Abstract
Animal models of pathogenic infection are needed to evaluate candidate compounds for the development of anti-infectious drugs. Dermatophytes are pathogenic fungi that cause several infectious diseases. We established a silkworm dermatophyte infection model to evaluate anti-fungal drugs. Injection of conidia of the dermatophyte Arthroderma vanbreuseghemii into silkworms was lethal. A. vanbreuseghemii conidia germinated in liquid culture were more potent against silkworms than non-germinated conidia. Germinated conidia of other dermatophytes, Arthroderma benhamiae, Trichophyton rubrum, and Microsporum canis, also killed silkworms. Injection of heat-treated germinated A. vanbreuseghemii conidia did not kill silkworms, suggesting that only viable fungi are virulent. Injecting terbinafine or itraconazole, oral drugs used clinically to treat dermatophytosis, into the silkworm midgut had therapeutic effects against infection with germinated A. vanbreuseghemii conidia. When silkworms were injected with A. vanbreuseghemii expressing enhanced green fluorescent protein (eGFP), mycelial growth of the fungus was observed in the fat body and midgut. Injection of terbinafine into the silkworm midgut, which corresponds to oral administration in humans, inhibited the growth of A. vanbreuseghemii expressing eGFP in the fat body. These findings suggest that the silkworm infection model with eGFP-expressing dermatophytes is useful for evaluating the therapeutic activity of orally administered anti-fungal agents against dermatophytes.
Introduction
Pathogenic fungi cause several infectious diseases, such as superficial cutaneous fungal infection and serious deep infection1. Superficial cutaneous fungal infection affects a quarter of the human population worldwide, and the prevalence is increasing2. Dermatophytes, pathogenic fungi, can cause serious deep infection in immunocompromised individuals3,4. Anti-fungal drugs against dermatophytosis in humans are administered orally or applied topically. The complete cure rate of dermatophytosis of the toenail by treatment with the topical medicine efinaconazol, however, is lower than 17.8–22.2%5. Furthermore, oral medicines used clinically against fungal diseases interact with many drugs and may cause serious side effects6,7. Therefore, the development of novel, effective anti-fungal drugs against dermatophytosis with fewer side effects is needed.
Most chemicals that exhibit antimicrobial activity in vitro do not have therapeutic effects in animal models because of their in vivo pharmacokinetics and toxicity. Therefore, evaluation of therapeutic effects in animal models is necessary for the development of clinically effective anti-fungal agents. Mammalian animals such as the guinea pig are often used as fungal skin infection models8. The high costs and ethical issues associated with using a large number of mammalian animals to screen for novel therapeutic agents against dermatophytosis, however, are prohibitive.
The silkworm is a useful animal model to evaluate the therapeutic effects of anti-bacterial and anti-viral drugs before testing the compounds in mammalian animals9,10. Silkworms are much less expensive to rear and maintain than mammalian animals, and their experimental use partly avoids the ethical issues. The silkworm size is relatively large and thus sufficient for different compound administration routes, such as intra-hemolymph and intra-midgut injection using a tuberculin syringe11. Intra-midgut injection in silkworms corresponds to oral administration in mammals. Amphotericin B, which is not orally available for human patients, also does not exhibit therapeutic effects in silkworms when administered into the intestine12. The median effective dose (ED50) of antibiotics in silkworms is consistent with that in mammals13. Furthermore, the pharmacokinetic parameters of antibiotics, such as the half-life in blood and protein binding capacities are also consistent between silkworms and mammals14,15. The LD50 value, the amount of reagents required to kill 50% of animals, of chemicals against silkworms also correlates well with that in mammals16,17, and therefore quantitative evaluation of both the toxic and therapeutic effects of candidate compounds can be simultaneously performed silkworms. By using the silkworm infection model as a second screening tool after an initial in vitro screening, we recently discovered a novel antibiotic from soil bacteria, lysocin E, that is therapeutically effective in a mouse model of systemic Staphylococcus aureus infection10.
Silkworm systemic infection models of five fungal species, Candida albicans, C. tropicalis, C. glabrata, Cryptococcus neoformans, and Aspergillus fumigatus, have been established13,18–21. These silkworm infection models can be used to quantitatively evaluate the therapeutic efficacy of anti-fungals. Using a silkworm infection model with the filamentous fungus Aspergillus fumigatus, we discovered ASP2397, a compound that has therapeutic effects in a mouse infection model20. This finding suggests that silkworms are useful for identifying novel anti-fungal drugs. To evaluate anti-fungal drugs against dermatophytes, we aimed to establish a novel silkworm infection model.
For evaluation of the therapeutic efficacy of drugs against dermatophytes, it is important to be able to quantitatively assess dermatophyte growth in animals. Dermatophytes grow as filamentous structures called hyphae, collectively referred to as a mycelium. The mycelial growth of dermatophytes contributes to disease progression22–24. Quantitative evaluation of mycelial growth in animals is difficult, however, because a single hypha may comprise numerous cells25. To overcome this problem, we aimed to establish a system to quantitatively evaluate dermatophyte growth in vivo. Fluorescence imaging is a simple and effective method of quantitatively evaluating cell growth and mobility in vivo 26. Green fluorescent protein (GFP) is a reported protein that is stable in vivo 27. Dermatophytes expressing enhanced GFP (eGFP) were established28. We hypothesized that mycelial growth could be evaluated based on detection of the fluorescence of eGFP-expressing dermatophytes in animals. Here we describe a silkworm infection model with dermatophytes expressing eGFP, and demonstrate that the infection model is useful for evaluating the therapeutic effects of terbinafine, a drug used clinically to treat dermatophyte infection.
Results
Killing of silkworms by injection of dermatophyte conidia
Previous studies of silkworm infection models of C. albicans, C. glabrata, C. tropicalis, C. neoformans, and Aspergillus fumigatus demonstrated that rearing temperatures after fungal injection profoundly affect the results29. The appropriate temperatures for infectious experiments are 27 °C for C. albicans and C. tropicalis; 30 °C for A. fumigatus; and 37 °C for C. glabrata and C. neoformans. Therefore, to establish the dermatophyte infection model, we first performed experiments at different temperatures. At 27 °C and 30 °C, all silkworms injected with the conidia of Arthroderma vanbreuseghemii dermatophytes died within 100 h, whereas all silkworms injected with saline were alive (Fig. 1a,b). On the other hand, at 37 °C, all silkworms injected with saline died, resulting in a small difference in the survival period between injection with saline and injection with dermatophytes (Fig. 1c). Subsequent infectious experiments with dermatophyte conidia were conducted at 30 °C, the temperature at which silkworm death was dependent on dermatophyte injection.
Figure 1.
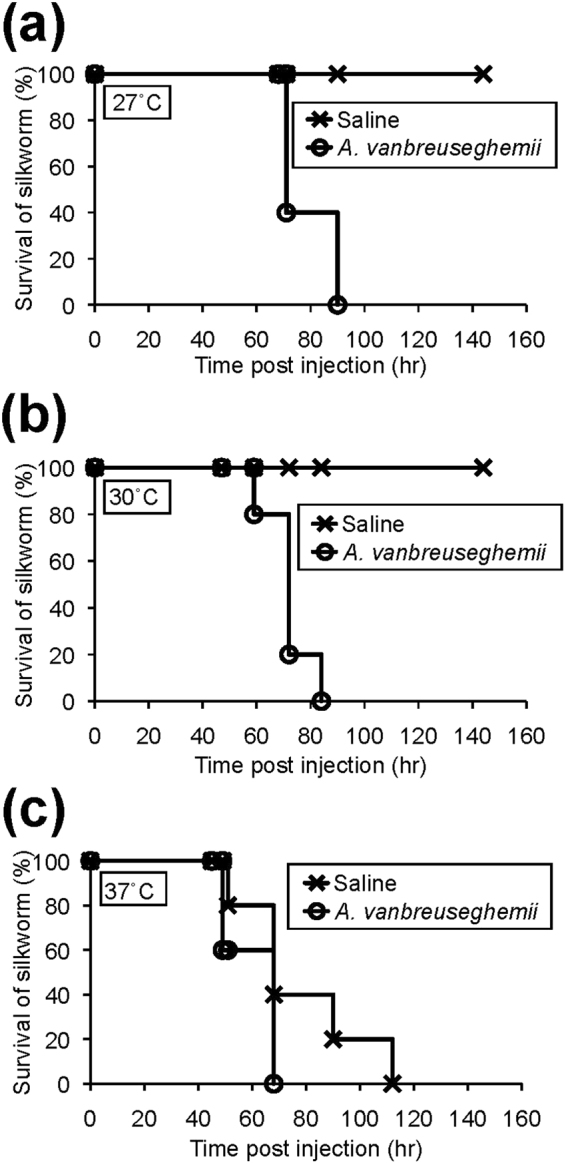
Effect of temperature on the pathogenicity of the Arthroderma vanbreuseghemii TIMM2789 dermatophytes in silkworms. Conidia (4 × 106) of A. vanbreuseghemii TIMM2789 dermatophytes were injected into the silkworm hemolymph. Survival of the animals at 27 °C (a), 30 °C (b), and 37 °C (c) was monitored. n = 5/group.
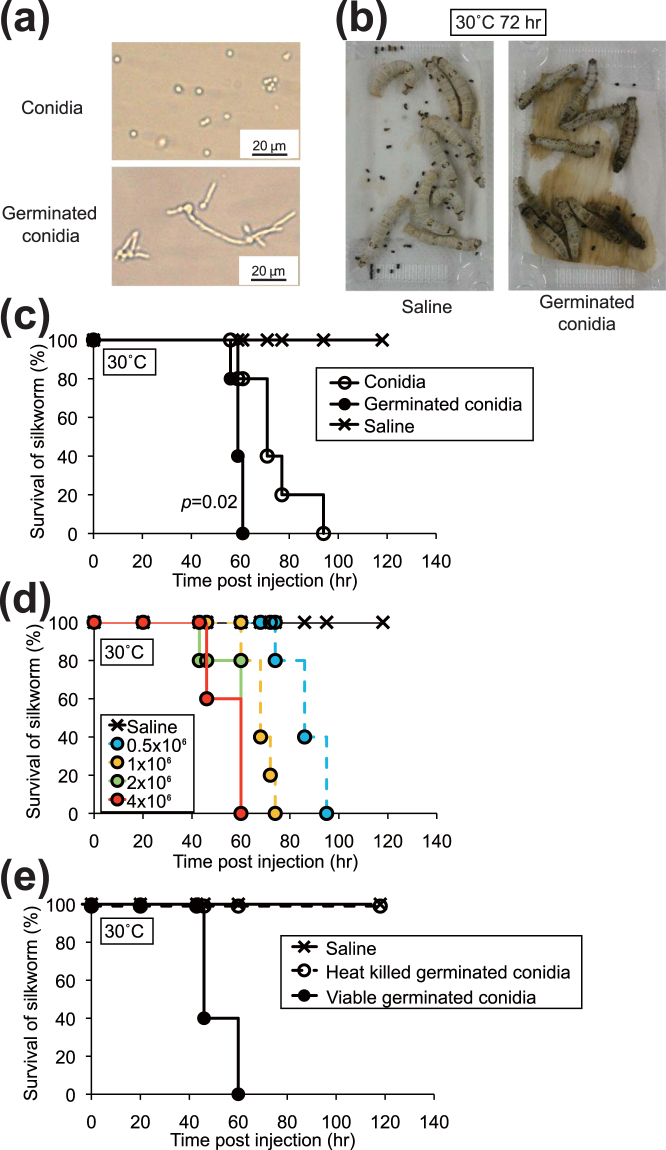
Dermatophyte conidia germinate in rich nutrient media, and form hyphae. Hyphal growth of dermatophytes contributes to the progression of infectious disease22–24. We tested whether conidia germinating in liquid media were more lethal to silkworms than those without germination. Germinated conidia were formed 1 day after incubation in liquid media (Fig. 2a) and the germinated conidia killed silkworms (Fig. 2b). Silkworms were killed more quickly by injection of germinated conidia than by injection of non-germinated conidia (Fig. 2c). The lethality of germinated conidia in silkworms was dose-dependent, ranging from 0 to 4 × 106 cells/larva (Fig. 2d). On the other hand, heat treatment of germinated conidia at 121 °C for 15 min abolished their lethal effects in silkworms (Fig. 2e).
Figure 2.
Infection of silkworms with germinated conidia of A. vanbreuseghemii TIMM2789 dermatophytes. (a) Microscopic observation of conidia of A. vanbreuseghemii TIMM2789 dermatophytes (upper panel) and germinated conidia cultured at 28 °C for 26 h (lower panel). (b) Silkworms reared at 30 °C for 72 h after injection of 4 × 106 of germinated conidia (right). Saline control (left). n = 10/group. (c) Conidia of A. vanbreuseghemii TIMM2789 dermatophytes were cultured at 28 °C for 26 h (germinated conidia). Conidia (4 × 106) or germinated conidia (4 × 106) were injected into the silkworm hemolymph, and the silkworms were reared at 30 °C. Survival of the animals was monitored. n = 5/group. (d) Conidia of A. vanbreuseghemii TIMM2789 dermatophytes were cultured at 28 °C for 26 h. Dermatophytes (0.5–4 × 106) were injected into the silkworms, and the silkworms were reared at 30 °C. Survival of the animals was monitored. n = 5/group. (e) Conidia of A. vanbreuseghemii TIMM2789 dermatophytes were cultured at 28 °C for 24 h. Germinated conidia (8 × 106) or samples autoclaved at 121 °C for 15 min (heat-killed germinated conidia) were injected into silkworms, and silkworms were reared at 30 °C. Survival of the animals was monitored. n = 5/group.
Next, we examined the virulence of other dermatophytes in silkworms. Several fungal species cause dermatophytosis in humans30. Among them, Trichophyton rubrum is clinically isolated with the highest frequency. A. benhamiae and Microsporum canis also cause dermatophytosis. Silkworms were also killed by injection into the hemolymph of the germinated conidia of T. rubrum, A. benhamiae, and M. canis (Fig. 3, Table 1). These findings suggest that the silkworm infection model is useful for quantitatively evaluating dermatophyte virulence.
Figure 3.
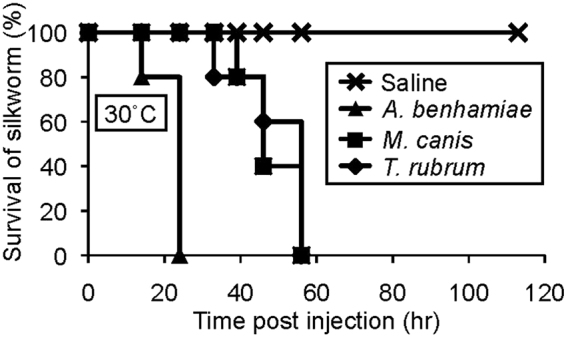
Pathogenicity of germinated conidia of Trichophyton rubrum, A. benhamiae, and Microsporum canis dermatophytes to silkworms. Conidia of Trichophyton rubrum, A. benhamiae, and Microsporum canis dermatophytes were cultured at 28 °C for 21 h. Conidia (2 × 106) were injected into silkworms, and the silkworms were reared at 30 °C. Survival of the animals was monitored. n = 5/group.
Table 1.
LD50 values of dermatophytes against silkworms.
| Strains | LD50 ± SD (×105 germinated spore/silkworm) |
|---|---|
| A. vanbreuseghemii TIMM2789 | 9 ± 1 |
| A. benhamiae IHEM20161 | 0.6 ± 0.1 |
| M. canis 20080 | 2 ± 0.6 |
| T. rubrum CBS118892 | 5 ± 2 |
Germinated conidia were injected into the silkworm hemolymph, and the silkworms were reared at 30 °C. Survival of the animals was monitored after 72 hours (n = 5/group). LD50 values of dermatophytes were determined from three independent experiments.
Evaluation of therapeutic effects of anti-fungal drugs using the silkworm infection model
We next examined whether anti-fungal drugs can be quantitatively evaluated using the silkworm dermatophyte-infection model. Oral administration of terbinafine or itraconazole is effective in human patients31,32. Intra-hemolymph injection of terbinafine or itraconazole extended silkworm life compared with silkworms injected with A. vanbreuseghemii alone (Fig. 4a). Injection of terbinafine or itraconazole into the midgut also had therapeutic effects against A. vanbreuseghemii infection (Fig. 4b). The ED50 values of terbinafine and itraconazole following intra-midgut injection were 11 ± 3 and 21 ± 3 μg/kg, respectively, against infection with A. vanbreuseghemii (Table 2). These results suggest that the therapeutic effects of anti-fungal drugs can be quantitatively evaluated using the silkworm dermatophyte infection model.
Figure 4.
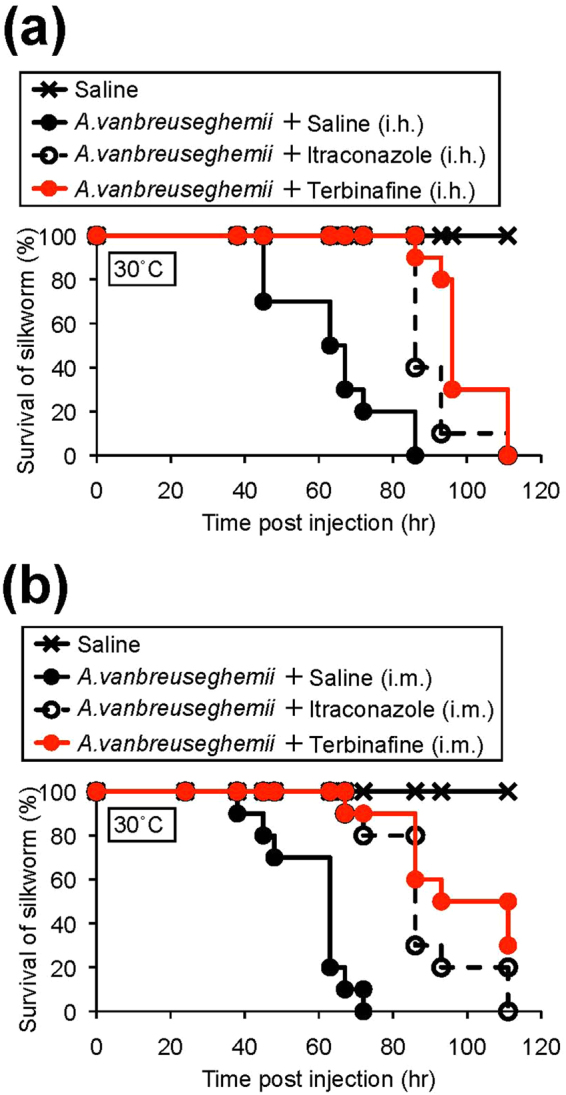
Therapeutic effects of terbinafine and itraconazole in silkworms infected with A. vanbreuseghemii TIMM2789 dermatophytes. Conidia of A. vanbreuseghemii TIMM2789 dermatophytes were cultured at 28 °C for 24 h. Cultured conidia (4 × 106) were injected into the silkworm hemolymph, followed by injection of 20 μg of terbinafine or 50 μg of itraconazole into hemolymph (i.h.: intra-hemolymph injection) or midgut (i.m.: intra-midgut injection). The silkworms were reared at 30 °C, and survival of the animals was monitored. n = 10/group.
Table 2.
MIC and ED50 values of terbinafine and itraconazole against A. vanbreuseghemii TIMM2789.
| MIC80 (μg·ml−1) | ED50 of i.h. (mg·kg−1 of larva) | ED50 of i.m. (mg·kg−1 of larva) | |
|---|---|---|---|
| Terbinafine | 0.013 | 5.3 ± 2.9 | 11 ± 3 |
| Itraconazole | 0.060 | 17 ± 6 | 21 ± 3 |
Germinated conidia were injected into the silkworm hemolymph, followed by injection of terbinafine or itraconazole into the hemolymph (i.h.) or midgut (i.m.). Silkworms were reared at 30 °C. Survival of the animals was monitored after 72 hours (n = 3/dose). ED50 values of anti-fungal agents were determined from three independent experiments.
Fluorescence imaging analysis of dermatophyte infection
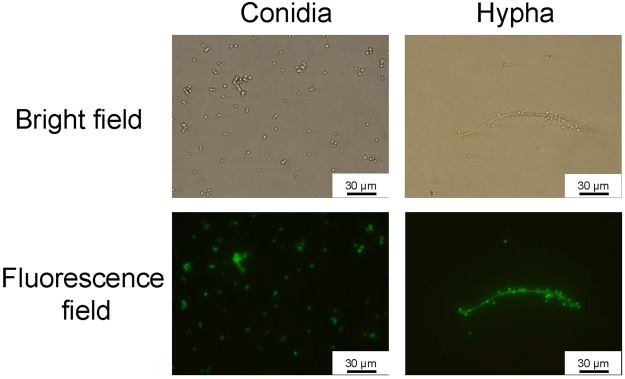
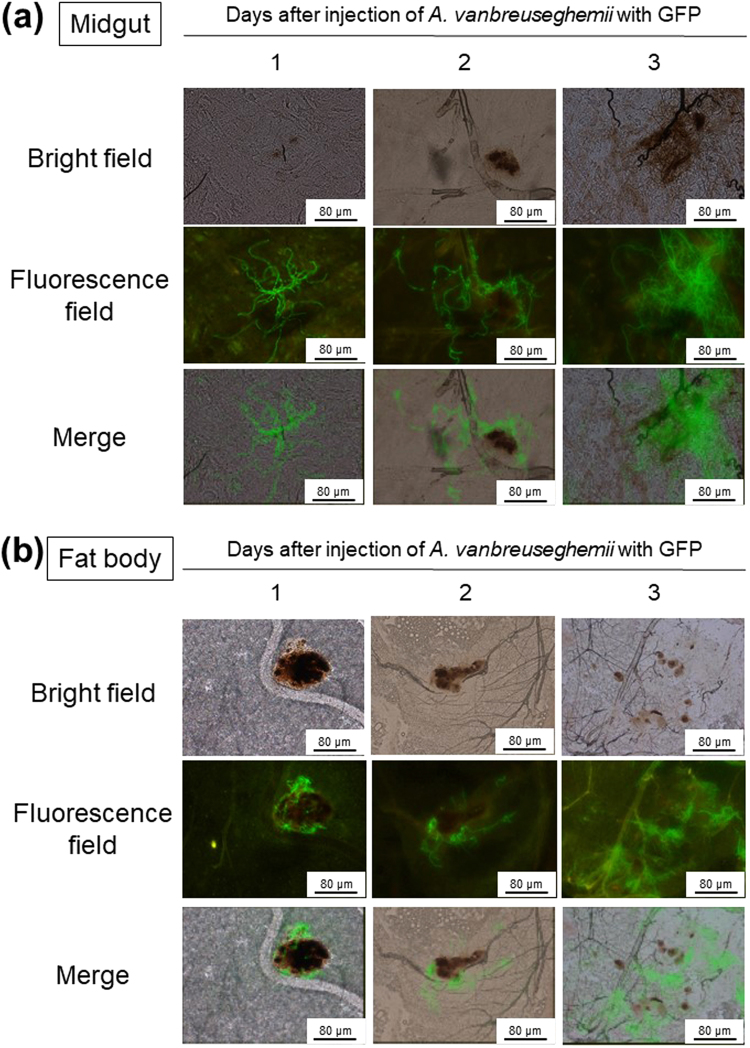
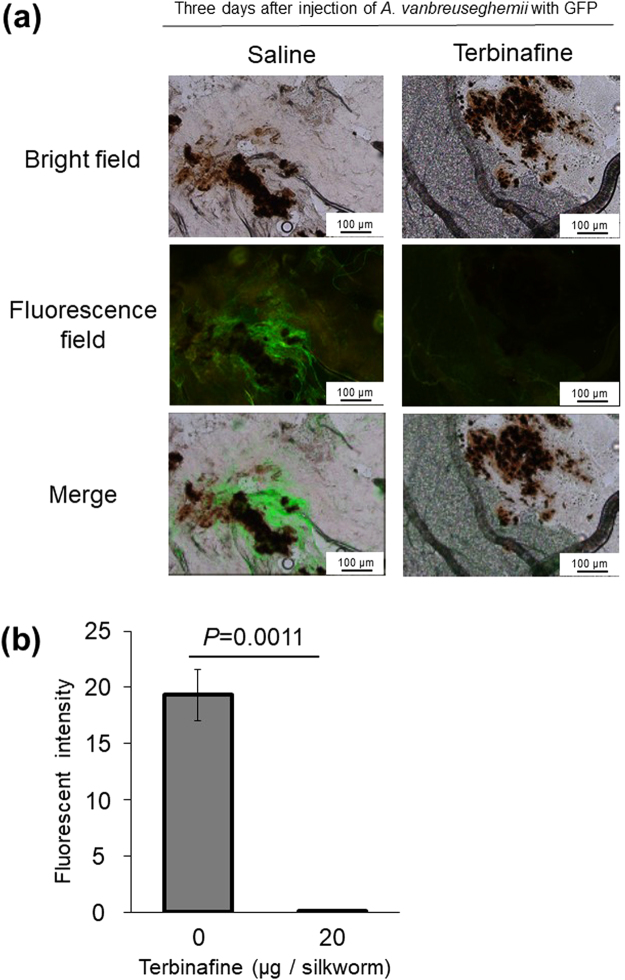
To observe the growth of dermatophytes in silkworms, we established a fluorescence imaging system with dermatophytes expressing eGFP. We confirmed the fluorescence of the conidia and hypha of dermatophytes expressing eGFP (Fig. 5). After injecting the conidia of dermatophytes expressing eGFP, mycelial growth was observed in the silkworm midgut and fat body (Fig. 6). Melanization, an insect immune response, was observed as brown spots in isolated silkworm tissues infected with dermatophytes and as brown filaments in the trachea, a silkworm respiratory organ. Injection of terbinafine inhibited the mycelial growth of dermatophytes expressing eGFP in the fat body at day 3 (Fig. 7a). Growth of the mycelia in the fat body was quantified by measuring fluorescence per weight of the organ and mycelial growth was inhibited in the terbinafine-injected group (Fig. 7b).
Figure 5.
Microscopic observation of A. vanbreuseghemii dermatophytes expressing eGFP. Left panels: Conidia of A. vanbreuseghemii expressing eGFP. Right panels: Hyphae of A. vanbreuseghemii expressing eGFP. Upper panels: bright field. Lower panels: fluorescence field.
Figure 6.
Fluorescence imaging of tissues of silkworms infected with A. vanbreuseghemii dermatophytes expressing eGFP. Conidia (5 × 106) of A. vanbreuseghemii expressing eGFP were injected into silkworms. The silkworms were reared at 30 °C for 1, 2, and 3 days. Silkworms were dissected and the midgut (a) or fat body (b) was isolated. Small pieces of fat body and midgut were placed on glass slides and pressed flat by cover slips. The tissues were observed under an inverted microscope (in brightfield, under excitation light, and merged images).
Figure 7.
Therapeutic effects of terbinafine on silkworms infected with A. vanbreuseghemii dermatophytes expressing eGFP. Conidia (5 × 106) of A. vanbreuseghemii dermatophytes expressing eGFP were injected into silkworms, followed by injection of terbinafine (20 μg) into the silkworm midgut. The silkworms were reared at 30 °C for 3 days. (a) Small pieces of fat body were placed on glass slides and pressed flat by cover slips. The samples were observed under a microscope (in brightfield, under excitation light, and merged images). (b) The fat body tissue was homogenized by sonication and the centrifuged pellet was suspended in water. The suspended sample (0.5 μl) was dropped on a microscope slide and dried. The samples were observed under a fluorescence microscope. After obtaining an image of the homogenized tissue, the fluorescence intensity of the sample area was determined. Florescence intensity/μg ∙ tissue protein is presented. n = 3/group.
Discussion
The present study demonstrated that injection of dermatophytes into silkworm hemolymph killed silkworms. By injecting A. vanbreuseghemii expressing eGFP into the silkworms, we were able to observe that terbinafine inhibited the mycelial growth of the dermatophyte. These findings suggest that the silkworm dermatophyte infection model is useful for evaluating the therapeutic effects of anti-fungal agents.
Although a dermatophyte infection model using mammals has been proposed, dermatophytes did not have lethal effects in that model. Injection of dermatophytes kills the larvae of Galleria mellonella, a lepidopteran insect like silkworm33. In the Galleria mellonella system, heat-killed dermatophytes were still lethal, indicating that the virulence of the dermatophytes is not due to the growth of the pathogens. In such a system, it is difficult to evaluate the therapeutic effects of compounds that exhibited anti-fungal activity in vitro. We demonstrated that injection of dermatophytes into silkworms is only lethal if the fungi are viable. Furthermore, anti-fungal drugs inhibited the growth and lethality of the dermatophyte in the silkworms. These findings indicate that mycelial growth is required for the dermatophyte to be pathogenic in silkworms. This is an advantage of the silkworm system compared with the G. mellonella system for evaluating the therapeutic effectiveness of anti-fungal reagents.
The cells of filamentous fungi grow as tube-like structures comprising multiple cells25, and therefore it is difficult to estimate cell number by counting the colony-forming units. Fluorescence imaging allows us to observe and quantify pathogens in animals34,35. Infected silkworms were used to evaluate anti-bacterial and anti-fungal drugs by monitoring host survival. By using pathogens expressing eGFP, we were able to observe proliferation of the pathogens in the host as well as suppression of the proliferation by the administration of the anti-fungal drugs against dermatophytosis. This is the first report of the use of fluorescence to detect the growth of pathogens expressing eGFP in silkworms, and as an indicator for evaluating drug efficacy in silkworms.
We demonstrated that germinated conidia had higher pathogenicity than non-germinated conidia in a silkworm dermatophyte infection model. Moreover, mycelial growth of dermatophytes expressing eGFP was observed in silkworm organs, such as the fat body and midgut. Mycelial growth in the silkworms was inhibited by intra-midgut administration of terbinafine. Terbinafine targets squalene epoxidase in dermatophytes and inhibits growth36. Phagocytosed dermatophytes elongate their hyphae inside macrophages, leading to rupture of the macrophage membrane in vitro 24. Based on ex vivo observations that dermatophyte hyphae invade the host tissue22,23, and in vitro observations that phagocytosed dermatophytes elongate their hyphae inside macrophages, leading to rupture of the macrophage membrane24, mycelial growth of dermatophytes is considered to be important for dermatophytosis. Our understanding of the molecular mechanisms of mycelial growth in vivo, however, is limited. Previous studies demonstrated that the use of silkworm infection models and mutants of pathogens are useful for determining the genes responsible for pathogenicity. In bacteria, virulence genes of S. aureus, pseudomonas aeruginosa, and Bacillus cereus were identified using silkworms by screening mutants with attenuated lethality in silkworms37–39. The gene encoding PTS1 of C albicans and the cyb2 gene of C. glabrate were identified as a novel virulence gene or adaptive gene, respectively, in fungi based on screening the fungal mutants using the silkworm infection models18,19. Genetic techniques to construct mutant libraries have been established in dermatophytes28. Therefore, the silkworm dermatophyte infection model will be useful for determining the factors responsible for the mycelial growth of dermatophytes in animals.
We found that the silkworm was killed by injection of four different species of dermatophytes, including T. rubrum, which is the most frequently isolated dermatophyte from patients with dermatophytosis. In a mammalian model using the guinea pig, T. rubrum causes less virulence than the other three dermatophytes, and establishing a reproducible system has proved difficult8. T. rubrum exhibited a similar LD50 as A. vanbreuseghemii and M. canis in the silkworm infection model, suggesting that the silkworm infection model could be useful for screening agents effective against T. rubrum.
The silkworm dermatophyte-infection model is useful for evaluating orally administered agents with therapeutic effects. Fluconazole, an orally administered anti-fungal used clinically, had therapeutic effects following midgut injection in the silkworm infection model with C. neoformans. On the other hand, midgut injection of amphotericin B, which is not orally available to human patients due to intestinal absorption problems, did not exhibit therapeutic effects in silkworms infected with C. neoformans 12. Intra-midgut injection in silkworms corresponds with oral administration in humans12. In the present study, intra-midgut injection of the anti-fungal medicines terbinafine and itraconazole, which are administered orally to human patients, had therapeutic effects in silkworm. The ED50 values of terbinafine and itraconazole were 11 ± 3 and 21 ± 3 mg/kg, respectively, in the silkworm infection model. The recommended daily oral dose of terbinafine and itraconazole for human tinea pedis (dermatophyte foot infection) is 250 mg and 400 mg/person, respectively40. Given that the average weight of humans is 60 kg, the calculated values of terbinafine and itraconazole are 4.2 and 6.7 mg/kg, respectively, for humans. Therefore, the therapeutically effective doses of these agents in the silkworm were consistent with the effective doses in humans. The results suggest that the silkworm model is highly valuable for screening orally and therapeutically effective drugs against dermatophytosis.
In conclusion, the silkworm dermatophyte infection model is a quantitative in vivo evaluation system for orally administered anti-fungal drugs. Using dermatophytes expressing eGFP allowed us to investigate mycelial growth in the animal. Many active anti-fungal substances have been reported41–44. From these candidates, therapeutically effective agents may be discovered using this silkworm model as a second screening tool. This system might contribute to reducing the number of mammalian animals used for discovering novel anti-dermatophyte drugs.
Methods
Dermatophytes used in this study
Four species of dermatophytes, A. vanbreuseghemii, A. benhamiae, Microsporum canis, and T. rubrum, were used in this study (Table 3). Conidia of dermatophytes were stocked in vials with silica gel at −80 °C.
Table 3.
Dermatophyte strains used in this study.
Culture method
Dermatophyte conidia stocked at −80 °C were spread on modified 1/10 Sabouraud agar (Bacto peptone 0.2%, glucose 0.1%, KH2PO4 0.1%, MgSO4 · 7H2O 0.1%, Bacto agar 1.5%, pH unadjusted), and incubated for 7–14 days at 28 °C. After incubation, 0.05% Tween80 was added to the plate, and the conidia were collected. The number of conidia was counted using a hemocytometer under a microscope. Conidia were cultured in Sabouraud medium (Bacto peptone1%, glucose 4%) with shaking at 28 °C for 20–28 h and used as germinated conidia.
Silkworm rearing
Eggs of Fu x Yo x Tukuba ∙ Ne were purchased from Ehime-Sanshu Co., Ltd. (Japan), disinfected, and hatched at 25–27 °C. The silkworms were fed an artificial diet, Silkmate 2 S, containing antibiotics purchased from Nihon-Nosan Co., Ltd. (Japan). Fifth instar larvae were used in the infection experiments.
Silkworm infection experiments
Silkworm fifth instar larvae were fed 1.5 g artificial diet (without antibiotics, Nihon-Nosan Co., Ltd., Japan) overnight. A suspension (50 μl) of the dermatophyte conidia or germinated conidia was injected into the silkworm hemolymph using a 1-ml tuberculin syringe (Terumo Medical Corporation, USA). To evaluate the therapeutic effects of anti-fungal agents, germinated conidia (4 × 106) were injected into the silkworm hemolymph, and then various concentrations of the anti-fungal agents (50 μl) dissolved in saline were injected into the silkworm hemolymph or midgut. To determine the ED50 values, three silkworms were injected for each dose of the anti-fungal agents, and the doses were created by 2-fold serial dilutions.
Construction of A. vanbreuseghemii expressing eGFP
A binary vector, pAg1h-eGFP, for the production of A. vanbreuseghemii expressing eGFP was constructed as follows. A genomic DNA fragment containing the upstream region (1.9 kb) of the A. vanbreuseghemii dipeptidyl peptidase V (DPPV) gene was amplified by polymerase chain reaction (PCR) with a pair of primers, AvDPPV-F1 (AAGACTAGTGACAATGACCCACAGGGCAAG) and AvDPPV-R1 (TGCGGGCCCTGTGAATGGAGCTAAGTTAATAGC). A DNA fragment containing the eGFP gene and the termination sequence of the Aspergillus nidulans tryptophan C gene (Accession No. ×02390; TtrpC) were also amplified from pCHSH75-GFP/TtrpCby PCR with a pair of primers28, eGFP-F1 (CGAACGGGCCCATGGTGAGCAAGGGCGAGGA) and TtrpC-R1 (AAGCTACTCGAGAAAGAAGGATTACCTCTAA). The two amplified fragments were double-digested with SpeI/ApaI or ApaI/XhoI, and inserted into the SpeI/ApaI and ApaI/XhoI sites within pAg1-hph 45, respectively, to generate pAg1h-eGFP. The pAg1h-eGFP was introduced into the wild-type A. vanbreuseghemii strain TIMM2789 using the Agrobacterium tumefaciens-mediated transformation method described previously28. After co-cultivation, the transformants were screened on Sabouraud agar containing 100–300 μg/ml hygromycin B. The desired transformants were selected by Southern blotting analysis and fluorescence microscopic observation. The transformant AvT-EGFP7 used in this study was confirmed to harbor two copies of the eGFP gene in the chromosomes.
Imaging of mycelial growth in silkworms
Conidia of A. vanbreuseghemii expressing eGFP were injected into silkworms, followed by injection with terbinafine (20 μg in saline) into the silkworm midgut. The silkworms were reared at 30 °C. Fat bodies and midguts were isolated after 1, 2, and 3 days. Small pieces of isolated tissues were placed on glass slides and pressed flat by cover slips46. The samples were examined under a microscope with bright light or ultraviolet light using a microscope equipped with fluorescent lens (Olympus). For quantitative evaluation of the fluorescence, fat body tissues were homogenized by sonication and centrifuged pellets (13 krpm, 3 min) were suspended in 10 μl of water. Suspended samples (0.5 μl) were dropped on a microscope slide, dried, and photographed under a fluorescence microscope. Fluorescence of the images was determined by Image J software (ImageJ 1.47t; National Institutes of Health, Bethesda, MD).
MIC determination
The MIC was determined according to the CLSI M38-A2 method. Conidia (2 × 103) were incubated with 2-fold serial dilutions of anti-fungal agents at 35 °C for 4 days, and MIC80 (concentration required to inhibit growth by 80%) was determined.
Anti-fungal agents
Terbinafine was purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Itraconazole was obtained from Sigma-Aldrich Corporation (St. Louis, MO, USA). The compounds were dissolved in dimethyl sulfoxide at a concentration of 10 mg/ml as stock solution. The stock solution was diluted with saline for therapeutic assay using the silkworms.
Statistical analysis
Survival curves were determined for each group based on the log-rank test, and p-values were determined. The log-rank test was performed for statistical processing using Prism software (GraphPad Software, Inc.).
Acknowledgements
We thank Kana Hashimoto, Miki Takahashi, and Mari Maeda (Genome Pharmaceuticals Institute Co., Ltd, Tokyo, Japan) for their technical assistance rearing the silkworms. This project was supported by JSPS KAKENHI grant number JP15H05783 (Scientific Research (S) to KS).
Author Contributions
M.I. conceived of the project, performed all the experiments, analyzed the data, and drafted the paper. Y.M. conceived of the project, designed the research, and wrote the paper. T.Y. constructed the dermatophyte expressing eGFP and wrote the paper. S.A. contributed to establish the silkworm infection model with dermatophytes expressing eGFP by fruitful discussion. K.S. wrote the paper.
Competing Interests
K.S. has an advisory role at the Genome Pharmaceuticals Institute Co., Ltd (Tokyo, Japan). The other authors declare no conflict of interest.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Garber G. An overview of fungal infections. Drugs. 2001;61(Suppl 1):1–12. doi: 10.2165/00003495-200161001-00001. [DOI] [PubMed] [Google Scholar]
- 2.Havlickova B, Czaika VA, Friedrich M. Epidemiological trends in skin mycoses worldwide. Mycoses. 2008;51(Suppl 4):2–15. doi: 10.1111/j.1439-0507.2008.01606.x. [DOI] [PubMed] [Google Scholar]
- 3.Rouzaud C, et al. Severe dermatophytosis and acquired or innate immunodeficiency: a review. Journal of Fungi. 2015;2:4. doi: 10.3390/jof2010004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lanternier F, et al. Deep dermatophytosis and inherited CARD9 deficiency. N Engl J Med. 2013;369:1704–1714. doi: 10.1056/NEJMoa1208487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lipner SR, Scher RK. Efinaconazole in the treatment of onychomycosis. Infect Drug Resist. 2015;8:163–172. doi: 10.2147/IDR.S69596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Katz HI. Drug interactions of the newer oral antifungal agents. The British journal of dermatology. 1999;141(Suppl 56):26–32. doi: 10.1046/j.1365-2133.1999.00011.x. [DOI] [PubMed] [Google Scholar]
- 7.Amichai B, Grunwald MH. Adverse drug reactions of the new oral antifungal agents–terbinafine, fluconazole, and itraconazole. International journal of dermatology. 1998;37:410–415. doi: 10.1046/j.1365-4362.1998.00496.x. [DOI] [PubMed] [Google Scholar]
- 8.Shimamura T, Kubota N, Shibuya K. Animal model of dermatophytosis. J Biomed Biotechnol. 2012;2012:125384. doi: 10.1155/2012/125384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Orihara Y, et al. A silkworm baculovirus model for assessing the therapeutic effects of antiviral compounds: characterization and application to the isolation of antivirals from traditional medicines. The Journal of general virology. 2008;89:188–194. doi: 10.1099/vir.0.83208-0. [DOI] [PubMed] [Google Scholar]
- 10.Hamamoto H, et al. Lysocin E is a new antibiotic that targets menaquinone in the bacterial membrane. Nat Chem Biol. 2015;11:127–133. doi: 10.1038/nchembio.1710. [DOI] [PubMed] [Google Scholar]
- 11.Ishii M, Matsumoto Y, Sekimizu K. Usefulness of silkworm as a host animal for understanding pathogenicity of Cryptococcus neoformans. Drug Discov Ther. 2016;10:9–13. doi: 10.5582/ddt.2016.01015. [DOI] [PubMed] [Google Scholar]
- 12.Matsumoto Y, et al. Quantitative evaluation of cryptococcal pathogenesis and antifungal drugs using a silkworm infection model with Cryptococcus neoformans. J Appl Microbiol. 2012;112:138–146. doi: 10.1111/j.1365-2672.2011.05186.x. [DOI] [PubMed] [Google Scholar]
- 13.Hamamoto H, et al. Quantitative evaluation of the therapeutic effects of antibiotics using silkworms infected with human pathogenic microorganisms. Antimicrob Agents Chemother. 2004;48:774–779. doi: 10.1128/AAC.48.3.774-779.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Panthee S, Paudel A, Hamamoto H, Sekimizu K. Advantages of the silkworm as an animal model for developing novel antimicrobial agents. Frontiers in Microbiology. 2017;8:373. doi: 10.3389/fmicb.2017.00373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fujiyuki T, Imamura K, Hamamoto H, Sekimizu K. Evaluation of therapeutic effects and pharmacokinetics of antibacterial chromogenic agents in a silkworm model of Staphylococcus aureusinfection. Drug Discov Ther. 2010;4:349–354. [PubMed] [Google Scholar]
- 16.Usui K, et al. Acute oral toxicity test of chemical compounds in silkworms. Drug Discov Ther. 2016;10:57–61. doi: 10.5582/ddt.2016.01025. [DOI] [PubMed] [Google Scholar]
- 17.Hamamoto H, Tonoike A, Narushima K, Horie R, Sekimizu K. Silkworm as a model animal to evaluate drug candidate toxicity and metabolism. Comparative biochemistry and physiology. Toxicology & pharmacology: CBP. 2009;149:334–339. doi: 10.1016/j.cbpc.2008.08.008. [DOI] [PubMed] [Google Scholar]
- 18.Ueno K, et al. Intestinal resident yeast Candida glabrata requires Cyb2p-mediated lactate assimilation to adapt in mouse intestine. PLoS One. 2011;6:e24759. doi: 10.1371/journal.pone.0024759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hanaoka N, et al. Identification of the putative protein phosphatase gene PTC1 as a virulence-related gene using a silkworm model of Candida albicans infection. Eukaryot Cell. 2008;7:1640–1648. doi: 10.1128/EC.00129-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakamura I, et al. Discovery of a new antifungal agent ASP2397 using a silkworm model of Aspergillus fumigatus infection. J Antibiot (Tokyo) 2017;70:41–44. doi: 10.1038/ja.2016.106. [DOI] [PubMed] [Google Scholar]
- 21.Ishii M, Matsumoto Y, Sekimizu K. Usefulness of silkworm as a model animal for understanding the molecular mechanisms of fungal pathogenicity. Drug Discov Ther. 2015;9:234–237. doi: 10.5582/ddt.2015.01052. [DOI] [PubMed] [Google Scholar]
- 22.Kaufman G, Horwitz BA, Duek L, Ullman Y, Berdicevsky I. Infection stages of the dermatophyte pathogen Trichophyton: microscopic characterization and proteolytic enzymes. Med Mycol. 2007;45:149–155. doi: 10.1080/13693780601113618. [DOI] [PubMed] [Google Scholar]
- 23.Duek L, Kaufman G, Ulman Y, Berdicevsky I. The pathogenesis of dermatophyte infections in human skin sections. J Infect. 2004;48:175–180. doi: 10.1016/j.jinf.2003.09.008. [DOI] [PubMed] [Google Scholar]
- 24.Campos MR, Russo M, Gomes E, Almeida SR. Stimulation, inhibition and death of macrophages infected with Trichophyton rubrum. Microbes Infect. 2006;8:372–379. doi: 10.1016/j.micinf.2005.07.028. [DOI] [PubMed] [Google Scholar]
- 25.Clemons KV, Stevens DA. Conventional or molecular measurement of Aspergillus load. Med Mycol. 2009;47(Suppl 1):S132–137. doi: 10.1080/13693780802213340. [DOI] [PubMed] [Google Scholar]
- 26.Hoffman RM. The multiple uses of fluorescent proteins to visualize cancer in vivo. Nat Rev Cancer. 2005;5:796–806. doi: 10.1038/nrc1717. [DOI] [PubMed] [Google Scholar]
- 27.Zimmer M. Green fluorescent protein (GFP): applications, structure, and related photophysical behavior. Chem Rev. 2002;102:759–781. doi: 10.1021/cr010142r. [DOI] [PubMed] [Google Scholar]
- 28.Yamada T, Makimura K, Uchida K, Yamaguchi H. Reproducible genetic transformation system for two dermatophytes, Microsporum canis and Trichophyton mentagrophytes. Med Mycol. 2005;43:533–544. doi: 10.1080/13693780500057619. [DOI] [PubMed] [Google Scholar]
- 29.Ishii, M., Matsumoto, Y., Nakamura, I. & Sekimizu, K. Silkworm fungal infection model for identification of virulence genes in pathogenic fungus and screening of novel antifungal drugs. Drug Discov Ther, doi:10.5582/ddt.2016.01080 (2017). [DOI] [PubMed]
- 30.Marconi VC, Kradin R, Marty FM, Hospenthal DR, Kotton CN. Disseminated dermatophytosis in a patient with hereditary hemochromatosis and hepatic cirrhosis: case report and review of the literature. Med Mycol. 2010;48:518–527. doi: 10.3109/13693780903213512. [DOI] [PubMed] [Google Scholar]
- 31.Evans EG, Sigurgeirsson B. Double blind, randomised study of continuous terbinafine compared with intermittent itraconazole in treatment of toenail onychomycosis. The LION Study Group. Bmj. 1999;318:1031–1035. doi: 10.1136/bmj.318.7190.1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Drake LA, et al. Oral terbinafine in the treatment of toenail onychomycosis: North American multicenter trial. J Am Acad Dermatol. 1997;37:740–745. doi: 10.1016/S0190-9622(97)70111-7. [DOI] [PubMed] [Google Scholar]
- 33.Achterman RR, Smith AR, Oliver BG, White TC. Sequenced dermatophyte strains: growth rate, conidiation, drug susceptibilities, and virulence in an invertebrate model. Fungal Genet Biol. 2011;48:335–341. doi: 10.1016/j.fgb.2010.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hoffman, R. M. In Biomedical Optics (BiOS) 2008. 68680E-68680E-68687 (International Society for Optics and Photonics).
- 35.Manicassamy B, et al. Analysis of in vivo dynamics of influenza virus infection in mice using a GFP reporter virus. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:11531–11536. doi: 10.1073/pnas.0914994107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Favre B, Ryder NS. Characterization of squalene epoxidase activity from the dermatophyte Trichophyton rubrum and its inhibition by terbinafine and other antimycotic agents. Antimicrob Agents Chemother. 1996;40:443–447. doi: 10.1128/aac.40.2.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kaito C, et al. Silkworm pathogenic bacteria infection model for identification of novel virulence genes. Mol Microbiol. 2005;56:934–944. doi: 10.1111/j.1365-2958.2005.04596.x. [DOI] [PubMed] [Google Scholar]
- 38.Chieda Y, et al. Pathogenicity of gacA mutant of Pseudomonas aeruginosa PA01 in the silkworm, Bombyx mori. FEMS microbiology letters. 2005;244:181–186. doi: 10.1016/j.femsle.2005.01.032. [DOI] [PubMed] [Google Scholar]
- 39.Tran SL, et al. Haemolysin II is a Bacillus cereus virulence factor that induces apoptosis of macrophages. Cell Microbiol. 2011;13:92–108. doi: 10.1111/j.1462-5822.2010.01522.x. [DOI] [PubMed] [Google Scholar]
- 40.Gupta AK, Cooper EA. Update in antifungal therapy of dermatophytosis. Mycopathologia. 2008;166:353–367. doi: 10.1007/s11046-008-9109-0. [DOI] [PubMed] [Google Scholar]
- 41.Magnusson J, Strom K, Roos S, Sjogren J, Schnurer J. Broad and complex antifungal activity among environmental isolates of lactic acid bacteria. FEMS microbiology letters. 2003;219:129–135. doi: 10.1016/S0378-1097(02)01207-7. [DOI] [PubMed] [Google Scholar]
- 42.De Lucca AJ, Walsh TJ. Antifungal peptides: novel therapeutic compounds against emerging pathogens. Antimicrob Agents Chemother. 1999;43:1–11. doi: 10.1128/aac.43.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hammer KA, Carson CF, Riley TV. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J Appl Microbiol. 2003;95:853–860. doi: 10.1046/j.1365-2672.2003.02059.x. [DOI] [PubMed] [Google Scholar]
- 44.Vicente MF, Basilio A, Cabello A, Pelaez F. Microbial natural products as a source of antifungals. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2003;9:15–32. doi: 10.1046/j.1469-0691.2003.00489.x. [DOI] [PubMed] [Google Scholar]
- 45.Yamada T, et al. Agrobacterium tumefaciens-mediated transformation of the dermatophyte, Trichophyton mentagrophytes: an efficient tool for gene transfer. Med Mycol. 2009;47:485–494. doi: 10.1080/13693780802322240. [DOI] [PubMed] [Google Scholar]
- 46.Inagaki Y, et al. Fluorescence imaging for a noninvasive in vivo toxicity-test using a transgenic silkworm expressing green fluorescent protein. Sci Rep. 2015;5:11180. doi: 10.1038/srep11180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Uchida R, et al. Nosokomycins, new antibiotics discovered in an in vivo-mimic infection model using silkworm larvae. I: Fermentation, isolation and biological properties. J Antibiot (Tokyo) 2010;63:151–155. doi: 10.1038/ja.2010.9. [DOI] [PubMed] [Google Scholar]
- 48.Symoens F, et al. The dermatophyte species Arthroderma benhamiae: intraspecies variability and mating behaviour. J Med Microbiol. 2013;62:377–385. doi: 10.1099/jmm.0.053223-0. [DOI] [PubMed] [Google Scholar]
- 49.White TC, Oliver BG, Graser Y, Henn MR. Generating and testing molecular hypotheses in the dermatophytes. Eukaryot Cell. 2008;7:1238–1245. doi: 10.1128/EC.00100-08. [DOI] [PMC free article] [PubMed] [Google Scholar]