Abstract
Background
In malaria elimination settings, the very low levels of transmission now being attained present challenges that demand new strategies to identify and treat low-density infections in both symptomatic and asymptomatic populations. Accordingly, passive case detection activities need to be supplemented by active case detection (ACD) strategies with more sensitive diagnostic tools. Malaria rapid diagnostic tests (RDTs) have provided low- and middle-income countries with unprecedented access to malaria diagnostics. Nevertheless, conventional RDTs miss a potentially important proportion of sub-microscopic infections. Therefore, new combination highly sensitive (HS-)RDTs, able to detect low parasite densities and identify all infected individuals, could support countries implementing ACD strategies for radical cure to accelerate malaria elimination. To address this need, an on-line survey was conducted to gather information from malaria control programme representatives to guide the development of next-generation RDTs.
Results
Most of respondents confirmed that ACD was a common activity in their programmes (56/75; 75%). Although microscopy was the preferred method in case management and reactive case detection, RDTs were the primary diagnostic tests used in proactive case detection (31/75; 41%). In terms of preferences for species detection in a new combination HS-RDT, data was not one-directional. Survey respondents slightly preferred the Pf/Pv/Pan combination (42%; 21/50), while Pf/Pan was more popular among end-users. Survey respondents also valued a low-cost (< $1.00 USD), lightweight and portable test, able to detect asymptomatic infections and differentiate species, as well as provide immediate results that could be interpreted with the naked eye. In addition, respondents were open to new tests and even to replace the existing ones for ACD (63%; 47/75).
Conclusions
This survey provided valuable information on the use and current limitations of ACD, on the primary product characteristics for a next-generation combination HS-RDT to support ACD and radical cure, and on the potential adoption of such a test, if available, to support malaria elimination.
Keywords: Malaria, Diagnostics, Rapid test, Elimination, Survey, Active case detection
Background
Malaria continues to be a significant public health concern, responsible for approximately 212 million new cases and 429,000 deaths in 2015 [1]. Nevertheless, scale-up of malaria control efforts has contributed to tremendous reductions in disease burden and, as a result, elimination is currently considered a target in numerous countries. Between 2010 and 2015, global new malaria cases and mortality rates fell by 21 and 29%, respectively [1] and during the same period, the World Health Organization (WHO) declared 17 countries and territories no longer endemic or having zero indigenous malaria cases [2]. In 2015, the World Health Assembly endorsed the ambitious goal of achieving worldwide malaria elimination in 10 countries by 2020 and 35 by 2030 [3]. There is currently a strong global commitment to malaria elimination, and malaria eradication has been embraced by the Roll Back Malaria Partnership and by WHO as a long-term reality [4].
With enough resources, elimination can be achieved with current tools in countries with reasonably good economies, infrastructure, and health systems [5]. Nevertheless, the very low levels of transmission now being attained in many countries present new challenges that demand new strategies, such as active case detection (ACD), to identify and treat infections among populations who do not seek treatment [6]. ACD is defined by WHO as the ‘detection by health workers of malaria cases at community and household levels, sometimes in population groups that are considered at high risk. ACD can consist of screening for fever followed by parasitological examination of all febrile patients or as parasitological examination of the target population without prior screening for fever’ [7].
Accordingly, different ACD strategies have been developed to find subclinical infections in populations at risk and foci of transmission that may become important as elimination is approached [8, 9]. These tactics are generally split into two broad types: reactive case detection (RACD) and proactive case detection (PACD). RACD is an active surveillance method that is triggered by passively detected cases. It involves screening households or individuals within a specified area, typically a pre-determined radius around a locally acquired case, with the goal of preventing further malaria transmission by identifying additional symptomatic or asymptomatic infections [10, 11]. PACD consists of targeted or mass screening to search for cases in the community, which may include screening to find cases that are symptomatic or asymptomatic without the trigger of passively detected cases [12, 13].
Inherent to the change in focus that occurs as countries progress from control to elimination is the need to interrupt transmission which requires identification and treatment of parasite carriers with sub-microscopic infections that are still capable of contributing to transmission [6, 14, 15]. In this sense, one of the main challenges identified by the malERA Consultative Group on Diagnoses and Diagnostics was to develop more sensitive tests capable of identifying very low parasite densities in asymptomatic individuals in field settings for mass screening and treatment [15].
The WHO currently recommends malaria diagnosis either by microscopy or rapid diagnostic test (RDTs) in patients with suspected malaria prior to treatment [1]. Conventional RDTs are adequate for diagnosing people with symptomatic malaria. They have facilitated access to malaria diagnosis outside health facilities in peripheral communities beyond the reach of microscopy and have the potential to become the test format of choice for ACD due to their portability and ease of use [12, 16]. Nonetheless, current RDTs cannot detect the low-level blood-stage malaria infections that can be detected by more sensitive but complex methods such as nucleic acid amplification techniques (NAATs) [17].
Whether more sensitive tests will make a significant impact on malaria elimination is still unclear [18], a new combination highly sensitive (HS-)RDTs, able to detect low-density parasitaemia due to any Plasmodium species and to identify all infected individuals, could support countries implementing strategies to accelerate the elimination of all forms of malaria. More specifically, in ACD, a combination HS-RDT could detect people infected with parasite densities below the limit of detection of current RDTs, prior to the development of symptoms and before transmission to others in their community. Compared with ACD using existing diagnostics, a combination HS-RDT could be more cost-effective and practical because programmes could detect a greater proportion of infections, spending less time in the field tracking every case and launching easier-to-implement screening and treatment campaigns (i.e. minimal infrastructure requirements, less labour-intensive).
To define how to improve next-generation RDTs, it is necessary to understand where current diagnostics are falling short, in particular in supporting ACD activities and to identify the primary customer preferences. Therefore, an on-line survey of malaria control programme representatives from endemic countries was conducted to gather information to support the development of a new combination HS-RDT for elimination, in an informed and validated way.
Methods
Survey design, data sources and analysis methods are described below.
Data collection
Survey with national malaria control programmes
An on-line, anonymous survey consisting of 20 questions was conducted between August and September 2016. The survey was accompanied by a description of survey objectives and definitions of ACD strategies. It inquired about current practices for malaria control and elimination (i.e. PCD, ACD), use of currently available diagnostic tests, and preferred characteristic for improved RDTs. The survey was available in English, French and Spanish. Invitations to participate were sent by email to 334 individuals from 104 malaria endemic countries included in FIND’s database. The target audience was national malaria programmes, as well as local institutions working closely with them on implementation of control and elimination strategies.
End-user survey
An email (in English and Spanish, as needed), including a brief questionnaire with 6 questions, was sent to 28 national or regional diagnostics supervisors from 14 different malaria endemic countries in November 2016. Questions focused on test type preferences and on the challenges in current malaria RDT use, in particular common operator errors.
WHO data
Published data collected by the WHO/Global Malaria Programme for the 2015 World Malaria Report was also used for the analysis. This dataset included information regarding prevalence of Plasmodium vivax and the current phase of WHO’s malaria programme in WHO countries (control, pre-elimination, elimination, prevention of re-introduction, and malaria free), and ACD policies.
Analysis
Data analysis involved translation to English, formatting, and merging survey responses with a data set of basic statistics on the countries (WHO region, WHO programme phase, and P. vivax prevalence). Absolute and relative number of responses was determined for each question, providing counts and percentages, respectively. Aggregated tables, as well as pie and column charts were generated to represent results. Major differences when comparing survey data with information available from WHO (e.g. region, programme phase, and P. vivax prevalence), are highlighted below in “Results” and “Discussion” sections.
Results
Survey demographics
Replies were received from 75 (22%) of the 334 contacted individuals, representing a total of 48 malaria endemic countries. Almost three quarters of the participants in the survey were based in the WHO AFRO and PAHO regions (31/75; 41% and 25/75; 33%, respectively; Fig. 1a). Most of the represented countries were those classified as being in the “Control” WHO programme phase (63/75; 84%; Fig. 1b). Questionnaire respondents were mostly from national malaria control programmes (NMCPs) and ministries of health (MoH) (50/75; 67%; Fig. 1c), as well as from countries where P. vivax is prevalent (38/73; 52%; Fig. 1d).
Fig. 1.
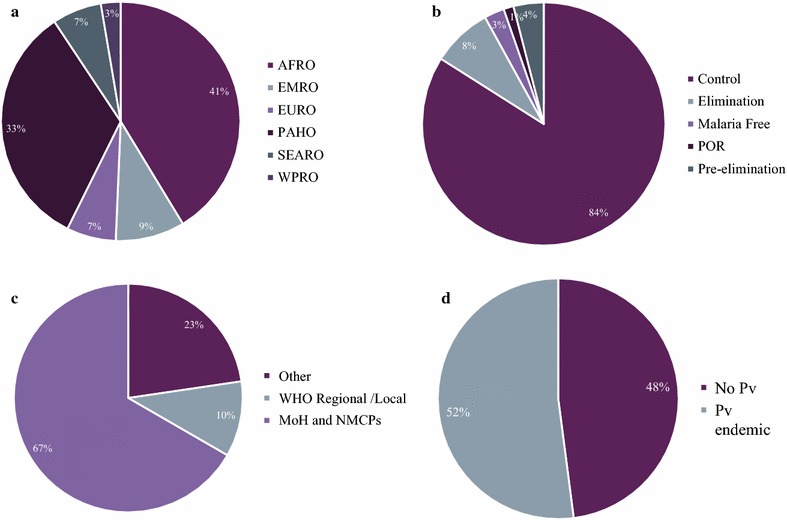
Survey demographics. a Respondents by region: African (AFRO), Americas (PAHO), Europe (EURO), the Eastern Mediterranean (EMRO), South-East Asia (SEARO) and the Western Pacific (WPRO). b Represented countries by WHO programme phase: control, pre-elimination, elimination, prevention of re-introduction (POR), and malaria free. c Respondents by affiliation: Ministries of Health (MoH) and National Malaria Control Programmes (NMCPs) from WHO regional offices and others (i.e. researchers, non-profit organizations). d Countries where Plasmodium vivax (Pv) is present or absent. All values are represented in percentages
Results were also compared with World Malaria Report (WMR) data [1] to understand how representative the survey data were. EMRO (9% in the questionnaire versus 14% in the WMR) and Asian regions (2–5% vs 10–10% in the WMR; SEARO-WPRO, respectively) were underrepresented while PAHO (33% vs 19% in the WMR) was overrepresented. Countries in prevention of reintroduction and pre-elimination phases were also underrepresented in the survey (1 and 4% vs 7 and 8% in the WMR, respectively), while ‘control’ countries were highly represented (84% vs 70% in the WMR). Countries with P. vivax prevalence were approximately equally represented in both data sets (42% vs 47% in the WMR).
Current diagnostics use
Inquiry about the use of malaria diagnostic tests showed that microscopy was the primary diagnostic test used (132/300; 44%), particularly in countries with P. vivax prevalence (80/152; 53%). Survey results indicated that microscopy was the preferred method in case management activities (49/75; 65% of all respondents), with RDTs (24/75; 32%) as a second test. Regarding the primary RDT type used, Pf/Pan test was the most commonly used test (28/75; 37%). Nonetheless, in countries with P. vivax prevalence, the Pf/Pv test was the most commonly used test (17/38; 45%), predominantly in the PAHO region.
Even though the majority of participating countries were classified by WHO as “control phase” countries (63/75; 84%), many respondents indicated that their programmes were performing ACD activities (56/75; 75%). Most of these countries (53/56; 95%) would perform more ACD activities if they had the capacity and resources. The biggest obstacle to performing more ACD was lack of sufficient budget (23/62; 37%), followed by lack of human resources (13/52; 25%) and transportation (12/59; 20%).
Microscopy (34/75; 45%) was slightly more common than RDTs (29/75; 39%) for RACD. In contrast, the primary diagnostic test used for PACD was RDT (31/75; 41%). Molecular testing was rare and only used in a few countries for surveys (13/75; 17%) and for ACD (2/75; 3%). Differences among regions were only observed in Africa, where RDTs were the primary diagnostic test used for RACD (18/31; 58%).
Interestingly, survey respondents were largely satisfied with their current diagnostic method for ACD (40/53; 75% were either “somewhat satisfied” or “extremely satisfied”). Nevertheless, satisfaction was slightly lower among those who rely on microscopy rather than RDTs for ACD: 30% were dissatisfied with microscopy (7/23) compared with 17% dissatisfied with RDTs (4/23). As shown in Fig. 2, the greatest limitation of malaria diagnostics for ACD was their lack of suitability for parasite detection in asymptomatic carriers, followed by their limited sensitivity for non-Plasmodium falciparum and P. falciparum species, as well as the requirement for extensive quality assurance and quality control mechanisms.
Fig. 2.
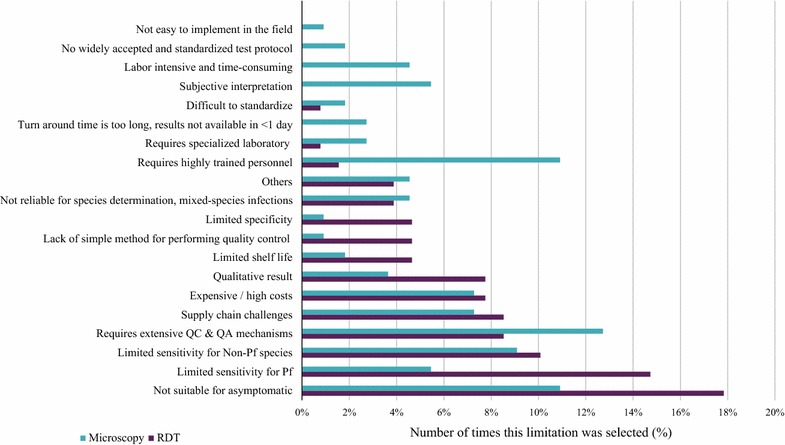
Limitations of the primary diagnostic used by program for ACD (all survey respondents). Respondents were asked to select up to three limitations. Responses were further analysed to split answers from “RDT” and “Microscopy” users. “Other” included: financial hardship, lack of confidence in suppliers, storing conditions, test acquisition, human resources, and lack of government political commitment. Columns represented the number of times that a given limitation was selected in percentage values. QC quality control, QA quality assurance
In this survey of end-users, the greatest challenge when using RDTs was to produce high quality tests that could withstand the conditions of transport, storage and use, particularly in high-temperature and high-humidity settings, with limited logistics and infrastructure. An improvement in some kit components (e.g. buffer volume, volume mark in pipettes, non-transparent cartridge color) and reagents, as well as extensive quality control and assurance mechanisms, were also seen as key factors for the success of RDT testing in the field.
Preferred product characteristics for a new combination HS-RDT
Survey participants were asked to provide suggestions regarding their desired product characteristics for a new combination HS-RDT for ACD activities to accelerate malaria elimination (Table 1). Survey respondents were asked to assume that a new combination HS-RDT would effectively detect all species, (i.e. even those with gene deletions), would have a limit of detection ten times better than current RDTs, and would be a WHO-prequalified lateral flow assay.
Table 1.
Key product characteristics according to survey respondents
| Attribute | Preferences | Comments |
|---|---|---|
| Species detection combination |
Pf/Pv/Pan
Pf/Pan |
The majority of participants preferred a Pf/Pv/Pan combination (42%; 21/50) while others were split between a Pf/Pan (36%; 18/50) and a Pf/Pv (18%; 9/50) test In the end-user survey, the Pf/Pan test was preferred (74%; 14/19) |
| Price | Less than $1.00 USD | Most survey respondents selected $0.50–0.99 USD as a reasonable price (52%; 35/67) |
| Shelf life and stability | 24 month shelf life at < 40 °C | The preferred shelf life was 2 years (79%; 59/75). Among different temperature options (30, 35, 40, 45 °C) preferred requirement was 40 °C (39%; 29/75) |
| Sample type | Peripheral whole blood from finger-prick is acceptable | Respondents were more open to saliva sampling (74% would use it for case management, ACD or both), but were less open to urine, breath or transdermal sample types (60, 54 and 49% respectively would use it for case management, ACD or both) |
| Packaging | Single use kits | Single kits, instead of bulk packaging with shared buffer, was most common (71%; 53/75) |
| Reader | Optional | Respondents indicated that the use of a reader was a moderate priority: 49% (37/75) of participants answered that it was “very likely” that their programme would implement an RDT with a reader for ACD. Among the capabilities provided by a reader, “enhanced sensitivity” was the most valued (65%; 49/75) |
The percentages represent the total number of times the particular benefit was selected compared to the total number of selections made for this question. The absolute number of responses is also provided
In terms of performance, when asked about the importance of other features for a diagnostic test for ACD, respondents underscored, as “definitely need”, the ability to detect most sub-microscopic infection (39/53; 74%), accurate speciation (37/53; 70%) and high specificity (34/53; 64%). A portable test with rapid turnaround time that can be used in the community, with minimal training needs and infrastructure level, were priorities from an operational point of view (Fig. 3).
Fig. 3.
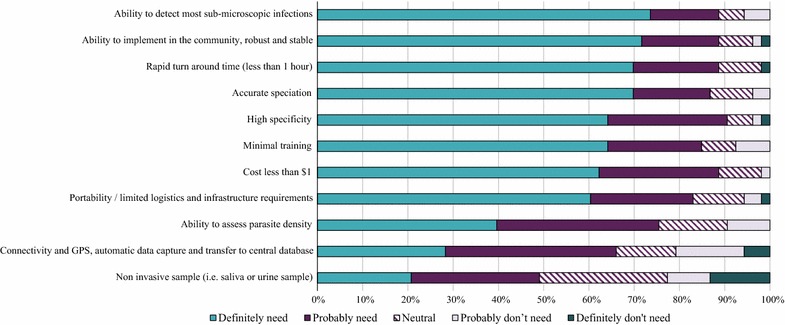
Importance of various features of a new combination HS-RDT for active case detection. Respondents were asked to select the importance of each feature in terms of “definitely need”, “probably need”, “neutral”, “probably don’t need”; and “definitely don’t need”. Columns represented the percentage of importance for each feature. GPS global positioning system
Potential adoption
Assuming that price was not an issue, respondents were favorable to implementing a combination HS-RDT ten times more sensitive than today’s best combination RDTs; 63% (47/75) and 29% (22/75) answered either “extremely likely” or “very likely” to implement it, respectively. From those, the following potential ACD activities where the new test would be used were indicated: PACD in high risk populations (19%; 56/293); reactive case detection (18%; 53/293); surveys with provision of treatment (15%; 45/293); PACD of high risk locations (15%; 43/293); screening pregnant women (14%; 41/293); and border screening (14%; 40/293).
Of respondents that would adopt the new highly sensitive RDT for ACD, almost half indicated they would “largely replace existing tests for ACD with combination HS-RDTs”, while the remainder pointed out they would “partially replace their existing tests with combination HS-RDTs” (46%; 32/69 in both cases). Beyond ACD and surveys, respondents would consider using the new test for travelers (27%; 50/188), while antenatal care (24%; 45/188) was also considered.
The survey also asked respondents what, in their opinion, would be the main benefits of a combination HS-RDT: ease of use/minimal user training (46/262; 18%), followed by an improved sensitivity for P. falciparum (44/262; 17%) and non-P. falciparum species (38/262; 15%) were the main benefits selected most often by participants.
Discussion
The results of the online survey allowed us to understand the perceived limitations of existing tests for ACD, preferred key product characteristics for a new combination HS-RDT and how this test might be adopted if available.
Considering that this survey was based on voluntary participation, the response rates were satisfactory, in particular the rates achieved for the main survey target were high: NMCP/MoH represented two-thirds of respondents and are central to the decision-making process for adoption of a new product. Overall, the survey was representative of malaria endemic countries, when comparing survey responses with WMR data [1]. Only a few discrepancies in terms of regions and programme phase representation were observed.
Despite the fact that the WHO guidance is not clear on when to perform ACD or the benefits of this strategy [8, 19, 20], ACD is a common activity [21, 22] and many countries have policies in place for various forms of ACD [1]. In agreement with this, only a quarter of survey respondents were not currently implementing ACD activities. For those performing ACD, microscopy was the preferred method. Nevertheless, RDT use was dominant in the African region as shown in other studies [23, 24]. For PACD, RDTs were more common than microscopy, and again, this was driven by African respondents. Well-known examples of large-scale ACD activities using primarily microscopy has been reported in countries from other WHO regions, such as Brazil [11].
Although the survey indicated that a high number of countries are performing ACD and using RDTs for such activity, it has been reported that testing volumes overall are small and that in fact microscopy dominates ACD [25]. Nevertheless, the data highlighted that respondents using RDTs for ACD were largely satisfied with their diagnostic method, compared with those using microscopy. In addition, and in agreement with other authors [6, 14, 15], the lack of sensitivity for parasite detection in asymptomatic carriers was pointed out as the greatest limitation of current diagnostics for ACD, particularly among RDT users. In this regard, some form of ACD using a more sensitive rapid diagnostic tool would be useful, whether it be for parasite clearance or information gathering.
In terms of the preferred product characteristics for a new combination HS-RDT, survey respondents vary in their preferences for the species detection combination. Overall, the data regarding the optimal combination of test lines for a new highly sensitive multi-species RDT was not one-directional. Although there was a slight preference for a more sensitive Pf/Pv/Pan test, particularly in elimination countries, this preference was less dominant for countries with P. vivax prevalence, where a Pf/Pv was prioritized (e.g. PAHO), and for the AFRO and EMRO regions, where a Pf/Pan test was preferred. Nonetheless, a RDT with three test lines was inconsistent with consumer’s desire for low prices and with end-user feedback about difficulties of interpreting a test with multiple lines. Furthermore, technical feasibility and cost of producing this type of test would be challenging.
Because test type (i.e. the combination of species detection test bands on the RDT) is a critical component of a new combination HS-RDT, additional data analysis was conducted to better understand the options. In the limited survey of end-users, a Pf/Pan test was preferred. The general feeling was that a combination HS-RDT with just two test lines—including one for Pan—was easy to interpret with the proper training and able to detect all species. Bell et al. [26] also referred to a Pf/Pan test as the optimum requirement in P. falciparum-predominant areas and the minimum one in mixed P. falciparum/non-P. falciparum areas for parasite screening activities. Furthermore, Pf/Pan and Pf/Pv combination tests already exist and are widely used for case management. Nonetheless, for ACD and elimination purposes, the first option, a Pf/Pan test, would not allow targeted radical cure of P. vivax and Plasmodium ovale infections while the second option, a Pf/Pv test, would miss infections due to other Plasmodium species. Specific detection of P. vivax and P. ovale infections is needed in order to provide targeted radical cure and eliminate all forms of malaria. Additional analysis for product interpretation, advantages and disadvantages of different possible combination tests is presented in Table 2.
Table 2.
Analysis of test type for a combination HS-RDT to support ACD
| Test type | Interpretation and treatment | Advantages | Disadvantages | Comments |
|---|---|---|---|---|
| Pan/Pf/Pv |
Pan(+)/Pf(+)/Pv(+) → ACT + PQ Pan(+)/Pf(+)/Pv(−) → ACT Pan(+)/Pf(−)/Pv(+) → CQ + PQ Pan(+)/Pf(−)/Pv(−) → CQ |
Detects all species Differentiates Pf Allows targeted Pv radical cure |
Does not target Po radical cure | Helpful for surveillance in drug resistant areas because differentiates Pf
Difficult interpretation by end user Technically challenging |
| Pan/Pf |
Pan(+)/Pf(+) → ACT Pan(+)/Pf(−) → CQ(+PQ) |
Detects all species Differentiates Pf |
Pan(+)/Pf(+) could be a mixed infection requiring PQ Pan(+)/Pf(−) would not receive PQ if Pv not confirmed |
Helpful for surveillance in drug resistant areas because differentiates Pf
29% of current RDT volume market Common, familiar format |
| Pf/Pv |
Pf(+)/Pv(+) → ACT + PQ Pf(+)/Pv(−) → ACT Pf(−)/Pv(+) → CQ + PQ |
Differentiates Pf
Allows targeted Pv radical cure |
Does not detect Pm/Po/Pk
Does not target Po radical cure |
Helpful for surveillance in drug resistant areas because differentiates Pf
6% of current RDT volume market |
| Pf/Pvom |
Pf(+)/Pvom(+) → ACT + PQ Pf(+)/Pvom(−) → ACT Pf(−)/Pvom(+) → CQ + PQ |
Differentiates Pf
Allows targeted Pv and Po radical cure |
Over-treatment of Pm with PQ Does not detect Pk |
Helpful for surveillance in drug resistant areas because differentiates Pf
Commercial product available |
| Pan/Pvo |
Pan(+)/Pvo(+) → ACT + PQ Pan(+)/Pvo(−) → ACT |
Detects all species Allows targeted Pv and Po radical cure |
Does not differentiate Pf | Shift from CQ to ACT for non-falciparum
Limited surveillance data on species; speciation at reference lab required |
| Pan only | Pan(+) → ACT + PQ | Detects all species | Over-treatment of Pf/Pm/Pk with PQ Does not differentiate Pf |
Shift from CQ to ACT for non-falciparum
Limited surveillance data on species; speciation at reference lab required |
This analysis assumes that all test lines have the same limit of detection required to detect sub-microscopic infections and that glucose-6-phosphate dehydrogenase (G6PD) deficiency testing is available and done when required
HS-RDT (high sensitive rapid diagnostic test); ACD (Active Case Detection); Pan (all Plasmodium species); Pf (Plasmodium falciparum); Pv (P. vivax); Pvom (P. vivax, P. ovale, and P. malariae); Pvo (P. vivax and P. ovale); Pk (P. knowlesi); ACT (artemisinin-combination therapy); PQ (primaquine); CQ (chloroquine); NAATs (nucleic acid amplification techniques)
Similar to other studies and work on the optimal diagnostic tests for malaria elimination [15, 25–27], survey respondents also valued a low-cost (< $1.00 USD), lightweight and portable test, with the ability to detect asymptomatic infections and different species, as well as provide immediate results that could be interpreted with the naked eye (although a reader would be acceptable). Long-term stability and resistance to high temperatures, as well as packaging of tests into individual kits, were also important to survey respondents.
Despite a relatively high satisfaction rate with existing tests for ACD, respondents were favorable to new tests and even to replacing existing ones. Particularly, MoH and NMCPs considered many potential ACD activities where they would use the new test, including for PACD in high-risk populations, RACD, and in surveys. In an ideal scenario, a highly sensitive field diagnostic test would increase effectiveness of targeted or mass screen-and-treat campaigns to near effectiveness of adequate mass drug administration campaigns while avoiding overtreatment of uninfected individuals [15, 28].
Recently, and after completing the survey, a new HRP2-specific highly sensitive RDT was launched into the market (Alere™ malaria Ag Pf ultrasensitive, catalog number #05FK140). While research data on the potential role and impact of this new P. falciparum-specific HS-RDT for ACD is still not available, this test is in line with most of the requirements highlighted by survey respondents and meets some of the key product characteristics, such as low-cost (USD$0.99), packaging (single use kit) and type of samples used (finger-prick blood, 5 µL).
Study limitations
It is important to note that this study had limitations and is thus not the sole consideration in developing product attributes for a new diagnostic test. The sample size for the survey was limited and probably those who were interested in RDTs were more likely to participate. Some regions and endemicity levels were over-represented and for some questions results were not unidirectional. In addition, perspectives from end-users were certainly biased toward case management, given the challenges of identifying and communicating remotely with end-users who regularly conduct ACD. Finally, the occurrence of mistakes in the survey analysis cannot be discounted, although large efforts were invested in accurate translation and detailed cross-checks to solve any detected discrepancies or omissions.
Conclusions
The ideal diagnostic test for malaria elimination does not exist yet, but this does not stop countries from implementing malaria elimination strategies, such as ACD with the tools available today. An effective screening tool to support accelerating elimination needs to provide a result rapidly enough to enable prompt treatment of positive cases and to distinguish between species to guide treatment decisions. In this regard, a next generation combination HS-RDT able to detect sub-microscopic malaria infections (symptomatic or asymptomatic), as well as to differentiate species requiring radical cure (P. vivax and P. ovale), could contribute to meeting the ambitious timelines for malaria elimination.
Authors’ contributions
JD and IG have designed the study. AC and JD have developed the survey questionnaires and conducted the data analysis. AC has drafted the manuscript. All authors have reviewed the manuscript. All authors read and approved the final manuscript.
Acknowledgements
We would like to thank all survey respondents for their efforts in providing replies and data, and resolving any subsequent questions.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets supporting the findings presented in this manuscript cannot be shared in a publicly available repository, since data was received under a strict confidentiality agreement. In order to preserve confidentiality, all results presented in the manuscript are in the form of aggregated figures.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Not applicable.
Funding
This survey has been conducted with funding from Bill & Melinda Gates Foundation Grant No. OPP1116774.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- ACD
active case detection
- AFRO
WHO Regional Office for Africa
- EURO
WHO Regional Office for Europe
- EMRO
WHO Regional Office for the Eastern Mediterranean
- FIND
Foundation for Innovative New Diagnostics
- HS
highly sensitive
- MoH
Ministry of Health
- NAATs
nucleic acid amplification techniques
- NMCP
National Malaria Control Programme
- PACD
proactive case detection
- PAHO
Pan American Health Organization
- Pan
all Plasmodium species
- Pf
Plasmodium falciparum
- POR
prevention of re-introduction
- Pv
Plasmodium vivax
- RDT
rapid diagnostic test
- RACD
reactive case detection
- SEARO
WHO Regional Office for South-East Asia
- USD
United States Dollar
- WHO
World Health Organization
- WMR
World Malaria Report
- WPRO
WHO Regional Office for the Western Pacific
Footnotes
Ana Campillo and Jen Daily contributed equally to this work
Contributor Information
Ana Campillo, Email: ana.campillo@finddx.org.
Jennifer Daily, Email: jenniferanndaily@gmail.com.
Iveth J. González, Email: Iveth.Gonzalez@finddx.org
References
- 1.WHO. World malaria report. Geneva: World Health Organization; 2016. http://apps.who.int/iris/bitstream/10665/252038/1/9789241511711-eng.pdf?ua=1.
- 2.WHO. A framework for malaria elimination. Geneva: World Health Organization; 2017. http://apps.who.int/iris/bitstream/10665/254761/1/9789241511988-eng.pdf?ua=1.
- 3.WHO. Global technical strategy for malaria 2016–2030. Geneva: World Health Organization; 2015. http://apps.who.int/iris/bitstream/10665/176712/1/9789241564991_eng.pdf?ua=1.
- 4.Roll Back Malaria Partnership. The global malaria action plan for a malaria free world. Geneva; 2008.
- 5.Plowe CV. The Jeremiah Metzger lecture: new additions to the toolbox for global malaria eradication. Trans Am Clin Climatol Assoc. 2014;125:271–280. [PMC free article] [PubMed] [Google Scholar]
- 6.Alonso PL, Brown G, Arevalo-Herrera M, Binka F, Chitnis C, Collins F, et al. A research agenda to underpin malaria eradication. PLoS Med. 2011;8:e1000406. doi: 10.1371/journal.pmed.1000406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.WHO. Malaria terminology. Geneva: World Health Organization; 2017. http://apps.who.int/iris/bitstream/10665/208815/1/WHO_HTM_GMP_2016.6_eng.pdf.
- 8.WHO RBMP. Disease surveillance for malaria elimination—an operational manual. Geneva: World Health Organization; 2012. http://apps.who.int/iris/bitstream/10665/44852/1/9789241503334_eng.pdf.
- 9.Tietje K, Hawkins K, Clerk C, Ebels K, McGray S, Crudder C, et al. The essential role of infection–detection technologies for malaria elimination and eradication. Trends Parasitol. 2014;30:259–266. doi: 10.1016/j.pt.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 10.Lindblade KA, Steinhardt L, Samuels A, Kachur SP, Slutsker L. The silent threat: asymptomatic parasitemia and malaria transmission. Expert Rev Anti Infect Ther. 2013;11:623–639. doi: 10.1586/eri.13.45. [DOI] [PubMed] [Google Scholar]
- 11.Macauley C. Aggressive active case detection: a malaria control strategy based on the Brazilian model. Soc Sci Med. 2005;60:563–573. doi: 10.1016/j.socscimed.2004.05.025. [DOI] [PubMed] [Google Scholar]
- 12.Sutcliffe CG, Kobayashi T, Hamapumbu H, Shields T, Mharakurwa S, Thuma PE, et al. Reduced risk of malaria parasitemia following household screening and treatment: a cross-sectional and longitudinal cohort study. PLoS ONE. 2012;7:e31396. doi: 10.1371/journal.pone.0031396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yangzom T, Gueye C, Namgay R, Galappaththy GNL, Thimasarn K, Gosling R, et al. Malaria control in Bhutan: case study of a country embarking on elimination. Malar J. 2012;11:9. doi: 10.1186/1475-2875-11-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Britton S, Cheng Q, McCarthy JS. Novel molecular diagnostic tools for malaria elimination: a review of options from the point of view of high-throughput and applicability in resource limited settings. Malar J. 2016;15:88. doi: 10.1186/s12936-016-1158-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.The malERA Consultative Group on Diagnoses A research agenda for malaria eradication: diagnoses and diagnostics. PLoS Med. 2011;8:e1000396. doi: 10.1371/journal.pmed.1000396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stresman GH, Kamanga A, Moono P, Hamapumbu H, Mharakurwa S, Kobayashi T, et al. A method of active case detection to target reservoirs of asymptomatic malaria and gametocyte carriers in a rural area in Southern Province, Zambia. Malar J. 2010;9:265. doi: 10.1186/1475-2875-9-265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hemingway J, Shretta R, Wells TNC, Bell D, Djimdé AA, Achee N, et al. Tools and strategies for malaria control and elimination: what do we need to achieve a grand convergence in malaria? PLoS Biol. 2016;14:e1002380. doi: 10.1371/journal.pbio.1002380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tanner M, Greenwood B, Whitty CJM, Ansah EK, Price RN, Dondorp AM, et al. Malaria eradication and elimination: views on how to translate a vision into reality. BMC Med. 2015;13:167. doi: 10.1186/s12916-015-0384-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.WHO. The role of mass drug administration, mass screening and treatment, and focal screening and treatment for malaria. Geneva: World Health Organization; 2015. http://www.who.int/malaria/mpac/mpac-sept2015- erg-mda-report.pdf.
- 20.WHO Evidence Review Group. Mass drug administration, mass screening and treatment and focal screening and treatment for malaria. Geneva: World Health Organization; 2015. http://www.who.int/malaria/mpac/mpac-sept2015-erg-mda-report.pdf.
- 21.Moonen B, Cohen JM, Snow RW, Slutsker L, Drakeley C, Smith DL, et al. Operational strategies to achieve and maintain malaria elimination. Lancet. 2010;376:1592–1603. doi: 10.1016/S0140-6736(10)61269-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.WHO . A field manual for low and moderate endemic countries. Geneva: World Health Organization; 2007. [Google Scholar]
- 23.PATH . Market opportunities for new diagnostics to support malaria elimination. Seattle: PATH; 2014. [Google Scholar]
- 24.Gueye CS, Sanders KC, Galappaththy GN, Rundi C, Tobgay T, Sovannaroth S, et al. Active case detection for malaria elimination: a survey among Asia Pacific countries. Malar J. 2013;12:358. doi: 10.1186/1475-2875-12-358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Program for Appropriate Technology in Health; PATH. Target product profile: point-of-care malaria infection detection test for rapid detection of low-density, subclinical malaria infections. 2014. http://sites.path.org/dx/files/2012/11/DIAMETER_IDT_TPP_FINAL_forwebsite.pdf.
- 26.Bell D, Fleurent AE, Hegg MC, Boomgard JD, McConnico CC. Development of new malaria diagnostics: matching performance and need. Malar J. 2016;15:406. doi: 10.1186/s12936-016-1454-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ebels KB, Clerk C, Crudder CH, McGray S, Magnuson K, Tietje K, et al. Incorporating user needs into product development for improved infection detection for malaria elimination programs. In: IEEE glob. humanit. technol. conf. 2014. p. 555–60. http://ieeexplore.ieee.org/document/6970338/.
- 28.von Seidlein L. The failure of screening and treating as a malaria elimination strategy. PLoS Med. 2014;11:e1001595. doi: 10.1371/journal.pmed.1001595. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets supporting the findings presented in this manuscript cannot be shared in a publicly available repository, since data was received under a strict confidentiality agreement. In order to preserve confidentiality, all results presented in the manuscript are in the form of aggregated figures.


