Abstract
Free human kallikrein 2 (free-hK2) and hK2 pro-form (pro-hK2) have been found to be increased in tumor tissues and serum from patients with prostate cancer. We established semiautomatic assays for free-hK2 and pro-hK2 using a research version of the Beckman Coulter ACCESS2 system. Serum samples from a cohort of 189 men undergoing radical prostatectomy for known high-risk disease were assayed for Prostate-Specific Antigen (PSA), free-PSA, free-hK2, and pro-hK2. Univariate Cox regression and multivariate models were used to predict both Gleason scores and progression-free survival (PFS). Free-hk2 levels ≥80 ng/L were predictive of both Gleason scores ≥7 (p = 0.04) and PFS (p = 0.03). PSA ≥8.0 µg/L also was predictive of PFS (p = 0.02). However, neither % free-PSA nor pro-hK2, when treated as continuous or cutoff variables were associated with Gleason score or PFS. Multivariable models showed that clinical stage T1c versus T2/T3, Gleason score ≥7, and PSA ≥8.0 µg/L or clinical stage T1c versus T2/T3, Gleason scores ≥7, and free-hK2 ≥80 ng/L were among the best models predicting PFS. Both free-hK2 and PSA in conjunction with clinical stage and Gleason score are good predictors of PFS in prostate cancer.
Keywords: Free-hK2, pro-hK2, PSA, prostate cancer, progression-free survival
Introduction
Prostate cancer has been detected earlier and in most cases at a treatable stage following the introduction of PSA as a noninvasive diagnostic test.1 However, prostate cancer is still the second leading cause of cancer death in men in the United States.2 Risk group and staging, based on the Gleason score, PSA levels at biopsy, and other tests are the main factors predictive of prognosis in men diagnosed with prostate cancer. During recent years, there has been active investigation for new markers to complement the PSA screening that would better differentiate high-risk cancers and identify those patients who are less likely to respond to conventional treatment and who could most benefit from multimodal or alternative therapies. Several tissue-based gene signatures have been developed to predict biochemical relapse or metastasis after radical prostatectomy (RP). Decipher® (GenomeDx Biosciences, Vancouver, Canada) is a 22 gene-tissue expression assay, which can be used to predict the 5-year risk of metastatic prostate cancer after RP. Other reverse transcription polymerase chain reaction (RT-PCR) tissue-based gene signature assays for prediction of recurrence are Myriad Genetics Prolaris Score, Oncotype DX® Genomic Prostate Score™ (GPS), and NF-kappa B Activated Recurrence Predictor 21 (NARP21).3 Several proteins and a new generation of markers in blood and urine, such as micro-RNA, circulating DNA, and circulating tumor cells, are under investigation for prostate cancer diagnosis and prognosis.3
hK2, a member of the kallikrein family of proteins, has been shown to be a predictive marker for prostate cancer progression, in some studies more so than serum PSA.4–6 Several hK2 forms are detected in serum, including free-hK2 and hK2-ACT.4–5,7 Free-hK2 is comprised of pro-hK2 and mature free-hK2, which are both increased in prostate cancer.4,6 Free-hK2 is associated with a higher Gleason score.4 An increase in free-hK2 after radiation therapy can predict recurrence.8 In this study, we developed a sensitive semiautomatic immunoassay to specifically detect pro-hK2 in human serum and verified the analytical performance of a published immunoassay for free-hK2.4 We evaluated the role of these markers for predicting prostate cancer progression using a cohort of 189 men with high-risk prostate cancer.
Material and methods
Analytic assays
Measurement of free-PSA and PSA
Free-PSA and PSA were measured by Hybritech PSA and free-PSA immunoassays (Beckman Coulter, Brea, California, USA, Access2).
Free-hK2 immunoassay
Free-hK2 was measured by a two-step sequential immunoassay using a research version of the ACCESS2 immunoassay system (Beckman Coulter) as previously described with small modifications.4 In the first step, 75 µL of serum reacted with 150 ng of biotinylated H499 antibody and 50 µL of 0.6 mg/mL of paramagnetic particles at 37°C for 30 min. After washing, the paramagnetic beads were incubated with alkaline phosphatase labelled H599 antibody for 30 min. Six calibrators (0, 5, 15, 30, 300, and 3000 ng/L) were made by dilution of recombinant mature hK2 into PSA zero diluent (Beckman Coulter). Cross-reactivity with PSA and Prostate Specific Antigen-Antichymotrypsin (PSA-ACT) is negligible; cross-reactivity with hK2-ACT is 2%.
Pro-hK2 assay development
Recombinant pro-hK2 A217V (Hybritech, San Diego, California, USA) was immunopurified from the condition medium of recombinant cell lines using HK1G586.1 immunoaffinity column (which recognizes pro-hK2 and mature hK2) as previously described.9 The concentration of purified pro-hK2 A217V was determined by absorbance at 280 nm.10 Six calibrators (0, 8, 31, 125, 500, and 2000 ng/L) were made by addition of recombinant pro-hK2 A217V into human serum albumin (HSA) matrix (0.1 M Hepes, 0.15 M sodium chloride, 7% HSA (Sigma A3782), 0.3% Plurafac A-38 (BASF), 0.1% sodium azide). Diluted standards were aliquoted and stored at −80°C.
Pro-hK2 was measured by an immunoassay similar to free-hK2 with two main modifications. In order to increase the sensitivity, the biotinylated capture antibody and alkaline phosphatase detection antibody were incubated off-line with serum for 18–20 h. To minimize interferences due to human anti-mouse and heterophilic antibodies in the serum, a preincubation with blockers was added to the process. Two hundred microliters of serum or standards were incubated with 75 µL of blocking solution containing heat-inactivated mouse serum (Hybritech), HBR-1, HBR-6, HBR-23 (Scantibodies laboratories, Santee, California, USA), and MAB-33-IgG polymer (Roche Diagnostics Corp, Indianapolis, Indiana, USA) for 1 h at room temperature (RT) with rotation. Biotinylated BT464.3 anti-pro-hK2 antibody and alkaline phosphatase HK1H 599.1 antibody were added and incubated with constant rotation at RT for 18–22 h. A one-step format on the Beckman Coulter ACCESS 2 system was used to capture and measure the antigen–antibody complexes; 200 µL of serum–blockers–antibodies mix was incubated with 50 µL of paramagnetic-streptavidin particles at 37°C for 20 min. The complex was washed and developed with substrate (dioxetane phosphate), which produced a chemiluminescent signal that was quantified in the research version of ACCESS 2 (Beckman Coulter).
A grade 2 polynomial curve math model was used for standardization. The amount of free-hK2 or pro-hK2 in the sample was determined from the signal/background ratios (measured relative light units (RLU)/RLUs of zero standard) by means of the calibration curve. The 2000 ng/L was excluded from the standardization curve when pro-hK2 values in the serum samples were lower than 500 ng/L.
Pro-hK2 assay and free-hK2 assay analytical performance
The detection limit was determined by assaying 20 replicates of HSA matrix zero diluent and 20 replicates of 4, 6, and 8 ng/L calibrators of pro-hK2, or 20 replicates of PSA zero diluent and 20 replicates of 2, 3, and 4 ng/L of hK2. The limit of detection was taking 2.5 standard deviation (SD) of 20 replicates of the zero calibrator (in RLUs) and using lineal regression with the zero and pro-hK2 or hK2 calibrators to calculate the limit of detection in nanograms per liter. The limit of quantification (LOQ) was the minimum concentration measured with imprecision <15%.
The within run imprecision was calculated measuring 20 replicates of three different levels of serum pools within a run. The between run imprecision (Coefficient of variation [CV] = 100 × SD/mean) was assessed by measuring three serum pools in 20 independent analytical runs over approximately 2 months. We assessed linearity after dilution by serially diluting patient serums in standard diluent.
Cross-reactivity of relevant analytes was assessed by measuring PSA, PSA-ACT, hK2, and hK2-ACT diluted in HSA matrix zero diluent. The concentrations tested by pro-hK2 assay were as follows: PSA, and PSA-ACT, 150 µg/L; hK2, 9 ng/L; and hK2-ACT, 10 µg/L.
To study the stability of pro-hK2 nine-patient samples kept at −80°C were defrosted and assayed within 4.5 h (time zero). We compared the initial values with readings of serum stored at RT for 1 day and at 40°C for 1 and 7 days. For reassessment of free-hK2 immunoassay, we compared the analytic performance characteristics with those previously published.4 We also compared values with 100 serum samples previously measured for free-hK2 in 2010, and the correlation between both measurements was evaluated using the Spearman test.
Human studies
We used de-identified male and female serum for validation studies. One hundred twelve serum samples from men with negative biopsies for prostate cancer were used to evaluate the relation between pro-hK2 and age.
We measured markers in a cohort consisted of 189 men who underwent open or robotic RP for prostate cancer at the Mayo Clinic from 2004 to 2008 and were considered high risk for recurrence. PSA and free-PSA were previously measured in all samples, and free-hK2 was previously measured in approximately half of the samples. Free-hK2 and pro-hK2 were measured in all available samples (172 samples) in 2015 with prior measurements used for six patients who did not have adequate residual sample. High risk was defined as a PSA >20 µg/L, a Gleason score of 8 or higher, seminal vesicle invasion (SVI), or a GPS score of ≥10 at RP. All analyses were limited to the 178 men who had PSA, % free-PSA, and free-hK2 data to allow for a matched comparison of PSA and hK2 data.
These studies were approved by the Mayo Clinic Institutional Review board.
Statistical methods
The primary objective was to evaluate whether free-hk2 and pro-hk2 are predictive of time to progression in high-risk prostate cancer patients undergoing RP. The primary endpoint was progression-free survival (PFS). Progression was defined as the occurrence of PSA progression as defined by American Urology Association/American Society for Radiation Oncology (AUA/ASTRO) guidelines,11 local recurrence, systemic progression or death. Local recurrence or systemic progression was defined as demonstrable local or distant metastasis on imaging studies or via biopsy. Time to progression is defined as the time to first occurrence of PSA progression, local or regional progression or death. Patients without an event were censored at the time of last contact. Cox proportional hazard models were used to assess free-hK2, pro-hK2, PSA, % free-PSA, age, Gleason score, GPS, SVI, and T stage to predict PFS.
Due to the skewness of the laboratory markers, the logarithmic values of these markers were determined and used in the Cox proportional hazard modeling. In addition to assessing the association of the lab markers by taking the logarithmic value, dichotomous cut points were also investigated by first splitting each continuous covariant into quartiles and choosing the cut point that best maximized the difference between the progression and nonprogression groups. In addition, a few alternate cut points were explored. For the multivariate analysis, only factors thought to be clinically relevant or with a p value of ≤0.2 in univariate analysis were considered as candidates in the multivariate model.
A secondary objective was to evaluate the association of the free-hK2 and pro-hK2 with Gleason score. Gleason score was dichotomized into two groups (<7 vs. ≥7). Wilcoxon rank sum and χ 2 tests were used to assess the association of free-hK2, pro-hK2, PSA, and % free-PSA with Gleason score. Various cut points of the laboratory markers were also assessed using above mentioned methods.
All tests were two-sided, with p values ≤0.05 considered statistically significant. Correlation between assays was evaluated using Spearman correlation test. Statistical analyses were performed by using SAS v9.3 (SAS Institute, Cary, North Carolina, USA).
Results
hK2 and pro-hK2 immunoassay analytical performance
We reanalyzed the analytical performance of the automated assay for free-hK2 and developed a semiautomatic assay for pro-hK2 using a research version of the Beckman Coulter ACCESS2 system.
Table 1 summarizes the analytical performance of free-hK2 and pro-hK2 assays.
Table 1.
Assay performance.
| Performance | Level | Free-hK2, ng/L | Pro-hK2, ng/L |
|---|---|---|---|
| Within run imprecision | Low | Mean = 18 | Mean = 26 |
| CV = 9.5% | CV = 3.1% | ||
| Medium | Mean = 41 | Mean = 82 | |
| CV = 2.9% | CV = 2.3% | ||
| High | Mean = 236 | Mean = 521 | |
| CV = 3.2% | CV = 2.2% | ||
| Between run imprecision | Low | Mean = 17 | Mean = 18 |
| CV = 8.8% | CV = 11.6% | ||
| Medium | Mean = 41 | Mean = 91 | |
| CV = 4.6% | CV = 4.1% | ||
| High | Mean = 226 | Mean = 524 | |
| CV = 7.2% | CV = 7.3% | ||
| Limit of detection | LOD | 2.1 | 3.5 |
| Limit of quantitation | LOQ | 4.0 | 8.0 |
hK2: human kallikrein 2.
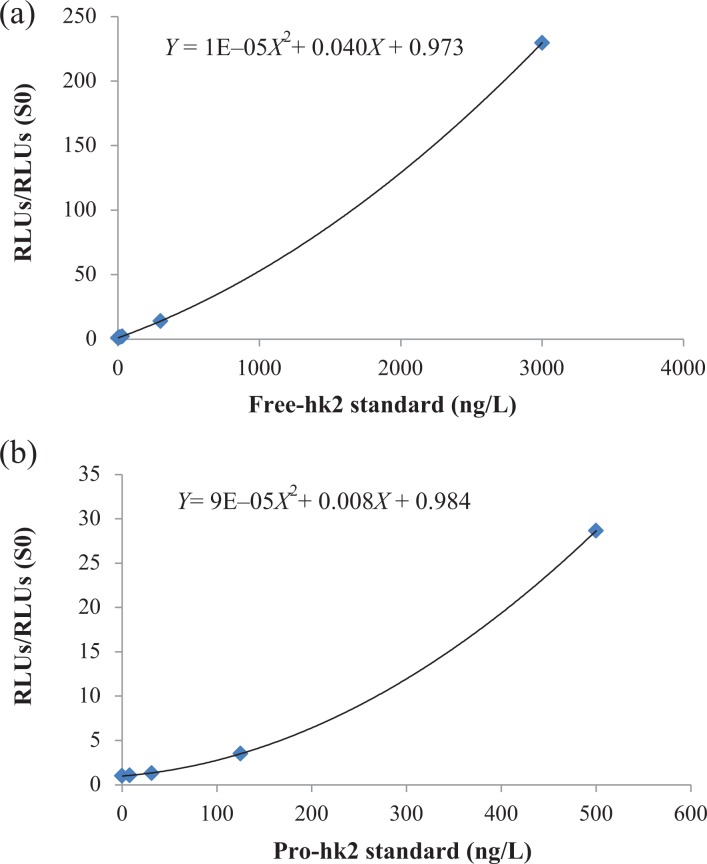
Because the concentration of pro-hK2 in circulation is very low, an immunoassay to measure it must have very low levels of detection and be highly reproducible. We developed a very sensitive semiautomatic immunoassay to measure pro-hK2, with a LOQ five times lower than that reported in an immunoassay described previously.6 This increase in sensitivity was required to measure pro-hK2 in serum samples within the gray range of PSA between 2 and 10 µg/L where the results of the pro-hK2 assay could have clinical relevance. Twenty-eight percent of the samples in the range of PSA of 2–10 µg/L had concentrations of pro-hK2 that were lower than the LOQ of 8 ng/L compared to 85% of the samples that would be under a LOQ previously described of 40 ng/L.6 Standard curves for free-hK2 and pro-hK2 are shown in Figure 1. Serum from females and males with and without known prostate disease was used to validate the Pro-hK2 assay. Pro-hK2 was under the LOQ in the 14 female serum samples tested. The mean value for Pro-hK2 in male serums with unknown prostate cancer was 22 ± 21 ng/L. Six male samples were diluted with standard diluent (HSA matrix), and the dilutions were measured with pro-hK2 assay. The average recovery by dilution was 96%. Three serum samples were diluted with PSA zero diluent and measured with free-hK2 assay. The average recovery by dilution was 106%. Cross-reactivity of pro-hK2 assay with PSA, PSA-ACT, and hK2-ACT was under the limit of detection and cross-reactivity with recombinant hK2 was of 0.07% in weight basis indicative that cross-reactivity with potentially interfering proteins is negligible.
Figure 1.
Average calibration curves for the free-hK2 (a) and pro-hK2 (b) ACCESS 2 research assays. The assays are calibrated with recombinant purified hK2 or pro-hK2. The y-axis represents the ratio of the RLU at various levels to the RLU for the zero calibrator. The solid lines represent the grade 2 polynomial regression curves. hK2: human kallikrein 2; RLU: relative light units.
Stability studies showed that pro-hK2 is stable. The mean recovery for samples stored at RT for 24 h was 93 ± 6%, for samples stored at 4°C for 1 day was 97 ± 5%, and for samples kept at 4°C for 1 week was 105 ± 7%.
Free-hK2 assay equally detected recombinant hK2 and recombinant pro-hK2. Free-hK2 values correlated well with previous measurements performed 5 years earlier with an immunoassay previously validated9 (Spearman correlation coefficient = 0.92, p < 0.001).
There was no significant correlation between free-hK2 and age (Spearman coefficient = 0.16, p = 0.10) and a poor correlation between pro-hK2 and age (Spearman coefficient = 0.25, p = 0.007) for men without prostate cancer (but including men with benign prostatic hypertrophy; n = 111, age range = 50–85 years).
Cohort study for prediction of PFS
Clinical-pathologic features of the cohort study are as follows: the median age was 63 years (interquartile range (IQR): 57–68) and predominantly Caucasian (93%). Fifty-one patients (29%) had Gleason scores <7 and 76 (42%) patients had clinical T stage of T1c at initial diagnosis. The median PSA value was 8.6 (IQR: 5.8–14.3). Median follow-up for the entire cohort was 5.7 years (IQR: 2.1–7.8).
Free-hK2 and pro-hK2 correlated poorly with PSA. The Spearman correlation coefficient between free-hK2 and PSA was 0.24 and pro-hK2 and PSA was 0.30. The hK2 and phK2 spearman correlation coefficient was 0.63.
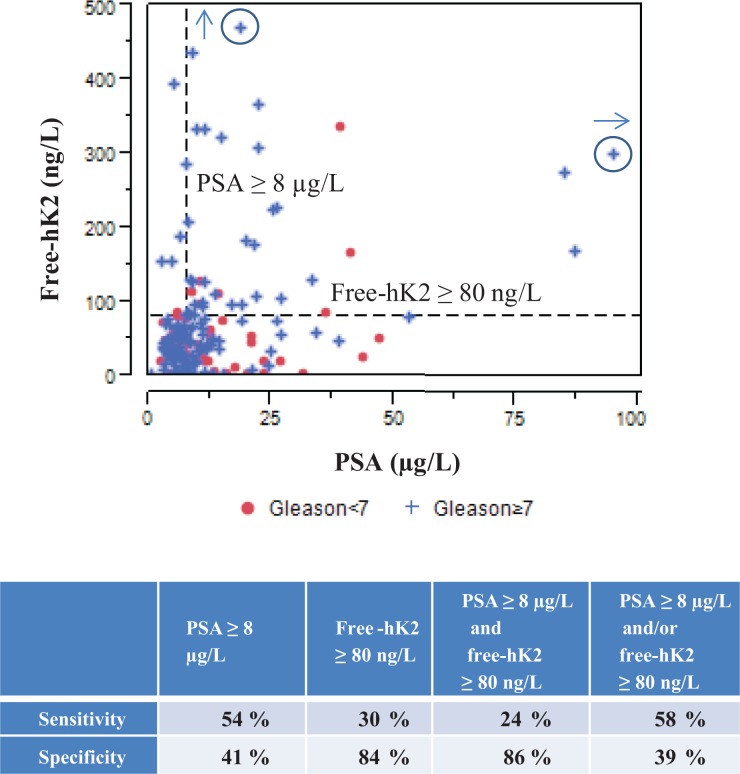
Table 2 summarizes the association of free-hK2, pro-hK2, PSA, and % free-PSA with Gleason score ≥7. Patients with free-hK2 ≥80 ng/L were more likely to have a high Gleason score (30% vs. 16%; p = 0.04). It is interesting to note that the observed median PSA was 10.4 µg/L in Gleason <7 group compared to 8.4 in the Gleason ≥7 group (not statistically different; p = 0.20). Figure 2 shows the categorical dot plot of free-hK2 versus PSA for Gleason scores (Gleason <7 and Gleason ≥7), the table underneath the graph summarizes the specificity and sensitivity for the prediction of Gleason score ≥7 by free-hK2 ≥80 ng/L and PSA ≥8 µg/L. Free-hK2 ≥80 is highly specific for prediction of high Gleason score (84%) compared to PSA ≥8 µg/L.
Table 2.
Association of hK2 and PSA with Gleason score.
| Gleason <7 (N = 51) | Gleason ≥7 (N = 126) | p Value | |
|---|---|---|---|
| PSA (µg/L) | 0.20 | ||
| N | 51 | 126 | |
| Median | 10.4 | 8.4 | |
| Q1, Q3 | 6.0, 17.7 | 5.8, 13.2 | |
| % Free-PSA | 0.76 | ||
| N | 51 | 126 | |
| Median | 11.7 | 11.2 | |
| Q1, Q3 | 7.8, 15.6 | 8.0, 14.9 | |
| Free-hK2 (ng/L) | 0.07 | ||
| N | 51 | 126 | |
| Median | 42.0 | 51.2 | |
| Q1, Q3 | 20.7, 65.1 | 28.0, 96.0 | |
| Pro-hK2 (ng/L) | 0.22 | ||
| N | 50 | 120 | |
| Median | 20.0 | 22.0 | |
| Q1, Q3 | 7.0, 34.0 | 9.5, 47.0 | |
| Free-hK2 ≥80 ng/L | 16% | 30% | 0.04 |
hK2: human kallikrein 2.
Figure 2.
Categorical dot plot of free-hK2 versus PSA discriminated by Gleason score <7 and Gleason score ≥7. The table underneath the graph shows the specificity and sensitivity for prediction of Gleason score ≥7 by free-hK2 ≥80 ng/L and PSA ≥8µg/L. The circled plus marks represent off-scale measurements. hK2: human kallikrein 2.
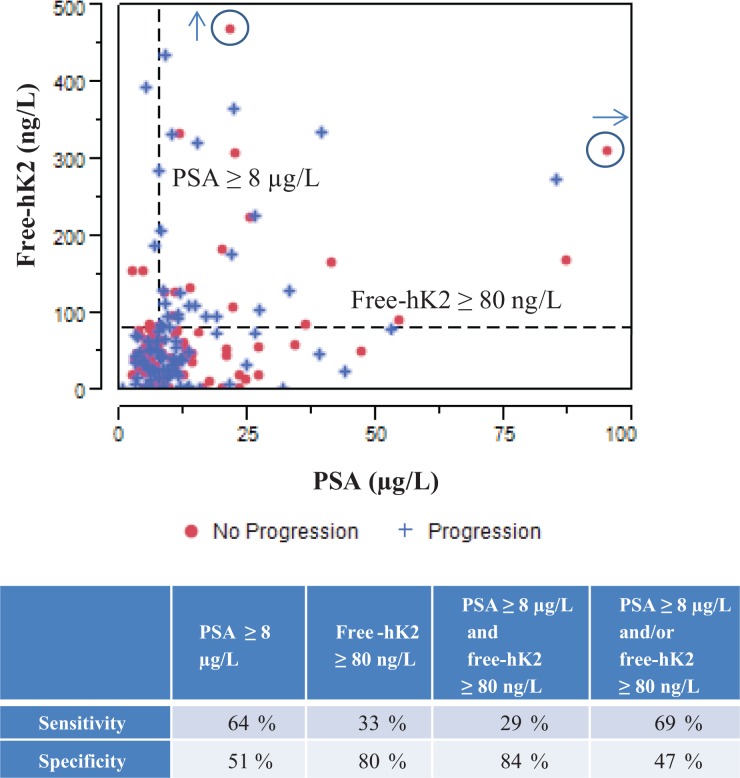
Overall univariate Cox regression analysis show that clinical stage (T1c vs. T2/T3, hazard ratio (HR) = 0.44; p ≤ 0.001), PSA ≥8 µg/L (HR = 1.72; p = 0.02), free-hK2 ≥80 ng/L (HR = 1.68; p = 0.03), GPSM (HR = 1.21; p = 0.002), Gleason score ≥7 (HR = 2.34; p = 0.003), and SVI (HR = 2.00; p = 0.002) were all significantly associated with PFS (Table 3). Figure 3 shows the categorical dot plot of free-hK2 vs. PSA for prostate cancer progression. The table underneath the graph summarizes the specificity and sensitivity for the prediction of prostate cancer progression by free-hK2 ≥80 ng/L and PSA ≥8 µg/L. Free-hK2 ≥80 has a higher specificity but a reduced sensitivity for the prediction of progression respect to PSA ≥8 µg/L. In the subgroup of patients with T1c disease, PSA ≥8 µg/L had elevated sensitivity and specificity of 80% and 59%, respectively.
Table 3.
Association of hK2 and PSA with PFS.
| N | Events | HR | 95% CI | p Value | |
|---|---|---|---|---|---|
| Age at surgery | 178 | 84 | 1.00 | (0.97, 1.03) | 0.81 |
| Clinical stage | |||||
| T1c | 76 | 25 | 0.44 | (0.27, 0.70) | <0.001 |
| T2 or T3 | 102 | 59 | |||
| Gleason score | |||||
| ≥7 | 125 | 69 | 2.34 | (1.34, 4.10) | 0.003 |
| <7 | 51 | 15 | |||
| SVI | |||||
| Yes | 55 | 34 | 2.00 | (1.29, 3.09) | 0.002 |
| No | 123 | 50 | |||
| GPSM | 178 | 84 | 1.21 | (1.07, 1.36) | 0.002 |
| PSA (log, µg/L) | 178 | 84 | 1.07 | (0.82, 1.40) | 0.60 |
| PSA (µg/L) | |||||
| ≥8.0 | 100 | 54 | 1.72 | (1.10, 2.68) | 0.02 |
| <8.0 | 78 | 30 | |||
| % Free-PSA (log) | 178 | 84 | 1.00 | (0.66, 1.49) | 0.98 |
| Free-hK2 (log, ng/L) | 178 | 84 | 1.10 | (0.89, 1.37) | 0.38 |
| Free-hK2 (ng/L) | |||||
| ≥80 | 47 | 28 | 1.68 | (1.07, 2.64) | 0.03 |
| <80 | 131 | 56 | |||
| Pro-hK2 (log, ng/L) | 171 | 81 | 1.04 | (0.86, 1.26) | 0.69 |
| Multivariate model with clinical stage, Gleason score, and PSA | |||||
| Clinical stage | |||||
| T1c | 76 | 25 | 0.53 | (0.32, 0.88) | 0.01 |
| T2 or T3 | 100 | 59 | |||
| Gleason score | |||||
| ≥7 | 125 | 69 | 1.90 | (1.05, 3.44) | 0.03 |
| <7 | 51 | 15 | |||
| PSA | |||||
| ≥8.0 | 98 | 54 | 1.72 | (1.10, 2.69) | 0.02 |
| <8.0 | 78 | 30 | |||
| Comparable multivariate model using free-hK2 instead of PSA with clinical stage and Gleason score | |||||
| Clinical stage | |||||
| T1c | 76 | 25 | 0.51 | (0.31, 0.83) | 0.006 |
| T2 or T3 | 100 | 59 | |||
| Gleason score | |||||
| ≥7 | 125 | 69 | 1.77 | (0.98, 3.18) | 0.06 |
| <7 | 51 | 15 | |||
| Free-hK2 (ng/L) | |||||
| ≥80 | 45 | 28 | 1.63 | (1.03, 2.57) | 0.04 |
| <80 | 131 | 56 | |||
hK2: human kallikrein 2; PFS: progression-free survival; HR: hazard ratio; CI: confidence interval; SVI: seminal vesicle invasion.
Figure 3.
Categorical dot plot of free-hK2 versus PSA discriminated by patients who did not have prostate cancer progression (no progression) and patients who progressed (progression). The table underneath the graph shows the specificity and sensitivity for prediction of prostate cancer progression by free-hK2 ≥80 ng/L and PSA ≥8 µg/L. The circled plus marks represent off-scale measurements. hK2: human kallikrein 2.
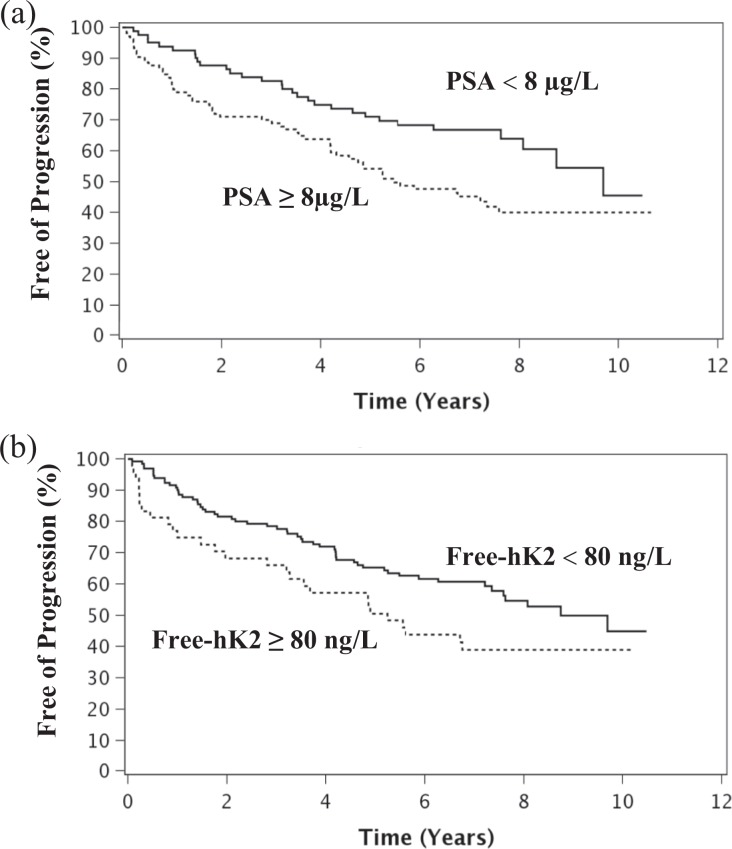
Kaplan–Meier curves of PFS by PSA ≥8 µg/L and free-hK2 ≥80 ng/L are shown in Figure 4(a) and (b), respectively. Table 3 displays the final multivariate model which includes clinical stage (T1c vs. T2/T3, HR = 0.53; p = 0.01), Gleason score ≥7 (HR = 1.90; p = 0.03), and PSA ≥8.0 µg/L (HR = 1.72; p = 0.02). Adding the free-hK2 ≥80 ng/L covariate to this model was not statistically significant (p = 0.82). However, the overall significance of the three variable Cox model with clinical stage, Gleason score, and hK2 ≥80 ng/L was nearly identical to clinical stage, Gleason score with PSA model (−2 log likelihood ratio test p value < 0.001 for both models).
Figure 4.
(a) Kaplan–Meier curves of PFS by PSA (PSA ≥8 µg/L represented by the dotted line and PSA <8 µg/L by the solid line). Of 100 total patients with PSA ≥8 µg/L, 54 progressed; and of 78 patients with PSA <8 µg/L, 30 progressed. Patients with PSA ≥8 µg/L had shorter PFS time (log-rank p value = 0.01). (b) Kaplan–Meier curves of PFS by free-hK2, with free-hK2 ≥80 ng/L (dotted line) and free-hK2 <80 (solid line). Of 47 total patients with free-hK2 ≥80 ng/L, 28 progressed; and of 131 patients with Free-hK2 <8 µg/L, 56 progressed. Patients with free-hK2 ≥80 ng/L had shorter PFS time (log-rank p value = 0.03). PFS: progression-free survival; hK2: human kallikrein 2.
Clinical stage (T1c vs. T2/T3, HR = 0.32; p = 0.001), PSA ≥8 µg/L (HR = 2.51; p = 0.003), and hK2 ≥80 ng/L (HR = 2.38; p = 0.01) were all significantly associated with PFS for patients with baseline PSA <10 µg/L. The final multivariate model included clinical stage (T1c vs. T2/T3, HR = 0.36; p = 0.004) and PSA ≥8.0 (HR = 2.08; p = 0.02). As occurred with the whole cohort Cox modeling, adding the free-hK2 ≥80 ng/L covariate to this model was not statistically significant (p = 0.09). However, the overall significance of the two variables Cox model with clinical stage and hK2 ≥80 ng/L was almost identical to clinical stage with PSA model (−2 log likelihood ratio test p value <0.001 for both models).
Discussion
Improved blood and urine markers used alone or in combination that better differentiate more aggressive cancers and predict progression and response to therapy are needed. High rates of recurrence in certain subsets of patients (almost 50% in this retrospective cohort study) suggest the need for more intensive and multimodal therapies. Markers that add predictive value for relapse after primary prostate cancer therapy may help select patients for clinical trials. In this study, we evaluated whether free-hK2 and pro-hK2 in serum could add information for clinical decision-making for prostate cancer after RP in a group of high-risk prostate cancer patients.
It was previously shown that the correlation between free-hK2 and PSA is low.4 We also found that in our cohort study, both free-hK2 and pro-hK2 poorly correlate with PSA, suggesting that free-hK2 and pro-hK2 might have independent clinical value and complement the information obtained by PSA.
Immunoassay methods to measure kallikreins in serum could lead to major differences in the results due to antibody specificity and epitope recognition, matrix effect, and complex formation with other molecules. Traceable methods to measure PSA and hK2 using mass spectroscopy could help better improve and standardized immunoassays to measure markers in biological samples.12
Neither pro-hK2 nor % free-PSA predicted progression. The lack of association between pro-hK2 and PFS could be due to a relative large proportion of samples under the limit of detection or with values in the lower end where the variability of the assay is higher and there is a potential for influence of interferences. Also, we cannot rule out some degradation or auto processing of pro-hK2 due to storage of frozen serum samples at −80°C.
Univariate Cox regression analysis shows high association of clinical stage and Gleason score with relapse. We also found that both PSA ≥8 µg/L and free-hK2 ≥80 ng/L significantly predicted recurrence. Free-hK2 showed to be more specific but less sensitive than PSA to predict prostate cancer progression. Although free-hK2 was not an independent covariate in multivariate Cox regression models, the significance of a three variable Cox model with clinical stage, Gleason score, and free-hK2 ≥80 ng/L was almost identical to a model of clinical stage, Gleason score with PSA ≥8 µg/L. Thus, either PSA or free-hK2 in conjunction with Gleason score and clinical stage could predict prostate cancer progression. Multivariable Cox models including PSA and clinical stage or free-hK2 and clinical stage also show good prediction of PFS in samples with PSA <10 ng/ml. Prospective studies should be performed to further validate the utility of free-hK2 as a predictive marker. Additionally, whether an increase in free-hK2 after prostatectomy better predicts relapse than preoperative free-hK2 should also be evaluated. Patients with free-hK2 ≥80 ng/L were more likely to have Gleason score higher or equal to 7. None of the other variables evaluated showed statistically significant association with high Gleason score. These results suggest that free-hK2 in serum could be a good marker for high-risk prostate tumors. It has been shown that the prostate cancer index is associated with Gleason score ≥7 and early relapse.13–17 Additionally, preoperative plasma transforming growth factor beta-1 (TGF-β1) and serum interleukin (IL)-6 and sIL-6R have been associated with poor clinical outcome and disease progression.18 Whether free-hK2 adds to the prediction value of these markers to discriminate higher risk tumors and recurrence should be evaluated in future studies. Free-hK2 in combination with other circulating markers together with sensitive imaging methods may increase the possibilities for curative success and reduce overtreatment of prostate cancer patients.
Footnotes
Declaration of conflicting interests: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was conducted in compliance with Mayo Clinic conflict of interest policies. Mayo Clinic and Dr Klee have received royalties of less than the federal threshold for significant financial interest from the licensing of technology to Beckman Coulter, Inc.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this study was provided by the Mayo Clinic Prostate SPORE grant NIH/NCI CA091956 and Beckman Coulter, Inc. to Dr Klee.
References
- 1. Schroder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet 2014; 384: 2027–2035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Siegel RL, Miller KD,, Jemal A. Cancer statistics, 2015. CA Cancer J Clin 2015; 65: 5–29. [DOI] [PubMed] [Google Scholar]
- 3. Locke JA, Black PC. Next generation biomarkers in prostate cancer. Front Biosci (Landmark Ed) 2016; 21: 328–342. [DOI] [PubMed] [Google Scholar]
- 4. Grauer LS, Finlay JA, Mikolajczyk SD, et al. Detection of human glandular kallikrein, hK2, as its precursor form and in complex with protease inhibitors in prostate carcinoma serum. J Androl 1998; 19: 407–411. [PubMed] [Google Scholar]
- 5. Klee GG, Goodmanson MK, Jacobsen SJ, et al. Highly sensitive automated chemiluminometric assay for measuring free human glandular kallikrein-2. Clin Chem 1999; 45: 800–806. [PubMed] [Google Scholar]
- 6. Vaisanen V, Eriksson S, Ivaska KK, et al. Development of sensitive immunoassays for free and total human glandular kallikrein 2. Clin Chem 2004; 50: 1607–1617. [DOI] [PubMed] [Google Scholar]
- 7. Saedi MS, Hill TM, Kuus-Reichel K, et al. The precursor form of the human kallikrein 2, a kallikrein homologous to prostate-specific antigen, is present in human sera and is increased in prostate cancer and benign prostatic hyperplasia. Clin Chem 1998; 44: 2115–2119. [PubMed] [Google Scholar]
- 8. Spratt DE, Evans MJ, Davis BJ, et al. Androgen receptor upregulation mediates radioresistance after ionizing radiation. Cancer Res 2015; 75: 4688–4696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kumar A, Goel AS, Hill TM, et al. Expression of human glandular kallikrein, hK2, in mammalian cells. Cancer Res 1996; 56: 5397–5402. [PubMed] [Google Scholar]
- 10. Mikolajczyk SD, Millar LS, Marker KM, et al. Ala217 is important for the catalytic function and autoactivation of prostate-specific human kallikrein 2. Eur J Biochem 1997; 246: 440–446. [DOI] [PubMed] [Google Scholar]
- 11. Valicenti RK, Thompson I, Jr, Albertsen P, et al. Adjuvant and salvage radiation therapy after prostatectomy: American Society for Radiation Oncology/American Urological Association Guidelines. Int J Radiat Oncol Biol Phys 2013; 86: 822–828. [DOI] [PubMed] [Google Scholar]
- 12. Klee EW, Bondar OP, Goodmanson MK, et al. Serum concentrations of prostate-specific antigen measured using immune extraction, trypsin digestion, and tandem mass spectrometry quantification of LSEPAELTDAVK peptide. Arch Pathol Lab Med 2014; 138: 1381–1386. [DOI] [PubMed] [Google Scholar]
- 13. Jansen FH, van Schaik RH, Kurstjens J, et al. Prostate-specific antigen (PSA) isoform p2PSA in combination with total PSA and free PSA improves diagnostic accuracy in prostate cancer detection. Eur Urol 2010; 57: 921–927. [DOI] [PubMed] [Google Scholar]
- 14. Catalona WJ, Partin AW, Sanda MG, et al. A multicenter study of [-2]pro-prostate specific antigen combined with prostate specific antigen and free prostate specific antigen for prostate cancer detection in the 2.0 to 10.0 ng/ml prostate specific antigen range. J Urol 2011; 185: 1650–1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Fossati N, Buffi NM, Haese A, et al. Preoperative prostate-specific antigen isoform p2PSA and its derivatives, %p2PSA and prostate health index, predict pathologic outcomes in patients undergoing radical prostatectomy for prostate cancer: results from a multicentric European prospective study. Eur Urol 2015; 68: 132–138. [DOI] [PubMed] [Google Scholar]
- 16. de la Calle C, Patil D, Wei JT, et al. Multicenter evaluation of the prostate health index to detect aggressive prostate cancer in biopsy naive men. J Urol 2015; 194: 65–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lughezzani G, Lazzeri M, Buffi NM, et al. Preoperative prostate health index is an independent predictor of early biochemical recurrence after radical prostatectomy: results from a prospective single-center study. Urol Oncol 2015; 33: 337:e7–e14. [DOI] [PubMed] [Google Scholar]
- 18. Shariat SF, Kattan MW, Traxel E, et al. Association of pre- and postoperative plasma levels of transforming growth factor beta(1) and interleukin 6 and its soluble receptor with prostate cancer progression. Clin Cancer Res 2004; 10: 1992–1999. [DOI] [PubMed] [Google Scholar]