Introduction
The abscopal effect involves shrinkage and/or remission of distant, unirradiated disease following local irradiation.1 It is rare and mechanistically remains under study, although recent case reports have seen incidence of the effect increasing with administration of immunomodulatory agents, such as the CTLA-4 inhibitor ipilimumab.2, 3, 4, 5, 6, 7 The abscopal effect appears to occur in response to radiation therapy and is immune system mediated,8 although research has predominantly focused on the use of conventional photon irradiation. Particle irradiation offers unique dose distributive benefits, concentrating dose in target tissue while sparing healthy tissue in a manner theoretically superior to photon.9 Meanwhile, heavy-ion irradiation, such as with carbon ions, offers additional biological effects owing to a superior linear energy transfer delivered on a per-particle basis.10 Carbon-ion radiation therapy (CIRT) has been used successfully in the treatment of recurrent rectal cancer.11
Here, we present 2 cases of patients who demonstrated an abscopal effect response following carbon-ion irradiation for metastatic recurrent colorectal cancer.
Case report 1
A 75-year-old man with recurrent colorectal cancer presented to his local hospital in November 2007 with a primary sigmoid colorectal cancer with liver metastasis (T4N2M1 stage IV according to the American Joint Committee on Cancer Staging Manual, 6th edition12). A high anterior resection was performed with partial hepatectomy. Pathological evaluation confirmed well-differentiated tubular adenocarcinoma, with TNM staging of T4N2M1, stage IV. In January 2010, he was diagnosed with a rectal cancer (T2N0, p-stage II), and ultra-low anterior resection was performed with colostomy. In November 2010, he presented with right abdominal pain. Computed tomography (CT) and positron emission tomography (PET)-CT demonstrated a 35-mm mass in the left side of the abdomen, with another 15-mm mass near and invading into the right common iliac artery. This was diagnosed as recurrence because of the gradual increase in size on serial imaging without changes in image or patient condition that would be associated with an infectious or inflammatory response (Fig 1). The patient was recommended and subsequently refused systemic chemotherapy, and was therefore referred to our institute for consideration of CIRT. The recurrent tumor near the right common iliac artery could not be treated because of its proximity to the small intestine. Because the patient presented with flank pain, it was determined to irradiate the 35-mm left-sided mass, with a surgical spacer later implanted to enable treatment of the 15-mm right common iliac artery mass. CIRT was delivered with a dose of 73.6 Gy (relative biological effectiveness [RBE]) in 16 fractions over 28 days in January 2011, derived from a clinical trial on using CIRT to treat rectal cancer recurrence.11 Treatment consisted of a single horizontal port and an off-vertical anterior and posterior port (Fig 2). PET-CT 1 month following treatment demonstrated notable reduction in both the treated and untreated tumor masses, with notable decrease of fludeoxyglucose accumulation (Fig 3). The patient's posttreatment course identified a lung metastasis on follow-up in February 2012; leucovorin calcium, fluorouracil, and oxaliplatin were administered. In June 2014, the patient had notable thrombocytopenia and was diagnosed with myelodysplastic syndrome. The patient died at 46 months post-CIRT because of myelodysplastic syndrome, with no recurrence or expansion of the 15-mm untreated mass near the right common iliac artery (Fig 4).
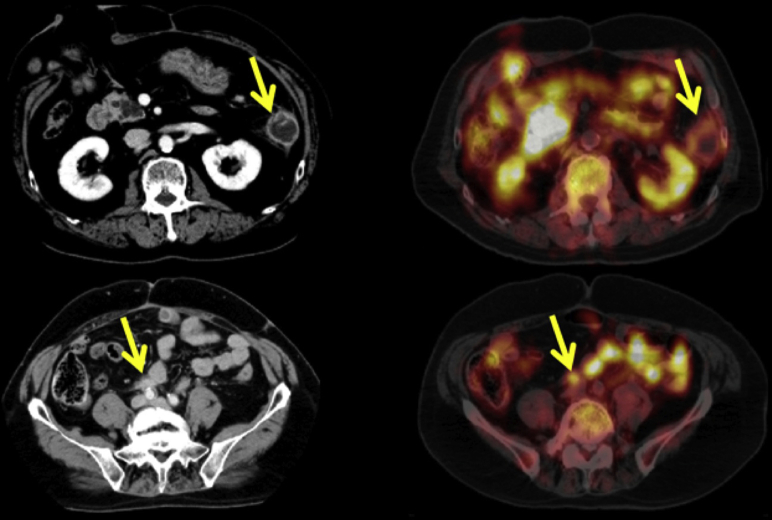
Figure 1.
Computed tomography (left) and positron emission tomography images displaying case 1's 35-mm left abdominal colorectal cancer recurrence (top) and the 15-mm nodule noted near the right common iliac artery (bottom).
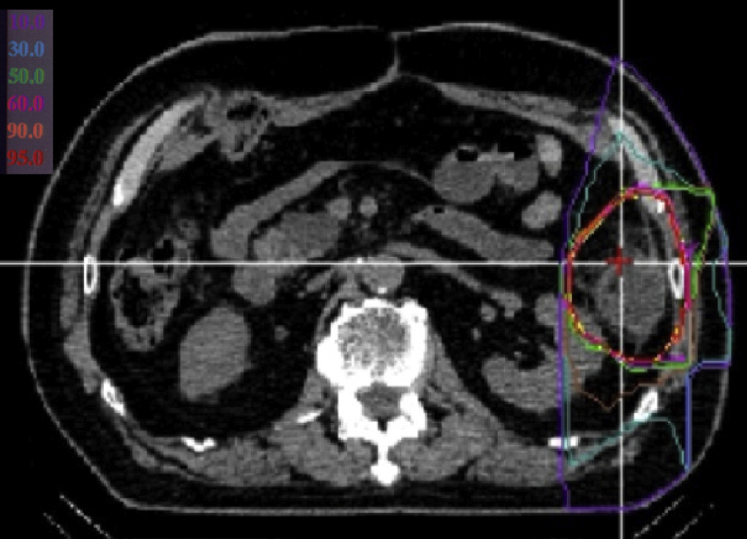
Figure 2.
Carbon-ion radiation therapy of the 35-mm left abdominal mass. Treatment was 73.6 Gy (relative biological effectiveness) in 16 fractions. One horizontal port and 2 off-vertical ports were used, indicated by red arrows.
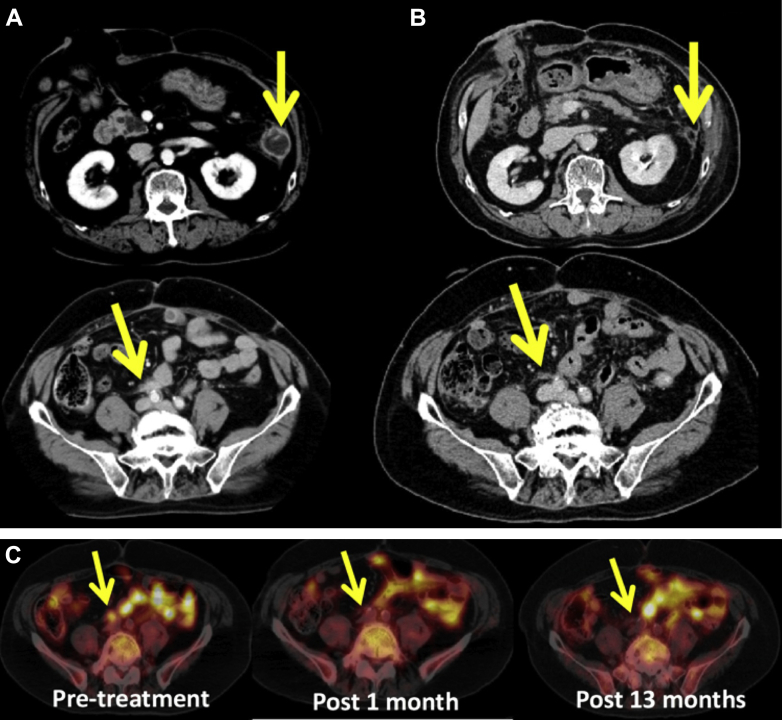
Figure 3.
(A) Pretreatment sizing of the left abdominal mass (top) and nodule in the right iliac artery region (bottom). The left abdominal mass was irradiated. (B) One month following treatment, regression is seen in both the irradiated mass (top) and nonirradiated nodule (bottom). (C) Positron emission tomography scans comparing pretreatment, 1 month posttreatment, and 13 months posttreatment, for the right iliac artery nodule.
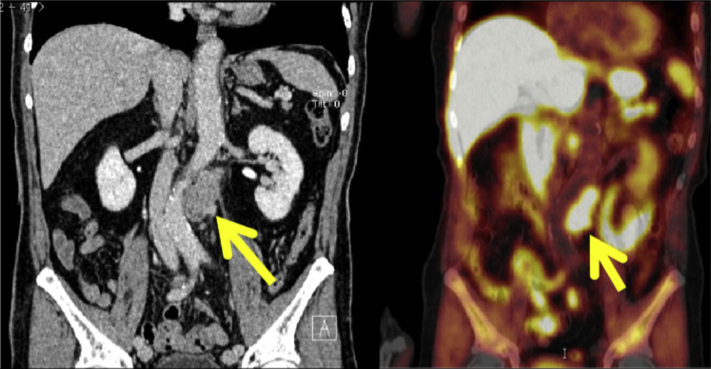
Figure 4.
Computed tomography scan of the recurrence of the patient's prior colorectal cancer in a lymph node near the abdominal aorta.
Case report 2
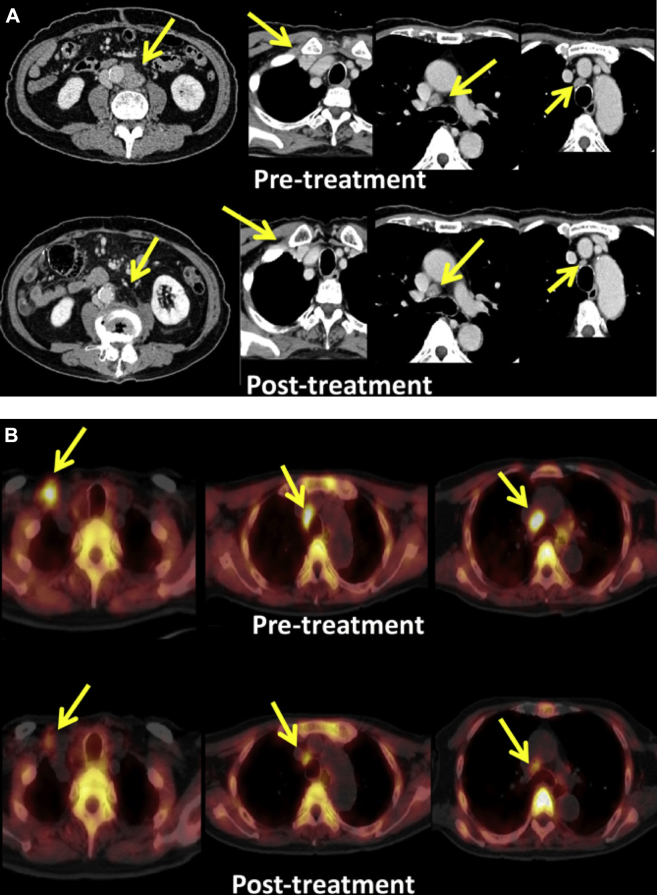
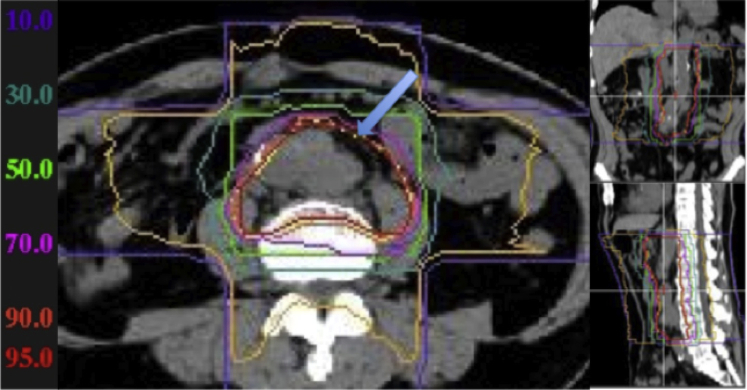
An 85-year-old man with recurrence of colorectal cancer presented to a local hospital in April 2008 with an ascending colon cancer (T3N1M0 stage IIIB12). A right hemicolectomy was performed. Adjuvant chemotherapy was not administered because of the patient's age and condition. In February 2009, the patient presented with back pain, and on CT a 45 mm disease recurrence was discovered in a lymph node near the abdominal aorta (Fig 4), with 10 mm masses seen under the mediastinum and right clavicle, respectively (Fig 5). In April 2009, the patient was referred to our institute for consideration of CIRT. A total of 50.4 Gy (RBE) in 12 fractions over 21 days was performed on the aortic lymph node, in line with an ongoing clinical trial treating isolated para-aortic lymph node recurrences (forthcoming). This consisted of opposing vertical and horizontal ports (Fig 6). Following treatment, the treated lymph node as well as the untreated right subclavian node shrank noticeably on follow-up imaging, whereas the mediastinal node remained stable. No additional treatment was administered. The patient is still alive today, 92 months following treatment, with no progression of disease.
Figure 5.
(A) Computed tomography and (B) positron emission tomography scans demonstrating before (top) and 13 months after (bottom) treatment changes. The aortic lymph node and nonirradiated 10-mm clavicular mass are both diminished.
Figure 6.
Two horizontal and 2 vertical ports were used for irradiation of the aortic lymph node, with 50.4 Gy (relative biological effectiveness) delivered in 12 fractions.
Discussion and conclusions
Here, 2 patients exhibiting an abscopal-like effect following carbon-ion irradiation have been presented, with disease remission of 46 months in case 1, and remission and stable disease of 92 months in case 2. In case 1, a lung metastasis was seen following treatment and chemotherapy was administered; however, reduction in size of the nonirradiated tumor was noted at 1 month postirradiation, before administration of chemotherapy. For case 2, no additional treatment was used, and the reduction of the clavicular tumor is easily noted on PET. Between the patients, abscopal effects were both seen in lymph nodes “downstream” of the treated site; however, delivered dose as well as method varied between the 2 patients. At our institute, recurrent lymph nodes are treated with 52.8 Gy (RBE), whereas other recurrences are treated with 73.6 Gy (RBE); it is notable that both doses lead to an abscopal-like effect. Whether this indicates an underlying susceptibility of these 2 patients' disease or specific characteristic of these patients' immune systems or if the generation of the abscopal effect reflected any advantage of the carbon-ion beam is difficult to discern.
The abscopal effect was reported for the first time in 195313; thereafter, clinical reports have been seen for numerous disease histologies treated with conventional irradiation, including malignant lymphoma,14 hepatocellular carcinoma,15 cervical carcinoma,16 melanoma,6 and others. Abscopal effects for colorectal cancer have been seen in mouse models,17, 18, 19 although there appear to be no clinical reports at the time of this writing. Numerous clinical trials to investigate combination radiation therapy-immunotherapy are registered with ClinicalTrials.gov,20 but a mechanism for the effect remains elusive.
Current research suggests the postirradiation development of an in situ tumor vaccine21 leading in rare cases to a systemwide response to tumor tissue. This involves potentiation of the target tumor by irradiation, and induction of a robust response of CD8+ effector T cells to the target tumor. Radiation is both immunosuppressing and immunostimulating; following irradiation, tumors may translocate a variety of recognizable antigens to their surface, such as calreticulin, potentiating recognition, and response by the immune system. We have reviewed this previously.22 Gameiro and colleagues have recently produced evidence suggesting that cells surviving irradiation exhibit common T-cell sensitivities, which may elaborate on expanded immune system activity to the postirradiation primary tumor.23 Comparatively, other work has investigated enhanced immune response when immunotherapy is combined with high-dose ablative photon radiation therapy; this may suggest that combination CIRT-immunotherapy may further potentiate abscopal-type reactions.24 Nonetheless, the mechanism by which out-of-field secondary disease is revealed to the immune system remains undetermined.
Full abscopal effects have been seen with and without immunotherapy, although the addition of immunotherapy appears to significantly improve the rate.4 Previous experimental trials with CIRT in mice have demonstrated metastasis suppression as well as abscopal effects,25, 26 but this is the first report of abscopal effects following heavy-ion irradiation in human patients. Further, this did not include usage of immunotherapy. How this mechanism may relate to abscopal effects seen in photon irradiation and how this may be further enhanced through the use of immunotherapy remain to be seen. In general, further work is needed to better elucidate the mechanism by which the abscopal effect operates.
Footnotes
Conflicts of interest: None.
References
- 1.Siva S., MacManus M.P., Martin R.F. Abscopal effects of radiation therapy: A clinical review for the radiobiologist. Cancer Lett. 2015;356:82–90. doi: 10.1016/j.canlet.2013.09.018. [DOI] [PubMed] [Google Scholar]
- 2.Grimaldi A.M., Simeone E., Giannarelli D. Abscopal effects of radiotherapy on advanced melanoma patients who progressed after ipilimumab immunotherapy. Oncoimmunology. 2014;3:e28780. doi: 10.4161/onci.28780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Golden E.B., Demaria S., Schiff P.B. An abscopal response to radiation and ipilimumab in a patient with metastatic non-small cell lung cancer. Cancer Immunol Res. 2013;1:365–372. doi: 10.1158/2326-6066.CIR-13-0115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tang C., Wang X., Soh H. Combining radiation and immunotherapy: A new systemic therapy for solid tumors? Cancer Immunol Res. 2014;2:831–838. doi: 10.1158/2326-6066.CIR-14-0069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Formenti S.C., Demaria S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. J Natl Cancer Inst. 2013;105:256. doi: 10.1093/jnci/djs629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Postow M.A., Callahan M.K., Barker C.A. Immunologic correlates of the abscopal effect in a patient with melanoma. N Engl J Med. 2012;366:925–931. doi: 10.1056/NEJMoa1112824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stamell E.F., Wolchok J.D., Gnjatic S. The abscopal effect associated with a systemic anti-melanoma immune response. Int J Radiat Oncol Biol Phys. 2013;85:293–295. doi: 10.1016/j.ijrobp.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Demaria S., Ng B., Devitt M.L. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int J Radiat Oncol Biol Phys. 2004;58:862–870. doi: 10.1016/j.ijrobp.2003.09.012. [DOI] [PubMed] [Google Scholar]
- 9.Tsujii H., Kamada T., Shirai T. Carbon-ion radiotherapy. In: Tsuji H., Kawahara K., editors. Springer; Tokyo: 2014. [Google Scholar]
- 10.Tinganelli W., Durante M., Hirayama R. Kill-painting of hypoxic tumours in charged particle therapy. Sci Rep. 2015;5:17016. doi: 10.1038/srep17016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamada S., Kamada T., Ebner D.K. Carbon-ion radiotherapy for pelvic recurrence of rectal cancer. Int J Radiat Oncol Biol Phys. 2016;96:93–101. doi: 10.1016/j.ijrobp.2016.04.022. [DOI] [PubMed] [Google Scholar]
- 12.American Joint Committee on Cancer . AJCC Cancer Staging Manual. In: Greene F.L., Page D.L., Fleming I.D., editors. Springer New York; New York, NY: 2002. [Google Scholar]
- 13.Mole R.H. Whole body irradiation; radiobiology or medicine? Br J Radiol. 1953;26:234–241. doi: 10.1259/0007-1285-26-305-234. [DOI] [PubMed] [Google Scholar]
- 14.Antoniades J., Brady L.W., Lightfoot D.A. Lymph-angiographic demonstration of abscopal effect in patients with malignant-lymphomas. Int J Radiat Oncol Biol Phys. 1977;2:141–147. doi: 10.1016/0360-3016(77)90020-7. [DOI] [PubMed] [Google Scholar]
- 15.Okuma K., Yamashita H., Niibe Y. Abscopal effect of radiation on lung metastases of hepatocellular carcinoma: a case report. J Med Case Rep. 2011;5:111. doi: 10.1186/1752-1947-5-111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takaya M., Niibe Y., Tsunoda S. Abscopal effect of radiation on toruliform para-aortic lymph node metastases of advanced uterine cervical carcinoma–A case report. Anticancer Res. 2007;27:499–503. [PubMed] [Google Scholar]
- 17.Strigari L., Mancuso M., Ubertini V. Abscopal effect of radiation therapy: Interplay between radiation dose and p53 status. Int J Radiat Biol Radiol Phys. 2014;90:248–255. doi: 10.3109/09553002.2014.874608. [DOI] [PubMed] [Google Scholar]
- 18.Dewan M.Z., Galloway A.E., Kawashima N. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin Cancer Res. 2009;15:5379–5388. doi: 10.1158/1078-0432.CCR-09-0265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Deng L., Liang H., Burnette B. Irradiation and anti-PD-L1 treatment synergistically promote antitumor immunity in mice. J Clin Invest. 2014;124:687–695. doi: 10.1172/JCI67313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kang J., Demaria S., Formenti S. Current clinical trials testing the combination of immunotherapy with radiotherapy. J ImmunoTher Cancer. 2016;4:51. doi: 10.1186/s40425-016-0156-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Durante M., Brenner D.J., Formenti S.C. Does heavy ion therapy work through the immune system? Int J Radiat Oncol Biol Phys. 2016;96:934–936. doi: 10.1016/j.ijrobp.2016.08.037. [DOI] [PubMed] [Google Scholar]
- 22.Ebner D.K., Tinganelli W., Helm A. The immunoregulatory potential of particle radiation in cancer therapy. Front Immunol. 2017;8:293. doi: 10.3389/fimmu.2017.00099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gameiro S.R., Malamas A.S., Bernstein M.B. Tumor cells surviving exposure to proton or photon radiation share a common immunogenic modulation signature, rendering them more sensitive to T cell-mediated killing. Int J Radiat Oncol Biol Phys. 2016;95:120–130. doi: 10.1016/j.ijrobp.2016.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bernstein M.B., Krishnan S., Hodge J.W. Immunotherapy and stereotactic ablative radiotherapy (ISABR): A curative approach? Nat Rev Clin Oncol. 2016;95:120–130. doi: 10.1038/nrclinonc.2016.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shimokawa T., Ma L., Ando K. The future of combining carbon-ion radiotherapy with immunotherapy: Evidence and progress in mouse models. Int J Particle Ther. 2016;3:61–70. doi: 10.14338/IJPT-15-00023.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Matsunaga A., Ueda Y., Yamada S. Carbon-ion beam treatment induces systemic antitumor immunity against murine squamous cell carcinoma. Cancer. 2010;116:3740–3748. doi: 10.1002/cncr.25134. [DOI] [PubMed] [Google Scholar]