in HBSS with 14% glycerol (Fig. 4B), and in HBSS with 10% DMSO (Fig. 4C). Any combination of Lpg and ELp (or Lpg[cpa] and ELp[cpa]) shown to be within the contour was correlated to the water transport data in that media with an R2 value of >0.96. Note that the contour for the media containing 14% glycerol was almost completely within the contour corresponding to 10% DMSO. This suggested that the membrane permeability parameters obtained in the presence of 10% DMSO could predict the volumetric response of the X. helleri sperm cells in the presence of 14% glycerol accurately, while the converse was not true.
Fig. 4.
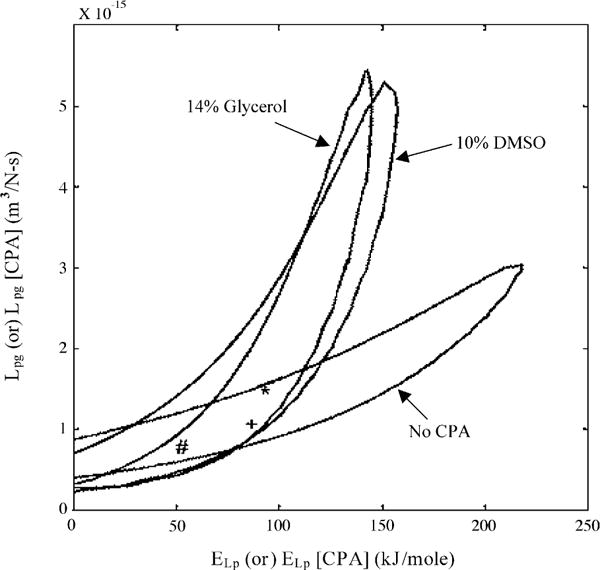
Contour plots of the goodness of fit parameter R2 (= 0.96) for water transport response in X. helleri sperm cells in three different media: HBSS with no CPAs (A), HBSS with 14% glycerol (B), and HBSS with 10% DMSO (C). Note that the best-fit parameters (Table 2) are represented within the contours by a “#” (absence of CPAs), “+” (with 14% glycerol) and “*” (with 10% DMSO). The membrane permeability at 0 °C, Lpg (or Lpg[cpa]) (m3/Ns) is plotted on the y-axis while the apparent activation energy of the membrane, ELp (or ELp[cpa]) (kJ/mol) is plotted on the x-axis.
3.3. Water transport simulations
Water transport simulations obtained using the best-fit parameters (Table 2) in Eq. (3) are shown for a variety of cooling rates (5–100 °C/min) in Fig. 5. From the simulations, the amount of trapped water (a lower bound on the potential intracellular ice) was computed as a ratio of the volume of the water trapped inside the sperm cell at temperature, T (~ − 30 °C) (where intracellular ice formation could occur by homogenous or volume catalysed nucleation) [36] to the initial sperm water volume, [(V − Vf)/(Vi − Vf)] where V is the volume after water transport ceases (at ~ − 30 °C), Vi is the initial (isotonic) sperm cell volume and Vf is the final (osmotically inactive) sperm cell volume. In the absence of CPAs, for cooling rates of ≤ 60, 80, and 100 °C/min, the trapped water volume was ≤ 0.5, 0.53, and 12.4% of the initial osmotically active water volume, and the corresponding end volumes were ≤ 0.6069Vi, 0.6071Vi, and 0.6539Vi, respectively (Fig. 5A). In the presence of 14% glycerol, for cooling rates of ≤ 20, 40, 60, 80, and 100 °C/min, the trapped water volume was ≤ 2.6, 6.2, 48.3, 59.5, and 66.9% of the initial osmotically active water volume, and the corresponding end volumes were ≤ 0.7027Vi, 0.7132Vi, 0.8418Vi, 0.8761Vi, and 0.8986Vi, respectively (Fig. 5B). And finally in the presence of 10% DMSO, for cooling rates of ≤ 20, 40, 60, 80, and 100 °C/min, the trapped water volume was ≤ 1.6, 6.4, 19.6, 51.2, and 59.9% of the initial osmotically active water volume, and the corresponding end volumes were ≤ 0.6824Vi, 0.6981Vi, 0.7406Vi, 0.8425Vi, and0.8705Vi, respectively (Fig. 5C).
Fig. 5.
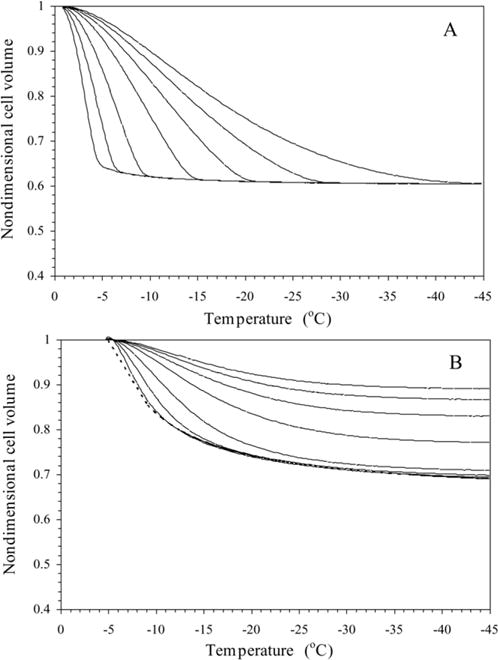
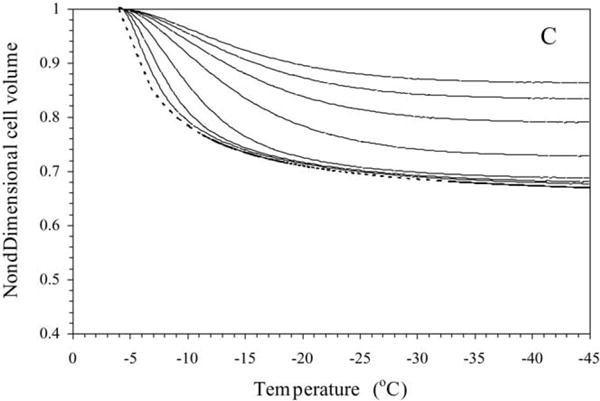
Volumetric response of X. helleri sperm cells at various cooling rates as a function of sub-zero temperature using the best-fit membrane permeability parameters (Table 1). The changes in the normalized cell volume (V/Vo) are shown as a function of temperature for different cooling rates in X. helleri sperm cell suspensions without CPAs (A), with 14% glycerol (B), and with 10% DMSO (C). The water transport curves represent the model-simulated response for different cooling rates (from left to right: 5, 10, 20, 40, 60, 80, and 100 °C/min). The model-simulated equilibrium cooling response obtained is shown as a dotted line. The sub-zero temperatures are shown along the x-axis while the non-dimensional volume is plotted along the y-axis.
As described earlier, the cooling rate which optimizes the response to freezing and thawing of any cellular system can be defined as the fastest cooling rate in a given media that proceeds without forming damaging intracellular ice [38]. Generally, intracellular ice formation is defined as damaging and lethal if more than 10–15% of the initial intracellular water is involved [48]. We defined the optimal cooling rate as the rate at which 5% of the initial osmotically active water volume was trapped within the cells at − 30 °C. The simulations (Fig. 5) obtained using the best-fit parameters show that the optimal cooling rate in HBSS as 89 °C/min, in HBSS with 14% glycerol as 29 °C/min, and in HBSS with 10% DMSO as 32 °C/min (Table 2). It is important to note if intracellular ice formation occurs by a heterogeneous or a surface-catalyzed nucleation mechanism [36] (generally between − 5 and − 20 °C for a variety of single cells), which our model does not predict, then potentially even more water would be trapped in the sperm cells than predicted by water transport alone (i.e. the lower bound of intracellular ice discussed above). Thus, the optimal cooling rates (Tables 2 and 3) based on the lower bound of intracellular ice formation would be over-estimated.
Table 3.
Predicted sub-zero membrane permeability parameters and the optimal rates of freezing for X. helleri sperm cells in the presence of extracellular ice and cryoprotective agents (CPAs)
| Assumed inactive cell volume (Vb) | Freezing media | Lpg or Lpg[cpa] × 1015 m3/Ns (μm/min atm) | ELp or ELp[cpa] kJ/mol (kcal/mol) | Goodness of fit, R2 value | Predicted optimal cooling rate (°C/min) |
|---|---|---|---|---|---|
| 0.4Vo | HBSS | 1.13 (0.0067) | 48.90 (11.68) | 0.997 | 90 |
| HBSS with 14% glycerol | 1.82 (0.0108) | 104.60 (25.01) | 0.997 | 25 | |
| HBSS with 10% DMSO | 2.45 (0.0145) | 119.00 (28.45) | 0.997 | 24 | |
| 0.8Vo | HBSS | 0.42 (0.0025) | 62.70 (15.00) | 0.996 | 71 |
| HBSS with 14% glycerol | 0.41 (0.0024) | 41.70 (9.96) | 0.962 | 20 | |
| HBSS with 10% DMSO | 0.46 (0.0027) | 43.20 (10.32) | 0.966 | 25 |
3.4. Parameter sensitivity analysis—effect of varying Vb
The value of the osmotically inactive cell volume, Vb of mammalian sperm cells has been reported to be 0.6Vo (the value used in this and other studies) and to range from as low as 0.23Vo to as high as 0.75Vo [24–30,39,40]. To estimate the effect of varying the osmotically inactive cell volume on the measured membrane permeability parameters (Lpg and ELp or Lpg[cpa] and ELp[cpa]), the value of Vb was increased to 0.8Vo and decreased to 0.4Vo. The DSC data were correspondingly modified (using Eq. (2)) and the modified DSC water transport data were curve fitted to the water transport model (Eqs. (3) and (4)) using the non-linear least squares curve-fitting technique described previously. The predicted values of the membrane permeability parameters (Lpg and ELp) using a value of 0.4Vo and 0.8Vo as the osmotically inactive cell volume, are shown in Table 3. Although the predicted membrane permeability parameters using Vb of 0.4Vo and 0.8Vo were different from each other and the values obtained using Vb of 0.6Vo (Table 2), the predicted rates of optimal freezing are not that different and are within ± 20% of one another. Thus, the variation in the value of Vb did not alter our model predicted optimal rates of freezing X. helleri sperm cell across a broad range of Vb values. Further studies are clearly needed to understand this lack of sensitivity in the value of the model predicted optimal cooling rates for X. helleri sperm cells to the assumed value of Vb.
To further illustrate the effect of varying the osmotically inactive cell volume on the predicted membrane permeability parameters, contour plots (similar to those shown in Fig. 4) were generated for an assumed Vb values of 0.6Vo and 0.4Vo. A comparison of contour space for the three different values of Vb (0.4Vo, 0.6Vo, and 0.8Vo) are shown in Fig. 6A (for HBSS with no CPAs), Fig. 6B (HBSS with 14% glycerol), and Fig. 6C (HBSS with 10% DMSO). In the presence of glycerol (Fig. 6B), and DMSO (Fig. 6C) the contour plots obtained with an inactive cell volume of 0.8Vo were completely enclosed by the contour obtained with an assumed Vb value of 0.6Vo, and a considerable region of the contours obtained with an assumed Vb value of 0.4Vo were also encompassed by the contours obtained with an assumed Vb value of 0.6Vo. This overlap suggests that the permeability parameters obtained with a Vb value of 0.6Vo can accurately predict the behavior of X. helleri sperm cells with a Vb value of either 0.8Vo or 0.4Vo, while the converse was not true. However, in the absence of CPAs (Fig. 6A), no precise conclusions could be drawn on the effect of assumed Vb on predicted membrane permeability parameters, although there seemed to be a trend towards lowering the range of best-fit activation energy values (ELp) with increasing Vb.
Fig. 6.
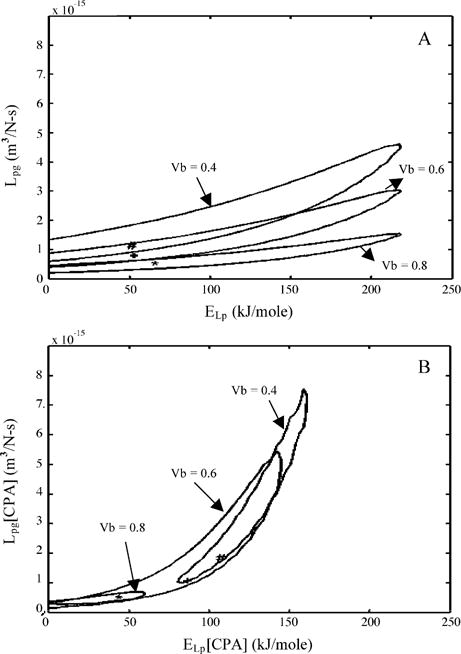
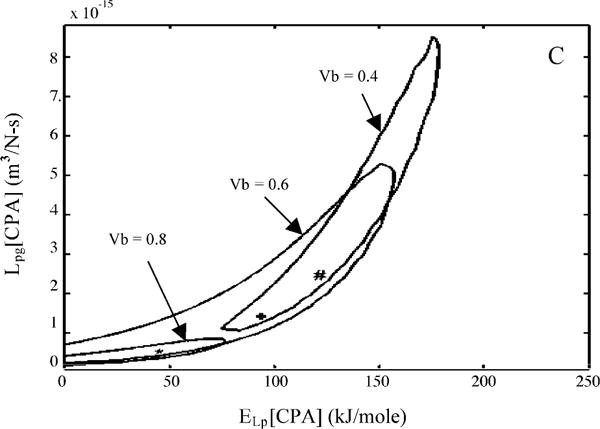
Contour plots of the goodness of fit parameter R2 (= 0.96) for water transport response in X. helleri sperm cells in three different media: HBSS with no CPAs (A), HBSS with 14% glycerol (B), and HBSS with 10% DMSO (C). Within each figure, the three contour correspond to the three assumed values of osmotically inactive cell volume, Vb (0.4Vo, 0.6Vo, and 0.8Vo). Note that the best-fit parameters (Tables 2 and 3) are represented within the contours by a “#” (Vb = 0.4Vo), “+” (Vb = 0.6Vo) and “*” (Vb = 0.8Vo). The membrane permeability at 0 °C, Lpg (or Lpg[cpa]) (m3/Ns) is plotted on the y-axis while the apparent activation energy of the membrane, ELp (or ELp[cpa]) (kJ/mol) is plotted on the x-axis.
3.5. Parameter sensitivity analysis—effect of varying assumed cell dimensions (or surface area to volume ratio)
To account for errors and uncertainities in the measured X. helleri sperm cell dimensions, we also investigated the effect of varying the surface area to volume ratio (S:V) by 50% on the predicted membrane permeability parameters. The geometric model was appropriately modified in the water transport model (Eqs. (3) and (4)) and the corresponding best-fit permeability parameters (Lpgor Lpg[cpa] and ELp or ELp[cpa]) were obtained using the non-linear least squares curve-fitting technique as previously described (data not shown). As expected, the membrane permeability parameters obtained by increasing the S:V ratio were lower than those shown in Tables 2 and 3, and the permeability parameters obtained by decreasing the S:V ratio were higher. This inverse relationship between S:V ratio and the predicted membrane permeability parameters (Lpg or Lpg[cpa] and ELp or ELp[cpa]) was not surprising, because Eq. (3) shows that the change in the cell volume as a function of temperature (dV/dT) is proportional to the product of Lp and Ac. Thus, for a given change in the cell volume as a function of temperature (dV/dT), an increase in Ac (or S:V ratio) will cause a corresponding decrease in the predicted value of Lp, where Lp = f(Lpg, ELp) as shown in Eq. (4) and vice-versa. Thus, any changes to the geometrical model of the X. helleri sperm cell (specifically membrane surface area, Ac) would manifest themselves with corresponding changes to the membrane permeability parameters predicted by the model (Lpg or Lpg[cpa] and ELp or ELp[cpa]).
Additional numerical simulations were also performed assuming the new S:V ratios and the corresponding water transport parameters in the water transport model (Eqs. (3) and (4)). These simulations were analyzed to predict the optimal cooling rate as described earlier (i.e. the rate at which 5% of the initial osmotically active water volume is trapped within the cells at − 30 °C). The simulations (data not shown) showed that the optimal cooling rate in HBSS as 108 °C/min, in HBSS with 14% glycerol as 24 °C/min, and in HBSS with 10% DMSO as 38 °C/min (for an assumed Vb value of 0.6Vo and a S:V ratio of 10.2). The corresponding values with an assumed Vb value of 0.6Vo and a S:V ratio of 3.4 were 72 °C/min (in HBSS), 36 °C/min (in HBSS with 14% glycerol), and 26 °C/min (in HBSS with 10% DMSO). These values are comparable (± 25%) to those obtained earlier using a S:V ratio of 6.8 (Table 2).
4. Discussion
The best-fit parameters obtained in this study using the DSC volumetric shrinkage data during freezing of X. helleri spermatozoa appeared to be lower than the reported suprazero (above zero and in the absence of extracellular ice) permeability values for mammalian species [40]. A similar lowering of the sperm cell water permeability parameter values was also found previously by Devireddy and colleagues for mouse [24], human [25], horse [26,27], canine [28], boar [29], and Pacific oyster [30]. This discrepancy between the membrane permeabilities may be associated with possible changes in the sperm cell plasma membrane during suprazero cooling. These changes could include either a lipid phase transition between 0 and 4 °C, or cold shock damage or “chilling” injury during cooling. The presence of extracellular ice further alters the cell membrane transport properties [43,45]. In general, for mammalian cells, the average activation energy obtained in the presence of extracellular ice is approximately twice as large as that for studies conducted in unfrozen solutions at higher temperatures [43]. These changes in membrane transport properties might be associated with a variety of thermotropic (temperature dependent) phase phenomena. For example, the temperature reduction that induces solidification in the extracellular medium may lead to lyotropic (i.e. independent of cooling rate) membrane phase changes and corresponding alterations of water transport and also membrane fluidity [40]. Further studies are needed to elucidate the precise mechanism by which extracellular ice alter membranes during a freezing process.
A comparison of the membrane permeability parameters reported earlier for C. gigas sperm cells in the presence of HBSS (1000 mOsm/kg) and 8% DMSO [30] with the values obtained in the present study for X. helleri sperm cells in the presence of HBSS (300 mOsm/kg) and 10% DMSO revealed the following differences: the apparent activation energies (different by a factor of 2) and the reference membrane permeability (different by a factor of 4) were higher for X. helleri sperm cells. Additionally, the reported optimal rates of freezing of C. gigas haploid and diploid sperm cells (~53 °C/min and ~44 °C/min) with 8% DMSO were higher (~27–40%) than the corresponding values with 10% DMSO for X. helleri sperm cells (23–32 °C/min). Clearly, the relative importance of the effect of extracellular ice and concentration of CPAs on the predicted membrane permeability parameters (Lpg or Lpg[cpa] and ELp or ELp[cpa]) and on the optimal rates of cooling is dependent on the cell type and the choice of freezing media.
The DSC technique was used to obtain water transport data and water permeability parameters (Lpg or Lpg[cpa] and ELp or ELp[cpa]) of X. helleri sperm cells in three different cryoprotective media. There was a large increase (35–80%) in the measured value of reference membrane permeability, Lpg (or Lpg[cpa]) and the activation energy, ELp (or ELp[cpa]) obtained in the presence and absence of CPAs (with an assumed Vb values of 0.6Vo (Table 2) or 0.4Vo (Table 3). However, when the assumed value was increased to 0.8Vo, the converse was found to be true, i.e. adding either glycerol or DMSO decreased (~35%) the measured value of the activation energy, ELp (or ELp[cpa]) while the values of reference membrane permeability, Lpg (or Lpg[cpa]) were not effected either due to the addition of glycerol (a difference of 3%) or DMSO (a difference of 7%) when compared to values obtained in the absence of CPAs (Table 3).
The best-fit permeability parameters were incorporated into a model of water transport to predict optimal cooling rates for X. helleri sperm cells (Tables 2 and 3). The optimal rates in the presence of 14% glycerol or 10% DMSO were smaller (~70%) than the predicted optimal values in their absence. This is surprising, as the presence of CPAs would be expected to increase the ability of the cell membrane to dehydrate at faster cooling rates as shown for mouse and human sperm cell suspensions [24,25] and mammalian (rat) hepatocytes [45]. However, a decrease in the predicted optimal cooling rates in the presence of CPAs was observed in sperm cells of stallion [26], boar [29], and Pacific oyster [30]. Further studies are needed to elucidate the precise mechanism by which CPAs alter membranes during and after freezing and thawing process.
The predicted optimal rates of freezing X. helleri sperm cells obtained in the present study ranged from 71 to 90 °C/min in HBSS, 20 to 30 °C/min in HBSS with 14% glycerol, and 24 to 32 °C/min in HBSS with 10% DMSO (Tables 2 and 3). The optimal values in HBSS with 14% glycerol were comparable to the experimentally determined optimal rate of freezing reported recently for X. helleri sperm cells [23]. The highest post-thaw motility occurred when the X. helleri sperm cells were suspended in HBSS with 14% glycerol were cooled at rates ranging from 20 to 35 °C/min from 5 to −80 °C before plunging into liquid nitrogen, and thawed at 40 °C in a water bath for 7 s [23]. However, Huang et al. [23] reported the highest post-thaw motility (~77%) at a cooling rate of 20 °C/min. This cooling rates is lower (~30%) than the optimal rate obtained assuming Vb = 0.6Vo (Table 2) and close (~2%) to the predicted optimal cooling rate assuming Vb = 0.8Vo (Table 3). As stated earlier, the predicted optimal rates using our model are probably over-estimated due to the non-inclusion of heterogeneous or a surface-catalyzed nucleation mechanism. Clearly, the close agreement between the numerically predicted and empirically determined optimal rate of freezing suggests that the membrane permeability parameters presented in this study will help to establish cryopreservation of X. helleri sperm cells on a firm biophysical basis.
In conclusion, the water transport (volumetric shrinkage) data for X. helleri sperm cells in the presence of extracellular ice and CPAs (glycerol and DMSO) during freezing was obtained in this study using the DSC technique at a cooling rate of 20 °C/min. The measured water transport data in the presence and absence of CPAs was curve-fitted to a model of water transport (Eqs. (3) and (4)), to predict membrane permeability parameters (Lpg and ELp or Lpg[cpa] and ELp[cpa]). The measured “best fit” permeability parameters ranged from, Lpg = 0.409 × 10−15 to 2.47 × 10−15 m3/Ns (0.0024–0.0145 μm/min atm) and ELp = 41.65–118.98 kJ/mol (9.96–28.45 kcal/mol). The permeability parameters obtained in this study predict an optimal rate of freezing for X. helleri sperm cells ranging from 20 to 35 °C/min, which agrees with empirically determined optimal experimental rate (20 °C/min) of freezing X. helleri sperm cells [23].
Acknowledgments
This work was supported in part by a grant from the Louisiana Board of Regents (LEQSF 2002-05-RD-A-03) to RD, and grants from the National Institutes of Health and the USDA-SBIR program to TRT. This manuscript was approved for publication by the Director of the Louisiana Agricultural Experiment Station as manuscript number 04-11-0491.
References
- 1.Meyer A. The evolution of sexually selected traits in male swordtail fishes (Xiphophorus:Poeciliidae) Heredity. 1997;79:329–37. [Google Scholar]
- 2.Meyer A, Morrissey JM, Schartl M. Recurrent origin of a sexually selected trait in Xiphophorus fishes inferred from a molecular phylogeny. Nature. 1994;368:539–42. doi: 10.1038/368539a0. [DOI] [PubMed] [Google Scholar]
- 3.Kallman KD. The sex determining mechanism of the poeciliid fish, Xiphophorus montezumae, and the genetic control of the sexual maturation process and adult size. Copeia. 1983;3:755–69. [Google Scholar]
- 4.Kallman KD. A new look at sex determination in poeciliid fishes. In: Turner BJ, editor. Evolutionary genetics of fishes. New York: Plenum Publishing Corporation; 1984. pp. 95–171. [Google Scholar]
- 5.Tiersch TR, Chandler RW, Kallman KD, Wachtel SS. Estimation of nuclear DNA content by flow cytometry in fishes of the genus Xiphophorus. Comp Biochem Physiol B. 1989;94:465–8. doi: 10.1016/0305-0491(89)90182-x. [DOI] [PubMed] [Google Scholar]
- 6.Breuckmann A, Paris F, Schreibman Martin P, Bluem V. Immunoreactive gonadotropin-releasing hormone (GnRH) in the brain and pituitary of adult and juvenile swordtails (Xiphophorus helleri, Teleostei, Poeciliidae) J Morphol. 1996;230:55–67. doi: 10.1002/(SICI)1097-4687(199610)230:1<55::AID-JMOR5>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 7.Flynn KM, Schreibman MP, Yablonsky-Alter E, Banerjee SP. Sexually dimorphic development and binding characteristics of NMDA receptors in the brain of the platyfish. Gen Comp Endocrinol. 1999;115:282–91. doi: 10.1006/gcen.1999.7317. [DOI] [PubMed] [Google Scholar]
- 8.Beaugrand JP, Goulet C. Distinguishing kinds of prior dominance and subordination experiences in males of green swordtail fish (Xiphophorus helleri) Behav Processes. 2000;50:131–42. doi: 10.1016/s0376-6357(00)00096-6. [DOI] [PubMed] [Google Scholar]
- 9.Hoefler CD, Morris MR. A technique for the temporary application and augmentation of pigment patterns in fish. Ethology. 1999;105:431–8. [Google Scholar]
- 10.Rosenthal GG, Evans CS. Female preference for swords in Xiphophorus helleri reflects a bias for large apparent size. Proc Natl Acad Sci USA. 1998;95:4431–6. doi: 10.1073/pnas.95.8.4431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Trainor BC, Basolo AL. An evaluation of video playback using Xiphophorus helleri. Anim Behav. 2000;59:83–9. doi: 10.1006/anbe.1999.1289. [DOI] [PubMed] [Google Scholar]
- 12.De Wolf W, Seinen W, Hermens JLM. N-Acetyltransferase activity in rainbow trout liver and in vitro biotransformation of chlorinated anilines and benzenes in fish. Xenobiotica. 1993;23:1045–56. doi: 10.3109/00498259309057042. [DOI] [PubMed] [Google Scholar]
- 13.Paris F, Paassen U, Bluem V. Sperm storage of the female swordtail (Xiphophorus helleri) Verh Ges Ichthyol. 1998;1:157–65. [Google Scholar]
- 14.Dove AD. Richness patterns in the parasite communities of exotic poeciliid fishes. Parasitology. 2000;120:609–23. doi: 10.1017/s0031182099005958. [DOI] [PubMed] [Google Scholar]
- 15.Schmahl G, Schmidt H, Ritter G. The control of ichthyophthiriasis by a medicated food containing quinine: efficacy tests and ultrastructure investigations. Parasitol Res. 1996;82:697–705. doi: 10.1007/s004360050188. [DOI] [PubMed] [Google Scholar]
- 16.Hogarth PJ. Protection of the developing foetus from immunological attack in Xiphophorus helleri. J Reprod Fertil. 1970;23:532. doi: 10.1530/jrf.0.0230532. [DOI] [PubMed] [Google Scholar]
- 17.McConnell TJ, Godwin UB, Norton SF, Nairn RS, Kazianis S, Morizot DC. Identification and mapping of two divergent, unlinked major histocompatibility complex class II B genes in Xiphophorus fishes. Genetics. 1998;149:1921–34. doi: 10.1093/genetics/149.4.1921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schartl M. Platyfish and swordtails: a genetic system for the analysis of molecular mechanisms in tumor formation. Trends Genet. 1995;11:185–9. doi: 10.1016/S0168-9525(00)89041-1. [DOI] [PubMed] [Google Scholar]
- 19.Nairn RS, Kazianis S, McEntire BB, Della CL, Walter RB, Morizot DC. A CDKN2-like polymorphism in Xiphophorus LG V is associated with UV-B-induced melanoma formation in platyfish-swordtail hybrids. Proc Natl Acad Sci USA. 1996;93:13042–7. doi: 10.1073/pnas.93.23.13042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rana KJ. Cryopreservation of fish spermatozoa Cryopreservation and freeze-drying protocols. Methods Mol Biol. 1995;38:151–65. doi: 10.1385/0-89603-296-5:151. [DOI] [PubMed] [Google Scholar]
- 21.Huang C, Dong Q, Tiersch TR. Sperm cryopreservation of a live-bearing fish, the platyfish Xiphophorus couchianus. Theriogenology. 2004;62:971–89. doi: 10.1016/j.theriogenology.2003.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Huang C, Dong Q, Walter RB, Tiersch TR. Initial studies on sperm cryopreservation of a live bearing fish, the green swordtail Xiphophorus helleri. Theriogenology. 2004;62:179–94. doi: 10.1016/j.theriogenology.2003.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang C, Dong Q, Walter RB, Tiersch TR. Sperm cryopreservation of green swordtail Xiphophorus helleri, a fish with internal fertilization. Cryobiology. 2004;48:295–308. doi: 10.1016/j.cryobiol.2004.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Devireddy RV, Swanlund DJ, Roberts KP, Bischof JC. Sub-zero water permeability parameters of mouse spermatozoa in the presence of extracellular ice and cryoprotective agents. Biol Reprod. 1999;61:764–75. doi: 10.1095/biolreprod61.3.764. [DOI] [PubMed] [Google Scholar]
- 25.Devireddy RV, Swanlund DJ, Roberts KP, Pryor JL, Bischof JC. The effect of extracellular ice and cryoprotective agents on the water permeability parameters of human sperm plasma membrane during freezing. Hum Reprod. 2000;15:1125–35. doi: 10.1093/humrep/15.5.1125. [DOI] [PubMed] [Google Scholar]
- 26.Devireddy RV, Olin T, Swanlund DJ, et al. Cryopreservation of equine spermatozoa: optimal cooling rates in the presence and absence of cryoprotective agents. Biol Reprod. 2002;66:222–31. doi: 10.1095/biolreprod66.1.222. [DOI] [PubMed] [Google Scholar]
- 27.Devireddy RV, Swanlund DJ, Alghamdi AS, et al. Measured effect of collection and cooling conditions on the motility and the water transport parameters at subzero temperatures of equine spermatozoa. Reproduction. 2002;124:643–8. [PubMed] [Google Scholar]
- 28.Thirumala S, Ferrer MS, Al-Jarrah A, Eilts BE, Paccamonti DL, Devireddy RV. Cryopreservation of canine spermatozoa: theoretical prediction of optimal cooling rates in the presence and absence of cryoprotective agents. Cryobiology. 2003;47:109–24. doi: 10.1016/j.cryobiol.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 29.Devireddy RV, Fahrig B, Godke RA, Leibo SP. Subzero water transport characteristics of boar spermatozoa confirm observed optimal cooling rates. Mol Reprod Dev. 2004;67:446–57. doi: 10.1002/mrd.20041. [DOI] [PubMed] [Google Scholar]
- 30.He Y, Dong Q, Tiersch TR, Devireddy RV. Variation in the membrane transport properties and predicted optimal rates of freezing for spermatozoa of diploid and tetraploid pacific oyster Crassostrea gigas. Biol Reprod. 2004;70:1428–37. doi: 10.1095/biolreprod.103.025296. [DOI] [PubMed] [Google Scholar]
- 31.Yuan S, Diller KR. Study of freezing biological systems using optical differential scanning calorimeter. Proceedings of ASME summer BED conference. 2001;50:117–8. [Google Scholar]
- 32.Diller KR. New techniques in cryomicroscopy. Cryobiology. 2002;45:250–1. [Google Scholar]
- 33.Mazur P. Kinetics of water loss from cells at subzero temperatures and the likelihood of intracellular freezing. J Gen Physiol. 1963;47:347–69. doi: 10.1085/jgp.47.2.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mazur P. Cryobiology: the freezing of biological systems. Science. 1970;168:939–49. doi: 10.1126/science.168.3934.939. [DOI] [PubMed] [Google Scholar]
- 35.Meryman HT. Review of biological freezing. In: Meryman HT, editor. Cryobiology. New York: Academic Press; 1966. pp. 1–114. [Google Scholar]
- 36.Toner M. Nucleation of ice crystals in biological cells. In: Steponkus PL, editor. Advances in low-temperature biology. Greenwich: JAI Press; 1993. pp. 1–52. [Google Scholar]
- 37.Lovelock JE. Haemolysis of human red blood-cells by freezing and thawing. Biochim Biophys Acta. 1953;10:414–26. doi: 10.1016/0006-3002(53)90273-x. [DOI] [PubMed] [Google Scholar]
- 38.Mazur P, Leibo SP, Chu EHY. A two-factor hypothesis of freezing injury. Exp Cell Res. 1972;71:255–345. doi: 10.1016/0014-4827(72)90303-5. [DOI] [PubMed] [Google Scholar]
- 39.Devireddy RV, Raha D, Bischof JC. Measurement of water transport during freezing in cell suspensions using a differential scanning calorimeter. Cryobiology. 1998;36:124–55. doi: 10.1006/cryo.1997.2071. [DOI] [PubMed] [Google Scholar]
- 40.Devireddy RV, Bischof JC. Recent advances in cryobiology using calorimetry. In: Kakac S, Smirnov H, Mila MR, editors. Low temperature and cryogenic refrigeration. Dordrecht, The Netherlands: Kluwer Academic Publishers; 2003. pp. 265–94. [Google Scholar]
- 41.Kedem O. Katchalsky Thermodynamic analysis of the permeability of biological membranes to non-electrolytes. Biochim Biophys Acta. 1958;27:229–46. doi: 10.1016/0006-3002(58)90330-5. [DOI] [PubMed] [Google Scholar]
- 42.McCaa C, Diller KR, Aggarawal SJ, Takahashi T. Cryomicroscopic determination of the membrane osmotic properties of human monocytes at subfreezing temperatures. Cryobiology. 1991;28:391–9. doi: 10.1016/0011-2240(91)90046-q. [DOI] [PubMed] [Google Scholar]
- 43.McGrath JJ. Membrane transport properties. In: McGrath JJ, Diller KR, editors. Low temperature biotechnology: emerging applications and engineering contributions. New York: ASME Press; 1988. pp. 273–330. [Google Scholar]
- 44.Levin RL, Cravalho EG, Huggins CG. A membrane model describing the effect of temperature on the water conductivity of erythrocyte membranes at subzero temperatures. Cryobiology. 1976;13:415–29. doi: 10.1016/0011-2240(76)90097-3. [DOI] [PubMed] [Google Scholar]
- 45.Smith DJ, Schulte M, Bischof JC. The effect of dimethylsulfoxide on the water transport response of rat hepatocytes during freezing. ASME J Biomech Eng. 1998;120:549–58. doi: 10.1115/1.2834744. [DOI] [PubMed] [Google Scholar]
- 46.Bevington PR, Robinson DK. Data reduction and error analysis for the physical sciences. New York: McGraw-Hill; 1992. pp. 107–24. [Google Scholar]
- 47.Montgomery DC, Runger GC. Applied statistics and probability for engineers. New York: John Wiley & Sons Inc; 1994. pp. 471–529. [Google Scholar]
- 48.Mazur P. Equilibrium, quasi-equilibrium, and nonequilibrium freezing of mammalian embryos. Cell Biophys. 1990;17:53–92. doi: 10.1007/BF02989804. [review] [DOI] [PubMed] [Google Scholar]


