Abstract
Poor solubility of milk protein concentrates (MPCs) is a key deterrent factor in their wider applications in the food industry as compared to other protein-rich dried products such as casein, caseinates and whey protein concentrates and isolates. Apart from the processing factors, the protein content of a MPC also decides its solubility. Solubility is a pre-requisite property of MPCs on which its other functional properties are majorly depended. Further, there is a confusion about the term MPC itself in the literature. An attempt has been made to describe MPC and provide an understanding on the manufacture of MPCs. Further, mechanisms of insolubility, factors affecting solubility of MPCs and an insight into the recently evolved strategies for overcoming the challenges related to their poor heat stability and solubility have been reviewed. Potential applications of MPC to be utilized as a novel ingredient in food industry are also outlined.
Keywords: Ultrafiltration, Diafiltration, Spray drying, Solubility, Functional properties, Milk protein concentrate
Introduction
Milk protein concentrates (MPCs) and Milk protein isolates (MPIs) are two protein rich milk powders that differ from each other on the basis of their protein content (dry matter basis). Milk protein powders with a protein content below 90% on dry matter basis (DM basis) are called ‘MPCs’, while those having a protein content ≥90%, are known as MPIs. Productions of both MPC and MPI in research laboratories were started around 1990. According to Kelly (2011), earlier MPC would have been known as ‘co-precipitates’, a term which was initially used by Everette (1952) for the coagulated mass of total milk proteins (casein and whey protein) obtained after their ‘co-precipitation’ at high temperature (≥90 °C) under acidic conditions with or without addition of CaCl2 in the milk. The concept and product could not attract global interest due to limited functional properties, especially solubility. MPC and MPI are also dairy based milk protein powders that differ significantly from skimmed milk powder (SMP) and whole milk powder (WMP) in terms of their proximate composition and physicochemical and functional properties. High-quality milk protein is concentrated in the form of MPC, in a ratio as it originally occurs in fresh milk. With concentrated milk proteins, MPCs have significantly low lactose and mineral content as compared to their counter parts like SMP and WMP. Ultrafiltration (UF) is a pressure-driven membrane process employed to concentrate milk proteins in the retentate, while removing lactose and soluble salts into the permeate. Proteins in MPC remain in almost undenatured state, retaining native functional properties owing to the application of moderate temperatures.
Definite standards for proper identification of MPC still do not exist in the food industry worldwide (Patel and Patel 2014). Mistry (2013) reported a broad range of compositional contents and functional characteristics of various MPCs. MPCs have been classified as second generation dairy ingredients (Havea 2006; Tong and Smithers 2013) containing protein ranging from 40 to 89% on a DM basis. The present classification of MPCs namely MPC-56, MPC-70 and MPC-85, is similar to the existing classification of whey protein powders the associated number represents the protein content of that particular MPC on a DM basis. Wide variations in the protein content of MPC are still present, but Sikand et al. (2011) broadly categorized these powders into 3 main types: (a) low-protein powder (≤40% protein content), (b) medium-protein powder (60–70% protein content), and, (c) high-protein powder (≥80% protein content). Thus, the protein content of any MPC may range from ≥40 to ≤89%. Most common MPCs are of the type MPC-42, MPC-70, MPC-80, and MPC-85 (Patel and Patel 2014). The protein content of a MPC is inversely proportional to the lactose and mineral (ash) content in other words protein is purified by reducing the lactose and mineral salts contents. As milk protein ingredients, MPCs are being used in several products like cheese, cultured dairy products, nutritional products, protein standardization, recombined dairy products, infant milk formula, ice-cream, dairy-based beverages, sports drinks and health-related products (Alvarez et al. 2005; Fang et al. 2012; Guiziou 2013; McCarthy et al. 2014; Yanjun et al. 2014). A recent review article focuses on functional properties of MPCs (Uluko et al. 2015), this review outlines their market potential, processes involved in their production, heat stability of UF retentates, factors affecting the solubility and different interventions such as physical, chemical and enzymatic methods employed to improve the solubility of MPCs. The aim of this manuscript is to provide an understanding on the manufacture of milk protein concentrates and an insight into the recently evolved strategies for overcoming the challenges related to their heat stability and solubility.
Market potential of milk protein concentrate
MPC is gaining popularity worldwide rapidly. Lagrange et al. (2015) reported that official data for the annual global production of MPC is not available except for the United States of America. The U.S. market size was estimated as 50,000–55,000 metric tons (MT) for MPC-42 and MPC-56, followed by 17,000–18,000 MT for MPC-70, MPC-85, and MPIs (U.S. Dairy Export Council 2012). In the last decade, the demand of MPC has grown rapidly, which was supported by its annual global production. In the year 2000, the worldwide production of MPC was only 40,000 MT that increased to 270,000 MT by the year 2012. U.S. imported about 76,600 MT MPC in the year 2006 to meet its domestic demands against its minimal production. According to Patel and Patel (2014), in the year 2013, U.S. produced about 45,900 MT of MPC, yet they had to import 55.0 MT of MPC, mainly from New Zealand, European Unions and Australia. As per the technical report of U.S. Dairy Export Council and the Dairy Research Institute, MPCs are considered as concentrated and potent source of quality proteins for the enhancement of functional, nutritional and sensorial properties in various food products. According to Guiziou (2013), high protein ingredients market that was earlier dominated by casein, caseinates, WPC and WPI, is presently being driven by MPCs owing to their established nutritional and functional properties. In the year 2014, American Dairy Products Institute (ADPI) and U.S. Dairy Export Council (USDEC) jointly applied for Generally Recognized as Safe (GRAS) notification to utilize MPCs and MPIs as a food constituents for better functional and nutritional properties in an array of food products, except infant formulations (Patel and Patel 2014). Presently, New Zealand is the top producer and exporter of MPCs in the world followed by EU. Application wise, MPC procurement is mainly dominated by processed cheese products. Experts forecasted that along with other milk powders, higher quantity of MPCs will be produced in key manufacturing areas of the world (Dairy Export Council 2012). It has been estimated that production of MPCs will grow more than 40,000 MT by the year 2020 and the same may expand its market volume by displacing casein in specific applications (Lagrange et al. 2015).
Production of milk protein concentrate
MPCs are usually prepared adapting ultrafiltration (UF), diafiltration (DF) followed by optional evaporation of the retentate or skim milk concentrate prior to spray drying. MPCs have relatively more undenatured proteins as these processes do not involve severe heat treatment and pH adjustment. Better stability and good solubility are pre-requisites for many functional properties of high protein powders, however, MPC particularly at higher protein content fail to yield desired solubility and hence other functional properties. The same situation has been reported as a major technological challenge against their wide applications (Yanjun et al. 2014). Widely adopted method of MPC production is presented in Fig. 1a. The various processes used and parameters maintained during the conversion of skimmed milk into MPCs, their impact on solubility of MPCs as well as various technological interventions used to enhance the solubility during the last 25 years has been reviewed in the following sections.
Fig. 1.
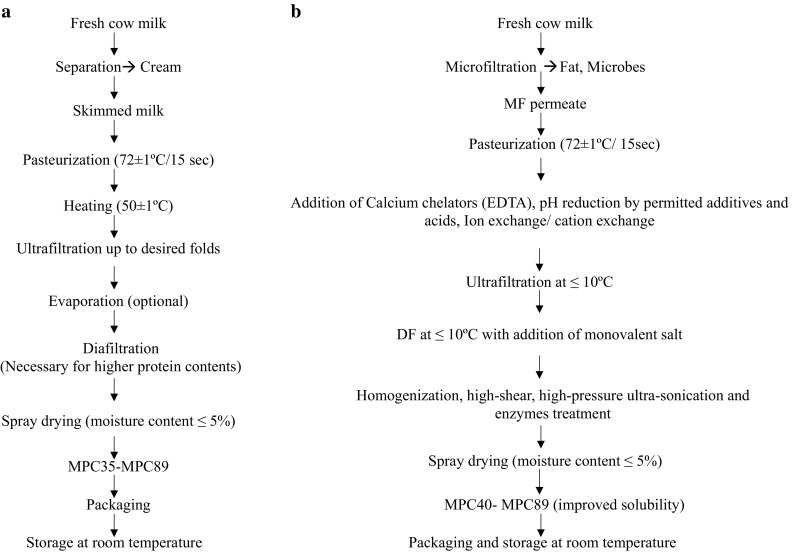
Production of MPCs using a existing method and, b proposed methods including different interventions for solubility improvement
Raw material and heat treatment
The use of high quality skimmed milk that is having a low total microbial and spore counts is essential to produce superior quality MPC (Mistry 2013). Either fresh defatted milk or reconstituted non-fat milk, mostly pasteurized using high temperature short time (HTST) system, has been used (Augustin et al. 2012; Mao et al. 2012; Crowley et al. 2014; Luo et al. 2015) to produce MPC. Udabage et al. (2012) thermized skim milk samples at 65 °C/15 s and studied high pressure induced changes in milk and in the concentrate made out of such milk. Rajagopalan and Cheryan (1991) suggested the use of microfiltration process for the complete removal of milk fat to produce MPCs and MPIs with excellent functional properties. Cao et al. (2016) reported use of pasteurized skim milk (85 °C/15 s) followed by UF and conventional evaporation. Li and Corredig (2014) heated the UF/DF retentates up to 80 °C/15 min for the first time to study the effect of pH as well as heat treatment on calcium release during acidification process and buffering capacity. It was reported that applied heat treatment did not show any alteration either in buffering capacity or in calcium release on lower concentration factors (1× and 2×). However, it resulted in increased release of calcium in soluble phase at higher concentration factors (4× UF and DF) compared to unheated milk.
Ultrafiltration and diafiltration of skim milk
UF is the most commonly used process for the production of MPCs (Harper 2011). In UF, separation of the solution molecules takes place on the basis of their size, shape as well as their charge and affinity towards the membrane (Bastian et al. 1991; Aimar et al. 1998). Its molecular weight cut-off (MWCO) ranges from 1 to 200 kDa with ~0.01 µm pore size and <10 bar operational pressure (Rosenberg 1995). During skim milk ultrafiltration, low molecular weight substances like water, lactose, soluble salts and vitamins pass through the membrane into permeate, while high molecular weight compounds like fat, if any, and proteins are retained and thus concentrated by UF membrane (Cheryan 1998; Singh 2007; Mistry 2011). Diafiltration (DF) is usually employed to improve the separation ability of the feed material through the addition of suitable solvent. Being universal solvent for the food systems, water is mostly added to retentate for dilution purpose, which dissolves soluble constituents and make them easily permeable through the membrane (Hausmann et al. 2013). DF can be operated in batch and continuous mode using the same UF membrane so as to achieve higher concentration of proteins, which is not possible with UF alone (Smith 2013). DF reduces lactose (<5 g/100 g) and increases protein content in the retentate and hence in the resultant MPC (>65 g/100 g) powder (Guiziou 2013). It is also used to adjust the buffering capacity and mineral content of UF retentate (Ramchandran and Vasiljevic 2013). Recently Cao et al. (2016) used nanofiltration (NF) membrane process for the production of MPC.
Protein content of cow skim milk retentate at 2.11, 3.00, 3.94 and 5.48 UF folds were 4.62, 7.23, 9.23 and 16.00%, respectively (Tripathy 2005). Vora (2008) conducted experiments in a pilot scale tubular UF plant using 50 kDa ZrO2 membrane and concluded that cow skim milk can be economically concentrated by this membrane up to 5.16-fold concentration (~20.70% protein), after that, flux approaches almost zero. Behaviour of UF and DF with skim milk as feed and employed process variables as reported in the literature during the conversion of skim milk into MPCs is shown in Table 1.
Table 1.
Process parameters and behaviour of ultrafiltration and diafiltration during skim milk processing
| Milk type and protein content (%) | UF parameters | DF parameters | Protein in MPC (%) | References | |||||
|---|---|---|---|---|---|---|---|---|---|
| MWCO (kDa) | Temp. (°C) | Folds | Protein (%) | Temp. (°C) | Folds | Protein (%) | |||
| CM, 3.3 | 50 | 50 | NM | 90 | Rajagopalan and Cheryan (1991) | ||||
| CM, 3.2 | 50 | 38 | 5 | 15 | 32 | 6 | 18.9 | 83.9 | Mistry and Hassan (1991) |
| CM, 3.3 | 50 | 35 | 5 | 15 | 30 | 5 | 20 | 83.47 | Mistry and Pulgar (1996) |
| BM, 3.96 | NM | 50 | 4.15 | NM | 67.50 | Patel and Mistry (1997) | |||
| CM, 3.46 | 10 | 10 | NM | NM | 56, 70, 85 | Bhaskar et al. (2003) | |||
| CM, NM | 20 | 44 | NM | 20% TS | NM | 62 | El-Samragy et al. (1993) | ||
| CM, 3.5 | 10 | 50 | 5 | 22.9% TS | NM | NM | 24.9% TS | 80–83 | Augustin et al. (2012) |
| CM, 3.43 | 10 | 5.9–19.7 | 6 | 13.61 | NM | 6 | 16.18 | 80.9 | Gualco (2010) |
| CM, 3.4 | NM | 50 | 3 | NM | 50 | 6 | 15.55% TS | 80 | Yanjun et al. (2014) |
| CM, NM | 10-UF 20-NF |
NM | NM | 61.21 | Cao et al. (2016) | ||||
| CM, NM | 20 | 15, 30, 50 | 5 | 15.4–16.4 | NM | NM | Luo et al. (2015) | ||
CM cow milk, BM buffalo milk, TS total solids, NM not mentioned, MWCO molecular weight cut-off, UF ultrafiltration, DF diafiltration, MPC milk protein concentrate
Effect of milk pH, temperature and diafiltration on calcium removal and flux
Milk is usually ultrafiltered either ≤10 °C or in the temperature range of 40–50 °C (Kelly 2011, Guiziou 2013; Liu et al. 2014). Membrane flux and temperature are inversely proportional to the viscosity (Cheryan 1998). Solubility of calcium is higher at lower temperature as higher calcium content passes into permeate at lower temperature, however it reduces the flux drastically (Knights 2013). Protein and calcium phosphate are the two well-known membrane foulants. Protein concentration during ultrafiltration at higher temperature precipitates more calcium phosphate in membrane pores and also promotes bacterial growth in the retentate (Rice 2008; Walstra et al. 2006). At 4 °C, maximum dissociation of casein micelles was observed in 5.2–5.3 pH range, but the same was highest in 5.5–5.6 pH range during ultrafiltration at 50 °C (Dalgleish and Law 1988). Moreover, significant reduction in natural pH of milk, markedly reduces membrane flux, but increases minerals removal due to dissolution of casein micelles (St-Gelais et al. 1991, Mao et al. 2012; Sikand et al. 2013). This is supported by the work of Luo et al. (2015), in which skim milk was ultrafiltered at three temperatures i.e. 15, 30 or 50 °C. It was observed that higher calcium content was passed into permeate at 15 °C as compared to 30 or 50 °C and resulted in less fouling of the membrane. Moreover, flux reduction rate was equal at 15 and 30 °C but it decreased rapidly at 50 °C for initial 3 h and then become constant.
Combined effect of temperature, acidification and diafiltration on flux and composition of skim milk retentate and permeate
St-Gelais et al. (1991) studied combined effects of temperature, acidification and DF on the composition of skim milk retentate and permeate. They prepared different samples such as (a) T-50: control, skim milk sample stored and ultrafiltered at 50 °C; (b) T-4: stored and ultrafiltered at 4 °C; (c) TA-50: stored at 4 °C, pH adjusted to 5.6 using HCl and ultrafiltered at 50 °C and; (d) TA-4: stored and ultrafiltered at 4 °C, followed by pH adjustment to 5.3. Diafiltered samples were prepared by the reconstitution of respective UF retentates (T-50D, T-4D, T-A50D, and T-A4D) with deionized water of the desired temperature. All the UF and DF samples were concentrated up to 5× concentration. Their research revealed that calcium content and buffering capacity of 5× skim milk retentate could be modified by adjusting the pH of acidification, temperature and the UF process. Removal of calcium was high when the milk was ultrafiltered at 4 °C, diafiltered with acidification at 50 or 4 °C but the permeate flux particularly on these runs was drastically reduced and decreased the effective plant capacity.
Eshpari et al. (2014) added 3.25 g/L glucono-δ-lactone (GDL) to reduce the pH of milk to 6.0 before its UF and DF. They produced MPC-65 employing UF whereas MPC-80 using combination of UF and DF from the skim milk having reduced pH (6), while control samples were manufactured using normal skim milk having natural pH (6.6). They observed that partial acidification of milk, significantly decreased the calcium content of MPC-80 from 1.84 ± 0.03 to 1.59 ± 0.03 g per 100 g of powder without affecting its chemical composition, particle size and particle density but increased its solubility as desired.
The degree of concentration by UF is bound to influence the composition of retentate. Proximate composition of cow and buffalo skimmed milks and their retentates has been shown in Table 2 (Kumar 2011). It can be seen from the table that the number of folds of increase in fat and protein contents of retentates is similar to that of the number of folds of concentration by ultrafiltration. However, as one can expect, with an increase in the degree of concentration by UF, the lactose content decreased. The increased calcium contents of the retentates was attributed to concentration of colloidal calcium present in skimmed milk during UF process.
Table 2.
Composition of cow and buffalo skim milks and retentates
Source: Kumar (2011)
| Constituents (%) | Cow skim milk | 5× UF retentate | Buffalo skim milk | 4× UF retentate |
|---|---|---|---|---|
| Total solids | 8.27 ± 0.25 | 20.60 ± 0.66 | 9.93 ± 0.58 | 23.63 ± 0.30 |
| Fat | 0.01 ± 0.01 | 0.05 ± 0.01 | 0.01 ± 0.01 | 0.04 ± 0.01 |
| Proteins | 3.14 ± 0.26 | 14.94 ± 0.39 | 4.20 ± 0.29 | 17.31 ± 0.26 |
| Lactose | 4.50 ± 0.36 | 3.94 ± 0.38 | 4.95 ± 0.25 | 4.20 ± 0.18 |
| Total ash | 0.63 ± 0.07 | 1.60 ± 0.13 | 0.75 ± 0.06 | 1.99 ± 0.09 |
| Calcium | 0.17 ± 0.01 | 0.62 ± 0.03 | 0.22 ± 0.03 | 0.70 ± 0.14 |
Mean ± S.E. (n = 7)
Improvement in heat stability of ultra-filtrated retentate
Heat stability commonly known as heat coagulation time (HCT) of liquid milks, evaporated and concentrated milks or retentates in case of membrane processing is the resistance shown by a fixed quantity of the sample (2 mL, in a closed glass tube) in minutes towards its heat coagulation at a particular temperature. It is well documented that the classical heat stability test is performed at 140 °C for liquid milks while the same is conducted at either 130 or 120 °C for concentrated milks and membrane retentates in a hot oil bath. Higher HCT means better suitability of these milks for processing at elevated temperatures owing to adequate proteins stability and vice versa. As compared to liquid and concentrated milks, the HCT-pH profile of UF/DF retentates are different due to shift in delicate ionic equilibrium between serum and micelles of milk during protein concentration (Holt et al. 1981).
Factors affecting heat stability of milk protein concentrate
Ultrafiltration
UF of skim milk has been reported to reduce the HCT of retentates. Comparative studies on the heat stability of cow skim milk concentrates (18.4% TS, at 130 °C) prepared in conventional rotary evaporator at 40 °C and the retentate produced by UF at 50 °C were conducted by Sweetsur and Muir (1980). It was reported that heat stability decreased with increase in concentration of both the samples. Below 18.4% TS, heat stability of both samples were similar, however above 18.4% TS level, the heat stability of UF concentrate was two times higher than that of conventional concentrate. In a series of experiments, it was observed that as the TS levels were increased from 18 to 25%, differences between the heat stabilities of both samples further increased. Effect of forewarming at 90 °C/10 min on the HCT-pH profiles of these samples showed that forewarming doubled the heat stability of the conventional concentrate, but had little effect on the stability of the UF concentrate which was ascribed to its significantly lower salt content (Sweetsur and Muir 1980).
Diafiltration
DF was used to enhance the heat stability of cow skim milk UF retentate by Sweetsur and Muir (1985). Significant increase in heat stability of the retentate was observed that ranged between 30 and 70%. Using higher water to concentrate ratio for the DF of fat containing concentrate, an increase in HCT was reported compared to its control value (29 min) with 42, 52 and 66 min, for 1:1, 2:1 and 4:1 water to concentrate ratio, respectively. Eshpari et al. (2014) used skim milk (pH ~6.6) to produce control samples of MPC-65 and MPC-80 as well as experimental samples using ultrafiltration and diafiltration, respectively. Reconstituted sample (5% wt/wt solids) produced from only ultrafiltration showed better heat stability (107 min) compared to the sample produced by diafiltration (2 min).
Samples of MPC-65 and MPC-80 were produced from normal milk (pH ~6.6) and glucono-δ-lactone (GDL) added (3.25 g/L, pH ~6.0) milk employing ultrafiltration and diafiltration, respectively. Reconstituted samples (5% wt/wt solids) of MPC-65 and MPC-80 produced from normal milk showed better heat stability of 107 and 2.40 min, respectively compared to samples produced from GDL added milk (pH ~6.0) with HCT values of only 0.5 and 0.2 min (Eshpari et al. 2014). Although, when compared at same pH (6.9), HCT values of GDL added and control MPC samples were at par with each other at 120 °C.
Addition of stabilizing salts
Effect of different stabilizing salts such as 200 mg/100 mL mixture of sodium phosphate/disodium phosphate (NaH2PO4/Na2HPO4, 2:1 w/w) and similar amount of trisodium citrate addition on the heat stability of UF retentate containing 20.9% TS subjected to either forewarming (90 °C/10 min) or no forewarming was studied by Sweetsur and Muir (1980). Heat stability of control samples (without forewarming and no added stabilizing salts) had maximum heat stability in acidic pH range. Both stabilizing salts increased heat stability of no forewarming samples but failed to increase heat stability of forewarmed sample. Tripathy (2005) and Vora (2008) reported that addition of 0.5% mixture of monosodium and disodium phosphate in 2:1 (w/w) ratio improved heat stability of fat containing UF retentate from 14 to 68 min. Heat stability of reconstituted casein micelle was determined by Le Ray et al. (1998) by the addition of different stabilizing salts like sodium chloride (NaCl), calcium chloride (CaCl2), and magnesium chloride (MgCl2) at the rate of 4.15, 3.77, 2.12 mol/kg, respectively; di-sodium and tri-sodium citrate at pH 7.4; at the rate 1.25 mol/kg and NaH2PO4 and Na2HPO4 at pH 7.4; 0.45 mol/kg. The reconstituted casein micelles were heat treated at 95 °C/for 10, 20 and 30 min. It was concluded that while the addition of CaCl2, and MgCl2 exhibited detrimental effects, but addition of NaCl, sodium citrate and sodium phosphate significantly increased the heat stability of casein micelles.
Spray drying of milk protein concentrate
MPCs have been manufactured using single and multi-stage spray driers equipped with pressure or rotary atomizers. Inlet and outlet air temperatures were also widely varied as shown in Table 3. Singh (2007) reported that several factors like inlet air temperature, degree of concentration, feed temperature, size of droplets and temperature of the air-powder mixture exiting from the drier, increases the protein denaturation and aggregation. Singh and Newstead (1992) reported that due to rapidity of spray drying, temperature of milk droplets does not exceed 70 °C until they have lost almost all of their water. At the end of drying, temperature of the droplets reaches to the outlet air temperature, hence, the outlet air temperature is a critical parameter to safe guard the dry milk products from heat damage. Spray drying was reported to result in minimum serum protein denaturation without apparent loss of immunoglobulin G and only 3–7% loss of serum albumin (Oldfield et al. 2005). Further, variation in inlet and outlet air temperatures (200/100 or 160/89 °C) did not have any major effect on the denaturation of serum proteins.
Table 3.
Details of Feed and spray driers used in production of various milk protein concentrates
| Protein % or %TS in feed | Single/multi stage | Atomizer details | Inlet air temp. (°C) | Outlet air temp. (°C) | Evaporation capacity (kg water/h) | References |
|---|---|---|---|---|---|---|
| 18.9% protein | Single | Rotary | 120–125 | 80–85 | 16–18 | Mistry and Hassan (1991) |
| 20% protein | Single | Rotary | 127–130 | 78–86 | NM | Mistry and Pulgar (1996) |
| 16.44% protein | Single | Centrifugal | 180 | 85 | 20–25 | Patel and Mistry (1997) |
| 85% protein DM basis | Three | Pressure, 0.8 mm orifice dia | 200 | 90 | NM | Havea (2006) |
| 22.9, 24.9, 31.4% TS | Single | Rotary | 175, 190 | 75, 90 | NM | Augustin et al. (2012) |
| Two | NM | 170 | 70, FBD 70 | NM | ||
| 17.5% TS | Two | Nozzle, 200 bar | 180 | 63, FBD 50 | 80 | Baldwin and Truong (2007) |
| 17% protein | Single | Single stream monodisperse atomizer | 178, 155, 107, 77 | 103, 93, 68, 54 | NM | Fang et al. (2012) |
| 15.55% TS | Single | NM | 130 | 65 | NM | Yanjun et al. (2014) |
Fang et al. (2012) developed direct relation between the spray drying temperature and solubility of mono-disperse MPC particles. Mono-dispersion resulted in particles with similar size and shape due to similar heating history. Dissolution test revealed more reliable results due to elimination of morphological and size differences. These powder particles were generated through a pilot scale microfluidic spray dryer using different inlet air temperatures such as 77, 107, 155 and 178 °C. Solubility and the extent of protein denaturation in MPC was determined using Focused beam reflectance measurement (FBRM) and polyacrylamide gel electrophoresis (PAGE). MPC particles produced at lower inlet air temperatures were spherical, but those produced at high inlet air temperature had deflated morphology, respectively. Increase in inlet air temperature not only increased the protein denaturation, but also decreased the solubility of MPC particles as revealed by FBRM results. Further, results of SDS-PAGE revealed that casein formed the insoluble material rather than whey protein. It was concluded that MPC with better functional properties can be produced using lower spray drying temperatures through the effective contact of sprayed particles with the drying air as well as further moisture removal in second drying stage. Proximate composition of different MPC as reported in the literature is shown in Table 4.
Table 4.
Proximate composition of different types of milk protein concentrates
| MPC | Protein | Lactose | Fat | Ash | Moisture | Ca | Na (g/100 g) | References |
|---|---|---|---|---|---|---|---|---|
| MPC 35 | 35.7, 35.4 | 53.0, 49.6 | 0. 50, 3.5 | 7.7, 8.1 | 3.6, 3.4 | 1.24, – | 0.045, – | Jimenez-Flores and Kosikowski (1986) and Crowley et al. (2014) |
| MPC50 | 49.8, 49.9 | 38.0, 35.8 | 0.53, 2.7 | 7.7, 7.8 | 4.5, 3.8 | 1.64 | 0.034 | Jimenez-Flores and Kosikowski (1986) and Crowley et al. (2014) |
| MPC56 | 57.1 | 30.1 | 1.3 | 7.7 | 3.8 | – | – | Jimenez-Flores and Kosikowski (1986) and Singh (2007) |
| MPC 60 | 56.4, 60.8 | 31.9, 24.5 | 0.51, 3 | 7.8, 7.7 | 3.9, 4 | 1.85, – | 0.028, – | Jimenez-Flores and Kosikowski (1986) and Crowley et al. (2014) |
| MPC 65 | 64.4, 65.45 | 24.0, 19.5 | 0.50, 2.38 | 7.8, 8.1, | 3.9, 3.5 | 2.00, 1.43 | 0.025, – | Jimenez-Flores and Kosikowski (1986) and Eshpari et al. (2014) |
| MPC 70 | 70.0, 68.8, 70 | 17.0, 18, 16, | 1.4, 1.6, 2 | 7.2, 8, 8 | 7.2, 3.6 4 | Singh (2007), Crowley et al. (2014) and Boer (2014) | ||
| MPC75 | 75 | 19.5 | 1.5 | 7.5 | 5.5 | Augustin et al. (2011) | ||
| MPC 80 | 75, 80, 80.9, 81.4, 79.1, 80, 80, | –, –, 0.31, –, 6.4, 6.5, 6 | 1.59, 1.35, 4.16, 1.40, 2.2, 2, 2 | 6.21, 7.42, 6.9, 7.8, 7.7, 6.5, 8 | 4.29, 4.42, 5.2, 4.30, 4.6, 5, 4 | –, –, 1.07 mg/ml, – | –, –, 0.108, – | Sikand et al. (2012, 2013), McCarthy et al. (2014), Crowley et al. (2014), Augustin et al. (2011) and Boer (2014) |
| MPC85 | 84.8, 84.8–85.6, 85, 82.4, 87.8, 84 | 4.6, 3.2– 3.8, 3, 4, 3.8, 1.4 |
1.7, 1–1.3, 1, 1.6, 1.9 | 7.3, 2.6–3.1, 7, 7.3, 7.5 | –, .8–4.2, 4, 4.7, 6.3, 4.8 | 2.30 | – | Anema et al. (2006), Havea (2006), Singh (2007), Mimouni et al. (2009, 2010) and Crowley et al. (2014) |
| MPC88 | 88 | 0.74 | 2.27 | 7.05 | 5.33 | – | – | Mistry (2002) |
| MPC90 | 85.9 | 0.8 | 1.9 | 7.6 | 4.2 | Crowley et al. (2014) |
Solubility of milk protein concentrate
Solubility is a key functional property of protein-rich powders and the same is interrelated with other functional properties like foaming, emulsification, gelling and so on. Moreover, UF, DF and spray drying, which are main processes of MPC production, have been reported to alter the salt equilibrium between colloidal and soluble phases of the protein stabilization system that may ultimately induce a detrimental impact on the milk proteins environment (Mistry and Hassan 1991; Singh 2007). Such alteration may adversely affect the important functional properties like solubility. MPC powders with higher protein content were reported to have poor solubility restricting their use in a number of potential food applications (De Castro-Morel and Harper 2002; Fang et al. 2012). De Castro-Morel and Harper (2002) collected 37 samples of different MPCs from 10 countries and examined for their basic functional properties and reported that correlation between protein content and solubility, viscosity or foaming was not observed in majority of the samples. The observed variation in the functional properties of different MPC samples, particularly in solubility was identified as the biggest hurdle in capturing the global market potential. Further, a study conducted by NIZO group on 32 commercial MPC samples, collected from international market clearly pointed that the poor solubility of MPC-80 continued to be a problem (Huppertz et al. 2010). Solubility, either in true solution or colloidal dispersion, is a pre-requisite to obtain optimum functional performance of components in foods (Damodaran 1996). Poor solubility of MPC negatively affects other functional properties (Baldwin 2010). Singh (2011) reported that different functional properties of MPC like solubility, emulsification, foaming, water binding, viscosity, gelling, heat and acid stability and freeze thaw stability are almost at par with the functional properties of calcium caseinate (except gelation) but poor than that of sodium caseinate, WPC and WPI. Therefore, improving the solubility of MPC powders remains a key challenge to improve its functionality and use.
Mechanism of insolubility development
As compared to sodium caseinate, WPC and WPI, higher proportion of milk solids remains undissolved during the reconstitution of MPC in water at 20 °C. This ‘insoluble’ portion can be easily seen at the bottom of the container as sediment. Many researchers have proposed different mechanisms responsible for MPC insolubility, which includes (a) network of cross-linked (αs–β–Cn) proteins was developed on the surface of the MPC powders, that acts as a barrier in transport of water and ultimately inhibits the hydration of the MPC particles, (b) movement of residual fat on the surface of powder particle (c) and slow release of casein micelles from dispersed powder due to increased cross linking (Mimouni et at. 2010). McKenna (2000) used 6 month old samples of MPC-85, which were stored at 20 °C to isolate the insoluble material followed by its analysis using microscopic techniques. Comparatively large size particles (~100 mm) were observed in the insoluble fraction, particularly made of casein micelles, which were held together through some protein–protein interactions. Such particles were not segregated even in their dispersion made in warm water (45 °C for 30 min). The development of insoluble fraction in protein rich systems is attributed to the covalent and non-covalent bonds based interactions (Havea et al. 1998). The former consists of inter- and intra-molecular di-sulphide bonds formed through sulphydryl–disulphide interchange or sulphydryl oxidation reactions (Gupta and Reuter 1992; Monahan et al. 1995). Non-covalent interactions includes hydrophobic, hydrogen, ionic and other weak interactions that contribute to the formation of insoluble protein material (Havea et al. 1998). Characterization of protein components as well as the type of inter-protein interactions responsible to form undissolved material in MPC powders manufactured in a pilot plant was carried out using PAGE by Havea (2006). It was observed that quantity of such insoluble material present in MPC samples was not only high but also varied notably. Further, it was reported that hydrophobic association of casein molecules with some contribution from minor whey proteins was the principle reason. The insoluble content continues to increase during the entire storage period of MPC powders. Disulphide-linked aggregates made of beta lactoglobulin (β-lg) and kappa casein (k-casein) did not contribute in the formation of such insoluble material. Baldwin (2010) reported that insolubility reaction can take place on the particle surface, between casein micelles or at the scale of the protein molecules but detailed scientific data for the complete elucidation of insolubility development mechanism are awaited.
Key factors contributing for insolubility of milk protein concentrate
Different processing factors like pH, protein and mineral contents of skim milk, its heating and holding prior to UF beyond pasteurization temperature; temperature and pH of skim milk during UF as well as mode of protein concentration (only UF; UF and DF; UF and evaporation; UF, DF and conventional evaporation) combinedly decides the relative quantity of calcium in the permeate and UF retentate. Moreover, inlet–outlet air temperatures used during spray drying, type of dryer and atomizer, storage conditions (temperature, time, relative humidity) and temperature of water during rehydration of MPC has significant impact on its solubility (Havea 2006; Mimouni et al. 2010; Richard et al. 2012). Optimum temperature to decrease hydration time of MPC was reported to be 50 °C (Fang et al. 2011). Augustin et al. (2012) reported that the solubility also depends on the method of production of MPCs. Further, the increase in TS of ultrafiltered concentrates (23% TS) either by diafiltration up to 25% TS or evaporation to 31% TS, decreased the solubility of MPC powders containing 80–83 g protein per 100 g powder. Subsequent, increase in TS of UF concentrate by classical evaporation (>31% TS), resulted in higher reduction in solubility.
Approaches to enhance solubility of milk protein concentrate
The solubility enhancement of MPC powders is a key challenge as compared to other protein-rich dairy powders (Table 5). It may be improved during production and storage of MPC from skim milk by controlling process parameters like temperature, pH, UF concentration ratio, DF and evaporation employed. Extent of heating to which UF concentrate was treated prior to drying and the conditions of entire spray drying process are vital (De Castro-Morel and Harper 2002, Schuck 2009). For the enhancement of solubility of MPC powders in cold water, different processing interventions were reported that includes the addition of monovalent ions before drying (Carr 2002), application of a cation exchanger (Bhaskar et al. 2003), reduction in pH of skim milk by acidification and its subsequent UF/DF and incorporation of calcium chelators or sequestering substances to reduce the calcium content of UF retentate (Schuck et al. 2002; Bhaskar et al. 2003). Recently, Udabage et al. (2012) reported that subjecting protein-rich concentrate to physical treatments like high hydrostatic pressure (HHP) to modify the protein structures was also results in better solubility of the resultant MPC powders. All these approaches are discussed briefly in the following section.
Table 5.
Key functional properties of different high protein milk based powders.
Source: Singh (2011), reproduced with the permission of Elsevier, License Number:4040051019961
| Functional properties | Caseinates | Whey protein rich products | Milk protein concentrates (MPC) | ||
|---|---|---|---|---|---|
| Sodium | Calcium | Whey protein concentrate (WPC) | Whey protein isolate (WPI) | ||
| Solubility | ××× | × | ××× | ××× | ×× |
| Emulsification | ××× | × | ×× | ×× | × |
| Foaming | ××× | × | ×× | ×× | × |
| Water binding | ××× | × | × | × | × |
| Viscosity | ××× | × | × | × | × |
| Gelation | – | – | ××× | × | – |
| Heat stability | ××× | × | × | × | × |
| Acid stability | × | × | ××× | ××× | × |
| Freeze–thaw stability | ××× | × | × | × | × |
×poor, ××good, ×××excellent
Calcium removal by cation exchanger, acidification and chelating agents
Bhaskar et al. (2003) developed a protocol to produce MPC and MPI (min 70 g protein/100 g powder) in dried or aqueous solution with the removal of calcium in the range of 30–100% as compared to control MPC. For calcium removal, approaches such as (a) adjustment of skim milk pH in 4–12 range using 3.3% citric acid and subjecting it to either cation or in ion exchanger in the sodium and/or potassium form, (b) initially acidification of milk to pH 4.6–6, followed by immediate protein enrichment as well as calcium removal by ultrafiltration or by a combination of ultrafiltration and diafiltration processes, (c) addition of different chelators such as citric acid, EDTA and phosphates or polyphosphates. MPC-85 samples with 33, 50 and 85% calcium reduction were produced and stored for 36 days at 20 and 40 °C. The solubility of these samples were approximately same (100%) at both storage temperatures during the entire storage period.
Another method was also developed to produce soluble MPCs. The method involves decreasing the pH of DF retentate to below 5.8 using H2SO4 and re-subjecting the retentate to DF using demineralized water until the percentage of permeate solids reached to 0.1%. This was followed by adjusting the pH to 6.7 with 5% NaOH solution and subsequent drying to obtain MPC powder. It was reported that the extent of calcium removal was highly affected by the degree of acidification. The depletion of calcium in the end product was 30 and 45% at pH-5.8 and 5.4, respectively. MPCs produced through DF under acidic conditions had 100% solubility. However, the solubility was reported to reduce continuously during storage. In another study conducted by Eshpari et al. (2014), skim milk pH was adjusted to 6 before subjecting to ultrafiltration and diafiltration by the addition of 3.25 g/L GDL and reported an increase in the solubility of MPC-80 due to significant decrease in its calcium content.
Application of high pressure treatment
High pressure processing (HPP) also called as high hydrostatic pressure (HHP) or ultra-high pressure processing (UHP) is an alternative food preservation method in which the food materials that containing water molecules are subjected to pressures in the range of 100–800 MPa with or without packaging, in a closed system to get tailored changes in physico-chemical, sensorial and functional properties of the treated foods. Suitability of high pressure treatment at 10–60 °C temperature and 100–400 MPa applied pressure range was studied by Udabage et al. (2012) to increase the solubility of MPC-85. Skim milk was pasteurized (HTST system) followed by either subjecting it to high pressure and produce UF–DF retentate or producing UF–DF retentate followed by subjecting it to high pressure prior to spray drying. It was reported that up to 77% solubility was obtained in the MPC sample, produced from the concentrate treated with 200 MPa at 40 °C prior to spray drying as compared to control having 66% solubility at 20 °C. Enhancement in the solubility was attributed to the altered surface composition of non-micellar casein in the milk induced by high pressure treatment before drying. Also, better solubility of MPC upon reconstitution in milk compared to water was observed due to higher mineral-salt content present in milk. Reconstitution of MPC was found to improve its solubility at higher temperatures. Moreover, high pressure treatment of protein-rich concentrate before its spray drying is one of the additive free potential option to improve solubility as well as storage stability of both MPCs and MPIs.
Sikand at el. (2012) studied the effect of powder source (MPC-75, and MPC-80), solvent type (water and UF permeate), reconstitution temperature (4 and 37 °C) and homogenization pressure (0 and 13,800 kPa) on the solubility of MPC samples. It was reported that solubility of MPC-80 was observed to be better at 37 °C reconstitution temperature followed by its homogenization at 13,800 kPa that produced smaller particles compared to the sample reconstituted at 37 °C without homogenization.
Application of high-shear treatment
High-shear treatment is a novel method of food processing in which the properties of food are altered by subjecting it to severe shearing. Effect of different high shear treatments as well as drying conditions on the solubility of MPCs were studied by Augustin et al. (2012) using 3 different sources such as (a) fresh skim milk, (b) reconstituted milk produced from commercial SMP and (c) commercial UF concentrate. Initially, skim milk was pasteurized (HTST) followed by its UF (10 kDa membrane) at 50 °C to get 22.9% TS which was further concentrated to 24.9 and 31.4% TS employing DF and conventional evaporation in scraped surface evaporator, respectively. These concentrates were later dried in a single stage spray drier equipped with rotary atomizer using 175–195 and 75–90 °C as inlet outlet air temperatures, respectively. Increase in insolubility index (ml/50 ml) of MPC samples produced from only UF, UF and DF; UF and evaporation, dried at either 170/75 °C or 195/90 °C were 4.9, 5.7; 11, 12; and 13.3, 20.5, respectively. In another part of the study, UF concentrate (~22% TS) from reconstituted pasteurized skim milk was obtained and subjected to three high-shearing treatments namely homogenization (300/100 bar), microfluidization (800 bar) and ultrasonication (24 kHz, 160 ml/min @ 600 W) followed by their spray drying (175/75 °C) to manufacture the respective MPC powders. MPC produced from the same UF concentrate at similar drying conditions without subjecting to any high-shear treatment was considered as control. Homogenization, microfluidization and ultrasonication treatment given to UF, DF retentates prior to spray drying was reported to result that resulted in significant improvement in the nitrogen solubility index (NSI) of MPC-82 from 70.14% (control) to 74.46, 89.52 and 74.69% respectively. Further with MPI-91, improved solubility was observed in the MPCs produced from the UF concentrated microfluidized at 1200 bar employing single stage drying as compared to the other MPCs produced from UF concentrate treated at 400 and 800 bar and dried either in single stage or double stage dryer. They study concluded that microfluidization is a capable physical method to enhance solubility of MPC or MPI powders. However, mode of retentate production and it’s pre-treatment before drying as well as the type of dryer employed, and drying variables were reported to have significant effect on the solubility of MPCs.
Application of ultrasound
High-intensity power ultrasound (power range 10–1000 Wcm2, frequency range 20–1000 kHz), has been reported to physically disrupt the material with promotion of chemical reactions (Mason 1998). In food industry, it has been used for many applications including homogenization and shearing. Yanjun et al. (2014) utilized this particular property of power ultrasound to enhance the solubility of MPC-80. In a batch system, about 750 ml of the retentate (15.50% TS) obtained from the DF process was sonicated using high-intensity power ultrasound for 0.5, 1, 2 and 5 min using high-grade titanium alloy probe (14.5 cm × 5 cm diameter) prior to its spray drying. It was reported that significant increase in the solubility of the MPC-80 was observed. The solubility of treated MPC was reported to increase from 35.78 to 88.30% indicating about 147% increase due to high-intensity power ultrasound when treated for 5 min. It was also concluded by the authors that enhancement in the functionally of MPC is directly proportional to the duration of the power ultrasound pre-treatment of the retentate.
Addition of monovalent salts
Effect of NaCl addition during DF on the solubility, hydrophobicity, and disulfide bonds of MPC-80 powder was studied by Mao et al. (2012). MPC-80 was produced from the retentates obtained after the addition of 0, 50, 100, and 150 mM NaCl. As compared to control (55%), solubility of MPC-80 obtained from 100 and 150 mM NaCl added retentates was found to be completely soluble. DF may result in the modification of surface hydrophobicity, reduction in intermolecular disulphide bonds, and an associated decrease in mean particle size of caseins. Moreover, addition of NaCl could modify the strength of hydrophobic interactions and sulfhydryl-disulfide interchange reactions and thereby affecting the protein aggregation and as a result the solubility of MPCs. In another study, different samples of MPC-80 namely MPC-80, without any salt addition (MPC80-C), with 150 mM KCl (MPC80-K) and 150 mM NaCl (MPC80-Na) salts addition in the UF retentate during its DF process (Sikand et al. 2013). The impact of these salts addition on the solubility and turbidity was studied and also compared the contents of minerals and protein in supernatants of ultra-centrifuged samples with that of control sample. It was reported that lower solubility (53%) was observed in control sample as compared to MPC80-Na or MPC80-K (100%) while the reverse trend was observed for turbidity. Protein and calcium contents were reported to be lower in the supernatants of ultra-centrifuged samples of MPC80-C (2.3%; 0.35 mg mL−1) as compared to MPC80-Na (3.8%; 0.63 mg/mL) and MPC80-K (3.7%; 0.67 mg mL−1).
Enzymatic treatments
Banach et al. (2013) treated MPC-80 with different enzymes to improve its solubility and other functional properties. Using digestive enzymes such as chymotrypsin, trypsin, pepsin and cysteine protease-papain produced two hydrolysates without bitterness were produced by controlling the degree of hydrolysis. Solubility of MPC-80 was improved in the pH range of 4.6–7.0. Treatment of MPC with suitable enzymes in controlled conditions can improve their solubility. Moreover, information related to enzymatic modification particularly of MPC are scanty. A proposed method for the production of MPC with improved solubility has been shown in Fig. 1b.
Combined effect of pH adjustment, diafiltration and homogenization
Meena et al. (2016) reported that pH, HCT and ζ-potential of cow milk-based control (5× UFR, TS ~22.33%); homogenized (5× HUFR, 2000/500 psi); diafiltered (5× Na-K, diafiltered with 75 mM concentration of NaCl and KCl each in 1:1 ratio) and pH adjusted (5× DSP 6.5 pH adjusted with 10% solution of Na2HPO4) retentates were 6.41, 6.41, 6.34, 6.50; 5.45, 1.45, 1.05, 81 min and -0.598, -1.007, -18.500 and -0.568 mV, respectively. The adjustment of pH of 5× HUFR between 6.1-7.0 with 10% solution of NaH2PO4 and Na2HPO4 significantly (P < 0.01) improved its HCT particularly in 6.5–6.7 pH range. These retentates were subsequently spray dried to obtain MPC60-C, MPC60-H, MPC60-Na–K and MPC60-DSP powders and studied for their physiochemical, reconstitution, functional and rheological properties by Meena et al. (2017). The solubility of fresh MPC60-C, MPC60-H and MPC60-Na–K powders was at par with each other, but lower than MPC60-DSP. However, at 25 ± 1 °C, the solubility of treated powders was remained significantly (P < 0.05) higher than control powder after sixty days of storage.
Application of milk protein concentrate as a food ingredient
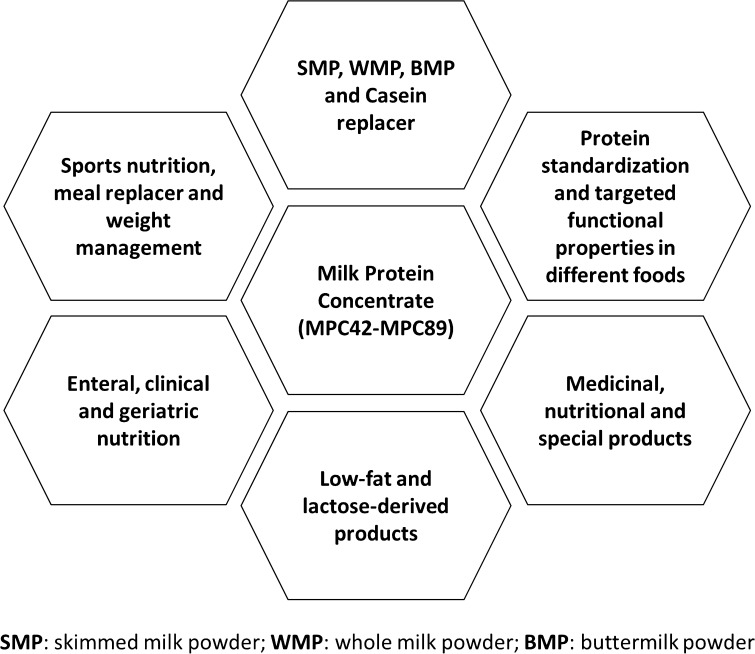
MPCs are produced using ultrafiltration technique without subjecting the milk to harsh processing conditions like high-heat treatment and change in its pH. Thus, the resultant product had casein to whey protein ratio equal to that of whole milk and proteins remains in their native state which enables MPCs to deliver the desired functional properties. Moreover, MPC had less milk fat, lactose and mineral content, which makes it suitable for the application as a quality ingredient in an array of special food products. MPCs are generally converted into powder form (Faka et al. 2009), which are used to meet different aims in several dairy products that take up 25% of the dairy market (Luo et al. 2015). They can be used to replace SMP, WMP, buttermilk powder (BMP) and caseins. Flexibility in their protein and lactose contents led MPCs to be suitable ingredients for dairy and non-dairy applications. As an ingredient, MPC now has wider applications in food industry in the production of beverages, cheese, confectionary, yogurt, ice-cream, high-protein bars, and dairy beverages (Fang et al. 2011; Loveday et al. 2009; Francolino et al. 2010; Giroux et al. 2010). Patel and Patel (2014) reported that MPC can be utilized in protein standardization, reduced fat and lactose product formulations, geriatric nutrition, follow-up formula, medicinal, nutritional and specialty products, development of drinks and food for sports nutrition, meal replacement and weight management. It can also be used in soups and sauces, meat products, bakery products, confectionary, chocolate, coffee whitener, desserts, whipped toppings, recombined milk, enteral and clinical nutrition. Thus, MPC as an ingredient has immense potential to tailor the various properties of existing food products and also has a wide scope for its utilization in next generation foods owing to its unique nutritional, sensorial, physical and functional merits. Various potential uses of MPC powders have been shown in Fig. 2.
Fig. 2.
Potential uses of milk protein concentrates
Future prospectives
The importance and impact of different factors such as the quality of skim milk, conditions of ultrafiltration and diafiltration and their effect on flux, effect of milk acidification, evaporation, addition of minerals and stabilizing salts, sonication, homogenization and high-shearing, dryer type, atomizing systems and drying conditions as well as relation of these parameters to insolubility of MPCs were reviewed. Functional properties of MPCs are crucial for their wider use in food and other industries. Suitable standards for the proper identification of MPCs are required internationally to produce and trade this quality ingredient. Although few mechanisms of insolubility development has been proposed, still full elucidation of the phenomena of insolubility development in protein rich products is required. Simple and innovative scientific interventions like addition of different permissible additives or constituents in the milk or retentate before spray drying to enhance solubility and can easily be adopted by the industries without changing their existing production lines. Potential of different techniques either individually or in combination such as enzyme treatment coupled alternative processing techniques like microwave heating, pulse electric field, infrared heating or ohmic heating, sonication, high-shear and high-pressure processing needs further scientific data and evidences to test their efficacy in solubility enhancement and improvement in other techno-functional properties. The development of precise testing tools for easy comparative analysis is also imperative.
References
- Aimar P, Taddei C, Lafaille JP, Sanchez V. Mass transfer limitations during ultrafiltration of cheese whey with inorganic membranes. J Membr Sci. 1998;38:203. doi: 10.1016/S0376-7388(00)82420-5. [DOI] [Google Scholar]
- Alvarez VB, Wolters CL, Vodovotz Y, Ji T. Physical properties of ice cream containing milk protein concentrates. J Dairy Sci. 2005;88:862–871. doi: 10.3168/jds.S0022-0302(05)72752-1. [DOI] [PubMed] [Google Scholar]
- Anema SG, Pinder DN, Hunter RJ, Hemar Y. Effects of storage temperature on the solubility of milk protein concentrate (MPC85) Food Hydrocoll. 2006;20:386–393. doi: 10.1016/j.foodhyd.2005.03.015. [DOI] [Google Scholar]
- Augustin MA, Oliver CM, Hemar Y. Casein, caseinates and milk protein concentrates. In: Chandan RC, Kalaria A, editors. Dairy ingredients for food processing. 1. Oxford: Wiley; 2011. pp. 161–178. [Google Scholar]
- Augustin MA, Sanguansri P, Williams R, Andrews H. High shear treatment of concentrates and drying conditions influence the solubility of milk protein concentrate powders. J Dairy Res. 2012;79:459–468. doi: 10.1017/S0022029912000489. [DOI] [PubMed] [Google Scholar]
- Baldwin AJ. Insolubility of milk powder products—a mini review. Dairy Sci Technol. 2010;90:169–179. doi: 10.1051/dst/2009056. [DOI] [Google Scholar]
- Baldwin AJ, Truong GNT. Development of insolubility in dehydration of dairy milk powders. Food Bioprod Process. 2007;85:202–208. doi: 10.1205/fbp07008. [DOI] [Google Scholar]
- Banach JC, Lin Z, Lamsal BP. Enzymatic modification of milk protein concentrate and characterization of resulting functional properties. LWT-Food Sci Technol. 2013;54:397–403. doi: 10.1016/j.lwt.2013.06.023. [DOI] [Google Scholar]
- Bastian ED, Collinge SK, Ernstrom CA. Ultrafiltration: partitioning of milk constituents into permeate and retentate. J Dairy Sci. 1991;74:2423. doi: 10.3168/jds.S0022-0302(91)78417-8. [DOI] [Google Scholar]
- Bhaskar GV, Singh H, Blazey ND (2003) Milk protein products and processes. US Patent US 2003/0096036
- Boer RD. From milk by-products to milk ingredients: upgrading the cycle. Hoboken: Blackwell; 2014. [Google Scholar]
- Cao J, Wang G, Wu S, Zhang W, Liu C, Li H, Li Y, Zhang L. Comparison of nanofiltration and evaporation technologies on the storage stability of milk protein concentrates. Dairy Sci Technol. 2016;96:107–121. doi: 10.1007/s13594-015-0244-3. [DOI] [Google Scholar]
- Carr A (2002) Monovalent salt enhances solubility of milk protein concentrate. PCT Patent WO 02/096208
- Cheryan M. Ultrafiltration and microfiltration handbook. Boca Raton: CRC Press; 1998. [Google Scholar]
- Crowley SV, Gazi I, Kelly AL, Huppertz T, O’Mahony JA. Influence of protein concentration on the physical characteristics and flow properties of milk protein concentrate powders. J Food Eng. 2014;135:31–38. doi: 10.1016/j.jfoodeng.2014.03.005. [DOI] [Google Scholar]
- Dalgleish DG, Law AJR. pH-induced dissociation of bovine casein micelles. I. Analysis of liberated caseins. J Dairy Res. 1988;55:529–538. doi: 10.1017/S0022029900033306. [DOI] [Google Scholar]
- Damodaran S. Functional properties. In: Nakai S, Modler HW, editors. Food proteins: properties and characterization. 1. New York: VCH Publishers; 1996. pp. 167–234. [Google Scholar]
- De Castro-Morel M, Harper WJ. Basic functionality of commercial milk protein concentrates. Milchwissenschaft. 2002;57:367–370. [Google Scholar]
- El-Samragy YA, Hansen CL, Mcmahon DJ. Production of ultrafiltered skim milk retentate powder. 1. Composition and physical properties. J Dairy Sci. 1993;76:388–392. doi: 10.3168/jds.S0022-0302(93)77357-9. [DOI] [PubMed] [Google Scholar]
- Eshpari H, Tong PS, Corredig M. Changes in the physical properties, solubility, and heat stability of milk protein concentrates prepared from partially acidified milk. J Dairy Sci. 2014;97:7394–7401. doi: 10.3168/jds.2014-8609. [DOI] [PubMed] [Google Scholar]
- Everette SC (1952) Casein–lactalbumin co-precipitate. US Patent, 2623038-19521223
- Faka M, Lewis MJ, Grandison AS, Hilton D. The effect of free Ca2+ on the heat stability and other characteristic of low-heat skim milk powder. Int Dairy J. 2009;19:386–392. doi: 10.1016/j.idairyj.2008.12.006. [DOI] [Google Scholar]
- Fang Y, Selomulya C, Ainsworth S, Palmer M, Chen XD. On quantifying the dissolution behaviour of milk protein concentrate. Food Hydrocolloid. 2011;25:503–510. doi: 10.1016/j.foodhyd.2010.07.030. [DOI] [Google Scholar]
- Fang Y, Rogers S, Selomulya C, Chen XD. Functionality of milk protein concentrate: effect of spray drying temperature. Biochem Eng J. 2012;62:101–105. doi: 10.1016/j.bej.2011.05.007. [DOI] [Google Scholar]
- Francolino S, Locci F, Ghiglietti R, Iezzi R, Muccheti G. Use of milk protein concentrate to standardize milk composition in Italian citric Mozzerella cheese making. LWT-Food Sci Technol. 2010;43:310–314. doi: 10.1016/j.lwt.2009.08.007. [DOI] [Google Scholar]
- Giroux HJ, Houde J, Britten M. Use of heated milk protein-sugar blends as antioxidant in dairy beverages enriched with linseed oil. LWT-Food Sci Technol. 2010;43:1373–1378. doi: 10.1016/j.lwt.2010.05.001. [DOI] [Google Scholar]
- Gualco SJ (2010) Effect of sodium chloride addition during diafiltration on the solubility of milk protein concentrate. http://digitalcommons.calpoly.edu/theses/427/. Accessed on 19 Feb 2016
- Guiziou GG. Concentrated milk and powders. In: Tamime AY, editor. Membrane processing: dairy and beverage applications. 1. Hoboken: Wiley; 2013. pp. 128–140. [Google Scholar]
- Gupta VK, Reuter H. Protein solubility and gelation behavior of whey protein concentrates prepared by ultrafiltration of sweet cheese whey. Neth Milk Dairy J. 1992;46:89–100. [Google Scholar]
- Harper WJ. Dairy ingredients in non-dairy foods. In: Fuquay JW, Fox PF, McSweeney PLH, editors. Encyclopaedia of dairy science. 2. San Diego: Academic Press; 2011. pp. 125–134. [Google Scholar]
- Hausmann A, Duke MC, Demmer T (2013) Principles of membrane filtration. In: Tamime AY (ed) Membrane processing: dairy and beverage applications, 1st edn. Wiley, Hoboken, pp 17–47
- Havea P. Protein interactions in milk protein concentrate powders. Int Dairy J. 2006;16(5):415–422. doi: 10.1016/j.idairyj.2005.06.005. [DOI] [Google Scholar]
- Havea P, Singh H, Creamer LK, Campanella OH. Electrophoretic characterization of the protein products formed during heat treatment of whey protein concentrate solutions. J Dairy Res. 1998;65:79–91. doi: 10.1017/S0022029997002641. [DOI] [Google Scholar]
- Holt C, Dalgleish DG, Jenness R. Inorganics constituents of milk. 2. Calculation of the ion equilibrium in milk diffusate and comparison with experiment. Anal Biochem. 1981;113:154–163. doi: 10.1016/0003-2697(81)90059-2. [DOI] [PubMed] [Google Scholar]
- Huppertz T, Alting A, Slangen C, Floris R (2010) Milk protein concentrate functionality (oral presentation).http://www.wds2010.com/delegates/presentations/09tue/05-Session3_2-Thom%20Huppertz.pdf. Accessed 9 Sept 2012
- Jimenez-Flores R, Kosikowski FV. Properties of ultrafilteed skim milk retentate powders. J Dairy Sci. 1986;69:329–339. doi: 10.3168/jds.S0022-0302(86)80410-6. [DOI] [Google Scholar]
- Kelly P. Milk protein products. In: Fuquay JW, Fox PF, McSweeney PLH, editors. Encyclopaedia of dairy science. 2. San Diego: Academic Press; 2011. pp. 848–854. [Google Scholar]
- Knights RJ (2013) Milk protein concentrates. US 2013/0287892 A1
- Kumar S (2011) Studies on the preparation of dairy whitener employing ultrafiltration Ph.D. Thesis. National Dairy Research Institute, Karnal, India
- Lagrange V, Whitsitt D, Burris C. Global market for dairy proteins. J Food Sci. 2015;80:A16–A22. doi: 10.1111/1750-3841.12801. [DOI] [PubMed] [Google Scholar]
- Le Ray CL, Maubois JL, Gaucheron F, Brulé G, Pronnier P, Garnier P. Heat stability of reconstituted casein micelle dispersions: changes induced by salt addition. Lait. 1998;78:375–390. doi: 10.1051/lait:1998437. [DOI] [Google Scholar]
- Li Y, Corredig M. Calcium release from milk concentrated by ultrafiltration and diafiltration. J Dairy Sci. 2014;97:5294–5302. doi: 10.3168/jds.2013-7567. [DOI] [PubMed] [Google Scholar]
- Liu DZ, Weeks MG, Dunstan DE, Martin GJO. Alterations to the composition of casein micelles and retentate serum during ultrafiltration of skim milk at 10 and 40°C. Int Dairy J. 2014;35:63–69. doi: 10.1016/j.idairyj.2013.10.017. [DOI] [Google Scholar]
- Loveday SM, Hindmarsh JP, Creamer LK, Singh H. Physicochemical changes in a model protein bar during storage. Food Res Int. 2009;42:798–806. doi: 10.1016/j.foodres.2009.03.002. [DOI] [Google Scholar]
- Luo X, Ramchandran L, Vasiljevic T. Lower ultrafiltration temperature improves membrane performance and emulsifying properties of milk protein concentrates. Dairy Sci Technol. 2015;95:15–31. doi: 10.1007/s13594-014-0192-3. [DOI] [Google Scholar]
- Mao XY, Tong PS, Gualco S, Vink S. Effect of NaCl addition during diafiltration on the solubility, hydrophobicity, and disulfide bonds of 80% milk protein concentrate powder. J Dairy Sci. 2012;95:3481–3488. doi: 10.3168/jds.2011-4691. [DOI] [PubMed] [Google Scholar]
- Mason TJ. Power ultrasound in food processing—the way forward. In: Povey MJW, Mason TJ, editors. Ultrasound in food processing. London: Thomson Science; 1998. pp. 105–126. [Google Scholar]
- McCarthy NA, Kelly PM, Maher PG, Fenelon MA. Dissolution of milk protein concentrate (MPC) powders by ultrasonication. J Food Engg. 2014;126:142–148. doi: 10.1016/j.jfoodeng.2013.11.002. [DOI] [Google Scholar]
- McKenna AB (2000) Effects of processing and storage on the reconstitution properties of whole milk and ultrafiltered skim milk powders, Ph.D. Thesis, Massey University, Palmerston North, New Zealand
- Meena GS, Singh AK, Borad S, Raju PN. Effect of concentration, homogenization and stabilizing salts on heat stability and rheological properties of cow skim milk ultrafiltered retentate. J Food Sci Technol. 2016;53(11):3960–3968. doi: 10.1007/s13197-016-2388-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meena GS, Singh AK, Arora S, Borad S, Sharma R, Gupta VK. Physico-chemical, functional and rheological properties of milk protein concentrate 60 as affected by disodium phosphate addition, diafiltration and homogenization. J Food Sci Technol. 2017;54(6):1678–1688. doi: 10.1007/s13197-017-2600-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mimouni A, Deeth HC, Whittaker AK, Gidley MJ, Bhandari BR. Rehydration process of milk protein concentrate powder monitored by static light scattering. Food Hydrocolloid. 2009;23:1958–1965. doi: 10.1016/j.foodhyd.2009.01.010. [DOI] [Google Scholar]
- Mimouni A, Deeth HC, Whittaker AK, Gidley MJ, Bhandari BR. Investigation of the microstructure of milk protein concentrate powders during rehydration: alterations during storage. J Dairy Sci. 2010;93:463–472. doi: 10.3168/jds.2009-2369. [DOI] [PubMed] [Google Scholar]
- Mistry VV. Manufacture and application of high milk protein powder. Lait. 2002;82:515–522. doi: 10.1051/lait:2002028. [DOI] [Google Scholar]
- Mistry VV. Membrane processing in cheese manufacture. In: Fuquay JW, Fox PF, McSweeney PLH, editors. Encyclopaedia of dairy science. 2. San Diego: Academic Press; 2011. pp. 618–624. [Google Scholar]
- Mistry VV. Cheese. In: Tamime AY, editor. Membrane processing: dairy and beverage applications. 1. Hoboken: Wiley; 2013. pp. 194–210. [Google Scholar]
- Mistry VV, Hassan HN. Delactosed high milk protein powder. 2. Physical and functional properties. J Dairy Sci. 1991;74:3716–3723. doi: 10.3168/jds.S0022-0302(91)78562-7. [DOI] [Google Scholar]
- Mistry VV, Pulgar JB. Physical and storage properties of high milk protein powder. Int Dairy J. 1996;6:195–203. doi: 10.1016/0958-6946(95)00002-X. [DOI] [Google Scholar]
- Monahan FJ, German JB, Kinsella JE. Effect of pH and temperature on protein unfolding and thiol/disulphide interchange reactions during heat-induced gelation of whey proteins. J Agric Food Chem. 1995;43:46–52. doi: 10.1021/jf00049a010. [DOI] [Google Scholar]
- Oldfield DJ, Singh H, Taylor MW. Effect of preheating and other process parameters on whey protein reactions during skim milk powder manufacture. Int Dairy J. 2005;15:501–511. doi: 10.1016/j.idairyj.2004.09.004. [DOI] [Google Scholar]
- Patel RS, Mistry VV. Physicochemical and structural properties of ultrafiltered buffalo milk and milk powder. J Dairy Sci. 1997;80:812–817. doi: 10.3168/jds.S0022-0302(97)76002-8. [DOI] [Google Scholar]
- Patel H, Patel S (2014) Milk protein concentrates: manufacturing and applications- technical report.http://www.usdairy.com/~/media/usd/public/mpc_tech_report_final.pdf. Accesed on 21 Feb 2016
- Rajagopalan N, Cheryan M. Total protein isolate from milk by ultrafiltration: factors affecting product composition. Dairy Sci. 1991;74:2435–2439. doi: 10.3168/jds.S0022-0302(91)78418-X. [DOI] [Google Scholar]
- Ramchandran L, Vasiljevic T. Whey processing. In: Tamime AY, editor. Membrane processing: dairy and beverage applications. 1. Hoboken: Wiley; 2013. pp. 193–205. [Google Scholar]
- Rice G (2008) Membrane separation of calcium salts from dairy ultrafiltration permeates, Ph.D. thesis, The University of Melbourne, Melbourne
- Richard B, Toubal M, Le Page JF, Nassar G, Radziszewski E, Nongaillard B, Debreyne P, Schuck P, Jeantet R, Delaplace G. Ultrasound tests in a stirred vessel to evaluate the reconstitution ability of dairy powders. Innov Food Sci Emerg Technol. 2012;16:233–242. doi: 10.1016/j.ifset.2012.06.007. [DOI] [Google Scholar]
- Rosenberg M. Current and future applications for membrane processes in the dairy industry. Trends Food Sci Technol. 1995;6:12–19. doi: 10.1016/S0924-2244(00)88912-8. [DOI] [Google Scholar]
- Schuck P. Understanding the factors effectively affecting spray-dried dairy powder properties and behaviour. In: Corredig M, editor. Dairy-derived ingredients, food and nutraceutical uses. 1. Cambridge: Woodhead Publishing Limited; 2009. pp. 24–50. [Google Scholar]
- Schuck P, Davenel A, Mariette F, Briard V, Méjean S, Piot M. Rehydration of casein powders: effects of added mineral salts and salt addition methods on water transfer. Int Dairy J. 2002;12:51–57. doi: 10.1016/S0958-6946(01)00090-5. [DOI] [Google Scholar]
- Sikand V, Tong PS, Roy S, Saona LER, Murray BA. Solubility of commercial milk protein concentrates and milk protein isolates. J Dairy Sci. 2011;94:6194–6202. doi: 10.3168/jds.2011-4477. [DOI] [PubMed] [Google Scholar]
- Sikand V, Tong P, Vink S, Walker J. Effect of powder source and processing conditions on the solubility of milk protein concentrates 80. Milchwissenschaft. 2012;67:300–303. [Google Scholar]
- Sikand V, Tong PS, Roy S, Rodriguez-Saona LE, Murray BA. Effect of adding salt during the diafiltration step of milk protein concentrate powder manufacture on mineral and soluble protein composition. Dairy Sci Technol. 2013;93:401–413. doi: 10.1007/s13594-013-0110-0. [DOI] [Google Scholar]
- Singh H. Interactions of milk proteins during the manufacture of milk powders. Lait. 2007;87:413–423. doi: 10.1051/lait:2007014. [DOI] [Google Scholar]
- Singh H. Functional properties of milk proteins. In: Fuquay JW, Fox PF, McSweeney PLH, editors. Encyclopaedia of dairy science. 2. San Diego: Academic Press; 2011. pp. 887–893. [Google Scholar]
- Singh H, Newstead DF. Aspects of proteins in milk powder manufacture. In: Fox PF, editor. Advanced dairy chemistry, vol. 1: proteins. 1. England: Elsevier; 1992. pp. 735–765. [Google Scholar]
- Smith K. Commercial membrane technology. In: Tamime AY, editor. Membrane processing: dairy and beverage applications. 1. Hoboken: Wiley; 2013. pp. 52–71. [Google Scholar]
- St-Gelais D, Hache S, Gros-louis M. Combined effects of temperature, acidification, and diafiltration on composition of skim milk retentate and permeate. J Dairy Sci. 1991;75:1167–1172. doi: 10.3168/jds.S0022-0302(92)77863-1. [DOI] [Google Scholar]
- Sweetsur AWM, Muir DD. Effect of concentration by ultrafiltration on the heat stability of skim milk. J Dairy Res. 1980;47:327–335. doi: 10.1017/S002202990002121X. [DOI] [Google Scholar]
- Sweetsur AWM, Muir DD. Optimization of the heat stability of concentrated milks prepared by Ultrafiltration. Milchwissenschaft. 1985;40:334–337. [Google Scholar]
- Tong PS, Smithers GW. The future of dairy ingredients: critical considerations that will underpin future success. In: Smithirs GW, Augustin MA, editors. Advances in dairy ingredients. 1. Hoboken: Wiley; 2013. pp. 313–317. [Google Scholar]
- Tripathy P (2005) Studies on the formulation of non-fat dairy whitener based on ultrafiltered buffalo skim milk M.Sc. Thesis, Institute of food technology, Bundelkhand University, Jhansi, (U.P)
- Udabage P, Puvanenthiran A, Yoo JA, Versteeg C, Augustin MA. Modified water solubility of milk protein concentrate powders through the application of static high pressure treatment. J Dairy Res. 2012;79:76–83. doi: 10.1017/S0022029911000793. [DOI] [PubMed] [Google Scholar]
- Uluko H, Liu L, Lv JP, Zhang SW. Functional characteristics of milk protein concentrates and their modification. Crit Rev Food Sci Nutr. 2015 doi: 10.1080/10408398.2012.758625. [DOI] [PubMed] [Google Scholar]
- U.S. Dairy Export Council . Global follow-on formula, growing-up milk. Arlington: Dairy Export Council; 2012. p. 64. [Google Scholar]
- Vora H (2008) Studies on the formulation of cow milk dairy whitener using ultrafiltration process. M. Tech. Thesis, National Dairy Research Institute (Deemed University), Karnal, Haryana, India
- Walstra P, Wouters JTM, Geurts TJ. Dairy science and technology. 2. Boca Raton: CRC Press; 2006. Milk components; pp. 17–108. [Google Scholar]
- Yanjun S, Jianhang C, Shuwen Z, Hongjuan L, Jing L, Lu L, Uluko H, Yanling S, Wenming C, Wupeng G, Jiaping L. Effect of power ultrasound pre-treatment on the physical and functional properties of reconstituted milk protein concentrate. J Food Eng. 2014;124:11–18. doi: 10.1016/j.jfoodeng.2013.09.013. [DOI] [Google Scholar]