Abstract
Background & Aims
Mucosal healing, determined by histologic analysis, is a potential therapeutic target for patients with ulcerative colitis (UC). However, the histologic features of tissue normalization, as an outcome of treatment, have not been well described. We examined the prevalence and predictive values of normalization of the colonic mucosa, based on histologic analysis (histologic normalization) in patients with UC, and determined its association with risk of clinical relapse, compared with histologic disease quiescence and endoscopic mucosal healing.
Methods
We performed a retrospective study of 646 patients with confirmed UC who underwent colonoscopy at a tertiary medical center from August 2005 through October 2013. We reviewed reports from pathology analyses of random mucosal biopsies from each colon segment, and categorized them into 3 groups based on histology findings: (1) normalization (completely normal mucosa with no features of chronicity present), (2) quiescence (crypt atrophy or branching without signs of active inflammation including erosions, abscesses, or focal neutrophil infiltration), or (3) active disease (epithelial infiltration by neutrophils, crypt abscesses, erosions, or ulceration). Histology findings were compared with clinical and endoscopic findings. We assessed variables associated with histology findings and, in patients in clinical remission (Simple Clinical Colitis Activity Index score ≤2 and subscore of ≤1 for stool frequency or rectal bleeding), predictive values for clinical relapse at follow-up evaluations 6 months later or more were calculated.
Results
Of the 646 patients included in the study, 60% had endoscopic mucosal healing, 40% had histologic quiescence, and 10% had histologic normalization. The level of agreement between mucosal and histologic activity was moderate (agreement for 68% of samples; k = 0.50; P < .001). On multivariate analysis, only proctitis associated with histologic normalization (P = .002). Of 310 patients in clinical remission at initial review, 25% had a clinical relapse, after a median time of 16 months (interquartile range, 10–23 months). Histologic normalization was independently associated with increased odds of relapse-free survival compared with histologic quiescence (hazard ratio, 4.31; 95% confidence interval, 1.48–12.46; P = .007) and histologic activity (hazard ratio, 6.69; 95% confidence interval, 2.16–20.62; P = .001); mucosal healing was not associated with increased odds of relapse-free survival compared with no mucosal healing (hazard ratio, 1.02; 95% confidence interval, 0.56–1.85; P = .954).
Conclusions
Histologic normalization of colonic mucosa can be used as a clinical endpoint for patients with UC. We associated histologic normalization with increased odds of relapse-free survival compared to endoscopic healing or histologic quiescence. Further studies are needed to determine whether histologic normalization should be a goal of treatment for patients with UC.
Keywords: histopathology, mucosal healing, inflammatory bowel disease, normalization
INTRODUCTION
Ulcerative colitis (UC) is a chronic inflammatory disease characterized by periods of disease activity alternating with periods of quiescence. The primary treatment goal in UC has been to limit these periods of activity and maintain clinical remission, traditionally defined as cessation of rectal bleeding and normalization of stool frequency.1 In recent years endoscopic mucosal healing (MH) in UC has been associated with improved clinical outcomes compared to achieving clinical remission alone.2–5 In fact, MH has been associated with prolonged remission, fewer hospitalizations and colectomies and an improved quality of life.2–8
Despite such improved outcomes associated with MH, up to 40% of patients with MH have persistent histologic inflammatory activity.3,9–12 Therefore, there is interest in the significance of histologic remission, as a “deeper” marker of disease control compared with MH. Although more research is needed into the practicality and validity of histologic assessment, it is known that reduced histologic activity is associated with decreased risks of relapse, hospitalization, corticosteroid use, colectomy, and colorectal neoplasia.3,9–18
It has been a common understanding that, following a diagnosis of UC, histologic abnormalities of the mucosa persist. This idea has been so established that when histologic grading has been described in UC, normalization has not generally been defined as an outcome that is distinct from quiescence; and histologic healing is generally described simply as absence of active inflammation.3,9,14,19 Furthermore, in 2 recently described histologic indices, the focus has been on short-term treatment responsiveness and architectural abnormalities have been excluded in their assessment.20,21
Although there are infrequent descriptions of histologic normalization in the literature,22 it remains incompletely defined in the context of clinical outcomes. In addition, patient, disease, and treatment characteristics associated with histologic normalization have not been studied, and it had not been determined if complete histologic normalization (CHN) is associated with improved clinical outcomes compared to histologic remission or endoscopic MH alone.
The aims of this study were to examine the prevalence and predictors of histologic normalization in patients with confirmed UC, and to determine if normalization is associated with improved clinical outcomes compared with ongoing histologic activity, histologic quiescence, and endoscopic MH.
METHODS
A retrospective case-control study was performed and approved by the institutional review board (13-1063). All patients who underwent colonoscopy at University of Chicago between August 2005 and October 2013 for UC were identified by 1 or more of the International Classification of Diseases, Ninth revision, Clinical Modification codes for UC (556.0, 556.2, 556.3, 556.5, 556.6, 556.8, 556.9). Patients were eligible for inclusion if they had an established diagnosis of UC at the time of this “follow-up” colonoscopy and documentation of previous complete colonoscopy and segmental biopsies obtained in at least the rectum, left colon, and right colon that showed chronic changes (with or without acute changes) consistent with a histologic diagnosis of UC more than 1 year prior. Patients who had inadequate documentation, had undergone a colectomy, or had confirmed Clostridium difficile infection at time of follow-up colonoscopy were excluded.
Medical Records Abstraction
Endoscopy reports were retrieved through the electronic documentation system for endoscopic reports (Provation, Minneapolis, MN). Demographic, clinical, histologic, and biochemical data were collected from our electronic medical record system (EPIC, Verona, WI), including age of diagnosis, disease duration, smoking history, and previous and current use of anti-inflammatory agents and/or immunosuppressant therapy (steroids, immunomodulators, anti–tumor necrosis factor agents) at time of follow-up colonoscopy.
Endoscopic and Histologic Assessment
The bowel was divided into 3 segments as per the Montreal classification for UC23: (1) proctitis (E1, rectum only), (2) left-sided (E2, rectum to splenic flexure), or (3) extensive colitis (E3, disease proximal to splenic flexure). Disease extent was determined using a modified Montreal classification in that, rather than endoscopic appearance, histology was used on most proximal biopsy showing evidence of disease, whether acute or chronic inflammation, or chronic architectural changes (ie, crypt branching/shortening, decreased crypt densities, and irregular mucosal surfaces). Maximal disease extent was determined at any colonoscopy performed over the patient’s history. The endoscopic and histologic severity and number of biopsies taken from each segment at follow-up endoscopy were recorded.
Endoscopic Mucosal Assessment
An academic inflammatory bowel disease (IBD) expert gastroenterologist with minimum 5 years’ experience performed all endoscopies, during which endoscopic photographs were obtained from each segment of bowel, with targeted photographs of areas of mucosal activity. An independent reviewer classified patients into 3 distinct groups of endoscopic grade of inflammation using the endoscopic subscore of the Modified Mayo Disease Activity Index.24 A score of 0 (no friability, granularity, and intact vascular pattern) was classified as normal, 1 (mild erythema or decreased vascular pattern) as quiescent mucosa, and a score of ≥2 (any of moderate or marked erythema, absent vascular pattern, friability, erosions, ulceration, or contact/spontaneous bleeding) as mucosal activity. Endoscopic MH was defined by either completely normal or quiescent mucosa (Modified Mayo Disease Activity Index endoscopic subscore ≤ 1).
Histologic Assessment
As is routine in this unit, random mucosal biopsies were obtained from each colon segment, targeting the area of most significant mucosal disease activity. Two pathologists who specialize in gastrointestinal histology routinely assess all biopsies and report histology using a standardized scale that includes histologically normal, quiescent, mild, moderate, or severe disease. We reviewed these pathologic reports and categorized histology specimens into 3 distinct groups using the modified Riley score as described by Bryant et al16, but with the subcategorization of histologic remission into histologic normalization and histologic quiescence based on the absence or presence of architectural changes, respectively. Based on the maximum inflammation score at each segment, patients were categorized as (1) histologic normalization: completely normal mucosa with no features of chronicity present; (2) histologic quiescence: features of chronicity including crypt atrophy or branching but no active inflammation, such as erosions, crypt abscesses, or focal neutrophil infiltration; and (3) histologic activity: presence of any epithelial infiltration by neutrophils, crypt abscesses, erosions or ulceration.
As previously reported, the inter-observer agreement for interpretation of UC histology between our pathologists using a 6-point scale that classifies varying severities of inflammatory activity (including normal) was very good (kappa=0.6).13 In this study, an additional 150 samples were re-graded by one of the expert pathologists (JH) who was blinded to the prior official reads of these specimens; 50 were histologically normal, 50 quiescent and 50 had active histology. All 150 samples were interpreted correctly (kappa=1.0).
The primary outcome of CHN was defined by normalization of mucosa without histologic features of chronicity in all bowel segments on follow-up colonoscopy in a patient with previous structural changes on biopsy consistent with UC.
Assessment of Clinical Relapse-Free Survival
Patients in clinical remission at follow-up colonoscopy with ≥ 6 months of follow-up at the University of Chicago from this colonoscopy until September 2014 were included in a separate analysis of clinical relapse-free survival. At each patient clinic visit, the Simple Clinical Colitis Activity Index was calculated.25 Clinical remission was defined as Simple Clinical Colitis Activity Index ≤2 and subscore of ≤ 1 for stool frequency or rectal bleeding as determined from physician records. Clinical relapse-free survival was defined as time from colonoscopy to period of clinical relapse, with clinical relapse defined at clinical follow-up as Simple Clinical Colitis Activity Index >2, subscore >1 for stool frequency or rectal bleeding, or medication escalation for symptoms, hospitalization for UC relapse, or colectomy for refractory UC.
Statistical Analysis
Continuous variables were summarized using medians and interquartile ranges. Categorical variables were expressed as percentage and number of cohort. Cohen kappa coefficient (k) was calculated to measure agreement between mucosal and histologic activity.
Univariate analysis of baseline characteristics was performed to identify predictive factors for CHN. The Mann-Whitney U test and analysis of variance were used to compare continuous variables, and Pearson’s chi-squared test was used to compare categorical variables. Multivariate analysis to identify independent factors associated with histological outcomes was performed using logistic regression.
Kaplan-Meier analysis was performed to compare clinical relapse-free survival in those with and without CHN and MH and log-rank statistics were performed to compare sub-groups of interest. Cox proportional hazard regression analysis was performed to identify independent predictors of clinical regression.
All variables with P < .20 on univariate analysis were retained and integrated into the multivariate models. A 2-sided P ≤ .05 was considered statistically significant. All data analyses were performed using Stata 12.0 (StataCorp, College Station, TX).
RESULTS
Patients
A total of 646 patients fulfilled the entry criteria and were included in the analysis. Baseline characteristics at time of follow-up colonoscopy are shown in Table 1. Using endoscopic criteria, 40% (n = 261) had endoscopic mucosal activity, 35% (n = 228) mucosal quiescence, and 24% (n = 157) mucosal normalization on follow-up colonoscopy. Using histologic criteria, 50% (n = 321) had ongoing activity, 40% (n = 260) histologic quiescence, and 10% (n = 65) had CHN.
Table 1.
Clinical Characteristics at Baseline
| Baseline characteristics (n = 646) | Median (IQR) or Percentage (n) | |
|---|---|---|
| Age at diagnosis of UC (years) | 29 (22–41) | |
| Gender (male) | 50.2% (n=324) | |
| Greatest disease extent seen: | ||
| Proctitis | 10.1% (n=65) | |
| Left-Sided | 30.5% (n=197) | |
| Pancolitis | 59.4% (n=384) | |
| Duration of disease (years) | 13 (7–22) | |
| Smoking status (current smoker) | 6.6% (n=41) | |
| Endoscopic mucosal disease: | ||
| Mucosal normalization | 157 (24.3%) | Mucosal healing:385 (59.6%) |
| Mucosal quiescence | 228 (35.3%) | |
| Mucosal activity | 261 (40.4%) | |
| Histologic disease: | ||
| Histologic normalization | 65 (10.1%) | Histologic healing: 325 (50 3%) |
| Histologic quiescence | 260 (40.2%) | |
| Histologic activity | 321 (49.7%) | |
| Medications, n(%) | ||
| Oral steroid exposure | 394 (66.8%) | |
| Current oral steroid | 54 (98.6%) | |
| 5-ASA exposure | 637 (99.5%) | |
| Current 5-ASA | 517 (81.6%) | |
| Immunomodulator exposure | 294 (48.1%) | |
| Current immunomodulator | 190 (30.1%) | |
| Previous cyclosporine salvage | 25 (4.2%) | |
| Anti-TNF exposure | 109 (18.1%) | |
| Current anti-TNF | 82 (13.0%) | |
The level of agreement between mucosal and histologic activity was moderate (68%; k = 0.50; P < .001). A total of 12% (19 of 157) of patients with mucosal normalization had histologic activity and 27% (61 of 228) of patients with mucosal quiescence had histologic activity. No patient (0 of 65) with histologic normalization had mucosal activity but 8% (20 of 260) of patients with histologic quiescence had mucosal activity.
Complete Histologic Normalization
A total of 10% (n = 65) of patients had complete normalization of their histology in all segments that had previously shown changes (ie, CHN). The mean number of biopsies taken at each endoscopy was 20 (standard deviation, 9.5) and the number of biopsies taken was not significantly different in patients achieving CHN and in those who did not (Supplementary Table 1).
CHN was identified in 9% (n = 35) of patients with extensive colitis, 8% (n = 15) with left-sided disease, and 23% (n = 15) with proctitis alone. By univariate analysis, CHN was associated with less extensive disease at baseline (P = .001), disease duration >10 years (P = .029), and negatively associated with previous steroid (P = .041) and anti–tumor necrosis factor therapy (P = .031) (Table 2). On multivariate analysis, a diagnosis of proctitis compared with left-sided (E2) (adjusted odds ratio, 3.63; 95% confidence interval, 1.56–8.46; P = .003) and extensive (E3) colitis (adjusted odds ratio, 2.81; 95% confidence interval, 1.32–5.96; P = .007) remained significantly and independently associated with CHN. There was a trend for patients with disease duration of more than 10 years to achieve CHN (adjusted odds ratio, 1.81; 95% confidence interval, 0.98–3.35; P = .058).
Table 2.
Univariate Analysis of Predictors of Complete Histologic Healing in UC
| Characteristic (n= 646) | CHN (n=65) | No CHN (n=581) | P value |
|---|---|---|---|
| Age, y, n (%) | |||
| ≤ 16 y | 11 (17%) | 49 (9%) | 0.071* |
| 17–39 y | 39 (61%) | 357 (63%) | |
| ≥ 40 y | 14 (22%) | 160 (28%) | |
| Gender, m, n(%) | 40 (46%) | 294 (51%) | 0.496 |
| Current smoker, n (%) | 5 (8%) | 36 (6%) | 0.666 |
| Disease extent at baseline, n (%) | |||
| E1 | 15 (23%) | 50 (9%) | 0.001** |
| E2 | 15 (23%) | 182 (31%) | |
| E3 | 35 (54%) | 349 (60%) | |
| Disease > 10 y, n (%) | 47 (73%) | 336 (59%) | 0.029** |
| Oral steroid Exposure, n (%) | 33 (55%) | 361 (68%) | 0.041** |
| 5-ASA monotherapy, n (%) | 40 (63%) | 291 (51%) | 0.082 |
| Previous immunomodulator, n (%) | 22 (36%) | 272 (50%) | 0.033** |
| Current immunomodulator, n (%) | 15 (23%) | 175 (31%) | 0.220 |
| Previous cyclosporine salvage, n (%) | 4 (7%) | 21 (4%) | 0.340 |
| Previous anti-TNF, n (%) | 5 (8%) | 104 (19%) | 0.031** |
| Current anti-TNF Rx, n (%) | 5 (8%) | 77 (14%) | 0.192* |
| Current immunomodulator and anti-TNF, n (%) | 4 (6%) | 26 (5%) | 0.555 |
Incorporated into multivariate analysis as p-value < 0.2
Significant p-value < 0.05
Clinical Relapse-Free Survival
A total of 310 patients who were in clinical remission at follow-up colonoscopy were assessed for clinical relapse-free survival. Baseline characteristics are shown in Table 3. Using endoscopic criteria, 25% (n = 80) had ongoing endoscopic mucosal activity, 41% (n = 127) quiescence, and 33% (n = 103) mucosal normalization. Using histologic criteria, 35% (n = 108) had histologic activity, 51% (n = 157) quiescence, and 15% (n = 45) CHN.
Table 3.
Baseline Characteristics of Those in Clinical Remission at Baseline
| Patients | N=310 |
|---|---|
| Sex, n (%), male | 159 (51.3%) |
| Age, median (IQR), y | 48.4 y (36.6–58.8) |
| Age of diagnosis, median (IQR), y | 29 y (22–41) |
| Active smoking status, n (%) | 21 (6.8%) |
| Duration of disease, median (IQR), y | 14 y (1–52 or 8–49) |
| Disease extent, n (%) | |
| E1 | 34 (11.0%) |
| E2 | 89 (28.7%) |
| E3 | 187 (60.3%) |
| Endoscopic mucosal disease activity, n(%) | |
| Endoscopic mucosal activity | 80 (25.0%) |
| Endoscopic mucosal quiescence | 127 (41.0%) |
| Endoscopic mucosal normalization | 103 (33.2%) |
| Histologic disease activity, n(%) | |
| Histologic activity | 108 (34.8%) |
| Histologic quiescence | 157 (50.7%) |
| Complete Histologic normalization | 45 (14.5%) |
| Medications, n (%) | |
| Past oral steroid exposure | 185 (66.3%) |
| Current oral steroids | 15 (4.9%) |
| Past 5-ASA exposure | 303 (99.3%) |
| Current 5-ASA therapy | 242 (79.3%) |
| Past IMM therapy exposure | 156 (52.9%) |
| Current Immunomodulator (IMM) therapy | 110 (36.2%) |
| Past anti-TNF therapy exposure | 57 (19.5%) |
| Current Anti-TNF therapy | 49 (16.2%) |
| Past cyclosporine salvage therapy | 12 (4.1%) |
Median follow-up was 22 (interquartile range, 14–34) months and 25% (n = 77) patients experienced clinical relapse at median time 16 (interquartile range, 10–23) months. Patients with CHN had lower rates of clinical relapse compared with those with histologic quiescence and activity (Figure 1A) and patients with endoscopic MH had lower rates of clinical relapse rates compared with those with mucosal activity (Figure 1B). In patients who had MH and were in clinical remission, histologic normalization remained protective against clinical relapse (Figure 1C).
Figure 1. Kaplan-Meier analysis of effect of endoscopic mucosal and histologic activity on clinical relapse-free survival.

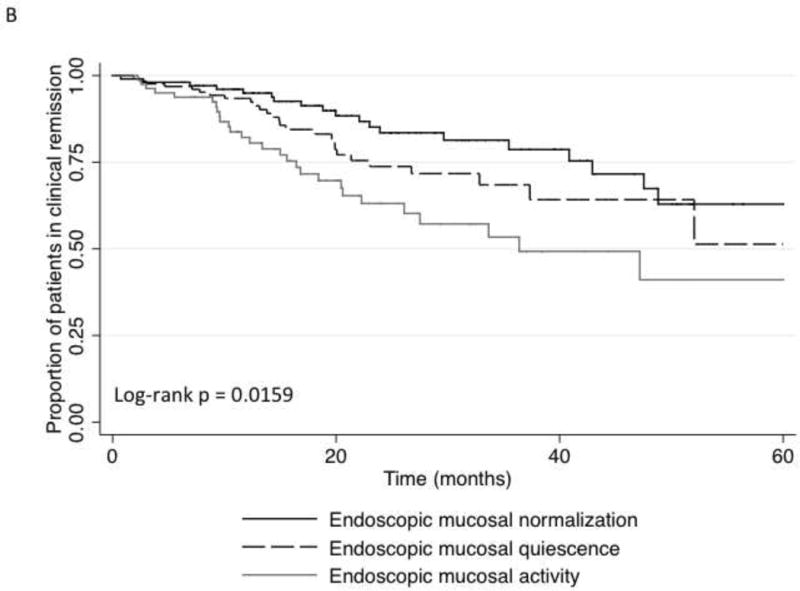
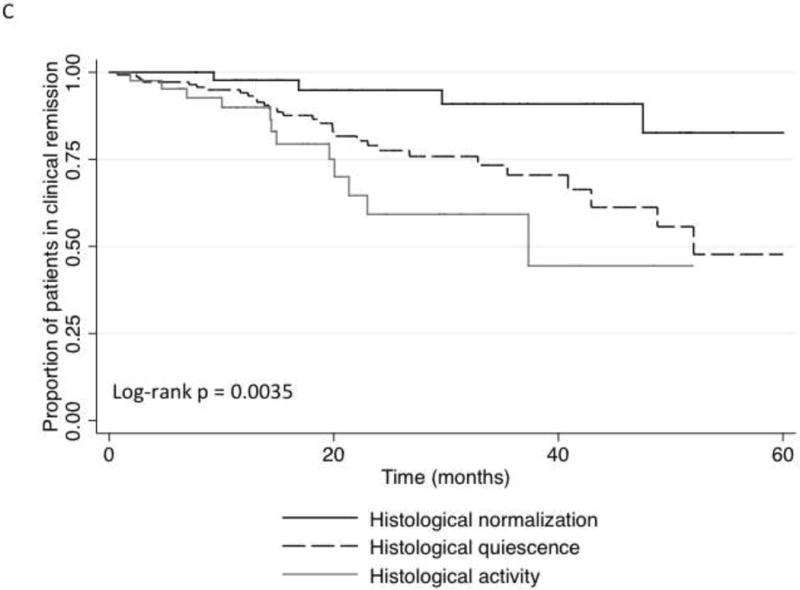
(A) Clinical relapse-free survival versus histologic healing. (B) Clinical relapse-free survival versus endoscopic mucosal healing. (C) Clinical relapse-free survival versus histologic healing in patients with endoscopic mucosal healing.
By univariate analysis, the only factors associated with improved clinical relapse-free survival were the achievement of endoscopic MH compared with no MH, and CHN compared with both histologic quiescence and histologic activity (Table 4). By multivariate analysis, only CHN compared with histologic quiescence (hazard ratio, 4.31 [1.48–12.46]; P = .007), CHN compared with histologic activity (hazard ratio, 6.69 [2.16–20.62]; P = .001), and no previous exposure to cyclosporine (P = .034) predicted clinical relapse-free survival (Table 4). MH was not independently associated with lower rate of clinical relapse.
Table 4.
Univariate and Multivariate Analysis For Factors Associated With Clinical- Relapse Free Survival
| Risk Factor | Univariate analysis: HRa (95% CI) | P valueb | Multivariate analysis: HRa (95% CI) | P valueb |
|---|---|---|---|---|
| Sex (Female) | 1.15 (0.74–1.81) | 0.533 | ||
| Older age at colonoscopy | 0.99 (0.97–1.00) | 0.152* | 0.99 (0.98–1.01) | 0.408 |
| Older age of diagnosis | 0.99 (0.98–1.01) | 0.436 | ||
| Current smoker | 1.19 (0.51 – 2.75) | 0.687 | ||
| Longer duration of disease | 0.99 (0.96–1.01) | 0.301 | ||
| Disease extent: | ||||
| E2 vs E1 | 1.33 (0.59–2.98) | 0.487 | ||
| E3 vs E1 | 1.41 (0.66–3.01) | 0.369 | ||
| Endoscopic Mucosal Healing: | ||||
| -Quiescent disease vs Normalization | 1.56 (0.87–2.76) | 0.131 | ||
| -Active disease vs Normalization | 2.44 (1.37–4.36) | 0.002 | ||
| No endoscopic mucosal healing | 1.93 (1.21 – 3.07) | 0.006** | 1.02 [0.56–1.85] | 0.954 |
| Histologic healing: | ||||
| -Quiescent disease vs Normalization | 3.79 (1.34 – 10.68) | 0.012** | 4.31 [1.48–12.46] | 0.007** |
| -Active disease vs Normalization | 6.76 (2.39 – 19.14) | < 0.001** | 6.69 [2.16–20.62] | 0.001** |
| oral steroids | 0.72 (0.23–2.30) | 0.582 | ||
| 5-ASA therapy | 1.19 (0.67–2.14) | 0.555 | ||
| anti-TNF therapy | 0.88 (0.44–1.78) | 0.724 | ||
| Dual therapy (IMM + anti-TNF) | 1.42 (0.61–3.27) | 0.416 | ||
| IMM therapy | 1.37 (0.86–2.17) | 0.183* | 1.21 (0.72–2.04) | 0.469 |
| Past anti-TNF therapy exposure | 0.81 (0.42–1.54) | 0.522 | ||
| Past IMM therapy exposure | 1.32 (0.83–2.09) | 0.236 | ||
| Past cyclosporine salvage therapy | 2.34 (0.99–5.52) | 0.052* | 2.71 [1.07–6.82] | 0.034** |
| Past oral steroid exposure | 1.08 (0.67–1.74) | 0.749 | ||
Hazard ratio for each risk factor in Cox model (estimate and 95% CI)
Significance level
significant P-value < 0.05)
Incorporated into multivariate analysis as P-value < 0.2
CI, confidence interval
DISCUSSION
We have demonstrated that histologic normalization of the colon in UC is possible and is characterized by statistically superior clinical relapse-free survival. A total of 1 in 10 of our cohort achieved CHN.
This is the first study to describe complete normalization of histology in UC. A study by Kleer and Appelman26 did demonstrate that areas of histologic chronic colitis became normal at some point in 22 of 41 patients (54%). However, this study only described the rate of normalization of a single point in the bowel, and did not look at complete normalization of the bowel or examine patient characteristics that predicted this normalization.
Most descriptions of histologic normalization have been reported as rectal sparing. Levine et al27 found that, of 24 asymptomatic patients with UC, 2 (8%) normalized their rectal biopsy on follow-up. Odze et al22 looked at 14 patients treated with either mesalamine or placebo enemas and found that, in patients on mesalamine rectal therapy, 36% of rectal biopsies normalized (defined as “complete absence” of any of the characteristic features of chronic UC). However, only 1 patient (7%) normalized all their rectal biopsies. Finally, Bernstein et al28 showed that 2 of 39 (5%) patients with UC have histologic evidence of absolute rectal sparing at some point during their disease. None of these studies looked at proximal colon histologic normalization or patient or disease characteristics associated with histologic normalization.
We found that CHN was associated with less extensive disease. It has previously been described that extensive colitis is a risk factor for more complicated disease outcomes with the rate of colectomy in these patients of about 19% at 10 years compared to 5% of those who have proctitis.29
Our study demonstrates that histologic normalization is associated with improved clinical outcomes when compared with histologic quiescence and activity, and is more predictive of improved outcomes than endoscopic MH alone or histologic quiescence alone. Several studies have now confirmed the value of histologic features of colitis predicting clinical relapse in UC.9,11,12,16,18 Riley et al9 found in 82 patients with UC that an acute inflammatory cell infiltrate, crypt abscess, and mucin depletion were significantly higher in those who subsequently relapsed within 12 months. Bitton et al11 reported on 74 patients in clinical and endoscopic remission with rectal biopsy specimens and demonstrated that basal plasmacytosis was associated with UC relapse with a hazard rate of 4.5. In addition, Feagins et al18 described 51 patients in clinical remission and reported that basal lymphoplasmacytosis, erosions/ulceration of the epithelial, or moderate to marked architectural distortion significantly predicted clinical flares by 6 and 12 months and was more accurate at predicting flares compared with endoscopic assessment alone. Finally, Bryant et al16 demonstrated that histologic remission predicted corticosteroid use and acute severe colitis requiring hospitalization over 6 years and, similar to our study, endoscopic MH did not. Although our data confirm the importance of histologic healing in improving clinical outcomes in patients with UC over and beyond that of endoscopic MH, it is novel in that we identify histologic normalization as a stronger predictor of a decreased risk of relapse in patients with either quiescent endoscopic mucosal or histologic UC.
Despite our findings, one must be guarded in translating these associations into clinical practice. The findings cannot yet justify increasing therapy for the sole purpose of achieving histologic normalization. It is important, however, to acknowledge that this level of “deeper remission” is associated with improved outcomes. Patients who achieve histologic normalization can be informed of their improved prognosis and may represent a cohort requiring less stringent clinical surveillance and follow-up. Furthermore, whether this is a subgroup of patients that may benefit from stable de-escalation of medical therapy or require less intensive cancer surveillance remains to be determined. Finally, similar to other recent reports,12,16 a total of 27% of patients in this study with mucosal quiescence had persistent microscopic inflammation. Because histologic assessment seems superior to endoscopic assessment in predicting clinical outcomes, this disparity between histologic and endoscopic outcomes indicates that one should consider incorporating histologic surveillance into clinical practice.
This study lends further evidence to the importance of histologic assessment in prognosticating a patient’s future disease course, and has implications for the design of future clinical trials. In fact, the United States Food and Drug Administration has pointed to the possibility of requiring documentation of histologic disease activity at inclusion and as an outcome measure in future clinical trials.30 Clarification of a standardized and validated reporting system for histologic disease severity is needed in UC. There are current international efforts being undertaken to develop such an index.20,21 This study suggests any such index should include histologic normalization as a separate and independent grade.
There are several notable strengths and several limitations to this study. As with any retrospective analysis, there may be inaccuracies in data collection. However, the extensive experience of the involved clinicians and overlapping data sources (electronic records, endoscopy, and pathology reports) should minimize this limitation. The generalizability of the data is also uncertain, because this is a single-center study based in a tertiary hospital setting where experts in the area of IBD manage patients. It is unclear whether this selection issue would make normalization of histology more or less likely; although patients may be treated for their disease more aggressively, they also most likely represent a more complex range of patients with more severe disease phenotypes compared with the general community. The tertiary nature of the setting is also in part its strength, because there is standardized reporting for endoscopy and pathology at the center. In addition, although we do use a standardized approach to sampling the mucosa in our patients with UC, it is possible that these results may represent a sampling bias. We believe this limitation was minimized based on the fact that there was no significant difference in biopsy number per patient when comparing cases and control subjects (histologic normalization compared with no normalization). There is also minimal variability between biopsies within each colonic segment with the same percentage of intrasegment biopsies with the same histologic inflammatory activity score being 80% or greater across all segments of the large bowel.31 Furthermore, although our histologic activity and quiescence definitions are similar to those previously described by others,3,14,16,32 the histologic scale used here to assess histologic normalization has not undergone independent validation. Finally, given the limitations of this retrospective review, dose and duration of medication exposures were not obtained. Although understanding more about therapies and how they may achieve histologic normalization is of interest, we suggest that this should be assessed in future trials, along with the potential for controlled de-escalation of therapies in patients who achieve CHN.
In conclusion, we demonstrate that histologic normalization is an outcome in patients with UC and have found that it occurs more often in association with less extensive disease. We demonstrate for the first time that CHN in UC is associated with improved clinical outcomes and provide further evidence that, despite the introduction and search for other predictive biomarkers in IBD, traditional histopathology may well be the most reliable. We propose that histologic assessment of disease activity should be part of endoscopic assessment in IBD. In addition, future standardized and validated histologic indices include histologic normalization as a unique outcome and encourage these findings to be incorporated into future clinical trials and clinical practice.
Supplementary Material
Acknowledgments
Financial/Grant Support: Funded in part by the “Digestive Disease Research Core Center of the University of Chicago (DK42086)”.
Abbreviations
- CHN
Complete Histologic Normalization
- MH
Mucosal Healing
- SCCAI
Simple Clinical Colitis Activity Index
Footnotes
Competing Interests/Disclosures: There are no author financial disclosures or relevant conflicts of interest for any author
Specific author contributions:
Britt Christensen: Study concept and design, acquisition of the data, analysis and interpretation of the data, drafting of the manuscript, statistical analysis
Jon Erich: Acquisition of data and drafting of the manuscript
Olufemi Kassim: Acquisition of data
Stephen B. Hanauer: Study concept and design and critical revision of the manuscript Peter R. Gibson: Study concept and design, drafting of the manuscript, critical revision of the manuscript and study supervision
Jerrold R. Turner: Pathology review and critical revision of the manuscript
John Hart: Pathology review and critical revision of the manuscript
David T. Rubin: Study concept and design, study supervision, drafting of the manuscript and critical revision of the manuscript
References
- 1.D’Haens G, Sandborn WJ, Feagan BG, et al. A review of activity indices and efficacy end points for clinical trials of medical therapy in adults with ulcerative colitis. Gastroenterology. 2007;132:763–86. doi: 10.1053/j.gastro.2006.12.038. [DOI] [PubMed] [Google Scholar]
- 2.Neurath MF, Travis SP. Mucosal healing in inflammatory bowel diseases: a systematic review. Gut. 2012;61:1619–35. doi: 10.1136/gutjnl-2012-302830. [DOI] [PubMed] [Google Scholar]
- 3.Truelove SC, Richards WC. Biopsy studies in ulcerative colitis. Br Med J. 1956;1:1315–8. doi: 10.1136/bmj.1.4979.1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Froslie KF, Jahnsen J, Moum BA, et al. Mucosal healing in inflammatory bowel disease: results from a Norwegian population- based cohort. Gastroenterology. 2007;133:412–22. doi: 10.1053/j.gastro.2007.05.051. [DOI] [PubMed] [Google Scholar]
- 5.Colombel JF, Rutgeerts P, Reinisch W, et al. Early mucosal healing with infliximab is associated with improved long-term clinical outcomes in ulcerative colitis. Gastroenterology. 2011;141:1194–201. doi: 10.1053/j.gastro.2011.06.054. [DOI] [PubMed] [Google Scholar]
- 6.Sandborn WJ, Rutgeerts P, Feagan BG, et al. Colectomy rate comparison after treatment of ulcerative colitis with placebo or infliximab. Gastroenterology. 2009;137:1250–1260. doi: 10.1053/j.gastro.2009.06.061. [DOI] [PubMed] [Google Scholar]
- 7.Feagan BG, Reinisch W, Rutgeerts P, et al. The effects of infliximab therapy on health-related quality of life in ulcerative colitis patients. Am J Gastroenterol. 2007;102:794–802. doi: 10.1111/j.1572-0241.2007.01094.x. [DOI] [PubMed] [Google Scholar]
- 8.Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am J Gastroenterol. 2015;110:1324–28. doi: 10.1038/ajg.2015.233. [DOI] [PubMed] [Google Scholar]
- 9.Riley SA, Mani V, Goodman MJ, et al. Microscopic activity in ulcerative colitis: what does it mean? Gut. 1991;32:174–8. doi: 10.1136/gut.32.2.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bessissow T, Lemmens B, Ferrante M, et al. Prognostic value of serologic and histologic markers on clinical relapse in ulcerative colitis patients with mucosal healing. Am J Gastroenterol. 2012;107:1684–92. doi: 10.1038/ajg.2012.301. [DOI] [PubMed] [Google Scholar]
- 11.Bitton A, Peppercorn MA, Antonioli DA, et al. Clinical, biological, and histologic parameters as predictors of relapse in ulcerative colitis. Gastroenterology. 2001;120:13–20. doi: 10.1053/gast.2001.20912. [DOI] [PubMed] [Google Scholar]
- 12.Park S, Abdi T, Gentry M, et al. Histological Disease Activity as a Predictor of Clinical Relapse Among Patients With Ulcerative Colitis: Systematic Review and Meta-Analysis. Am J Gastroenterol. 2016;111:1692–701. doi: 10.1038/ajg.2016.418. [DOI] [PubMed] [Google Scholar]
- 13.Rubin DT, Huo D, Kinnucan JA, et al. Inflammation is an independent risk factor for colonic neoplasia in patients with ulcerative colitis: a case-control study. Clin Gastroenterol Hepatol. 2013;11:1601–8. doi: 10.1016/j.cgh.2013.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gupta RB, Harpaz N, Itzkowitz S, et al. Histologic inflammation is a risk factor for progression to colorectal neoplasia in ulcerative colitis: a cohort study. Gastroenterology. 2007;133:1099–1105. doi: 10.1053/j.gastro.2007.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hefti MM, Chessin DB, Harpaz NH, et al. Severity of inflammation as a predictor of colectomy in patients with chronic ulcerative colitis. Dis Colon Rectum. 2009;52:193–7. doi: 10.1007/DCR.0b013e31819ad456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bryant RV, Burger DC, Delo J, et al. Beyond endoscopic mucosal healing in UC: histological remission better predicts corticosteroid use and hospitalisation over 6 years of follow-up. Gut. 2016;65(3):408–14. doi: 10.1136/gutjnl-2015-309598. [DOI] [PubMed] [Google Scholar]
- 17.Peyrin–Biroulet L, Bressenot A, Kampman W. Histologic remission: the ultimate therapeutic goal in ulcerative colitis? Clin Gastroenterol Hepatol. 2014;12:929–934. doi: 10.1016/j.cgh.2013.07.022. [DOI] [PubMed] [Google Scholar]
- 18.Feagins LA, Melton SD, Iqbal R, et al. Clinical implications of histologic abnormalities in colonic biopsy specimens from patients with ulcerative colitis in clinical remission. Inflamm Bowel Dis. 2013;18:1477–82. doi: 10.1097/MIB.0b013e318281f4ae. [DOI] [PubMed] [Google Scholar]
- 19.Geboes K, Riddell R, Öst A, et al. A reproducible grading scale for histological assessment of inflammation in ulcerative colitis. Gut. 2000;47:404–409. doi: 10.1136/gut.47.3.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Marchal-Bressenot A, Salleron J, Boulagnon-Rombi C, et al. Development and validation of the Nancy histological index for UC. Gut. 2015 doi: 10.1136/gutjnl-2015-310187. [DOI] [PubMed] [Google Scholar]
- 21.Mosli MH, Feagan BG, Zou G, et al. Development and validation of a histological index for UC. Gut. 2015 doi: 10.1136/gutjnl-2015-310393. [DOI] [PubMed] [Google Scholar]
- 22.Odze R, Antonioli D, Peppercorn M, et al. Effect of topical 5- aminosalicylic acid (5-ASA) therapy on rectal mucosal biopsy morphology in chronic ulcerative colitis. Am J Surg Pathol. 1993;17:869–75. doi: 10.1097/00000478-199309000-00002. [DOI] [PubMed] [Google Scholar]
- 23.Satsangi J, Silverberg M, Vermeire S, et al. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut. 2006;55:749–753. doi: 10.1136/gut.2005.082909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Scherl EJ, Pruitt R, Gordon GL, et al. Safety and efficacy of a new 3.3 g bid tablet formulation in patients with mild-to-moderately-active ulcerative colitis: a multicenter, randomized, double-blind, placebo- controlled study. Am J Gastroenterol. 2009;104:1452–1459. doi: 10.1038/ajg.2009.83. [DOI] [PubMed] [Google Scholar]
- 25.Walmsley RS, Ayres RC, Pounder RE, et al. A simple clinical colitis activity index. Gut. 1998;43:29–32. doi: 10.1136/gut.43.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kleer CG, Appelman HD. Ulcerative colitis: patterns of involvement in colorectal biopsies and changes with time. Am J Surg Pathol. 1998;22:983–9. doi: 10.1097/00000478-199808000-00008. [DOI] [PubMed] [Google Scholar]
- 27.Levine TS, Tzardi M, Mitchell S, et al. Diagnostic difficulty arising from rectal recovery in ulcerative colitis. J Clin Pathol. 1996;49:319–23. doi: 10.1136/jcp.49.4.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bernstein CN, Shanahan F, Anton PA, et al. Patchiness of mucosal inflammation in treated ulcerative colitis: a prospective study. Gastrointest Endosc. 1995;42:232–7. doi: 10.1016/s0016-5107(95)70097-8. [DOI] [PubMed] [Google Scholar]
- 29.Solberg IC, Lygren I, Jahnsen J, et al. Clinical course during the first 10 years of ulcerative colitis: results from a population-based inception cohort. Scand J Gastroenterol. 2009;44:431–440. doi: 10.1080/00365520802600961. [DOI] [PubMed] [Google Scholar]
- 30.U.S. Department of Health and Human Services Food and Drug Adminstration Center for Drug Evaluation and Research (CDER) Ulcerative Colitis: Clinical Trial Endpoints Guidance for Industry. Available: http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm515143.pdf [Accessed 4th September 2016]
- 31.Mikolajczyk AE, Watson S, Ackerman MT, et al. Assessment of the Degree of Variation of Histologic Inflammation in Ulcerative Colitis. Gastroenterology. 2014;146:S-232. doi: 10.1093/ibd/izac070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hanauer S, Schwartz J, Robinson M, et al. Mesalamine capsules for treatment of active ulcerative colitis: results of a controlled trial. Am J Gastroenterol. 1993;88:1188–97. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


