Abstract
Disruption of the cooperative balance between osteoblasts and osteoclasts causes various bone disorders, some of which are because of abnormal osteoclast recruitment. Osteoporosis, one of the bone disorders, is not effectively treated by currently available medicines. In addition to the development of novel drugs for palliative treatment, the exploitation of novel compounds for preventive treatment is important in an aging society. Quercetin, a major flavonoid found in many fruits and vegetables, has been expected to inhibit cancer and prevent several diseases because of its anti-inflammatory and estrogenic functions. It has been reported that quercetin has the potential to reduce bone resorption, but the mechanism by which this compound affects the differentiation of osteoclasts remains unknown. Here, using a bone marrow cell-based in vitro osteoclast differentiation system from bone marrow cells, we found that the ability of quercetin to inhibit osteoclastogenesis was related to its estrogenic activity. The inhibition was partially blocked by a specific antagonist for the nuclear receptor estrogen receptor α, but a specific antagonist of the membrane-type receptor GPR30 completely ablated this inhibition. Furthermore, quercetin suppressed the transient increase of Akt phosphorylation induced by the stimulation of macrophage colony-stimulating factor and receptor activator of NF-κB ligand with no effect on MAPK phosphorylation, suggesting exquisite crosstalk between cytokine receptor and G-protein coupled receptor signaling. These results indicate the important role of GPR30 in osteoclast differentiation and provide new insights to the development of new treatments for osteoporosis.
Abbreviations: RANKL, receptor activator of NF-κB ligand; M-CSF, macrophage colony-stimulating factor; QOL, quality of life; GPCR, G-protein coupled receptor; BMC, bone marrow cell; TRAP, tartrate-resistant acid phosphatase
Keywords: Osteoclast, Osteoporosis, Flavonoid, Quercetin, Estrogen, GPR30
Highlights
-
•
Flavonoids can prevent osteoporosis, but the precise mechanism remains unknown.
-
•
The major flavonoid quercetin inhibited osteoclastogenesis.
-
•
The inhibition was entirely rescued by GPR30 antagonism.
-
•
GPR30 has a key role in mechanism of osteoclastogenesis prevention by quercetin.
1. Introduction
Osteoclasts are primary cells responsible for bone resorption, and these cells are of hematopoietic stem cell origin. Precursors of osteoclasts have been demonstrated to share properties with the monocyte/macrophage cell lineage [1], [2]. Since the receptor activator of NF-κB ligand (RANKL) was discovered as an essential cytokine for osteoclast differentiation in the presence of macrophage colony-stimulating factor (M-CSF) [3], [4], [5], the molecular mechanisms of osteoclast differentiation and function have been extensively studied [6]. Drugs developed in these studies are expected to cure bone diseases such as bone metastasis, osteopetrosis, and osteoporosis; some of these drugs are currently in use [7].
In particular, osteoporosis is a disease for which treatments have become more important in an aging society for reasons such as increasing population of elderly patients and the close relationship of osteoporosis to the quality of life of patients. Disruption of the balance between bone formation by osteoblasts and bone resorption by osteoclasts results in osteoporosis. Treatments for osteoporosis include vitamin D3, calcium, and estrogen supplementation as well as bisphosphonates and anti-RANKL antibody administration [8]; various compounds are expected to have efficacy, particularly as preventive treatments.
Flavonoids are a class of the compounds expected to have preventive effects against osteoporosis [9]. They are secondary metabolites of plants and the most common group of polyphenols in the human diet. In plants, they are important for guarding against oxidant damage and attacks by insects and microbes. In humans, many benefits of flavonoids related to metabolic syndrome, cancer, brain health, and immune system have been suggested by recent researches. Quercetin is a major flavonoid found in many fruits and vegetables, including onion, caper, and apple; flavonoids may have preventive effects against some diseases and inhibitory effects against some cancers [10], [11]. These properties have been considered to be attributable to its anti-inflammatory or anti-oxidant action, but recent studies suggest that quercetin has further effects such as estrogenic activity [12].
Several studies illustrated that quercetin reduced bone resorption by suppressing the differentiation of osteoclasts [13], [14], [15]. In these studies, NF-κB, AP-1 [14], and NFATc1 [15] were involved in the effects of quercetin, whereas the effects on the estrogen receptor (ER) were controversial. These genes are known as the master genes of osteoclastogenesis, and it may be a natural consequence that these genes are affected by the suppression of osteoclast differentiation. Moreover, the receptors to which quercetin binds are unknown. Examination of these receptors and the downstream signal transduction is important to elucidate the precise mechanisms of the effects of quercetin.
In this study, we demonstrate the effect of quercetin on osteoclastogenesis using an in vitro osteoclast differentiation system using bone marrow-derived monocytes/macrophages. Suppression of osteoclast differentiation partially recovered because of ERα antagonism and entirely because of GPR30 antagonism. The membrane-type G-protein coupled receptor (GPCR) GPR30 was constantly expressed during osteoclast differentiation, and the suppression of this differentiation by a GPR30 agonist was similar to the suppression by quercetin. Signal molecule analysis revealed that quercetin and a GPR30 agonist suppressed the phosphorylation of Akt. Thus, the results of this study indicate the importance of GPCR signaling in addition to the cytokine signals, such as M-CSF and RANKL in osteoclast cells.
2. Materials and methods
2.1. Reagents and antibodies
Quercetin was purchased from Wako Pure Chemical (Osaka, Japan). The classical ERα/β antagonist ICI182,780, the GPR30 agonist G-1, and the GPR30 antagonist G-15 were purchased from Tocris Bioscience (Bristol, UK).
Anti-phospho-Akt, anti-Akt, anti-phospho-MAPK, and anti-MAPK antibodies were purchased from Cell Signaling Technology (Danvers, MA). Anti-β-actin antibody and HRP-conjugated anti-rabbit antibody were purchased from IMGENEX (San Diego, CA) and Santa Cruz Biotechnology (San Diego, CA) respectively.
2.2. In vitro osteoclastogenesis
Bone marrow-derived monocyte/macrophage osteoclast precursors were isolated as described previously [16], [17]. Briefly, bone marrow cells (BMCs) were flushed out from the bone marrow cavity of the femora and tibiae of 4–12-week-old C57BL/6 J male mice (Kyudo, Tosu, Japan) and suspended in α-MEM (Wako) supplemented with 10% fetal bovine serum (FBS; Nichirei, Tokyo, Japan), 100 U/ml penicillin, and 20,000 U/ml Leukoprol (Kyowa, Tokyo, Japan) as a source of M-CSF and cultured for 3 days in Petri dishes in a humidified atmosphere of 5% CO2. Then, after the removal of nonadherent cells and stromal cells by washing with PBS and by subsequent incubation for 5 min in 0.25% Trypsin/ 0.05% EDTA, the monocytes/macrophages were harvested in α-MEM/10% FBS by vigorous pipetting. The isolated monocytes/macrophages were seeded as osteoclast progenitors and cultured in α-MEM /10% FBS with M-CSF (equivalent of 20 ng/ml) and recombinant mouse soluble RANKL (sRANKL, 10 ng/ml, Wako) and/or other agents. The culture medium was exchanged every 2 days.
After culturing, osteoclasts were fixed in methanol and stained for tartrate-resistant acid phosphatase (TRAP) activity using a leukocyte acid phosphatase kit (Sigma-Aldrich, St. Louis, MO) according to the manufacturer's instructions. The TRAP activities of osteoclasts were measured with 20 μl of culture medium incubated for 4 min at 37 °C with 80 μl of buffer (40 mM NaCl, 4 mM EDTA, 0.2% Triton X-100, 400 mM acetic acid, 1.76 mg/ml l-ascorbic acid, 9.2 mg/ml sodium tartrate dihydrate, 3.6 mg/ml p-nitrophenylphosphate disodium) and terminated by the addition of 100 μl of 300 mM NaOH; the absorbance at 405 nm was measured using a Multiskan FC plate reader (Thermo Fischer Scientific, Waltham, MA).
Animal experiments were conducted in accordance with the institutional guidelines of Kagoshima University for the care and use of experimental animals and approved by the Animal Research Committee of Kagoshima University.
2.3. Semi-quantitative RT-PCR
Total RNA was extracted with Sepazol RNA I Super G (Nacalai Tesque, Kyoto, Japan), and 0.1 μg of the total RNA was used as a template for cDNA synthesis with a ReverTraAce qPCR RT Kit (Toyobo, Osaka, Japan). The sequences of the primers used for PCR were as follows:
GPR30 forward 5′-TGGTCCTTGTTTTCTTCATCTGCTG-3′ and reverse 5′-CACAGCACTGCTGAACCTGACCTCT-3′;
ERα forward 5′-CAGGGAGAAGAGTTTGTGTGCCTCA-3′ and reverse 5′-TCAGATCGTGTTGGGGAAGCCCTCT-3′;
ERβ forward 5′-GATGAGGGGAAGTGCGTGGAAGGGA-3′ and reverse 5′- TCACTGTGACTGGAGGTTCTGGGAG-3′;
β-actin forward 5′-TCACCCACACTGTGCCCATCTAC-3′ and reverse 5′-GAGTACTTGCGCTCAGGAGGAGC-3′.
2.4. Western blot analysis
After various stimulations, the cells were washed twice with cold PBS and extracted in lysis buffer [50 mM Tris–HCl (pH 7.5), 150 mM NaCl, 1% Nonidet P-40, 0.1% sodium deoxycholate, 1 mM sodium fluoride, 1 mM sodium vanadate, and a protease inhibitor cocktail (Nacalai Tesque)]. The extracts were centrifuged at 13,000 rpm for 10 min, and the supernatants were used for analysis. Samples containing equal amounts of protein were subjected to SDS-PAGE, and the proteins separated in the gel were electrotransferred onto polyvinylidene difluoride membranes. After blocking with 5% skim milk, the membrane was immunoblotted with the indicated antibodies, and subsequently with peroxidase-conjugated anti-rabbit IgG antibody. Immunoreactive proteins were visualized with Chemi-Lumi One Super (Nacalai Tesque) according to the manufacturer's instructions.
2.5. Statistical analyses
Statistical analyses were performed using SPSS Statistics (IBM). Data were analyzed by one-way analysis of variance followed by Tukey post hoc tests. For all graphs, data are shown as means±standard deviation (SD). Statistical significance is indicated as follows: *, p < 0.01; **, p < 0.05.
3. Results
3.1. Inhibition of osteoclast differentiation by quercetin
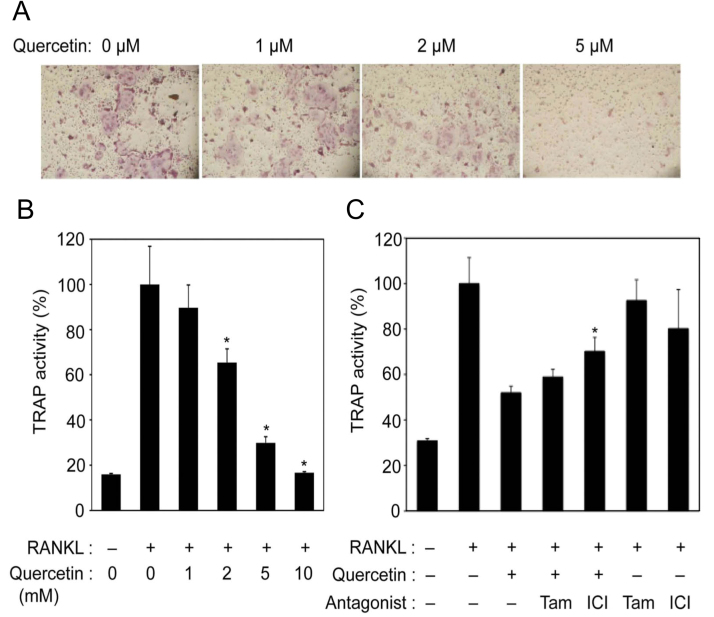
To examine the role of quercetin in osteoclastogenesis, we first cultured osteoclast precursors with sRANKL and quercetin at several concentrations. As shown in Fig. 1A, when osteoclast precursors were cultured with sRANKL in the absence of quercetin, TRAP-positive mononuclear preosteoclasts appeared on day 2 and began to fuse with each other to form multinucleated osteoclasts on days 3–4. In contrast, quercetin addition decreased the number of TRAP-positive multinucleated cells in a concentration-dependent manner. For more detailed analysis, we measured TRAP activity in the culture medium. Quercetin addition also decreased TRAP activities in the culture medium (Fig. 1B). Compared with the control, TRAP activities decreased by 35, 70, and 83% in the presence of 2, 5, and 10 μM of quercetin, respectively.
Fig. 1.
Suppression of in vitro osteoclastogenesis in the presence of quercetin. A. Osteoclast precursors obtained from bone marrow cells treated with macrophage colony-stimulating factor (M-CSF) for 3 days were cultured with quercetin at the indicated concentrations in the presence of M-CSF (20 ng/ml) and soluble receptor activator of NF-κB ligand (sRANKL) (10 ng/ml) for 3 days. After culturing, the cells were stained for tartrate-resistant acid phosphatase (TRAP) activity. B. Osteoclast precursors were cultured as indicated in A. TRAP activity in the culture medium was measured (mean±SD of four experiments; *, p<0.01 vs. positive control containing sRANKL without quercetin). C. Osteoclast precursors were cultured under differentiation conditions with 10 μM quercetin and 1 μM tamoxifen (Tam) or 1 μM ICI182,780 (ICI) for 3 days. After culturing, TRAP activity in the culture medium was measured [mean±SD of six experiments; *, p<0.01 vs. RANKL (+), quercetin (+), and antagonist (−)].
Many studies reported that quercetin has anti-oxidant activity, although estrogenic effects have also been reported in recent studies [12], [18]. There is no information regarding the estrogenic activity of quercetin in osteoclast cells; thus, we assessed whether quercetin functioned as an estrogenic agent in osteoclastogenesis using ER antagonists. As observed in Fig. 1A-B, osteoclast precursors were cultured with quercetin and/or the ER antagonists tamoxifen and ICI182,780 (Fig. 1C). There were no significant effects between tamoxifen or ICI182,780 and positive controls in the TRAP assay. TRAP activity decreased by quercetin addition, and this decline did not recover by tamoxifen addition. Upon the addition of another antagonist, ICI182,780, the decrease in TRAP activity recovered (p<0.01) but the recovery was only partially (approximately 38%).
3.2. Expression and function of the membrane-type ER GPR30 in osteoclasts
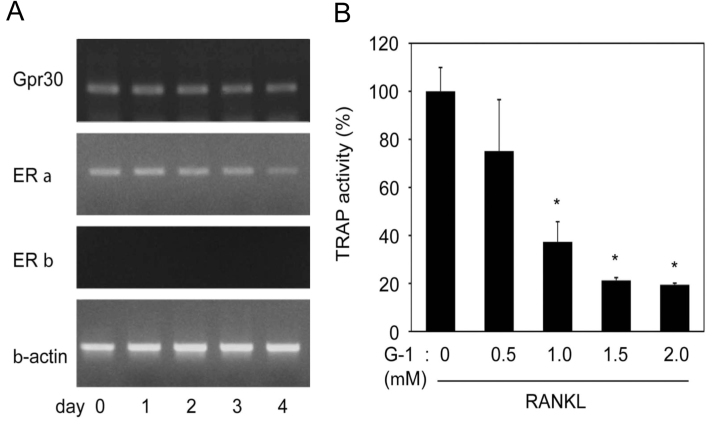
As tamoxifen and ICI182,780 antagonized the activities of estrogen, particularly its nuclear receptors ERα and ERβ, and partially reversed the effects of quercetin, we examined the involvement of another ER, GPR30/GPER1, in osteoclastogenesis. Carmeci et al. identified that GPR30 is a GPCR [19], and Heino et al. reported its expression in bone tissue [20]. As the expression profile of GPR30 during osteoclast differentiation has not been reported, we examined the expression of GPR30. Osteoclast precursors were cultured with M-CSF and sRANKL; samples were prepared sequentially for semi-quantitative RT-PCR to determine the expression of GPR30, ERα, ERβ and β-actin. As observed for ERα, GPR30 was constantly expressed throughout the osteoclastogenesis (Fig. 2A). ERβ expression was not observed, in accordance with the lack of the role of ERβ in osteoclast function [21].
Fig. 2.
Involvement of GPR30 in osteoclastogenesis. A. Bone marrow-derived monocyte/macrophage osteoclast precursors were cultured with macrophage colony-stimulating factor (M-CSF; 20 ng/ml) and soluble receptor activator of NF-κB ligand (sRANKL; 10 ng/ml) for the indicated periods with the ascertainment of osteoclast maturation at day 4. The expressions of GPR30, ERα, ERβ, and β-actin were determined by semi-quantitative RT-PCR in which the respective cDNAs were not saturated. B. Osteoclast precursors were cultured with M-CSF, sRANKL and the indicated concentration of G-1, a specific agonist of GPR30. Following osteoclast maturation, tartrate-resistant acid phosphatase (TRAP) activity in the culture medium was measured (mean±SD of four experiments; *, p<0.01 vs. cells cultured without G-1).
With the observation of GPR30 expression during osteoclastogenesis, we subsequently examined the involvement of GPR30 in osteoclastogenesis. As illustrated in Fig. 1, osteoclast precursors from BMCs were differentiated in the presence of the specific GPR30 agonist G-1. Similar to the case of quercetin, TRAP activity in the culture medium decreased by the addition of G-1 (Fig. 2B), with significant loss of multinucleated mature osteoclasts (data not shown). It is noteworthy that the differentiation was inhibited by a remarkably low concentration of a specific GPR30 agonist, suggesting the participation of GPR30 in osteoclastogenesis.
3.3. Effect of quercetin via GPR30 in osteoclasts
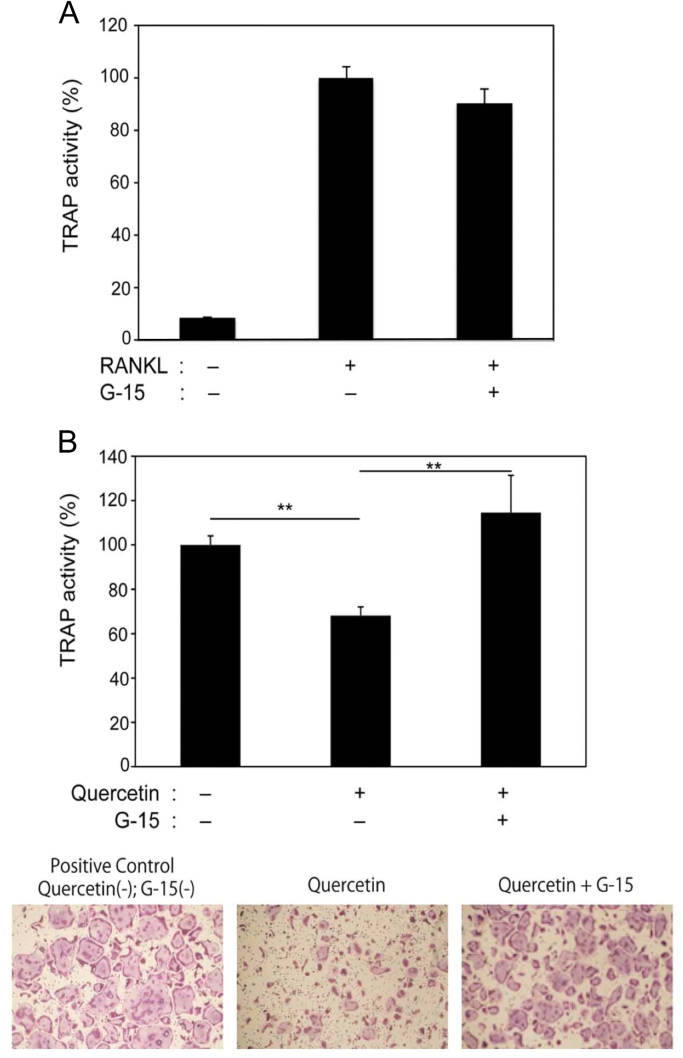
These results revealed that osteoclastogenesis was suppressed by both quercetin and a GPR30 pathway. Next, we analyzed whether quercetin suppressed the differentiation of osteoclasts via the GPR30 signaling pathway using its specific antagonist G-15. There was no significant effect of G-15 alone on the positive control (Fig. 3A). However, the addition of G-15 clearly reversed the suppression induced by quercetin (Fig. 3B), demonstrating that the direct binding of quercetin to GPR30 is important for osteoclastogenesis. TRAP images of the recovery effect by G-15 are shown in the lower part of Fig. 3B.
Fig. 3.
Reversal of quercetin-induced suppression of osteoclastogenesis by a specific GPR30 antagonist. A. Bone marrow-derived monocyte/macrophage osteoclast precursors were cultured with macrophage colony-stimulating factor (M-CSF; 20 ng/ml) and soluble receptor activator of NF-κB ligand (sRANKL; 10 ng/ml) with or without G-15 (10 µM), a specific antagonist of GPR30. B. Preosteoclasts treated as described in A were cultured with or without quercetin (5 μM) and G-15 (10 μM). Following osteoclast maturation, tartrate-resistant acid phosphatase (TRAP) activity in the culture medium was measured (mean±SD of eight experiments; **, p<0.05), and the images of the TRAP stain are shown in the lower parts of B.
3.4. Signals from GPR30 in osteoclast precursors
Since the identification of the master cytokine, RANKL [3], and the master transcription factor, NFATc1 [22], the mechanisms by which many signal molecules regulate various processes have been elucidated. However, signals from GPCRs in osteoclasts remain unknown. To disclose the mechanism by which the signal pathway in osteoclastogenesis was influenced by GPR30, we examined the activation of Akt and MAPK, which are involved in important signaling pathways in osteoclast differentiation and survival. To examine the molecular mechanisms in osteoclastogenesis, we cultured preosteoclasts with M-CSF and RANKL for 2 days.
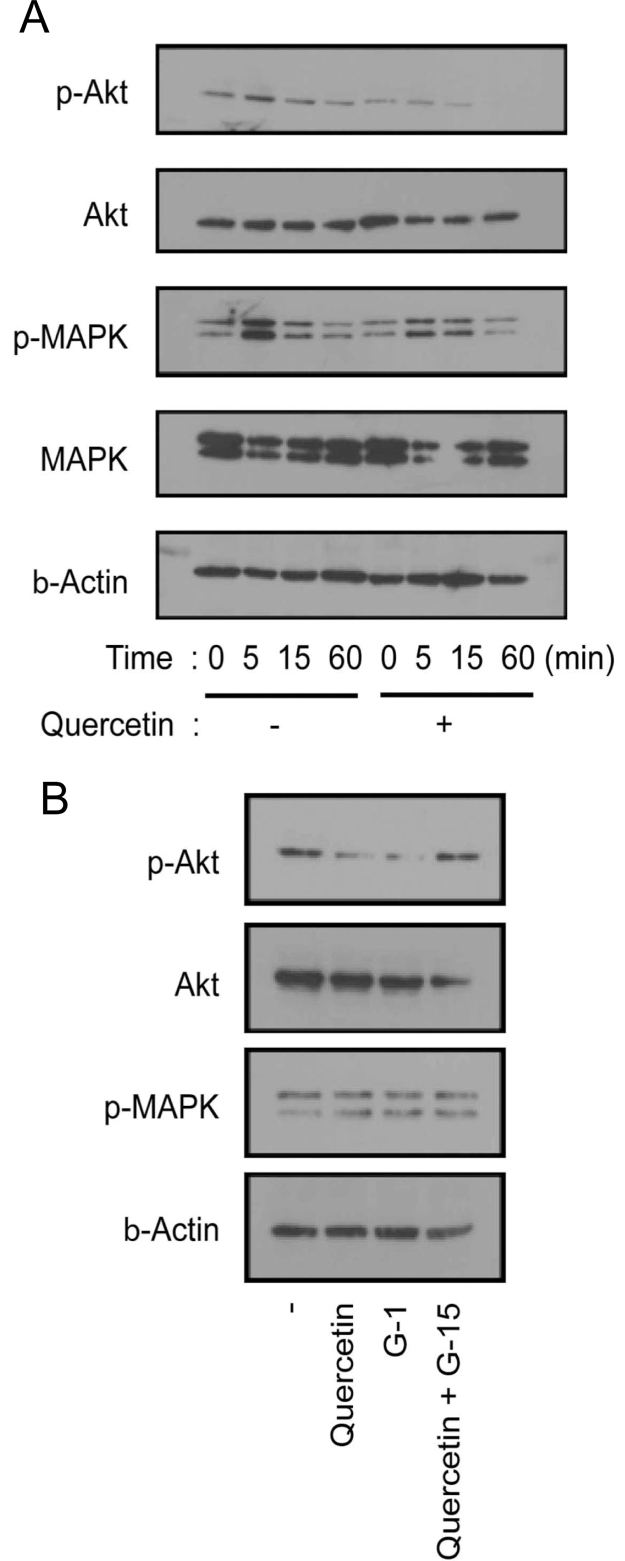
The preosteoclasts were starved in serum free medium for 3 h and stimulated by M-CSF and sRANKL with or without quercetin. As shown in Fig. 4A, temporal phosphorylation of Akt and MAPK were observed upon stimulation in the absence of quercetin. The addition of quercetin resulted in the disappearance of the transient phosphorylation of Akt with no or a modest effect on MAPK. Moreover quercetin suppressed the baseline level phosphorylation of Akt at 60 min. This result suggested the possibility that the phosphorylation of Akt in response to cytokine signaling was impaired by GPCR signaling.
Fig. 4.
Effects on signal molecules by quercetin and ligands of GPR30. A. Preosteoclasts, which are osteoclast precursor cells, were treated for 2 days with macrophage colony-stimulating factor (M-CSF; 20 ng/ml) and soluble receptor activator of NF-κB ligand (sRANKL; 10 ng/ml), starved in serum-free medium for 3 h, and stimulated by M-CSF (20 ng/ml) and sRANKL 10ng/ml) with or without quercetin (10 μM) for the indicated time period. After stimulation, the cells were extracted, and equal amounts of the cell lysates were subjected to western blot analysis. B. Preosteoclasts treated as described in A were stimulated with quercetin (10 μM) and G-1 (10 μM) or quercetin (10 μM) and G-15 (10 μM) for 15 min. The cells were extracted and equal amounts of the cell lysates were subsequently subjected to western blot analysis.
As previously mentioned, quercetin inhibited the osteoclastogenesis via GPR30 signaling. To confirm the involvement of GPR30 in the action of quercetin, we analyzed Akt phosphorylation using GPR30 antagonists and agonists. Preosteoclasts cultured with M-CSF and RANKL were stimulated with or without quercetin and GPR30 ligands for 15 min. As shown in Fig. 4B, similar to quercetin, the GPR30 agonist suppressed Akt phosphorylation. Further, the GPR30 antagonist G-15 rescued the quercetin-induced suppression of Akt phosphorylation. In contrast, the phosphorylation of MAPK was not changed in any case. Taken together, the abovementioned results obtained in this study suggest that quercetin inhibited osteoclastogenesis via GPR30, and that the phosphorylation of Akt is involved in this activity.
4. Discussion
In this study, we demonstrated that quercetin suppressed osteoclastogenesis induced by RANKL from osteoclast precursors in a concentration-dependent manner. We particularly focused on the estrogenic activity of quercetin and found that the suppression of osteoclastogenesis was not completely reversed by ERα and ERβ antagonists but by an antagonist for the novel membrane-type ER GPR30. GPR30 was expressed during osteoclastogenesis, and a GPR30 agonist suppressed osteoclastogenesis in a manner similar to that of quercetin but at an extremely low concentration. Moreover, a GPR30 antagonist reversed the inhibition induced by quercetin, reinforcing our hypothesis that quercetin functions via the GPR30 pathway in osteoclastogenesis. Our analyses of signal molecules indicated that Akt phosphorylation was inhibited by the addition of quercetin and a GPR30 agonist, suggesting that Akt participates in the quercetin-mediated suppression of osteoclastogenesis. Ford et al. described the involvement of GPR30 on bone mineralization by using Gpr30 knockout mice, but they reported no information about the mechanism of signal transduction [23], and the function of GPR30 remains unresolved. In fact, Martensson et al. demonstrated that deletion of GPR30 reduced bone growth but reported that there was no effect on the skeleton of male mice [24]. Additionally, Windahl et al. showed that estrogen treatment had no effect of reduced growth plate height on Gpr30 KO mice [25]. Our in vitro results together with their in vivo studies help to clarify the functions of GPR30.
GPR30 is a member of the GPCR family [19], which is considered as the most successful target for the drug discovery. Garland described in his review that the analyses of approved drugs typically quote 30% to 50% of all medications as exerting their effects via members of this family [26]. He also noted that GPCRs are still a mainstay for new drugs, as seen by examination of recent regulatory approvals in the United States. There were 21, 30, and 39 new molecular entities approved by the Food and Drug Administration in 2010, 2011, and 2012, respectively. Of these, 5 of 21 (2010), 5 of 30 (2011), and 7 of 39 (2012) target GPCR, giving a total of 17 of 90 (19%) over the 3 years. Despite the importance of drug exploitation for osteoporosis, studies of the roles of GPCRs in osteoclasts have not advanced, excluding studies on parathyroid hormone and calcitonin, which are the essential hormones for bone metabolism. The crucial participation of RANKL, a TNF family cytokine, in osteoclastogenesis has been indicated in several studies; however, as revealed in our results, the additional signals from other receptors must not be ignored.
Osteoporosis is a disorder caused by an imbalance between bone-forming osteoblasts and bone-resorbing osteoclasts. In the treatment of osteoporosis, bisphosphonates are frequently administered as they are the inhibitors of osteoclast functions. Regardless of their benefits, in a subset of patients receiving these drugs, osteonecrosis of the jaws has emerged as a significant complication [27]. Osteonecrosis induced by the adverse effect of bisphosphonates is called bisphosphonate-related osteonecrosis of the jaw (BRONJ). The inhibition of osteoclast function can also inhibit normal bone turnover, preventing local microdamage from normal mechanical loading or injury being repaired; this ultimately results in bone necrosis. The complete blockage of osteoclastogenesis may be an etiology of BRONJ; thus the mild regulation of osteoclasts such as that by quercetin may be important for the treatment of osteoporosis.
In addition to palliative treatment, preventive care is also important for osteoporosis. Regarding preventive care, agents with few harmful actions are required. In this context, flavonoids such as quercetin have useful characteristics. As more studies investigate the functions of flavonoids in other cell types, their usefulness will increase. It is desired that the effects of flavonoids are comprehensively assessed in all cell types. In this study, we could not show direct binding of quercetin to GPR 30, though it has been reported that quercetin stimulated c-fos expression via GPR30 in breast cancer cells [28]. It is well known that the G protein-coupled receptor activates the Akt/c-Fos signal pathway. Therefore, it is a possible mechanism that the action of quercetin via GPR30 could influence Akt signaling. Our results indicate that the phosphorylation of Akt but not of MAPK is affected by quercetin and GPR30 signal pathways; however the precise mechanism remains unknown. Akt may function in almost all the cell types; thus the investigation of the effects of agents on Akt phosphorylation may provide useful information.
In conclusion, the results presented in this study provide novel insights into the effects of quercetin on osteoclastogenesis, particularly the involvement of GPR30 and the effect on the phosphorylation of Akt. Because of the direct binding of quercetin to GPR30, further investigation of quercetin and other flavonoids is needed, but quercetin has the potential to be a useful therapeutic for osteoporosis.
Acknowledgments
We thank Shouko Kato for secretarial assistance. This work was supported by grants-in-aid for scientific research (No. 22791792 to MM) from the Japanese Society for the Promotion of Science. The authors declare that they have no conflicts of interest regarding the contents of this article.
MM and TS designed the study and wrote the paper. MM, TT, and KT performed and analyzed the experiments. KT, MM, MS and TS reviewed the results and discussed the manuscript, and revised the manuscript. All authors approved the final version of the manuscript.
Footnotes
Transparency data associated with this article can be found in the online version at http://dx.doi.org/10.1016/j.bbrep.2016.10.013i.
Appendix A. Transparency document
Supplementary material
References
- 1.Felix R., Cecchini M.G., Hofstetter W., Elford P.R., Stutzer A., Fleisch H. Impairment of macrophage colony-stimulating factor production and lack of resident bone marrow macrophages in the osteopetrotic op/op mouse. J. Bone Miner. Res. 1990;5:781–789. doi: 10.1002/jbmr.5650050716. [DOI] [PubMed] [Google Scholar]
- 2.Kodama H., Yamasaki A., Nose M., Niida S., Ohgame Y., Abe M., Kumegawa M., Suda T. Congenital osteoclast deficiency in osteopetrotic (op/op) mice is cured by injections of macrophage colony-stimulating factor. J. Exp. Med. 1991;173:269–272. doi: 10.1084/jem.173.1.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yasuda H., Shima N., Nakagawa N., Yamaguchi K., Kinosaki M., Mochizuki S., Tomoyasu A., Yano K., Goto M., Murakami A., Tsuda E., Morinaga T., Higashio K., Udagawa N., Takahashi N., Suda T. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. USA. 1998;95:3597–3602. doi: 10.1073/pnas.95.7.3597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lacey D.L., Timms E., Tan H.L., Kelley M.J., Dunstan C.R., Burgess T., Elliott R., Colombero A., Elliott G., Scully S., Hsu H., Sullivan J., Hawkins N., Davy E., Capparelli C., Eli A., Qian Y.X., Kaufman S., Sarosi I., Shalhoub V., Senaldi G., Guo J., Delaney J., Boyle W.J. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–176. doi: 10.1016/s0092-8674(00)81569-x. [DOI] [PubMed] [Google Scholar]
- 5.Kong Y.Y., Yoshida H., Sarosi I., Tan H.L., Timms E., Capparelli C., Morony S., Oliveira-dos-Santos A.J., Van G., Itie A., Khoo W., Wakeham A., Dunstan C.R., Lacey D.L., Mak T.W., Boyle W.J., Penninger J.M. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature. 1999;397:315–323. doi: 10.1038/16852. [DOI] [PubMed] [Google Scholar]
- 6.Asagiri M., Takayanagi H. The molecular understanding of osteoclast differentiation. Bone. 2007;40:251–264. doi: 10.1016/j.bone.2006.09.023. [DOI] [PubMed] [Google Scholar]
- 7.Boyce B.F., Rosenberg E., de Papp A.E., Duong L.T. The osteoclast, bone remodelling and treatment of metabolic bone disease. Eur. J. Clin. Investig. 2012;42:1332–1341. doi: 10.1111/j.1365-2362.2012.02717.x. [DOI] [PubMed] [Google Scholar]
- 8.Russell R.G.G. Pharmacological diversity among drugs that inhibit bone resorption. Curr. Opin. Pharmacol. 2015;22:115–130. doi: 10.1016/j.coph.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 9.Prasain J.K., Carlson S.H., Wyss J.M. Flavonoids and age-related disease: risk, benefits and critical windows. Maturitas. 2010;66:163–171. doi: 10.1016/j.maturitas.2010.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Carvalho K.M., Morais T.C., de Melo T.S., de Castro Brito G.A., de Andrade G.M., Rao V.S., Santos F.A. The natural flavonoid quercetin ameliorates cerulein-induced acute pancreatitis in mice. Biol. Pharm. Bull. 2010;33:1534–1539. doi: 10.1248/bpb.33.1534. [DOI] [PubMed] [Google Scholar]
- 11.Dajas F. Life or death: neuroprotective and anticancer effects of quercetin. J. Ethnopharmacol. 2012;143:383–396. doi: 10.1016/j.jep.2012.07.005. [DOI] [PubMed] [Google Scholar]
- 12.van der Woude H., Ter Veld M.G.R., Jacobs N., van der Saag P.T., Murk A.J., Rietjens I.M.C.M. The stimulation of cell proliferation by quercetin is mediated by the estrogen receptor. Mol. Nutr. Food Res. 2005;49:763–771. doi: 10.1002/mnfr.200500036. [DOI] [PubMed] [Google Scholar]
- 13.Woo J.T., Nakagawa H., Notoya M., Yonezawa T., Udagawa N., Lee I.S., Ohnishi M., Hagiwara H., Nagai K. Quercetin suppresses bone resorption by inhibiting the differentiation and activation of osteoclasts. Biol. Pharm. Bull. 2004;27:504–509. doi: 10.1248/bpb.27.504. [DOI] [PubMed] [Google Scholar]
- 14.Wattel A., Kamel S., Prouillet C., Petit J.P., Lorget F., Offord E., Brazier M. Flavonoid quercetin decreases osteoclastic differentiation induced by RANKL via a mechanism involving NFκB and AP-1. J. Cell Biochem. 2004;92:285–295. doi: 10.1002/jcb.20071. [DOI] [PubMed] [Google Scholar]
- 15.Tsuji M., Yamamoto H., Sato T., Mizuha Y., Kawai Y., Taketani Y., Kato S., Terao J., Inakuma T., Takeda E. Dietary quercetin inhibits bone loss without effect on the uterus in ovariectomized mice. J. Bone Miner. Metab. 2009;27:673–681. doi: 10.1007/s00774-009-0088-0. [DOI] [PubMed] [Google Scholar]
- 16.Masuhara M., Sato T., Hada N., Hakeda Y. Protective protein/cathepsin A down-regulates osteoclastogenesis by associating with and degrading NF-κB p50/p65. J. Bone Miner. Metab. 2009;27:46–56. doi: 10.1007/s00774-008-0017-7. [DOI] [PubMed] [Google Scholar]
- 17.Okayasu M., Nakayachi M., Hayashida C., Ito J., Kaneda T., Masuhara M., Suda N., Sato T., Hakeda Y. Low-density lipoprotein receptor deficiency causes impaired osteoclastogenesis and increased bone mass in mice because of defect in osteoclastic cell-cell fusion. J. Biol. Chem. 2012;287:19229–19241. doi: 10.1074/jbc.M111.323600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Han D.H., Denison M.S., Tachibana H., Yamada K. Relationship between estrogen receptor-binding and estrogenic activities of environmental estrogens and suppression by flavonoids. Biosci. Biotechnol. Biochem. 2002;66:1479–1487. doi: 10.1271/bbb.66.1479. [DOI] [PubMed] [Google Scholar]
- 19.Carmeci C., Thompson D.A., Ring H.Z., Francke U., Weigel R.J. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics. 1997;45:607–617. doi: 10.1006/geno.1997.4972. [DOI] [PubMed] [Google Scholar]
- 20.Heino T.J., Chagin A.S., Savendahl L. The novel estrogen receptor G-protein-coupled receptor 30 is expressed in human bone. J. Endocrinol. 2008;197:R1–R6. doi: 10.1677/JOE-07-0629. [DOI] [PubMed] [Google Scholar]
- 21.Carnesecchi J., Vanacker J.M. Estrogen-related receptors and the control of bone cell fate. Mol. Cell. Endocrinol. 2015 doi: 10.1016/j.mce.2015.07.019. [DOI] [PubMed] [Google Scholar]
- 22.Takayanagi H., Kim S., Koga T., Nishina H., Isshiki M., Yoshida H., Saiura A., Isobe M., Yokochi T., Inoue J., Wagner E.F., Mak T.W., Kodama T., Taniguchi T. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell. 2002;3:889–901. doi: 10.1016/s1534-5807(02)00369-6. [DOI] [PubMed] [Google Scholar]
- 23.Ford J., Hajibeigi A., Long M., Hahner L., Gore C., Hsieh J.T., Clegg D., Zerwekh J., Oz O.K. GPR30 deficiency causes increased bone mass, mineralization, and growth plate proliferative activity in male mice. J. Bone Miner. Res. 2011;26:298–307. doi: 10.1002/jbmr.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Martensson U.E., Salehi S.A., Windahl S., Gomez M.F., Sward K., Daszkiewicz-Nilsson J., Wendt A., Andersson N., Hellstrand P., Grande P.O., Owman C., Rosen C.J., Adamo M.L., Lundquist I., Rorsman P., Nilsson B.O., Ohlsson C., Olde B., Leeb-Lundberg L.M. Deletion of the G protein- coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiol-stimulated insulin release in female mice. Endocrinology. 2009;150:687–698. doi: 10.1210/en.2008-0623. [DOI] [PubMed] [Google Scholar]
- 25.Windahl S.H., Andersson N., Chagin A.S., Martensson U.E., Caristen H., Olde B., Swanson C., Moverare-Skrtic S., Savedahl L., Lagerquist M.K., Leeb-Lundberg L.M., Ohlsson C. The role of the G protein-coupled receptor GPR30 in the effects of estrogen in ovariectomized mice. Am. J. Physiol. Endocrinol. Metab. 2009;296:E490–E496. doi: 10.1152/ajpendo.90691.2008. [DOI] [PubMed] [Google Scholar]
- 26.Garland S.L. Are GPCRs still a source of new targets? J. Biomol. Screen. 2013;18:947–966. doi: 10.1177/1087057113498418. [DOI] [PubMed] [Google Scholar]
- 27.Ruggiero S.L., Drew S.J. Osteonecrosis of the jaws and bisphosphonate therapy. J. Dent. Res. 2007;86:1013–1021. doi: 10.1177/154405910708601101. [DOI] [PubMed] [Google Scholar]
- 28.Maggiolini M., Vivacqua A., Fasanella G., Recchia A.G., Sisci D., Pezzi V., Montanaro D., Musti A.M., Picard D., Ando S. The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17β-estradiol and phytoestrogens in breast cancer cells. J. Biol. Chem. 2004;279:2700–27016. doi: 10.1074/jbc.M403588200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material