Abstract
There are limited data on the incidence and management of acute faecal incontinence with diarrhoea in the ICU. The FIRST™ Observational Study was undertaken to obtain data on clinical practices used in the ICU for the management of acute faecal incontinence with diarrhoea in Germany, UK, Spain and Italy. ICU-hospitalised patients ≥18 years of age experiencing a second episode of acute faecal incontinence with diarrhoea in 24 h were recruited, and management practices of acute faecal incontinence with diarrhoea were recorded for up to 15 days. A total of 372 patients had complete data sets; the mean duration of study was 6.8 days. At baseline, 40% of patients experienced mild or moderate-to-severe skin excoriation, which increased to 63% in patients with acute faecal incontinence with diarrhoea lasting >15 days. At baseline, 27% of patients presented with a pressure ulcer, which increased to 37%, 45% and 49% at days 5, 10 and 15, respectively. Traditional methods (pads, sheets and tubes) were more commonly used compared to faecal management systems during days 1–4 (76% vs. 47% faecal management system), while the use of a faecal management system increased to 56% at days 5–9 and 61% at days 10–15. At baseline, only 26% of nurses were satisfied with traditional management methods compared to 69% with faecal management systems. For patients still experiencing acute faecal incontinence with diarrhoea after 15 days, 82% of nurses using a faecal management systems to manage acute faecal incontinence with diarrhoea were satisfied or very satisfied, compared to 37% using traditional methods. These results highlight that acute faecal incontinence with diarrhoea remains an important healthcare challenge in ICUs in Europe; skin breakdown and pressure ulcers remain common complications in patients with acute faecal incontinence with diarrhoea in the ICU.
Keywords: Acute faecal incontinence, ICU, prevalence, skin breakdown, faecal management system
Introduction
Faecal incontinence presents a major challenge for healthcare professionals, and has been reported to affect a substantial number of patients in acute care.1 The condition is associated with a number of clinical issues, including the risk of perineal dermatitis and cross-contamination with pathogens responsible for healthcare-associated infections (HCAIs).1–3 The management of faecal incontinence in the acute care setting also requires considerable nursing time and treating the associated complications can result in increased hospital stays and hospital costs.4–7 Although there is an increasing body of evidence regarding the prevalence and consequences of chronic faecal incontinence in community and nursing homes, there is currently a lack of information regarding the prevalence and management of acute faecal incontinence with diarrhoea (AFId) in acutely or critically ill hospitalised patients, with only a small number of reports describing the specific problem of AFId in the ICU and its incidence.8–19 Similarly, there is little available data on the incidence of the clinical consequences of AFId in this setting.9–21
To better understand specific issues related to AFId in European ICUs, the FIRST™ (Faecal Incontinence Re-evaluation STudy) programme was initiated by a group of healthcare professionals involved in the management of critical care patients, supported by an educational grant from ConvaTec. The FIRST™ programme aims to gather data on AFId and raise awareness of the prevalence, associated clinical consequences and current management of the condition.22 As part of the initiative, a definition of AFId was agreed upon to distinguish it from other descriptions of faecal incontinence that are not relevant for the ICU. “AFId” is thus defined as at least two episodes of faecal incontinence with diarrhoea – liquid or semi-liquid stool (according to the Bristol Stool chart23) – in a 24-h time period. It should be noted that AFId does not refer to continent patients with diarrhoea who are still able to use toilet facilities.24
This initiative included the FIRST™ Survey, which aimed to provide a snapshot of AFId in Europe across ICUs in four European countries (Germany, Italy, Spain and UK).9,22 Importantly, the FIRST™ Survey identified low awareness of the clinical challenges associated with AFId and substantial variability in management strategies, possibly reflecting the absence of management recommendations for AFId in the countries surveyed. To address this, a set of expert recommendations was developed to aid carers in understanding AFId and its clinical consequences and to provide guidance on the most appropriate management options, depending on individual patient risk factors and comorbidities.24
Following the results of the FIRST™ Survey, which were published in 2012, a second phase of the FIRST™ programme was initiated – a prospective, observational, multicentre study to obtain data on current routine clinical practices in the management of AFId in the critical care setting in selected European countries through the use of site staff assessments. Here we present the results of the FIRST™ Observational Study, which provide insights into the real-world clinical challenges associated with AFId and its management in the ICU, which may also provide additional information and support for the use of expert consensus recommendations for the management of AFId in daily clinical practice.
Methodology
Study design
The primary objective of this non-interventional, observational study was to obtain data on current routine clinical practices in the management of AFId in Germany, Spain, Italy and the UK. Secondary objectives were to document the incidence and prevalence of AFId, to understand the consequences of AFId on the integrity of the perineal skin and the development of pressure ulcers (PUs), to document staff satisfaction with current management methods and to understand the total time spent managing AFId.
Patients and data collection
In August 2011, the initial recruitment of patients began in 46 sites across the four countries. Inclusion criteria were: male and female ICU hospitalised patients ≥18 years of age experiencing a second episode of AFId in 24 h. Other than the minimum age limit, there were no exclusion criteria as the study aim was to observe and record AFId management in real-life conditions. The primary and secondary objectives were monitored over an observational time period of 15 days, but patients left the study earlier if symptoms related to AFId resolved or they were discharged from the ICU. By the time the final patient was recruited in July 2012, 435 patients had been enrolled. Patient data were obtained prospectively by clinicians via an electronic data capture system using an electronic case report form (eCRF) provided for this study. A daily diary was also completed by nursing staff to record the number of patients with AFId, the number of new patients with AFId, the number of patients present in the ICU and the number of patients who left the ICU in order to determine incidence and prevalence. Only complete sets of data for each patient were used for the analysis. Written informed consent was obtained from all patients before enrolment. The institutional review board at each participating centre approved the study, which was conducted in accordance with the principles of Good Clinical Practice, the provisions of the Declaration of Helsinki, and other applicable local regulations.
AFId management methods
For the purpose of the analysis, patients were described as being treated with either traditional management methods, comprising pads, sheets, faecal collectors and tubes, or with a faecal management system (FMS). This was an observational study, which did not evaluate the outcome of an intervention and only recorded outcomes when they occurred.
Data analyses
Descriptive analyses were performed for both the primary and secondary objectives. Categorical variables were reported using frequency tables. Continuous variables were summarised using means, standard deviations, and minimum and maximum values. In the event that a continuous variable was assessed over time, changes in the variable were reported using change from baseline values (visit value − baseline value). No statistical inferences were derived for this descriptive study.
The FIRST™ Observational Study advisory board
The study concept and design of the FIRST™ Observational Study was guided and supported by an international board of experts from across Europe. Each member is directly involved in the management and care of critically ill patients.
Results
Patient characteristics and disposition
Of the 435 patients who were enrolled, complete data sets were available for 372 patients and included in the analysis; any unresolved data entries for any patient were classified as “incomplete”. Table 1 shows the distribution of patients per centre and per country. The mean age was 60.6 years, and 60% of patients were male. The mean weight of patients was 77.2 kg and mean BMI was 26.9.
Table 1.
Patient distribution.
| Country | Centres (n) | Patients enrolled (n) | Patients analysed (n) |
|---|---|---|---|
| Germany | 10 | 88 | 85 |
| Spain | 13 | 127 | 91 |
| Italy | 13 | 142 | 125 |
| UK | 10 | 78 | 71 |
| Total | 46 | 435 | 372 |
The mean time of the patient in the study was 6.8 days; only 12.9% of patients remained for the full 15-day study period (Table 2). The main reason for early exit from the study was cessation of AFId (58.6%) and discharge from the ICU (28.1%). Surgical and medical patients were included (Table 3). The proportion of patients with enteral feeding (≥80%) and mechanical ventilation (>75%) remained high throughout the 15-day study period, while the percentage of patients on antibiotics tended to decrease, from 88% on day 1, 83% on day 5, 75% on day 10 to 73% on day 15.
Table 2.
Duration of study and reasons for early cessation.
| % Patients (n = 372) | |
|---|---|
| Patients completing all 15 study days | 12.9 |
| Primary reason for discontinuation | |
| Death | 10.2 |
| Cessation of AFId | 58.6 |
| Discharge from ICU | 28.1 |
| Other (hospital discharge, consent withdrawal, etc.) | 3.1 |
| Mean duration of AFId | 6.8 days |
Table 3.
Reasons for ICU hospitalisation.
| % Patients | |
|---|---|
| Pulmonary | 27.4 |
| Cardiovascular | 14.0 |
| Infection | 12.9 |
| Neurological | 11.0 |
| Gastrointestinal | 9.4 |
| Trauma | 8.3 |
| Other (metabolic, renal, malignancy, etc.) | 17.0 |
The baseline sequential organ failure assessment (SOFA) score (index of disease severity) was 7.2 for patients using an FMS at baseline and 6.7 for those using traditional methods. There was little variation in the SOFA score throughout the study (6.8 on days 1 and 5, 7.1 on day 10 and 7.5 on day 15). Baseline Braden scores (risk of PU) were 11.0 and 11.3 for those using FMS or traditional methods, respectively (Figure 1).
Figure 1.
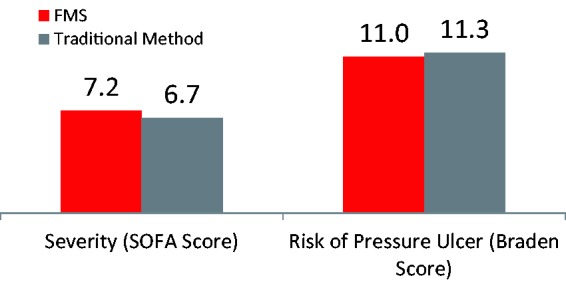
Baseline characteristics for patients managed with traditional methods or FMS.
FMS: faecal management system; SOFA: sequential organ failure assessment.
Incidence and prevalence of AFId
The incidence of AFId, calculated as the ratio of the number of patients with at least one incidence of faecal incontinence over the total time at risk among all patients, was 795 per 10,000 patient-days (Figure 2). The prevalence of AFId, the ratio of the number of patients with AFI over the number of patients in the ICU, was 17.4% (Figure 3). Some variations were observed in the incidence and prevalence between countries.
Figure 2.
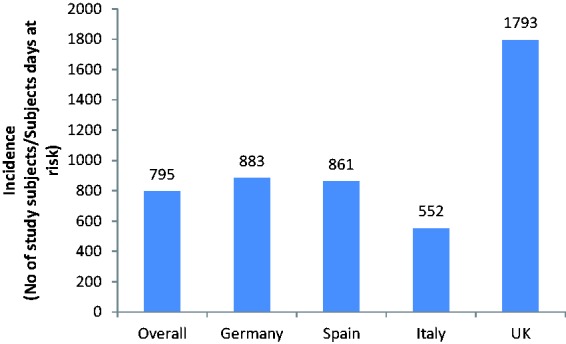
Incidence of AFId overall and for each country.
Figure 3.

Prevalence of AFId overall and for each country.
Skin integrity
At baseline, approximately 40% of patients overall experienced either mild or moderate-to-severe skin excoriation. This proportion increased in patients with AFId of longer duration (5–10 days); in patients with AFId lasting 15 days, mild skin excoriation was observed in approximately 37% of patients and moderate-to-severe excoriation in 25.5% (Figure 4). At baseline, 27% of patients presented with at least one PU in the sacral, perineal/buttocks area. This increased to 37%, 45% and 49% at days 5, 10 and 15, respectively. The risk of developing a PU was high for all patients throughout the study (range 11.0–12.4). No notable differences were seen in increased skin excoriation or increased risk of developing a PU between patients receiving traditional management vs. FMS over the course of the observational period.
Figure 4.
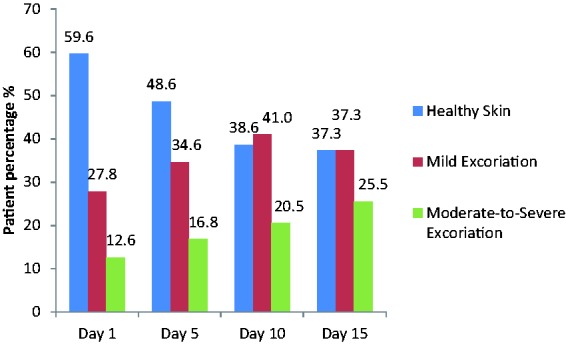
Skin integrity.
Management methods and nurse satisfaction
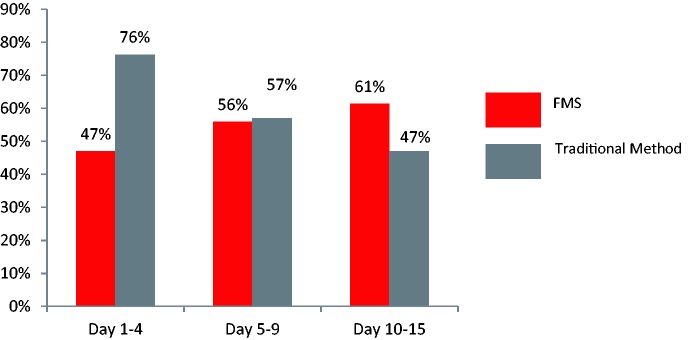
Various management methods were used over the course of the study: diapers and pads, incontinence sheets, faecal collectors, faeces-diverting tubes (all classed as traditional management methods) and FMSs. Overall, traditional methods, i.e. pads, sheets and tubes, were most commonly used to manage AFId during the initial days (days 1–4) of the study (76% traditional vs. 47% FMS). The use of FMS increased over the course of the observational period to 56% at days 5–9 and 61% at days 10–15 (Figure 5). The exact usage of FMS to manage AFId varied from country to country; however, Spain was found to implement FMS as the sole management method in only 4% of cases at baseline, whereas the UK implemented FMS with a much higher frequency, in around 20% of cases (data not shown).
Figure 5.
Management method.
FMS: faecal management system.
In the cases where FMS was implemented, nurses were more satisfied managing AFId than with traditional methods (Figure 6). At baseline, only 26% of nurses were satisfied with traditional management methods compared to 69% with FMS. For patients who were still experiencing AFId after 15 days, 82% of nurses using FMS to manage AFId were satisfied or very satisfied compared to 37% of nurses using traditional methods.
Figure 6.
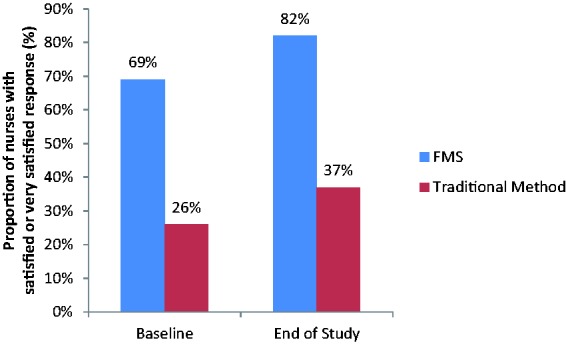
Nurse satisfaction with AFId management methods.
FMS: faecal management system.
Discussion
The results of the FIRST™ Observational Study highlight that AFId remains an important healthcare challenge in ICUs in Europe. In this observational study, the overall prevalence of AFId was 17.4% which is in broad agreement with the estimated prevalence of 9–37% reported in the FIRST™ Survey.22 Variation in the prevalence of AFId was observed between countries, as was also seen in the FIRST™ Survey.22 Wide variation in the reported prevalence of AFId in the acute care setting has been noted in previous investigations, ranging from 6.6 to 33% in two studies in the US,5,22 6.4% in a Spanish study10 and 22.4% in a UK-based study.9 While definitions in the two FIRST™ studies were standardised, the patient case-mix present in the ICU at any given moment and differences in the use of antibiotics or parenteral feeding can contribute to variations in the prevalence of AFId, in addition to other unquantifiable variances.
One of the primary goals in the management of AFId is maintenance of skin integrity. Importantly, at baseline, around 40% of patients already experienced either mild or moderate-to-severe skin excoriation. This percentage further increased in patients with long-lasting AFId. Moderate-to-severe excoriation and PUs in the sacral, perineal/buttocks areas were also prevalent. FMSs are specifically designed to protect the skin from contact with faecal waste that can cause skin breakdown, moisture wounds, and lead to the development of PUs.25 The majority of the patients in the study did not have an FMS until 5–9 days after the onset of AFId, and even after long-lasting AFId (>10 days) 39% still did not have an FMS in situ. In this observational study, no notable differences were seen in skin outcomes between management methods. However, the proportion of patients with an FMS increased over the course of the study compared with traditional methods. This might suggest that caregivers believe that an FMS is important in avoiding incontinence-associated complications and reducing the time spent managing episodes of diarrhoea in patients requiring longer stays in the ICU or may reflect the use of FMS for patients who are more critically ill. FMS use was slightly higher in patients with higher SOFA score at the start of the study. However, this observational study was not designed to investigate the reasons why a particular management method was chosen, or to investigate clinical outcomes associated with a particular management approach. Similarly, to avoid bias, it was not possible to collect data on reasons for changes in management during the observational period.
Use of FMS was associated with substantially higher satisfaction in managing AFId among nurses compared with traditional methods, particularly for long-lasting AFId. At baseline, only 26% of nurses reported satisfaction with traditional methods compared to 69% with an FMS. At the end of the study, the percentage of nurses reporting satisfaction with an FMS increased to 82%, while only 37% were satisfied with traditional care. Greater satisfaction with FMS may be related to the possibility that it has the potential to reduce the time spent managing AFId episodes and better control of unpleasant odour compared to traditional pads and sheets. In patients who are managed using an FMS, once the device is in place it is difficult to ascertain nursing time required for managing incontinence episodes. In this observational study, approximately 0.7 nursing hours per day were required to manage AFId episodes using traditional management methods. In the FIRST™ Survey it was found that approximately 10–20 min are spent managing an AFId episode, and most nurses estimated that three nursing staff were required.22 Thus, one patient experiencing five episodes of AFId in a 24-h period managed by three nursing staff for 15 min each episode would consume 3.75 h of nursing time.22 This highlights that, although the cost of pads may be low and the time for staff training short, the overall costs of managing frequent and persistent AFId episodes can be significant. Overall costs must take into consideration the time spent by care providers to manage each episode, the large number of replacement pads used, and the additional costs of managing the spread of infections through inadequately contained, infected faeces. In a previous study, it has already been noted that there is significant utilisation of healthcare resources, including nursing time, associated with the management of AFId that may not be widely recognised.6
The FIRST™ Survey reported a “low to moderate” appreciation of the problems associated with AFId among both nurses and physicians, and that despite a range of management methods available, around 50% of the respondents reported a lack of standard protocols or guidelines in their respective ICUs.22 Although not available at the time of this observational study, standard management plans or protocols could have a positive impact on reducing the clinical complications associated with AFId whilst ensuring the best use of time and resources. In this regard, a set of expert recommendations were published whilst this study was ongoing to aid carers managing AFId and its clinical consequences, and to help them to administer the most appropriate management options, depending on individual patient risk factors and comordibities.24 The treatment algorithm recommends that after diagnosis of AFId (a second episode of faecal incontinence with diarrhoea (liquid or semi-liquid stool, Bristol stool chart type 6--7) in 24 h), patients are categorised as:
those with good prognosis, and
those with, or at risk of developing, complications.
As the former group are well nourished/hydrated and are predicted to have a short hospital stay, they can be managed using traditional methods. The group having AFId with, or at risk of, developing complications includes those with a risk of skin breakdown in the perineal region; at risk of cross infection; reduced mobility; long-lasting diarrhoea; other clinical conditions. In such patients, an FMS is recommended whenever possible.
This study does have a number of limitations, particularly the limitations inherent in the design of an observational study, which may be subject to bias. The study was carefully designed to minimise potential bias, although clearly this reduces the possibility to correlate the observations with factors influencing management choices or impact on clinical outcomes. Another limitation of this study is the selection of hospitals and ICUs across the different countries, which may vary significantly. The aim was to ensure a cross-sectional selection of hospitals for example according to size and teaching status, but since only approximately 10 centres from each country were included it is challenging to be fully representative of hospital-based clinical practice in these countries, and this may also have an influence on the use or availability of different management methods for AFId. However, even with these limitations, this observational study does provide additional insights into current clinical practice regarding the management of AFId. Since the completion of this study, and with the availability of expert recommendations, it may now be timely to review how this increasing understanding of the impact and consequences of AFId in the ICU may have influenced management practices over the short term, and ideally help identify how this complex clinical condition could be better managed in the future.
Conclusions
The FIRST™ Observational Study provides additional confirmation that there are still clinical challenges and substantial variability in the management of AFId in Europe and that AFId remains an important healthcare challenge in ICUs in Europe; skin breakdown and PUs remain common complications in patients with AFId in the ICU. Variations in the observed results between countries also suggest that there are differences between European countries in how AFId is managed in ICUs. It is hoped that this additional observation data on current management of AFId, together with published expert recommendations,24 may help to further raise awareness of this important component of ICU management and improve the current management of AFId in the ICU.
Centres participating in the FIRST™ Observational Study
Germany
Universitätsmedizin Rostock
Hausanschrift, Schillingallee 35, Postfach 100888. 18057 Rostock
BG Berufsgenossenschaftliche Unfallklinik Tuebingen
BG Klinik Tübingen, Schnarrenbergstrasse 95, 72076 Tübingen
Städt. Klinikum Bogenhausen München
Englschalkinger Strasse 77, 81925 München
Universitätsklinikum Bonn
Sigmund-Freud-Str. 25 53127 Bonn
Marien-Hospital Aachen
Zeise 4 52066 Aachen
Universitätsklinikum Regensburg
Franz-Josef-Strauss-Allee 11, 93053 Regensburg
Universtitäsklinikum Magdeburg
Leipziger Str. 44, 39120 Magdeburg
Klinik Aschersleben-Stassfurt
Eislebener Str. 7a, 06449 Aschersleben
Universitätsklinikum Charité Berlin
Charité Campus Virchow-Klinikum, Augustenburger Platz 1, 13353 Berlin
Vivantes Neukölln, Berlin
Vivantes Netzwerk f. Gesundheit GmbH, Klinikum Neukölln, Rudowerstr. 48, 12351 Berlin
Italy
Azienda Ospedaliera Universitaria Pisana
Via Paradisa 2, 56126 Pisa
Azienda Ospedaliera Universitaria Integrata Verona (Borgo Roma)
Piazzale L. A. Scuro 10, 37135 Verona
Azienda Ospedaliera Universitaria Integrata Verona (Borgo Trento)
Piazzale A. Stefani 1, 37127 Verona
Azienda Ospedaliera San Filippo Neri
Piazza S. Maria Della Pieta' 5, 00135 Rome
Azienda Ospedaliera Careggi
Viale Morgagni 85, 50134 Florence
Ospedale Perrino
Strada Statale 7 Mesagne, 72100 Brindisi
Ospedale Monaldi
Via Leonardo Bianchi 1, 80131 Napoli
Azienda Ospedaliera San Martino
Largo Rosanna Benzi, 1 60132 Genoa
Policlinico S. Orsola Malpighi
Via Pietro Albertoni 15, 40121 Bologna
Azienda Ospedaliera Nuovo S: Giovanni Di Dio Firenze
Via Torre Galli 3, 50122 Florence
Ospedale Policlinico Tor Vergata
Viale Oxford 81, 00155 Rome
Ospedale S. Giovanni Battista “Le Molinette"
Corso Bramante 88/90, 10126 Turin
Azienda Di Rilievo Nazionale E Di Alta Specializzazione Civico G. Di Cristina Fatebenefratelli
Piazzale Leotta 4, 90127 Palermo
Spain
Hospital Universitari Vall D'Hebron
Passeig De La Vall d'Hebron, 119-129 08035, Barcelona
Hospital Universitario Puerta De Hierro
Majadahonda Manuel de Falla, 1. 2822.
Hospital Universitario La Paz
P° De La Castellana, 261 28046, Madrid
Hospital Universitari de Bellvitge
Calle Feixa Llarga S/N. L'Hospitalet De Llobregat. 08907, Barcelona
Hospital A Coruña
Xubias De Arriba S/N 15006 A Coruña
Hospital General Universitario De Elche
Camino De La Almazara 11, 3202 Elche, Alicante
Hospital Universitario Ramon Y Cajal
Ctra. De Colmenar Viejo Km. 9,100, 28034, Madrid
Hospital Xeral-Cies
Rua Pizarro 22, 36204, Vigo
Hospital Clinic De Barcelona
Calle Villaroel 170, 08036, Barcelona
Hospital La Fe
Avda Campanar 21, 46009, Valencia
Hospital Txagorritxu
Calle Jose Atxotegi S/N 01006, Vitoria
Hospital Universitario Central De Asturias
Calle Celestino Villamil, S/N 33006, Oviedo
Hospital Universitario Rio Hortega
Calle Dulzaina 2, 47012, Valladolid
Hospital Universitario Virgen Del Rocio
Avda. Manuel Siurot S/N. 41013, Seville
United Kingdom
Hull Royal Infirmary General Intensive Care Unit
Anlaby Road, Hull
Southampton General
Tremona Road, Southampton
Queen Alexandra
Cosham Portsmouth
Derriford Hospital
Crownhill, Plymouth, Devon
St James University Hospital
Leeds General Infirmary, Great George Street, Leeds
Leeds General Infirmary
Great George Street, Leeds
Musgrove Park Hospital
Taunton, Somerset
Royal Blackburn Hospital
Haslingden Road, Blackburn
Airedale
Skipton Rd, Steeton, Keighley, West Yorkshire
Warwick Hospital
Lakin Road, Warwick
Acknowledgement
™FIRST is a trademark of ConvaTec Inc.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was designed and fully funded by ConvaTec. Study centres were not limited to those using ConvaTec proprietary FMSs. Data collection and analysis was carried out by ConvaTec. Support for manuscript preparation was by IntraMed, Milan, with revision by the Steering Committee members and final review for approval.
References
- 1.Rees J, Sharpe A. The use of bowel management systems in the high-dependency setting. Br J Nursing 2009; 18: S19–S24. [DOI] [PubMed] [Google Scholar]
- 2.Bardsley A. More guidance on acute faecal incontinence is a priority. Continence UK Journal 2007 (Suppl): 4.
- 3.Padmanabhan A, Stern M, Wishin J, et al. Clinical evaluation of a flexible fecal incontinence management system. Amer J Crit Care 2007; 16: 384–393. [PubMed] [Google Scholar]
- 4.Barbut F, Petit JC. Epidemiology of clostridium difficile-associated infections. Clin Microbiol Infect 2001; 7: 405–410. [DOI] [PubMed] [Google Scholar]
- 5.Beitz JM. Fecal incontinence in acutely and critically ill patients: options in management. Ostomy Wound Manage 2006; 52: 56–58, 60, 62–66. [PubMed] [Google Scholar]
- 6.Echols J, Friedman BC, Mullins RF, et al. Clinical utility and economic impact of introducing a bowel management system. J Wound Ostomy Continence Nurs 2007; 34: 664–670. [DOI] [PubMed] [Google Scholar]
- 7.Binks R. Faecal management systems in acute and critical care. Continence, UK 2007; 1: 5–8. [Google Scholar]
- 8.Bliss DZ, Johnson S, Savik K, et al. Fecal incontinence in hospitalized patients who are acutely ill. Nurs Res 2000; 49: 101–108. [DOI] [PubMed] [Google Scholar]
- 9.Ousey K. An evaluation of the management of faecal incontinence in two intensive care units. Presented at the 7th Asia Pacific Nurses Convention (ASPAN), 30 June–2 July 2010, Singapore.
- 10.Izaguirre Guerricagoitia L, Truchuelo Aragón A. Prevalence of diarrhea in critical patients units in Spain: a multicenter study. Enferm Intensiva 2011; 22: 65–73. [DOI] [PubMed] [Google Scholar]
- 11.All Wales Guidelines for Faecal Management Systems. London, MA: Healthcare Ltd. Available at: http://www.welshwoundnetwork.org/files/6313/8555/6979/all_wales-faecal_systems.pdf (2010, accessed 25 September 2013).
- 12.Cutting KF and White RJ. Maceration of the skin and wound bed 1: its nature and causes. J Wound Care 2002; 11: 275–278. [DOI] [PubMed]
- 13.Beeckman D, Woodward S, Gray M. Incontinence-associated dermatitis: step-by-step prevention and treatment. Br J Community Nurs 2011; 26: 382–389. [DOI] [PubMed] [Google Scholar]
- 14.European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Prevention and treatment of pressure ulcers: quick reference guide. Available at: http://www.npuap.org/wp-content/uploads/2012/02/Final_Quick_Prevention_for_web_2010.pdf (2010, accessed 18 June 2015).
- 15.Drees M, Snydman DR, Schmid CH, et al. Antibiotic exposure and room contamination among patients colonized with vancomycin-resistant enterococci. Infect Control Hosp Epidemiol 2008; 29: 709–715. [DOI] [PubMed] [Google Scholar]
- 16.Sethi AK, Al-Nassir WN, Nerandzic MM, et al. Skin and environmental contamination with vancomycin-resistant Enterococci in patients receiving oral metronidazole or oral vancomycin treatment for Clostridium difficile-associated disease. Infect Control Hosp Epidemiol 2009; 30: 13–17. [DOI] [PubMed] [Google Scholar]
- 17.Bhalla A, Pultz NJ, Gries DM, et al. Acquisition of nosocomial pathogens on hands after contact with environmental surfaces near hospitalized patients. Infect Control Hosp Epidemiol 2004; 25: 164–167. [DOI] [PubMed] [Google Scholar]
- 18.McFarland LV, Mulligan ME, Kwok RY, et al. Nosocomial acquisition of Clostridium difficile infection. N Eng J Med 1989; 320: 204–210. [DOI] [PubMed] [Google Scholar]
- 19.Montejo JC. Enteral nutrition-related gastrointestinal complications in critically ill patients: a multicenter study. The Nutritional and Metabolic Working Group of the Spanish Society of Intensive Care Medicine and Coronary Units. Crit Care Med 1999; 27: 1447–1453. [DOI] [PubMed] [Google Scholar]
- 20.Junkin J, Selekof JL. Prevalence of incontinence and associated skin injury in the acute care inpatient. J Wound Ostomy Continence Nurs 2007; 34: 260–269. [DOI] [PubMed] [Google Scholar]
- 21.Driver DS. Perineal dermatitis in critical care patients. Crit Care Nurse 2007; 27: 42–46. quiz 47. [PubMed] [Google Scholar]
- 22.Bayón García C, Binks R, De Luca E, et al. Prevalence, management and clinical challenges associated with acute faecal incontinence in the ICU and critical care settings: The FIRST™ cross-sectional descriptive survey. Intensive Crit Care Nurs 2012; 28: 242–250. [DOI] [PubMed] [Google Scholar]
- 23.Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol 1997; 32: 920–924. [DOI] [PubMed] [Google Scholar]
- 24.Bayón García C, Binks R, De Luca E, et al. Expert recommendations for managing acute faecal incontinence with diarrhoea in the intensive care unit. J Intensive Care Society 2013; 14(Suppl 2): 1–9. [Google Scholar]
- 25.Keller BP, Wille J, van Ramshorst B, et al. Pressure ulcers in intensive care patients: a review of risks and prevention. Intensive Care Med 2002; 28: 1379–1388. [DOI] [PubMed] [Google Scholar]