Abstract
The microbiome can promote or disrupt human health by influencing both adaptive and innate immune functions. We tested whether bacteria that normally reside on human skin participate in host defense by killing Staphylococcus aureus, a pathogen commonly found in patients with atopic dermatitis (AD) and an important factor that exacerbates this disease. High-throughput screening for antimicrobial activity against S.aureus was performed on isolates of coagulase-negative Staphylococcus (CoNS) collected from the skin of healthy and AD subjects. CoNS strains with antimicrobial activity were common on the normal population but rare on AD subjects. A low frequency of strains with antimicrobial activity correlated with colonization by S.aureus. The antimicrobial activity was identified as previously unknown antimicrobial peptides (AMPs) produced by CoNS species including Staphylococcus epidermidis and Staphylococcus hominis. These AMPs were strain-specific, highly potent, selectively killed S.aureus, and synergized with the human AMP LL-37. Application of these CoNS strains to mice confirmed their defense function in vivo relative to application of nonactive strains. Strikingly, reintroduction of antimicrobial CoNS strains to human subjects with AD decreased colonization by S.aureus. These findings show how commensal skin bacteria protect against pathogens and demonstrate how dysbiosis of the skin microbiome can lead to disease.
Introduction
Antimicrobial peptides (AMPs) are essential immune defense molecules produced by neutrophils, paneth cells, mast cells, epithelial cells, and adipocytes (1–4). Because of their direct antimicrobial action, AMPs control growth of microorganisms that normally reside on epithelial surfaces, a community of microbes referred to as the “microbiome” (5). The skin microbiome is also regulated by other ecological factors such as humidity, temperature, pH, and lipid content (6). As a consequence of the environment established by the host, only a restricted collection of microbes can normally colonize the epithelial surface. However, in some disease states, an altered balance of the microbiota can occur, a condition known as dysbiosis. This state of dysbiosis may contribute to the disruption of immune homeostasis and increase disease symptoms. For example, atopic dermatitis (AD) is a disease characterized by dry, itchy, and inflamed skin that is frequently subject to infections of the skin and has been clearly shown to have an altered skin bacterial flora when compared to non-AD subjects (7, 8). Although it is not clear why this occurs, the skin of subjects with AD has a decreased capacity to express certain AMPs such as cathelicidins and β-defensins despite the presence of skin inflammation (9–13). This relative deficiency in AMPs has been proposed to result in an inability of the skin to resist growth of pathogens such as Staphylococcus aureus.
The high abundance of S.aureus in AD contributes to the pathophysiology of this disease and has been linked with immune dysfunction including T helper cell 2 lymphocyte skewing (14), reduced AMPs (12, 15), exacerbated allergic reactions (16, 17), and disruption of the skin barrier (18). Mice develop AD-like lesions when they are experimentally colonized by S.aureus (19, 20). However, in contrast to the exacerbation of disease and inflammation caused by S.aureus, other bacterial species found on normal skin appear to aid in normal immune homeostasis (21). For example, Staphylococcus epidermidis, a prevalent species of bacteria on healthy human skin, can diminish inflammation after injury (22), enhance development of cutaneous T cells (23–25), and promote expression of AMPs (23, 24, 26–28). Furthermore, some bacteria have been shown to produce their own AMPs that can synergize with AMPs produced by mammalian cells (29, 30).
Here, we hypothesized that normal human skin is populated by commensal bacteria that promote health by producing antimicrobial activity. Furthermore, we asked whether a deficiency of this community in AD might exacerbate disease by facilitating the survival of S.aureus. We report a discovery of a broad community of protective bacteria on normal human skin that provides host defense against pathogens. These observations offer a specific strategy for rational microbiome therapy.
Results
Bacteria on normal human skin inhibit S.aureus but are infrequent in AD
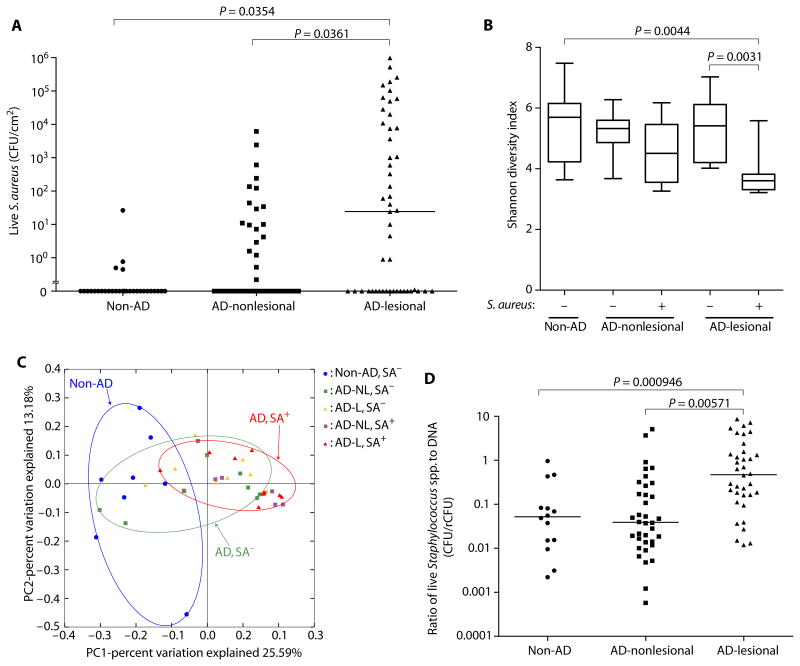
We performed 16S rRNA gene sequencing and culture-based analysis of skin swabs taken from healthy non-AD subjects and the lesional and nonlesional sites of subjects with AD (refer to table S1 for demographic data). A large proportion of subjects with AD were identified to be culture–positive for S.aureus on both their lesional and nonlesional skin (Fig. 1A). S.aureus culture–positive lesional skin had decreased bacterial diversity when compared to lesional skin that was S.aureus culture–negative (Fig. 1B and fig. S1). Principal coordinate analysis using weighted UniFrac distances suggested a similarity in the bacterial communities of non-AD controls and S.aureus–positive and S.aureus–negative AD subjects (Fig. 1C). The absolute abundance of total staphylococcal species on skin swabs was also evaluated by manual colony counting for colony-forming units (CFUs) on a selective egg yolk mannitol salt agar (fig. S2A) and by quantitative polymerase chain reaction (qPCR) of bacterial DNA with genus-specific 16S primers to yield a relative CFU (rCFU) based on DNA abundance (fig. S2B). Swabs to quantify live bacteria and bacterial DNA were simultaneously obtained from the area next to each other. The specificity of the Staphylococcus-specific primers was confirmed as shown in fig. S3. Both colony counting and 16S rDNA qPCR methods gave similar results from samples from AD lesional skin. However, these methods yielded different results on samples from normal skin where qPCR of DNA abundance detected about 10-fold higher rCFUs than live colony counting (Fig. 1D). One explanation for the larger amount of DNA recovered from normal skin compared to AD skin was that DNA recovered from normal skin was associated with bacteria that had been killed by the normal antimicrobial defense systems on the skin surface environment. This observation led us to hypothesize that bacteria residing on the skin of AD subjects had less antimicrobial activity than those on normal skin.
Fig. 1. Dysbiosis of the skin microbiome in AD is associated with S.aureus colonization.
(A) Live S.aureus CFUs on skin of non-AD subjects and on nonlesional and lesional skin of subjects with AD. Bar, median. (B) Shannon diversity index of bacterial community on skin from non-AD and S.aureus culture–positive or S.aureus culture– negative subjects with AD. Data are shown as box and whisker plots. (C) Principal coordinate analysis plot analysis displaying composition of bacterial communities on non-AD and S.aureus culture–positive or S.aureus culture–negative subjects with AD. SA+, S.aureus culture–positive; SA−, S.aureus culture–negative; L, lesional; NL, nonlesional. (D) Ratio of Staphylococcus spp. CFU abundance determined by live colony counting compared to rCFU of Staphylococcus determined by qPCR with species-specific primers. Bar, median. P values were calculated by two-tailed paired t test for lesional versus nonlesional samples or two-tailed independent t test for non-AD versus AD groups.
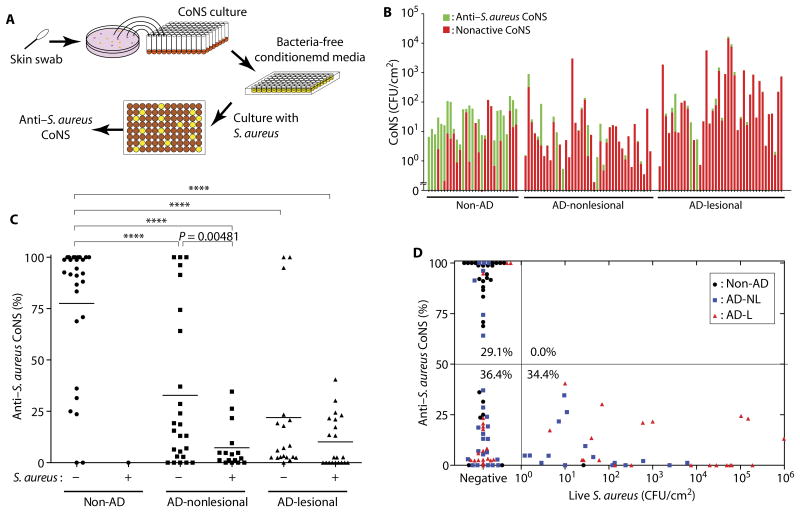
Previous studies have found that laboratory isolates of coagulase-negative Staphylococcus (CoNS) species can produce AMPs (29, 30). We therefore next sought to determine the frequency of CoNS strains that produce antimicrobial activity on normal skin. To detect antimicrobial activity, we randomly isolated about 80 individual CoNS isolates from each skin culture swab and then conducted an unbiased analysis of antimicrobial activity by measuring the capacity of the sterile conditioned supernatant of each individual isolate to inhibit S.aureus growth (Fig. 2A). A total of 2029 colonies were studied from the forearms of 30 healthy subjects, and 5695 colonies were studied from nonlesional and lesional skin of 49 subjects with AD. This functional screening revealed that most CoNS clones isolated from healthy subjects could inhibit S.aureus growth, but CoNS clones with antimicrobial activity were much less frequent on AD subjects and were lowest on individuals who were S.aureus culture–positive (Fig. 2, B and C). Low frequency of CoNS with antimicrobial activity was also seen in the only healthy subject that was S.aureus culture–positive. The measurement of frequency of clones with anti–S.aureus activity was stable in multiple skin swab samples from the same subjects over 14 days (fig. S4). No subject that was culture-positive for S.aureus had antimicrobial activity detected in more than 50% of their CoNS isolates (Fig. 2D). This suggested that a deficiency in the antimicrobial function of the CoNS bacterial community was strongly associated with colonization by S.aureus.
Fig. 2. S.aureus colonization correlates with a lack of antimicrobial activity in CoNS.
(A) Schematic of a high-throughput antimicrobial screening of CoNS against S.aureus.(B) CFU of CoNS without (red) or with antimicrobial activity against S.aureus (green). Each bar represents data from individual subjects. (C) Frequency of CoNS colonies with activity against S.aureus. Data are reported for 29 healthy subjects and 41 nonlesional or 40 lesional sites from AD subjects. Subjects that had less than 25 CoNS colonies per swab were not included. Each point represents the frequency that anti–S.aureus activity was detected in all colonies isolated from each individual. Bar, mean. ****P < 0.0001. P values were calculated by two-tailed independent t test. (D) Correlation between proportion of CoNS strains that have the capacity to inhibit S.aureus growth and abundance of live S.aureus CFU on the skin surface. Each dot represents data from one individual. Quadrants are divided on the basis of the frequency of antimicrobial CoNS (>50 or <50%) and detection of live S.aureus (<1or >1CFU/cm2). The proportion (%) of subjects in each quadrant to total subjects is shown. L, lesional; NL, nonlesional.
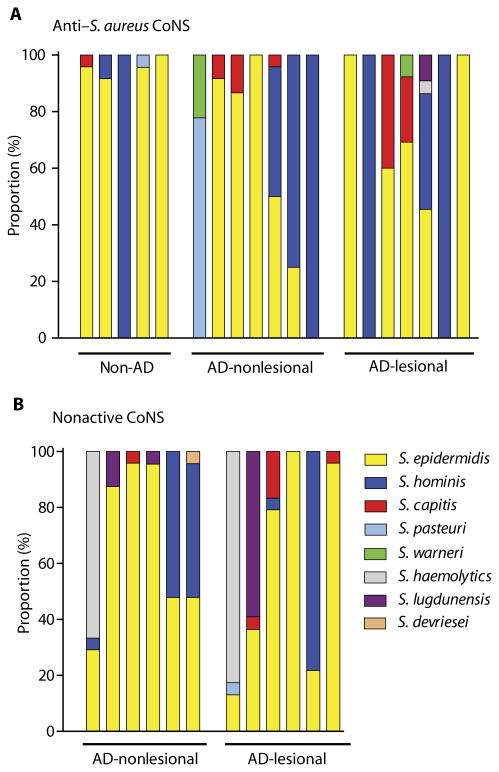
To identify the bacteria that had antimicrobial activity, full-length 16S rRNA gene sequencing was performed on active clones (refer to table S2 for the number of clones analyzed). The predominant species of CoNS with anti–S.aureus activity was either S. epidermidis or Staphylococcus hominis, although other species were also observed with this activity (Fig.3A and table S2, A and B).However, many clones of these CoNS species did not produce antimicrobial activity, thus demonstrating that the strain, and not only species identity, predicts antimicrobial function (Fig. 3B and table S2B).
Fig. 3. Antimicrobial activity is detected in diverse strains of CoNS and not predictable at the species level.
(A) Relative abundance of CoNS species with antimicrobial activity from five non-AD subjects and seven subjects with AD, who were randomly selected. Up to 48 CoNS isolates were randomly selected from each individual for 16S rRNA sequencing to identify species. Refer to table S2 for details. (B) Relative abundance of CoNS species without anti–S.aureus activity isolated from six subjects with AD. Refer to table S2 for details.
Application of antimicrobial CoNS strains to skin inhibits S.aureus
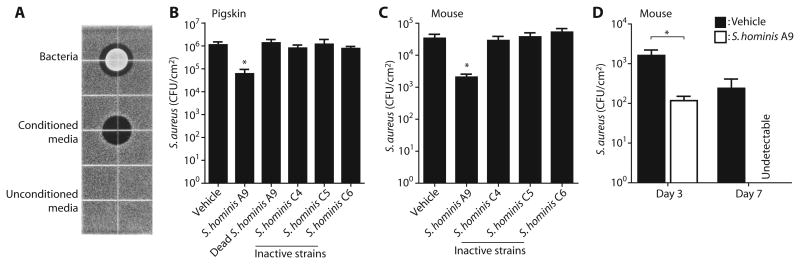
To confirm that the antimicrobial function detected through random screening in vitro could be reproduced under the physiologically relevant conditions of the skin, the S. hominis clone A9 was selected for further analysis. This strain was selected because S. hominis was frequently detected in our screen, and the function of S. hominis on human skin was unknown. S. hominis A9 grown on agar generated a clear zone of inhibition of S.aureus growth, thus confirming that this strain spontaneously secreted activity into culture medium at a level sufficient to directly inhibit S.aureus (Fig. 4A). To directly test whether this clone could reduce colonization by S.aureus on the skin, we applied S. hominis A9 or an equal density of control S. hominis isolates without antimicrobial activity to the surface of sanitized pigskin (31) on which a defined amount of S.aureus had first been applied. The amount of S. hominis applied was similar to the estimated density of bacteria on normal human skin (1 × 105 CFU/cm2). A significant decrease in S.aureus was seen after a single application of S. hominis A9 at this density (P = 0.0276; Fig. 4B). Application of S. hominis A9 that was killed before application or application of control S. hominis strains that did not show antimicrobial activity did not reduce S.aureus survival. Similarly, after colonization of the back skin of mice with a known amount of S.aureus, a single application of S. hominis A9 reduced S.aureus but control strains of S. hominis had no effect (Fig. 4C). Furthermore, twice-a-day applications of S. hominis A9 for 1 week completely eliminated S.aureus colonization compared to vehicle (Fig. 4D). Vehicle treatment did not affect S.aureus survival on the skin surface in comparison to untreated control (fig. S5). These observations show that S. hominis A9 produced anti–S.aureus activity when present on the skin and prompted further biochemical and genetic identification of the molecule(s) responsible for this activity.
Fig. 4. Colonization by an antimicrobial CoNS strain is protective against S.aureus.
(A)Anti–S.aureus activity secreted from a representative antimicrobial S. hominis strain (A9) isolated from a non-AD subject. A live colony of S. hominis A9 (upper), conditioned medium (10 μl) from an S. hominis A9 overnight culture (middle), or unconditioned medium [tryptic soy broth (TSB)] served as a negative control were applied on TSB agar containing S.aureus. The black area represents zone of growth inhibition of S.aureus.(B and C) Effect of S. hominis on the survival of S.aureus on ex situ pigskin (B) or S. hominis applied to live mouse skin (C). S.aureus was first applied to skin as described in Materials and Methods. The action of S. hominis A9 was compared to controls, including UV-killed and washed A9 or live S. hominis strains that do not produce AMP activity in solution assay (C4, C5, and C6). All CoNS bacteria were applied at 1 × 105CFU/cm2. Data represent means ± SEM of data from five pigskin sheets (B) or six mice (C). (D) Effect of multiple applications of S. hominis A9 on survival of S.aureus on mouse skin. S. hominis A9 strain or vehicle was applied twice a day to mouse back skin colonized by S.aureus over the indicated periods. Skin swabs were collected to count S.aureus survival. Data represent means ± SEM from 10 (day 3) or 5 (day 7) independent mice. *P < 0.05 by two-tailed independent t test.
Commensal bacteria on human skin produce AMPs
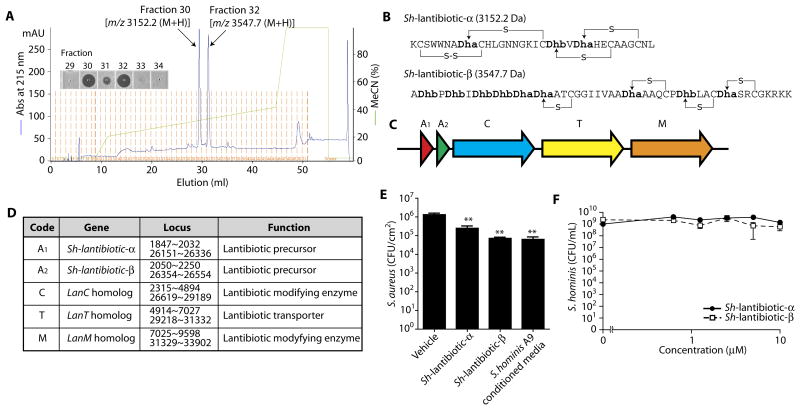
Reverse-phase chromatography revealed two independent peaks with antimicrobial activity in the conditioned medium of S. hominis A9 (Fig.5A and fig. S6). Edman degradation, whole-genome sequencing, and genome-guided matrix-assisted laser desorption/ionization–time-of-flight (MALDI-TOF)/TOF analyses predicted the structures for the mature forms of these AMPs as lantibiotics (Fig. 5B and fig. S7). These proteins were encoded within a gene cluster that included lanM, lanC, and lanT homologs (Fig.5,C and D), and were duplicated within the genome, but not detected within the genome of another S. hominis strain previously sequenced (32). Because these S. hominis AMPs had similar structure to known lantibiotics (33), we refer to them henceforth as Sh-lantibiotic-α and Sh-lantibiotic-β. To validate the antimicrobial function of Sh-lantibiotic-α and Sh-lantibiotic- β on skin, we applied 0.5 nmol of purified Sh-lantibiotics to sanitized pigskin to which S.aureus had been previously applied. Direct application of these Sh-lantibiotic peptides to skin, or application of sterile conditioned culture medium from S. hominis A9, significantly inhibited S.aureus (P < 0.01; Fig. 5E). In contrast, these Sh-lantibiotics did not act against S. hominis A9 itself at concentrations up to 10 μM(Fig. 5F). S. hominis A9 strongly suppressed growth of multiple strains of S.aureus, including MRSA strain USA 300, and clinical isolates from subjects with AD but did not affect growth of other species frequently isolated from normal human skin, including Propionibacterium acnes, S. epidermidis, and Corynebacterium minutissimum (fig. S8).
Fig. 5. S. hominis A9 isolated from normal human skin produces unique lantibiotics.
(A) Reverse-phase high-performance liquid chromatography elution profile of peptides purified from culture supernatant of S. hominis A9 strain. The insert is a radial diffusion activity against S.aureus from the indicated fractions. Molecular mass of fractions 30 and 32 was measured by MALDI-TOF mass spectrometry (MS) (fig. S4). (B) Amino acid sequence and predicted thioester bonds from the two lantibiotics purified from S. hominis A9. Dha, 2,3-didehydroalanine; Dhb, (Z)-2,3-didehydrobutyrine. (C) Organization of the gene cluster encoding Sh-lantibiotic precursors and lanti-biotic biosynthetic genes in S. hominis A9. (D) List of lantibiotic-related genes, gene locus, and putative functions. (E) Effect of application of Sh-lantibiotics (0.5 nmol) or conditioned medium from S. hominis A9 (50 μl) on survival of S.aureus on pigskin. Data represent means ± SEM of four independent assays. **P < 0.01 by two-tailed independent t test. (F) Dose-response curves for the antimicrobial activity of Sh-lantibiotic-α and Sh-lantibiotic-β against S. hominis A9 strain. Data represent means ± SEM of triplicate assays. m/z, mass/charge ratio.
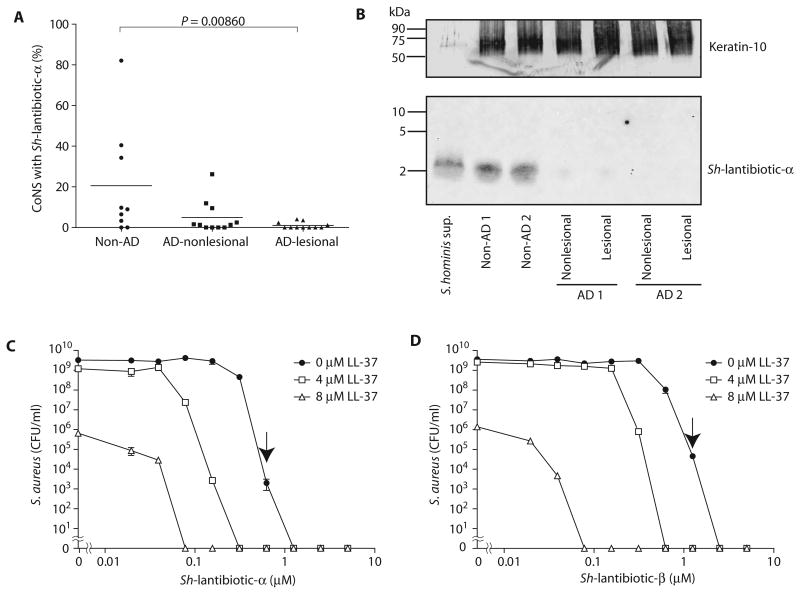
Identification of the genes encoding Sh-lantibiotics permitted us to screen subjects by direct colony PCR to determine the frequency of this AMP in the CoNS community residing on the skin of human subjects. PCR analysis was performed on 632 CoNS colonies from the skin of 9 randomly selected normal subjects and 1713 CoNS colonies from 11 randomly selected individuals with AD. These results showed that the Sh-lantibiotic-α genes were common in the bacterial community on normal subjects but not on AD subjects (Fig. 6A), a finding consistent with our functional screening results shown earlier in Fig. 2. Western blot analysis using mouse polyclonal immunoglobulin G raised against Sh-lantibiotic-α peptide showed that the peptide could be detected on the skin of non-AD subjects that were PCR-positive for the Sh-lantibiotic-α gene, but not on AD subjects that were PCR-negative (Fig. 6B and fig. S9), thus confirming that the protein for this lantibiotic was constitutively expressed on human skin and correlated with the presence of the gene. These Sh-lantibiotics were likely not the only antimicrobials produced by the skin microbiome because genome sequencing of additional clones selected by functional screening revealed other putative lantibiotic and bacteriocin genes in other active isolates (fig. S10). We also identified previously known lantibiotics, such as epidermin and Pep-5 (34), in the genomes of some S. epidermidis clones with anti–S.aureus activity isolated from normal skin (fig. S11). Lantibiotic-related genes were not found in the genomes of strains that did not exhibit antimicrobial activity such as S. hominis C4, C5, and C6.
Fig. 6. Sh-lantibiotics are commonly found on healthy human skin and synergize with a host AMP.
(A) Frequency of detecting Sh-lantibiotic-α by colony PCR using gene-specific primers in CoNS isolates from human skin. Each point represents analysis of one individual. Bar, mean. P value was calculated by Wilcoxon-Mann-Whitney test. (B) Detection of Sh-lantibiotic-α peptide by Western blotting from extracts of skin swabs taken from two non-AD subjects who were colonized by bacteria having the Sh-lantibiotic-α gene and two AD subjects who were PCR-negative for the Sh-lantibiotic-α gene. S. hominis culture supernatant was loaded as a positive control. A total of 20 mg of protein was loaded in each lane. The uncropped image is shown in fig. S9. The membrane was restained with antibody against cytokeratin-10, a predominant protein in the stratum corneum, as a loading control. (C and D) Dose-response curves for the antimicrobial activity of Sh-lantibiotic-α (C) and Sh-lantibiotic-β (D) against S.aureus and their synergistic antimicrobial activity with human LL-37. Data represent means ± SEM of triplicate assays. Arrow shows minimal bactericidal concentration.
Sh-lantibiotics synergize with a human AMP
The human skin expresses several human AMPs, and therefore, these host AMPs will be present together with the bacterial Sh-lantibiotics on the skin surface. This shared location raised the potential that the bacterial AMPs may act with human AMPs to defend against pathogens. To examine this, we combined each Sh-lantibiotic with LL-37, a mammalian AMP of the cathelicidin family. Sh-lantibiotic-α and Sh-lantibiotic-β showed strong activity alone and had superior activity against S.aureus compared to known human AMPs (Fig. 6, C and D) (35). LL-37 alone (8 μM) decreased S.aureus survival by >3-log, whereas 4 μM LL-37 alone did not affect S.aureus survival in vitro. Combination with 4 and 8 μM LL-37, concentrations consistent with those measured on human skin, enhanced the antimicrobial activity of each Sh-lantibiotic by 4-fold (with 4 μM LL-37) and 16-fold (Sh-lantibiotic-α with 8 μM LL-37) or 32-fold (Sh-lantibiotic-β with 8 μM LL-37), respectively, suggesting synergistic action between prokaryotic and host AMPs.
Application of bacteria with antimicrobial activity decreased S.aureus in AD
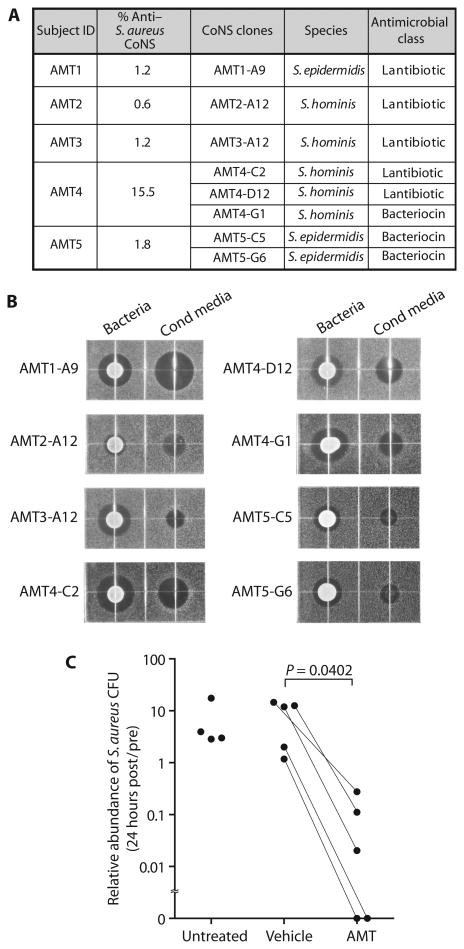
To investigate the capacity of commensal bacteria to inhibit S.aureus on human skin, we tested the effect of applying these bacteria to subjects with AD. Five AD subjects who were S.aureus culture–positive were recruited to participate in this study (table S3). Strains with antimicrobial activity were infrequent within the total CoNS community on these AD subjects, but rare clones could be isolated if sufficient CoNS colonies were screened. Only a single S. epidermidis or S. hominis strain with antimicrobial activity was isolated from the three subjects. In these cases, a single strain of CoNS was formulated (refer to Fig. 7A). Three and two antimicrobial S. hominis or S. epidermidis strains were isolated from two AD subjects. The whole genome of each active clone was sequenced, and clusters of lantibiotic- or bacteriocin-related genes were identified in all active clones (Fig. 7A and fig. S10). All selected clones and their conditioned media were confirmed to produce a zone of inhibition of S.aureus growth by radial diffusion assay (Fig. 7B). These clones with antimicrobial activity were then expanded and formulated in a cream vehicle base (Cetaphil lotion), and a single dose was applied in a double-blind fashion to the forearm of each subject, whereas vehicle alone was applied to the contralateral arm (autologous transplant) (refer to fig. S12 for study design). Bacteria were applied to the skin for a final concentration of 1 × 105 CFU/cm2, a density similar to previous assessments of the abundance of bacteria on normal human skin. Subjects were instructed not to wash for 24 hours after application. S.aureus was measured at each site before and 24 hours after application of active CoNS transplant or vehicle. To assess potential vehicle effects, we left some subjects untreated. Twenty-four hours after initiation of the study, no significant difference in S.aureus abundance was detected between untreated individuals and individuals treated with vehicle alone (Fig. 7C), although in the absence of washing, a trend toward an increase in bacterial abundance from baseline was seen in both untreated and vehicle-treated groups (fig. S13). In contrast, a single application of antimicrobial CoNS strain(s) significantly decreased S.aureus abundance compared to vehicle (P = 0.0402; Fig. 7C and fig. S13). These data confirmed that CoNS strains with antimicrobial activity can directly suppress S.aureus colonization on human subjects with AD.
Fig. 7. Transplantation of antimicrobial CoNS reduces survival of S.aureus on human skin.
(A) Characterization of CoNS clones used for autologous micro-biome transplant (AMT). Antimicrobial class of each clone was identified by whole-genome sequencing (refer to fig. S11 for more details). (B) Radial diffusion assay for anti–S.aureus activity secreted from each active CoNS strain used for AMT. Radial diffusion assay of bacteria and conditioned medium was conducted as described previously. (C) Effect of transplantation of antimicrobial CoNS or vehicle on the survival of S.aureus on the skin of five subjects with AD. S.aureus survival was measured by colony counting before transplant (pre) and 24 hours after a single application of bacteria (post). Application and analysis were done in a blinded fashion, and samples are from the contralateral arm of subjects treated with the identical vehicle containing antimicrobial bacteria or vehicle only. Data of AMT or placebo treatment (vehicle) are compared with data from four untreated control subjects with AD. P value was calculated by two-tailed paired t test.
Discussion
AD is a common and severe skin disorder that harbors an abnormal community of bacteria on the skin. In particular, patients with AD are frequently colonized by S.aureus that then exacerbates the disease by further promoting inflammation (7, 8). We hypothesized that the commensal bacteria residing on normal skin might contribute to host defense by producing molecules that inhibit colonization by S.aureus. Furthermore, we examined whether the dysbiosis seen in AD may be functionally important because of a loss of these protective strains. We observed that several different bacterial species on healthy human skin produce anti–S.aureus activity and that bacteria with this activity were far less frequent on AD subjects. The relative absence of commensal bacterial strains producing antimicrobial activity was most evident on individuals colonized by S.aureus. Indeed, no subject was colonized by S.aureus if they also had a normal abundance of CoNS bacteria that produced antimicrobial activity, and application of antimicrobial CoNS strains to animal or human skin greatly reduced S.aureus colonization. Together, these observations show that specific bacteria within the human skin microbiome defend against S.aureus.
In normal skin, culture-based methods for estimating CFU detected a lower abundance of bacteria than DNA-based methods. This was not likely to be inherent to the techniques themselves because the two methods yielded similar results when samples were taken from AD lesional skin. There are several potential explanations for this observation, including differences in capacity to sample bacterial DNA on AD lesional skin compared to healthy skin, or other products in the samples influencing the assay results. However, it is also possible that more bacteria on normal skin are dead than on AD lesional skin. Indeed, we have shown here that normal skin has more effective surface antimicrobial activity than AD skin. AMPs such as LL-37, β-defensin-2, and β-defensin-3 also have lesser expression in inflamed skin of AD patients than in inflamed skin of normal subjects, but the constitutive expression of these AMPs is low in noninflamed skin (10–13, 36). Therefore, the increased capacity of noninflamed normal skin to kill bacteria is not likely due to the expression of these host AMPs. Alternatively, the higher antimicrobial activity generated by the commensal CoNS community on normal skin can explain this increased surface antimicrobial action. In this model, the microbiome provides the first line of defense, whereas the host innate immune system provides the second line of defense, which is activated only after the surface is damaged to trigger host AMP production.
Unbiased functional selection of commensal CoNS strains that were prevalent on human skin permitted identification of previously unknown AMPs. We report purification and validation of two potent lantibiotics in a common commensal strain of S. hominis. These lantibiotics were constitutively secreted by bacteria at concentrations that are sufficient to kill S.aureus on the skin surface, detectable on the surface of normal human skin, and highly synergistic with LL-37, an important human AMP (2, 37). The synergistic action between prokaryotic and host AMPs suggests that the microbe-derived molecules should be considered as a component of the normal skin immune defense system. Several other bacterial species were also found to produce antimicrobial activity. Although Gram-negative bacteria have been rarely detected on normal human skin, it has been reported recently that selective culture systems could successfully expand some Gram-negative bacteria from healthy donors and that these could suppress S.aureus growth on AD subjects (38). It is unclear whether the presence of such Gram-negative bacteria influences normal host defense. Furthermore, other potential activities detected in laboratory strains of bacteria such as the capacity to inhibit biofilm formation (39) may also play a role in normal skin protection. A limitation of this current study is that a complete catalog of protective bacteria cannot be identified at this time. A wide range of microbes may have this function, and factors that influence their survival are not currently understood. Overall, these data suggest that the human skin microbiome could provide a diverse array of beneficial factors that may require functional screening to first detect.
The Sh-lantibiotics identified here showed selective activity against S.aureus but did not inhibit the growth of S. hominis and S. epidermidis. Other isolates of CoNS also exerted selective antimicrobial activity against S.aureus but not against bacterial species frequently found on the healthy skin. Previously identified AMPs from S. epidermidis were also known to exert selective killing, a logical behavior if the cell is to resist killing itself (30). The mechanism responsible for selective killing by bacterial AMPs may be due to differences in the capacity to disrupt the microbial cell membrane. The capacity for selective killing of pathogenic bacteria over the normal microflora is highly desirable because it will help to shape the normal bacterial community. This illustrates the potential in further analysis of the host defense function of the human skin microbiome and is consistent with observations that expression of a bacteriocin in the intestinal microbiome can outcompete pathogens at the epithelial interface (40). Because we found that antimicrobial function of skin bacteria is not predictable at the species level, more detailed metagenomic (41), transcriptomic (42), proteomic, and metabolomic analyses (43, 44) will provide a more complete understanding of the diverse antibacterial defense system of the skin.
We have shown here in both animal and human systems that topical application of rationally selected bacterial strains can decrease S.aureus abundance. This approach is inherently superior to current pharmaceutically derived antibiotics because they should not disrupt cutaneous homeostasis by nonspecific killing of the normal microflora. On the basis of our observations, nonspecific antibacterial action by common pharmaceutical antibiotics could kill protective strains of CoNS and might therefore enhance the potential for recolonization by S.aureus. The selective activity of antimicrobials derived from normal flora would avoid this nonspecific effect and illustrates why bacteriotherapy that kills S.aureus but does not inhibit other beneficial CoNS strains could be superior. Thus, application of a single selective CoNS AMP may permit emergence of multiple different AMPs, an environment less likely to promote antibiotic resistance to any single antibiotic. Long-term protection could be achieved if the applied antimicrobial bacteria could successfully colonize on the skin surface. We demonstrated that repeated application of active CoNS for 1 week completely eliminated S.aureus on mice. The benefits of long-term/multiple applications of antimicrobial bacteria on the skin of subjects with AD remain to be determined.
Enhancing the presence of certain CoNS strains could benefit the patient by also exerting anti-inflammatory actions. For example, S. epidermidis has been shown to be anti-inflammatory (22, 25). Given the complex nature of AD, ideal bacteriotherapy would include both targeting repair of the intrinsic epidermal barrier and optimizing the immune defense functions provided by the resident bacteria. Some members of normal skin flora may be able to do both (22–26, 29, 30). Extended periods of bacteriotherapy will be necessary to evaluate this effect.
In conclusion, we report here evidence that the community of bacteria residing on normal human skin provides protection against S.aureus. A lack of this activity on the skin of subjects with AD is strongly associated with increased colonization by S.aureus.It is not clear at present whether this dysfunction of the bacterial community in AD was an intrinsic result of the disease itself or whether it is attributed to extrinsic factors in subjects with AD such as environmental exposure or the action of other members of the AD bacterial community to suppress commensal microbes that are beneficial and in turn resist S.aureus. The presence of S.aureus may also be contributing to the suppression of the protective strains of CoNS identified here. However, with the identification of unique antimicrobials and bacterial strains from healthy human skin, it is possible to exploit this information in diagnostically and therapeutically useful ways. Such observations will further define the important mutualistic relationship that exists between humans and some bacteria at the epithelial interface.
Materials and Methods
Study design
This study was designed to test the hypothesis that the skin commensal bacteria contribute to the host defense by producing molecules that can kill S.aureus. Adults with AD and age-matched non-AD subjects were recruited from the University of California, San Diego (UCSD), La Jolla, CA, and the National Jewish Health (NJH), Denver, CO (demographic data are shown in table S1). All subjects avoided any treatments and therapies that may potentially affect skin microbiome before sample collection (refer to the Supplementary Materials for sampling collection criteria). Collection of surface bacteria was done from lesional skin on the antecubital fossa and from nonlesional skin of the upper arm. Microbial communities on the skin surface of healthy subjects and patients with AD were evaluated by culture-based colony counting, qPCR, and 16S rRNA community sequencing. Antimicrobial activity of CoNS isolated from the skin of healthy subjects and patients with AD was measured against S.aureus by high-throughput screening. The species of CoNS with or without anti–S.aureus activity were identified by 16S rRNA sequence. Antimicrobial activity produced by representative CoNS strain isolated from a non-AD healthy subject was characterized by liquid chromatography, MALDI-TOF/TOF MS, Edman end-terminal sequencing, and genome sequencing. To test the therapeutic potential of CoNS strains with anti–S.aureus activity, active strains were applied at physiologically relevant density on ex situ pigskin or mouse skin colonized by S.aureus. To further investigate the antimicrobial activity of CoNS in human, CoNS strains with anti–S.aureus activity were autologously reapplied to the lesional skin of one arm of the AD subject, whereas vehicle only was applied to the contralateral arm. All treatment was conducted in a double-blinded fashion and unblinded after all results were analyzed. No power analysis was performed to predetermine sample size. The number of experimental replications is provided in the figure legends. The details of all protocols and all primary data for experiments where n < 20 (table S5) are provided in the Supplementary Materials.
Human subjects
All experiments involving human subjects were carried out according to the institutional review board protocols approved by UCSD (project no. 071032) and NJH (HS-2581). Informed consent was obtained from all subjects.
Screening for antimicrobial activity
Up to 84 individual isolated colonies of CoNS from each skin site were randomly picked and cultured in TSB at 37°C overnight. Each plate also contained wells with a nonantimicrobial strain of S. epidermidis (ATCC1457) as negative control and a potent antimicrobial strain of S. hominis (A9 strain) as positive control. The antimicrobial activity of sterile conditioned medium prepared from each CoNS culture was evaluated by mixing with 1 × 104 CFU of S.aureus (ATCC35556). Antimicrobial strains were defined as those that suppressed S.aureus growth after 22 hours to less than 50% (I50) of growth seen in negative controls.
Transplantation of antimicrobial CoNS on mice and ex situ pigskin
All experiments involving live animal work were in accordance with the approval of the Institutional Animal Care and Use Guidelines of the University of California, San Diego (protocol no. S09074). Fresh-frozen pigskin was purchased as sheets and was sanitized by 3% chloroxylenol and rinsed with sterile phosphate-buffered saline. For mouse experiments, the backs of female C57BL6 mice (6 weeks old) were shaved. S.aureus was then epicutaneously applied on the dorsal skin of mice or pigskin at the indicated CFU density. S. hominis A9 strain or inactive strains were subsequently applied on the skin for 20 hours. Live bacteria were harvested by swabbing to measure S.aureus survival.
Autologous microbiome transplant
The approach of AMT for subjects with AD has been officially approved by the U.S. Food and Drug Administration, and this protocol has been filed as an investigational new drug application (UCSD approval no. 15786). This trial has been registered at ClinicalTrials.gov (NCT01959113). AD subjects who are S.aureus carriers on the lesional sites of both antecubital fossa were screened. CoNS strains with anti–S.aureus activity were screened from the nonlesional site and identified by 16S rRNA sequencing. Each CoNS strain was expanded in TSB overnight and formulated at 1 × 107 CFU/g in Cetaphil lotion (Galderma). When multiple strains were included, an equal CFU of each CoNS was formulated at a total concentration of 1 × 107 CFU/g. One arm was treated with AMT formulation to get CoNS (1 × 105 CFU/cm2) on the target skin. The other arm received an equal amount of moisturizer only. S.aureus CFU was measured before and 24 hours after transplant.
Statistical analysis
Paired t tests (two-tailed) were used to compare lesional to nonlesional samples within AD subjects, and independent t tests (two-tailed) were used to compare non-AD to AD samples. For nonnormally distributed variables such as CoNS with Sh-antibiotic-α frequency, non-parametric approaches such as Wilcoxon-Mann-Whitney tests for non-AD to AD samples and Wilcoxon signed-rank tests for lesional to nonlesional samples within AD subjects were used. Longitudinal mixed models of frequency of antimicrobial CoNS and the ratio of live Staphylococcus to Staphylococcus DNA over time were also fit. Each model included lesion type, visit, and their interaction term as fixed effects, whereas a compound symmetry structure was used to account for correlation between samples obtained from the same subject at multiple time points. Frequency of antimicrobial CoNS used a cumulative logit link and multinomial distribution of categorized percentages (≤20, 21–79, ≥80) to account for a bimodal distribution. Statistical analyses were performed using SAS software (version 9.3) and R software (version 3.1.1).
Supplementary Material
Fig. S1. Analysis of the skin bacterial community by 16S rRNA gene sequencing and its relationship to the capacity to culture S.aureus from the skin.
Fig. S2. Abundance of live Staphylococcus and Staphylococcus-specific DNA on the skin of non-AD and AD subjects.
Fig. S3. Specificity of Staphylococcus genus–specific primers for real-time qPCR.
Fig. S4. Anti–S.aureus activity in CoNS is stable over time.
Fig. S5. Effect of vehicle treatment on the survival of S.aureus on ex situ pigskin or live mouse skin.
Fig. S6. MALDI-TOF MS analysis for two AMPs purified from S. hominis.
Fig. S7. Representation of amino acid losses in genome-guided MALDI-TOF/TOF analysis for Sh-lantibiotic-β.
Fig. S8. Skin isolate strains of CoNS exert selective antimicrobial activity against S.aureus.
Fig. S9. Detection of Sh-lantibiotic-α peptide by Western blotting from extracts of skin swabs taken from two non-AD subjects and two AD subjects.
Fig. S10. List of hypothetical antimicrobial genes identified in anti–S.aureus CoNS clones used for AMT.
Fig. S11. List of previously known lantibiotic genes identified in antimicrobial S. epidermidis clones isolated from non-AD skin.
Fig. S12. Study design of AMT therapy for patients with AD.
Fig. S13. Effect of transplantation of antimicrobial CoNS or vehicle on the survival of S.aureus on the skin of five subjects with AD.
Table S1. Clinical characteristics of AD and non-AD subjects.
Table S2. Proportion of CoNS species identified with antimicrobial and nonantimicrobial activity.
Table S3. Clinical characteristics of AD subjects receiving AMT.
Table S4. Sequences of PCR primers.
Table S5. Primary data (provided as an Excel file).
Acknowledgments
Funding: This work was funded by the Atopic Dermatitis Research Network (ADRN) (NIH/NIAID 1 U19 AI117673-02 to R.L.G. and D.Y.M.L.), the Rho Federal Systems Division, and NIH grants (R21AR067547, R01AR06781, R01AI052453, and R01AI118816 to R.L.G.). S.N. is supported by an NIH grant (1TL1TR001443).
Footnotes
Author contributions: T.N. and R.L.G. conceived and designed this study and wrote the manuscript. T.R.H., A.M.T., K.A.C., T.Y., F.S., P.T., J.S., D.Y.M.L., and R.L.G. procured clinical samples. K.A. and G.D. coordinated the clinical study. T.N., T.H.C., S.N., and P.F.K. analyzed data from clinical samples. T.N., A.B., A.V.M., and P.C.D. assisted in the identification of AMPs. H.L., J.-N.K., and K.Z. performed genome sequencing and analysis of CoNS strains. A.G. and S.R.G. conducted microbiome 16S rRNA sequencing. T.N., R.L.G., A.L., and G.D. evaluated and analyzed the data and conducted statistical analysis. D.Y.M.L. and R.L.G. directed this project as the principal investigators of ADRN.
Competing interests: T.N. and R.L.G. are co-inventors of UCSD technology related to the bacterial AMPs discussed herein, and R.L.G. is a cofounder and consultant of MatriSys Bioscience (La Jolla, CA), a company that is developing skin bacteriotherapy.
Data and materials availability: The 16S rRNA sequence data for this study have been published in the DDBJ Sequence Read Archive (http://trace.ddbj.nig.ac.jp/dra/index_e.html) under the accession code DRA005329.
References and Notes
- 1.Nakatsuji T, Gallo RL. Antimicrobial peptides: Old molecules with new ideas. J Invest Dermatol. 2012;132:887–895. doi: 10.1038/jid.2011.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gallo RL, Hooper LV. Epithelial antimicrobial defence of the skin and intestine. Nat Rev Immunol. 2012;12:503–516. doi: 10.1038/nri3228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang Lj, Guerrero-Juarez CF, Hata T, Bapat SP, Ramos R, Plikus MV, Gallo RL. Innate immunity. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science. 2015;347:67–71. doi: 10.1126/science.1260972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clevers HC, Bevins CL. Paneth cells: Maestros of the small intestinal crypts. Annu Rev Physiol. 2013;75:289–311. doi: 10.1146/annurev-physiol-030212-183744. [DOI] [PubMed] [Google Scholar]
- 5.Schommer NN, Gallo RL. Structure and function of the human skin microbiome. Trends Microbiol. 2013;21:660–668. doi: 10.1016/j.tim.2013.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grice EA, Kong HH, Conlan S, Deming CB, Davis J, Young AC, NISC Comparative Sequencing Program. Bouffard GG, Blakesley RW, Murray PR, Green ED, Turner ML, Segre JA. Topographical and temporal diversity of the human skin microbiome. Science. 2009;324:1190–1192. doi: 10.1126/science.1171700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kong HH, Oh J, Deming C, Conlan S, Grice EA, Beatson MA, Nomicos E, Polley EC, Komarow HD, NISC Comparative Sequence Program. Murray PR, Turner ML, Segre JA. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22:850–859. doi: 10.1101/gr.131029.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Leyden JJ, Marples RR, Kligman AM. Staphylococcus aureus in the lesions of atopic dermatitis. Br J Dermatol. 1974;90:525–530. doi: 10.1111/j.1365-2133.1974.tb06447.x. [DOI] [PubMed] [Google Scholar]
- 9.Mallbris L, Carlén L, Wei T, Heilborn J, Nilsson MF, Granath F, Ståhle M. Injury downregulates the expression of the human cathelicidin protein hCAP18/LL-37 in atopic dermatitis. Exp Dermatol. 2010;19:442–449. doi: 10.1111/j.1600-0625.2009.00918.x. [DOI] [PubMed] [Google Scholar]
- 10.Ong PY, Ohtake T, Brandt C, Strickland I, Boguniewicz M, Ganz T, Gallo RL, Leung DYM. Endogenous antimicrobial peptides and skin infections in atopic dermatitis. N Engl J Med. 2002;347:1151–1160. doi: 10.1056/NEJMoa021481. [DOI] [PubMed] [Google Scholar]
- 11.Hata TR, Kotol P, Boguniewicz M, Taylor P, Paik A, Jackson M, Nguyen M, Kabigting F, Miller J, Gerber M, Zaccaro D, Armstrong B, Dorschner R, Leung DYM, Gallo RL. History of eczema herpeticum is associated with the inability to induce human β-defensin (HBD)-2, HBD-3 and cathelicidin in the skin of patients with atopic dermatitis. Br J Dermatol. 2010;163:659–661. doi: 10.1111/j.1365-2133.2010.09892.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Howell MD, Boguniewicz M, Pastore S, Novak N, Bieber T, Girolomoni G, Leung DYM. Mechanism of HBD-3 deficiency in atopic dermatitis. Clin Immunol. 2006;121:332–338. doi: 10.1016/j.clim.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 13.Howell MD, Gallo RL, Boguniewicz M, Jones JF, Wong C, Streib JE, Leung DYM. Cytokine milieu of atopic dermatitis skin subverts the innate immune response to vaccinia virus. Immunity. 2006;24:341–348. doi: 10.1016/j.immuni.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 14.Allakhverdi Z, Comeau MR, Jessup HK, Yoon BRP, Brewer A, Chartier S, Paquette N, Ziegler SF, Sarfati M, Delespesse G. Thymic stromal lymphopoietin is released by human epithelial cells in response to microbes, trauma, or inflammation and potently activates mast cells. J Exp Med. 2007;204:253–258. doi: 10.1084/jem.20062211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sieprawska-Lupa M, Mydel P, Krawczyk K, Wojcik K, Puklo M, Lupa B, Suder P, Silberring J, Reed M, Pohl J, Shafer W, McAleese F, Foster T, Travis J, Potempa J. Degradation of human antimicrobial peptide LL-37 by Staphylococcus aureus-derived proteinases. Antimicrob Agents Chemother. 2004;48:4673–4679. doi: 10.1128/AAC.48.12.4673-4679.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vu AT, Baba T, Chen X, Le TA, Kinoshita H, Xie Y, Kamijo S, Hiramatsu K, Ikeda S, Ogawa H, Okumura K, Takai T. Staphylococcus aureus membrane and diacylated lipopeptide induce thymic stromal lymphopoietin in keratinocytes through the Toll-like receptor 2–Toll-like receptor 6 pathway. J Allergy Clin Immunol. 2010;126:985–993.e3. doi: 10.1016/j.jaci.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 17.Nakamura Y, Oscherwitz J, Cease KB, Chan SM, Muñoz-Planillo R, Hasegawa M, Villaruz AE, Cheung GYC, McGavin MJ, Travers JB, Otto M, Inohara N, Núñez G. Staphylococcus d-toxin induces allergic skin disease by activating mast cells. Nature. 2013;503:397–401. doi: 10.1038/nature12655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller LS, Cho JS. Immunity against Staphylococcus aureus cutaneous infections. Nat Rev Immunol. 2011;11:505–518. doi: 10.1038/nri3010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kobayashi T, Glatz M, Horiuchi K, Kawasaki H, Akiyama H, Kaplan DH, Kong HH, Amagai M, Nagao K. Dysbiosis and Staphylococcus aureus colonization drives inflammation in atopic dermatitis. Immunity. 2015;42:756–766. doi: 10.1016/j.immuni.2015.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakatsuji T, Chen TH, Two AM, Chun KA, Narala S, Geha RS, Hata TR, Gallo RL. Staphylococcus aureus exploits epidermal barrier defects in atopic dermatitis to trigger cytokine expression. J Invest Dermatol. 2016;136:2192–2200. doi: 10.1016/j.jid.2016.05.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gallo RL, Nakatsuji T. Microbial symbiosis with the innate immune defense system of the skin. J Invest Dermatol. 2011;131:1974–1980. doi: 10.1038/jid.2011.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lai Y, Di Nardo A, Nakatsuji T, Leichtle A, Yang Y, Cogen AL, Wu ZR, Hooper LV, Schmidt RR, von Aulock S, Radek KA, Huang CM, Ryan AF, Gallo RL. Commensal bacteria regulate Toll-like receptor 3–dependent inflammation after skin injury. Nat Med. 2009;15:1377–1382. doi: 10.1038/nm.2062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Naik S, Bouladoux N, Linehan JL, Han SJ, Harrison OJ, Wilhelm C, Conlan S, Himmelfarb S, Byrd AL, Deming C, Quinones M, Brenchley JM, Kong HH, Tussiwand R, Murphy KM, Merad M, Segre JA, Belkaid Y. Commensal–dendritic-cell interaction specifies a unique protective skin immune signature. Nature. 2015;250:104–108. doi: 10.1038/nature14052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Naik S, Bouladoux N, Wilhelm C, Molloy MJ, Salcedo R, Kastenmuller W, Deming C, Quinones M, Koo L, Conlan S, Spencer S, Hall JA, Dzutsev A, Kong H, Campbell DJ, Trinchieri G, Segre JA, Belkaid Y. Compartmentalized control of skin immunity by resident commensals. Science. 2012;337:1115–1119. doi: 10.1126/science.1225152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Laborel-Preneron E, Bianchi P, Boralevi F, Lehours P, Fraysse F, Morice-Picard F, Sugai M, Sato'o Y, Badiou C, Lina G, Schmitt AM, Redoules D, Casas C, Davrinche C. Effects of the Staphylococcus aureus and Staphylococcus epidermidis secretomes isolated from the skin microbiota of atopic children on CD4+ T cell activation. PLOS ONE. 2015;10:e0144323. doi: 10.1371/journal.pone.0141067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lai Y, Cogen AL, Radek KA, Park HJ, Macleod DT, Leichtle A, Ryan AF, Di Nardo A, Gallo RL. Activation of TLR2 by a small molecule produced by Staphylococcus epidermidis increases antimicrobial defense against bacterial skin infections. J Invest Dermatol. 2010;130:2211–2221. doi: 10.1038/jid.2010.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wanke I, Steffen H, Christ C, Krismer B, Götz F, Peschel A, Schaller M, Schittek B. Skin commensals amplify the innate immune response to pathogens by activation of distinct signaling pathways. J Invest Dermatol. 2011;131:382–390. doi: 10.1038/jid.2010.328. [DOI] [PubMed] [Google Scholar]
- 28.Li D, Lei H, Li Z, Li H, Wang Y, Lai Y. A novel lipopeptide from skin commensal activates TLR2/CD36-p38 MAPK signaling to increase antibacterial defense against bacterial infection. PLOS ONE. 2013;8:e58288. doi: 10.1371/journal.pone.0058288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cogen AL, Yamasaki K, Muto J, Sanchez KM, Crotty Alexander L, Tanios J, Lai Y, Kim JE, Nizet V, Gallo RL. Staphylococcus epidermidis antimicrobial δ-toxin (phenolsoluble modulin-g) cooperates with host antimicrobial peptides to kill group A Streptococcus. PLOS ONE. 2010;5:e8557. doi: 10.1371/journal.pone.0008557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cogen AL, Yamasaki K, Sanchez KM, Dorschner RA, Lai Y, MacLeod DT, Torpey JW, Otto M, Nizet V, Kim JE, Gallo RL. Selective antimicrobial action is provided by phenol-soluble modulins derived from Staphylococcus epidermidis, a normal resident of the skin. J Invest Dermatol. 2010;130:192–200. doi: 10.1038/jid.2009.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rubinchik E, Dugourd D, Algara T, Pasetka C, Friedland HD. Antimicrobial and antifungal activities of a novel cationic antimicrobial peptide, omiganan, in experimental skin colonisation models. Int J Antimicrob Agents. 2009;34:457–461. doi: 10.1016/j.ijantimicag.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 32.Jiang S, Zheng B, Ding W, Lv L, Ji J, Zhang H, Xiao Y, Li L. Whole-genome sequence of Staphylococcus hominis, an opportunistic pathogen. J Bacteriol. 2012;194:4761–4762. doi: 10.1128/JB.00991-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McAuliffe O, Ross RP, Hill C. Lantibiotics: Structure, biosynthesis and mode of action. FEMS Microbiol Rev. 2001;25:285–308. doi: 10.1111/j.1574-6976.2001.tb00579.x. [DOI] [PubMed] [Google Scholar]
- 34.Fontana MBC, de Bastos MdC, Brandelli A. Bacteriocins Pep5 and epidermin inhibit Staphylococcus epidermidis adhesion to catheters. Curr Microbiol. 2006;52:350–353. doi: 10.1007/s00284-005-0152-5. [DOI] [PubMed] [Google Scholar]
- 35.Dorschner RA, Lin KH, Murakami M, Gallo RL. Neonatal skin in mice and humans expresses increased levels of antimicrobial peptides: Innate immunity during development of the adaptive response. Pediatr Res. 2003;53:566–572. doi: 10.1203/01.PDR.0000057205.64451.B7. [DOI] [PubMed] [Google Scholar]
- 36.Lande R, Gregorio J, Facchinetti V, Chatterjee B, Wang YH, Homey B, Cao W, Wang YH, Su B, Nestle FO, Zal T, Mellman I, Schröder JM, Liu YJ, Gilliet M. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449:564–569. doi: 10.1038/nature06116. [DOI] [PubMed] [Google Scholar]
- 37.Nizet V, Ohtake T, Lauth X, Trowbridge J, Rudisill J, Dorschner RA, Pestonjamasp V, Piraino J, Huttner K, Gallo RL. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature. 2001;414:454–457. doi: 10.1038/35106587. [DOI] [PubMed] [Google Scholar]
- 38.Myles IA, Williams KW, Reckhow JD, Jammeh ML, Pincus NB, Sastalla I, Saleem D, Stone KD, Datta SK. Transplantation of human skin microbiota in models of atopic dermatitis. JCI Insight. 2016;1:e86955. doi: 10.1172/jci.insight.86955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Iwase T, Uehara Y, Shinji H, Tajima A, Seo H, Takada K, Agata T, Mizunoe Y. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization. Nature. 2010;465:346–349. doi: 10.1038/nature09074. [DOI] [PubMed] [Google Scholar]
- 40.Kommineni S, Bretl DJ, Lam V, Chakraborty R, Hayward M, Simpson P, Cao Y, Bousounis P, Kristich CJ, Salzman NH. Bacteriocin production augments niche competition by enterococci in the mammalian gastrointestinal tract. Nature. 2015;526:719–722. doi: 10.1038/nature15524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Oh J, Byrd AL, Deming C, Conlan S, NISC Comparative Sequencing Program. Kong HH, Segre JA. Biogeography and individuality shape function in the human skin metagenome. Nature. 2014;514:59–64. doi: 10.1038/nature13786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kang D, Shi B, Erfe MC, Craft N, Li H. Vitamin B12 modulates the transcriptome of the skin microbiota in acne pathogenesis. Sci Transl Med. 2015;7:293ra103. doi: 10.1126/scitranslmed.aab2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bouslimani A, Porto C, Rath CM, Wang M, Guo Y, Gonzalez A, Berg-Lyon D, Ackermann G, Moeller Christensen GJ, Nakatsuji T, Zhang L, Borkowski AW, Meehan MJ, Dorrestein K, Gallo RL, Bandeira N, Knight R, Alexandrov T, Dorrestein PC. Molecular cartography of the human skin surface in 3D. Proc Natl Acad Sci U S A. 2015;112:E2120–E2129. doi: 10.1073/pnas.1424409112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kaddurah-Daouk R, Baillie RA, Zhu H, Zeng ZB, Wiest MM, Nguyen UT, Wojnoonski K, Watkins SM, Trupp M, Krauss RM. Enteric microbiome metabolites correlate with response to simvastatin treatment. PLOS ONE. 2011;6:e25482. doi: 10.1371/journal.pone.0025482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fadrosh DW, Ma B, Gajer P, Sengamalay N, Ott S, Brotman RM, Ravel J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome. 2014;2:6. doi: 10.1186/2049-2618-2-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Edgar RC. Search and clustering orders of magnitude faster than BLAST. Bioinformatics. 2010;26:2460–2461. doi: 10.1093/bioinformatics/btq461. [DOI] [PubMed] [Google Scholar]
- 48.DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K, Huber T, Dalevi D, Hu P, Andersen GL. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol. 2006;72:5069–5072. doi: 10.1128/AEM.03006-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics. 2011;27:2194–2200. doi: 10.1093/bioinformatics/btr381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang Q, Garrity GM, Tiedje JM, Cole JR. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol. 2007;73:5261–5267. doi: 10.1128/AEM.00062-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Paulson JN, Stine OC, Bravo HC, Pop M. Differential abundance analysis for microbial marker-gene surveys. Nat Methods. 2013;10:1200–1202. doi: 10.1038/nmeth.2658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Carter CH. Egg yolk agar for isolation of coagulase-positive staphylococci. J Bacteriol. 1960;79:753–754. doi: 10.1128/jb.79.5.753-754.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Parisi JT, Hamory BH. Simplified method for the isolation, identification, and characterization of Staphylococcus epidermidis in epidemiologic studies. Diagn Microbiol Infect Dis. 1986;4:29–35. doi: 10.1016/0732-8893(86)90053-2. [DOI] [PubMed] [Google Scholar]
- 54.Cole JR, Wang Q, Fish JA, Chai B, McGarrell DM, Sun Y, Brown CT, Porras-Alfaro A, Kuske CR, Tiedje JM. Ribosomal Database Project: Data and tools for high throughput rRNA analysis. Nucleic Acids Res. 2014;42:D633–D642. doi: 10.1093/nar/gkt1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kersten RD, Yang YL, Xu Y, Cimermancic P, Nam SJ, Fenical W, Fischbach MA, Moore BS, Dorrestein PC. A mass spectrometry–guided genome mining approach for natural product peptidogenomics. Nat Chem Biol. 2011;7:794–802. doi: 10.1038/nchembio.684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Medema MH, Blin K, Cimermancic P, de Jager V, Zakrzewski P, Fischbach MA, Weber T, Takano E, Breitling R. antiSMASH: Rapid identification, annotation and analysis of secondary metabolite biosynthesis gene clusters in bacterial and fungal genome sequences. Nucleic Acids Res. 2011;39:W339–W346. doi: 10.1093/nar/gkr466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Li B, Sher D, Kelly L, Shi Y, Huang K, Knerr PJ, Joewono I, Rusch D, Chisholm SW, van der Donk WA. Catalytic promiscuity in the biosynthesis of cyclic peptide secondary metabolites in planktonic marine cyanobacteria. Proc Natl Acad Sci U S A. 2010;107:10430–10435. doi: 10.1073/pnas.0913677107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tanca A, Palomba A, Deligios M, Cubeddu T, Fraumene C, Biosa G, Pagnozzi D, Addis MF, Uzzau S. Evaluating the impact of different sequence databases on metaproteome analysis: Insights from a lab-assembled microbial mixture. PLOS ONE. 2013;8:e82981. doi: 10.1371/journal.pone.0082981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zaiou M, Nizet V, Gallo RL. Antimicrobial and protease inhibitory functions of the human cathelicidin (hCAP18/LL-37) prosequence. J Invest Dermatol. 2003;120:810–816. doi: 10.1046/j.1523-1747.2003.12132.x. [DOI] [PubMed] [Google Scholar]
- 60.Paule SM, Pasquariello AC, Hacek DM, Fisher AG, Thomson RB, Jr, Kaul KL, Peterson LR. Direct detection of Staphylococcus aureus from adult and neonate nasal swab specimens using real-time polymerase chain reaction. J Mol Diagn. 2004;6:191–196. doi: 10.1016/S1525-1578(10)60509-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Iwase T, Hoshina S, Seki K, Shinji H, Masuda S, Mizunoe Y. Rapid identification and specific quantification of Staphylococcus epidermidis by 5′ nuclease real-time polymerase chain reaction with a minor groove binder probe. Diagn Microbiol Infect Dis. 2008;60:217–219. doi: 10.1016/j.diagmicrobio.2007.08.009. [DOI] [PubMed] [Google Scholar]
- 62.Matsuda K, Tsuji H, Asahara T, Matsumoto K, Takada T, Nomoto K. Establishment of an analytical system for the human fecal microbiota, based on reverse transcription-quantitative PCR targeting of multicopy rRNA molecules. Appl Environ Microbiol. 2009;75:1961–1969. doi: 10.1128/AEM.01843-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jiang H, Dong H, Zhang G, Yu B, Chapman LR, Fields MW. Microbial diversity in water and sediment of Lake Chaka, an athalassohaline lake in northwestern China. Appl Environ Microbiol. 2006;72:3832–3845. doi: 10.1128/AEM.02869-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig. S1. Analysis of the skin bacterial community by 16S rRNA gene sequencing and its relationship to the capacity to culture S.aureus from the skin.
Fig. S2. Abundance of live Staphylococcus and Staphylococcus-specific DNA on the skin of non-AD and AD subjects.
Fig. S3. Specificity of Staphylococcus genus–specific primers for real-time qPCR.
Fig. S4. Anti–S.aureus activity in CoNS is stable over time.
Fig. S5. Effect of vehicle treatment on the survival of S.aureus on ex situ pigskin or live mouse skin.
Fig. S6. MALDI-TOF MS analysis for two AMPs purified from S. hominis.
Fig. S7. Representation of amino acid losses in genome-guided MALDI-TOF/TOF analysis for Sh-lantibiotic-β.
Fig. S8. Skin isolate strains of CoNS exert selective antimicrobial activity against S.aureus.
Fig. S9. Detection of Sh-lantibiotic-α peptide by Western blotting from extracts of skin swabs taken from two non-AD subjects and two AD subjects.
Fig. S10. List of hypothetical antimicrobial genes identified in anti–S.aureus CoNS clones used for AMT.
Fig. S11. List of previously known lantibiotic genes identified in antimicrobial S. epidermidis clones isolated from non-AD skin.
Fig. S12. Study design of AMT therapy for patients with AD.
Fig. S13. Effect of transplantation of antimicrobial CoNS or vehicle on the survival of S.aureus on the skin of five subjects with AD.
Table S1. Clinical characteristics of AD and non-AD subjects.
Table S2. Proportion of CoNS species identified with antimicrobial and nonantimicrobial activity.
Table S3. Clinical characteristics of AD subjects receiving AMT.
Table S4. Sequences of PCR primers.
Table S5. Primary data (provided as an Excel file).