Abstract
Background
Preterm (PT) infants are at greater risk for severe influenza infection and experience decrements in long-term antibody responses to vaccines. This may related to defects in antibody secreting cell (ASC) generation.
Objective
To investigate the relationships among the frequencies of influenza-specific antibody secreting cells ASC numbers and subsets and antibody responses to influenza vaccines (IV) among PT and full-term (FT) infants.
Design/Methods
We enrolled 11 former PT (≤32 weeks’ gestation, ≤1500 grams’ birth weight) and 11 FT infants, 6 to 17 months of age, receiving their first influenza immunizations. Infants received two doses of inactivated trivalent (T)IV or quadrivalent (Q)IV during the 2012–2013 and 2013–2014 influenza seasons, respectively, at 0 and 28 days, and blood was drawn at 0, 10, 35, and 56 days and 9 months. Vaccine-specific antibody was measured by hemagglutination inhibition (HAI) at 0 and 56 days and 9 months, vaccine-specific ASC numbers by enzyme linked immunospot (ELISPOT) at 10 and 35 days, and ASC subsets by flow cytometry at 0, 10 and 35 days.
Results
PT infants had post-vaccine HAI titers to all 4 vaccine strains at least equal to FT infants at 56 days and 9 months after beginning immunization. Influenza-specific ASC ELISPOT responses at 35 days were higher among PT than FT infants (median 100 v. 30 per 106 PBMC, p = 0.04). ASC numbers at 35 days were positively correlated with serum HAI titers at 56 days (ρ=0.50–0.80). There were no statistical differences between PT and FT infants in the frequency of five ASC subsets and no specific ASC subset correlated with durability of serum antibody titers.
Conclusion(s)
Influenza-specific ASC numbers in both FT and PT infants correlated with peak antibody titers, but ASC subsets did not correlate with durability of antibody response.
Keywords: Influenza vaccine, premature infant, antibody-secreting cell, antibody titer, plasma cell
Introduction
Infants, particularly preterm (PT) infants, are at a greater risk for severe influenza infection and related complications [1]. PT infants are born with immature immune systems, including defects in B-cell function, yet it is unclear how prematurity affects the quality of the cellular vaccine responses or when immune function matures after birth [2]. Compared to full term (FT) infants, extremely PT infants have decrements in serum titers to a number of vaccines that can persist at least to school age [3–18]. Although PT and FT infants exhibit similar acute serological responses to influenza vaccines [19], no studies have assessed long term immunogenicity or cellular immunity following influenza vaccine among PT infants.
After vaccination, protective antibody titers originate from naïve B cell differentiation into antibody secreting cells (ASC) and memory B cells in infants. In adults, most circulating ASC demonstrate vaccine antigen specificity with little bystander response [20], and influenza-specific ASC frequency correlates with the rise in protective antibody titers [21]. Among adults, there is significant heterogeneity in the ASC (CD19+CD27hiCD38hi) subsets after vaccination that include CD138+ and CD138neg subsets [22, 23]. We have also identified CD19neg ASC populations after vaccination that resemble a recently described human bone marrow long-lived plasma cell (LLPC) phenotype by cell surface markers [24].
We evaluated the relationships among the frequencies of influenza-specific antibody secreting cells ASC numbers and subsets and antibody responses to influenza vaccines (IV) among PT and full-term (FT) infants.
Materials and Methods
Subjects
In this prospective study, infants were recruited during the 2012–2013 and 2013–2014 influenza vaccine seasons at the University of Rochester. We enrolled very-low-birth-weight (≤ 1500 grams at birth), PT (≤ 32 weeks’ gestation at birth) infants and normal-birth-weight (> 2500 grams at birth), FT (35 0/7 to 41 6/7 weeks’ gestation at birth) infants into the study. Subjects were 6–17 months chronological age, previously un-immunized for influenza, and eligible for influenza immunization. Subjects with known immunodeficiency, concurrent systemic corticosteroid administration or physician-diagnosed influenza illness were excluded. The Institutional Review Board approved the study, and informed parental permission was obtained.
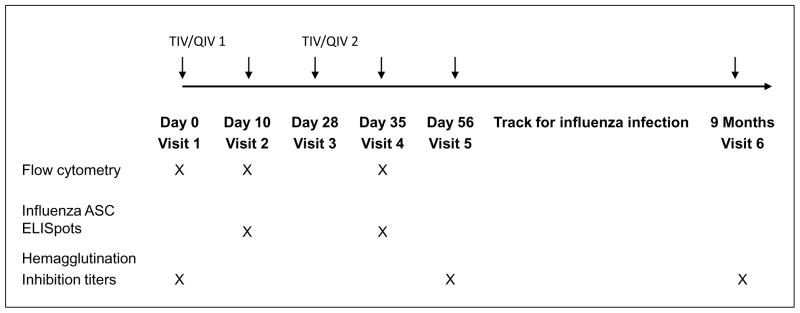
Children had six study visits over 9 months, at 0 (Visit 1), 10 (Visit 2), 28 (Visit 3), 35 (Visit 4) and 56 (Visit 5) days, and 9 months (Vsiit 6) from the first dose of influenza vaccine (Figure 1). Children received two doses (prime/boost) by intramuscular thigh injection with a 1-inch needle of trivalent (TIV) (H1N1, H3N2, B-Yamagata, 2012–2013 season, multiple lots) or quadrivalent (QIV) (H1N1, H3N2, B-Yamagata, B-Victoria, 2013–2014 season, multiple lots), inactivated, split-virion influenza vaccine (Fluzone®, Sanofi Pasteur, Swiftwater, PA), about 4 weeks apart. TIV components were A/California/7/2009 (H1N1), A/Victoria/361/2011 (H3N2), and B/Wisconsin/1/2010 (Yamagata lineage). QIV components were A/California/7/2009 (H1N1)-like virus, A/Victoria/361/2011 (H3N2)-like virus, B/Massachusetts/2/2012-like virus (B/Yamagata lineage), and B/Brisbane/60/2008-like virus (Victoria lineage). Since only QIV contained the B/Victoria strain, B/Victoria-related assay results are reported only for children receiving QIV. Blood was obtained at five time points (Figure 1). An interim medical history, focused on influenza symptoms, was obtained at each study visit.
Figure 1.
Schematic of study design. TIV = Trivalent influenza vaccine, QIV = quadrivalent influenza vaccine. Arrows denote visits. X = blood draw performed for indicated analysis.
Peripheral blood mononuclear cell (PBMC) isolation
PBMC were isolated as previously described [25]. Briefly, plasma was first separated by centrifugation at room temperature. The blood, diluted 1:2 with phosphate buffered saline was centrifuged over a Ficoll gradient. The buffy coat layer was transferred to a 15-mL tube, and the cells were washed with HBSS. Cells were counted and viability assessed by Trypan Blue exclusion. All enzyme-linked immunospot (ELISPOT) and flow cytometry assays were done on fresh cells.
Influenza hemagglutination inhibition assay (HAI)
Sera were separated and frozen at −80°C until analysis. Influenza antibody levels were measured by microtiter hemagglutination inhibition (HAI) test, as previously described [19]. Briefly, reagents for the HAI test were obtained from the WHO Collaborating Centers for Influenza, Centers for Disease Control and Prevention. Hemagglutination titer for each influenza antigen was determined and diluted to contain 4 HA units in 0.025-mL of virus suspension. H1N1, H3N2, B/Victoria, and B/Yamagata antigens were used. Serum titers were determined by the highest dilution of serum capable of inhibiting hemagglutination. A serum control was run on each plate, and all assays within a vaccine season were run using the same lot of antigen.
Antigen-specific ASC ELISPOT
The frequency of influenza antigen-specific ASCs was measured by ELISPOT, as previously described [21]. Briefly, 96-well ELISPOT plates were coated for 2 hours at room temperature with the following: TIV for the 2012–2013 cohort (6 μg/mL, Sanofi Pasteur), QIV for the 2013–2014 cohort (8 μg/mL, Sanofi Pasteur), goat anti-human IgG (5 μg/mL, Invitrogen), tetanus toxoid (2 μg/ml, Calbiochem) or Bovine Serum Albumin (2% in PBS, Fisher), diluted in PBS. Plates were then washed and blocked with RPMI + fetal bovine serum + antibiotic/antimycotic for 2 hours at 37°C with 5% CO2. Plates were incubated at 37°C with 8% CO2 for 18–20 hours with 300,000, 100,000 or 10,000 PBMC in duplicate. Cells were then aspirated and plates washed with PBS with 0.1% Tween. Bound antibodies were detected with alkaline phosphatase-conjugated goat-anti-human IgG (1 μg/mL, Jackson Immunoresearch) for 2 hours and washed with PBS/Tween. Plates were developed with Vector Alkaline Phosphatase Substrate Kit III. Spots in each well were counted using a CTL Immunospot reader.
Polychromatic flow cytometry
An aliquot of PBMC were stained with anti-human Ki67-FITC, IgD-PE, CD20-Cy5PE, CD3/CD14-Cy5.5PE, CD19-Cy7PE, CD38-V450 (PacBlue), CD138-APC, and CD27-APCeFluor780 and analyzed by flow cytometry (LSR II, Biosciences, Franklin Lakes, NJ) using FlowJo software (Tree Star, Inc., Ashland, OR) as previously described [24].
Statistical analysis
All statistical analyses were performed using SAS (version 9.4, SAS Institute Inc, Cary, NC). The Fisher’s exact test was used for categorical variables while t-test or Wilcoxon Rank Sum test was used for continuous variables. HAI titers were log-transformed to satisfy the normality assumption of the models. Spearman correlation coefficient (ρ) was used to evaluate the correlation between outcome variables such as change in HAI titers, ASC frequencies measured by ELISPOT and ASC subset cells. HAI titers measured at different time points were also compared between study groups using repeated measure Analysis of Covariance (ANCOVA) after controlling for subject gender, baseline weight and age. Since the data were collected from two vaccine seasons, the potential batch effect of vaccines was examined by including the interaction with season. The association between the ASC subset cell populations and the change in HAI titers was also investigated using ANCOVA. All analyses were 2-sided at the 0.05 level of significance.
Results
Over two years, 11 premature infants (7 in 2012–2013, 4 in 2013–2014) and 11 full-term infants (3 in 2012–2013, 8 in 2013–2014) were enrolled. No infant was reported to have an influenza-like illness either before or during the study. However, one full-term subject from the 2012–2013 season was excluded from analyses due to serological evidence of prior B/Victoria influenza infection. All PT infants and 8 FT infants completed all visits, but small volumes from blood draws limited the sample available for some analyses. The denominator for each assay is given in the appropriate Table or Figure.
Subject characteristics are shown in Table 1. PT infants were of similar postnatal age, but weighed less, than FT infants. The time between the first and second doses of vaccine was similar between the two groups. No infant suffered a vaccine-related or study-related adverse event.
Table 1.
Vaccination characteristics among subjects with postvaccination titers available.
| PT Group (n=11) | FT Group (n=10) | |
|---|---|---|
| Gestational age at birth (weeks) | 27.3 ± 1.8 | 39.2 ± 1.7 |
| Birth weight (grams) | 961 ± 280 | 3450 ± 408 |
| Sex (male) | 6 (55%) | 4 (40%) |
| Age at first vaccination (months) | 8.1 ± 1.7 | 8.0 ± 2.4 |
| Age (corrected for gestation) at first vaccination (months) | 5.2 ± 1.8 | 7.8 ± 2.5 |
| Weight at first vaccination (kg) | 6.3 ± 0.8 | 8.1 ± 0.6 |
| Time from first to second vaccine dose (weeks) | 5.1 ± 0.4 | 5.4 ± 0.9 |
| Time from first vaccine dose (Visit 1) to Visit 6 (months) | 8.9 ± 0.8 | 8.7± 0.8 |
Results are presented as mean ± standard deviation (SD) or N (%)
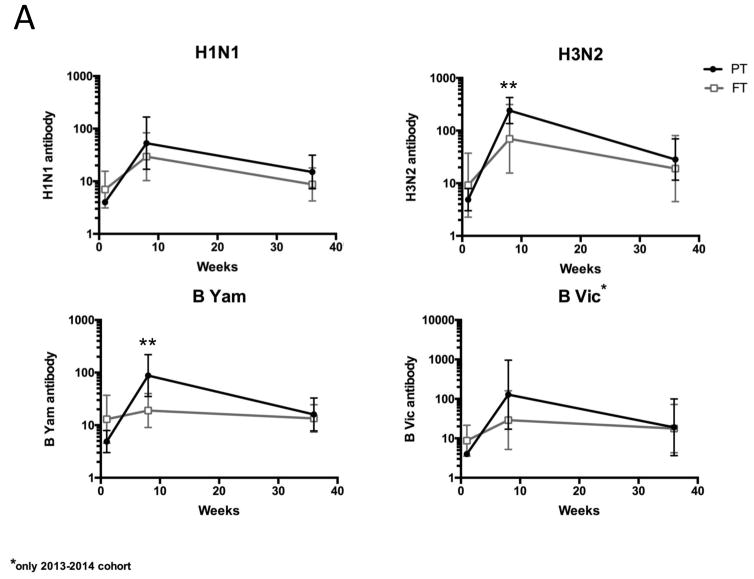
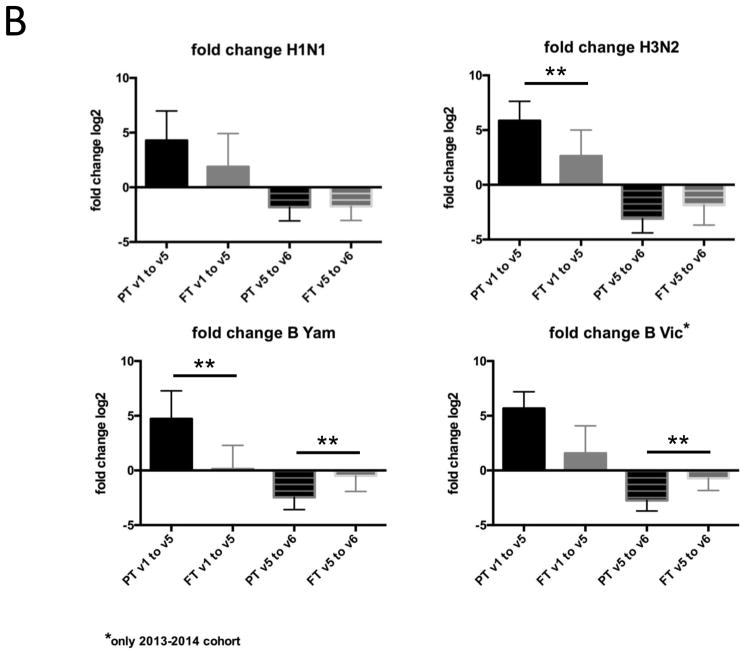
When HAI geometric mean titers (GMT) for H3N2 and B/Yamagata were higher in PT than FT children at one month after the boost (Visit 5) (Figure 2A). There were no differences between groups children reaching nominal protective antibody titers of either 1:32 or 1:64, other than a difference in B/Yamagata titers ≥1:64 favoring PT infants at Visit 5 (Table 2)[26]. PT infants had a larger fold change in antibody titer compared to FT infants following two vaccine doses for H3N2 and B/Yamagata, and a larger fall in titer of B/Yamagata and B/Victoria from the peak titer (Visit 5) to 9 months (Visit 6) (Figure 2B).
Figure 2.
Hemagglutination inhibition (HAI) titers. Serum was collected and analyzed at Visit 1 (0 days), Visit 5 (56 days) and Visit 6 (9 months). Pre-vaccine titers (Visit 1) were available for 7 preterm (PT) and all full-term (FT) infants and post-vaccine titers (Visits 5 and 6) available for all PT and 8 FT infants. B Yam = B/Yamagata. B Vic = B/Victoria. *B/Victoria assessed only in 2013–14 cohort (when it was a component of vaccine).
A. Geometric mean titers (GMT) with 95% confidence intervals (CI). GMT for H3N2 and B Yam differed between PT and FT infants at Visit 5. **P<0.05.
B. Fold change in titers between visits ± standard deviation (SD). v1 = Visit 1, v5 = Visit 5, v6 = Visit 6. Fold change in titers from Visits 1 to Visit 5 differed between PT and FT for H3N2 and B Yamagata and from Visits 5 and 6 for B/Yamagata and B/Victoria. **P<0.05.
Table 2.
Proportion of Children with Postvaccine Antibody Titer ≥1:32 or ≥1:64.
| Visit #5 (Day 56) | Visit #6 (9 Months) | |||||
|---|---|---|---|---|---|---|
| PT | FT | P* | PT | FT | P* | |
| Titer ≥1:32 | ||||||
| H1N1 | 9/11 (82%) | 3/8 (38%) | 0.07 | 3/11 (27%) | 2/8 (25%) | 1.0 |
| H3N2 | 11/11 (100%) | 6/8 (75%) | 0.16 | 6/11 (55%) | 2/8 (25%) | 0.35 |
| B/Yamagata | 9/11 (82%) | 3/8 (38%) | 0.07 | 4/11 (36%) | 1/8 (13%) | 0.34 |
| B/Victoria | 4/4 (100%) | 3/7 (43%) | 0.19 | 2/4 (50%) | 3/7 (43%) | 1.0 |
| Titer ≥1:64 | ||||||
| H1N1 | 6/11 (55%) | 3/8 (38%) | 0.65 | 2/11 (18%) | 0/8 (0%) | 0.49 |
| H3N2 | 11/11 (100%) | 6/8 (75%) | 0.16 | 5/11 (45%) | 1/8 (13%) | 0.18 |
| B/Yamagata | 9/11 (82%) | 2/8 (25%) | 0.02 | 2/11 (18%) | 1/8 (13%) | 1.00 |
| B/Victoria | 3/4 (75%) | 2/7 (29%) | 0.24 | 1/4 (25%) | 2/7 (29%) | 1.00 |
PT = preterm, FT = full term
P values by Fisher exact test
Since we had previously shown that chronological age explained differences between PT and FT influenza HAI titers [19], we performed secondary analyses for the HAI results controlling for age, weight and sex. When controlled for co-variates, the HAI titer GMT did not differ statistically significantly between PT and FT infants for any strain at any time. Evaluation also showed no effect of season on titer and no interaction of titer and season (p>0.25 for all analyses).
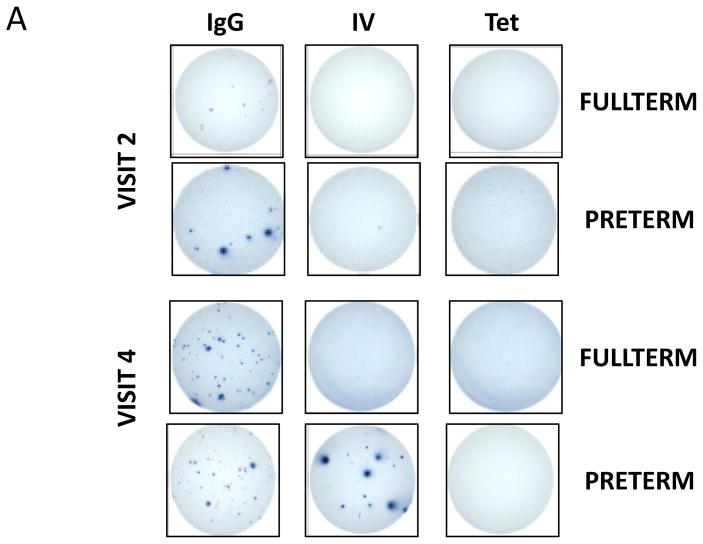
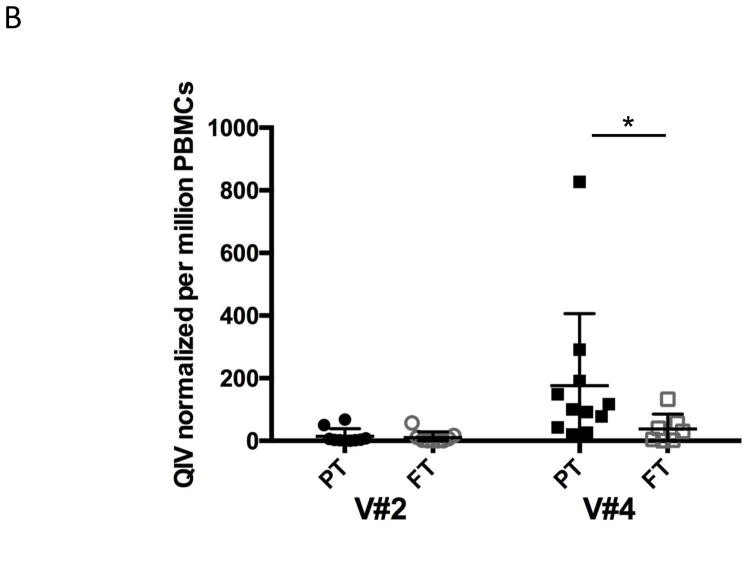
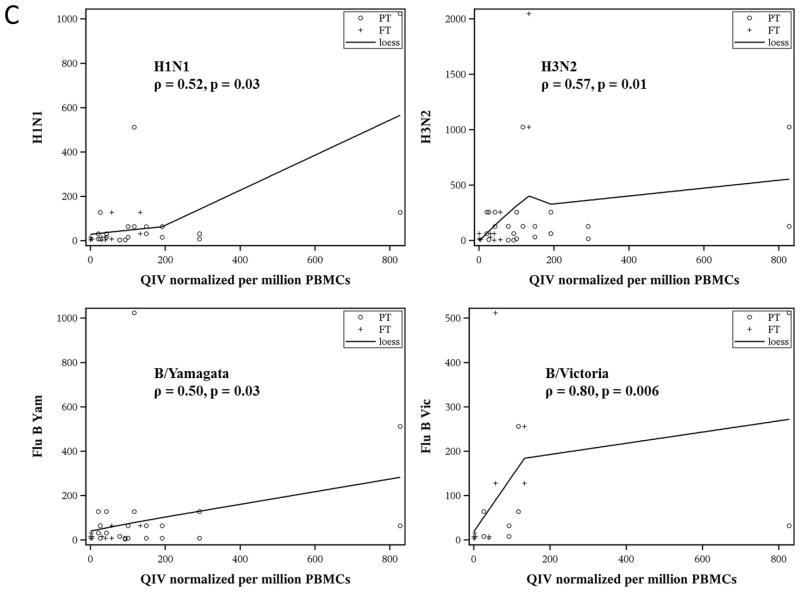
Representative ELISPOT wells from PT and FT infants are shown in Figure 3A. Quality and size of the spots did not differ significantly between the two groups. ASC ELISPOT frequencies were higher among PT than FT infants at Visit 4 (seven days after the second dose of vaccine) (Figure 3B). Additionally, ASC frequencies at Visit 4 correlated with HAI titers for all four strains at Visit 5 (Figure 3C). ASC frequencies at Visit 4 also correlated with individual antibody titer to the H1N1 (ρ = 0.60, p = 0.008) and B/Victoria (ρ = 0.65, p = 0.04) HAI titers at nine months (Visit 6).
Figure 3.
Frequency of influenza antigen-specific antibody secreting cells (ASCs) measured by Enzyme-linked immunospot (ELISPOT) assay, as measured at Visits 2 (10 days) and 4 (35 days).
A. Representative wells from preterm and full-term infants at each Visit. IgG = IgG-coated well, IV = influenza-vaccine-coated wells (TIV for 2012–13, QIV for 2013–14), Tet = tetanus-coated wells.
B. Spots per million peripheral blood mononuclear cells (PBMC), normalized to bovine serum albumin (BSA) background at Visits 2 (V#2) and 4 (V#4). Preterm (PT) ASC numbers were higher than full-term (FT) numbers at Visit 4. Each square represents a separate child. Mean (central bar) and standard deviation (whiskers) are shown. Standard deviation extends below axis in some cases. *P = 0.04.
C. Correlation between antigen-specific ASC frequency measured at Visit 4 and hemagglutination inhibition (HAI) titers measured at Visit 5 (56 days). ASC frequency and titer were modestly correlated for all serotypes. Each symbol represents a separate child (° = Preterm, + = Full-term). LOESS smoothing curves (loess) are shown to illustrate relationships. ρ = Spearman’s rho correlation coefficient.
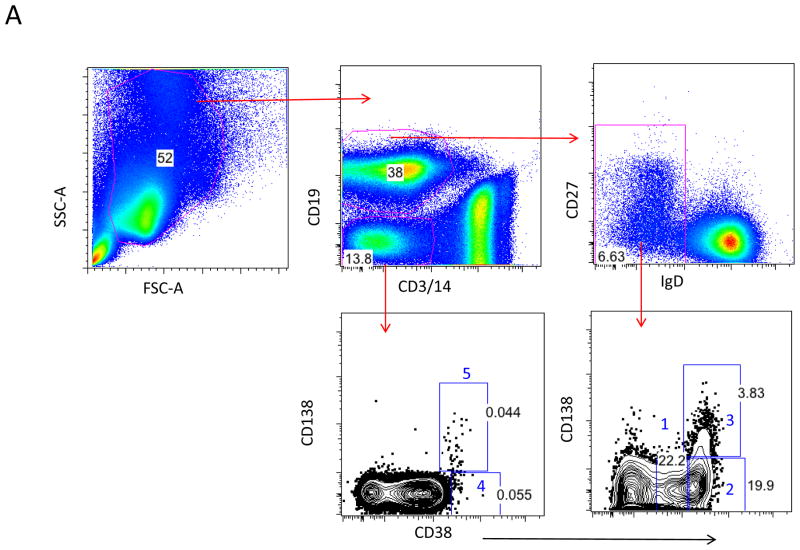
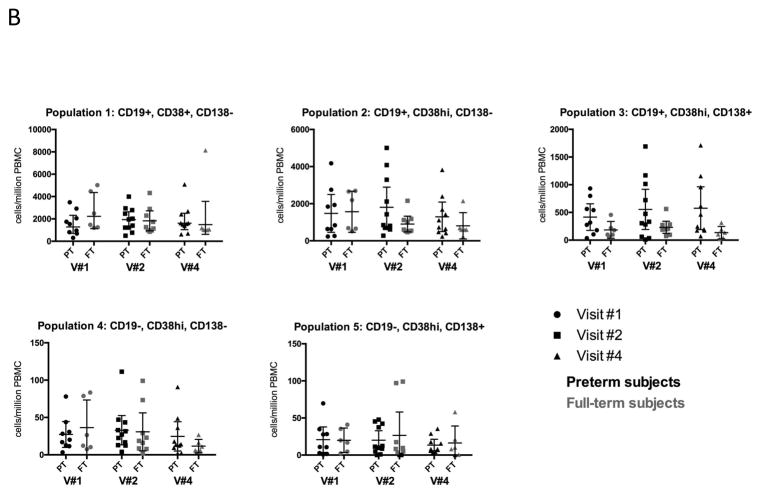
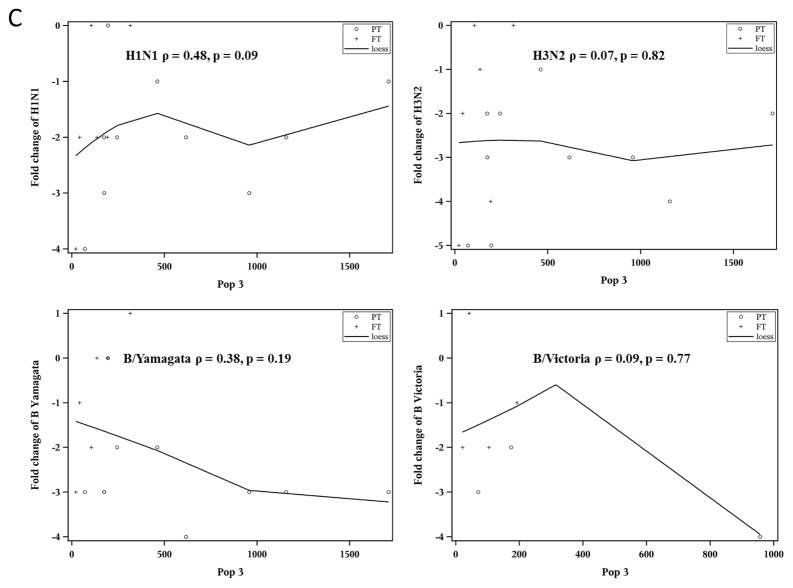
Although mature long-lived plasma cells in the human bone marrow express high levels of CD138 and a fraction of the circulating ASC also can express CD138 in adults [24, 27], little was known if ASC from both PT and FT infants can express CD138. We identified five distinct ASC subsets using CD19, CD38, and CD138 similar to markers used in human BM. Population (Pop) 1 cells were identified as CD19+CD38+CD138neg and contains both B cells and early ASCs. Two CD19+ subsets included pop 2: CD19+CD38hiCD138neg and pop 3: CD19+CD38hiCD138+ and two CD19neg subsets included pop 4:CD19negCD38hiCD138neg and pop 5: CD19negCD38hiCD138+ (Figure 4A). Both FT and PT infants expressed CD138+ ASC subsets (pop 3& 5) although the majority of ASC subsets after vaccination in both FT and PT was pop 2: CD19+CD138neg (Figure 4B). There were no differences in frequency of any ASC subset at any time between PT and FT infants. Although the frequency of one ASC subset (pop 3: CD19+CD38hiCD138+) appeared higher among PT at the peak of the ASC responses (Visit 4), this difference did not reach statistical significance (p = 0.052). There was no correlation between the frequency of any ASC subset at the peak of the cellular ASC responses (Visit 4) and the rise in HAI titer for any strain one month after vaccination (data not shown) or the decay between the peak and at 9 months (Figure 4C).
Figure 4.
ASC cell sub-populations, as measured by flow cytometry.
A. Flow cytometry gating strategy. Cell populations 1–5 are described in Methods.
B. Frequency of cell populations in preterm (PT) and full-term (FT) infants. Center bar represents mean and whiskers represent 95% confidence interval. Each symbol represents a separate child. There were no statistically significant differences in numbers of cells in any population per million peripheral blood mononuclear cells (PBMC) between FT and PT infants at any of the 3 visits (V).
C. Correlation between frequency of CD19+ CD38hi CD138+ cells (Population 3) measured by flow cytometry at Visit 4 (35 days) and change in hemagglutination inhibition (HAI) titers measured at Visits 5 (56 days) and 6 (9 months). Each symbol represents a separate child (° = Preterm, + = Full-term). LOESS smoothing curves (loess) are shown to illustrate relationships. ρ = Spearman’s correlation coefficient. There were no statistically significant correlations between Population 3 frequency and HAI titer change for any strain.
Discussion
In this study, we validated similar influenza HAI titers in both PT and FT infants after the recommended initial influenza vaccine series. PT infants had higher peak HAI titers than FT infants to some vaccine antigens, and the peak serum titers correlated with the vaccine-specific ASC frequencies in all infants. There were few differences in the proportions of infants who reached protective titers, which are in any case only approximations of vaccine effectiveness [26, 28]. Both PT and FT infants had significant heterogeneity of the ASC subsets after the first influenza vaccine. By flow cytometry, we were able to identify 5 distinct ASC populations in the blood after vaccination, which are similar to findings in adults (unpublished data). This is among the first studies to demonstrate that heterogeneity of ASC subsets (both CD19+ and CD19neg and CD138+ and CD138neg subsets) occur in PT infants, suggesting that gestational age does not affect generation of heterogeneity of ASC subsets. We did not see a correlation between any particular ASC subset and durability of HAI titers.
The elevated humoral response of the PT infant group is consistent with our previous findings that PT infants had higher serum antibody titers than FT infants after immunization with influenza, MMR and varicella vaccines [19, 29]. Multivariate analysis showed that factors such as gender, weight and postnatal age explain some of the differences between FT and PT infant titers, a finding in keeping with our previous work [19]. PT infants had lower baseline HAI titers prior to vaccination, which may have been a result of limited placental transmission of maternal antibodies [30, 31]. Lower initial antibody titers may have limited antibody blocking of vaccine antigen, resulting in the higher rises of HAI titers and larger vaccine-specific ASC frequencies seen in the PT infants.
Human long-lived plasma cells (LLPC) are responsible for long-term maintenance of protective serum antibodies [32–34], and our group has recently identified the human LLPC in the bone marrow by showing long-lived viral responses in only one (CD19negCD38hi CD138+) plasma cell subset [24]. This LLPC compartment is morphologically and transcriptionally distinct compared to the short-lived bone marrow (BM) plasma cells, with distinct vacuoles, tighter chromatin and increased number of autophagosomes and a unique autophagy signature [24]. Although the goal of human vaccines is to generate and sustain LLPC, the mechanisms of how human LLPC formation are not well understood.
The presence of specific B cell populations in the blood of aged individuals has been correlated to poor vaccine responses to influenza, which may be due in part to altered T helper cell function [35, 36]. Furthermore, the CD4+ T cell response to Streptococcus pneumoniae and Haemophilus influenzae vaccines are lower in pediatric subjects than in adults [37]. This could be partially explained by differences in soluble factors that are found at different levels in childrens than in adults, including increased IL-10 and decreased TNF-α [38, 39]. Thus, alterations in cellular phenotype may affect vaccine response in both aged individuals and children born preterm.
Distinct CD19neg and CD138+ ASC subsets are found in the blood of adults after highly effective vaccines such as tetanus toxoid (with a 10-year antibody half life), suggesting that heterogeneity of ASC subsets are important for long-lived vaccine responses [22, 23]. To our surprise, all ASC subsets were found among very PT infants and, despite lack of correlation with ASC subsets, we were able to measure influenza HAI titers at 9 months after immunization.
In this study, despite the higher peak of serum antibodies, PT infants had a larger fall in antibody titer by 9 months post-vaccine than FT infants, resulting in similar HAI values among FT and PT infants at 9 months. These results suggest that PT infants may not generate LLPC as efficiently as FT infants to sustain protective antibody titers. However, ASC heterogeneity in subsets was similar between PT and FT infants suggesting extrinsic mechanistic differences beyond ASC may be responsible for persistence of plasma cell survival in the FT infants. One explanation could be differences in migratory capacity of ASC to BM survival niches, since CXCR4 has been shown to be important for LLPC generation [40]. Another difference could be an immature BM microenvironment in PT compared to FT infants, which could play a critical role plasma cell survival. More studies are needed with particular focus on understanding differences in BM microenvironments between PT and FT infants.
This study has several limitations. The sample size was small, giving limited power for the analyses and raising the possibility of missing small differences in ASC populations associated with antibody durability. The study population was drawn from two influenza vaccine seasons, with an imbalance of PT and FT infants between seasons. However, the three shared vaccine components (H1N1, H3N2 and B/Yamagata) were similar between seasons, and we previously found no differences between vaccine responses distributed over two seasons [19]. Further, additional analysis suggested no effect of season on the results. Despite attempts to rule out influenza virus exposure among enrollees, one infant showed serological evidence of prior influenza infection. Other infants could have had asymptomatic influenza infections during the study. Baseline titers allowed us to rule out pre-existing influenza exposure, but we could not completely rule out illness acquired during the study.
Although our study focused on influenza-IgG class-switched ASC, the HAI assay and ASC subsets evaluated all isotypes. Since antibody type and specificity mediate different downstream biological effects (e.g., antibody-dependent cell-mediated cytotoxity or interferance with intracellular uncoating of viral particles) [41, 42], our study did not specifically address the potentially differential effects of various isotypes.
This study supports the suggestion that by 6 months of age, the immunological immaturity of vaccine responses displayed by PT infants at birth and thereafter has largely resolved [3–18]. The similarity to FT infants in ASC numbers and ASC subpopulations provides further supporting evidence for the relative immunological maturity of 6-month-old former extremely PT infants. However, questions of protective antibody durability and LLPC generation in PT infants remain.
In conclusion, this study explored the relationship among antibody titers, influenza-specific ASC, and ASC subsets measured by flow cytometry in PT and FT infants. It was unique in the use of multiple comprehensive assessments at many time points over months. Influenza vaccination among premature infants produces strong and durable antibody responses and remains one of the most effective methods of preventing severe influenza and its complications among this vulnerable population.
Highlights.
Influenza-specific antibody-secreting cells numbers were elevated among premature infants
Antibody-secreting cells numbers correlated with hemagglutination inhibition titers
Five antibody-secreting cell subsets were identified in both premature and full term infants
Acknowledgments
This research was supported in part by NHLBI R01 HL097141-01, 1R01 AI121252, P01 AI125180-01, P01 A1078907-04. We thank Heidie Huyck for help with specimen processing, Ken Schnabel for technical support with HAI measurements, and the University of Rochester Vaccine Evaluation Unit (John Treanor, Director) and Doreen Francis for supporting subject visits. We thank Elizabeth Werner, Dee Maffett, Cassandra Horihan and Rosemary Jensen for assistance with study coordination and Michael O’Reilly and Paige Lawrence for collaboration in obtaining the funding that made this work possible. Above all, we thank the children and their parents who participated in the study.
Abbreviations
- ASC
antibody secreting cells
- ELISPOT
enzyme-linked immunospot
- FT
full-term
- GA
gestational age
- HAI
hemagglutination inhibition
- LLPC
long-lived plasma cells
- PBMC
peripheral blood mononuclear cell
- PT
preterm
- QIV
quadravalent inactivate influenza vaccine
- TIV
trivalent inactivated influenza vaccine
Footnotes
Conflicts of interest: None
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Resch B, Kurath-Koller S, Eibisberger M, Zenz W. Prematurity and the burden of influenza and respiratory syncytial virus disease. World J Pediatr. 2016;12:8–18. doi: 10.1007/s12519-015-0055-x. [DOI] [PubMed] [Google Scholar]
- 2.Sharma AA, Jen R, Butler A, Lavoie PM. The developing human preterm neonatal immune system: a case for more research in this area. Clin Immunol. 2012;145:61–8. doi: 10.1016/j.clim.2012.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Washburn LK, O’Shea TM, Gillis DC, Block SM, Abramson JS. Response to Haemophilus influenzae type b conjugate vaccine in chronically ill premature infants. Journal of Pediatrics. 1993;123:791–4. doi: 10.1016/s0022-3476(05)80862-5. [DOI] [PubMed] [Google Scholar]
- 4.Greenberg DP, Vadheim CM, Partridge S, Chang SJ, Chiu CY, Ward JI. Immunogenicity of Haemophilus influenzae type b tetanus toxoid conjugate vaccine in young infants. The Kaiser-UCLA Vaccine Study Group. Journal of Infectious Diseases. 1994;170:76–81. doi: 10.1093/infdis/170.1.76. [DOI] [PubMed] [Google Scholar]
- 5.Munoz A, Salvador A, Brodsky NL, Arbeter AM, Porat R. Antibody response of low birth weight infants to Haemophilus influenzae type b polyribosylribitol phosphate-outer membrane protein conjugate vaccine. Pediatrics. 1995;96:216–9. [PubMed] [Google Scholar]
- 6.Heath PT, Booy R, McVernon J, Bowen-Morris J, Griffiths H, Slack MP, et al. Hib vaccination in infants born prematurely. Arch Dis Child. 2003;88:206–10. doi: 10.1136/adc.88.3.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Berrington JE, Cant AJ, Matthews JN, O’Keeffe M, Spickett GP, Fenton AC. Haemophilus influenzae type b immunization in infants in the United Kingdom: effects of diphtheria/tetanus/acellular pertussis/Hib combination vaccine, significant prematurity, and a fourth dose. Pediatrics. 2006;117:e717–24. doi: 10.1542/peds.2005-0348. [DOI] [PubMed] [Google Scholar]
- 8.Losonsky GA, Wasserman SS, Stephens I, Mahoney F, Armstrong P, Gumpper K, et al. Hepatitis B vaccination of premature infants: a reassessment of current recommendations for delayed immunization. Pediatrics. 1999;103:e14. doi: 10.1542/peds.103.2.e14. [DOI] [PubMed] [Google Scholar]
- 9.Patel DM, Butler J, Feldman S, Graves GR, Rhodes PG. Immunogenicity of hepatitis B vaccine in healthy very low birth weight infants. Journal of Pediatrics. 1997;131:641–3. doi: 10.1016/s0022-3476(97)70078-7. [DOI] [PubMed] [Google Scholar]
- 10.Linder N, Handsher R, German B, Sirota L, Bachman M, Zinger S, et al. Controlled trial of immune response of preterm infants to recombinant hepatitis B and inactivated poliovirus vaccines administered simultaneously shortly after birth. Arch Dis Child Fetal Neonatal Ed. 2000;83:F24–7. doi: 10.1136/fn.83.1.F24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sood A, Singh D, Mehta S, Midha V, Kumar R. Response to hepatitis B vaccine in preterm babies. Indian J Gastroenterol. 2002;21:52–4. [PubMed] [Google Scholar]
- 12.Freitas da Motta MS, Mussi-Pinhata MM, Jorge SM, Tachibana Yoshida CF, Sandoval de Souza CB. Immunogenicity of hepatitis B vaccine in preterm and full term infants vaccinated within the first week of life. Vaccine. 2002;20:1557–62. doi: 10.1016/s0264-410x(01)00493-5. [DOI] [PubMed] [Google Scholar]
- 13.Omenaca F, Garcia-Sicilia J, Garcia-Corbeira P, Boceta R, Romero A, Lopez G, et al. Response of preterm newborns to immunization with a hexavalent diphtheria-tetanus-acellular pertussis-hepatitis B virus-inactivated polio and Haemophilus influenzae type b vaccine: first experiences and solutions to a serious and sensitive issue. Pediatrics. 2005;116:1292–8. doi: 10.1542/peds.2004-2336. [DOI] [PubMed] [Google Scholar]
- 14.O’Shea TM, Dillard RG, Gillis DC, Abramson JS. Low rate of response to enhanced inactivated polio vaccine in preterm infants with chronic illness. Clinical Research and Regulatory Affairs. 1993;10:49–57. [Google Scholar]
- 15.D’Angio CT, Maniscalco WM, Pichichero ME. Immunologic response of extremely premature infants to tetanus, Haemophilus influenzae, and polio immunizations. Pediatrics. 1995;96:18–22. [PubMed] [Google Scholar]
- 16.Oligbu G, Hsia Y, Folgori L, Collins S, Ladhani S. Pneumococcal conjugate vaccine failure in children: A systematic review of the literature. Vaccine. 2016;34:6126–32. doi: 10.1016/j.vaccine.2016.10.050. [DOI] [PubMed] [Google Scholar]
- 17.Khalak R, Pichichero ME, D’Angio CT. Three-year follow-up of vaccine response in extremely preterm infants. Pediatrics. 1998;101:597–603. doi: 10.1542/peds.101.4.597. [DOI] [PubMed] [Google Scholar]
- 18.Kirmani KI, Lofthus G, Pichichero ME, Voloshen T, D’Angio CT. Seven-year follow-up of vaccine response in extremely premature infants. Pediatrics. 2002;109:498–504. doi: 10.1542/peds.109.3.498. [DOI] [PubMed] [Google Scholar]
- 19.D’Angio CT, Heyne RJ, Duara S, Holmes LC, O’Shea TM, Wang H, et al. Immunogenicity of trivalent influenza vaccine in extremely low-birth-weight, premature versus term infants. Pediatr Infect Dis J. 2011;30:570–4. doi: 10.1097/INF.0b013e31820c1fdf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee FE, Halliley JL, Walsh EE, Moscatiello AP, Kmush BL, Falsey AR, et al. Circulating human antibody-secreting cells during vaccinations and respiratory viral infections are characterized by high specificity and lack of bystander effect. J Immunol. 2011;186:5514–21. doi: 10.4049/jimmunol.1002932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Halliley JL, Kyu S, Kobie JJ, Walsh EE, Falsey AR, Randall TD, et al. Peak frequencies of circulating human influenza-specific antibody secreting cells correlate with serum antibody response after immunization. Vaccine. 2010;28:3582–7. doi: 10.1016/j.vaccine.2010.02.088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Medina F, Segundo C, Campos-Caro A, Gonzalez-Garcia I, Brieva JA. The heterogeneity shown by human plasma cells from tonsil, blood, and bone marrow reveals graded stages of increasing maturity, but local profiles of adhesion molecule expression. Blood. 2002;99:2154–61. doi: 10.1182/blood.v99.6.2154. [DOI] [PubMed] [Google Scholar]
- 23.Rebhahn JA, Roumanes DR, Qi Y, Khan A, Thakar J, Rosenberg A, et al. Competitive SWIFT cluster templates enhance detection of aging changes. Cytometry A. 2016;89:59–70. doi: 10.1002/cyto.a.22740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Halliley JL, Tipton CM, Liesveld J, Rosenberg AF, Darce J, Gregoretti IV, et al. Long-Lived Plasma Cells Are Contained within the CD19(−)CD38(hi)CD138(+) Subset in Human Bone Marrow. Immunity. 2015;43:132–45. doi: 10.1016/j.immuni.2015.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee FE, Walsh EE, Falsey AR, Lumb ME, Okam NV, Liu N, et al. Human infant respiratory syncytial virus (RSV)-specific type 1 and 2 cytokine responses ex vivo during primary RSV infection. J Infect Dis. 2007;195:1779–88. doi: 10.1086/518249. [DOI] [PubMed] [Google Scholar]
- 26.Ohmit SE, Petrie JG, Cross RT, Johnson E, Monto AS. Influenza hemagglutination-inhibition antibody titer as a correlate of vaccine-induced protection. J Infect Dis. 2011;204:1879–85. doi: 10.1093/infdis/jir661. [DOI] [PubMed] [Google Scholar]
- 27.Qian Y, Wei C, Eun-Hyung Lee F, Campbell J, Halliley J, Lee JA, et al. Elucidation of seventeen human peripheral blood B-cell subsets and quantification of the tetanus response using a density-based method for the automated identification of cell populations in multidimensional flow cytometry data. Cytometry B Clin Cytom. 2010;78(Suppl 1):S69–82. doi: 10.1002/cyto.b.20554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Benoit A, Beran J, Devaster JM, Esen M, Launay O, Leroux-Roels G, et al. Hemagglutination Inhibition Antibody Titers as a Correlate of Protection Against Seasonal A/H3N2 Influenza Disease. Open Forum Infect Dis. 2015;2:ofv067. doi: 10.1093/ofid/ofv067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.D’Angio CT, Boohene PA, Mowrer A, Audet S, Menegus MA, Schmid DS, et al. Measles-mumps-rubella and varicella vaccine responses in extremely preterm infants. Pediatrics. 2007;119:e574–9. doi: 10.1542/peds.2006-2241. [DOI] [PubMed] [Google Scholar]
- 30.van den Berg JP, Westerbeek EA, Berbers GA, van Gageldonk PG, van der Klis FR, van Elburg RM. Transplacental transport of IgG antibodies specific for pertussis, diphtheria, tetanus, haemophilus influenzae type b, and Neisseria meningitidis serogroup C is lower in preterm compared with term infants. Pediatr Infect Dis J. 2010;29:801–5. doi: 10.1097/inf.0b013e3181dc4f77. [DOI] [PubMed] [Google Scholar]
- 31.Resch B, Resch E, Muller W. Should respiratory care in preterm infants include prophylaxis against respiratory syncytial virus infection? The case in favour. Paediatr Respir Rev. 2013;14:130–6. doi: 10.1016/j.prrv.2012.12.005. [DOI] [PubMed] [Google Scholar]
- 32.Amanna IJ, Carlson NE, Slifka MK. Duration of humoral immunity to common viral and vaccine antigens. N Engl J Med. 2007;357:1903–15. doi: 10.1056/NEJMoa066092. [DOI] [PubMed] [Google Scholar]
- 33.Fairfax KA, Kallies A, Nutt SL, Tarlinton DM. Plasma cell development: from B-cell subsets to long-term survival niches. Semin Immunol. 2008;20:49–58. doi: 10.1016/j.smim.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 34.Oracki SA, Walker JA, Hibbs ML, Corcoran LM, Tarlinton DM. Plasma cell development and survival. Immunol Rev. 2010;237:140–59. doi: 10.1111/j.1600-065X.2010.00940.x. [DOI] [PubMed] [Google Scholar]
- 35.Frasca D, Diaz A, Blomberg BB. Activation-Induced Cytidine Deaminase and Switched Memory B Cells as Predictors of Effective In Vivo Responses to the Influenza Vaccine. Methods Mol Biol. 2015;1343:107–14. doi: 10.1007/978-1-4939-2963-4_9. [DOI] [PubMed] [Google Scholar]
- 36.Zhang Y, Wang Y, Zhang M, Liu L, Mbawuike IN. Restoration of Retarded Influenza Virus-specific Immunoglobulin Class Switch in Aged Mice. J Clin Cell Immunol. 2016:7. doi: 10.4172/2155-9899.1000403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sharma SK, Roumanes D, Almudevar A, Mosmann TR, Pichichero ME. CD4+ T-cell responses among adults and young children in response to Streptococcus pneumoniae and Haemophilus influenzae vaccine candidate protein antigens. Vaccine. 2013;31:3090–7. doi: 10.1016/j.vaccine.2013.03.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pettengill MA, van Haren SD, Levy O. Soluble mediators regulating immunity in early life. Front Immunol. 2014;5:457. doi: 10.3389/fimmu.2014.00457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Salinas GF, De Rycke L, Barendregt B, Paramarta JE, Hreggvidstdottir H, Cantaert T, et al. Anti-TNF treatment blocks the induction of T cell-dependent humoral responses. Ann Rheum Dis. 2013;72:1037–43. doi: 10.1136/annrheumdis-2011-201270. [DOI] [PubMed] [Google Scholar]
- 40.Beck TC, Gomes AC, Cyster JG, Pereira JP. CXCR4 and a cell-extrinsic mechanism control immature B lymphocyte egress from bone marrow. J Exp Med. 2014;211:2567–81. doi: 10.1084/jem.20140457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xiang K, Ying G, Yan Z, Shanshan Y, Lei Z, Hongjun L, et al. Progress on adenovirus-vectored universal influenza vaccines. Hum Vaccin Immunother. 2015;11:1209–22. doi: 10.1080/21645515.2015.1016674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bai Y, Ye L, Tesar DB, Song H, Zhao D, Bjorkman PJ, et al. Intracellular neutralization of viral infection in polarized epithelial cells by neonatal Fc receptor (FcRn)-mediated IgG transport. Proc Natl Acad Sci U S A. 2011;108:18406–11. doi: 10.1073/pnas.1115348108. [DOI] [PMC free article] [PubMed] [Google Scholar]