Abstract
The reproductive success of men with mitochondrial disease is to date unreported. We compared the age- and era-adjusted reproductive success of 94 British male patients with mitochondrial disease to that of the UK general male population. The reproductive success of men with mitochondrial disease was 65% of that in the general population (95% confidence interval: 53% 79%), and the effect magnitude was related to the disease severity. This contrasts with the two previous studies finding that the reproductive success of women with mitochondrial DNA disease is not impaired. We conclude that the reproductive health of men with mitochondrial disease merits increased clinical attention.
Introduction
Mitochondrial disorders are a common form of inherited disease in the adult population.1 Previous studies suggest that the reproductive success of women harbouring pathogenic mitochondrial DNA (mtDNA) variants is not impaired.2, 3 However, population genetic theory suggests that male reproduction is particularly vulnerable to the mitochondrial dysfunction related to mtDNA variation,4 although there are no reports on the reproductive success of men with mitochondrial disorders. We investigated the reproductive success of men with mitochondrial disease in a large patient cohort.
Materials and methods
We assessed the reproductive success among adult men with genetically defined mitochondrial disease, either due to function-affecting variants in mtDNA or nuclear DNA (nDNA), identified from the Medical Research Council Mitochondrial Disease Patient Cohort Study UK database in January 2015. Described mtDNA and nDNA variants have been submitted to the publicly available ClinVar (https://www.ncbi.nlm.nih.gov/clinvar/) database.
Data collection
Eligibility criteria were a definite genetic mitochondrial disease diagnosis, UK residency, minimal age of 18 years, and ability to give informed consent. Subjects were asked to complete a questionnaire interrogating the number, sex and birth year of children, and factors associated with male fertility and the likelihood of having children, including level of education, marital status, diagnosis of diabetes mellitus, and smoking. The patients were asked not to report adopted or foster children. The patient recruitment flow chart is depicted in Supplementary Figure S1.
Statistical analyses
We calculated age-specific fertility rates and the total reproductive rate to estimate reproductive success. General population male fertility data were obtained from ONS.5 These are based on birth registration data.6 For births solely registered by mother (6.6% of all births in England and Wales in 2007), the father’s age was estimated using ONS jointly registered births outside marriage data. We included only men born in or after 1944 (N=101), the earliest year with ONS data. There were seven pairs of known-related individuals, among these 101 were present at the time of analysis, and we randomly excluded the other from each pair, leaving 94 unrelated individuals for the final analyses. For calculations of fertility after 2013, the rates for 2013 were used (extrapolation justified by the stable population fertility).
We calculated the fertility across the age range 15–64 years, analogous to the general fertility rate (live births per 1000 male population aged 15–64 years) reported by the ONS.5 The person-years contributed by each subject were summed and formed the denominator by which the total number of births to all fathers over the period was divided. We calculated fertility rates for the entire fertile age range and in 5-year age groups. A directly comparable reference fertility rate was calculated using the 2013 ONS data.5 The cohort and reference fertility rates were tested for difference with an exact Poisson two-sided test, using the ‘exactci’ package in R.7 We report the relative ratio of the cohort and ONS fertility rates with 95% Poisson confidence intervals.
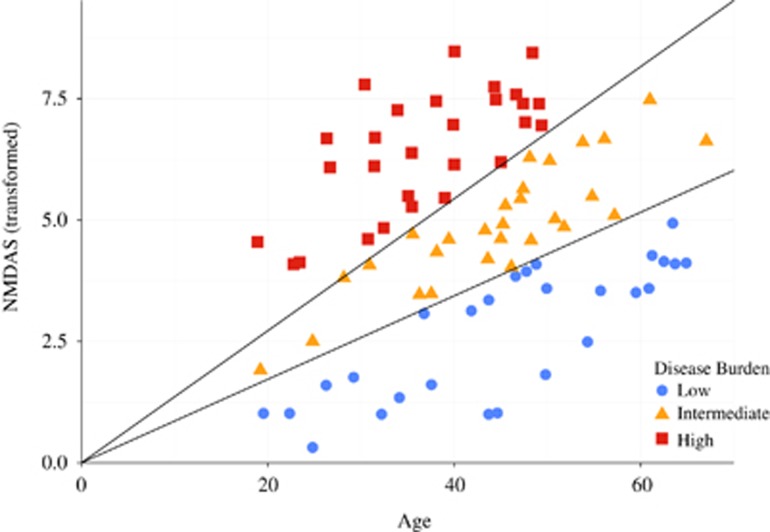
To examine lifetime fertility and age-specific fertility by disease state, the subjects were grouped into low/medium/high-disease burden categories. The clinical severity of mitochondrial disease was assessed with the Newcastle Mitochondrial Disease Adult Scale (NMDAS) score,8 available for 82 of the 94 subjects. Box–Cox9 identified the square root as the optimal transformation of the NMDAS score (NMDAST) in regression against age. The cohort divided into three equally sized groups segregate the subjects by NMDAS score progression (Figure 1).
Figure 1.
Segregation of patients by NMDAS score into three groups according to disease burden. Both age and the square root-transformed NMDAS score are used to define the disease burden groups.
To estimate the effect of education, the cohort was segregated into those who had completed upper secondary/pre-university education or university (N=41) and those who had not (N=53). For diabetes, we considered those currently with diabetes mellitus (N=27) vs not (N=67). For smoking, we considered ever (current or former) (N=45) vs never smokers (N=49). For each group comparison, the reproductive success was calculated relative to an age- and era-weighted general population fertility rate. For statistical comparison of fertility between groups, a two-sample exact Poisson test for two proportions was used. For each test, the age and era-weighted general population fertility rates for each group were examined to verify that there were no substantive differences between the group fertility profiles.
The effect of marital status was determined by estimating the proportion of those currently single (N=26) compared with all other statuses (married, cohabiting, separated, divorced, and widowed) (N=68). We evaluated whether the proportion of single males in the cohort was different from an age-weighted proportion as calculated from the ONS general population data.10
Ethical considerations
This study was approved and performed under the ethical guidelines issued by our institution for clinical studies. Written informed consent was obtained from all subjects.
Results
The reproductive success of men with mitochondrial disease was 0.65 (95% CI: 0.53; 0.79) relative to general UK male population (Table 1), and similar in mtDNA and nDNA subgroups (Table 2; Supplementary Table S1). Men with more severe disease had lower relative reproductive success than those more mildly affected (Figure 1; Table 1), which was not observed in women harbouring mtDNA variants.3
Table 1. Age-specific and overall relative reproductive success of men with mitochondrial disease.
| Disease burden | No. patients | Age low | Age high | Births | Person-years | GFR pop | Ratio | Ratio L | Ratio U | P value |
|---|---|---|---|---|---|---|---|---|---|---|
| All patients | ||||||||||
| 15 | 19 | 4 | 470 | 0.0108 | 0.7860 | 0.2142 | 2.0125 | 0.8500 | ||
| 20 | 24 | 17 | 468 | 0.0631 | 0.5755 | 0.3353 | 0.9215 | 0.0180 | ||
| 25 | 29 | 22 | 452 | 0.0996 | 0.4885 | 0.3062 | 0.7396 | 2.22E−04 | ||
| 30 | 34 | 36 | 403 | 0.0950 | 0.9402 | 0.6585 | 1.3017 | 0.7918 | ||
| 35 | 39 | 13 | 368 | 0.0608 | 0.5806 | 0.3092 | 0.9929 | 0.0464 | ||
| 40 | 44 | 5 | 325 | 0.0267 | 0.5755 | 0.1869 | 1.3431 | 0.2720 | ||
| 45 | 49 | 0 | 269 | 0.0091 | 0.0000 | 0.0000 | 1.5011 | 0.1713 | ||
| 50 | 54 | 0 | 167 | 0.0024 | 0.0000 | 0.0000 | 9.2989 | 1 | ||
| 55 | 59 | 0 | 110 | 0.0010 | 0.0000 | 0.0000 | 33.1035 | 1 | ||
| 60 | 64 | 0 | 79 | 0.0005 | 0.0000 | 0.0000 | 97.5063 | 1 | ||
| 97 | 3111 | 0.0480 | 0.6495 | 0.5267 | 0.7923 | 6.2 × 10−06 | ||||
| By disease burden | ||||||||||
| Low | 27 | 32 | 923 | 0.046 | 0.7544 | 0.516 | 1.065 | 0.1183 | ||
| Medium | 27 | 29 | 978 | 0.0478 | 0.6203 | 0.4155 | 0.8909 | 0.0073 | ||
| High | 28 | 23 | 712 | 0.0528 | 0.6118 | 0.3878 | 0.918 | 0.0146 | ||
| By genotype | ||||||||||
| mtDNA | 73 | 76 | 2364 | 0.0489 | 0.6569 | 0.5176 | 0.8222 | 0.0001 | ||
| nDNA | 21 | 21 | 747 | 0.0450 | 0.6252 | 0.3870 | 0.9557 | 0.0275 | ||
Abbreviations: GFR, general fertility rate; mtDNA, mitochondrial DNA; nDNA, nuclear DNA.
Table 2. Genetic causes of mitochondrial disease among the study participants.
| Nuclear DNA | mtDNA | ||||||
|---|---|---|---|---|---|---|---|
| Gene | Function | N | Prevalence | Variant | Effect | N | Prevalence |
| All | 21 | 2.9 × 10−5 | All | 73 | 9.6 × 10−5 | ||
| POLG | mtDNA polymerase | 6 | 0.3 × 10−5 | m.3243A>G | tRNA leucine 1 | 36 | 3.5 × 10−5 |
| OPA1 | Mitochondrial dynamics | 6 | 0.4 × 10−5 | Single mtDNA deletion | 20 | 1.5 × 10−5 | |
| TWNK | mtDNA helicase | 5 | 0.7 × 10−5 | m.8344A>G | tRNA lysine | 6 | 0.2 × 10−5 |
| GFER | Mitochondrial biogenesis | 1 | n/a | m.7989T>C | COX subunit | 1 | n/a |
| RRM2B | mtDNA maintenance | 1 | 0.2 × 10−5 | m.8839G>C | ATP synthase F0 subunit 6 | 1 | n/a |
| SPG7 | Mitochondrial protein paraplegin | 1 | 0.8 × 10−5 | m.8993T>C | ATP synthase F0 subunit 6 | 1 | 0.1 × 10−5 |
| YARS2 | Mitochondrial tRNA synthetase | 1 | n/a | m.8993T>G | ATP synthase F0 subunit 6 | 1 | 0.05 × 10−5 |
| m.9185T>C | ATP synthase F0 subunit 6 | 1 | n/a | ||||
| m.12271T>C | tRNA leucine 2 | 1 | n/a | ||||
| m.12283A>G | tRNA leucine 2 | 1 | 0.05 × 10−5 | ||||
| m.13051A>G | Complex I subunit | 1 | n/a | ||||
| m.13513G>A | Complex I subunit | 1 | n/a | ||||
| m.14674T>C | tRNA glutamic acid | 1 | n/a | ||||
| m.14709T>C | tRNA glutamic acid | 1 | 0.1 × 10−5 |
Abbreviations: ATP, adenosine triphosphate; COX, cytochrome c oxidase; mtDNA, mitochondrial DNA; prevalence, prevalence among affected adults (Gorman et al.1); tRNA, transfer RNA.
The proportion of single males in our cohort was 24.8% (95% CI: 17.1–34.3%), whereas the proportion of single males in the general population, weighted according to the age and birth year profile of our cohort, was 16.3% (P=0.027). The marriage status-adjusted general fertility rate showed that when accounting for marital status, the reproductive success of our cohort was 0.73 (95% CI: 0.59; 0.88) relative to the general UK male population (Table 1).
The disease severity as reflected in the NMDAS score did not differ between men who participated in the study and those who did not (P=0.269). Because the low/medium/high NMDAS groups were not age-matched, the effect of disease severity on reproductive success could not be statistically evaluated by direct comparison. However, each group could be compared with the age and era-matched general population and these comparisons showed an inverse relationship between reproductive success and disease severity (Figure 1; Table 1). Among patients in this study, smoking status (P=1.000), education level (P=0.186), and having diabetes mellitus (P=0.806), were not associated with the number of recorded births.
Discussion
We found that the reproductive success of men with mitochondrial disease was significantly compromised. This contrasts with the previous reports that reproductive success in women with inherited mitochondrial DNA variants does not differ from general population,3 using the same methodology exploiting UK population data to calculate age and era-adjusted relative fertility rates.
We have not employed Bonferroni or any other multiple testing adjustment in our analyses for well-documented reasons, and particularly, as we are testing a priori hypotheses;11 however, the number of statistical tests used do open up the potential for more frequent type I statistical errors, and this should be taken into account while interpreting the findings.
The element of uncertainty attached to the assignment of children to their biological fathers is common knowledge; however, there is no reason to suggest that this would differ between our data and the ONS data, or in regard to reporting children born outside of marriage or children from previous unions.12
The severity of mitochondrial disease as reflected by the NMDAS scores did not differ between study participants and those choosing not to participate, lending support to the contention that the present results were not due to sampling bias related to disease severity. As 14 out of 76 (18%) patient exclusions were due to cognitive impairment, which is very likely to decrease reproductive success, our results may in fact under-estimate the effect.
The high proportion of singles among men with mitochondrial disease may reflect their relative social isolation and suggests that they are less attractive mating partners. Marriage status is not an ideal proxy for being in a relationship: however, the adjusted fertility rate suggests that the decreased reproductive success observed cannot be simply explained by decreased likelihood of marriage.
Evolutionary theory provides an overarching framework for understanding sex differences in different physiological and disease phenotypes. Sex-specific natural selection favours traits that increase the survival or reproductive success of individuals of the respective sex. As mtDNA is passed only from mother to offspring, adaptive evolution of mtDNA is not affected by any possible male-specific phenotypes.4 Previous studies suggest that mtDNA haplogroups are associated with asthenozoospermia among men with fertility problems,13 and high levels of the mtDNA variant affecting function m.3243A>G are associated with low sperm motility.14 In future, it would be of interest to correlate reproductive success with mtDNA variant heteroplasmy levels in various tissues. The observed sex difference in reproductive success, in the context of mitochondrial dysfunction, indicates existing animal data on sexual dimorphism with regard to mitochondrial function may be relevant to human biology.15
The focus of previous research on reproductive issues in mitochondrial disease has largely been on women. We suggest that the reproductive health of men with mitochondrial disease merits increased clinical attention. Future work will address the detailed mechanisms leading to impaired reproductive success in men with mitochondrial disease.
Acknowledgments
This work was supported by the Wellcome Trust. MHM is supported by the Sigrid Jusélius Foundation. We wish to express our gratitude to nurse specialist Sister Catherine Feeney, Sister Alexandra Bright, and Ms Rebekah Taylor for their expert assistance.
Footnotes
Supplementary Information accompanies this paper on European Journal of Human Genetics website (http://www.nature.com/ejhg)
The authors declare no conflict of interest.
Supplementary Material
References
- Gorman GS, Schaefer AM, Ng Y et al: Prevalence of nuclear and mitochondrial DNA mutations related to adult mitochondrial disease. Ann Neurol 2015; 77: 753–759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moilanen JS, Majamaa K: Relative fitness of carriers of the mitochondrial DNA mutation 3243A>G. Eur J Hum Genet 2001; 9: 59–62. [DOI] [PubMed] [Google Scholar]
- Gorman GS, Grady JP, Ng Y et al: Mitochondrial donation—how many women could benefit? N Engl J Med 2015; 372: 885–887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank SA, Hurst LD: Mitochondria and male disease. Nature 1996; 383: 224. [DOI] [PubMed] [Google Scholar]
- Office for National Statistics (UK). Further parental characteristics, England and Wales, 2013 http://www.ons.gov.uk/ons/rel/vsob1/further-parental-characteristics—england-and-wales/2013/index.html. Accessed 5 April 2017.
- Office for National Statistics (UK). Population Trends, No. 136, 2009 http://www.ons.gov.uk/ons/rel/population-trends-rd/population-trends/no—136—summer-2009/index.html. Accessed 5 April 2017.
- R Core Team. R: A Language and Environment for Statistical Computing [Computer Program]. Vienna, Austria: R Foundation for Statistical Computing, 2014. http://www.r-project.org. Accessed 5 April 2017.
- Schaefer AM, Phoenix C, Elson JL, McFarland R, Chinnery PF, Turnbull DM: Mitochondrial disease in adults: a scale to monitor progression and treatment. Neurology 2006; 66: 1932–1934. [DOI] [PubMed] [Google Scholar]
- Box GE, Cox DR: An analysis of transformations. J R Stat Soc B Stat Methodol 1964; 26: 211–252. [Google Scholar]
- Office for National Statistics (UK). ONS General Lifestyle Survey 2011. Chapter 5: Marriage and cohabitation, 2011. http://webarchive.nationalarchives.gov.uk/20160105160709/http://ons.gov.uk/ons/rel/ghs/general-lifestyle-survey/2011/rpt-chapter-5.html. Accessed 5 April 2017.
- Perneger TV: What's wrong with Bonferroni adjustments. BMJ 1998; 316: 1236–1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyner K, Peters HE, Hynes K, Sikora A, Taber JR, Rendall MS: The quality of male fertility data in major US surveys. Demography 2012; 49: 101–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz-Pesini E, Lapeña AC, Díez-Sánchez C et al: Human mtDNA haplogroups associated with high or reduced spermatozoa motility. Am J Hum Genet 2000; 67: 682–696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiropoulos J, Turnbull DM, Chinnery PF: Can mitochondrial DNA mutations cause sperm dysfunction? Mol Hum Reprod 2002; 8: 719–721. [DOI] [PubMed] [Google Scholar]
- Innocenti P, Morrow EH, Dowling DK: Experimental evidence supports a sex-specific selective sieve in mitochondrial genome evolution. Science 2011; 332: 845–848. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.