Highlights
-
•
Effects of silica nanoparticles (SiO2-NPs) were investigated in Huh7 cells.
-
•
SiO2-NPs induced ER stress response and activated MAPK pathway.
-
•
SiO2-NPs induced inflammatory reaction by induction of TNF-α.
-
•
Activation of MAPK may lead to activation of AP-1 complex.
Keywords: Silica nanoparticles, Endoplasmic reticulum stress, MAPK, TNF-α, Huh7 cells
Abstract
Humans may be exposed to engineered silica nanoparticles (SiO2-NPs) but potential adverse effects are poorly understood, in particular in relation to cellular effects and modes of action. Here we studied effects of SiO2-NPs on cellular function in human hepatoma cells (Huh7). Exposure for 24 h to 10 and 50 μg/ml SiO2-NPs led to induction of endoplasmic reticulum (ER) stress as demonstrated by transcriptional induction of DNAJB9, GADD34, CHOP, as well as CHOP target genes BIM, CHAC-1, NOXA and PUMA. In addition, CHOP protein was induced. In addition, SiO2-NPs induced an inflammatory response as demonstrated by induction of TNF-α and IL-8. Activation of MAPK signalling was investigated employing a PCR array upon exposure of Huh7 cells to SiO2-NPs. Five of 84 analysed genes, including P21, P19, CFOS, CJUN and KSR1 exhibited significant transcriptional up-regulation, and 18 genes a significant down-regulation. Strongest down-regulation occurred for the proto-oncogene BRAF, MAPK11, one of the four p38 MAPK genes, and for NFATC4. Strong induction of CFOS, CJUN, FRA1 and CMYC was found after exposure to 50 μg/ml SiO2-NPs for 24 h. To analyse for effects derived from up-regulation of TNF-α, Huh7 cells were exposed to SiO2-NPs in the presence of the TNF-α inhibitor sauchinone, which reduced the induction of the TNF-α transcript by about 50%. These data demonstrate that SiO2-NPs induce ER stress, MAPK pathway and lead to inflammatory reaction in human hepatoma cells. Health implications of SiO2-NPs exposure should further be investigated for a risk assessment of these frequently used nanoparticles.
1. Introduction
Engineered silica nanoparticles (SiO2-NPs) are among the most popular nanoparticles. They are applied in industrial manufacturing, and applications including cosmetics, foods, medicines, drug delivery etc. Increased production and usage lead to augmented exposure of humans via oral intake and inhalation. Despite their widespread use, the potential health implications as well as molecular modes of action are not sufficiently known.
SiO2-NPs enter cells and induce time- and size-dependent cytotoxicity at high concentrations by induction of oxidative stress [1], membrane damage, as well as disturbed calcium homeostasis [2], [3]. In human intestinal Caco-2 cells, SiO2-NPs induced genotoxicity and pro-inflammatory reactions [4]. In human lung cells, SiO2-NPs induce oxidative stress and inflammation [5]. The induction of pro-inflammatory responses after exposure to SiO2-NPs was also found in human lung epithelial cells, human macrophages [6] and in the rat lung [7]. Recently, we have shown that SiO2-NPs induce endoplasmic reticulum (ER) stress in human hepatoma cells [8], pro-inflammatory cytokine TNF-α and activate MAPK pathways [9].
The ER stress response is activated when folding and export of proteins is perturbed under different cellular stress conditions. Accumulation of non- or misfolded proteins in the ER induces an adaptive response, the unfolded protein response (UPR), that reduces protein de novo synthesis and enhances the production of chaperones that facilitate protein folding and enhances the proteosomal degradation of misfolded proteins [10]. There are three distinct pathways triggered by ER stress namely protein kinase like ER kinase (PERK), inositol-requiring protein 1 (IRE1) and activating transcription factor 6 (ATF6), all of which induce the expression of different genes aiming to restore the normal function of the ER, and in case it fails, to induce apoptosis [11]. All three pathways are controlled through the chaperone binding immunoglobulin protein (BIP) (or also called 78 kDa glucose-regulated protein, GRP78). The ER stress response can be assessed by selective markers, such as induction of the chaperone BIP, splicing of XBP-1 mRNA, phosphorylation of eIF2α and induction of CHOP. The UPR has important consequences, including apoptosis. It accompanies acute and chronic liver diseases and plays a significant role in liver pathogenesis [12]. Additionally ER stress can activate NFƙB [13] leading to the expression of interferons (INFs) Type I and inflammatory cytokines like TNF-α [14], which is involved in the inflammatory response, apoptosis, cell proliferation and cell differentiation.
Inflammatory and immune responses are regulated by multiple signalling pathways. Among them are the NFƙB and mitogen-activated protein kinase (MAPK) signalling pathways, which include many proteins including MAPK (originally called the extracellular signal-regulated kinase1/2 ERK1/2), p38, CREB, CMYC and CJUN N-terminal kinase (JNK) pathways. MAPK regulates the activities of many transcription factors. It is important in the cellular stress response, modulates a variety of inflammatory responses [15] and apoptosis, and plays a role in cancer development.
Based on our previous findings that SiO2-NPs induced the expression of ER stress markers [8], [9], and that SiO2-NPs induced TNF-α expression and activated the MAP kinases regulated transcription factors CJUN and CMYC [9], here we aimed to further explore and deepen our understanding on ER stress, on induction of inflammatory response, and its consequences such as MAPK activation following exposure to SiO2-NPs. To this end, we exposed human hepatoma cells Huh7 to SiO2-NPs followed by a detailed investigation of ER stress response. Exposure concentrations were selected based on the previous study on cytotoxicity of SiO2-NPs [8]. The used concentrations were non-toxic, therefore all detected effects are real effects and not unspecific toxic reactions. We analysed the expression of the chaperone DNAJB9, the ATF-4 target gene GADD34 and the pro-apoptotic gene CHOP, the expression of Il-8 as a marker for inflammation, as well as the expression of various genes belonging to the MAPK signalling pathway. Our study shows that all of these pathways are affected by exposure to SiO2-NPs, suggesting that further toxicological assessment of human exposure is needed for safety assessment of these nanoparticles.
2. Materials and methods
2.1. Chemicals
SiO2-NPs, sauchinone and SP600125 were purchased from Sigma-Aldrich (Buchs, Switzerland), anti-CHOP antibody was purchased from BioConcept (Allschwil, Switzerland), and anti-actin antibody was purchased from Fisher Scientific (Reinach, Switzerland), the secondary anti-mouse antibody IRDye 680RD was purchased from Lucerna-Chem (Lucerne, Switzerland).
2.2. Huh7 cells
The human hepatoma cell line Huh7 was kindly provided by Markus Heim, University Hospital Basel, Switzerland. Cells were grown in DMEM with GlutaMAX™ (LuBioScience, Lucerne, Switzerland) supplemented with 10% FCS for cultivation and without FCS for exposure experiments in a humidified incubator with 5% CO2 at 37 °C. Cells were usually split every 4 days and sub-cultured at split ratios of about 1:6.
2.3. Characterization of the SiO2-NPs
Surface characteristics, size distribution and appearance were previously reported [8].
2.4. RNA isolation, reverse transcription, and quantitative (q)PCR
Total RNA was isolated from Huh7 cells using Trizol reagent (Sigma-Aldrich, Buchs, Switzerland) according to the manufacturer’s instructions. RNA was reverse transcribed by Moloney murine leukemia virus reverse transcriptase (Promega Biosciences, Inc., Wallisellen, Switzerland) in the presence of random hexamers (Roche, Rotkreuz, Switzerland) and deoxynucleoside triphosphate (Sigma-Aldrich, Buchs, Switzerland). The reaction mixture was incubated for 5 min at 70 °C and then for 1 h at 37 °C. The reaction was stopped by heating at 95 °C for 5 min. qPCR was performed based on SYBR green fluorescence (SYBR green PCR master mix; Roche, Rotkreuz, Switzerland). The sequences of the used primers are shown in Table 1. The amplification conditions were 95 °C for 5 min. For initial denaturing, 40 cycles of 95 °C for 30 s for denaturing, 61 °C for 60 s for annealing and elongation. A melting curve was run afterwards. The difference in the cycle threshold (ΔCT) value was derived by subtracting the CT value for GAPDH, which served as an internal control, from the CT value for the target genes. All reactions were run in duplicates using a BioRad real time PCR machine (CFX 96 Real Time System). mRNA expression levels of target genes were expressed as a several fold increase according to the formula 2ΔCT (not exposed)–ΔCT (exposed) in which “not exposed” are solvent control samples. Due to graphical reasons the fold change is converted into Log2 values.
Table 1.
Sequences of used qPCR primers.
| Primer | Forward (5′ → 3′) | Reverse (5′ → 3′) |
|---|---|---|
| GAPDH | GAAGGTGAAGGTCGGAGTC | GAAGATGGTGATGGGATTTC |
| ATF4 | AGT GGC ATC TGT ATG AGC CCA | GCT CCT ATT TGG AGA GCC CCT |
| BiP | CGA GGA GGA GGA CAA GAA GG | GAC CTT GAA CGG CAA GAA CT |
| XBP-1s | TGC TGA GTC CGC AGC AGG TG | GCT GGC AGG CTC TGG GGA AG |
| NOXA | ATT ACC GCT GGC CTA CTG TG | GTG CTG AGT TGG CAC TGA AA |
| CHAC-1 | CCT GAA GTA CCT GAA TGT GCG AGA | GCA GCA AGT ATT CAA GGT TGT GGC |
| TNF-α | CAG CCT CTT CTC CTT CCT GA | TGAGGTACAGACCCTCTGAT |
| FRA-1 | CAG GCG GAG ACT GAC AAA CTG | TCC TTC CGG GAT TTT GCA GAT |
| CHOP | GGA GCA TCA GTC CCC CAC TT | TGT GGG ATT GAG GGT CAC ATC |
| CFOS | CCG GGG ATG CCT CTC TTA CT | CCAGGTCCGTGCAGAAGTC |
| PUMA | CGA CCT CAA CGC ACA GTA CG | AGG CAC CTA ATT GGG CTC CAT |
| CJUN | TCC AAG TGC CGA AAA AGG AAG | CGA GTT CTG AGC TTT CAA GGT |
| BIM | GTA TTC GGT TCG CTG CGT TC | CGC AGG CTG CAA TGG TCT AC |
| CMYC | TGA GGA GAC ACC GCC CAC | CAA CAT CGA TTT CTT CCT CAT CTT |
| GADD34 | CCC AGA AAC CCC TAC TCA TGA TC | GCC CAG ACA GCC AGG AAA T |
| DNAJB9 | TCG GCA TCA GAG CGC CAA ATC A | ACC ACT AGT AAA AGC ACT GTG TCC AAG |
| JUND | TCA TCA TCC AGT CCA ACG GG | TTC TGC TTG TGT AAA TCC TCC AG |
2.5. RT2 profiler™ PCR array human MAP kinase signalling pathway
Sample preparations and the experimental procedures were performed according to manufacturer’s instructions. Total RNA was isolated from Huh7 cells using the RNeasy kit (QIAGEN, Basel, Switzerland). cDNA was obtained using the RT2 first strand kit (QIAGEN, Basel, Switzerland) using 500 ng RNA as input for both the control and the treated sample. The qPCR performed was based on SYBR green fluorescence and the provided RT2 SYBR Green master mix was used. The amplification conditions were as described above. The obtained data were analysed using the QIAGEN web portal.
2.6. Preparation of cell extracts and immunoblotting
Cells were homogenized in 50 μl of lysis buffer (50 mM Tris, 150 mM NaCl, 15 mM EDTA, 0.1% Triton X-100 and 1 mM phenylmethylsulfonyl fluoride) incubated for 20 min on ice, centrifuged at 14,000 rpm for 5 min. Protein concentrations were determined with Thermo Scientific BCATM protein assay kit (Fish Scientific, Wohlen, Switzerland). Immunoblotting was performed as described [16]. Membranes were incubated with the primary antibodies anti-CHOP and anti-actin and with the secondary fluorescent anti-mouse antibody. To detect the CHOP and actin band, the membranes were scanned with Licor Odyssey scanner (Bad Homburg, Germany). Analysis to determine the intensity of the CHOP and actin bands was performed with Image J software.
2.7. Measurement of TNF − α protein
The amount of TNF-α protein in the cell culture supernatant was measured using the Cymax human TNF-α ELISA kit from Adipogen (Liestal, Switzerland) according to the manufacturer’s protocol.
2.8. Statistical analysis
Differences between treatments were assessed by one way ANOVA followed by a Bonferroni’s multiple comparison test to compare treatment means with respective controls. Results are given as means ± standard error of means. Differences were considered statistically significant with one asterisk at p = 0.01–0.05, two asterisks at p = 0.001–0.01 and three asterisks at p = 0.0001–0.001.
3. Results
3.1. Characterisation of SiO2-NPs
We characterized the used SiO2-NPs before [8]. They had a surface area/weight ratio of 115 m2 g−1, and size of 273 nm. Their surface was negatively charged (−12.7 mV). Nanoparticle tracking analysis showed that 50% of the SiO2-NPs had a size smaller than 100 nm, 40% a size between 100 and 200 nm, and 10% of the particles had sizes larger than 200 nm. As most of the particles showed a size smaller than 100 nm, potential biological activity can be expected.
3.2. Induction of ER stress
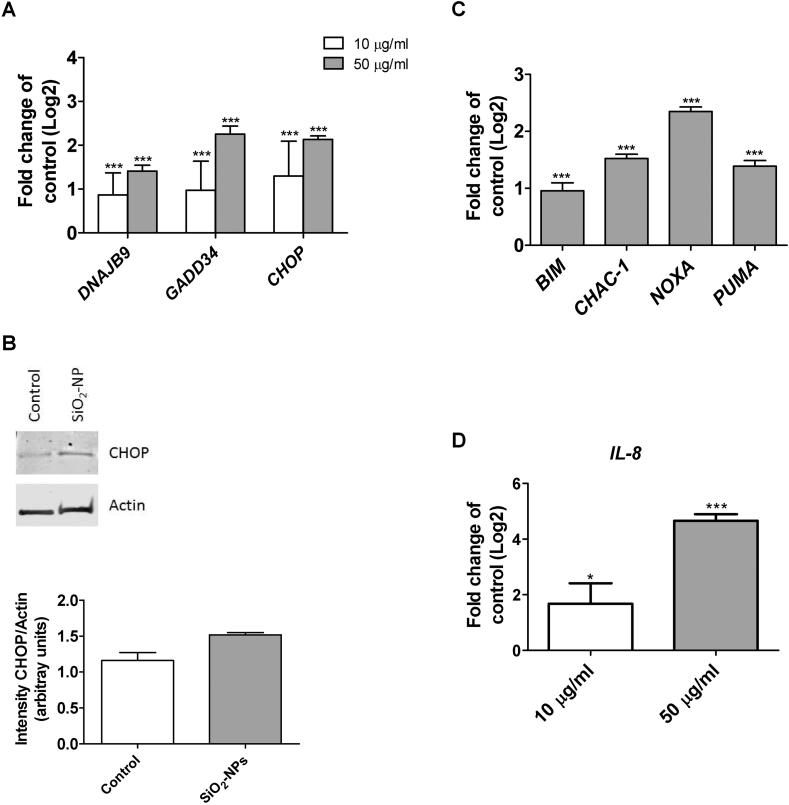
Exposure of Huh7 cells to SiO2-NP induced ER stress, as demonstrated by dose-related transcriptional induction of ER stress genes, DNAJB9, GADD34 and CHOP upon exposure to 10 and 50 μg/ml SiO2-NPs after 24 h (Fig. 1A). Additionally, CHOP protein was induced after 24 h exposure to 50 μg/ml SiO2-NPs (Fig. 1B).
Fig. 1.
Induction of ER stress and inflammation in response to SiO2-NP exposure of Huh7 cells. A: Abundance of DNAJB9, GADD34 and CHOP transcripts following exposure to 10 (white bars) and 50 μg/ml (grey bars) SiO2-NPs for 24 h. B: Huh7 cells were exposed to 50 μg/ml SiO2-NPs for 24 h followed by Western Blot analysis for CHOP and actin (above), and analysis of the Western Blot signal intensities (below). C: Expression of CHOP target genes BIM, CHAC-1, NOXA and PUMA in Huh7 cells following exposure to 50 μg/ml SiO2-NPs for 24 h D: Abundance of IL-8 transcript in Huh7 cells following exposure to 50 μg/ml SiO2-NPs for 24 h. Shown are results of three independent experiments +/−SEM. Significant differences are marked with one asterisk at p = 0.01–0.05, two asterisks at p = 0.001–0.01 and three asterisks at p = 0.0001–0.001.
As CHOP plays an important role in the induction of apoptosis under prolonged ER stress, we further investigated the expression of CHOP target genes. The transcripts of BIM, CHAC-1, NOXA and PUMA were significantly induced upon 24 h exposure to 50 μg/ml SiO2-NPs (Fig. 1C). In addition to ER stress, SiO2-NPs induced an inflammatory response by induction of TNF-α and IL-8; there was a dose-related induction of the IL-8 transcript after 24 h exposure to 10 and 50 μg/ml SiO2-NPs (Fig. 1D).
3.3. Activation of MAPK signalling
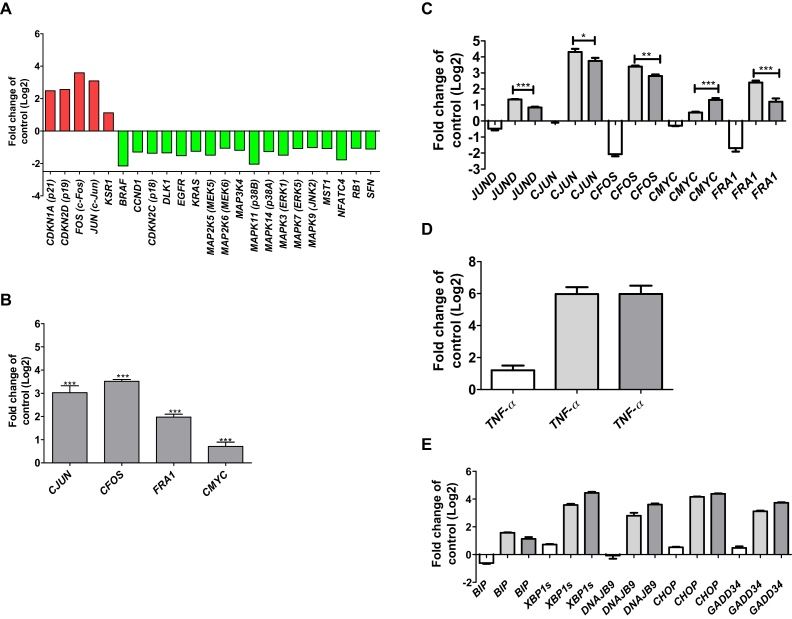
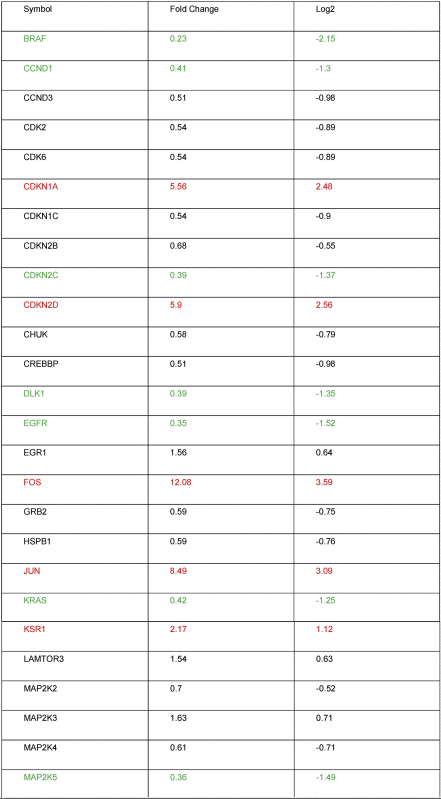
To investigate molecular effects of SiO2-NPs on the activation of MAP kinases, a MAPK signalling pathway PCR array was performed. Table 2 shows that five of the totally 84 analysed genes exhibited a significant transcriptional up-regulation, and 18 genes a significant down-regulation upon exposure of Huh7 cells to SiO2-NPs. Of the five up-regulated genes, P21 and P19 encode for inhibitors for the cyclin D and the CDK4/6 complex; CFOS and CJUN are down stream of ERK and JNK kinases, and KSR1 promotes MEK and RAF phosphorylation. Of the 18 down-regulated genes, strongest down-regulation occurred for the proto-oncogene BRAF, MAPK11, one of the four p38 MAPK genes, and for NFATC4, which is important for T-cell development (Fig. 2A).
Table 2.
Alteration of expressed genes from the MAPK array. Significantly up-regulated genes are shown in red, significantly down-regulated genes in green.
 |
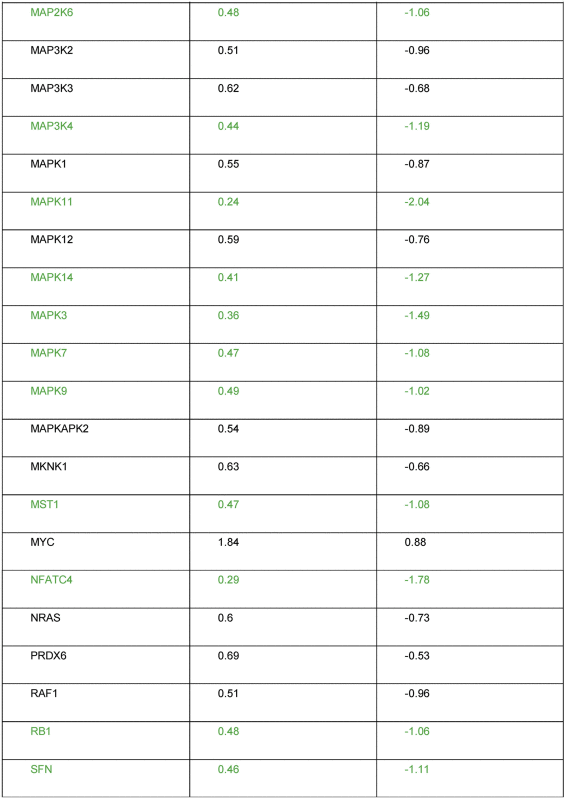 |
Fig. 2.
Activation of MAPK signalling in Huh7 cells in response to SiO2-NPs. A: Transcripts from the human MAP Kinase Signaling Pathway RT2 Profiler™ PCR Array showing significant up- and down-regulation in Huh7 cells following exposure to 50 μg/ml SiO2-NPs after 24 h. B: Abundance of CJUN, CFOS, FRA1 and CMYC transcripts in Huh7 cells following exposure to 50 μg/ml SiO2-NPs for 24 h C: Abundance of JUND, CJUN, CFOS, FRA1 and CMYC transcripts. D: Abundance of TNF-α transcript. E: Abundance of BIP, XBP-1s, DNAJB9, CHOP and GADD34 transcripts in Huh7 cells following co-exposure to 50 μg/ml SiO2-NPs and 25 μM JNK inhibitor SP600125 for 24 h. Key for C, D and E: white bars: 25 μM SP600; light grey bars: 50 μg/ml SiO2-NPs; dark grey bars: 25 μM SP600 and 50 μg/ml SiO2-NPs. Shown are results of three independent experiments +/−SEM. Significant differences are marked with one asterisk at p = 0.01–0.05, two asterisks at p = 0.001–0.01 and three asterisks at p = 0.0001–0.001.
To verify the results of the PCR array, transcriptional expression of CFOS and CJUN was assessed after SiO2-NP exposure. There was a strong induction of both genes after exposure to 50 μg/ml SiO2-NPs for 24 h (Fig. 2B). In addition, the expression of another downstream target of MAPKs was assessed. FRA1 showed an up-regulation upon SiO2-NP exposure (Fig. 2B). Compared to the other three analysed MAPK targets, CMYC showed a significant but weaker up-regulation (Fig. 2B).
To analyse for the effects driven by MAPK activation, Huh7 cells were co-exposed to 50 μg/ml SiO2-NPs and to 25 μM of the JNK inhibitor SP600125 for 24 h. There was a significant down-regulation of the JUND, CJUN, CFOS and FRA1 transcripts. In contrast, CMYC transcript showed a significant up-regulation (Fig. 2C). The partial inhibition of the MAPK signalling had no effect on the expression of the TNF-α transcript (Fig. 2D). There seems to be no direct link between MAPKs and ER stress. The up-regulation of BIP, XBP1s, DNAJB9, CHOP and GADD34 transcripts in response to SiO2-NPs was not significantly changed, when partially inhibiting the MAPK signalling (Fig. 2E).
3.4. Effects driven by SiO2-NPs induced TNF-α
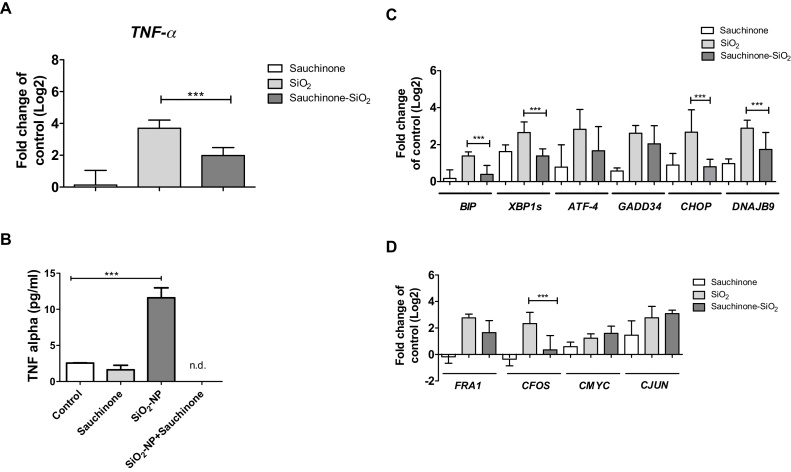
To analyse for the effects derived from up-regulation of TNF-α, Huh7 cells were exposed to SiO2-NPs in the presence of the TNF-α inhibitor sauchinone. Sauchinone reduced the induction of the TNF-α transcript upon SiO2-NP exposure by about 50% (Fig. 3A). On protein level, however, no TNF-α was detectable after co-exposure of Huh7 cells to SiO2-NPs and sauchinone (Fig. 3B). Furthermore, to address the question, whether SiO2-NPs induced ER stress, and in turn TNF-α, or vice versa, the expression of ER stress genes upon co-exposure of Huh7 cells to SiO2-NPs and sauchinone was assessed. The reduction in the TNF-α level lead to a significant reduction of transcripts of ER stress genes BIP, XBP–1s and DNAJB9 (Fig. 3C). Furthermore, transcripts of ATF-4, GADD34 and CHOP were lowered in the presence of sauchinone (Fig. 3C).
Fig. 3.
Alterations in gene expression driven by TNF-α. Abundance of TNF-α transcript (A), and TNF-α protein (B), of BIP, XBP1s, ATF-4, GADD34, CHOP and DNAJB9 transcripts (C), and FRA1, CFOS, CMYC and CJUN transcripts (D) in Huh7 cells following co-exposure to 50 μg/ml SiO2-NPs and 25 μM sauchinone for 24 h. Shown are results of three independent experiments +/- SEM. Significant differences are marked with one asterisk at p = 0.01–0.05, two asterisks at p = 0.001–0.01 and three asterisks at p = 0.0001–0.001.
The effects of TNF-α on MAPK expression were investigated by analysing the expression of FRA1, CFOS, CMYC and CJUN. The transcript of CFOS was significantly reduced in the presence of sauchinone, the other three analysed transcripts did not show any significant changes (Fig. 3D).
4. Discussion
4.1. Induction of ER stress response
In the present study, we analysed in detail the induction of ER stress and evaluated the consequences of the induction of TNF-α and activation of MAPK signalling pathway by SiO2-NPs at exposure concentrations of 10 and 50 μg/ml. Based on a Dutch food consumption survey [17], the daily up-take of amorphous SiO2-NPs as food additives was estimated to be 9.4 mg per kg body weight [18]. This would make a daily uptake of 658 mg SiO2-NPs for a 70 kg weight man. In vitro systems mimicking human gastric digestion showed that 80% of orally ingested SiO2-NPs withstand gastric dissolution and display still nano-size range [19]. Based on these data, our effects at 10 and 50 μg/ml SiO2-NPs are relevant regarding human health issues. In previous studies, we demonstrated the induction of various ER stress markers in Huh7 cells upon exposure to SiO2-NPs [8], [9].
The induction of ER stress is also known for other nanoparticles. Titanium dioxide nanoparticles induced ER stress and inflammation in the lung of mice after inhalation [20]. Silver nanoparticles induced ER stress in different cell lines and in lung, liver and kidneys of mice after intratracheal instillation [21]. Zinc oxide nanoparticles induced ER stress in liver tissue of mice [22]. The induction of ER stress by zinc oxide nanoparticles was also observed in vitro and can be regarded as an early biomarker for nanoparticle toxicity [23]. These findings suggest that the induction of ER stress is a mode of action of different nanoparticles.
In addition to the common ER stress markers BIP and XBP-1s, here we also analysed the abundance of the DNAJB9 and GADD34 transcripts. DNAJB9 is a chaperone that is involved in the ER-associated degradation (ERAD) of misfolded proteins. GADD34 plays a role in dephosphorylation of the translation initiation factor eIF-2α and is a promoter of apoptosis and CHOP, a gene which plays a role in apoptosis. Exposure of Huh7 cells to SiO2-NP resulted in a strong induction of GADD34 and CHOP, and a weaker but significant induction of DNAJB9 (Fig. 1A). GADD34 is a very interesting protein as it plays a role in the induction or promotion of various cancers, including colon cancer in mice by induction of pro inflammatory mediators like TNF-α, and by induction of epithelial proliferation [24]. Up-regulation of GADD34 was noted in the liver of mice exposed to diethylnitrosamine. It induced the up-regulation of pro inflammatory cytokines and expression of the oncogene C-MYC leading to the development of hepatocellular cancer [25].
In general, it is still unclear, whether ER stress induces TNF-α expression, or whether TNF-α induces ER stress. In mouse embryonic fibroblasts, ER stress induced TNF-α [26]. In another study, ER stress induced IRE1α activation regulated the production of inflammatory cytokines, such as TNF-α [27]. Exposure of bone marrow stem cells to alcohol induced up-regulation of BIP, ATF-4, CHOP and TNF-α. Simultaneous silencing of ATF-4 and CHOP prevented the induction of TNF-α [28]. On the other hand, induction of the unfolded protein response upon TNF-α exposure was shown in murine fibrosarcoma cells [29].
The induction of a pro-inflammatory response by SiO2-NPs was shown in human lung cells and murine macrophages, where a strong transcriptional induction of the pro-inflammatory cytokines TNF-α, IL-6 and IL-8 was noted [6]. Another link between ER stress and induction of inflammatory cytokines is the activation of intracellular NOD receptors, normally activated by binding to bacterial structures, through ER stress, followed by induction of inflammatory cytokines. The increased activity of the enzyme IRE1α in response to ER stress, leads to the formation of a complex between the TRAF2, NOD1 and NOD2, initiating inflammation through RIP2 activation [30].
In our study, we aimed to clarify this question by exposure of Huh7 cells to SiO2-NPs and the TNF-α inhibitor sauchinone. Indeed, sauchinone reduced the up-regulation of TNF-α mRNA upon SiO2-NP exposure about 50% (Fig. 3A) and eliminated the TNF-α protein (Fig. 3B). As a consequence, the induction of the ER stress marker gene transcripts were significantly reduced (Fig. 3B). This leads to the conclusion that SiO2-NPs first led to induction of TNF-α, and subsequently to induction of ER stress.
4.2. Activation of MAPK signalling
To investigate the effect of SiO2-NPs on the MAPK signalling pathway, the expression of 84 different MAPK signalling related genes was assessed. Strong up-regulation was found for CDKN1A/P21, CDKN2D/P19, CFOS and CJUN, and a weaker up-regulation of KSR1 (Fig. 2A). This strong induction of MAPK target genes gives a strong hint that the MAP kinases are activated upon SiO2-NP exposure even if the phosphorylation of the MAP kinases was not assessed. CDKN1A/P21 is a tumor suppressor gene involved in the regulation of cell cycle progression in the G1 phase. It also plays a crucial role in the development of hepatocellular cancer. Hepatitis B virus X protein (HBx) induces hepatocarcinogenesis by induction of P21 overexpression [31].
CJUN plays a role in several cellular processes including proliferation, cell survival and cell transformation [32], [33]. In human hepatocellular cancer CJUN is activated suggesting an important oncogenic function [34]. CJUN prevents apoptosis by antagonizing p53 activity, and thus contributes to the early stage of human hepatocellular cancer development [35], or liver tumor initiation [36]. In human liver biopsies from hepatocellular carcinoma patients infected with hepatitis C virus, protein kinase R lead to an overexpression of CFOS and CJUN promoting proliferation of hepatocellular carcinoma [37].
In our study, SiO2-NPs led to a strong induction of CJUN transcript (Figs. 2A, B). In the presence of the JNK inhibitor SP600125, induction was significant repressed. Furthermore, the expression of CFOS and FRA1 was significantly repressed by SP600125 (Fig. 2C). The inhibition of JNK had no effects on the abundance of the TNF-α transcript and on the activation of ER stress (Figs. 2D and E). This leads to the conclusion that these two events are up-stream of the MAPK activation upon SiO2-NP exposure.
The inhibition of TNF-α by sauchinone had a weaker effect on the activation of MAPKs. Only CFOS was significantly reduced (Fig. 3D). We hypothesize that the induction of ER stress is responsible for the activation of the MAPKs. To further clarify this point, experiments with TNF-α blocking antibodies should be done in future experiments. It is known that ER stress can activate MAPKs, and as a consequence, induce the activation of the AP-1 complex [38]. Consequently, ER stress may be involved in chronic liver diseases.
SiO2-NPs were shown in different cells to induce reactive oxygen species (ROS) and oxidative stress. We previously demonstrated this reaction in Huh7 cells in response to SiO2-NPs coated with 1% silver [8]. Induction of ROS and oxidative stress was also found in kidney cells [1], human lung cells [5], murine lung epithelial cells [39], and murine macrophages [40]. As oxidative stress can lead to the induction of ER stress [41], induction of oxidative stress by SiO2-NPs could be an additional mechanisms responsible for the activation of ER stress.
5. Conclusions
Our data show a clear induction of an inflammatory response by IL-8 and TNF-α induction upon SiO2-NP exposure. One of the consequences of elevated TNF-α levels is the induction of ER stress, which in turn can activate the MAPKs leading to the activation of the AP-1 complex. More research is needed to analyse the link between SiO2-NP exposure, induction of chronic inflammation, induction of ER stress, and induction of MAPK in relation to health impacts.
Acknowledgement
We thank Thamar Looser, FHNW, for her Master thesis contribution.
References
- 1.Passagne I., Morille M., Rousset M., Pujalté I., L'azou B. Implication of oxidative stress in size-dependent toxicity of silica nanoparticles in kidney cells. Toxicology. 2012;299(2–3):112–124. doi: 10.1016/j.tox.2012.05.010. [DOI] [PubMed] [Google Scholar]
- 2.Napierska D., Thomassen L.C., Lison D., Martens J.A., Hoet P.H. The nanosilica hazard: another variable entity. Part. Fibre Toxicol. 2010;7(1):39–71. doi: 10.1186/1743-8977-7-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ariano P., Zamburlin P., Gilardino A., Mortera R., Onida B., Tomatis M., Ghiazza M., Fubini B., Lovisolo D. Interaction of spherical silica nanoparticles with neuronal cells: size dependent toxicity and perturbation of calcium homeostasis. Small. 2011;7(6):766–774. doi: 10.1002/smll.201002287. [DOI] [PubMed] [Google Scholar]
- 4.Tarantini A., Lanceleur R., Mourot A., Lavault M.T., Casterou G., Jarry G., Hogeveen K., Fessard V. Toxicity, genotoxicity and proinflammatory effects of amorphous nanosilica in the human intestinal Caco-2 cell line. Toxicol. In Vitro. 2015;29(2):398–407. doi: 10.1016/j.tiv.2014.10.023. [DOI] [PubMed] [Google Scholar]
- 5.Sahu D., Vijayaraghavan R., Kannan G.M. Silica nanoparticle induces oxidative stress and provokes inflammation in human lung cells. Experiment. Nanosci. 2015;10(13):983–1000. [Google Scholar]
- 6.Panas A., Marquardt C., Nalcaci O., Bockhorn H., Baumann W., Paur H.R., Mülhopt S., Diabaté S., Weiss C. Screening of different metal oxide nanoparticles reveals selective toxicity and inflammatory potential of silica nanoparticles in lung epithelial cells and macrophages. Nanotoxicology. 2013;7(3):259–273. doi: 10.3109/17435390.2011.652206. [DOI] [PubMed] [Google Scholar]
- 7.Brown D.M., Kanase N., Gaiser B., Johnston H., Stone V. Inflammation and gene expression in the rat lung after instillation of silica nanoparticles: effect of size, dispersion medium and particle surface charge. Toxicol. Lett. 2014;224(1):147–156. [PubMed] [Google Scholar]
- 8.Christen V., Fent K. Silica nanoparticles and silver-doped silica nanoparticles induce endoplasmatic reticulum stress response and alter cytochrome P4501A activity. Chemosphere. 2012;87(4):423–434. doi: 10.1016/j.chemosphere.2011.12.046. [DOI] [PubMed] [Google Scholar]
- 9.Christen V., Camenzid M., Fent K. Silica nanoparticles induce endoplasmic reticulum stress response, oxidative stress and activate the mitogen-activated protein kinase (MAPK) signaling pathway. Toxicol. Rep. 2014;1:1143–1151. doi: 10.1016/j.toxrep.2014.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harding H.P., Calfon M., Urano F., Novoa I., Ron D. Transcriptional and translational control in the Mammalian unfolded protein response. Annu. Rev. Cell Dev. Biol. 2002;18:575–599. doi: 10.1146/annurev.cellbio.18.011402.160624. [DOI] [PubMed] [Google Scholar]
- 11.Rutkowski D.T., Kaufman R.J. A trip to the ER: coping with stress. Trends Cell. Biol. 2004;14:20–28. doi: 10.1016/j.tcb.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 12.Dara L., Ji C., Kaplowitz N. The contribution of ER stress to liver diseases. Hepatology. 2011;53:1752–1763. doi: 10.1002/hep.24279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lin W.C., Chuang Y.C., Chang Y.S., Lai M.D., Teng Y.N., Su I.J. Endoplasmic reticulum stress stimulates p53 expression through NF-κB activation. PLoS One. 2012;7:e39120. doi: 10.1371/journal.pone.0039120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Akira S., Uematsu S., Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 15.DiDonato J.A., Mercurio F., Karin M. NFκB and the link between inflammation and cancer. Immunol. Rev. 2012;246:379–400. doi: 10.1111/j.1600-065X.2012.01099.x. [DOI] [PubMed] [Google Scholar]
- 16.Duong F.H., Filipowicz M., Tripodi M., La Monica N., Heim M.H. Hepatitis C virus inhibits interferon signaling through up-regulation of protein phosphatase 2A. Gastroenterology. 2004;126(1):263–277. doi: 10.1053/j.gastro.2003.10.076. [DOI] [PubMed] [Google Scholar]
- 17.C. Kistenmaker, M. Bouman, K. Hulshof, Consumption of separate products by Dutch population groups-Dutch national food consumption survey 1997–1998. Zeist, TNO-Nutrition Food Res Institute, TNO-Report, 98, 812, (1998).
- 18.Winkler H.C., Suter M., Naegeli H. Critical review of the safety assessment of nano-structured silica additives in food. Nanobiotechnology. 2016;14:44. doi: 10.1186/s12951-016-0189-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Peters R., Kramer E., Oomen A.G., Herrera Rivera Z.E., Oegema G., Tromp P.C. Presence of nano-sized silica during in vitro digestion of foods containing silica as a food additive. ACS Nano. 2012;6:2441–2451. doi: 10.1021/nn204728k. [DOI] [PubMed] [Google Scholar]
- 20.Yu K.N., Sung J.H., Lee S., Kim J.E., Kim S., Cho W.Y., Lee A.Y., Park S.J., Lim J., Park C., Chae C., Lee J.K., Lee J., Kim J.S., Cho M.H. Inhalation of titanium dioxide induces endoplasmic reticulum stress-mediated autophagy and inflammation in mice. Food. Chem. Toxicol. 2015;85:106–113. doi: 10.1016/j.fct.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 21.Huo L., Chen R., Zhao L., Shi X., Bai R., Long D., Chen F., Zhao Y., Chang Y.Z., Chen C. Silver nanoparticles activate endoplasmic reticulum stress signaling pathway in cell and mouse models: the role in toxicity evaluation. Biomaterials. 2015;61:307–315. doi: 10.1016/j.biomaterials.2015.05.029. [DOI] [PubMed] [Google Scholar]
- 22.Yang X., Shao H., Liu W., Gu W., Shu X., Mo Y., Chen X., Zhang Q., Jiang M. Endoplasmic reticulum stress and oxidative stress are involved in ZnO nanoparticle-induced hepatoxicity. Toxicol. Lett. 2015;234(1):40–49. doi: 10.1016/j.toxlet.2015.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen R., Huo L., Shi X., Bai R., Zhang Z., Zhao Y., Chang Y., Chen C. Endoplasmic reticulum stress induced by zinc oxide nanoparticles is an earlier biomarker for nanotoxicological evaluation. ACS Nano. 2014;8(3):2562–2574. doi: 10.1021/nn406184r. [DOI] [PubMed] [Google Scholar]
- 24.Tanaka Y., Ito S., Oshino R., Chen N., Nishio N., Isobe K. Effects of growth arrest and DNA damage-inducible protein 34 (GADD34) on inflammation-induced colon cancer in mice. Br. J. Cancer. 2015;113(4):669–679. doi: 10.1038/bjc.2015.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen N., Nishio N., Ito S., Tanaka Y., Sun Y., Isobe K. Growth arrest and DNA damage-inducible protein (GADD34) enhanced liver inflammation and tumorigenesis in a diethyl-nitrosamine (DEN)-treated murine model. Cancer Immunol. Immunother. 2015;64(6):777–789. doi: 10.1007/s00262-015-1690-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu P., Han Z., Couvillon A.D., Kaufman R.J., Exton J.H. Autocrine tumor necrosis factor alpha links endoplasmic reticulum stress to the membrane death receptor pathway through IRE1α-mediated NF-κB activation and down-regulation of TRAF2 expression. Mol. Cell. Biol. 2006;26(8):3071–3084. doi: 10.1128/MCB.26.8.3071-3084.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim S., Joe Y., Kim H.J., Kim Y.S., Jeong S.O., Pae H.O., Ryter S.W., Surh Y.J., Chung H.T. Endoplasmic reticulum stress-induced IRE1α activation mediates cross-talk of GSK-3β and XBP-1 to regulate inflammatory cytokine production. J. Immunol. 2015;194(9):4498–4506. doi: 10.4049/jimmunol.1401399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen Y., Gao H., Yin Q., Chen L., Dong P., Zhang X., Kang J. ER stress activating ATF4/CHOP-TNF-α signaling pathway contributes to alcohol-induced disruption of osteogenic lineage of multipotential mesenchymal stem cell. Cell. Physiol. Biochem. 2013;32(3):743–754. doi: 10.1159/000354476. [DOI] [PubMed] [Google Scholar]
- 29.Xue X., Piao J.H., Nakajima A., Sakon-Komazawa S., Kojima Y., Mori K., Yagita H., Okumura K., Harding H., Nakano H. Tumor necrosis factor alpha (TNFalpha) induces the unfolded protein response (UPR) in a reactive oxygen species (ROS)-dependent fashion, and the UPR counteracts ROS accumulation by TNFalpha. J. Biol. Chem. 2005;280(40):33917–33925. doi: 10.1074/jbc.M505818200. [DOI] [PubMed] [Google Scholar]
- 30.Keestra-Gounder A.M., Byndloss M.X., Seyffert N., Young B.M., Chávez-Arroyo A., Tsai A.Y., Cevallos S.A., Winter M.G., Pham O.H., Tiffany C.R., de Jong M.F., Kerrinnes T., Ravindran R., Luciw P.A., McSorley S.J., Bäumler A.J., Tsolis R.M. NOD1 and NOD2 signalling links ER stress with inflammation. Nature. 2016;532(7599):394–397. doi: 10.1038/nature17631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yano M., Ohkoshi S., Aoki Y.H., Takahashi H., Kurita S., Yamazaki K., Suzuki K., i S. Yamagiwa, Sanpei A., Fujimaki S., Wakai T., Kudo S.E., Matsuda Y., Aoyagi Y. Hepatitis B virus X induces cell proliferation in the hepatocarcinogenesis via up-regulation of cytoplasmic p21 expression. Liver Int. 2013;33(8):1218–1229. doi: 10.1111/liv.12176. [DOI] [PubMed] [Google Scholar]
- 32.Jochum W., Passegué E., Wagner E.F. AP-1 in mouse development and tumorigenesis. Oncogene. 2001;20(19):2401–2412. doi: 10.1038/sj.onc.1204389. [DOI] [PubMed] [Google Scholar]
- 33.Vogt P.K. Jun, the oncoprotein. Oncogene. 2001;20(19):2365–2377. doi: 10.1038/sj.onc.1204443. [DOI] [PubMed] [Google Scholar]
- 34.Yuen M.F., Wu P.C., Lai V.C., Lau J.Y., Lai C.L. Expression of c-Myc, c-Fos, and c-jun in hepatocellular carcinoma. Cancer. 2001;91(1):106–112. doi: 10.1002/1097-0142(20010101)91:1<106::aid-cncr14>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 35.Eferl R., Ricci R., Kenner L., Zenz R., David J.P., Rath M., Wagner E.F. Liver tumor development. c-Jun antagonizes the proapoptotic activity of p53. Cell. 2003;112(2):181–192. doi: 10.1016/s0092-8674(03)00042-4. [DOI] [PubMed] [Google Scholar]
- 36.Min L., Ji Y., Bakiri L., Qiu Z., Cen J., Chen X., Chen L., Scheuch H., Zheng H., Qin L., Zatloukal K., Hui L., Wagner E.F. Liver cancer initiation is controlled by AP-1 through SIRT6-dependent inhibition of surviving. Nat. Cell Biol. 2012;11:1203–1211. doi: 10.1038/ncb2590. [DOI] [PubMed] [Google Scholar]
- 37.Watanabe T., Hiasa Y., Tokumoto Y., Hirooka M., Abe M., Ikeda Y., Matsuura B., Chung R.T., Onji M. Protein kinase R modulates c-Fos and c-Jun signaling to promote proliferation of hepatocellular carcinoma with hepatitis C virus infection. PLoS One. 2013;8(7):e67750. doi: 10.1371/journal.pone.0067750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Olivares S., Green R.M., Henkel A.S. Endoplasmic reticulum stress activates the hepatic activator protein 1 complex via mitogen activated protein kinase-dependent signaling pathways. PLoS One. 2014;9(7):e103828. doi: 10.1371/journal.pone.0103828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Decan N., Wu D., Williams A., Bernatchez S., Johnston M., Hill M., Halappanavar S. Characterization of in vitro genotoxic, cytotoxic and transcriptomic responses following exposures to amorphous silica of different sizes. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2016;796:8–22. doi: 10.1016/j.mrgentox.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 40.Lehman S.E., Morris A.S., Mueller P.S., Salem A.K., Grassian V.H., Larsen S.C. Silicananoparticle-generated ROS as a predictor of cellular toxicity: mechanistic insights and safety by design. Environ. Sci. Nano. 2016;3(1):56–66. doi: 10.1039/C5EN00179J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pallepati P., Averill-Bates D.A. Activation of ER stress and apoptosis by hydrogen per-oxide in HeLa cells: protective role of mild heat preconditioning at 40°C. Biochim. Biophys. Acta. 2011;1813(12):1987–1999. doi: 10.1016/j.bbamcr.2011.07.021. [DOI] [PubMed] [Google Scholar]