Abstract
Perfusion techniques have been used for centuries to visualize the circulation of tissues. Axolotl (Ambystoma mexicanum) is a species of salamander that has emerged as an essential model for regeneration studies. Little is known about how revascularization occurs in the context of regeneration in these animals. Here we report a simple method for visualization of the vasculature in axolotl via perfusion of 1,1’-Dioctadecy-3,3,3’,3’-tetramethylindocarbocyanine perchlorate (DiI). DiI is a lipophilic carbocyanine dye that inserts into the plasma membrane of endothelial cells instantaneously. Perfusion is done using a peristaltic pump such that DiI enters the circulation through the aorta. During perfusion, dye flows through the axolotl’s blood vessels and incorporates into the lipid bilayer of vascular endothelial cells upon contact. The perfusion procedure takes approximately one hour for an eight-inch axolotl. Immediately after perfusion with DiI, the axolotl can be visualized with a confocal fluorescent microscope. The DiI emits light in the red-orange range when excited with a green fluorescent filter. This DiI perfusion procedure can be used to visualize the vascular structure of axolotls or to demonstrate patterns of revascularization in regenerating tissues.
Keywords: Physiology, Issue 124, DiI, 1, 1'-Dioctadecy-3, 3, 3', 3'-tetramethylindocarbocyanine perchlorate, visualization, perfusion, vasculature, labelling, Ambystoma mexicanum, axolotl
Introduction
Visualization of vasculature plays a vital role in understanding the structure and function of organisms across many species. Starting in the 16th century with Leonardo da Vinci, models and graphic representations of the circulation have been studied1. Using waxes and rubber molds, tissues were perfused to create three-dimensional models of the vasculature, which allowed for the study of organogenesis and pathogenesis1,2. Resins and waxes were colored with dyes such as India Ink or carmine red to allow for their easy visualization1,2. However, these techniques caused many issues because their high viscosities prevented full perfusion of the tissue of interest1. As the field became more sophisticated, the use of confocal and electron microscopes came into play, moving the perfusion techniques away from cast-molds and toward liquid perfusions of the vasculature, some of which allowed for the perfusion and imaging of blood vessels without destroying the initial tissue3. DiI, a fluorescent carbocyanine dye, is one such stain that allows for the perfusion of animals without damage to the vascular tissue.
Carbocyanine dyes are lipophilic dyes that incorporate into cell membranes upon contact. These dyes allow for easy and instantaneous staining of vascular endothelial cells, which can then be viewed under a fluorescent confocal microscope. DiI moves via lateral diffusion in the lipid membrane of cells, as shown in the labeling and tracing of neurons4. Chemically, the two alkyl chains of DiI give the dye its high affinity for cell membranes, while two conjugated rings from a fluorochrome which is responsible for emitting a red wavelength when excited by green fluorescent light filters4. DiI has been utilized in many capacities, including successful labeling of the plasma membrane and both anterograde and retrograde labeling in neurons5,6. DiI has previously been used in perfusion protocols while visualizing the vasculature of mice7.
Axolotls (Ambystoma mexicanum) are salamanders that live exclusively in brackish lakes near Mexico City, Mexico. These animals have become an important model for understanding regenerative processes as they can regenerate full limbs, tail (including nerve cord), portions of the heart and other internal organs, and portions of the eye as adults8,9. Additionally, with the recent application of genetic tools in axolotls, unprecedented insight into the molecules and cells driving these processes is now possible8. The successful regeneration of an entire limb requires an extensive revascularization process, which may play a significant role in regeneration beyond simply the traditional functions of blood vessels in providing oxygen and nutrients. Understanding revascularization in the context of tissue regeneration is imperative. Axolotl blood vessels have previously been visualized using India Ink, and while the results were intriguing, this process has not been revisited in subsequent decades10. We sought to adapt a DiI perfusion protocol developed for use in mammals to allow for a complete perfusion and visualization of the axolotl vasculature7. This protocol describes the steps taken to successfully perfuse and subsequently visualize the axolotl circulation with a DiI staining technique. This procedure will allow for precise visualization of patent blood vessels in homeostatic tissues, as well as in regenerating tissues, and provides a novel method for visualization and analysis of the revascularization process in the axolotl.
Protocol
All axolotl experimentation was performed in accordance with Brigham and Women's Hospital's (BWH) Institutional Animal Care and Use Committee.
1. Set up Perfusion Experiment
Place an adult axolotl in a plastic container filled with 0.1% tricaine solution (MS222) for 15-20 min or until completely anesthetized. Ensure that the container is filled with enough tricaine solution such that the axolotl is completely submerged. Note: All procedures must be performed in accordance with institutional animal care guidelines. At BWH, an axolotl is considered fully anesthetized when it fails a foot-pinch test, meaning there is no reflexive movement when the foot is gently squeezed. Caution: Though tricaine is an anesthetic specifically used for aquatic organisms, direct skin contact with the tricaine solution should be avoided.
- Set up the axolotl perfusion station.
- Place the absorbent pad on a flat, level surface with the absorbent side facing up.
- Cut a hole in the polystyrene foam frame that is the appropriate size and shape for the anesthetized axolotl to lay in the supine position. Place the frame on the absorbent pad. Note: Some extra paper towels can be placed immediately under the frame for additional absorbency.
- Load the peristaltic pump with the perfusion tubing. Set the pump to a flow rate of 0.7 ml/min, flowing in the clockwise direction.
- Make the dilutent solution with 0.7x PBS and 5% glucose in a 1:4 mixture.
- Mix 10 mL of diluent solution with 200 µL of the DiI stock solution in a 50 mL conical tube. Cap and mix by inversion. Cover this tube with aluminum foil paper to protect the working solution from exposure to light. Note: Volumes should be changed according to the size of the axolotl proportionally. These values are for an approximately 15 cm axolotl (snout-to-tail length). Animals of this size may not have reached full sexual maturity so animal sex may not be determined at this time.
- Fill a 50 mL conical tube with 0.7x PBS. Note: PBS will be used for priming the loop and axolotl exsanguination.
- Attach the 27-gauge butterfly needle to the exit end of the perfusion tubing. Fold the butterfly wings onto each other and place in the clamp stand.
- Place the free end of the perfusion tubing in the 50 mL conical tube filled with 0.7x PBS and run the perfusion pump until the entire tubing is filled with solution. Pause the pump once the entire tubing is filled with PBS. Note: Be sure the tubing is free of air bubbles at all times, as these will cause air emboli in the axolotl and prevent full perfusion.
- Place a paper towel in the axolotl-shaped mold in the polystyrene foam frame. Using a transfer pipette, soak the towel with tricaine solution. Note: Cut a small square in the middle of the paper towel to allow for the drainage of fluids during the perfusion procedure.
- Place the anesthetized axolotl supine on the paper towel inside the polystyrene foam frame.
2. Opening the Axolotl Chest
Use surgical forceps to pinch the skin along the central axis of the axolotl's chest, just below the line of the shoulders. Pull up.
Use a scalpel to make a small incision where the skin has been pulled.
- Remove a square patch of skin over the chest in order to reveal two cartilage plates.
- Remove the skin to open a window over the thoracic cavity large enough to clearly see the heart and approximately 5 mm of the aorta branching off of the heart.
Carefully tear the connective tissue using forceps or the closed scissors in order to avoid cutting any major blood vessels.
Lift each cartilage plate individually using the forceps and excise them with the surgical scissors.
Carefully pinch the pericardium with the forceps, pull up, and puncture it using the surgical scissors; this incision should be just deep enough to puncture the very thin pericardium and should be large enough to allow for removal of the pericardium. Take care not to cut the heart.
Delicately remove the pericardium to expose the heart and the aorta. Note: Using a transfer pipette, periodically flush the chest cavity and the gills with tricaine solution to keep the area clear and keep the axolotl anesthetized.
3. Perfusion of the Axolotl
Place the clamp stand with the loaded butterfly needle next to the polystyrene foam frame, such that the arm of the clamp can easily be manipulated to insert the needle into the axolotl aorta. Point the needle's tip toward the rostral aspect of the animal during insertion and keep the needle parallel to the aorta to avoid puncturing it through the opposite side.
Turn on the peristaltic pump. 0.7x PBS should continue to flow through tubing.
- Insert the needle into the aorta.
- Slide the forceps under the aortic arch and pull up slightly to allow for easy access.
- Maneuver the needle-clamp combination such that the needle runs along the length of the aorta, pointing up towards the head. Insert the needle while using the forceps for support behind the aorta. Note: The needle should be inserted deep enough into the aorta to ensure that it will not slip out during perfusion. This may be about 5 mm for a 15 cm axolotl. Ensure that the needle is perfectly in line with the aorta to avoid complete puncture of the vessel. Through-and-through punctures can cause massive hemorrhage and decrease success rates of perfusion. Successful insertion can be confirmed by visible enlargement of the atria of the heart.
- Quickly lacerate one atrium with the scissors and allow blood to drain.
- Flush with tricaine solution to prevent blood accumulation and clot formation in the chest cavity.
Perfuse the axolotl with about 20-30 mL of PBS.The animal should change from light pink in color to white in a successful perfusion.
Pause the peristaltic pump and move the free end of the tubing into the 15 mL tube of DiI solution. Restart the pump, take care to avoid creating any air bubbles in the tubing.
Perfuse the axolotl with the entire working stock of DiI. Note: In a successful perfusion, the axolotl with change color to the bright pink of the DiI. This will be most noticeable in the gills.
Pause the pump after the perfusion with DiI is complete and place the free end of the tubing into 4% Paraformaldehyde (PFA) solution to fix the tissue. Restart the pump and perfuse at least 10 mL of PFA. Caution: PFA is toxic and should be handled and disposed of appropriately. Gloves and safety glasses should be worn, and solutions should be made inside a fume hood. Perfusion of the axolotl with PFA to fix the tissue results in the death of the animal.
4. Ending the Perfusion and Visualization Preparation
Stop the peristaltic pump and remove the needle from the axolotl aorta.
Place the axolotl on a plastic plate. Note: Using one half of a large Petri dish works well and allows for pouring a small amount of Tricaine or PBS onto the axolotl to keep its skin wet and improve visualization quality.
Dispose of all used materials in the appropriate waste bins. Clean surgical tools with 70% ethanol, disinfect using a glass-bead sterilizer between animals, and sterilize by autoclaving following the procedure. Flush tubing with the PBS solution and then drain, dry fully, and store for further use.
5. Visualization the Perfused Axolotl
Place the axolotl under a fluorescent confocal microscope.
Turn the lights off as visualization of the DiI-stained vessels is impeded by light.
Use a green fluorescence emission filter cube (e.g. ET-CY3) with the confocal microscope to visualize the vasculature of the axolotl. Use excitation light of wavelength 545 nm. Note: To obtain an image of high quality, the following parameters can be used: exposure for 1.1 s, gain of 1x, saturation of 1.0, magnification of 2X.
Representative Results
With DiI staining, the vasculature of the axolotl can be easily visualized. Blood vessels of animals perfused with the lipophilic dye are immediately visible under a fluorescent confocal microscope. Figure 1.1-1.5 is a schematic representation of the perfusion protocol. After perfusion with the bright pink dye, a successfully perfused axolotl will appear pink. Using a green fluorescent filter on a confocal microscope a red emission of the vascular network will appear. The DiI staining occurs in all body tissues when perfusion is successful, including the tail, limbs, gills, and eyes (Figure 2A, Figure 2B, Figure 2C, Figure 2D, repectively). Unsuccessful perfusions result in a lack of red-stained vasculature or in patchy staining of the vessels.
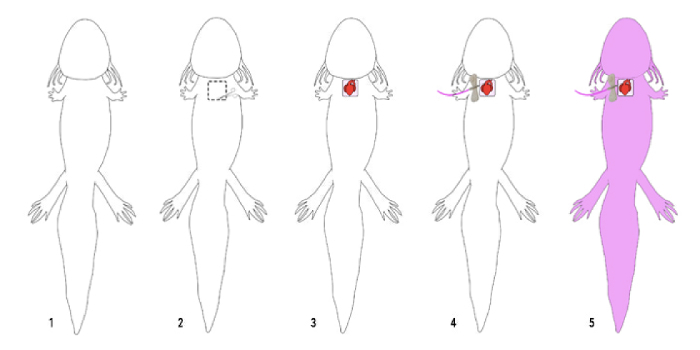
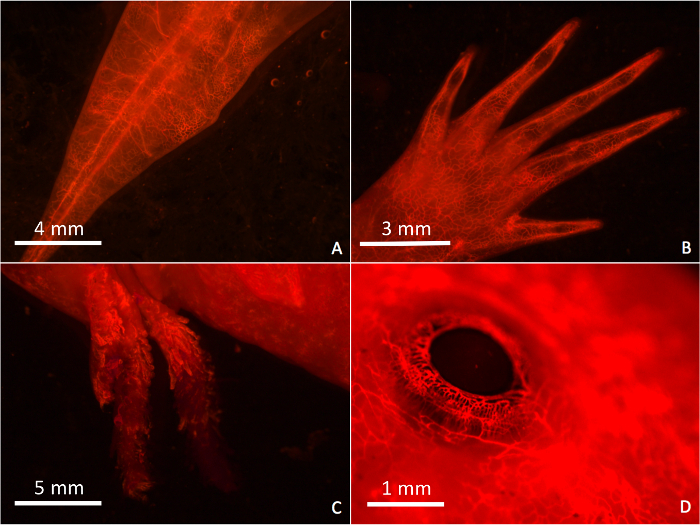
Figure 1: Schematic of the perfusion protocol. Axolotls successfully perfused with the lipophilic dye, DiI, demonstrate full staining of the vasculature upon imaging. 1: Full supine axolotl before perfusion experiment. 2: Opening the chest of the axolotl. 2: Axolotl with an open chest cavity. 3: Insertion of the 27 G butterfly needle into the aorta of the axolotl. 4: Tubing should first contain 0.7x PBS, then the DiI working solution, and lastly 4% PFA. 5: Fully perfused axolotls appear pink. Please click here to view a larger version of this figure.
Figure 2: Images of a Fully Perfused Axolotl. Images of the axolotl vasculature were taken using a fluorescent confocal microscope after successful perfusion with the DiI stain. 2A: Tail. 2B: Foot. 2C: Gills. 2D: Eye. Imaging is done using a confocal microscope with an green fluorescent emission filter cube. Magnification for images A, B, C, and D, are 1.74X, 2.16X, 1.18X, and 5.69X, respectively. Please click here to view a larger version of this figure.
Discussion
Visualization of the vasculature of the axolotl can be successfully accomplished via perfusion with the lipophilic carbocyanine dye, DiI. In this study, we describe a novel protocol for the perfusion of the axolotl with DiI using a peristaltic pump. We also show the subsequent visualization of the axolotl vasculature using a fluorescent confocal microscope. This protocol was an adaptation of the rodent DiI perfusion protocol seen in Li et al.7, however major differences between the rodent and the axolotl required a revision of the protocol to fit the axolotl model.
This study discusses a method of DiI perfusion of the axolotl in order to successfully visualize the vasculature. Differences in the anatomy and physiology between the salamander and rodent demand alterations in major aspects of the perfusion, including location of needle insertion, method of perfusion, and the reagents used. In order to achieve a successful perfusion, we limited the damage done to the vasculature of the axolotl. While opening the chest cavity, care was taken to fully expose the heart and aorta while avoiding any damage or lacerations to major blood vessels. Limiting the use of the surgical scissors prevented accidental clipping of major vessels while small incisions maintained control over the exposure of the heart and aorta. Success rates of perfusions also increased when the DiI needle was inserted through the aorta, rather than directly into the chambers of the heart. The axolotl, unlike the mouse, has a three chambered heart, containing only one ventricle with significantly less musculature than that of the mouse. Because of these differences, the location of needle insertion had to be moved to the more stable aorta. The aorta was determined to be the optimal location for insertion of the perfusion needle as it is large enough for puncture by a 27 G needle and has limited movement. Movement was minimized in order to avoid accidental removal or slipping of the perfusion needle or through-and-through puncture of the aorta. Cardiac perfusions using the ventricle as an insertion point proved to have a much lower rate of success than those with an aortic insertion point. Erroneous puncture of the vasculature often resulted in the formation of emboli or prevented perfusion, resulting in very low rates of successful vascular labeling. By using a clamp stand to hold the butterfly needle during perfusion, we have decreased its movement, therefore increasing the rate of successful perfusions. Additionally, due to the delicacy of the axolotl tissue, when compared to the mouse, a peristaltic perfusion pump was necessary, as opposed to the manual perfusion previously used. The use of this pump allowed for a hands-free approach to the axolotl perfusion to minimize mistaken puncture of the thin tissues. Perfusions were unsuccessful for many additional reasons, including through-and-through puncture, clotting, and embolism. In the case that the needle was inserted into the aorta and a second puncture was created through the posterior wall, the DiI solution would flow directly into the chest cavity rather than pass through the systemic circulation. Additionally, once blood exited the vasculature, it quickly formed a blood clot which could impede perfusion. Clots and air bubbles could also form in the vasculature, causing emboli which preclude successful perfusion. Lastly, this protocol incorporated reagents adjusted to fit the axolotl osmolality, which differs significantly from that of the mammal. Adaptation of this protocol and the significant changes made to fit the axolotl model will assist in the pursuit of understanding the process of revascularization of tissues during regeneration.
DiI, which is pink in color, will perfuse the animal and give it a bright pink hue. Successfully perfused axolotls became bright pink to the naked eye, with highly vascularized regions appearing more intensely stained. Perfused animals viewed with a fluorescent confocal microscope using a green filter can be visualized in the red-orange emission spectrum. Vasculature was best visualized in thinner tissues that minimized accidental DiI staining of non-vascular tissues. Perfusion of the tissue with 4% Paraformaldehyde (PFA) immediately after DiI perfusion should be done to fix the tissue.
DiI perfusions are end-point experiments for the axolotl. During the procedure, all of the animal’s blood is effectively drained and replaced with 0.7x PBS, followed immediately by DiI solution, and finally 4% PFA. This disrupts the axolotl’s ability to engage in the vital act of gas exchange and it loses the ability to oxygenate its body tissues. Due to this end-point nature, each perfusion captures only a single time-point of vascular growth, and the animal cannot be further perfused at a later time. Due to this time-limiting factor, multiple animals must be used in order to describe a time course of vascular development.
This DiI protocol, and the modifications applied to improve it, can be used to successfully label and visualize the vasculature of the axolotl. Since the axolotl is an essential model organism for the study of regeneration, successful perfusions open up opportunities to interrogate the process of angiogenesis during regeneration. The axolotl is a model organism for the study of regeneration because it is a neotenous animal and therefore retains a remarkable ability to regenerate throughout adulthood8. The revascularization process of regenerating tissues, however, is not well understood, therefore the adaptation of the DiI perfusion to the axolotl system presents opportunities to understand regeneration that were not available with the mammalian model. The perfusion of the axolotl using DiI is a novel technique for the study of revascularization of regenerating tissue in this animal model, therefore, this protocol can be further used to understand organogenesis during development and angiogenesis during disease as well as be utilized as an important tool during the study of regeneration.
Disclosures
The authors have nothing to disclose.
Acknowledgments
This research was supported by the Brigham & Women’s Hospital and the March of Dimes. The authors would like to thank all of the members of the Whited Lab for their support and advice.
References
- Giuvarasteanu I. Scanning electron microscopy of vascular corrosion casts - standard method for studying microvessels. Rom J Morphol Embryo. 2007;48(3):257–261. [PubMed] [Google Scholar]
- Hasan MR, Herz J, Hermann DM, Doeppner TR. Intravascular perfusion of carbon black ink allows reliable visualization of cerebral vessels. J Vis Exp. 2013. p. e4374. [DOI] [PMC free article] [PubMed]
- Minnich B, Lametschwandtner A. Scanning electron microscopy and vascular corrosion casting for the characterization of microvascular networks in human and animal tissues. Microscopy: Science, Technology, Applications, and Education. 2010;1:29–39. [Google Scholar]
- Honig M, Hume RI. DiI and DiO: versatile fluorescent dyes for neuronal labelling and pathway tracing. Trends Neurosci. 1989;13:333–335. [PubMed] [Google Scholar]
- Honig MG, Hume RI. Fluorescent carbocyanine dyes allow living neurons of identified origin to be studied in long-term cultures. J Cell Biol. 1986;103(1):171–187. doi: 10.1083/jcb.103.1.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz M, Agranoff BW. Outgrowth and maintenance of neurites from cultured goldfish retinal ganglion cells. Brain Res. 1981;206(2):331–343. doi: 10.1016/0006-8993(81)90535-7. [DOI] [PubMed] [Google Scholar]
- Li Y, Song Y, Zhao L, Gaidosh G, Laties AM, Wen R. Direct labeling and visualization of blood vessels with lipophilic carbocyanine dye DiI. Nat Protoc. 2008;3(11):1703–1708. doi: 10.1038/nprot.2008.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo TH, Kowalko JE, DiTommaso T, Nyambi M, Montoro DT, Essner JJ, Whited JL. Evidence of TALEN-mediated gene editing of an endogenous locus in axolotl. Regeneration. 2015;2(1):37–43. doi: 10.1002/reg2.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockes JP, Kumar A. Appendage Regeneration in Adult Vertebrates and Implications for Regenerative Medicine. Science. 2005;310(5756):1919–1923. doi: 10.1126/science.1115200. [DOI] [PubMed] [Google Scholar]
- Smith AR, Wolpert L. Nerves and angiogenesis in amphibian limb regeneration. Nature. 1975;257(5523):224–225. doi: 10.1038/257224a0. [DOI] [PubMed] [Google Scholar]


