Abstract
Background/Aim: The aim of this study was to assess the inÔidence of MSI in a large series of human hepatocellular carcinomas (HCC) with various etiologies. Materials and Methods: The MSI status was determined by polymerase chain reaction (PCR) using 5 mononucleotide and 13 CAn dinucleotide repeats. Results: None of the 122 HCC samples displayed an MSI-High phenotype, as defined by the presence of alterations at more than 30% of the microsatellite markers analyzed. Yet, limited microsatellite instability consisting in the insertion or deletion of a few repeat motifs was detected in 32 tumor samples (26.2%), regardless of the etiology of the underlying liver disease. MSI tended to be higher in patients with cirrhosis (p=0.051), possibly reflecting an impact of the inflammatory context in this process. Conclusion: Based on a large series of HCC with various etiologies, our study allowed us to definitely conclude that MSI is not a hallmark of HCC.
Keywords: Hepatocellular carcinoma, microsatellite instability, cirrhosis, inflammation
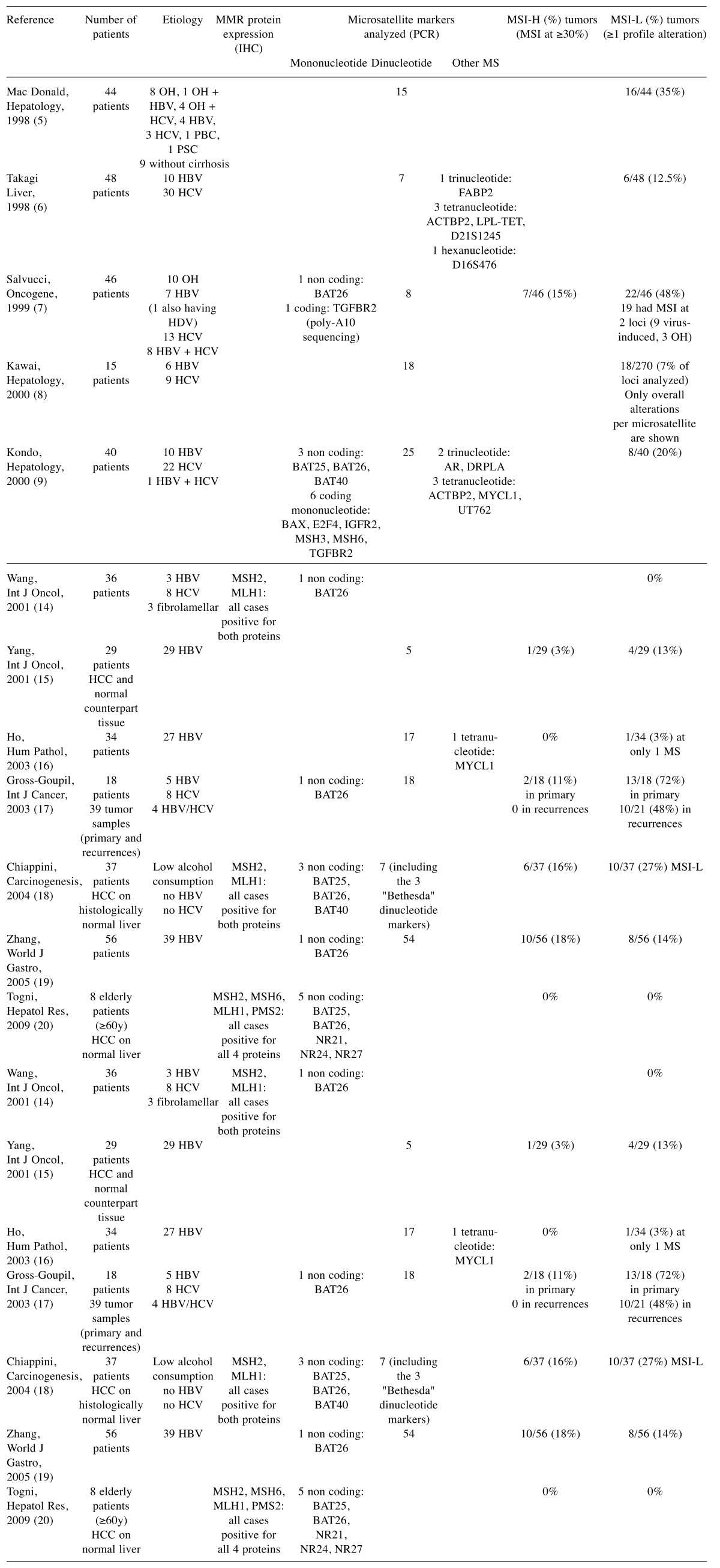
Hepatocellular carcinoma (HCC) is the fifth leading cause of cancer worldwide in men and the seventh in women. Major risk factors for HCC include infection with hepatitis B or C viruses (HBV or HCV), alcohol-induced liver disease. The distribution of risk factors for HCC is highly variable around the world. The HCC incidence is rising in Western countries, due to increasing HCV infection and non-alcoholic steatohepatitis (NASH) development (1). Several molecular classifications of HCC based on the comprehensive analysis of tumor genomes, epigenomes, and transcriptomes, have been established (2). These approaches led to identification of subgroups of tumors sharing recurrent alterations underlying the deregulation of cell cycle and signaling pathways (3), some of which being preferentially associated with a particular causative etiology (4). Several studies have investigated whether HCC might be associated with microsatellite instability (MSI), a well-known oncogenic pathway for other cancers such as colon and gastric cancers (Table I) (5-20). In these studies, the numbers of patients varied from eight to fifty-six (19,20); the etiology of HCC, as well as the techniques used to screen MSI were highly variable, leading to conflicting results with the frequency of MSI ranging from 0-48% (7,14,17).
Table I. Previous studies on MSI in HCC.
CH, Chronic hepatitis; HGDN, high-grade dysplastic nodule; LGDN, low-grade dysplastic nodule; PBC, primary biliary cirrhosis; PSC, primary sclerosing cholangitis.
MSI tumors accumulate mutations due to a defect in mismatch repair (MMR), a DNA repair system dedicated to correcting the errors made by polymerases during replication. The MSI phenotype was originally described in hereditary non-polyposis colorectal cancer, known as Lynch syndrome, in which MMR inactivation results from a monoallelic germline mutation of one of the MMR genes, most frequently MSH2 or MLH1, associated with the subsequent loss of the corresponding wild-type allele in the tumor (21,22). Rare Lynch cases have recently been shown to result from germline EPCAM/TACSTD1 gene deletions leading to MSH2 promoter hypermethylation and transcription inhibition (23). Sporadic MSI colon tumors are twice as frequent as familial cases and generally occur as a consequence of abnormal hypermethylation of MLH1 promoter (24). Interestingly, MSI may be further due to other molecular alterations arising in various pathological situations, notably when mutations occur at very high frequencies, outweighing the MMR capabilities. For example, MSI lymphomas have been reported in immunodeficient patients, due to HIV infection or to iatrogenic immunosuppressive treatment for transplantation (25). Low levels of MSI may also be favored by chronic inflammation or oxidative stress (26,27), as reported in pancreatitis (28) and ulcerative colitis, a chronic disease characterized by free radical stress and colon cancer proneness (29). Interestingly, microRNAs-155 (30) and -21 (31), two major miRNAs in hepatocellular oncogenesis that are also overexpressed in inflammatory bowel diseases (32), down-regulate the expression of MMR core proteins, MSH2 and MLH1 (33,34). Lastly, MMR protein expression defects and MSI have been reported in the inflammation-associated malignant transformation of endometriosis (35). Altogether, these observations indicate that some cases of HCC, especially those associated with a high degree of inflammation, might display some degree of MSI.
Microsatellites are simple sequence repeats of one to six nucleotides, which are particularly prone to insertion or deletion (indel) mutations during replication. In absence of a functional MMR system, indel mutations are left unrepaired and appear as novel alleles with altered lengths. Such novel alleles are classically looked for using various PCR techniques designed to detect MSI tumors. In 1997, an international consensus meeting recommended a panel of five markers, two mononucleotide and three dinucleotide repeat markers, to analyze MSI in colorectal tumors (36). Using this so-called "Bethesda panel", tumors were diagnosed as MSI-High (MSI-H) when displaying instability at two ore more of these markers (36). Later, a pentaplex PCR system comprising five mononucleotide repeats, and allowing 100% sensitivity and specificity with no need to match corresponding normal DNA, became the gold standard in MSI colorectal cancer screening (37,38). Analysis of MMR protein expression by immunohistochemistry (IHC) is also very popular, with MSH2 and MLH1 being performed in routine while MSH6 and PMS2 are analyzed in rare studies. In HCC, MSI has been investigated by IHC (14,18,20), and by PCR using highly variable sets of microsatellite loci (5,6,9-11,13-20,39) (Table I).
The aim of this study was to assess the MSI prevalence in a large series of HCC including various etiologies, representative of the Western HCC population, using the highly sensitive gold-standard mononucleotide pentaplex PCR method, together with 13 polymorphic dinucleotide markers.
Materials and Methods
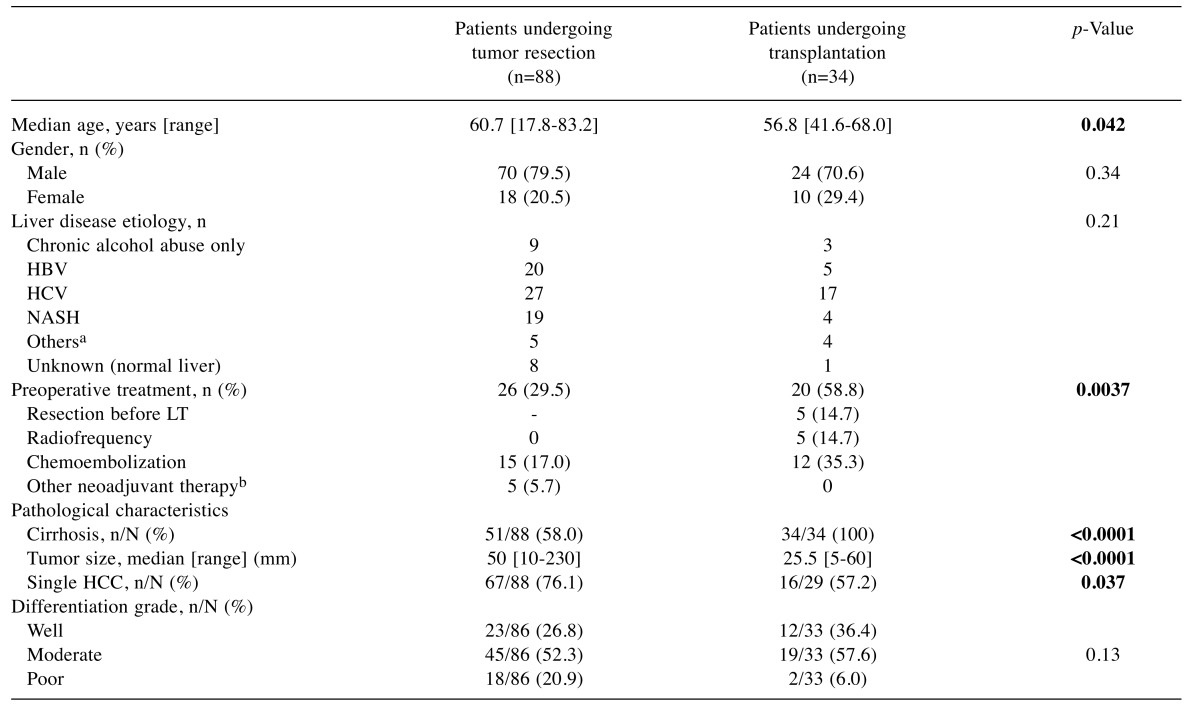
Patients and tumor samples. The study population consisted of 122 patients with histologically-proven HCC who underwent surgery at the Saint-Antoine Hospital, Paris. Each patient provided informed consent for research, and the study has been conducted in accordance with the French laws and regulations. Among them, 88 (72.1%) underwent liver tumor resection and 34 (27.9%) received liver transplantation. There was no significant difference in the underlying liver disease spectrum between the two groups of patients, which was related to chronic alcohol abuse only in 12 patients (9.8%), HCV infection in 44 patients (36%), HBV infection in 25 patients (20.5%), and NASH in 23 patients (18.9%). The diagnosis of cirrhosis based on histology was positive for 84/121 (69%) patients. The surrounding liver showed no sign of fibrosis at the initial pathological examination in 17 patients undergoing tumor resection (19.3%). The tumor size was the largest diameter measured by imaging. The major clinical and pathological characteristics of patients are summarized in Table II. Tumor samples obtained from resected or explanted livers with HCC were retrieved from the HUEP tumor biobank (Pathology department, Saint-Antoine hospital, Hôpitaux Universitaires Est Parisien, AP-HP).
Table II. Clinicopathological characteristics of patients with HCC in relation with surgical management.
aHemochromatosis (n=4), autoimmune hepatitis (n=1), hepatic glycogenosis (n=1), primary biliary cirrhosis (n=3); bSorafenib (n=4), GEMOX (n=1). HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; LT, liver transplantation; NASH, nonalcoholic steatohepatitis; PVE, portal vein embolization. The age and tumor size of patients who underwent tumor resection were compared to those receiving a liver transplant using the unpaired t-test with Welch’s correction. Tumor etiologies and differentiation grades were compared using the Chi-square test. All other clinicopathological characteristics were compared using two-sided Fisher’s exact test. p-Values below 0.05 considered as statistically significant are shown in bold.
DNA extraction and microsatellite instability analysis. DNA was extracted using the Nucleospin Tissue DNA isolation kit (Macherey Nagel) for the frozen samples (n=88), and the Express extract DNA extraction kit (Kappa Biosystems) for the paraffin-embedded samples (n=34), according to the manufacturers instructions. MSI analysis was carried out using 5 quasi-monomorphic mononucleotide repeats markers (BAT25, BAT26, NR21, NR22, NR24) co-amplified in a single pentaplex PCR reaction (40), and 13 highly polymorphic dinucleotide repeat markers (2p16: D2S123; 5q14: D5S107; 8p22: D8S1731, D8S261; 11q13: D11S937, D11S911; 17p12: D17S1791; 17q11: D17S1824, D17S1873; 18p11: D18S53, D18S1114, D18S1132; 18q21: D18S1127). For dinucleotide repeats, the number of markers that could be co-amplified varied from 2 to 5, depending on the amplification efficacy of each primer set and whether DNA was extracted from frozen or paraffin-embedded samples. Primer sequences, chromosome location and amplicon size of the mononucleotide and dinucleotide repeats analyzed are available upon request; forward primers were 5’-end-labeled with a fluorescent dye (FAM, HEX or Dragon Fly Orange, Eurogentec). Amplification of DNA extracted from frozen tumors, was performed using Taq DNA Polymerase (Qiagen), starting with an initial 3 min denaturation step at 95˚C, followed by 35 cycles at 95˚C for 30 sec, 60˚C for 30 sec and 72˚C for 30 sec, with a final extension at 72˚C for 1 min. DNA extracted from paraffin-embedded tumors were amplified using the KAPA2G Robust HotStart ReadyMix because it is efficient on crude DNA preparations possibly containing enzyme inhibitors, such as those obtained from paraffin-embedded samples (KAPA BIOSYSTEMS); the PCR conditions were the same except that the duration of each step was 15 sec. Adequate dilutions of the PCR products were run on an ABI PRISM® 3100 Genetic Analyzer (Applied Biosystems) using Performance Optimized Polymer-7 (Applied Biosystems) and HD400 ROX Size Standard (Applied Biosystems); allelic sizes were defined using the GeneMapper Software (Applied Biosystems).
Statistical analyses. The age and tumor size of patients who underwent tumor resection were compared to those receiving a liver transplant using the unpaired t-test with Welch’s correction. The tumor etiologies and differentiation grades were compared using the Chi-square test. All other clinicopathological characteristics were compared using the two-sided Fisher’s exact test. The numbers of alleles displaying altered profiles in tumors were compared using the unpaired t-test with Welch’s correction. The relationship between the level of MSI and the etiology or the differentiation grade was evaluated using the Kruskal-Wallis test. The impact of cirrhosis on the level of MSI was analyzed by a one-sided Mann-Whitney test. A two-sided Mann-Whitney test was used to evaluate the impact of tumor size or number on the level of MSI. p-Values below 0.05 were considered statistically significant.
Results
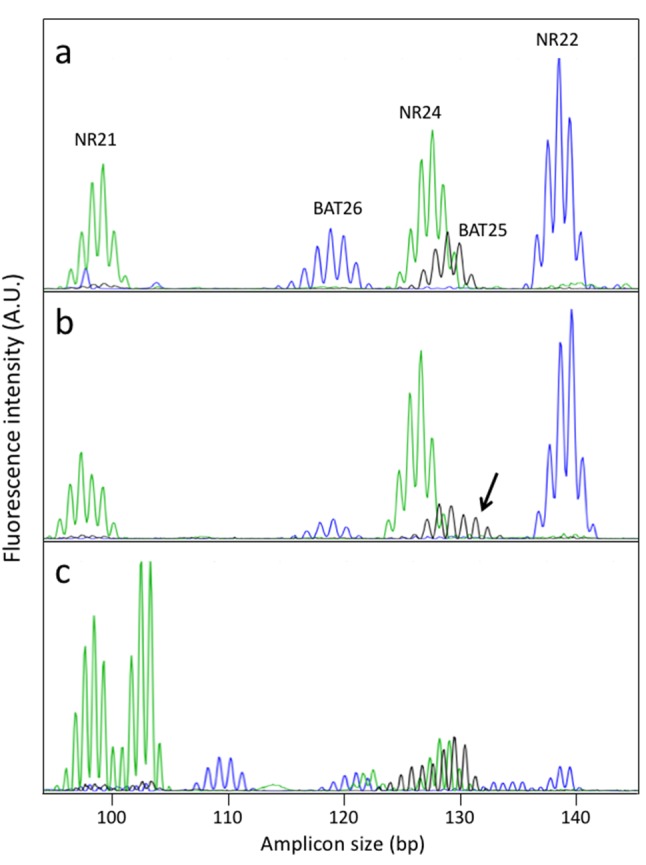
Abnormal profiles consisting in the insertion or deletion of few nucleotides have been observed in 8 tumors (1 at NR21, 1 at BAT26, and 6 at BAT25) (Table III). Figure 1 shows an example of the profiles of the mononucleotide repeats pentaplex PCR performed on tumors showing either a normal profile at all 5 markers (Figure 1a) or displaying an abnormal BAT25 profile (Figure 1b). The shifts occurring in BAT25 are less marked than in a typical MSI colorectal cancer that displays extensive instability, used as a positive control (Figure 1c). The possibility that atypical MS profiles were due to PCR artifacts arising early during the PCR was systematically ruled out by repeating the amplification reactions. The results obtained for tumors retrieved from patients who underwent tumor resection or liver transplantation are shown in Table III. The frequencies of abnormal profiles were comparable in frozen and paraffin-embedded samples (4/439 vs. 4/162, p=0.22; data can be retrieved from Table III). Among the 122 HCC tumors analyzed, none could be considered as displaying a typical MSI-H phenotype as defined by instability in at least 3 of the 5 mononucleotide repeats analyzed. Yet, 8 tumors displayed instability at 1 mononucleotide repeat.
Table III. MS profiles in tumor samples of patients with hepatocellular carcinoma. Normal profiles are shown in grey, abnormal profiles are shown in black, rectangles with a cross indicate that DNA could not be amplified, open rectangles indicate samples that have not been investigated. For each tumor sample, the number of markers analyzed, the number of abnormal profiles and the percentage of abnormal profiles are indicated in the last three columns. Tumor identifiers in the left column start with an R or a T to indicate whether patient underwent tumor resection (R) or transplantation (T); a P at the end indicates paraffin-embedded samples. Abbreviation: MS, microsatellite.
Figure 1. Examples of pentaplex PCR of mononucleotide repeats. (1a) Typical profile of a tumor devoid of microsatellite instability; (1b) HCC case displaying an atypical profile at the BAT25 marker, (1c) typical profile of an MSI colorectal tumor.
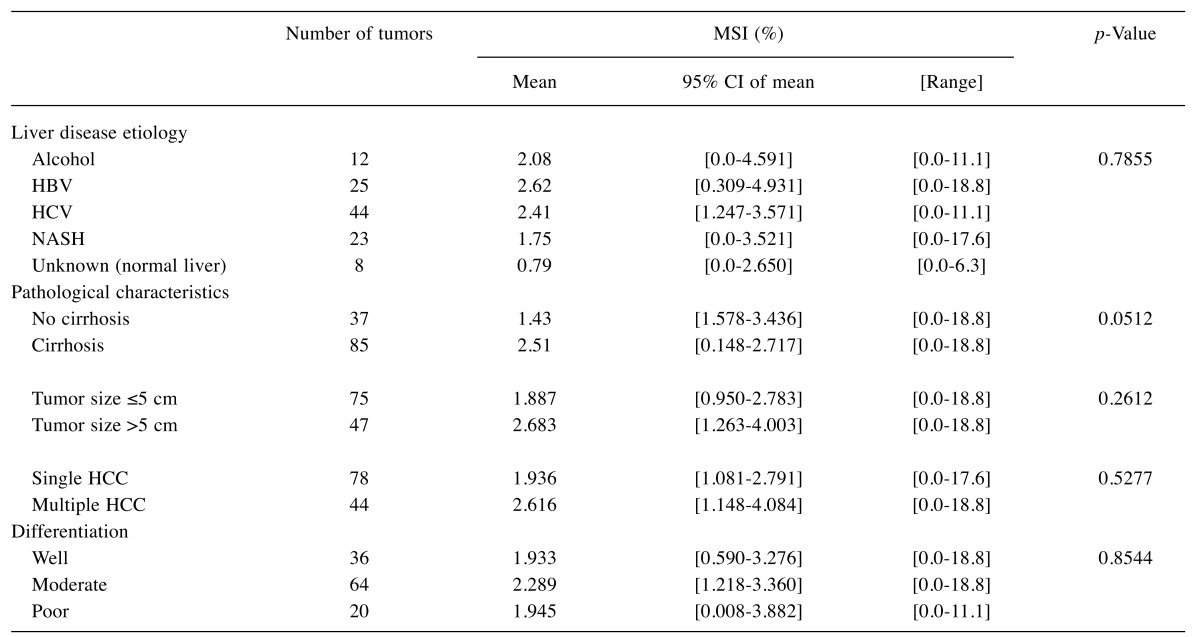
We further screened for instability at 13 dinucleotide repeated loci located on 8 different regions of the genome, with the size of the amplicons varying from 78 to 290 bp. The rates of abnormal profiles in dinucleotide repeats did not differ significantly when comparing frozen and paraffin-embedded tumors (p=0.19, data not shown). The majority of samples (90/122) displayed no detectable instability at any of the 13 dinucleotide repeat loci analyzed. At maximum, 3 abnormal profiles could be detected in 3 tumors (Table III), which is not enough to classify these tumors as MSI-H. According to the recommendations established for colorectal cancers, a tumor should display instability at ≥30-40% of dinucleotide repeats to be considered as MSI-H, meaning 4 out of the 13 markers analyzed in our conditions. Interestingly, low levels of MSI were observed in one-fourth of the tumors (38/122). Moreover, the level of MSI was not related to the etiology of the underlying liver disease, the differentiation grade, the size or number of liver tumors, but tended to be higher in patients with cirrhosis (p=0.051) (Table IV).
Table IV. Relationship between MSI levels and clinicopathological characteristics.
The relationship between the level of MSI and the etiology or the diffentiation grade was evaluated using the Kruskal-Wallis test. The impact of cirrhosis was analyzed using a one-sided Mann-Whitney test. A two-sided Mann-Whitney test was used to evaluate the impact of tumor size or number.
Discussion
The aim of our study was to appraise whether MSI could contribute to HCC oncogenesis, and if instability might be preferentially associated with a specific etiology and/or treatment. Previously published studies that addressed this question varied tremendously in terms of HCC etiology, a characteristic that depends largely on the geographical origin of the patients, and in the number of tumors analyzed that was between 8 and 56 (5,6,9-20,39). Furthermore, the type and number of markers also varied considerably from a single mononucleotide repeat marker (14) to 55 microsatellite markers, mostly dinucleotide repeated sequences (19); few studies also included trinucleotide, tetranucleotide and hexanucleotide repeats whose genetic stability may involve DNA repair pathways other than MMR (6,9,16). Most studies screened MSI using PCR technique, but the definition of MSI was not always clearly specified, which makes the comparisons between the published data even more hazardous. Reported MSI frequency in HCC varied between 0-48%, with a frequency of MSI-H tumors varying from 0-18% when taking into account the consensus definition for MSI-H tumors. Several reasons may contribute to the variability observed in the data published: (i) analyses of MSI profiles using highly polymorphic markers other than mononucleotide repeats are somehow tricky to interpret outside of well-trained laboratories and may lead to misinterpretation, (ii) the number of samples analyzed was often too limited to definitely establish the percentage of MSI, (iii) underlying etiology differed widely among studies. Lastly, the only study performed using the recently developed highly efficient mononucleotide pentaplex PCR system investigated no more than eight cases of HCC; besides these tumors all occurred in elderly patients with no chronic liver disease (20). Based on our expertise in analyzing microsatellite profiles (41-47), we investigated MSI in a large collection of HCC of various etiologies using robust and accurate PCR-based MSI testing techniques, both at mononucleotide repeats, using the gold-standard pentaplex PCR based on five nearly monomorphic mononucleotide markers, and at 13 dinucleotide repeats located on 8 different chromosomes.
MSH2/MSH6 complexes, known as MutSα, are able to efficiently bind to most base-base mismatches (with the exception of CC mismatches), and to loops of one or two nucleotides, whereas MutSβ, consisting of MSH2 and MSH3, preferentially repairs larger heteroduplexes loops, such as those resulting from indel of dinucleotide repeats. Excision of the newly replicated DNA fragment containing a mismatch involves MLH1/PMS2 complex, referred as MutLα, that acts upon binding of either MutSα or MutSβ. Thus, analyzing instability at both mononucleotide and dinucleotide repeated sequences is needed to investigate these partially redundant DNA repair systems. The gold-standard screening method for MSI in colorectal cancers is now based solely on the analysis of mononucleotide repeats, which are particularly prone to slippage during replication as they harbour high number of repeated motifs. Yet, this method is adequate in the case of colorectal cancers, as we know that MSI results from a defect in one of the component of MutSα or MutLα (MSH2, MSH6, MLH1, PMS2). However, in the case of HCC, we considered the possibility that MSI might arise through other mechanisms. Consequently, we included dinucleotide repeats in our panel to investigate the possibility of a defect in MutSβ.
Though none of the HCC analyzed could be classified as MSI-H, microsatellite instability arose at one locus or more in one-fourth of the HCC analyzed, without being associated with a particular underlying liver disease etiology. Mononucleotide and dinucleotide repeats were equally frequently altered when taking into consideration the number of markers of each type analyzed. Interestingly, the level of MSI tended to be higher in patients with cirrhosis, a difference that failed to reach statistical significance (p=0.051), but corroborates the existence of a link between inflammation and genetic instability (48).
Interestingly, a recent study showed that cells lacking the H3K36 trimethyltransferase SETD2 display MSI and an elevated spontaneous mutation frequency, characteristic of MMR deficient cells (49). Thus, it is tempting to propose that deleterious mutations of SETD2 such as those recently reported in a subset of HCC (2) may be responsible, at least in part, in microsatellite alterations arising in these tumors. Whether other mechanisms known to disturb the histone H3 methylation status, such as the HCV nonstructural protein 5A that has been shown to interact with the lysine methyltransferase SET and MYND domain-containing 3 (SMYD3) (50), may also induce MSI remains to be investigated on large series of HCV-related HCC.
In conclusion, our observations made with highly sensitive and specific panel markers on a large series of HCC indicate that low levels of genetic instability in both mononucleotide and dinucleotide repeated sequences occur quite commonly in HCC, independently of the etiology of the underlying liver disease. The molecular mechanisms inducing low MSI in HCC remain to be identified. They may differ among tumours, possibly involving chronic oxidative stress and inflammation that are hallmarks of liver cancers, or deleterious mutations in SETD2, as recently described in a minority of cases of HCC (2). Whether low MSI levels have an impact on the prognosis of HCC, as in the case of colorectal cancer, remains to be investigated.
Conflicts of Interest
The Authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript.
Acknowledgements
Claire GOUMARD was a recipient of a grant from the Institut National du Cancer (French National Cancer Institute). The funding source had no role in study design, data collection, data analysis, data interpretation or the writing of the report. The authors thank Pr Olivier ROSMORDUC and Dr Laëtitia FARTOUX for their contribution to the care of patients with HCC. They further thank the HUEP tumor biobank (Pathology Department, Saint-Antoine Hospital, Hôpitaux Universitaires Est Parisien, AP-HP) for providing good quality tumor samples, as well Sylvie DUMONT for her expert technical help in managing them (LUMIC histomorphology technological platform, UMS30, UPMC, Paris).
References
- 1.El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365:1118–1127. doi: 10.1056/NEJMra1001683. [DOI] [PubMed] [Google Scholar]
- 2.Schulze K, Imbeaud S, Letouze E, Alexandrov LB, Calderaro J, Rebouissou S, Couchy G, Meiller C, Shinde J, Soysouvanh F, Calatayud AL, Pinyol R, Pelletier L, Balabaud C, Laurent A, Blanc JF, Mazzaferro V, Calvo F, Villanueva A, Nault JC, Bioulac-Sage P, Stratton MR, Llovet JM, Zucman-Rossi J. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat Genet. 2015;47:505–511. doi: 10.1038/ng.3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hass HG, Vogel U, Scheurlen M, Jobst J. Gene-expression analysis identifies specific patterns of dysregulated molecular pathways and genetic subgroups of human hepatocellular carcinoma. Anticancer Res. 2016;36:5087–5095. doi: 10.21873/anticanres.11078. [DOI] [PubMed] [Google Scholar]
- 4.Shibata T, Aburatani H. Exploration of liver cancer genomes. Nat Rev Gastroenterol Hepatol. 2014;11:340–349. doi: 10.1038/nrgastro.2014.6. [DOI] [PubMed] [Google Scholar]
- 5.Macdonald GA, Greenson JK, Saito K, Cherian SP, Appelman HD, Boland CR. Microsatellite instability and loss of heterozygosity at DNA mismatch repair gene loci occurs during hepatic carcinogenesis. Hepatology. 1998;28:90–97. doi: 10.1002/hep.510280114. [DOI] [PubMed] [Google Scholar]
- 6.Takagi K, Esumi M, Takano S, Iwai S. Replication error frequencies in primary hepatocellular carcinoma: A comparison of solitary primary versus multiple primary cancers. Liver. 1998;18:272–276. doi: 10.1111/j.1600-0676.1998.tb00165.x. [DOI] [PubMed] [Google Scholar]
- 7.Salvucci M, Lemoine A, Saffroy R, Azoulay D, Lepere B, Gaillard S, Bismuth H, Reynes M, Debuire B. Microsatellite instability in european hepatocellular carcinoma. Oncogene. 1999;18:181–187. doi: 10.1038/sj.onc.1202279. [DOI] [PubMed] [Google Scholar]
- 8.Kawai H, Suda T, Aoyagi Y, Isokawa O, Mita Y, Waguri N, Kuroiwa T, Igarashi M, Tsukada K, Mori S, Shimizu T, Suzuki Y, Abe Y, Takahashi T, Nomoto M, Asakura H. Quantitative evaluation of genomic instability as a possible predictor for development of hepatocellular carcinoma: Comparison of loss of heterozygosity and replication error. Hepatology. 2000;31:1246–1250. doi: 10.1053/jhep.2000.7298. [DOI] [PubMed] [Google Scholar]
- 9.Kondo Y, Kanai Y, Sakamoto M, Mizokami M, Ueda R, Hirohashi S. Genetic instability and aberrant DNA methylation in chronic hepatitis and cirrhosis – a comprehensive study of loss of heterozygosity and microsatellite instability at 39 loci and DNA hypermethylation on 8 cpg islands in microdissected specimens from patients with hepatocellular carcinoma. Hepatology. 2000;32:970–979. doi: 10.1053/jhep.2000.19797. [DOI] [PubMed] [Google Scholar]
- 10.Maggioni M, Coggi G, Cassani B, Bianchi P, Romagnoli S, Mandelli A, Borzio M, Colombo P, Roncalli M. Molecular changes in hepatocellular dysplastic nodules on microdissected liver biopsies. Hepatology. 2000;32:942–946. doi: 10.1053/jhep.2000.18425. [DOI] [PubMed] [Google Scholar]
- 11.Roncalli M, Bianchi P, Grimaldi GC, Ricci D, Laghi L, Maggioni M, Opocher E, Borzio M, Coggi G. Fractional allelic loss in non-end-stage cirrhosis: Correlations with hepatocellular carcinoma development during follow-up. Hepatology. 2000;31:846–850. doi: 10.1053/he.2000.5790. [DOI] [PubMed] [Google Scholar]
- 12.Yamamoto H, Itoh F, Fukushima H, Kaneto H, Sasaki S, Ohmura T, Satoh T, Karino Y, Endo T, Toyota J, Imai K. Infrequent widespread microsatellite instability in hepatocellular carcinomas. Int J Oncol. 2000;16:543–547. doi: 10.3892/ijo.16.3.543. [DOI] [PubMed] [Google Scholar]
- 13.Dore MP, Realdi G, Mura D, Onida A, Massarelli G, Dettori G, Graham DY, Sepulveda AR. Genomic instability in chronic viral hepatitis and hepatocellular carcinoma. Hum Pathol. 2001;32:698–703. doi: 10.1053/hupa.2001.25593. [DOI] [PubMed] [Google Scholar]
- 14.Wang L, Bani-Hani A, Montoya DP, Roche PC, Thibodeau SN, Burgart LJ, Roberts LR. HMLH1 and hMSH2 expression in human hepatocellular carcinoma. Int J Oncol. 2001;19:567–570. [PubMed] [Google Scholar]
- 15.Yang EB, Zhao YN, Zhang K, Mack P. Microsatellite alterations in human hepatocellular carcinoma infected with hepatitis b virus: Associated with the elevation of serum alpha-fetoprotein. Int J Oncol. 2001;19:513–518. doi: 10.3892/ijo.19.3.513. [DOI] [PubMed] [Google Scholar]
- 16.Ho MK, Lee JM, Chan CK, Ng IO. Allelic alterations in nontumorous liver tissues and corresponding hepatocellular carcinomas from chinese patients. Hum Pathol. 2003;34:699–705. doi: 10.1016/s0046-8177(03)00175-8. [DOI] [PubMed] [Google Scholar]
- 17.Gross-Goupil M, Riou P, Emile JF, Saffroy R, Azoulay D, Lacherade I, Receveur A, Piatier-Tonneau D, Castaing D, Debuire B, Lemoine A. Analysis of chromosomal instability in pulmonary or liver metastases and matched primary hepatocellular carcinoma after orthotopic liver transplantation. Int J Cancer. 2003;104:745–751. doi: 10.1002/ijc.11017. [DOI] [PubMed] [Google Scholar]
- 18.Chiappini F, Gross-Goupil M, Saffroy R, Azoulay D, Emile JF, Veillhan LA, Delvart V, Chevalier S, Bismuth H, Debuire B, Lemoine A. Microsatellite instability mutator phenotype in hepatocellular carcinoma in non-alcoholic and non-virally infected normal livers. Carcinogenesis. 2004;25:541–547. doi: 10.1093/carcin/bgh035. [DOI] [PubMed] [Google Scholar]
- 19.Zhang SH, Cong WM, Xian ZH, Wu MC. Clinicopathological significance of loss of heterozygosity and microsatellite instability in hepatocellular carcinoma in China. World J Gastroenterol. 2005;11:3034–3039. doi: 10.3748/wjg.v11.i20.3034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Togni R, Bagla N, Muiesan P, Miquel R, O’Grady J, Heaton N, Knisely AS, Portmann B, Quaglia A. Microsatellite instability in hepatocellular carcinoma in non-cirrhotic liver in patients older than 60 years. Hepatol Res. 2009;39:266–273. doi: 10.1111/j.1872-034X.2008.00455.x. [DOI] [PubMed] [Google Scholar]
- 21.Jacob S, Praz F. DNA mismatch repair defects: Role in colorectal carcinogenesis. Biochimie. 2002;84:27–47. doi: 10.1016/s0300-9084(01)01362-1. [DOI] [PubMed] [Google Scholar]
- 22.Sijmons RH, Hofstra RM. Review: Clinical aspects of hereditary DNA mismatch repair gene mutations. DNA Repair (Amst) 2016;38:155–162. doi: 10.1016/j.dnarep.2015.11.018. [DOI] [PubMed] [Google Scholar]
- 23.Rumilla K, Schowalter KV, Lindor NM, Thomas BC, Mensink KA, Gallinger S, Holter S, Newcomb PA, Potter JD, Jenkins MA, Hopper JL, Long TI, Weisenberger DJ, Haile RW, Casey G, Laird PW, Le Marchand L, Thibodeau SN. Frequency of deletions of EPCAM (TACSTD1) in MSH2-associated lynch syndrome cases. J Mol Diagn. 2011;13:93–99. doi: 10.1016/j.jmoldx.2010.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kane MF, Loda M, Gaida GM, Lipman J, Mishra R, Goldman H, Jessup JM, Kolodner R. Methylation of the hMLH1 promoter correlates with lack of expression of hmlh1 in sporadic colon tumors and mismatch repair-defective human tumor cell lines. Cancer Res. 1997;57:808–811. [PubMed] [Google Scholar]
- 25.Duval A, Raphael M, Brennetot C, Poirel H, Buhard O, Aubry A, Martin A, Krimi A, Leblond V, Gabarre J, Davi F, Charlotte F, Berger F, Gaidano G, Capello D, Canioni D, Bordessoule D, Feuillard J, Gaulard P, Delfau MH, Ferlicot S, Eclache V, Prevot S, Guettier C, Lefevre PC, Adotti F, Hamelin R. The mutator pathway is a feature of immunodeficiency-related lymphomas. Proc Natl Acad Sci USA. 2004;101:5002–5007. doi: 10.1073/pnas.0400945101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gasche C, Chang CL, Rhees J, Goel A, Boland CR. Oxidative stress increases frameshift mutations in human colorectal cancer cells. Cancer Res. 2001;61:7444–7448. [PubMed] [Google Scholar]
- 27.Chang CL, Marra G, Chauhan DP, Ha HT, Chang DK, Ricciardiello L, Randolph A, Carethers JM, Boland CR. Oxidative stress inactivates the human DNA mismatch repair system. Am J Physiol Cell Physiol. 2002;283:C148–154. doi: 10.1152/ajpcell.00422.2001. [DOI] [PubMed] [Google Scholar]
- 28.Brentnall TA, Chen R, Lee JG, Kimmey MB, Bronner MP, Haggitt RC, Kowdley KV, Hecker LM, Byrd DR. Microsatellite instability and K-ras mutations associated with pancreatic adenocarcinoma and pancreatitis. Cancer Res. 1995;55:4264–4267. [PubMed] [Google Scholar]
- 29.Brentnall TA, Crispin DA, Bronner MP, Cherian SP, Hueffed M, Rabinovitch PS, Rubin CE, Haggitt RC, Boland CR. Microsatellite instability in nonneoplastic mucosa from patients with chronic ulcerative colitis. Cancer Res. 1996;56:1237–1240. [PubMed] [Google Scholar]
- 30.Han ZB, Chen HY, Fan JW, Wu JY, Tang HM, Peng ZH. Up-regulation of microRNA-155 promotes cancer cell invasion and predicts poor survival of hepatocellular carcinoma following liver transplantation. J Cancer Res Clin Oncol. 2012;138:153–161. doi: 10.1007/s00432-011-1076-z. [DOI] [PubMed] [Google Scholar]
- 31.Zhu Q, Wang Z, Hu Y, Li J, Li X, Zhou L, Huang Y. miR-21 promotes migration and invasion by the miR-21-PDCD4-AP-1 feedback loop in human hepatocellular carcinoma. Oncol Rep. 2012;27:1660–1668. doi: 10.3892/or.2012.1682. [DOI] [PubMed] [Google Scholar]
- 32.Svrcek M, El-Murr N, Wanherdrick K, Dumont S, Beaugerie L, Cosnes J, Colombel JF, Tiret E, Flejou JF, Lesuffleur T, Duval A. Overexpression of micrornas-155 and 21 targeting mismatch repair proteins in inflammatory bowel diseases. Carcinogenesis. 2013;34:828–834. doi: 10.1093/carcin/bgs408. [DOI] [PubMed] [Google Scholar]
- 33.Valeri N, Gasparini P, Fabbri M, Braconi C, Veronese A, Lovat F, Adair B, Vannini I, Fanini F, Bottoni A, Costinean S, Sandhu SK, Nuovo GJ, Alder H, Gafa R, Calore F, Ferracin M, Lanza G, Volinia S, Negrini M, McIlhatton MA, Amadori D, Fishel R, Croce CM. Modulation of mismatch repair and genomic stability by miR-155. Proc Natl Acad Sci USA. 2010;107:6982–6987. doi: 10.1073/pnas.1002472107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Valeri N, Gasparini P, Braconi C, Paone A, Lovat F, Fabbri M, Sumani KM, Alder H, Amadori D, Patel T, Nuovo GJ, Fishel R, Croce CM. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2) Proc Natl Acad Sci USA. 2010;107:21098–21103. doi: 10.1073/pnas.1015541107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fuseya C, Horiuchi A, Hayashi A, Suzuki A, Miyamoto T, Hayashi T, Shiozawa T. Involvement of pelvic inflammation-related mismatch repair abnormalities and microsatellite instability in the malignant transformation of ovarian endometriosis. Hum Pathol. 2012;43:1964–1972. doi: 10.1016/j.humpath.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 36.Boland CR, Thibodeau SN, Hamilton SR, Sidransky D, Eshleman JR, Burt RW, Meltzer SJ, Rodriguez-Bigas MA, Fodde R, Ranzani GN, Srivastava S. A National Cancer Institute workshop on microsatellite instability for cancer detection and familial predisposition: Development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res. 1998;58:5248–5257. [PubMed] [Google Scholar]
- 37.Buhard O, Cattaneo F, Wong YF, Yim SF, Friedman E, Flejou JF, Duval A, Hamelin R. Multipopulation analysis of polymorphisms in five mononucleotide repeats used to determine the microsatellite instability status of human tumors. J Clin Oncol. 2006;24:241–251. doi: 10.1200/JCO.2005.02.7227. [DOI] [PubMed] [Google Scholar]
- 38.Soreide K. High-fidelity of five quasimonomorphic mononucleotide repeats to high-frequency microsatellite instability distribution in early-stage adenocarcinoma of the colon. Anticancer Res. 2011;31:967–971. [PubMed] [Google Scholar]
- 39.Salvucci M, Lemoine A, Azoulay D, Sebagh M, Bismuth H, Reyns M, May E, Debuire B. Frequent microsatellite instability in post hepatitis B viral cirrhosis. Oncogene. 1996;13:2681–2685. [PubMed] [Google Scholar]
- 40.Suraweera N, Duval A, Reperant M, Vaury C, Furlan D, Leroy K, Seruca R, Iacopetta B, Hamelin R. Evaluation of tumor microsatellite instability using five quasimonomorphic mononucleotide repeats and pentaplex pcr. Gastroenterology. 2002;123:1804–1811. doi: 10.1053/gast.2002.37070. [DOI] [PubMed] [Google Scholar]
- 41.Foucault F, Praz F, Jaulin C, Amor-Gueret M. Experimental limits of PCR analysis of (CA)n repeat alterations. Trends Genet. 1996;12:450–452. doi: 10.1016/0168-9525(96)99996-5. [DOI] [PubMed] [Google Scholar]
- 42.Jacob S, Aguado M, Fallik D, Praz F. The role of the DNA mismatch repair system in the cytotoxicity of the topoisomerase inhibitors camptothecin and etoposide to human colorectal cancer cells. Cancer Res. 2001;61:6555–6562. [PubMed] [Google Scholar]
- 43.Fallik D, Borrini F, Boige V, Viguier J, Jacob S, Miquel C, Sabourin JC, Ducreux M, Praz F. Microsatellite instability is a predictive factor of the tumor response to irinotecan in patients with advanced colorectal cancer. Cancer Res. 2003;63:5738–5744. [PubMed] [Google Scholar]
- 44.Mongiat-Artus P, Miquel C, Van der Aa M, Buhard O, Hamelin R, Soliman H, Bangma C, Janin A, Teillac P, van der Kwast T, Praz F. Microsatellite instability and mutation analysis of candidate genes in urothelial cell carcinomas of upper urinary tract. Oncogene. 2006;25:2113–2118. doi: 10.1038/sj.onc.1209229. [DOI] [PubMed] [Google Scholar]
- 45.Miquel C, Jacob S, Grandjouan S, Aime A, Viguier J, Sabourin JC, Sarasin A, Duval A, Praz F. Frequent alteration of DNA damage signalling and repair pathways in human colorectal cancers with microsatellite instability. Oncogene. 2007;26:5919–5926. doi: 10.1038/sj.onc.1210419. [DOI] [PubMed] [Google Scholar]
- 46.Zaanan A, Cuilliere-Dartigues P, Guilloux A, Parc Y, Louvet C, de Gramont A, Tiret E, Dumont S, Gayet B, Validire P, Flejou JF, Duval A, Praz F. Impact of p53 expression and microsatellite instability on stage III colon cancer disease-free survival in patients treated by 5-fluorouracil and leucovorin with or without oxaliplatin. Ann Oncol. 2010;21:772–780. doi: 10.1093/annonc/mdp383. [DOI] [PubMed] [Google Scholar]
- 47.Zaanan A, Flejou JF, Emile JF, des Guetz G, Cuilliere-Dartigues P, Malka D, Lecaille C, Validire P, Louvet C, Rougier P, de Gramont A, Bonnetain F, Praz F, Taieb J. Defective mismatch repair status as a prognostic biomarker of disease-free survival in stage III colon cancer patients treated with adjuvant FOLFOX chemotherapy. Clin Cancer Res. 2011;17:7470–7478. doi: 10.1158/1078-0432.CCR-11-1048. [DOI] [PubMed] [Google Scholar]
- 48.Sesti F, Tsitsilonis OE, Kotsinas A, Trougakos IP. Oxidative stress-mediated biomolecular damage and inflammation in tumorigenesis. In Vivo. 2012;26:395–402. [PubMed] [Google Scholar]
- 49.Li F, Mao G, Tong D, Huang J, Gu L, Yang W, Li GM. The histone mark H3K36me3 regulates human DNA mismatch repair through its interaction with mutSalpha. Cell. 2013;153:590–600. doi: 10.1016/j.cell.2013.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Eberle CA, Zayas M, Stukalov A, Pichlmair A, Alvisi G, Muller AC, Bennett KL, Bartenschlager R, Superti-Furga G. The lysine methyltransferase SMYD3 interacts with hepatitis c virus NS5a and is a negative regulator of viral particle production. Virology. 2014;462-463:34–41. doi: 10.1016/j.virol.2014.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]