Abstract
The thoracic thymus is the primary vertebrate organ for T cell generation. Accessory cervical thymi have also been identified in humans and mice, and shown in mice to be independent functional organs that support T cell development. However, their origin and functional significance remain unclear. Here we show that cervical thymi in mice have two origins: delayed differentiation of endodermal precursors, and transdifferentiation of parathyroid-fated cells. Compared to thoracic thymus, parathyroid-origin cervical thymi (pCT) express low levels of the thymic epithelial cell-specific transcription factor FOXN1. Consequently, pCT form a distinct microenvironment that supports an atypical thymocyte development pathway, generating T cells with unconventional phenotypic characteristics. Our data demonstrate a transdifferentiation origin for a subset of cervical thymi, with specific functional consequences for T cell development.
Introduction
In addition to the main, thoracic thymus, cervical thymi located in the neck have been reported to be present in about 50% of both humans and mice 1,2, and have recently been shown to be a production site for functional T cells that can contribute to peripheral immunity 3,4. Cervical thymi in mice have also been reported to have an altered tissue-restricted antigen profile, which could lead to the production of auto-reactive T cells 3. Previous reports indicate that cervical thymus in mice are identifiable perinatally 3,4, and are frequently associated with the parathyroid and/or thyroid glands 3,4. Further analysis supported a ‘birthdate’ for cervical thymi 1 some time after E12.5 5, which is later than the initiation of thoracic thymus organogenesis at about E11. However, their cellular origin remains unknown.
The thymus and parathyroid glands have a common origin from 3rd pharyngeal pouch-derived primordia that separate into distinct organs during morphogenesis 6. We and others have previously shown that parathyroid cell clusters are also located outside of these two organs 7,8; our data showed that these ectopic parathyroid clusters arise as a normal consequence of organ separation, in both mice and humans 7. The location and timing of the appearance of cervical thymi and their possible functional differences compared to thoracic thymi make it important to determine their cellular origins and the molecular mechanisms controlling their development.
In the current study, we show that cervical thymi arise from two distinct precursor cell populations, both of which originate from the separation of the 3rd pharyngeal pouch-derived endodermal primordium into distinct thymus and parathyroid organs. We further show that a subset of cervical thymi arise from cells previously differentiating as parathyroid, and that this subset generate T cells that are functionally distinct from those made by the thoracic thymus. Thus, the generation and origins of cervical thymi have clear consequences for their function, and may provide a unique model for understanding the establishment and maintenance of cell fate.
Results
Cervical thymus development requires parathyroid organogenesis
We tested whether cervical thymus occurrence correlated with known mechanisms of thymus and parathyroid organ development using a genetic approach. Foxn1 is a transcription factor required for thymic epithelial cell (TEC) differentiation; whose loss of function causes the nude (nu) phenotype 9. We detected cervical thymi by expression of a Foxn1::GFP transgene in newborn Foxn1nu/nu mice, although they were consistently smaller and present at lower frequency than those in wild-type mice (Chi-squared test, p=0.0116) (Table 1). We also analyzed Gcm2 null mice, in which all parathyroid cells undergo apoptosis by E12 thus precluding organ separation 7, for the incidence of cervical thymi. No cervical thymi were detected among 33 Gcm2 null mutants (Chi-squared test, p=0.0001) (Table 1). These observations suggest that, like the thoracic thymus, initial cervical thymus formation does not require Foxn1, while subsequent development and growth does require Foxn1. However, unlike the thoracic thymus, normal parathyroid organogenesis is required for cervical thymus development.
Table 1.
Frequency of cervical thymus in neonatal Foxn1 or Gcm2 null mice1.
| Genotype | Number | Incidence Of Cervical Thymus2 |
|---|---|---|
| Foxn1 +/nu | 22 | 13/22 |
| Foxn1 nu/nu | 19 | 4/19 |
| Gcm2 +/+ | 44 | 21/44 |
| Gcm2 +/- | 61 | 24/61 |
| Gcm2 −/− | 33 | 0/33 |
The presence of one or more cervical thymus was scored in individual neonatal mice.
Frequencies were calculated as different from expected Mendelian ratios using the Chi-square test. Only Gcm1−/− mice showed frequencies significantly different from controls.
Cervical thymi originate in late fetal stages
The dependence of cervical thymus development on normal thymus-parathyroid organogenesis suggested three possible origins (Fig.1a). We have shown that the parathyroid domain tends to fragment during normal organ separation and migration, leaving clusters of Gcm2 and parathyroid hormone (Pth) positive parathyroid cells between the position of the parathyroid and the thymus after these organs separate 7. At some frequency, these small clusters of parathyroid cells could be converted to a thymus fate. It is also possible that a few Foxn1 positive cells are excluded from the encapsulated thymus domain during thymus-parathyroid organ separation. The final possibility is that cells that express neither the Foxn1 nor Gcm2 organ-specific markers within the common thymus-parathyroid primordia are released during organ separation and survive in the neck region, and can be induced by some mechanism to differentiate to a thymus fate.
Figure 1. The ontology of cervical thymi and their dependence on parathyroid organogenesis.
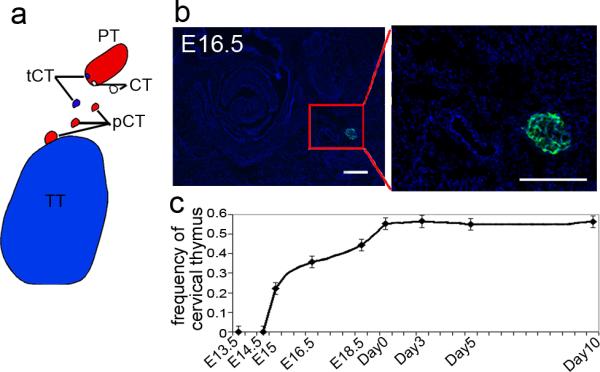
(a) Schematic of the possible origins of the cervical thymus. Red indicates either parathyroids (PT) or parathyroid-derived cervical thymi (pCT); blue indicates thoracic thymus (TT) and thoracic thymus-derived cervical thymi (tCT); white indicates nonthymus and non-parathyroid origin for cervical thymi (CT). (b) Cervical thymus detected on transverse section of E16.5 Foxn1::EGPF transgenic embryo (green, GFP; blue, DAPI). (c) Occurrence of cervical thymus begins at E15 embryonic stage; frequency increases with development until birth (n≥10 for each time point listed), (Error bars represent SEM, Scale bar=50μm).
Since cervical thymus development occurs later than that of the thoracic thymus 5, we determined at which stage we could first detect cervical thymi, analyzing Foxn1Cre;Rosa26YFP mice from E13.5 until 10 days after birth. The earliest stage at which we detected Foxn1+ cells outside the thoracic thymus was E15, at a frequency of 2/9 embryos, consistent with previously published data that concluded that cervical thymi first turn on Foxn1 after E12.5 and before E15.5 5. The frequency of cervical thymus clusters gradually increased through the later stages of fetal development, reaching a maximum of about 50% at newborn stage (Fig.1b, c). The timing of appearance of Foxn1-expressing cells in the cervical thymi is consistent with their arising from small clusters of Foxn1-negative cells created by the normal process of thymus-parathyroid separation.
A subset of cervical thymi originate from parathyroid cells
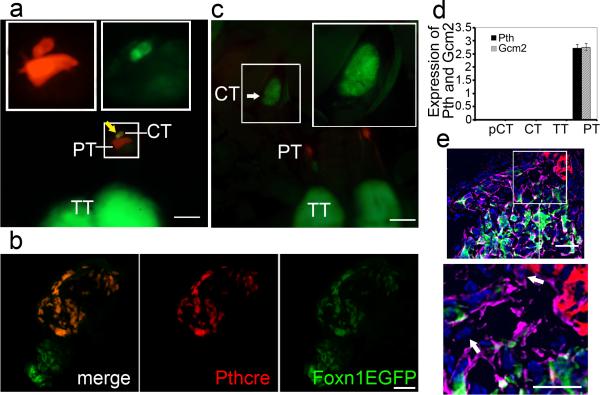
To test whether parathyroid cells might be the origin of cervical thymi, we used a Parathyroid Hormone (Pth)-Cre transgene to activate a fluorescent reporter, tdTomato, and used the Foxn1::EGFP transgene 10 or a FOXN1 antibody to track Foxn1 gene expression. In these mice, parathyroid origin epithelial cells are red (tdTomato+) while Foxn1-expressing thymus cells are green (EGFP+). This Pth-Cre transgene does not have activity in the thoracic thymus (Supplementary Fig. S1). At the newborn stage, two types of cervical thymi were observed: those in which cells were doubly positive for both red and green fluorescence (Fig.2a, b and Supplementary Fig. S2), which we designated as pCT and those with only green fluorescence (Foxn1-expressing) (Fig.2b,c). The pCT had a history of parathyroid fate lineage (red) but current Foxn1 expression (green), and had down-regulated their parathyroid differentiation program, as indicated by absence of the parathyroid-specific markers Gcm2 and Pth (Fig. 2d).
Figure 2. Parathyroid origin of a subset of cervical thymi.
(a, c) Whole mount images of 3 day old PthCre;CAG-td-Tomato;Foxn1::EGPF mice, parathyroid (PT) is red, thoracic thymus (TT) is green. Large boxes show high magnification of cervical thymi (CT) in small boxes. (Scale bar=200μm). (a) CT of parathyroid origin (yellow arrow) next to parathyroid. (b) Section of cervical thymi, one with green only, and one with green and red. (Scale bar=50μm). (c) Whole mount image of non-parathyroid origin CT (white arrow). (d) No expression of Pth and Gcm2 in cervical thymus regardless of origin. (N>3, Error bars represent SEM). (e) Epithelial (pankeratin-positive) cells at the thymus-parathyroid interface at E12.5 that express neither FOXN1 nor PTH (white arrow). Foxn1::EGPF, green; Pth-Cre activated CAG-td Tomato, red; pan-keratin, violet; DAPI, blue. (Scale bar=50μm).
Most cervical thymi appear to originate from delayed differentiation of endodermal precursors, since about 75% of cervical thymi (Table 2) arise from cells that express neither parathyroid markers nor Foxn1 prior to about E15.5. We will refer to this class as “CT”. Consistent with this conclusion, we identified a small number of epithelial cells at the interface between these two organ domains at E12, just prior to thymusparathyroid separation that express neither thymus nor parathyroid differentiation markers (Fig. 2e). While in the absence of a positive lineage marker we cannot conclusively identify these cells as the source of the non-parathyroid origin cervical thymi, their existence is consistent with this model. pCT arise from the transdifferentiation of parathyroid cells, which express Pth at early stages, but subsequently turn off the parathyroid program and switch to a thymus cell fate as shown by Foxn1 expression. We found that CT were broadly distributed between the thyroid and thymus in the neck region, similar to previous reports 3,4. In contrast, most pCT are located near the posterior thyroid, and thus closer to the parathyroid glands proper (Supplementary Fig. S3).
Table 2.
Frequency of different types of cervical thymi at postnatal day 5
| 1 CT/mouse | 2 CT/mouse | 3 CT/mouse | 4 CT/mouse | Total | |
|---|---|---|---|---|---|
| Number of mice | 23 | 10 | 3 | 1 | 37 |
| Total number of CT/pCT | 23 | 20 | 9 | 4 | 56 |
| Frequency of CT (%) | 78.6 | 75 | 77.8 | 100 | - |
| Frequency of pCT (%) | 21.7 | 25 | 22.2 | 0 | - |
pCT and CT have distinct thymocyte differentiation profiles
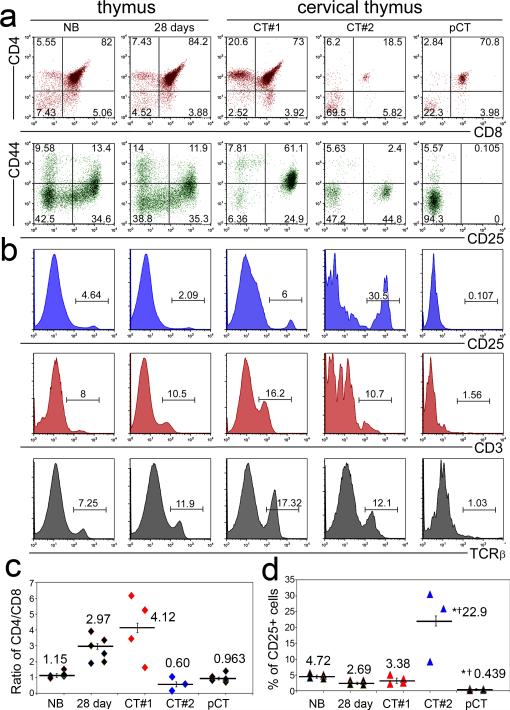
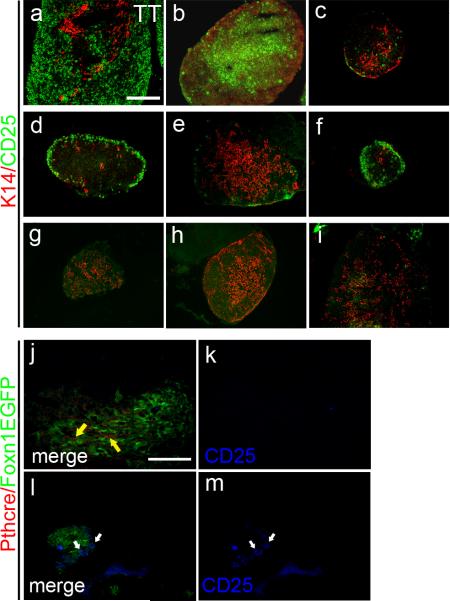
Given the two different origins for cervical thymi, we asked whether their cellular origin influences TEC differentiation and thus the ability to support thymocyte differentiation. We performed flow cytometric analysis of thymocyte differentiation on individual microdissected cervical thymi from postnatal day 5, and compared them to newborn and 28 day-old thoracic thymi. About 25% of cervical thymi showed an aberrant thymocyte differentiation profile, with a reduced CD4+:CD8+ ratio, and an absence of CD25+ immature thymocytes (Fig. 3 a-d). This phenotype was confirmed by immunohistochemistry for CD25, which showed variable CD25+ distribution and phenotypes compared to thoracic thymus (Fig. 4 a-i). Most strikingly, no CD25+ cells could be detected in about 25% of cervical thymi (Fig. 4g-i). This phenotype was not correlated with organ size, but did track with lineage, as all parathyroid-derived pCT that were assayed for CD25+ thymocytes specifically lacked these cells (Fig. 4j-m). In contrast, pCT contain increased frequencies of B, NK and granulocyte populations, while results for CT were similar to thoracic thymus (Supplementary Fig. S4). While CD25+ cells were consistently absent from pCT, the results for CT at 5 days postnatal fell into two categories, either similar to thoracic thymus (see Fig. 3, CT#1) to very high (CT#2). The frequency of CD25+ cells were consistently anti-correlated with the % DP cells (Fig. 3c, d), suggesting that these CT were less well developed.
Figure 3. Parathyroid-derived cervical thymus support atypical T cell development.
(a) Flow cytometric analysis of thoracic and cervical thymus at postnatal day 3 for CD4, CD8, CD25 and CD44 shows changes in the frequency of thymocyte subsets in cervical thymi (N>3). (b) Two classes of cervical thymi with either very low CD25 and TCRβ expression, or levels similar to thoracic thymus (N>3). Positive gates are indicated. (c) Ratio of CD4/CD8 is lower in parathyroid-derived cervical thymus, but shows variety in non-parathyroid derived cervical thymus, either similar/higher (CT#1, in red) or lower (CT#2, in blue) compared to thoracic thymus (N>3, Error bars represent SEM,). (d) Percentage of CD25+ cells is significantly lower in pCT, but is significantly higher in the CT#2 group of non-parathyroid derived cervical thymus (N>3; Error bars represent SEM, *significant relative to NB, t-Test, p<0.01 †significant relative to 28day, t-Test, p<0.01 ).
Figure 4. Parathyroid-derived cervical thymus cannot support normal T cell development.
(a) CD25+ cells (green) are widespread in E15.5 thoracic thymus. and in some cervical thymus (b, c). (d, e and f) CD25+ cells mainly locate at the sub-capsular area in some cervical thymi. (g, h and i) CD25+ cells are absent in some cervical thymus. (j, k) Parathyroid derived cervical thymus does not contain CD25+ cells (blue). (l, m) CD25+ cells are present in non-parathyroid derived cervical thymus. (Scale bar=100μm)
Analysis of TCRβ expression further supported these different CT and pCT phenotypes. Thymocytes from pCT consistently had lower frequencies of TCRβ+ thymocytes than those 28 day thoracic thymus, both when measured on total thymocytes and on subsets identified by CD4 and CD8 (Fig. 3b, Supplementary Fig. S5, and Table 3). Reduced TCRβ+ % was seen for all subsets except for CD8 SP, which looked similar to the NB stage. The levels of TCRβ were also significantly reduced on all subsets except for DPs (Table 3). All CT regardless of CD25+ or DP frequencies have similar TCRβ frequencies on individual subsets, and are generally similar to 28 day controls (Supplementary Fig. S5). However, both CD8+ and DN subsets displayed higher frequencies of TCRβ+ cells than either newborn or 28day thymus. The DN expression is particularly interesting, and could indicate either that cells have premature TCRβ surface expression or that more mature cells are down regulating their CD4 and/or CD8 expression. As CD3 expression tracked with TCRβ (Fig. 3b) and the TCRβ+ DN cells showed a biphasic structure in their levels of TCRβ expression, we lean towards the latter explanation.
Table 3.
Intensity and frequency of TCRβ expression on cervical thymus-derived thymocytes
| MFI1 of TCRb+ cells | % of TCRb+ cells | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| NB | 28days | CT#1 | CT#2 | pCT | NB | 28days | CT#1 | CT#2 | pCT | |
| Total | 68.1 | 81.9 | *156.3 | 94.7 | *†52.4 | 7.13 | 12.5 | *†17.74 | *14.1 | *†1.02 |
| CD4+ | 260.3 | 280 | 260.7 | †241.7 | *†144 | 99.7 | 99.5 | 99.5 | 99.4 | *†73.5 |
| CD8+ | 160 | 222.3 | †165.3 | †164 | †120.3 | 30.6 | 86 | *†96.3 | *†93 | †35.7 |
| CD4+8+ | 47.4 | 47.9 | 47.1 | 47.3 | 44.97 | 22.1 | 24.7 | †19.8 | 21.8 | *†11.5 |
| CD4-8- | 43.4 | 76.1 | *92.9 | *86.2 | *†95.2 | 6.8 | 18.3 | *†24.6 | *†26.6 | *†3.8 |
Median Fluorescence Intensity
significant at p<0.01 relative to NB (t-Test)
significant at p<0.01 relative to 28 days (t-Test)
pCT phenotypes are consistent with low Foxn1 expression
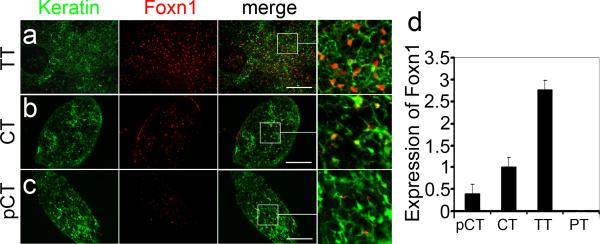
The thymocyte phenotypes observed in pCT, including selective absence of CD25+ thymocytes, reduced CD4+:CD8+ ratio, reduced TCR levels in the presence of DP and SP thymocytes, and increased frequencies of NK and B cells, are similar to the thoracic thymus phenotypes we previously reported for a severely hypomorphic allele of Foxn1, Foxn1Δ 11-14. Foxn1 hypomorphic alleles result in thymi with a variety of specific and detrimental effects on TEC differentiation and function 11-16. pCT had fewer FOXN1+ TECs, as well as reduced Foxn1 mRNA and protein levels per cell compared to both other cervical thymi and thoracic thymus (Fig. 5a-d), suggesting that reduced Foxn1 levels suppressed TEC differentiation in pCT, and that these defects underlie the thymocytes phenotypes in pCT. Furthermore, CT had average Foxn1 levels that were less than half that of thoracic thymus, although still twice that of pCT (Fig. 5b, d). These reduced levels may also underlie the milder but significant differences in thymocyte differentiation in CT.
Figure 5. Parathyroid-lineage cervical thymi have reduced Foxn1 levels at postnatal day 3.
(a) FOXN1 in thoracic thymus. (b) FOXN1 in non-parathyroid derived cervical thymus. (c) FOXN1 in parathyroid-derived cervical thymus is lower than in non-parathyroid derived cervical thymus. (d) Real-Time PCR analysis of Foxn1 expression in parathyroid-derived (pCT) and non-parathyroid-derived (CT) cervical thymus, thoracic thymus (TT) and parathyroids (PT) (N>3, Error bars represent SEM). (Scale bar=50μm).
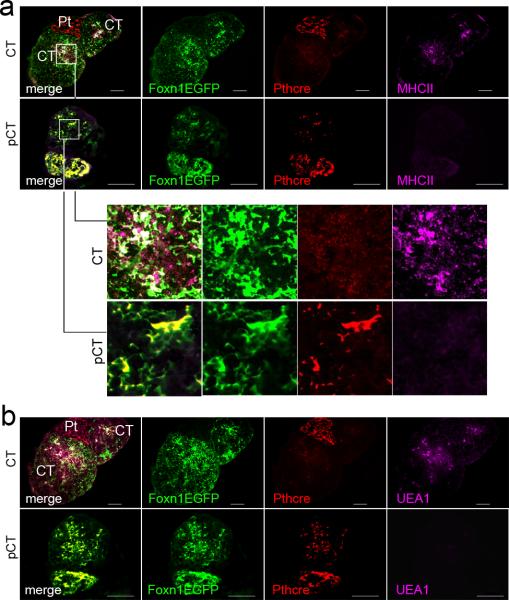
To determine whether the structure of the thymic microenvironment in CT and pCT was similar to that of Foxn1 hypomorphic thoracic thymi, we analyzed the expression of MHC class II and UEA-1, two TEC markers that are significantly reduced in Foxn1Δ/Δ mice 11. pCT (parathyroid lineage cervical thymi) expressed very low levels of MHC class II and were negative for UEA-1, compared to non-parathyroid-derived CT (Fig. 6a, b). This phenotype is similar to that of Foxn1Δ/Δ mice 11, and is consistent with the thymocyte differentiation defects shown above. These data support the conclusion that parathyroid-derived cervical thymi have reduced Foxn1 expression resulting in altered microenvironments, and thus produce thymocytes with functional differences similar to those made by Foxn1Δ/Δ hypomorphic thoracic thymi, but in wild-type mice.
Figure 6. Deficient differentiation of epithelial cells in parathyroid-derived cervical thymus.
(a) Expression of MHCII in parathyroid-derived cervical thymus (pCT; lower panels) is lower than in non-parathyroid derived cervical thymus (CT; upper panels). Higher magnification images of the areas indicated by boxes in the panels in (a) are shown below panel a. (b) UEA-1+ TECs are present in non-parathyroid derived cervical thymus (CT; upper panel) but not in parathyroid-derived cervical thymus (pCT; lower panel). (Scale bar=50μm).
Discussion
Our data present a naturally occurring instance of cell fate switching in vivo, that may be a model for understanding the basis for establishment and maintenance of cell fate. Our results showed that the earliest stage of cervical thymus occurrence is E15-16 days in wild type mice, when cervical thymi are very small (<10 cells). After E15, the incidence of cervical thymus gradually increased and reached a maximum of 50% at neonatal and later stages. This gradual increase in incidence suggests that cervical thymus expansion and differentiation is not induced by a single discrete event, but may have a stochastic or cumulative element. We did find that absence of Foxn1 can reduce but not eliminate cervical thymus formation, and that the cervical thymi that do form in nude mice are smaller than in wild-type mice. Our interpretation of these data is that Foxn1 is required for initial expansion of the cervical thymus, and that in its absence some cervical thymi may be too small to detect by these methods, or may not persist in the absence of Foxn1 function.
Our data clearly show that cervical thymi arise as a byproduct of normal thymus and parathyroid morphogenesis, and show that they can develop via two different differentiation pathways. As cervical thymi are absent in mice that lack parathyroids, parathyroid organogenesis is an absolute requirement for cervical thymus formation, even if they derive from delayed differentiation of endodermal precursors. We did identify cells at the thymus-parathyroid junction in the early primordium that express neither thymus nor parathyroid-specific markers. We surmise that these “unspecified” cells are only separated from the thymus during the process of thymus-parathyroid separation – in the absence of the parathyroid, there is no separation, so they would remain encapsulated within the thoracic thymus. As Foxn1 expression is turned on in cervical thymi after specification and initial primordium development occurs for the thoracic thymus, we do not have a marker to identify the non-parathyroid-derived cells that form the majority of cervical thymi prior to their expression of Foxn1, and thus cannot determine whether those cells have acquired a thymus fate or are at this point still unspecified to either organ lineage. Indeed, both types of cells could be present; the two subsets of CT (illustrated by CT#1 and CT#2) could be due to differing origins for these subtypes, namely whether the cells at time of activating thymus program were already specified to a thymus identity, as opposed to being in an unspecified third pouch progenitor condition. In the absence of a molecular marker that identifies thymus-fated cells prior to Foxn1 expression, we cannot distinguish these possibilities at present. Furthermore, only a subset of parathyroid-fated cells that separate from the main parathyroid convert to the cervical thymus fate. Thus, we cannot at this point easily investigate the initial stages of cervical thymus induction from either cell source. It is possible that differences in the local microenvironments (signaling, matrix components) in which these ectopic clusters are located in addition to differing intrinsic properties of the cell type of origin influence their ability to be induced to a thymus fate.
The differing “birthdates” of CT may contribute to the range of phenotypes observed within the non-parathyroid-derived CT. “Younger” CT should look more like fetal stage thymi with few DP cells and many more CD25+ cells (like the CT #2 example), while “older” CT should look more like postnatal thoracic thymi (like the CT #1 example). While it is not possible to know the age of a specific CT on dissection, this explanation does fit with our observation that the frequencies of DP and CD25+ cells are consistently anti-correlated in CT, and with the increasing frequency of CT across late fetal stages, indicating a range of birthdates. Varying Foxn1 levels could also contribute to phenotypic variability; the ability to effectively upregulate Foxn1 could vary with differing mechanisms of activating the program.
Our data clearly demonstrate that the phenotype of an individual cervical thymus depends on its cell type of origin; parathyroid-origin cervical thymi develop by an atypical path, correlated with a lower level of Foxn1. The distinct phenotype of parathyroid-derived pCT (low MHC Class II expression, absent UEA-1 staining, selective absence of CD25+ cells, low TCRαβ expression, reversed CD4:CD8 ratio, increased NK and B cell frequencies) is identical to the phenotype of the Foxn1Δ/Δ hypomorphic allele that we previously reported 11-13. This T cell phenotype is also similar to that of “T-selected” CD4 cells in humans, which have been shown to have distinct functional characteristics and may play a discrete protective role relative to other CD4+ T cells 17-19. Therefore, the parathyroid-origin cervical thymi can act as a normal source of this specialized subset of T cells that have been proposed to play a unique role in immune function. These results also have potential implications for human immunity, as the presence and type of cervical thymi could influence the frequency of this subset of T cells that has been implicated as providing added protection against infectious diseases 20. Furthermore, patients with Gcm2 mutations 21 should lack both CT and pCT, but specific clinical study of the immune status and phenotypes of these individuals has not been done. The fact that thymi arising from cell fate switching do not completely up regulate their new organ-specific program also has implications for efforts to develop protocols for cellular reprogramming, including for other endodermal organs such as liver and pancreas.
Methods
Mice
Male and female mice were analyzed at E13.5 to 10 days postnatal. Foxn1:eGFP mice were obtained from Dr. Thomas Boehm (Max-Planck Institute of Immunobiology, Freiburg, Germany). Foxn1Cre 22 and Gcm2 null mutant 23 mouse strains and genotyping protocols have been described previously. Rosa26YFP 24, Rosa-td-Tomato 25, and PthCre 26 strains were obtained from The Jackson Labs. All mice were genotyped using PCR. 5′-GAGCTGAAGGGCATCGACTT-3′ and 5′-CTTGTGCCCCAGGATGTTG-3′ were used as primers for Foxn1::eGFP, 5′GGACATGTTCAGGGATCGCCAGGCG-35′-GAGCTGAAGGGCATCGACTT-3′ and 5′-CTTGTGCCCCAGGATGTTG-3′ were used as primers for Foxn1Cre and PthCre, Used as primers for Gcm2 null, 5′-AAG ACC GCG AAG AGT TTG TC-3′, 5′-AAA GTC GCT CTG AGT TGT TAT-3′ and 5′-GGA GCG GGA GAA ATG GAT ATG-3′used as primers for Rosa26YFP, 5′-GAA TTA ATT CCG GTA TAA CTT CG-3′ , 5′-AAA GTC GCT CTG AGT TGT TAT-3′ and 5′-CCA GAT GAC TAC CTA TCC TC-3′ were used as primers for Rosa-td-Tomato. All experiments involving animals were approved by the Institutional IACUC at UGA.
mRNA analysis from individual thoracic and cervical thymi
Total RNA from individual cervical and thoracic thymi from 5-day-old C57BL/6 mice was isolated using the RNEasy kit (Qiagen). cDNA was synthesized from the total RNA and used as a template for real-time relative quantitation for Foxn1 (Invitrogen, Mm0043948_m1), Pth (Invitrogen, Mm00451600_g1), and Gcm2 (Invitrogen, Mm00492312_m1) using commercially available reagents (Invitrogen) on an Applied Biosystems 7500 Real Time PCR System.
Immunohistochemical procedures
Frozen mouse tissues (thoracic and cervical thymus) were sectioned (7mm) and fixed in 4% formaldehyde in 0.1 m phosphate buffer, pH 7.4 for 5 min at 4C, or in absolute acetone for 20 sec at −20 C. Tissues were rinsed with PBS, blocked with 10% donkey serum in 0.1 m phosphate buffer, pH 7.4 for 30 min at RT, and then incubated with anti-Foxn1 (Santa Cruz, G-20, 1:200), anti-B220 (BD, RA3-6B2, 1:100), anti-Cytokeratin (Sigma, C-11, 1:800), anti-CD31 (BD, MEC13.3, 1:100), anti-CD25 (BD, PC61, 1:100), anti-β5t (MBL, PD021, 1:200), anti-CD11c (BD, HL3, 1:100), anti-Aire (Santa Cruz, M-300, 1:200), anti-K5 (Covance, AF138, 1:1000), anti-K14 (Covance, AF64, 1:1000), or UEA1 lectin (Vector Labs, X0922, 1:400). In most experiments, the secondary detection was performed with donkey anti-primary species. For Pthcre;td-Tomato;Foxn1::EGFP observation, tissues were fixed in 4% formaldehyde in 0.1 m phosphate buffer, pH 7.4 for 5 min at 4C, wash with 5% sucrose/PBS buffer, pH 7.4 for one hour, 15% sucrose/PBS buffer, pH 7.4 for one hour then sectioned. Tissues were examined by fluorescent microscopy.
Flow cytometric analysis
Thymocytes harvested from individual thoracic and cervical thymus from 5 day-old mice were suspended at1xFACS buffer (1xPBS with 2% BSA and 0.1% NaN3) and incubated with saturating concentrations of the conjugated monoclonal antibodies CD4-Cy5/APC (Biolegend, GK1.5, 1:150), CD8-Cy7/APC (Biolegend, 53-6.7, 1:150), CD25-APC (BD, PC61, 1:150), and CD44-PerCP (Biolegend, IM-7, 1:150), CD3-PE (Biolegend, 145-2C11, 1:150) and TCR-FITC (BD, 0901, 1:150 ) at 4 C for 30 min, washed, and then fixed with 1% paraformaldehyde (EM Sciences, Ft. Washington, PA) before analysis on a FACS Caliber (Becton Dickinson and Co., San Jose, CA).
Statistics
Data are presented as the mean and SEM. Comparisons between two groups were made using Student's t test. P < 0.05 was considered significant. Chi-squared test was used to test for associations between nude and wild-type categories or Gcm2 mutants and wild-type. P<0.05 was considered significant.
Supplementary Material
Acknowledgements
This work was supported by NIH R21 AI075244 to NRM. We thank Julie Nelson in the CTEGD Flow Cytometry Facility at the University of Georgia, Qiaozhi Wei for assistance in collecting E12.5 embryos, and Thomas Boehm for providing Foxn1::EGFP mice. We also thank Ellen Richie and Clare Blackburn for reading our manuscript before submission.
Footnotes
Author contributions
JL performed the majority of the experiments; ZL performed the initial experiments establishing the system; SX participated in the design, execution, and analysis of flow cytometry studies; NM performed data analysis and experimental design; JL and NM wrote the manuscript.
Conflict of interests
The authors declare no competing financial interest.
References
- 1.Ashour M. Prevalence of ectopic thymic tissue in myasthenia gravis and its clinical significance. J Thorac Cardiovasc Surg. 1995;109:632–5. doi: 10.1016/S0022-5223(95)70343-8. [DOI] [PubMed] [Google Scholar]
- 2.Tovi F, Mares AJ. The aberrant cervical thymus. Embryology, Pathology, and clinical implications. Am J Surg. 1978;136:631–7. doi: 10.1016/0002-9610(78)90324-0. [DOI] [PubMed] [Google Scholar]
- 3.Dooley J, Erickson M, Gillard GO, Farr AG. Cervical thymus in the mouse. J Immunol. 2006;176:6484–90. doi: 10.4049/jimmunol.176.11.6484. [DOI] [PubMed] [Google Scholar]
- 4.Terszowski G, et al. Evidence for a functional second thymus in mice. Science. 2006;312:284–7. doi: 10.1126/science.1123497. [DOI] [PubMed] [Google Scholar]
- 5.Corbeaux T, et al. Thymopoiesis in mice depends on a Foxn1-positive thymic epithelial cell lineage. Proc Natl Acad Sci U S A. 2010;107:16613–8. doi: 10.1073/pnas.1004623107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gordon J, Manley NR. Mechanisms of thymus organogenesis and morphogenesis. Development. 2011;138:3865–78. doi: 10.1242/dev.059998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Z, et al. Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions. PLoS Genet. 2010;6:e1001251. doi: 10.1371/journal.pgen.1001251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dooley J, Erickson M, Farr A. Lessons from Thymic Epithelial Heterogeneity: FoxN1 and Tissue-Restricted Gene Expression by Extrathymic, Endodermally Derived Epithelium. J Immunol. 2009;183:5042–5049. doi: 10.4049/jimmunol.0901371. [DOI] [PubMed] [Google Scholar]
- 9.Nehls M, et al. Two genetically separable steps in the differentiation of thymic epithelium. Science. 1996;272:886–9. doi: 10.1126/science.272.5263.886. [DOI] [PubMed] [Google Scholar]
- 10.Rodewald HR. Thymus organogenesis. Annu Rev Immunol. 2008;26:355–88. doi: 10.1146/annurev.immunol.26.021607.090408. [DOI] [PubMed] [Google Scholar]
- 11.Su DM, Navarre S, Oh WJ, Condie BG, Manley NR. A domain of Foxn1 required for crosstalk-dependent thymic epithelial cell differentiation. Nat Immunol. 2003;4:1128–35. doi: 10.1038/ni983. [DOI] [PubMed] [Google Scholar]
- 12.Xiao S, Manley NR. Impaired thymic selection and abnormal antigen- specific T cell responses in Foxn1(Delta/Delta) mutant mice. PLoS One. 2010;5:e15396. doi: 10.1371/journal.pone.0015396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xiao S, Su DM, Manley NR. T cell development from kit-negative progenitors in the Foxn1Delta/Delta mutant thymus. J Immunol. 2008;180:914–21. doi: 10.4049/jimmunol.180.2.914. [DOI] [PubMed] [Google Scholar]
- 14.Xiao S, Su DM, Manley NR. Atypical memory phenotype T cells with low homeostatic potential and impaired TCR signaling and regulatory T cell function in Foxn1Delta/Delta mutant mice. J Immunol. 2007;179:8153–63. doi: 10.4049/jimmunol.179.12.8153. [DOI] [PubMed] [Google Scholar]
- 15.Chen L, Xiao S, Manley NR. Foxn1 is required to maintain the postnatal thymic microenvironment in a dosage-sensitive manner. Blood. 2009;113:567–74. doi: 10.1182/blood-2008-05-156265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nowell CS, et al. Foxn1 regulates lineage progression in cortical and medullary thymic epithelial cells but is dispensable for medullary sublineage divergence. PLoS Genet. 2011;7:e1002348. doi: 10.1371/journal.pgen.1002348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Choi EY, et al. Thymocyte-thymocyte interaction for efficient positive selection and maturation of CD4 T cells. Immunity. 2005;23:387–96. doi: 10.1016/j.immuni.2005.09.005. [DOI] [PubMed] [Google Scholar]
- 18.Li W, et al. An alternate pathway for CD4 T cell development: thymocyte- expressed MHC class II selects a distinct T cell population. Immunity. 2005;23:375–86. doi: 10.1016/j.immuni.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 19.Li W, et al. Thymic selection pathway regulates the effector function of CD4 T cells. J Exp Med. 2007;204:2145–57. doi: 10.1084/jem.20070321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li W, et al. Thymic selection pathway regulates the effector function of CD4 T cells. The Journal of experimental medicine. 2007;204:2145–57. doi: 10.1084/jem.20070321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ding C, Buckingham B, Levine MA. Familial isolated hypoparathyroidism caused by a mutation in the gene for the transcription factor GCMB. J Clin Invest. 2001;108:1215–20. doi: 10.1172/JCI13180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gordon J, et al. Specific expression of lacZ and cre recombinase in fetal thymic epithelial cells by multiplex gene targeting at the Foxn1 locus. BMC Dev Biol. 2007;7:69. doi: 10.1186/1471-213X-7-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gunther T, et al. Genetic ablation of parathyroid glands reveals another source of parathyroid hormone. Nature. 2000;406:199–203. doi: 10.1038/35018111. [DOI] [PubMed] [Google Scholar]
- 24.Chen SK, et al. Hematopoietic origin of pathological grooming in Hoxb8 mutant mice. Cell. 2010;141:775–85. doi: 10.1016/j.cell.2010.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.King P, Paul A, Laufer E. Shh signaling regulates adrenocortical development and identifies progenitors of steroidogenic lineages. Proc Natl Acad Sci U S A. 2009;106:21185–90. doi: 10.1073/pnas.0909471106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meir T, et al. Deletion of the vitamin D receptor specifically in the parathyroid demonstrates a limited role for the receptor in parathyroid physiology. Am J Physiol Renal Physiol. 2009;297:F1192–8. doi: 10.1152/ajprenal.00360.2009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.