Abstract
Background
In the pathophysiology of implant failure, metal ions and inflammation-driven osteoclasts (OC) play a crucial role. The aim of this study was to investigate whether vanadium (V) ions induce differentiation of monocytic OC precursors into osteoresorptive multinucleated cells. In addition, the influence of V ions on the activation and function of in vitro generated OC was observed.
Methods
Human monocytes and osteoclasts were isolated from peripheral blood monocytic cells (PBMCs). Exposition with increasing concentrations (0–3 μM) of V4+/V5+ ions for 7 days followed. Assessment of OC differentiation, cell viability, and resorptional ability was performed by standard colorimetric cell viability assay 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenil)-2H-tetrazolium (MTS), tartrate-resistant acid phosphatase (TRAP) expression, and functional resorption assays on bone slides during a period of 21 days.
Results
No significant differences were noted between V4+/V5+ ions (p > 0.05). MTS showed significant reduction in cellular viability by V concentrations above 3 μM (p < 0.05). V concentrations above 0.5 μM showed negative effects on OC activation/differentiation. Higher V concentrations showed negative effects on resorptive function (all p < 0.05) without affecting cell viability. V4+/V5+ concentrations below 3 μM have negative effects on OC differentiation/function without affecting cell survival.
Conclusion
Vanadium-containing implants may reduce implant failure rate by influencing osteoclast activity at the bone-implant interface. V-ligand complexes might offer new treatment options by accumulating in the bone.
1. Introduction
Metal-based implants have become essential and very successful treatment tools in dental and orthopedic trauma surgery. Implant failure rates are usually well below 5% for the first two years. The rates may increase up to 10–20% under local inflammatory conditions, including periodontitis and allergic reactions against the metal implant [1]. To date, surgical revision is the gold standard in treating implant failure. With an increasingly ageing population, implant failure with the consequent need of revision surgery will have a clinical and economic significance in the future.
Aseptic loosening (AL) is the leading cause of failure of total joint arthroplasty [2]. Aside from the well investigated and recognized role of wear debris (in the nanometer range) in the initiation and development of AL, over the last years, there has been increasing evidence that involvement of metal ions released by biocorrosion might influence AL by enhancing osteolysis and decreasing osseointegration [3, 4].
Various metal alloys used in metal implants contain vanadium (V), which gives the alloy favorable physicochemical and mechanical properties. Several in vitro and in vivo studies have investigated the bioactivity of V and different V compounds. It has been demonstrated that V compounds have an insulin and growth factor mimicking action [5, 6]. In addition, V compounds largely influence phosphatases, a group of enzymes mainly associated with the cell membrane [7]. Phosphatases, specifically the tartrate-resistant acid phosphatase (TRAP), play a paramount role in osteoclastic activities. Inhibiting TRAP might reduce the risk of AL. In vivo studies carried out to investigate the biodistribution of V compounds showed that V accumulates predominantly in the bone, kidney, spleen, and liver after 24 h of administration [8]. A recently published study has also shown that V compounds are able to regulate osteoblastic growth [9]. In particular, V ions released by biocorrosion from metal implants and accumulated in the bone could exert specific effects on bone turnover and more specifically on OC. Little is known about the potential biological effects of V ions on OC. In this study, we investigate the hypothesis that V4+ and V5+ ions may interfere with OC differentiation and activation using a well-established in vitro human OC model, as well as an in vitro bone resorption model.
2. Methods
2.1. Isolation of Peripheral Blood Monocytic Cells and Generation of Osteoclasts
The study protocol established by Lionetto et al. was used in the experiments [10]. Ficoll-gradient centrifugation (Amersham Biosciences, Uppsala, Sweden) was used to isolate pooled peripheral blood monocytic cells (PBMCs) from buffy coats of healthy blood donors. RPMI-1640 plus GlutaMAX™ medium (RPMI) (Gibco/Invitrogen, Auckland, New Zealand) supplemented with 5% human serum and 1% antibiotics (10,000 units/mL penicillin G sodium, 10,000 μg/mL streptomycin sulfate, and 25 μg/mL amphotericin B (Gibco standard medium)) was the culture medium for PBMCs at 37°C (humified, 5% CO2, in 25 cm2 tissue culture flasks (Sarstedt)). The nonadherent PBMCs were discarded and the adherent PBMCs were washed twice using 0.1 M phosphate-buffered saline (PBS, pH = 7.2), scraped off, and resuspended in standard medium after 1 h of culturing. These cells were used as monocytic cells (MCs) as described below.
To generate the OC, cell cultures of adherent PBMCs were supplemented with OC differentiation cytokines (10 ng/mL recombinant human macrophage-colony stimulating factor (hM-CSF) and 10 ng/mL recombinant human receptor activator of NF-κB ligand (hRANKL) (ReproTech, Rocky Hill, NJ, USA)).
2.2. Cell Culture Conditions
The MC, MC supplemented with OC differentiation cytokines, and mature OC were subsequently exposed to increasing concentrations (0 to 3 μM) of bis(maltolato)oxovanadium(IV) (BMOV) V4+ or oxidized bis(maltolato)oxovanadium(V) (oxBMOV) V5+ ions for 7 days. Cell viability was assessed using 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) colorimetric assay (Promega, Madison, WI, USA). The assays were repeated four times for each experimental condition, and the mean optical light density was recorded using a microplate reader (Labsystems, Helsinki, Finland) at an absorbance of 492 nm.
2.3. Detection of TRAP Using ELF97 and Flow Cytometry
The endogenous tartrate-resistant acid phosphatase (TRAP) activity was detected by using the phosphatase substrate ELF97 (Molecular Probes, Eugene, OR, USA) as described previously [11], to assess OC differentiation in the presence of the different concentrations of V4+ or V5+ ions. Cell fixation was performed after 7 days, using a culture medium containing 1% paraformaldehyde. After being washed twice with distilled water, the fixed cells were subsequently incubated with 200 μM ELF97 in a 110 mM acetate buffer staining solution for a period of 20 min. Cells were characterized for expression of surface markers by using fluorescence-labeled mouse monoclonal antibodies binding specifically to human HLA-DR (phycoerythrin (PE) fluorescent labeled; Becton Dickinson Biosciences, San Jose, CA, USA) and CD45 (PerCP fluorescent labeled; Becton Dickinson Biosciences). To account for the background signal and autofluorescence, isotype antibodies and unstained controls were used.
A FACS Vantage Cell Sorter with a UV laser was used for sample analysis. Approximately 105 gated cells were analyzed. The ELF97 was excited at 350 nm, and the TRAP-related signal was collected using a 530/30 band pass filter (515–545 nm). The PE and PerCP fluorochromes were excited using a 488 nm laser. The signal was collected using a 575/26 and a 675/20 band pass filter. The data was further analyzed using the Flowjo v8.5.3 software package (Treestar, Ashland, OR, USA).
2.4. Osteoclast Resorptive Function on the Dentine Slices
Evidence of OC function and bone resorptive activity was assessed using lacunar resorption assays on the dentine slices, as previously described [3]. Whale dentine slices (diameter 15 mm, thickness 0.5 mm) were cut from a sperm whale tooth, purchased from Kaempf (Osborne Park, Western Australia, collected prior to 1972, before the ban on whale hunting in Australia). Adherent cells (4 × 105 cells/well) were cultured on dentine slices. The slices were washed with water and left overnight in a 0.25% ammonium hydroxide solution after a culturing period of 21 days to remove all cellular material. Staining was performed with 1% toluidine blue dye in 1% sodium borate for 10 min. A Nikon Inverted Microscope Eclipse TE 300 (Nikon Instruments, Melville, NY, USA) and a Nikon CCD digital camera were used for documentation. The whole surface of the slice was examined. The DXM 1200F-ACT-1 image processing software (Nikon) was used to analyze the images. A dark-blue excavation on the dentine surface with a clear rim of unchanged and unstained original surface located between neighboring pits was defined as a resorption pit. The extent of resorption was determined from the number of pits formed and by calculating the total planar surface area of the resorption pits. The diameter of 30 pits/slice was measured and the mean value used to calculate the surface of the number of pits counted. The number of pits was counted using a single blinded observer.
2.5. Calculations and Statistical Analysis
The data was analyzed using the SPSS for Windows software package (v15.0; SPSS Inc., Chicago, IL). Independent sample t-tests were conducted to determine whether the mean ion concentrations were significantly greater than the minimum detection limits. A one-way ANOVA test was used to test the differences in the mean in the expression of the TRAP, as well as differences in the mean number of resorption pits and the surface area across the various cell cultures. A p value of p < 0.05 was considered to be statistically significant.
3. Results
3.1. Cell Viability
Nontoxic concentrations of V4+ and V5+ ions that did not greatly decrease the MC and OC viability were defined by MTS colorimetric assays. A significant reduction in MC and in vitro generated OC was seen at concentrations of V4+ or V5+ ions greater than 3 μM. No significant change in MC and OC viability was found when using concentrations up to 3 μM, indicating the toxic concentrations for MC and OC (Figure 1). Toxicity of V4+ and V5+ concentrations above 3 μM was additionally confirmed by Annexin-5 staining. No significant difference was found between V4+ and V5+ ions (data not shown) (p > 0.05). Subsequently, increasing concentrations (0 to 3 μM of V4+ or V5+) were defined as standard conditions for the experiments.
Figure 1.
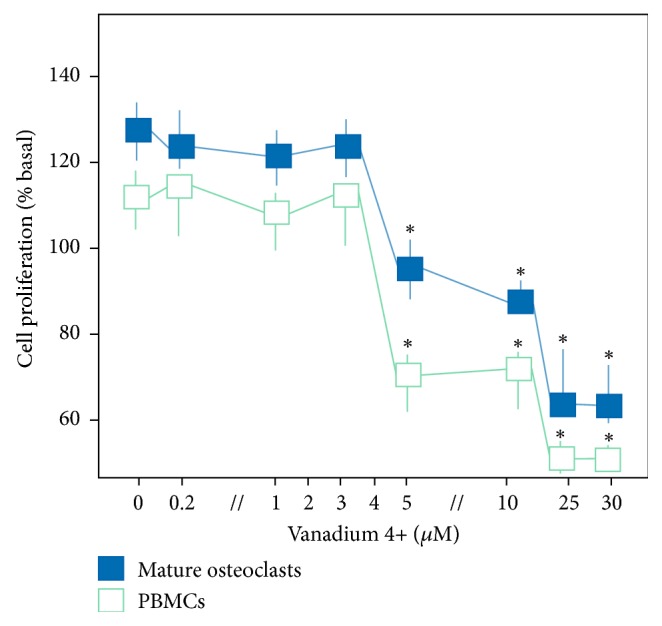
Assessment of cell viability. Cell proliferation (percentage of basal proliferation) of monocytic cells and osteoclasts under increasing concentration of vanadium 4+ ions. No differences were noted in the same concentrations of vanadium 5+ ions (p > 0.05). ∗p < 0.001.
3.2. Detection of TRAP Using ELF97 and Flow Cytometry (FACS)
All OC cultures were TRAP-positive while all the MC cultures supplemented with OC differentiation cytokines showed a significantly reduced expression of TRAP, indicating OC differentiation inhibition in the presence of V4+ or V5+ for all the concentrations (Figure 2). All cultures of MC exposed only to V4+ or V5+ (no supplementation with OC differentiation cytokines) showed no expression of TRAP. Quantitative FACS analysis showed that TRAP expression was significantly decreased in the OC cultures exposed to increasing concentrations of V4+ or V5+ ions compared with OC cultured without any V ions (p < 0.05) (Figure 2).
Figure 2.
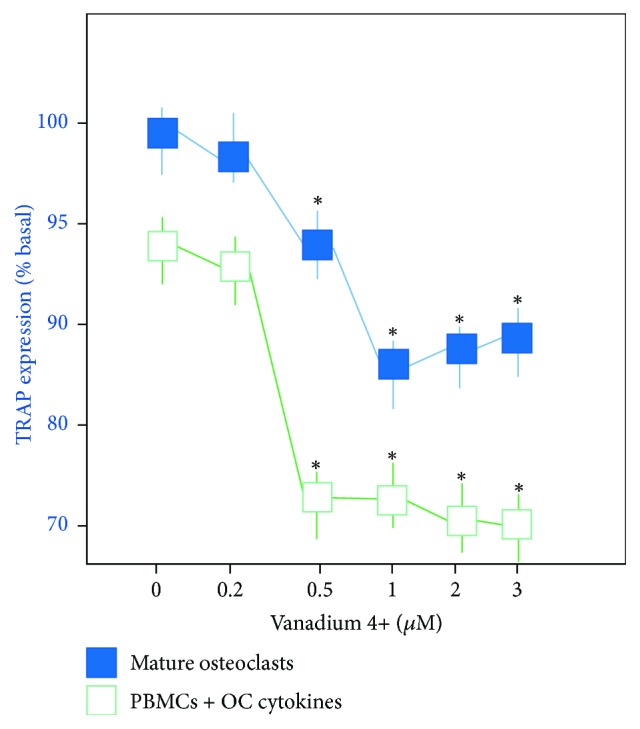
Quantitative analysis of TRAP expression by osteoclasts and monocytes supplemented with osteoclast differentiation cytokines in the presence of increasing concentrations (0 to 3 μM) of V4+ ions. Mean fluorescence intensity (MFI) indicates the average TRAP expression per cell. The MFI value indicates relative TRAP expression when compared with the control (MFI = 121). No differences were noted between the same concentrations of V4+ and V5+ ions (p > 0.05) (∗p < 0.05).
3.3. Qualitative and Quantitative Assessment of Osteoclastic Resorptive Function on the Dentine Slices
The resorptive function was assessed on the dentin slides after the discovery that V4+ and V5+ ions had an essential effect on OC differentiation for the selected concentrations.
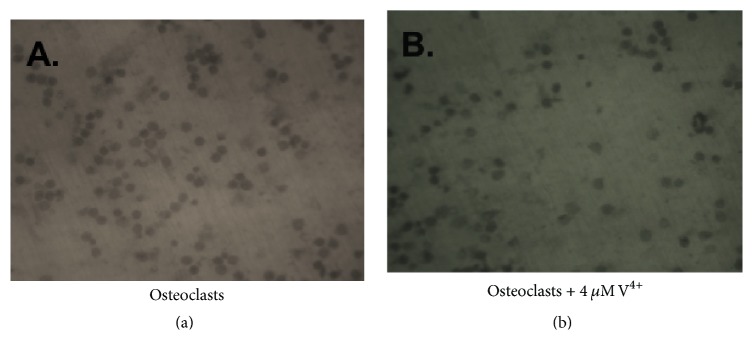
OC were cultured with and without V4+ or V5+ ions on dentin slides for a period of 21 days. The extent of lacunar resorption was analyzed (Figure 3). Between the different conditions, qualitative and quantitative differences in the patterns of absorption were seen. Compared to the control samples, exposition to increased V4+ or V5+ ions led to smaller sized resorption pits, while the resorption pits of untreated OC were larger. Osteoclasts incubated with the different concentrations of V4+ or V5+ ions showed a significantly decreased number of resorption pits and resorption area of the total dentine surface compared to untreated OC (Figure 4). Monocytes, as a negative control, showed only very few resorption pits (22 ± 3.9 pits/mm2) with small dentine area resorption (0.28%, Figure 4). Untreated OC showed an average of 801 ± 13.2 resorption pits/mm2 with a dentine resorption area of 10.19% (p < 0.001). Regarding the resorption area, no difference was detected between V4+ and V5+ ions (p > 0.05). Resorption features were not seen on the dentine slices incubated without cells.
Figure 3.
Assessment of bone resorption. The images show a representative example of resorption pits (dark spots) on dentine slides after 21 days of incubation under different culture conditions. (a) Untreated osteoclasts. (b) Osteoclasts + 4 μM vanadium 4+ ions.
Figure 4.
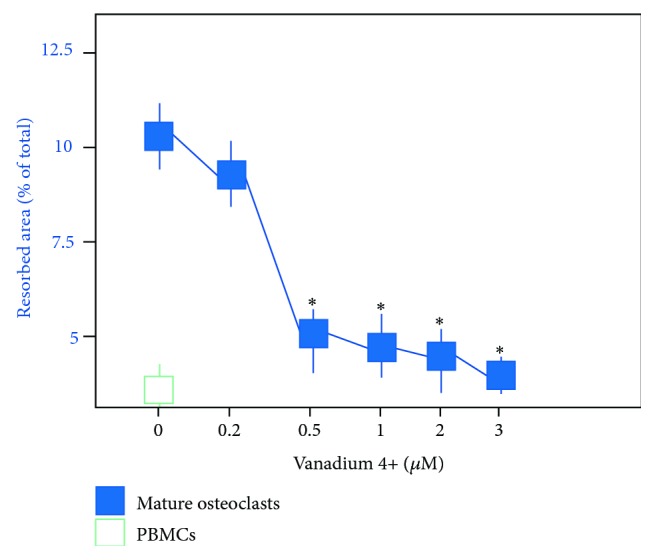
Histogram representing the mean area (percentage of total area ± SE) of resorption on dentine slides after 21 days in culture under different conditions. Note the significant decrease of dentin resorption by osteoclasts exposed to V4+ ions. No differences were noted between the same concentrations of V4+ and V5+ ions (p > 0.05) (∗p > 0.05).
4. Discussion
The hypothesis that V4+ and V5+ ions are able to interfere with OC differentiation and bone resorptive activity was clearly supported in this study. The hypothesis was based on previous data showing the strong effects of different metal ions on bone metabolism as well as the tendency of V ions/compounds to accumulate in bone cells [3, 4, 8].
The integrity of the skeleton as well as the osseous integration of intercortical metal implants (e.g., total joint arthroplasty) requires the regulated activity of bone-forming cells (osteoblasts) and bone-resorbing cells (OC). Numerous growth factors and cytokines with pro- and antiapoptotic effects are produced in the bone, regulating the balance between osteoformation and resorption [9, 11, 12]. Thus, imbalanced bone cell activity results in severe bone alterations and tissue loss leading in the worst cases to implant loosening without osseointegration. The term “osseointegration” is defined as incorporation of nonvital components in a predictive and reliable way into vital bone. This anchorage mechanism is specifically important if cementless implants are used [13, 14]. In addition, altered TRAP levels in bone pathologies like osteoporosis might compromise osseointegration [15].
Metal ions have been shown to affect bone turnover through a variety of both direct and indirect mechanisms. It has been already demonstrated that Ti4+ ions act directly on bone cells by enhancing OC differentiation [3]. In line with this, Co2+ and Cr3+ ions affected human osteoblasts by inhibiting the release of alkaline phosphatase, enhancing apoptosis, RANKL, and osteoprotegerin (OPG) expression, with enhanced osteoclastogenesis and osteolysis [16, 17]. Furthermore, metal ions induce elevated proinflammatory cytokine secretion such as interleukin- (IL-) 1α/β, IL-6, and tumor necrosis factor- (TNF-) α [4, 18]. TNF-α has a direct influence on OC precursors, whereas IL-6 and IL-1α/β act indirectly by increasing osteoblast RANKL and M-CSF expression, which directly drives osteoclastogenesis [4]. Several studies have reported the bioactivity of vanadate, vanadyl, and different V4+ complexes on bone metabolism [19–22]. Low concentrations (<25 μM) of V4+ compounds influence the proliferation and differentiation of osteoblast-like cells in culture, while higher concentrations have inhibitory and cytotoxic effects on osteoblasts. V compounds stimulate the protein tyrosine phosphorylation through the inhibition of protein tyrosine phosphatases (PTPases) [23]. The presented results confirm the cytotoxicity of high concentrations (>3 μM) of V4+ and V5+ ions on MC and OC in culture. This study demonstrates the inhibitory action of V4+ and V5+ ions at concentration < 3 μM on OC differentiation and activity in vitro. This is based on several observations: (a) significantly decreased expression of TRAP in cultures of MC supplemented with OC differentiation cytokines and exposed to V4+ or V5+ ions, (b) significantly decreased TRAP expression in cell cultures of mature and functional OC exposed to V4+ or V5+ ions, and (c) significantly reduced bone resorption by OC exposed to V4+ or V5+.
5. Conclusion
V-containing implants may reduce OC activity at the bone-implant interface and thereby decrease bone resorption and ultimately the rate of implant failure. Primary osseointegration is supported by suppressing local TRAP activity. Additionally, V complex development with different ligands might be an alternative strategy of use in skeletal tissue engineering especially when considering their tendency to accumulate in bone.
Acknowledgments
This study was supported by the International Team for Implantology (ITI) Center, Peter Merian-Weg 10, Basel, Switzerland.
Abbreviations
- AL:
Aseptic loosening
- ARCBS:
Australian Red Cross Blood Service
- Co:
Cobalt
- Cr:
Chrome
- MC:
Monocytic cells
- MTS:
3-(4,5-Dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenil)-2H-tetrazolium
- μM:
Micromol
- nm:
Nanometer
- OC:
Osteoclasts
- PBMC:
Peripheral blood monocytic cell
- PE:
Phycoerythrin
- TRAP:
Tartrate-resistant acid phosphatase
- V:
Vanadium
- BMOV:
Bis(maltolato)oxovanadium(IV)
- oxBMOV:
Oxidized bis(maltolato)oxovanadium(V).
Ethical Approval
Ethical approval for the use of human blood cells was granted by the Ethics Committee of the University of Western Australia (Agreement no. RA/4/1/1069) and the Australian Red Cross Blood Service (ARCBS), Perth, WA, Australia (Agreement no. 06-03WA-25).
Disclosure
The results of this study were presented at the 100th Annual Congress of the Swiss Society of Surgery.
Conflicts of Interest
All the authors confirm that there are no conflicts of interest.
References
- 1.Harris W. H. Osteolysis and particle disease in hip replacement: A review. Acta Orthopaedica. 1994;65(1):113–123. doi: 10.3109/17453679408993734. [DOI] [PubMed] [Google Scholar]
- 2.Cadosch D., Chan E., Gautschi O. P., Filgueira L. Metal is not inert: role of metal ions released by biocorrosion in aseptic loosening—current concepts. Journal of Biomedical Materials Research A. 2009;91(4):1252–1262. doi: 10.1002/jbm.a.32625. [DOI] [PubMed] [Google Scholar]
- 3.Cadosch D., Chan E., Gautschi O. P., Meagher J., Zellweger R., Filgueira L. Titanium IV ions induced human osteoclast differentiation and enhanced bone resorption in vitro. Journal of Biomedical Materials Research - Part A. 2009;91(1):29–36. doi: 10.1002/jbm.a.32183. [DOI] [PubMed] [Google Scholar]
- 4.Cadosch D., Sutanto M., Chan E., et al. Titanium uptake, induction of RANK-L expression, and enhanced proliferation of human T-lymphocytes. Journal of Orthopaedic Research. 2010;28(3):341–347. doi: 10.1002/jor.21013. [DOI] [PubMed] [Google Scholar]
- 5.Srivastava A. K., Mehdi M. Z. Insulino-mimetic and anti-diabetic effects of vanadium compounds. Diabetic Medicine. 2005;22(1):2–13. doi: 10.1111/j.1464-5491.2004.01381.x. [DOI] [PubMed] [Google Scholar]
- 6.Thompson K., Orvig C. Vanadium compounds in the treatment of diabetes. Metal Ions in Biological Systems. 2004;42:221–252. doi: 10.1201/9780203913703.ch7. [DOI] [PubMed] [Google Scholar]
- 7.Crans D. C. Antidiabetic, Chemical, and Physical Properties of Organic Vanadates as Presumed Transition-State Inhibitors for Phosphatases. Journal of Organic Chemistry. 2015;80(24):11899–11915. doi: 10.1021/acs.joc.5b02229. [DOI] [PubMed] [Google Scholar]
- 8.Cortizo A. M., Molinuevo M. S., Barrio D. A., Bruzzone L. Osteogenic activity of vanadyl(IV)-ascorbate complex: evaluation of its mechanism of action. International Journal of Biochemistry and Cell Biology. 2006;38(7):1171–1180. doi: 10.1016/j.biocel.2005.12.007. [DOI] [PubMed] [Google Scholar]
- 9.Tuan R. S. Biology of developmental and regenerative skeletogenesis. Clinical Orthopaedics and Related Research. 2004;427:S105–S117. doi: 10.1097/01.blo.0000143560.41767.ee. [DOI] [PubMed] [Google Scholar]
- 10.Lionetto S., Little A., Moriceau G., et al. Pharmacological blocking of the osteoclastic biocorrosion of surgical stainless steel in vitro. Journal of Biomedical Materials Research - Part A. 2013;101(4):991–997. doi: 10.1002/jbm.a.34402. [DOI] [PubMed] [Google Scholar]
- 11.Manolagas S. C. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocrine Reviews. 2000;21(2):115–137. doi: 10.1210/er.21.2.115. [DOI] [PubMed] [Google Scholar]
- 12.Kousteni S., Chen J.-R., Bellido T., et al. Reversal of bone loss in mice by nongenotropic signaling of sex steroids. Science. 2002;298(5594):843–846. doi: 10.1126/science.1074935. [DOI] [PubMed] [Google Scholar]
- 13.Brånemark R., Brånemark P-I., Rydevik B., Myers R. R. Osseointegration in skeletal reconstruction and rehabilitation. Journal of Rehabilitation Research & Development. 2001;38:1–4. [PubMed] [Google Scholar]
- 14.Piarulli G., Rossi A., Zatti G. Osseointegration in the elderly. Aging Clinical and Experimental Research. 2013;25(S1):59–60. doi: 10.1007/s40520-013-0103-0. [DOI] [PubMed] [Google Scholar]
- 15.Resmini G., Migliaccio S., Carbonare L. D., et al. Differential characteristics of bone quality and bone turnover biochemical markers in patients with hip fragility fractures and hip osteoarthritis: Results of a clinical pilot study. Aging Clinical and Experimental Research. 2011;23(2):99–105. doi: 10.1007/BF03351075. [DOI] [PubMed] [Google Scholar]
- 16.Dai M., Zhou T., Xiong H., Zou W., Zhan P., Fu W. Effect of metal ions Co2+ and Cr3+ on osteoblast apoptosis, cell cycle distribution, and secretion of alkaline phosphatase. Journal of Reparative and Reconstructive Surgery. 2011;25(1):56–60. [PubMed] [Google Scholar]
- 17.Dai M., Yuan X., Fan H., Cheng M., Ai J. Expression of receptor activator of nuclear factor kappaB ligand and osteoprotegerin of mice osteoblast induced by metal ions. Journal of Reparative and Reconstructive Surgery. 2010;24(3):292–295. [PubMed] [Google Scholar]
- 18.Wang J. Y., Wicklund B. H., Gustilo R. B., Tsukayama D. T. Titanium, chromium and cobalt ions modulate the release of bone-associated cytokines by human monocytes/macrophages in vitro. Biomaterials. 1996;17(23):2233–2240. doi: 10.1016/0142-9612(96)00072-5. [DOI] [PubMed] [Google Scholar]
- 19.Barrio D., Braziunas M., Etcheverry S., Cortizo A. Maltol complexes of vanadium (IV) and (V) Regulate in vitro alkaline phosphatase activity and osteoblast-like cell growth. Journal of Trace Elements in Medicine and Biology. 1997;11(2):110–115. doi: 10.1016/S0946-672X(97)80035-1. [DOI] [PubMed] [Google Scholar]
- 20.Etcheverry S. B., Barrio D. A., Cortizo A. M., Williams P. A. M. Three new vanadyl(IV) complexes with non-steroidal anti-inflammatory drugs (Ibuprofen, Naproxen and Tolmetin). Bioactivity on osteoblast-like cells in culture. Journal of Inorganic Biochemistry. 2002;88(1):94–100. doi: 10.1016/S0162-0134(01)00368-3. [DOI] [PubMed] [Google Scholar]
- 21.Etcheverry S. B., Williams P. A. M., Sálice V. C., Barrio D. A., Ferrer E. G., Cortizo A. M. Biochemical properties and mechanism of action of a vanadyl(IV) - Aspirin complex on bone cell lines in culture. BioMetals. 2002;15(1):37–49. doi: 10.1023/A:1013183910203. [DOI] [PubMed] [Google Scholar]
- 22.Barrio D. A., Williams P. A. M., Cortizo A. M., Etcheverry S. B. Synthesis of a new vanadyl(IV) complex with trehalose (TreVO): insulin-mimetic activities in osteoblast-like cells in culture. Journal of Biological Inorganic Chemistry. 2003:459–68. doi: 10.1007/s00775-002-0438-z. [DOI] [PubMed] [Google Scholar]
- 23.Tracey A. S., Gresser M. J. Interaction of vanadate with phenol and tyrosine: implications for the effects of vanadate on systems regulated by tyrosine phosphorylation. Proceedings of the National Academy of Sciences. 1986;83(3):609–613. doi: 10.1073/pnas.83.3.609. [DOI] [PMC free article] [PubMed] [Google Scholar]