Abstract
Bone homeostasis depends on the resorption of bone by osteoclasts and formation of bone by osteoblasts. Imbalance of this tightly coupled process can cause diseases such as osteoporosis. Thus, the mechanisms that regulate communication between osteoclasts and osteoblasts are critical to bone cell biology. It has been shown that osteoblasts and osteoclasts can communicate with each other through direct cell-cell contact, cytokines and extracellular matrix interaction. Osteoblasts can affect osteoclast formation, differentiation or apoptosis through several pathways, such as OPG/RANKL/RANK, LGR4/RANKL/RANK, Ephrin2/ephB4 and Fas/FasL pathways. Conversely, osteoclasts also influence formation of bone by osteoblasts via the d2 isoform of vacuolar (H+) ATPase (v-ATPase) V0 domain (Atp6v0d2), complement component 3a, semaphorin 4D or microRNAs. In addition, cytokine released from the resorbed bone matrix, such as TGF-β and IGF-1 also affects the activity of osteoblasts. Several reviews have been performed on the osteoblasts-osteoclasts communication. However, few reviews have shown the research advances in the recent years. In this review we summarized the current knowledge on osteoblast-osteoclast communication.
Keywords: bone remodeling, osteoblast, osteoclast, communication
Bone is in a constant state of remodeling, which is important for the maintenance of normal skeletal structure and function. Thus, imbalance can result in disease such as osteoporosis. Many types of cells and factors are involved in the process of bone remodeling. Osteoblast and osteoclast are the two main cells participating in those progresses (Matsuo and Irie, 2008). Osteoclasts are responsible for aged bone resorption and osteoblasts are responsible for new bone formation (Matsuoka et al., 2014). The resorption and formation is in stable at physiological conditions. However, when the balance is disturbed, bone architecture or function will be abnormal. Bone metabolism diseases, such as osteoporosis or osteopetrosis will occur (Zaidi, 2007).
Many cytokine, hormones and signaling pathway are involved in in bone remodeling (Matsuo and Irie, 2008). It has been shown that osteoblast and osteoclast can communicate with each other through direct cell-cell contact, cytokine or cell-bone matrix (Tamma and Zallone, 2012). The molecular mechanisms of cellular communication between osteoblast and osteoclast are one of the central issues in bone cell biology (Mundy and Elefteriou, 2006). The communication between osteoblast and osteoclast occur at various stages of differentiation (Matsuo and Irie, 2008). Earlier studies suggested that osteoblast could regulate osteoclast formation (Rodan and Martin, 1981). Subsequently, more and more studies showed that osteoclast can also affect osteoblast activity (Matsuoka et al., 2014; Wang et al., 2015).
Several reviews have been performed on the osteoblasts-osteoclasts interaction (Matsuo and Irie, 2008; Tamma and Zallone, 2012). However, few reviews were published in the recent years. In this short review, we highlight the major research progresses of communication between osteoblasts and osteoclasts.
The role of osteoblasts in osteoclast formation
Cell-cell contact
It has been shown that osteoblast can communicate with osteoclast via direct contact. When those two cells contact, they can form gap junctions and small water-soluble molecules can passage between the two cell types. Everts et al. (2002) have shown that bone lining cells, a subpopulation of the osteoblast, are in close contact with osteoclasts attached to bone. The initiation of osteoclastogenesis mainly depends on the interaction between those two cells (Everts et al., 2002; Matsuo and Irie, 2008). Transmission electron micrograph also showed that there are direct contact between mature osteoclasts and osteoblasts, and the basic multicellular unit is the site for cell-cell contact (Matsuo and Irie, 2008).
Ephrin2/ephB4
Bone cells and its progenitor both express ephs and ephrins(Tamma and Zallone, 2012). Zhao et al.(2006) showed that there were other bidirectional regulations between osteoblast and osteoclast. This regulation was mediated via transmembrane ligland, ephrin2, on osteoclast and receptor tyrosine kinase, ephB4, on osteoblast and osteocytes. Osteoblast could inhibit osteoclast formation via ephb4 and osteoclast could stimulate osteoblast differentiation via ephrin2. The forward signaling mediated by EPH (OC-OB) stimulates osteoblast differentiation and induces bone formation; the reverse signaling mediated by Ephrin (OB-OC) inhibits bone resorption. Furthermore, Zhao et al. (2006) showed that ephrinB2 signaling inhibits osteoclastogenesis by blocking induction of Fos and its transcriptional target Nfatc1, which is induced by RANKL. Additionally, EphB4 signaling is known to induce osteogenic regulatory factors, such as Dlx5, Osx and Runx2.
MSF/MCP-1
Osteoblasts can secrete macrophage colony-stimulating (M-CSF)(Lacey et al., 1994), which binds to its receptor c-Fms expressed on macrophages and osteoclasts (Wiktor-Jedrzejczak et al., 1990). The number of osteoclasts in M-CSF knock-out mice was obviously decreased compared with the wild-type mice. M-CSF is an important cytokine for survival, differentiation, cell migration and activity in macrophages and osteoclasts (Wiktor-Jedrzejczak et al., 1990; Kim and Kim, 2016). M-CSF also enhanced the differentiation of bone marrow precursors to osteoclast precursors and up-regulated the receptor activator of nuclear factor κB (RANK) expression in bone marrow precursors (Sambandam et al., 2010).
Osteoblasts also can produce monocyte chemoattractant protein-1 (MCP-1, or CCL2)(Graves et al., 1999), which is a candidate recruiter of osteoclast precursors (Li et al., 2007). MCP-1 binds with it receptor expressed in osteoclast precursors, and then the circulating osteoclast precursor position the bone surface to differentiate into active osteoclast. However, in normal bone MCP-1is not expressed. Inflammation can stimulate MCP-1 expression in osteoblast.
OPG/RANKL/RANK
Before the discovery of RANKL, OPG and RANK, many groups observed that osteoblasts produce cytokines to regulate osteoclast differentiation and formation.
In the 1990s, four groups independently found that OPG/RANKL plays a critical role during osteoclast formation (Boyce and Xing, 2008). First, they found that OPG protects against bone loss (Simonet et al., 1997; Yasuda et al., 1998a), and then they discovered its ligand (OPGL) using OPG as a probe (Lacey et al., 1998; Yasuda et al., 1998b). Further, OPGL was identified as a member of TNF super family, which is called receptor activator of nuclear factor-kb ligland (RANKL)(Anderson et al., 1997), or TNF-related activation induced cytokine (TRANCE)(Wong et al., 1997). RANK had been discovered by another group when they sequence cDNAs from a human bone marrow derived myeloid dendritic-cell cDNA library, and it was found to be the receptor for OPGL (Dougall et al., 1999).
OPG is also called osteoclastogenesis inhibitory factor (OCIF). OPG is produced by many cell types besides osteoblasts, such as cells in the heart, liver and spleen. It has been shown that B cells are the major source of OPG in the bone marrow of mice, which account for 64% of total bone marrow OPG production (Li et al., 2007). Osteopetrosis is observed in transgenic mice over-expressing OPG due to the lack of osteoclasts (Simonet et al., 1997), OPG knock-out mice have osteoporosis due to unregulated osteoclasts (Bucay et al., 1998). OPG, as a decoy receptor, can bind with RANKL and block its bind and activation with RANK (Boyle et al., 2003). Consequently, the main signaling pathway of osteoclast differentiation and activation were blocked. OPG expression in osteoblasts is regulated by amount of hormones, cytokines and growth factors (Theoleyre et al., 2004), such as estrogen, 1, 25(OH)2D3 and TNF.
RANKL is also called osteoclast differentiation factor (ODF), TNF-related activation-induced cytokine (TRANCE) and osteoprotegerin ligland(OPGL)(Lacey et al. 1998). The generally accepted nomenclature, RANKL, was established by the American Society of Bone Mineral Research in 2000 (The American Society for Bone and Mineral Research President’s Committee on Nomenclature). RANKL is highly expressed in osteoblasts, activated T cells, lymph nodes, thymus, mammary glands and lung, and lowly expressed in bone marrow(Kearns et al., 2008). RANKL is typically membrane-bound on osteoblasts (Kearns et al., 2008). The secreted protein is from the membrane form due to either proteolytic cleavage or alternative splicing (Ikeda et al. 2001). RANKL can bind with its receptor RANK, which is expressed on osteoclastic progenitor cells, and activates the down signaling pathways related with cell growth and differentiation (Boyle et al., 2003). A variety of cytokines, hormones and growth factors can regulate RANKL expression, including PTH, estrogen, and inflammatory cytokines. OPG/RANKL ratio critically influences the osteoclast formation.
RANK is a type I transmembrane protein member of the TNF receptor superfamily. It is highly expressed on membrane of osteoclast progenitor, mature osteoclasts and dendritic cells and mammary glands (Fata et al., 2000). The cancers with high bone metastatic potential also express RANK, such as breast and prostate cancer (Kim et al. 2006). RANK binds with its ligand RANKL, and then activates TNF receptor-associated factors (TRAFs) family, including TRAFs 2,5 and 6. However, TRAF6 alone can induce osteoclastogenesis (Kadono 2001) and is essential for the activation of intracellular signal transduction pathways(Kim et al., 1999). RANK/TRAF regulates osteoclast formation, activation and survival via different signaling pathways (Boyle et al., 2003), including JNK/AP-1, IκK/NF-κB, c-myc and calcineurin/NFATc1 for osteoclast formation; src and MKK6/p38/MITF for activation.
LGR4/RANKL/RANK
A recent study showed that leucine-rich repeat-containing G-protein-coupled receptor 4 (LGR4, also called GPR48) is also receptor for RANKL (Luo et al., 2016). RANKL can directly interact with human LGR4. They further confirmed that LGR4 was expressed in osteoclasts. The in vivo study indicated that low bone mass and osteoclast hyperactivation were observed in Lgr4-deficient mice. Number, surface area and size of osteoclasts were markedly higher in Lgr4-deficient mice compared with those in control mice. In vitro research demonstrated that Lgr4-deficient promoted osteoclasts formation and inhibited its apoptosis. Further data showed that the Lgr4 loss induced osteoclastogenesis did not depend on LGR4 ligand R-spondin1 or norrin. However, LGR4 inhibited RANKL binding to RANK in a dose-dependent manner, which indicated that LGR4 competes with RANK to bind with RANKL. The downstream signaling pathways of osteoclast differentiation and survival were attenuated. LGR4 may be act as another decoy receptor for RANKL.
Sema3A/Nrp
Semaphorins are axonal growth cone guidance molecules, which are involved in the interaction between osteoblasts and osteoclasts (Negishi-Koga et al., 2011). Hayashi et al. (Hayashi et al., 2012) identified some proteins secreted by osteoblast that inhibit osteoclast differentiation. They found that axon guidance molecule Sema3A may play important role. They further showed that the recombinant Sema3A could inhibit osteoclast differentiation in a dose-dependent manner. Sema3A mRNA can be detected in isolated osteoblasts and osteocytes. They showed that the inhibitory effects of Sema3A on osteoclast differentiation were abolished when NRP1 expression was knocked down by short shRNA. Furthermore, they observed that Sema3A could not inhibit osteoclast differentiation if Nrp1 gene lacking the Sema-binding site. Subsequently, they demonstrated that Sema3A inhibit osteoclastogenesis by binding to Nrp1. Sema3A induced inhibition on osteoclast differentiation may be mediated by the regulation of DAP12-induced ITAM signaling. Nrp1 can compete with TREM2 for PlxnA1 and acts as a suppressor of PlxnA1-TREM2-DAP12 induced signal.
Lysophosphatidic acid (LPA)
LPA is a potent bioactive phospholipid(Hosogaya et al., 2008). LPA is produced by several cells type, including activated platelets and osteoblasts (Sims et al., 2013). Osteoblast-derived LPA may regulate the activity of tumor cells in the skeletal microenviroment and osteoclasts (Panupinthu et al., 2008). LPA receptor, LPA1, LPA2, LPA4 and LPA5 is expressed at high or low level in osteoclasts (Del Fattore et al., 2008). McMichael et al. showed that LPA enhanced osteoclast progenitor cell fusion (McMichael et al., 2010) and LPA is required for osteoclastogenesis (David et al., 2010). LPA could regulate calcium signaling and induce nuclear accumulation of NFATc1 in osteoclast (Lapierre et al., 2010) which is important signaling pathways for osteoclast formation (Boyle et al., 2003). In addition, Lapierre et al. (2010) also showed that LPA could suppress osteoclast apoptosis and induce morphology change of mature osteoclasts. Further study showed that treatment with LPA resulted in increase in osteoclast size accompanied by increased number of nuclei (McMichael et al., 2010).
Osteoclast apoptosis induced by osteoblasts
Estrogen can induce osteoclast apoptosis (Kousteni et al., 2002). Nakamura et al. (Nakamura et al., 2007) showed that the estrogen-induced apoptosis of osteoclast are related with the induction of Factor associated suicide ligand (FasL) in these same cells. However, Krum and colleagues (Krum et al., 2008) found that that estrogen affects osteoclast survival through the upregulation of FasL in osteoblasts leading to the apoptosis of pre-osteoclasts. Furthermore, they found that tamoxifen and raloxifene also induce pre-osteoclasts by the same osteoblast-dependent mechanism. Garcia et al. (Garcia et al., 2013) also showed that 17b-estradiol (E2)-induced apoptosis of osteoclasts is mediated by cleavage and solubilization of osteoblast-expressed FasL. E2 could up-regulate matrix metalloproteinase-3 (MMP3) expression, and then MMP3 cleaves the full-length FasL into soluble FasL. In the presence of MMP3 specific inhibitor, extensive cleavage and soluble FasL concentrations were reduced. Recently, Wang et al. (2015) also showed that osteoblasts induce osteoclast apoptosis by FAS ligand (FASL)/FAS signaling. Conditional knockout FasL in osteoblast caused increase of osteoclast number and activity. Moreover, they indicated that the FasL expression were down-regulated in osteoblasts from ovariectomized osteoporotic mice compared with the sham control group. Then, they found that FasL knockout osteoblast from sham control showed weak capacity to induce osteoclast apoptosis and overexpression of FasL in OVX-derived osteoblast showed increase capacity to induce osteoclast apoptosis. Osteoblast-induced osteoclast apoptosis by FasL has an important role OVX osteoporosis. In addition, using osteoblast progenitor/osteoblast-specific FASL-deficient (FasL cKO) mice, they found that blockage of RANKL showed limited role to reduce the osteoclast activity in FasL cKO mice. Wang et al. (Wang et al., 2015) also showed that osteoclast progenitors have lower FAS expression compared with mature osteoclast. Osteoclast progenitors may be low sensitivity to osteoblast-induced apoptosis via FASL/FAS pathway.
The role of osteoclast in bone formation
Atp6v0d2
Atp6v0d2 is the abbreviation of d2 isoform of vacuolar (H+) ATPase (v-ATPase) V0 domain. It is highly expressed in osteoclasts (Rho et al., 2002). However, little is known about the functional importance of Atp6v0d2 (Rho et al., 2002). Lee et al. (2006) showed that marked increase in bone mass and decrease in bone marrow cavity space were observed in Atp6v0d2 −/− mice. Meanwhile, osteoclast surface extent and the number of multinucleated TRAP positive cells were reduced. However, the number of mononuclear TRAP+ cells was slightly increased. This data indicated that Atp6v0d2 −/− may influence osteoclast maturation, not differentiation. In addition, they found that Atp6v0d2 was related with the cell-cell fusion. The formation of mature giant osteoclasts was decreased in the absence of Atp6v0d2. Furthermore, a marked increase in osteoblasts and bone formation were also observed in Atp6v0d2 −/− mice. However, osteoblast differentiation and differentiation related gene expression was not changed. The authors speculated that increased osteoblast activity in Atp6v0d2 −/− mice may be due to osteoblast extrinsic factors produced by mutant osteoclasts or their precursors. Atp6v0d2 may have great potential as a target for the treatment of osteoporosis due to its role on osteoclasts and bone formation.
Complement Component 3a
Matsuoka et al. (2014) established a co-culture system of osteoblast and mature osteoclasts to the influence of osteoclasts on osteoblastogenesis. They purified a peptide whose sequence matched complement component 3(C3) from the conditional medium (CM) of mature osteoclasts. C3 progressively increased during osteoclast differentiation. They found that C3 bioactive fragment 3a could stimulate osteoblast differentiation, and that the C3a receptor was expressed in stromal cell lines, as well as in primary calvarial osteoblasts. When the C3 gene expression was knocked down in osteoclasts, the CM of osteoclast failed to stimulate the osteoblast differentiation. However, the alkaline phosphatase activity (ALP) activity of osteoblast was inhibited by the CM in the presence of C3aR antagonist, and C3aR antagonist alone did not affect ALP activity. These data all suggested that the C3a in the CM is responsible for the enhanced ALP activity. Furthermore, they showed that the ALP activity was increased if C3aR agonist was added to osteoblast culture. Subsequently, they found that C3a expression in bone is increased in OVX-treated mice with excessive osteoclast activation. C3aR antagonist inhibited the stimulation of bone formation in OVX mice. Bone loss and damage of trabecular architecture were also exacerbated. These results suggested that C3a may mediate the coupling of bone resorption to formation in a high turnover model.
Semaphorin 4D
Semaphorin 4D (Sema4D) is an axon guidance molecule. Negishi-Koga et al. (2011) showed that osteoclasts highly express Sema4D and Sema4D potently inhibits bone formation through its receptor plexin-B1 expressed by osteoblasts. Bone mass, trabecular thickness and bone strength were obviously increased in Sema4D −/− mice compared with wild-type mice. In addition, the osteoblast surface and bone formation rates were also markedly increased. However, the osteoclastic bone resorption was normal. These data suggested that the high bone mass in Sema4D −/− mice were due to the increase in osteoblastic bone formation. Further study showed that addition of Sema4D inhibited bone nodule formation, ALP activity and osteoblastic gene expression. Plexin-B1 is highly expressed during osteoblast differentiation. Plexin-B1 forms a receptor complex with erythroblastic leukemia viral oncogene homolog 2(ErbB2) in osteoblasts. Binding of Smea4d to Plexin-B1 induced the phosphorylation of ErB2 which play a role of kinase in osteoblasts. However, addition of Sema4D still slightly suppressed the bone formation in Plxnb1 −/− cells, which suggested that other receptors may partly involve in the effects of Sema4D on osteoblasts. Subsequently, they found that binding of Sema4D to Plexin-b1-ErbB2 receptor complex could induce RhoA activation, which mediates IRS-1 phosphorylation. The effects of Sema4D on osteoblast may be in part through an attenuation of IGF-1 signaling. In addition, Sema4D may induce osteoblast motility through RhoA activation and affect the localization of osteoblasts to a proper site. In vivo studies also demonstrated that Sema4D-specific antibody can protect against bone loss in OVX mice by promoting bone formation. Recent studies also showed that siRNA-Sema4D significantly increase bone volume, volume bone mineral density and number of osteoblasts in a rat model (Zhang et al., 2015, 2016).
Sclerostin
Sclerostin is a protein that in humans is encoded by the SOST gene (Balemans et al., 2001). Sclerostin is expressed in human long bones and cartilages and is expressed in the osteocyte and has anti-anabolic effects on bone formation (Winkler et al. 2003). Kusu and colleagues (2003) found that Sclerostin was also expressed in mouse osteoclasts. They further showed that Sclerostin bound to the BMP6 and BMP7 with high affinity and inhibited their activity in mouse MC3T3-E1 cells. However, Winkler et al. (2003) showed that no expression of SOST RNA was observed in human osteoclasts or human osteoclast progenitor cells. Ota et al. (2013) indicated that Sclerostin was expressed in osteoclasts in aged mice. The concentrated conditional media (CCM) from osteoclasts from 6-week or 12-month-old mice enhanced osteogenenesis. However, the CCM from 18- to 24-month old mice inhibited mineralization. Furthermore, they observed that Sclerostin mRNA and protein levels form the osteoclasts in 24-month old mice were significantly high than that in 6-week old mice. If anti-sclerostin antibody was added into the CCM, the suppression of mineralization by CCM was reversed. Osteoclasts from aged mice may have some different phenotype compared with young mice.
MicroRNA/exsomes
It has been shown that MicroRNAs also regulate osteogenic activity and osteoblastic bone formation (Lian et al., 2012). MiR-214-3p has been proved to play a crucial role in skeletal disorders, including suppressing osteogenic differentiation of myoblast cells (Shi et al. 2013) and bone formation (Wang et al., 2013). A recent study indicated that exosomal miR-214-3p produced by osteoclast may affect the osteoblastic bone formation (Li et al., 2016). Abundant miR-214-3P and exosomal miR-214-3p were detected in mature mouse osteoclasts but not in mature osteoblasts. They found that osteoclastic miR-214-3P associated with the reduced bone formation in elderly women with fractures and in OVX mice. Using an osteoclastic-specific miR-214-3P knock-in mice model, they also found that the bone mass was decreased and trabecular architecture was poorly organized compared with the wild-type mice. Histological analysis revealed that bone formation markers, such as mineral apposition rate and Ob.S/BS, in miR-214-3P knock-in (OC-miR-214-3p) mice were all significantly lower than that in wild-type mice. When osteoblasts were co-cultured with OC-miR-214-3p osteoclasts, the expression of osteoblast activity related genes were all significantly down-regulated compared with those in osteoblasts co-cultured with wild type osteoclasts. In addition, they showed that the osteoblast activity was inhibited if osteoclastic-specific miR-214-3P was transferred to. Furthermore, they demonstrated that exosomal miR-214-3p could transfer from osteoclasts to osteoblasts. They also showed that osteoclast-derived exosomal miR-214-3p could be taken up by osteoblasts in vivo. The mRNA levels of bone formation genes, such as Alp, Opn and Bsp were significantly lower in mice injected with OC-miR-214-3p exosomes. Bone formation was also markedly suppressed. Finally, they showed that the inhibition of osteoclastic-specific miR-214-3P enhanced bone formation in aging OVX mice. A recent study also showed that osteoclasts produced miRNA-enriched exosomes, and the exosomes transferred miR-214 into the osteoblasts to inhibit their activity (Sun et al., 2016). Osteoblast activity was inhibited in osteoclast-specific miR-214 transgenic mice. High miR-214 levels were observed in osteoporotic patients and OVX mice. This study also indicated that exsomes derived from osteoclasts recognized osteoblasts via ephrinA2/EphA2. These two studies suggest that osteoclasts may affect osteoblasts via exosomes.
In addition, Ephrin-EphB4 bidirectional regulation (Zhao et al., 2006), collagen triple helix repeat containing 1 (Cthrc 1) (Takeshita et al., 2013) and sphingosin-1-phosphate (S1P)(Ryu et al., 2006; Pederson et al., 2008) also play important role in the progress of osteoclast-osteoblast cross-talk. For Ephrin-EphB4, the forward signaling mediated by EPH (OC-OB) stimulates osteoblast differentiation and induce bone formation. Takeshita et al. (Takeshita et al., 2013) found that Cthrc1, a protein secreted by mature bone-resorbing osteoclasts, could stimulate osteogenesis. Osteoclast-specific deletion of Cthrc1 caused osteopenia due to reduced bone formation. Cthrc1 knock-out mice showed low bone mass, low bone formation rate and microstructural abnormalities. In addition, mature osteoclasts produce and secrete many factors, such as WNT10B, BMP6, the lipid S1P, Hepatocyte growth factor (Grano et al., 1996), Cathpsin K (Kiviranta et al., 2001; Lotinun et al., 2013), and Tartrate resistant acid phosphatase (TRAcP)(Teti, 2013) to stimulate osteoblast recruitment and survival. TGF-b and IGF-1 released from the bone matrix during osteoclastic bone resorption also play important role on osteoblast activity (Tang et al., 2009; Xian et al., 2012), such as RANKL expression and cell migration.
In summary, bone homeostasis is crucial for the normal function and structure of bone. The homeostasis depends on the cross-talk between osteoblasts and osteoclasts. Many cytokines, hormones and signaling pathway are involved in those progresses. OPG/RANKL/RANK pathway is one of the important advances in bone cell biology, which evidence that osteoblast can regulate osteoclast. Subsequent studies further showed that osteoclast also influence the activity of osteoblast or bone formation. The schematic presentation of osteoblast-osteoclast interaction was showed in Fig 1. Understanding the cross-talk between osteoblast and osteoclast is important for the discovery and development of novel anti-osteoporosis drugs. We believe that additional studies will reveal more and more novel molecules which mediate the communications between those two cells. Meanwhile, further studies are still needed to clarify the known signal transduction pathways.
Figure 1.
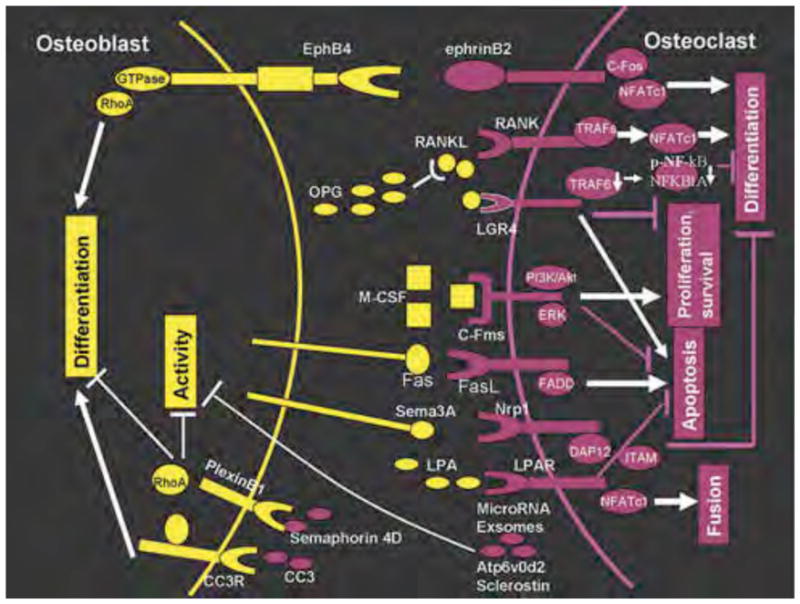
The schematic presentation of osteoblast–osteoclast interactions. Osteoblasts can enhanced the proliferation, differentiation, survival, apoptosis or fusion by macrophage colonystimulating factor (M-CSF)/C-Fms, receptor activator of nuclear factor-κB ligand (RANKL), lysophosphatidic acid/lysophosphatidic acid receptor (LPA/LPAR) and Fas/FasL (FADD, Fas-associated death domain). Osteoblasts also inhibit (⊥) the differentiation, proliferation or survival of osteoclast by osteoprotegerin (OPG), RANKL/LGR4 and Sema3A/Nrp1. Osteoclasts can promote osteoblasts differentiation by Complement Component 3a (CC3)/CC3 receptor (CC3R) and ephrinB2. D2 isoform of vacuolar (H+) ATPase (v-ATPase) V0 domain (Atp6v0d2), Semaphorin 4D, Sclerostin, MicroRNAs (miR-214 and miR-214-3p) or exsomes from osteoclast can inhibit (⊥) the differentiation or activity of osteoblast. There are also bidirectional interactions (EphB4-ephrinB2) between osteoblasts and osteoclasts.
Acknowledgments
This study was funded by Natural Science Foundation of Jiangsu Province (no. BK20161609), and NIH P30 AR069655.
References
- 1.The American Society for Bone and Mineral Research President’s Committee on Nomenclature. Proposed standard nomenclature for new tumor necrosis factor family members involved in the regulation of bone resorption. J Bone Miner Res. 2000;15:2293–6. doi: 10.1359/jbmr.2000.15.12.2293. [DOI] [PubMed] [Google Scholar]
- 2.Anderson DM, Maraskovsky E, Billingsley WL, Dougall WC, Tometsko ME, Roux ER, Teepe MC, DuBose RF, Cosman D, Galibert L. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature. 1997;390:175–9. doi: 10.1038/36593. [DOI] [PubMed] [Google Scholar]
- 3.Balemans W, Ebeling M, Patel N, Van Hul E, Olson P, Dioszegi M, Lacza C, Wuyts W, Van Den Ende J, Willems P, et al. Increased bone density in sclerosteosis is due to the deficiency of a novel secreted protein (SOST) Hum Mol Genet. 2001;10:537–43. doi: 10.1093/hmg/10.5.537. [DOI] [PubMed] [Google Scholar]
- 4.Boyce BF, Xing L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch Biochem Biophys. 2008;473(2):139–46. doi: 10.1016/j.abb.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–42. doi: 10.1038/nature01658. [DOI] [PubMed] [Google Scholar]
- 6.Bucay N, Sarosi I, Dunstan CR, Morony S, Tarpley J, Capparelli C, Scully S, Tan HL, Xu W, Lacey DL, et al. osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev. 1998;12:1260–8. doi: 10.1101/gad.12.9.1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.David M, Wannecq E, Descotes F, Jansen S, Deux B, Ribeiro J, Serre CM, Grès S, Bendriss-Vermare N, Bollen M, Saez S, Aoki J, Saulnier-Blache JS, Clézardin P, Peyruchaud O. Cancer cell expression of autotaxin controls bone metastasis formation in mouse through lysophosphatidic acid-dependent activation of osteoclasts. PLoS One. 2010;5:e9741. doi: 10.1371/journal.pone.0009741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Del Fattore A, Teti A, Rucci N. Osteoclast receptors and signaling. Arch Biochem Biophys. 2008;473:147–160. doi: 10.1016/j.abb.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 9.Dougall WC, Glaccum M, Charrier K, Rohrbach K, Brasel K, De Smedt T, Daro E, Smith J, Tometsko ME, Maliszewski CR, et al. RANK is essential for osteoclast and lymph node development. Genes Dev. 1999;13:2412–24. doi: 10.1101/gad.13.18.2412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Everts V, Delaisse JM, Korper W, Jansen DC, Tigchelaar-Gutter W, Saftig P, Beertsen W. The bone lining cell: its role in cleaning Howship’s lacunae and initiating bone formation. J Bone Miner Res. 2002;17:77–90. doi: 10.1359/jbmr.2002.17.1.77. [DOI] [PubMed] [Google Scholar]
- 11.Fata JE, Kong YY, Li J, Sasaki T, Irie-Sasaki J, Moorehead RA, Elliott R, Scully S, Voura EB, Lacey DL, et al. The osteoclast differentiation factor osteoprotegerin-ligand is essential for mammary gland development. Cell. 2000;103:41–50. doi: 10.1016/s0092-8674(00)00103-3. [DOI] [PubMed] [Google Scholar]
- 12.Garcia AJ, Tom C, Guemes M, Polanco G, Mayorga ME, Wend K, Miranda-Carboni GA, Krum SA. ERalpha signaling regulates MMP3 expression to induce FasL cleavage and osteoclast apoptosis. J Bone Miner Res. 2013;28:283–90. doi: 10.1002/jbmr.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grano M, Galimi F, Zambonin G, Colucci S, Cottone E, Zallone AZ, Comoglio PM. Hepatocyte growth factor is a coupling factor for osteoclasts and osteoblasts in vitro. Proc Natl Acad Sci U S A. 1996;93:7644–8. doi: 10.1073/pnas.93.15.7644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Graves DT, Jiang Y, Valente AJ. The expression of monocyte chemoattractant protein-1 and other chemokines by osteoblasts. Front Biosci. 1999;4:D571–80. doi: 10.2741/graves. [DOI] [PubMed] [Google Scholar]
- 15.Hayashi M, Nakashima T, Taniguchi M, Kodama T, Kumanogoh A, Takayanagi H. Osteoprotection by semaphorin 3A. Nature. 2012;485:69–74. doi: 10.1038/nature11000. [DOI] [PubMed] [Google Scholar]
- 16.Hosogaya S, Yatomi Y, Nakamura K, Ohkawa R, Okubo S, Yokota H, Ohta M, Yamazaki H, Koike T, Ozaki Y. Measurement of plasma lysophosphatidic acid concentration in healthy subjects: strong correlation withlysophospholipase D activity. Ann Clin Biochem. 2008;45:364–8. doi: 10.1258/acb.2008.007242. [DOI] [PubMed] [Google Scholar]
- 17.Ikeda T, Kasai M, Utsuyama M, Hirokawa K. Determination of three isoforms of the receptor activator of nuclear factor-kappaB ligand and their differential expression in bone and thymus. Endocrinology. 2001;142:1419–26. doi: 10.1210/endo.142.4.8070. [DOI] [PubMed] [Google Scholar]
- 18.Kadono YATO. TRAF6 expression promoted by RANKL/RANK signaling plays an important role in osteoclast differentiation. JBMR. 2001;16:s176. [Google Scholar]
- 19.Kearns AE, Khosla S, Kostenuik PJ. Receptor activator of nuclear factor kappaB ligand and osteoprotegerin regulation of bone remodeling in health and disease. Endocr Rev. 2008;29:155–92. doi: 10.1210/er.2007-0014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim HH, Lee DE, Shin JN, Lee YS, Jeon YM, Chung CH, Ni J, Kwon BS, Lee ZH. Receptor activator of NF-kappaB recruits multiple TRAF family adaptors and activates c-Jun N-terminal kinase. FEBS Lett. 1999;443:297–302. doi: 10.1016/s0014-5793(98)01731-1. [DOI] [PubMed] [Google Scholar]
- 21.Kim JH, Kim N. Signaling Pathways in Osteoclast Differentiation. Chonnam Med J. 2016;52:12–7. doi: 10.4068/cmj.2016.52.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim NS, Kim HJ, Koo BK, Kwon MC, Kim YW, Cho Y, Yokota Y, Penninger JM, Kong YY. Receptor activator of NF-kappaB ligand regulates the proliferation of mammary epithelial cells via Id2. Mol Cell Biol. 2006;26:1002–13. doi: 10.1128/MCB.26.3.1002-1013.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kiviranta R, Morko J, Uusitalo H, Aro HT, Vuorio E, Rantakokko J. Accelerated turnover of metaphyseal trabecular bone in mice overexpressing cathepsin K. J Bone Miner Res. 2001;16:1444–52. doi: 10.1359/jbmr.2001.16.8.1444. [DOI] [PubMed] [Google Scholar]
- 24.Kousteni S, Chen JR, Bellido T, Han L, Ali AA, O’Brien CA, Plotkin L, Fu Q, Mancino AT, Wen Y, et al. Reversal of bone loss in mice by nongenotropic signaling of sex steroids. Science. 2002;298:843–6. doi: 10.1126/science.1074935. [DOI] [PubMed] [Google Scholar]
- 25.Krum SA, Miranda-Carboni GA, Hauschka PV, Carroll JS, Lane TF, Freedman LP, Brown M. Estrogen protects bone by inducing Fas ligand in osteoblasts to regulate osteoclast survival. EMBO J. 2008;27:535–45. doi: 10.1038/sj.emboj.7601984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kusu N, Laurikkala J, Imanishi M, Usui H, Konishi M, Miyake A, Thesleff I, Itoh N. Sclerostin is a novel secreted osteoclast-derived bone morphogenetic protein antagonist with unique ligand specificity. J Biol Chem. 2003;278:24113–7. doi: 10.1074/jbc.M301716200. [DOI] [PubMed] [Google Scholar]
- 27.Lacey DL, Erdmann JM, Shima M, Kling S, Matayoshi A, Ohara J, Perkins SL. Interleukin 4 enhances osteoblast macrophage colony-stimulating factor, but not interleukin 6, production. Calcif Tissue Int. 1994;55:21–8. doi: 10.1007/BF00310164. [DOI] [PubMed] [Google Scholar]
- 28.Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T, Elliott R, Colombero A, Elliott G, Scully S, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–76. doi: 10.1016/s0092-8674(00)81569-x. [DOI] [PubMed] [Google Scholar]
- 29.Lapierre DM, Tanabe N, Pereverzev A, Spencer M, Shugg RP, Dixon SJ, Sims SM. Lysophosphatidic acid signals through multiple receptors in osteoclasts to elevate cytosolic calcium concentration, evoke retraction, and promote cell survival. J Biol Chem. 2010;285:25792–25801. doi: 10.1074/jbc.M110.109322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lee SH, Rho J, Jeong D, Sul JY, Kim T, Kim N, Kang JS, Miyamoto T, Suda T, Lee SK, et al. v-ATPase V0 subunit d2-deficient mice exhibit impaired osteoclast fusion and increased bone formation. Nat Med. 2006;12:1403–9. doi: 10.1038/nm1514. [DOI] [PubMed] [Google Scholar]
- 31.Li D, Liu J, Guo B, Liang C, Dang L, Lu C, He X, Cheung HY, Xu L, Lu C, et al. Osteoclast-derived exosomal miR-214-3p inhibits osteoblastic bone formation. Nat Commun. 2016;7:10872. doi: 10.1038/ncomms10872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li X, Qin L, Bergenstock M, Bevelock LM, Novack DV, Partridge NC. Parathyroid hormone stimulates osteoblastic expression of MCP-1 to recruit and increase the fusion of pre/osteoclasts. J Biol Chem. 2007;282:33098–106. doi: 10.1074/jbc.M611781200. [DOI] [PubMed] [Google Scholar]
- 33.Li Y, Toraldo G, Li A, Yang X, Zhang H, Qian WP, Weitzmann MN. B cells and T cells are critical for the preservation of bone homeostasis and attainment of peak bone mass in vivo. Blood. 2007;109:3839–48. doi: 10.1182/blood-2006-07-037994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lian JB, Stein GS, van Wijnen AJ, Stein JL, Hassan MQ, Gaur T, Zhang Y. MicroRNA control of bone formation and homeostasis. Nat Rev Endocrinol. 2012;8:212–27. doi: 10.1038/nrendo.2011.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lotinun S, Kiviranta R, Matsubara T, Alzate JA, Neff L, Luth A, Koskivirta I, Kleuser B, Vacher J, Vuorio E, et al. Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J Clin Invest. 2013;123:666–81. doi: 10.1172/JCI64840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Luo J, Yang Z, Ma Y, Yue Z, Lin H, Qu G, Huang J, Dai W, Li C, Zheng C, Xu L, Chen H, Wang J, Li D, Siwko S, Penninger JM, Ning G, Xiao J, Liu M. LGR4 is a receptor for RANKL and negatively regulates osteoclast differentiation and bone resorption. Nat Med. 2016;22:539–46. doi: 10.1038/nm.4076. [DOI] [PubMed] [Google Scholar]
- 37.Matsuo K, Irie N. Osteoclast-osteoblast communication. Arch Biochem Biophys. 2008;473:201–9. doi: 10.1016/j.abb.2008.03.027. [DOI] [PubMed] [Google Scholar]
- 38.Matsuoka K, Park KA, Ito M, Ikeda K, Takeshita S. Osteoclast-derived complement component 3a stimulates osteoblast differentiation. J Bone Miner Res. 2014;29:1522–30. doi: 10.1002/jbmr.2187. [DOI] [PubMed] [Google Scholar]
- 39.McMichael BK, Meyer SM, Lee BS. c-Src-mediated phosphorylation of thyroid hormone receptor-interacting protein 6 (TRIP6) promotes osteoclast sealing zone formation. J Biol Chem. 2010;285:26641–26651. doi: 10.1074/jbc.M110.119909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mundy GR, Elefteriou F. Boning up on ephrin signaling. Cell. 2006;126:441–3. doi: 10.1016/j.cell.2006.07.015. [DOI] [PubMed] [Google Scholar]
- 41.Nakamura T, Imai Y, Matsumoto T, Sato S, Takeuchi K, Igarashi K, Harada Y, Azuma Y, Krust A, Yamamoto Y, et al. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell. 2007;130:811–23. doi: 10.1016/j.cell.2007.07.025. [DOI] [PubMed] [Google Scholar]
- 42.Negishi-Koga T, Shinohara M, Komatsu N, Bito H, Kodama T, Friedel RH, Takayanagi H. Suppression of bone formation by osteoclastic expression of semaphorin 4D. Nat Med. 2011;17:1473–80. doi: 10.1038/nm.2489. [DOI] [PubMed] [Google Scholar]
- 43.Ota K, Quint P, Ruan M, Pederson L, Westendorf JJ, Khosla S, Oursler MJ. Sclerostin is expressed in osteoclasts from aged mice and reduces osteoclast-mediated stimulation of mineralization. J Cell Biochem. 2013;114:1901–7. doi: 10.1002/jcb.24537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Panupinthu N, Rogers JT, Zhao L, Solano-Flores LP, Possmayer F, Sims SM, Dixon SJ. P2X7 receptors on osteoblasts couple to production of lysophosphatidic acid: a signaling axis promotingosteogenesis. J Cell Biol. 2008;181:859–71. doi: 10.1083/jcb.200708037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pederson L, Ruan M, Westendorf JJ, Khosla S, Oursler MJ. Regulation of bone formation by osteoclasts involves Wnt/BMP signaling and the chemokine sphingosine-1-phosphate. Proc Natl Acad Sci U S A. 2008;105:20764–9. doi: 10.1073/pnas.0805133106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rho J, Altmann CR, Socci ND, Merkov L, Kim N, So H, Lee O, Takami M, Brivanlou AH, Choi Y. Gene expression profiling of osteoclast differentiation by combined suppression subtractive hybridization (SSH) and cDNA microarray analysis. DNA Cell Biol. 2002;21:541–9. doi: 10.1089/104454902320308915. [DOI] [PubMed] [Google Scholar]
- 47.Rodan GA, Martin TJ. Role of osteoblasts in hormonal control of bone resorption--a hypothesis. Calcif Tissue Int. 1981;33:349–51. doi: 10.1007/BF02409454. [DOI] [PubMed] [Google Scholar]
- 48.Ryu J, Kim HJ, Chang EJ, Huang H, Banno Y, Kim HH. Sphingosine 1-phosphate as a regulator of osteoclast differentiation and osteoclast-osteoblast coupling. EMBO J. 2006;25:5840–51. doi: 10.1038/sj.emboj.7601430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sambandam Y, Blanchard JJ, Daughtridge G, Kolb RJ, Shanmugarajan S, Pandruvada SN, Bateman TA, Reddy SV. Microarray profile of gene expression during osteoclast differentiation in modelled microgravity. J Cell Biochem. 2010;111:1179–87. doi: 10.1002/jcb.22840. [DOI] [PubMed] [Google Scholar]
- 50.Shi K, Lu J, Zhao Y, Wang L, Li J, Qi B, Li H, Ma C. MicroRNA-214 suppresses osteogenic differentiation of C2C12 myoblast cells by targeting Osterix. Bone. 2013;55:487–94. doi: 10.1016/j.bone.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 51.Simonet WS, Lacey DL, Dunstan CR, Kelley M, Chang MS, Luthy R, Nguyen HQ, Wooden S, Bennett L, Boone T, et al. Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell. 1997;89:309–19. doi: 10.1016/s0092-8674(00)80209-3. [DOI] [PubMed] [Google Scholar]
- 52.Sims SM, Panupinthu N, Lapierre DM, Pereverzev A, Dixon SJ. Lysophosphatidic acid: a potential mediator of osteoblast-osteoclast signaling in bone. Biochim Biophys Acta. 2013;1831:109–16. doi: 10.1016/j.bbalip.2012.08.001. [DOI] [PubMed] [Google Scholar]
- 53.Sun w, Zhao C, Li Y, Wang L, Nie G, Peng J, Wang A, Zhang P, Tian W, Li Q, Song J, Wang C, Xu X, Tian Y, Zhao D, Xu Z, Zhong G, Han B, Ling S, Chang Y, Li Y. Osteoclast-derived microRNA-containing exosomes selectively inhibit osteoblast activity. Cell Discovery. 2016 doi: 10.1038/celldisc.2016.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Takeshita S, Fumoto T, Matsuoka K, Park KA, Aburatani H, Kato S, Ito M, Ikeda K. Osteoclast-secreted CTHRC1 in the coupling of bone resorption to formation. J Clin Invest. 2013;123:3914–24. doi: 10.1172/JCI69493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tamma R, Zallone A. Osteoblast and osteoclast crosstalks: from OAF to Ephrin. Inflamm Allergy Drug Targets. 2012;11:196–200. doi: 10.2174/187152812800392670. [DOI] [PubMed] [Google Scholar]
- 56.Tang Y, Wu X, Lei W, Pang L, Wan C, Shi Z, Zhao L, Nagy TR, Peng X, Hu J, et al. TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat Med. 2009;15:757–65. doi: 10.1038/nm.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Teti A. Mechanisms of osteoclast-dependent bone formation. Bonekey Rep. 2013;2:449. doi: 10.1038/bonekey.2013.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Theoleyre S, Wittrant Y, Tat SK, Fortun Y, Redini F, Heymann D. The molecular triad OPG/RANK/RANKL: involvement in the orchestration of pathophysiological bone remodeling. Cytokine Growth Factor Rev. 2004;15:457–75. doi: 10.1016/j.cytogfr.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 59.Wang L, Liu S, Zhao Y, Liu D, Liu Y, Chen C, Karray S, Shi S, Jin Y. Osteoblast-induced osteoclast apoptosis by fas ligand/FAS pathway is required for maintenance of bone mass. Cell Death Differ. 2015;22:1654–64. doi: 10.1038/cdd.2015.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang X, Guo B, Li Q, Peng J, Yang Z, Wang A, Li D, Hou Z, Lv K, Kan G, et al. miR-214 targets ATF4 to inhibit bone formation. Nat Med. 2013;19:93–100. doi: 10.1038/nm.3026. [DOI] [PubMed] [Google Scholar]
- 61.Wiktor-Jedrzejczak W, Bartocci A, Ferrante AJ, Ahmed-Ansari A, Sell KW, Pollard JW, Stanley ER. Total absence of colony-stimulating factor 1 in the macrophage-deficient osteopetrotic (op/op) mouse. Proc Natl Acad Sci U S A. 1990;87:4828–32. doi: 10.1073/pnas.87.12.4828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Winkler DG, Sutherland MK, Geoghegan JC, Yu C, Hayes T, Skonier JE, Shpektor D, Jonas M, Kovacevich BR, Staehling-Hampton K, et al. Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. EMBO J. 2003;22:6267–76. doi: 10.1093/emboj/cdg599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wong BR, Rho J, Arron J, Robinson E, Orlinick J, Chao M, Kalachikov S, Cayani E, Bartlett FR, Frankel WN, et al. TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J Biol Chem. 1997;272:25190–4. doi: 10.1074/jbc.272.40.25190. [DOI] [PubMed] [Google Scholar]
- 64.Xian L, Wu X, Pang L, Lou M, Rosen CJ, Qiu T, Crane J, Frassica F, Zhang L, Rodriguez JP, et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat Med. 2012;18:1095–101. doi: 10.1038/nm.2793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yasuda H, Shima N, Nakagawa N, Mochizuki SI, Yano K, Fujise N, Sato Y, Goto M, Yamaguchi K, Kuriyama M, et al. Identity of osteoclastogenesis inhibitory factor (OCIF) and osteoprotegerin (OPG): a mechanism by which OPG/OCIF inhibits osteoclastogenesis in vitro. Endocrinology. 1998;139:1329–37. doi: 10.1210/endo.139.3.5837. [DOI] [PubMed] [Google Scholar]
- 66.Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci U S A. 1998;95:3597–602. doi: 10.1073/pnas.95.7.3597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zaidi M. Skeletal remodeling in health and disease. Nat Med. 2007;13:791–801. doi: 10.1038/nm1593. [DOI] [PubMed] [Google Scholar]
- 68.Zhang Y, Wei L, Miron RJ, Shi B, Bian Z. Anabolic bone formation via a site-specific bone-targeting delivery system by interfering with semaphorin 4D expression. J Bone Miner Res. 2015;30:286–96. doi: 10.1002/jbmr.2322. [DOI] [PubMed] [Google Scholar]
- 69.Zhang Y, Wei L, Miron RJ, Shi B, Bian Z. Bone scaffolds loaded with siRNA-Semaphorin4d for the treatment of osteoporosis related bone defects. Sci Rep. 2016;6:26925. doi: 10.1038/srep26925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhao C, Irie N, Takada Y, Shimoda K, Miyamoto T, Nishiwaki T, Suda T, Matsuo K. Bidirectional ephrinB2-EphB4 signaling controls bone homeostasis. Cell Metab. 2006;4:111–21. doi: 10.1016/j.cmet.2006.05.012. [DOI] [PubMed] [Google Scholar]


