Abstract
L-Carnitine functions to transport long chain fatty acyl CoAs into the mitochondria for degradation by β-oxidation. Treatment with L-carnitine can ameliorate metabolic imbalance in many inborn errors of metabolism. In recent years there has been considerable interest in the therapeutic potential of L-carnitine and its acetylated derivative acetyl-L-carnitine (ALCAR) for neuroprotection in a number of disorders including hypoxia-ischemia, traumatic brain injury, Alzheimer’s disease and in conditions leading to central or peripheral nervous system injury. There is compelling evidence from preclinical studies that L-carnitine and ALCAR can improve energy status, decrease oxidative stress and prevent subsequent cell death in models of adult, neonatal and pediatric brain injury. ALCAR can provide an acetyl moiety that can be oxidized for energy, used as a precursor for acetylcholine, or incorporated into glutamate, glutamine and GABA, or into lipids for myelination and cell growth. Administration of ALCAR after brain injury in rat pups improved long-term functional outcomes, including memory. Additional studies are needed to better explore the potential of L-carnitine and ALCAR for protection of developing brain as there is an urgent need for therapies that can improve outcome after neonatal and pediatric brain injury.
Keywords: L-carnitine, acetyl-L-carnitine, neuroprotection, neonatal hypoxia-ischemia, pediatric traumatic brain injury, metabolism, inborn errors of metabolism, carnitine shuttle
Introduction
L-Carnitine is a naturally occurring compound found in most, if not all, mammalian tissues including brain [1]. Although carnitine can be obtained in the diet and synthesized in kidney, liver and brain, it is considered a ‘conditionally essential’ nutrient for humans under specific circumstances when intracellular levels are low (e.g. premature infants, elderly patients, diabetes and genetic conditions resulting in primary or secondary carnitine deficiency) [2, 3].
L-Carnitine primarily functions to transport activated long chain fatty acids (long chain fatty acyl-CoAs) into the mitochondria for degradation by β-oxidation [1, 4–6]. Human plasma and tissues, including brain, contain free L-carnitine as well as acylated derivatives with varying length carbon chains, including the acetylated and palmitoylated derivatives [1].
In recent years there has been considerable interest in the therapeutic potential of L-carnitine and acetyl-L-carnitine (ALCAR) for neuroprotection [1, 4, 7–22]. Therapeutic efficacy of L-carnitine treatment for infants affected by some inborn errors of metabolism has been reported [1, 3, 21, 23–36]. A number clinical trials and case studies have reported efficacy of ALCAR for neuroprotection in conditions leading to central or peripheral nervous system injury in adults [1, 4, 7–22]. Although relatively few studies have determined the efficacy of acetyl-L-carnitine for neuroprotection in models of developmental brain injury, results from these studies are promising [12, 19, 20, 37–39].
L-Carnitine biosynthesis in humans
The carbon backbone of L-carnitine comes from 6-N-trimethyllysine, a product of protein degradation after lysosomal proteolysis [40, 41]. 6-N-trimethyllysine is converted to γ-butyrobetaine in a series of reactions catalyzed by enzymes ubiquitously present in tissues. γ-Butyrobetaine is then converted to L-carnitine in a reaction catalyzed by γ-butyrobetaine dioxygenase, an enzyme that is only expressed in kidney, liver, and brain [40, 41]. Therefore, complete endogenous carnitine biosynthesis occurs only in these tissues (kidney, liver, and brain), which can also form L-carnitine using γ-butyrobetaine imported from other cells that do not contain γ-butyrobetaine dioxygenase [40, 41]. The detailed biosynthetic pathway of L-carnitine is shown in Figure 1.
Figure 1. L-Carnitine biosynthesis in humans.
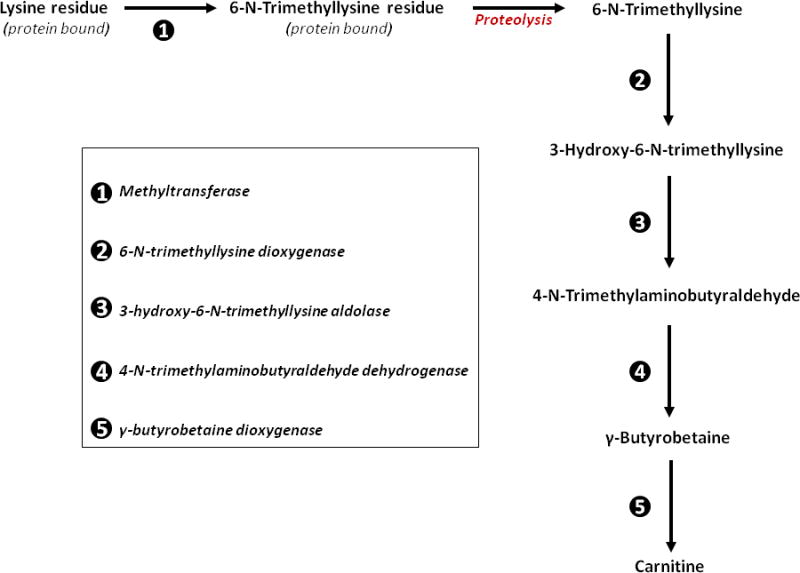
In the first step of L-carnitine biosynthesis, a lysine residue bound to some proteins is post-translationally methylated by a methyltransferase (enzyme 1) to form a 6-N-trimethyllysine residue. The methyl groups are transferred from S-adenosylmethionine yielding S-adenosylhomocysteine and the methylated lysine. After lysosomal proteolytic release of the 6-N-trimethyllysine residue, 6-N-trimethyllysine is then metabolized by 6-N-trimethyllysine dioxygenase (enzyme 2) leading to the formation of the hydroxylated metabolite, 3-hydroxy-6-N-trimethyllysine. 3-hydroxy-6-N-trimethyllysine aldolase (enzyme 3) splits 3-hydroxy-6-N-trimethyllysine into glycine plus 4-N-trimethylaminobutyraldehyde, which is further dehydrogenated to 4-N-trimethylaminobutyrate (also known as γ-butyrobetaine) by 4-N-trimethylaminobutyraldehyde dehydrogenase (enzyme 4). The enzymes mentioned in these steps are ubiquitously expressed; therefore, γ-butyrobetaine can be produced in many tissues. The last step in carnitine synthesis is the hydroxylation of γ-butyrobetaine by γ-butyrobetaine dioxygenase (enzyme 5) forming 3-hydroxy-4-N-trimethylaminobutyrate (carnitine). The presence of γ-butyrobetaine dioxygenase is restricted to kidney, liver, and brain, therefore the complete pathway for endogenous carnitine biosynthesis only occurs in these tissues.
L-Carnitine uptake and metabolic role in tissues
In the diet, carnitine is obtained primarily from red meat and dairy products [1]. There are also dietary supplements containing the L-isomer (L-carnitine) with high purity [42]. L-Carnitine and its corresponding derivative containing 2 carbons in the acyl moiety (acetyl-L-carnitine) are taken up into cells via the organic cation transporter novel 2 (OCTN2) which cotransports L-carnitine with Na+ [43, 44]. Uptake into brain also occurs primarily via the OCTN2 transporter [1, 44, 45]. There is evidence that the B0,+ transporter, a Na+/Cl− dependent amino acid/carnitine transporter, also has a role in L-carnitine and acetyl-L-carnitine uptake into brain [44, 46–48], and into astrocytes [49, 50]. Immunohistochemical studies show that labeling for OCTN 1, 2 and 3 is distributed in many regions of mouse brain and spinal cord in a pattern consistent with possible roles in modulating bioenergetics and cholinergic neurotransmission [51]. OCTN2 and OCTN3 mRNA and proteins are found in neurons obtained from adult and suckling rat brain [52]
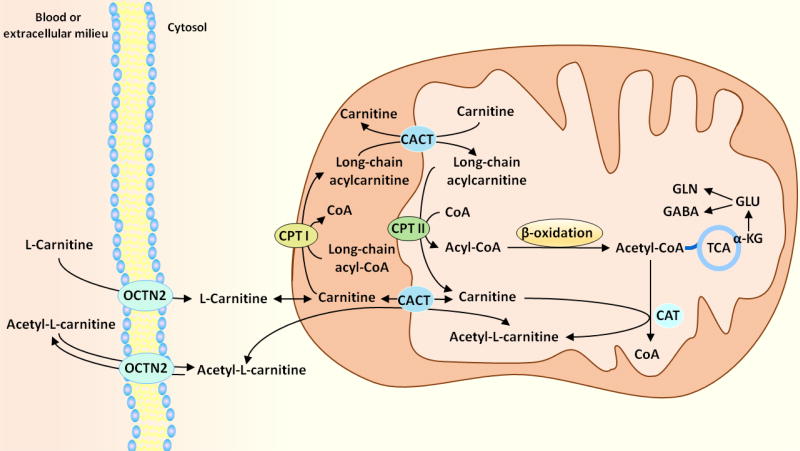
While medium- and short-chain fatty acids can freely enter mitochondria by diffusion through the membranes [1], L-carnitine has an essential role in the transfer of activated long chain fatty acids into the mitochondria in a series of reactions called the “carnitine shuttle”, so that they can undergo β-oxidation (Figure 2) [1, 43]. The enzyme acyl-CoA synthase converts long chain fatty acids to fatty acyl-CoAs, which are subsequently converted to acylcarnitines by the enzyme carnitine palmitoyltransferase I (CPT I) localized in the outer mitochondrial membrane [1, 43]. Acylcarnitines cross the inner mitochondrial membrane via a transporter, the carnitine/acylcarnitine translocase, in exchange for free L-carnitine [1, 43]. The enzyme carnitine palmitoyltransferase II (CPT II) which is localized in the inner mitochondrial membrane converts acylcarnitines back to acyl CoAs and free L-carnitine, which exits the mitochondria and serves as the substrate for CPT I to form more acylcarnitine [1, 43]. Therefore, the transfer of acyl moieties from fatty acyl-CoA esters to carnitine also replenishes intracellular free CoA that is crucial for intermediary metabolism [53].
Figure 2. The carnitine shuttle.
L-carnitine and acetyl-L-carnitine enter the cells from blood or extracellular milieu through the OCTN2 transporter. The enzyme acyl-CoA synthase (not shown) converts long chain fatty acids to fatty acyl-CoAs, which are subsequently converted to acylcarnitines by the enzyme carnitine palmitoyltransferase I (CPT I) localized in the outer mitochondrial membrane. Acylcarnitines cross the inner mitochondrial membrane via a transporter, the carnitine/acylcarnitine translocase (CACT), in exchange for free L-carnitine. The enzyme carnitine palmitoyltransferase II (CPT II), which is localized in the inner mitochondrial membrane, converts acylcarnitines back to acyl-CoAs and free L-carnitine, which exits the mitochondria and serves as the substrate for CPT I to form more acylcarnitine. Carbons from acyl-CoAs imported into the mitochondrial matrix through the carnitine shuttle can be oxidized for energy or metabolized via the TCA cycle and incorporated into glutamate, glutamine and GABA.
The carnitine shuttle is essential to prevent accumulation of long chain fatty acids and long chain acyl-CoAs which can be deleterious to cells [1, 43, 54]. The enzyme carnitine acetyltransferase (CAT) has a crucial role in the metabolic flexibility of cells, as it transfers a 2-carbon moiety from acetyl-CoA to L-carnitine, forming the membrane permeable compound acetyl-L-carnitine; this serves to regulate intracellular trafficking of carbons between mitochondria and cytosol [55]. In adult brain, the carbons from the acetyl moiety can be used to synthesize fatty acids, which are preferentially incorporated into structural lipids, rather than oxidized via β-oxidation [45, 56]. In developing brain the acetyl moiety may be oxidized for energy and incorporated into neurotransmitters and lipids [57].
The importance of the carnitine shuttle is underscored by the report that a polymorphism of the CPT II gene that leads to decreased enzyme activity may be associated with acute encephalopathy associated with influenza [58, 59] and that deficiency of the OCTN2 transporter can lead to neurological manifestations including cognitive impairment and seizures [51]. Two inherited diseases in the carnitine biosynthetic pathway have been reported (deficiency of 6-N-trimethyllysine dioxygenase and deficiency of γ-butyrobetaine dioxygenase), which result in only mildly decreased to normal carnitine levels in the affected patients [60, 61]. It is intriguing though, that despite the mild impact on L-carnitine levels, patients affected by these diseases have marked brain alterations such as nondysmorphic autism (deficiency of 6-N-trimethyllysine dioxygenase) [60] and microcephaly and speech delay (deficiency of γ-butyrobetaine dioxygenase) [61].
Neuroprotection afforded by L-carnitine supplementation in patients with carnitine deficiencies
Primary carnitine deficiency and defects in the carnitine shuttle in patients
Deficiency in the OCTN2 carnitine transporter is a rare inherited disease that leads to systemic primary carnitine deficiency [62, 63]. It is associated with depletion of intracellular carnitine, low serum carnitine concentrations and increased urinary excretion of carnitine and its derivatives [62, 63]. Patients normally respond to pharmacological doses of oral L-carnitine, particularly if supplementation is implemented prior to organ damage [64].
Defects of the enzymes and transporter involved in the transfer of long chain fatty acids from the cytoplasm into mitochondrial matrix (CPT I, CPT II and carnitine/acylcarnitine translocase) have also been described [42]. Treatment of these carnitine shuttle disorders is primarily based on avoidance of fasting and metabolic decompensation in the patients [63].
Secondary carnitine deficiency in patients
Secondary carnitine deficiency may arise from different causes, including acquired (prolonged utilization of some medications) or associated with inborn errors of metabolism (e.g. fatty acid oxidation disorders) [65]. Carnitine levels are normally less depleted in secondary carnitine deficiency when compared to OCTN2-deficient patients, and therefore smaller doses of L-carnitine can restore the carnitine levels in a shorter period of time than in patients with primary deficiency [41].
Disorders of fatty acid oxidation and organic acidemias can lead to secondary carnitine deficiency by trapping free carnitine by conjugating it with acyl moieties which accumulate in these conditions. It is postulated that clinical symptoms, including CNS complications such as convulsion, coma and lethargy, may be triggered by the accumulation of metabolites and their acyl-CoA derivatives that disrupt intermediary metabolism [66–69]. The hydrolysis of the acyl-CoA derivatives and subsequent accumulation of free organic acids can lead to severe acidosis that can be life threatening [3]. Competitive inhibition of carnitine/acetyl-L-carnitine reuptake by OCTN2 in kidney by the accumulating acylcarnitines (e.g. propionyl carnitine, etc.) contributes to the increased loss of carnitine by excretion in the urine [70]. There are reports showing improvement after L-carnitine therapy in patients with some organic acidemias, including propionic acidemia, methylmalonic acidemia, and glutaric acidemia type I [71–75]. More recent reports have demonstrated that L-carnitine supplementation as adjuvant therapy contributes to the amelioration of blood markers of oxidative damage in patients affected by phenylketonuria [76], maple syrup urine disease [77], and disorders of propionate metabolism [78].
Likewise, metabolites accumulated after long term utilization of pharmacological therapies, such as valproate [79] and the antibiotic cefditoren pivoxil [80], can be conjugated to carnitine and result in carnitine depletion. Secondary carnitine deficiency may also arise from other deleterious conditions such as hemodialysis or renal tubular dysfunction, which result in excessive loss of carnitine in urine. Secondary carnitine deficiency may also occur in malnutrition or prematurity, due to reduced intake or uptake of carnitine from the diet, or reduced reuptake in kidney [41, 81, 82]. Treatment with L-carnitine ameliorated symptoms of encephalopathy subsequent to long term use of valproate [79], and the antibiotic cefditoren pivoxil [80]. Case reports indicate that improvement with L-carnitine treatment was also seen in hyperammonemic encephalopathy caused by carnitine deficiency that manifested several years after gastrointestinal bypass surgery [83] and in encephalopathy secondary to gluten enteropathy [84]. Additionally, a randomized clinical trial reported some improvement in Friedreich’s ataxia patients treated with L-carnitine [85].
Studies using animal models provide insight into possible mechanisms of neuroprotection by L-carnitine
Ueno et al. [86] studied a model of chronic cerebral hypoperfusion in adult rats caused by permanent ligation of both common carotid arteries. Rats treated with an oral dose of 600 mg/kg L-carnitine after artery occlusion had significantly reduced escape latency in the Morris water maze when tested 28 days after surgery, but no improvement in motor performance compared to vehicle treated rats. Rats treated with L-carnitine had decreased oxidative DNA damage and lipid peroxidation [86], greater myelin sheath thickness and enhanced expression of oligodendrocyte markers after chronic hypoperfusion. At 28 days after onset of hypoperfusion, rats treated with L-carnitine had increased phosphorylated Akt and mammalian target of rapamycin (mTOR), as well as increased levels of phosphorylated high-molecular weight neurofilament (pNFH) compared to vehicle treated rats [86]. The authors proposed that L-carnitine “regulates the PTEN/Akt/mTOR signaling pathway, and enhances axonal plasticity while concurrently ameliorating oxidative stress and increasing oligodendrocyte myelination of axons” and suggested that this attenuated cognitive impairment after chronic hypoperfusion [86] However, the increased CPT I and CPT II protein levels in L-carnitine treated rats may also have protected myelin after chronic hypoperfusion.
Pretreatment of rats with L-carnitine (100mg/kg, injected intraperitoneally; i.p.) prior to injection of the metabolic inhibitor 3-nitropropionic acid (3-NPA) in adult Sprague Dawley rats attenuated the 3-NPA induced increase in expression of the uncoupling protein UCP-2 and dopamine D(1) receptor genes in striatum that were upregulated in response to impaired metabolism [87]. Yu et al. [88] found that pretreatment of mice with intraperitoneal injections of L-carnitine , suppressed both tonic and clonic seizures induced by pentylenetetrazol in a dose-dependent manner.
Pediatric models of brain injury
Wainwright et al. [19] demonstrated that pretreatment of 7 day old rat pups with L-carnitine (16 mmol/kg) 30 minutes prior to hypoxia-ischemia (HI) led to improved outcome at 7 and 28 days after HI. Pretreatment with L-carnitine led to significantly less tissue loss in the ipsilateral hemisphere, compared to vehicle controls at both 7 days and 28 days after HI. Dying neurons labeled with Fluro-Jade B were present in the hippocampus and cortex of vehicle treated pups after HI. In contrast, no cells labeled with Fluro-Jade B were present in the brain of rat pups pretreated with L-carnitine [19]. Interestingly there was no protection in rat pups treated with L-carnitine at 1 and 4 hours after HI [19]. The authors proposed that L-carnitine could prevent the accumulation of acyl-CoAs in mitochondria, which they hypothesized is a key early event involved in the pathophysiology of hypoxic-ischemic injury [19]. This hypothesis from Wainwright et al. [19] has merit, as accumulation of acyl-CoAs can inhibit the activity of some tricarboxylic acid (TCA) cycle enzymes, adenine nucleotide translocation and ATP formation [89–91]. L-Carnitine has been shown to reduce the level of acyl-CoAs in mitochondria by converting them to acylcarnitine esters [92]. Thus treatment with L-carnitine enables CPT I to transfer the acyl groups from acyl-CoAs to free carnitine, yielding acylcarnitine esters, which prevents the accumulation of, and subsequent damage from, high levels of acyl-CoAs [19].
An in vitro study by Rau et al. [54] determined the effects of oxygen glucose deprivation (OGD) on carnitine homeostasis and synaptic activity in hippocampal slice cultures from 7 day old rat brain. Interestingly, OGD led to decreased levels of CPT I and CPT II protein, a corresponding decrease in free carnitine and an increase in the ratio of acylcarnitine to free carnitine [54]. The changes in enzymes and carnitine homeostasis were ameliorated by treatment with L-carnitine for 2 hours prior to OGD [54]. Pretreatment with L-carnitine also attenuated superoxide production and HIF1α expression after OGD, and decreased apoptosis in neurons during reperfusion [54]. Treatment with L-carnitine maintained synaptic viability (determined by EPSP amplitude) within the hippocampus at 48 hours after OGD. This study underscores the vulnerability of CPT I, CPT II, and carnitine homeostasis to oxidative stress in a widely used in vitro model of ischemia and reperfusion [54]. Importantly, it demonstrated that treatment with L-carnitine attenuated oxidative stress and protected the levels of CPT I, CPT II, ATP and synaptic activity [54].
A number of studies have used the ratio of acylcarnitines to free carnitine as an index of carnitine homeostasis [19, 20, 53, 54, 93]. Abnormal ratios, reflecting impaired carnitine status and/or homeostasis, have been reported in plasma of infants and children with inborn errors of metabolism [93, 94], children with autism spectrum disorder [95], animal models of autism [96] and in brain tissue after OGD in vitro [54]. The studies from Wainwright and coworkers discussed above suggest that maintaining carnitine homeostasis in brain tissue can prevent dysfunction and death of neurons in models of hypoxic-ischemic injury [54]
Neuroprotection by acetyl-L-carnitine (ALCAR)
As noted above, ALCAR is one of the most common metabolites of carnitine found in plasma and tissues of humans and mammals [1]. ALCAR has documented neuroprotective effects and is also sold as a dietary supplement [7, 12, 13, 18, 22, 37, 97–99]. ALCAR has several properties that could have neuroprotective effects including providing carnitine and an acyl moiety that can be used for energy [57, 100], and for synthesis of acetylcholine [101], amino acid neurotransmitters [57] and lipids [102] as discussed in more detail below. ALCAR has been found to have anti-inflammatory effects [7], lead to stabilization of membranes [1], act as an antioxidant protecting against oxidative stress [37, 53, 103, 104], enhance the activity of nerve growth factor [105], and potentiate energy metabolism [57, 97], and cholinergic responses [1, 106]. ALCAR administration induced mitochondrial biogenesis in hypoxic rats [15], and increased mitochondrial mass after spinal cord injury [13].
Recent reports demonstrate that administration of ALCAR after injury can improve mitochondrial function [38], decrease swelling in brain after injury [37, 39], and prevent loss of tissue in pediatric injury models [12, 37, 39]. Long term administration of ALCAR improved energy status in healthy mouse brain [107].
Since ALCAR is metabolized to acetyl CoA, it has the potential to acetylate histones, which can modify gene expression [108, 109], and to acetylate proteins and enzymes, which can greatly modify activity [110–112].
Uptake and metabolism of ALCAR in brain
ALCAR enters the brain rapidly in primates [113] and rodents and is metabolized in mitochondria to free carnitine and acetyl-CoA [1, 57] as shown in Figure 3. Thus ALCAR provides both carnitine for the transport of fatty acids across mitochondrial membranes, and acetyl-CoA that can be incorporated into lipids [114], oxidized in the TCA cycle for energy production and incorporated into neurotransmitters [1, 57]. Scafidi et al. [57] determined incorporation of label in brain from the metabolism of [2-13C]acetyl-L-carnitine after i.p. injection in 21–22 day old rats. This key study showed that the acetyl moiety of ALCAR was metabolized for energy and incorporated into the carbon skeleton of the neurotransmitters GABA and glutamate in developing brain [57]. Unlike the pattern seen with other substrates, there was prolonged cycling of carbon from ALCAR in the TCA cycle, which was readily detected from the cycling ratios of GABA, glutamine and glutamate [57]. Scafidi et al. [57] also found very high metabolism of carbon from the acetyl moiety of 13C-ALCAR in the potentially neuroprotective pyruvate recycling pathway. This pathway is considered to be neuroprotective as it can provide pyruvate when glycolysis is inhibited [115–117]. Furthermore, the acetyl moiety from ALCAR can enter the TCA cycle when metabolism via the pyruvate dehydrogenase complex is impaired as occurs in hypoxia and traumatic brain injury [103, 118–122]. Overall, the data from Scafidi et al. [57] demonstrate that ALCAR metabolism in developing brain is unusual in that there is particularly high pyruvate recycling, and labeling of GABA, glutamine, glutamate and aspartate from metabolism in the TCA cycle increased from 30 minutes to 2 hours. Such a pattern is not found with metabolism of other substrates including glucose and acetate [122, 123]. The continued increase in metabolite labeling may be due to reutilization of the acetyl CoA subsequent to oxidation of fatty acids that were synthesized from the acetyl moiety of the labeled ALCAR. This possibility is supported by the findings of Ricciolini et al. [102] who showed that the incorporation of 14C into lipids was highest at 1 hour after injection of [1-14C]acetyl-L-carnitine into adult rat brain, and labeling was decreased at 3, 6 and 22 h after injection, suggesting that turnover of lipids containing carbons from ALCAR and reutilization of the carbons from ALCAR occurs in brain [102]. Studies with adult brain found that octanoate could support ~ 20% of oxidative metabolism [124].
Figure 3. Metabolism of ALCAR in brain.
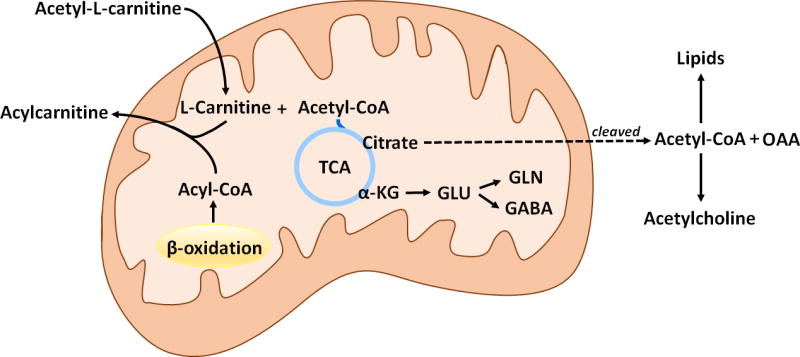
The mitochondrial membrane permeable acetyl-L-carnitine (ALCAR) is split in the mitochondrial matrix yielding acetyl-CoA and L-carnitine. Acetyl-CoA can be oxidized for energy via the tricarboxylic acid (TCA) cycle or incorporated into glutamate, glutamine or GABA. The citrate formed from the condensation of acetyl CoA and oxaloacetate (OAA) can also exit the mitochondria and following cleavage by citrate lyase it provides cytosolic OAA, and acetyl-CoA which can be used for lipid synthesis or as a precursor for acetylcholine. Free L-carnitine in the mitochondrial matrix can be used to form carnitine derivatives of acyl-CoA conjugates, therefore reducing their toxicity in conditions where the levels of these compounds are high (e.g., fatty acid oxidation disorders).
Ricciolini et al. [102] reported that almost 80 % of the label from 14C-ALCAR recovered in the lipid soluble fraction of brain was found in the polar lipid fraction that includes phospholipids and fatty acids, and ~ 20% was recovered in the neutral lipid fraction. The acetyl moiety from ALCAR was incorporated into saturated, monounsaturated and polyunsaturated fatty acids at 60%, 15% and 25%, respectively, indicating that it can be used for de novo synthesis of lipids and also elongation of polyunsaturated fatty acids derived from the diet [102].
Thus, the acetyl moiety from ALCAR can be oxidized for energy, serve as a precursor for acetylcholine, and be incorporated into amino acid neurotransmitters and lipids in brain [57, 102].
ALCAR alters the rate of glucose utilization in brain
Aureli et al. [125] showed that administration of ALCAR (100 mg/kg) by i.p. injection 20 minutes prior to injection of labeled glucose led to a reduction in the oxidation of [U-14C]glucose for energy and in the incorporation of 13C into amino acids and tricarboxylic acid cycle intermediates, consistent with sparing of glucose by use of the acetyl CoA from ALCAR for energy in brain. Interestingly, administration of ALCAR led to increased levels of proglycogen, a low molecular weight glycogen precursor, in brain compared to the levels in untreated rats [125].
Other studies showed that the intravenous administration of high doses of either 500 or 750 mg/kg ALCAR to awake adult Fischer-344 rats significantly increased uptake and phosphorylation of [14C]2-deoxy-D-glucose (14C-DG) used to determine the cerebral regional metabolic rate of glucose (rCMRglc) [126]. Administration of 500 or 750 mg/kg ALCAR prior to 14C-DG led to an increase in rCMRglc of 21–22% in 8 and 11 brain regions, respectively. The highest increases in rCMRglc were seen in the basal forebrain, septal and brainstem regions [126]. Although the mechanism is not known, acetyl CoA from ALCAR metabolism may have been used for synthesis of acetylcholine, which may have contributed to enhanced cholinergic neurotransmission [126]. In contrast to the increased glucose metabolism after ALCAR, the administration of carnitine (500 mg/kg) plus acetate (500 mg/kg) did not significantly alter the rCMRglc in any regions of brain [126]. This latter finding is consistent with reports that uptake and metabolism of acetate occurs primarily in astrocytes [123, 127], in contrast to ALCAR which is metabolized in both neurons and astrocytes [57].
Studies of ALCAR neuroprotection in adults
As noted above, clinical trials and case studies have reported efficacy of ALCAR for neuroprotection in conditions leading to central (CNS) or peripheral nervous system injury in adults [1, 4, 7–22]. Several studies in adult patients with probable Alzheimer’s disease reported that clinical trials of ALCAR therapy improved cognitive performance; however, a number of studies reported no improvement [126, 128] and refs therein. Studies with Alzheimer’s can be difficult to interpret since many used ALCAR in combination with lipoic acid or other therapies (reviewed in [128]). Calabrese et al. [129] reported that treatment of multiple sclerosis (MS) patients with ALCAR for 6 months resulted in decreased levels of reactive nitrogen species and protein nitration in CSF, and increased GSH levels and GSH/GSSG ratio compared to untreated MS subjects or patients with noninflammatory neurological conditions.
Animal Studies Using ALCAR Supplementation
Smeland et al. [107] determined the effect of chronic supplementation with about 0.5 g/kg ALCAR per day in drinking water of mice for 25 days. They found increased glucose levels and decreased [3-13C]lactate in both hippocampus and cortex, but no changes in the incorporation of 13C from metabolism of [1-13C]glucose into the amino acids glutamate, GABA and glutamine. However, the cortex of ALCAR treated mice had a higher total content of adenosine nucleotides and phosphocreatine, in conjunction with a higher ratio of phosphocreatine to creatine, all of which indicate increased energy levels. Mice supplemented with ALCAR had increased levels of noradrenaline and myo-inositol, and decreased GABA concentration in the hippocampus, and increased levels of serotonin in the cerebral cortex [107].
In preclinical studies supplementation with ALCAR improved learning and synaptic transmission in aged rats [130–132]. Other studies showed that high doses of ALCAR led to improvement after ischemia [133]. Ten minutes of forebrain ischemia in adult rats led to a significantly decreased number of intact neurons in hippocampal tissue, decreased concentrations of glutathione and ATP, and increased total nitrate/nitrite and thiobarbituric acid-reactive substances which accumulate in conditions of oxidative stress [133]. Rats treated with ALCAR (300 mg/kg) or propionylcarnitine (300 mg/kg) after ischemia had less loss of ATP and glutathione, more intact neurons, and decreased markers of oxidative stress and energy depletion in the hippocampal CA1 region 7 days after ischemia [133]. In a clinically relevant model of global ischemia after canine cardiac arrest, treatment with ALCAR reduced the amount of protein carbonyls which are formed after oxidative stress in brain [104].
Pediatric Models Using ALCAR Supplementation
Neonatal hypoxia ischemia, which occurs in ~1–4/1000 live births in the US, is a major cause of mortality and adverse neurodevelopmental outcome [134–137]. The effects of neonatal HI have been widely studied in the postnatal day 7 rat pup using the Rice-Vannucci method of carotid artery ligation combined with 8% O2 [37, 39, 138–145]. Our studies using 75 minutes exposure to 8% O2 result in a moderate injury and evidence of more impairment in male pups compared to females [39, 146, 147]. Our group has determined the neuroprotective effects of ALCAR (100 mg/kg administered by subcutaneous injection) at 0, 4, 24 and 48 hours after HI on postnatal day 7. In vivo imaging showed that treatment with ALCAR after HI led to a smaller lesion volume in brain determined at 3, 7 and 28 days after HI [39]. In vivo 1H-magnetic resonance spectroscopy (1H-MRS) in the same rat pups showed that treatment with ALCAR after HI improved lactate levels and maintained creatine concentration in the ipsilateral hippocampus compared to saline treated pups [37].
After HI on postnatal day 7 both male and female pups showed impairment in several measurements of social play; however, treatment with ALCAR did not rescue deficits in social play [39]. Treatment with ALCAR after HI led to improved performance on simple motor tests including negative geotaxis, which was impaired in both male and female pups [39]. Righting reflex and suspension on a dowel was impaired only in male pups after HI; ALCAR treatment improved performance on these tests [39]. Treatment with ALCAR after HI led to short term and long term improvement in novel object recognition in male pups compared to saline treated pups [39].
Using the same neonatal rat pup model, Demarest et al. [38] determined the effect of treatment with ALCAR on the functional impairment of mitochondria from brain at 20 hours after HI. Mitochondria from both hemispheres of male rat brain had more impairment of state 3 (Complex I dependent, ADP stimulated) respiration compared to mitochondria from female brain after HI, and the respiration was 30% lower in mitochondria from male brain after HI, than in sham rat pups [38]. Treatment of pups with ALCAR after HI prevented the impairment of Complex II dependent FCCP-uncoupled respiration in mitochondria isolated from the contralateral side of male brain and partially restored the FCCP uncoupled respiration changes in mitochondria from the contralateral side of female brain [38]. ALCAR administration after HI increased mitochondrial glutathione peroxidase activity in brain of male pups at 20 hours after injury. Treatment with ALCAR after HI decreased the significant increase in protein carbonyl formation that was found only in the brain of male pups in both hemispheres of the cerebral cortex, hippocampus and perirhinal cortex [38]. Using the postnatal day 7 rat pup model of HI described above, Demarest et al. [146] determined the effects of ALCAR on mitogenesis in brain. Pups treated with ALCAR after HI had a significant increase in the activity of citrate synthase in the ipsilateral hemisphere compared to sham pups and controls. HI led to a significant increase in the ratio of mitochondrial DNA to nuclear DNA (mtDNA/nDNA), in the ipsilateral side of brain in both male and female rat pups following HI [146]. This increase in mtDNA/nDNA ratio was prevented by treatment with ALCAR after HI; treated rats had ratios comparable to shams. Electron transport chain subunits of Complex I, II and IV were upregulated in female brain, but not in male brain following HI; treatment with ALCAR after HI had no effect on the level of the oxidative phosphorylation proteins [146].
Traumatic brain injury (TBI) is a major pediatric health problem with a rate of 500,000 per year in the US and leading to ~ 3000 pediatric deaths [134, 148]. Survivors of pediatric TBI frequently have long-term physical, social, psychological and cognitive impairments [149, 150] that can last into adulthood [151–153]. Additional therapies are needed to improve outcome after TBI in children. Scafidi et al. [12] found that young rats treated with ALCAR after controlled cortical impact traumatic brain injury (TBI) at 21–22 days of age had improved novel object recognition and motor function compared to rats treated with saline. Rats treated with ALCAR also had smaller lesion volumes at 7 days after TBI than rats treated with saline [12].
Thus treatment with ALCAR during the first 48 hours after HI on postnatal day 7, or after TBI on day 21–22 led to long-term protection of the ipsilateral hemisphere and improved behavioral outcome [12, 39]. It is important to note that there was no evidence that administration of ALCAR to 7 day old rat pups was harmful in either males or females studied until 35 days of age [39].
In recent years there have been increasing concerns about possible adverse effects of general anesthesia on the rapidly developing brains of infants and young children. Several studies have also reported that L-carnitine, and particularly ALCAR can protect the developing brain from deleterious effects of exposure to clinically used anesthetic agents [154–157]. Treatment with ALCAR protected from neuroinflammation and apoptosis resulting from anesthesia [154–157]. It is particularly important that some of these studies used newborn or very young nonhuman primates [154, 155, 157].
ALCAR and acetylcholine
A number of studies have shown that ALCAR administration maintains and/or increases acetylcholine levels in brain [101, 158–161]. De Simone et al. [158] found increased activity of choline acetyltransferase and expression of nerve growth factor (NGF) receptor in the striatum, and increased NGF protein levels in the hippocampus of rat pups after intracerebroventricular injection of ALCAR every other day from 0–21 days of age.
Maintaining acetylcholine levels is important as this neurotransmitter has a crucial role in learning and memory [162, 163]. Cholinergic pathways in the basal forebrain and hippocampus are necessary for attention, learning and memory [164]. Acetylcholine triggers hippocampal and cortical synaptic plasticity in part through astrocyte-neuron interactions [165]. The carnitine shuttle has a role in providing acetyl-CoA groups for acetylcholine synthesis (Figure 3), and in buffering the level of free coenzyme A in the cytosol which can inhibit acetylcholine synthesis via choline acetyltransferase [101].
Acetylcholine and/or cholinergic stimulation have an important role in protecting developing brain from inflammation. Furukawa et al. [166] demonstrated that pretreatment of 7 day old rat pups with the acetylcholinesterase inhibitor galantamine (5mg/kg injected i.p.) 2 hours prior to HI led to significantly reduced damage in the hippocampus and cortex at 7 days after injury [166]. Pretreatment of rat pups with galantamine also led to decreased microglial accumulation and less IL-1β in brain 7 days after HI. Another study by this group [167] reported that administration of multiple doses of the acetylcholine receptor agonist carbacol decreased microglial activation and inflammation, and decreased brain damage after hypoxia-ischemia in 7 day old rat pups. Furukawa et al. [168] suggested that cholinergic stimulation can ameliorate neonatal brain damage. Thus strategies to potentiate cholinergic transmission and/or acetylcholine levels could be neuroprotective in newborn brain.
Neuroprotection of developing brain
There is an urgent need for therapies that can improve outcome after neonatal and pediatric brain injury as current therapies are only partially effective [134, 135, 169–172]. Any therapies used must be safe for infants and children, and must also protect and/or support the complex and highly regulated process of normal brain development. Indeed, injury to the brain in children and/or infants can be particularly devastating because it is superimposed on the precisely orchestrated and energy demanding processes required for brain development [134]. The developing brain has high energy needs for basic cellular functions and for synthesis of neurotransmitters, nucleic acids, proteins, carbohydrates and lipids needed for cell growth and myelination [134]. Acute injury to pediatric brain can disrupt the complex and highly regulated normal developmental processes [134].
Effective neuroprotection involves attenuating inflammation, preventing energy failure, preventing oxidative damage to key cellular and mitochondrial proteins, and maintaining neuronal and glial functions and biosynthetic capabilities. Although L-carnitine fulfils some of these requirements, acetyl-L-carnitine fulfills these requirements and also has the additional capability to provide an acetyl-CoA that can be oxidized for energy, used as a precursor for acetylcholine, or incorporated into glutamate, glutamine, GABA, and into lipids for myelination and cell growth.
The capability of both L-carnitine and ALCAR treatment to decrease oxidative stress [37, 53, 103, 104] and subsequent oxidative DNA damage [86], and lipid peroxidation [86] is key to neuroprotection. [88]. Treatment with L-carnitine influenced signal transduction pathways in a preclinical adult brain injury model [86]; this has not been reported, and likely not yet studied with ALCAR. Both L-carnitine and ALCAR led to less loss of tissue in neonatal and/or young rats, supporting the efficacy for neuroprotection in developing brain [12, 19, 37, 39]. L-carnitine treatment protected the level of CPT I and CPT II, as well as oligodendrocytes and myelin [86]. The efficacy of ALCAR in protecting enzymes of the carnitine shuttle and myelin has not been addressed. Protection of oligodendrocytes, myelin and neurons would be particularly important after neonatal or pediatric brain injury.
Preclinical studies using a number of models of chronic and/or acute brain injury have shown that animals treated with ALCAR had improved energy metabolism [97] and mitochondrial respiration [147]. The ability of ALCAR to increase nerve growth factor [105] and enhance cholinergic activity may be particularly important for neuroprotection in developing brain [1, 106].
Protecting tissue from energy failure and oxidative stress that can lead to cell death is important; however, a crucial standard of preclinical efficacy is protection of functional outcomes. Thus, the evidence of preserved synaptic function [86] and long term improvement in learning in rats treated with L-carnitine after injury [86] is particularly important, and more studies determining functional outcomes are needed. In addition, more in vivo studies using treatment with L-carnitine after injury are needed since several studies used pretreatment and/or in vitro models.
There is compelling evidence that administration of ALCAR after injury in developing brain can attenuate injury and improve long term functional outcome [39]. The preclinical studies showing that treatment with ALCAR after perinatal HI, or after pediatric TBI improved motor function and both short and long term memory [12, 39] are particularly promising and translationally relevant. Additional studies using more complex learning paradigms would provide more insight into the potential therapeutic efficacy of ALCAR for protecting developing brain.
There is a crucial need for therapies that can lead to improved outcome after neonatal and pediatric brain injury as the currently used therapies are only partially effective [134, 135, 169–172]. Additional studies, particularly in vivo studies with long term functional outcomes are needed to better explore the potential of L-carnitine and ALCAR for protection of developing brain.
Acknowledgments
The authors would like to gratefully acknowledge and thank Bruna Klippel Ferreira for her excellent effort in preparing Figure 2 and Figure 3, and Dr. Jaylyn Waddell and Dr. Susanna Scafidi for their very helpful suggestions. Research described from Dr. McKenna’s laboratory was supported in part by NIH Grants 5P01 HD016596 and P01 HD085928. Dr. Ferreira’s research is supported by the "Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro", Coordination for the Improvement of Higher Education Personnel (CAPES) and National Council for Scientific and Technological Development (CNPq).
Abbreviations
- ALCAR
acetyl-L-carnitine
- OCTN2
organic cation transporter novel 2
- CPT I
carnitine palmitoyltransferase I
- CPT II
carnitine palmitoyltransferase II
- CAT
carnitine acetyltransferase
- MRS
magnetic resonance spectroscopy
- 13C-NMR
13C-nuclear magnetic resonance spectroscopy
- OGD
oxygen-glucose deprivation
- HI
hypoxia-ischemia
- TBI
traumatic brain injury
- 3-NPA
3-nitropropionic acid
- i.p.
intraperitoneal
- CSF
cerebrospinal fluid
- mTOR
mammalian target of rapamycin
- NGF
nerve growth factor
- pNFH
phosphorylated high-molecular weight neurofilament
- rCMRglc
cerebral regional metabolic rate of glucose
- TCA
tricarboxylic acid
References
- 1.Jones LL, McDonald DA, Borum PR. Acylcarnitines: role in brain. Prog Lipid Res. 2010;49:61–75. doi: 10.1016/j.plipres.2009.08.004. [DOI] [PubMed] [Google Scholar]
- 2.Marcovina SM, Sirtori C, Peracino A, Gheorghiade M, Borum P, Remuzzi G, Ardehali H. Translating the basic knowledge of mitochondrial functions to metabolic therapy: role of L-carnitine. Transl Res. 2013;161:73–84. doi: 10.1016/j.trsl.2012.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ribas GS, Vargas CR, Wajner M. L-carnitine supplementation as a potential antioxidant therapy for inherited neurometabolic disorders. Gene. 2014;533:469–76. doi: 10.1016/j.gene.2013.10.017. [DOI] [PubMed] [Google Scholar]
- 4.Alves E, Binienda Z, Carvalho F, Alves CJ, Fernandes E, de Lourdes Bastos M, Tavares MA, Summavielle T. Acetyl-L-carnitine provides effective in vivo neuroprotection over 3,4-methylenedioximethamphetamine-induced mitochondrial neurotoxicity in the adolescent rat brain. Neuroscience. 2009;158:514–23. doi: 10.1016/j.neuroscience.2008.10.041. [DOI] [PubMed] [Google Scholar]
- 5.Calabrese V, Giuffrida Stella AM, Calvani M, Butterfield DA. Acetylcarnitine and cellular stress response: roles in nutritional redox homeostasis and regulation of longevity genes. J Nutr Biochem. 2006;17:73–88. doi: 10.1016/j.jnutbio.2005.03.027. [DOI] [PubMed] [Google Scholar]
- 6.Cahova M, Chrastina P, Hansikova H, Drahota Z, Trnovska J, Skop V, Spacilova J, Malinska H, Oliyarnyk O, Papackova Z, Palenickova E, Kazdova L. Carnitine supplementation alleviates lipid metabolism derangements and protects against oxidative stress in non-obese hereditary hypertriglyceridemic rats. Appl Physiol Nutr Metab. 2015;40:280–91. doi: 10.1139/apnm-2014-0163. [DOI] [PubMed] [Google Scholar]
- 7.Zanelli SA, Solenski NJ, Rosenthal RE, Fiskum G. Mechanisms of ischemic neuroprotection by acetyl-L-carnitine. Ann N Y Acad Sci. 2005;1053:153–161. doi: 10.1196/annals.1344.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zaitone SA, Abo-Elmatty DM, Shaalan AA. Acetyl-L-carnitine and alpha-lipoic acid affect rotenone-induced damage in nigral dopaminergic neurons of rat brain, implication for Parkinson's disease therapy. Pharmacol Biochem Behav. 2012;100:347–60. doi: 10.1016/j.pbb.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 9.Wilson AD, Hart A, Brannstrom T, Wiberg M, Terenghi G. Delayed acetyl-L-carnitine administration and its effect on sensory neuronal rescue after peripheral nerve injury. J Plast Reconstr Aesthet Surg. 2007;60:114–8. doi: 10.1016/j.bjps.2006.04.017. [DOI] [PubMed] [Google Scholar]
- 10.Virmani A, Koverech A, Ali SF, Binienda ZK. Acetyl-L-Carnitine Modulates TP53 and IL10 Gene Expression Induced by 3-NPA Evoked Toxicity in PC12 Cells. Curr Neuropharmacol. 2011;9:195–9. doi: 10.2174/157015911795017182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Suchy J, Chan A, Shea TB. Dietary supplementation with a combination of alpha-lipoic acid, acetyl-L-carnitine, glycerophosphocoline, docosahexaenoic acid, and phosphatidylserine reduces oxidative damage to murine brain and improves cognitive performance. Nutr Res. 2009;29:70–4. doi: 10.1016/j.nutres.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 12.Scafidi S, Racz J, Hazelton J, McKenna MC, Fiskum G. Neuroprotection by acetyl-L-carnitine after traumatic injury to the immature rat brain. Dev Neurosci. 2010;32:480–487. doi: 10.1159/000323178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patel SP, Sullivan PG, Lyttle TS, Magnuson DS, Rabchevsky AG. Acetyl-L-carnitine treatment following spinal cord injury improves mitochondrial function correlated with remarkable tissue sparing and functional recovery. Neuroscience. 2012;210:296–307. doi: 10.1016/j.neuroscience.2012.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kocsis K, Knapp L, Meszaros J, Kis Z, Farkas T, Vecsei L, Toldi J. Acetyl-L-carnitine and oxaloacetate in post-treatment against LTP impairment in a rat ischemia model. An in vitro electrophysiological study. J Neural Transm (Vienna) 2015;122:867–72. doi: 10.1007/s00702-014-1343-7. [DOI] [PubMed] [Google Scholar]
- 15.Hota KB, Hota SK, Chaurasia OP, Singh SB. Acetyl-L-carnitine-mediated neuroprotection during hypoxia is attributed to ERK1/2-Nrf2-regulated mitochondrial biosynthesis. Hippocampus. 2012;22:723–736. doi: 10.1002/hipo.20934. [DOI] [PubMed] [Google Scholar]
- 16.Chan A, Shea TB. Effects of dietary supplementation with N-acetyl cysteine, acetyl-L-carnitine and S-adenosyl methionine on cognitive performance and aggression in normal mice and mice expressing human ApoE4. Neuromolecular Med. 2007;9:264–9. doi: 10.1007/s12017-007-8005-y. [DOI] [PubMed] [Google Scholar]
- 17.Barhwal K, Hota SK, Prasad D, Singh SB, Ilavazhagan G. Hypoxia-induced deactivation of NGF-mediated ERK1/2 signaling in hippocampal cells: neuroprotection by acetyl-L-carnitine. J Neurosci Res. 2008;86:2705–21. doi: 10.1002/jnr.21722. [DOI] [PubMed] [Google Scholar]
- 18.Ishii T, Shimpo Y, Matsuoka Y, Kinoshita K. Anti-apoptotic effect of acetyl-l-carnitine and I-carnitine in primary cultured neurons. Jpn J Pharmacol. 2000;83:119–124. doi: 10.1254/jjp.83.119. [DOI] [PubMed] [Google Scholar]
- 19.Wainwright MS, Mannix MK, Brown J, Stumpf DA. L-carnitine reduces brain injury after hypoxia-ischemia in newborn rats. Pediatr Res. 2003;54:688–95. doi: 10.1203/01.PDR.0000085036.07561.9C. [DOI] [PubMed] [Google Scholar]
- 20.Wainwright MS, Kohli R, Whitington PF, Chace DH. Carnitine treatment inhibits increases in cerebral carnitine esters and glutamate detected by mass spectrometry after hypoxia-ischemia in newborn rats. Stroke. 2006;37:524–30. doi: 10.1161/01.STR.0000198892.15269.f7. [DOI] [PubMed] [Google Scholar]
- 21.Roe CR, Millington DS, Maltby DA, Bohan TP, Hoppel CL. L-carnitine enhances excretion of propionyl coenzyme A as propionylcarnitine in propionic acidemia. J Clin Invest. 1984;73:1785–8. doi: 10.1172/JCI111387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang R, Zhang H, Zhang Z, Wang T, Niu J, Cui D, Xu S. Neuroprotective effects of pre-treatment with l-carnitine and acetyl-L-carnitine on ischemic injury in vivo and in vitro. Int J Mol Sci. 2012;13:2078–2090. doi: 10.3390/ijms13022078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vieira Neto E, Fonseca AA, Almeida RF, Figueiredo MP, Porto MA, Ribeiro MG. Analysis of acylcarnitine profiles in umbilical cord blood and during the early neonatal period by electrospray ionization tandem mass spectrometry. Braz J Med Biol Res. 2012;45:546–56. doi: 10.1590/S0100-879X2012007500056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schmidt-Sommerfeld E, Penn D, Kerner J, Bieber LL, Rossi TM, Lebenthal E. Quantitation of urinary carnitine esters in a patient with medium-chain acyl-coenzyme A dehydrogenase deficiency: effect of metabolic state and L-carnitine therapy. J Pediatr. 1989;115:577–82. doi: 10.1016/s0022-3476(89)80284-7. [DOI] [PubMed] [Google Scholar]
- 25.Roe CR, Hoppel CL, Stacey TE, Chalmers RA, Tracey BM, Millington DS. Metabolic response to carnitine in methylmalonic aciduria. An effective strategy for elimination of propionyl groups. Arch Dis Child. 1983;58:916–20. doi: 10.1136/adc.58.11.916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rashed MS, Ozand PT, Bucknall MP, Little D. Diagnosis of inborn errors of metabolism from blood spots by acylcarnitines and amino acids profiling using automated electrospray tandem mass spectrometry. Pediatr Res. 1995;38:324–31. doi: 10.1203/00006450-199509000-00009. [DOI] [PubMed] [Google Scholar]
- 27.Poorthuis BJ, Jille-Vlckova T, Onkenhout W. Determination of acylcarnitines in urine of patients with inborn errors of metabolism using high-performance liquid chromatography after derivatization with 4’-bromophenacylbromide. Clin Chim Acta. 1993;216:53–61. doi: 10.1016/0009-8981(93)90138-t. [DOI] [PubMed] [Google Scholar]
- 28.Okun JG, Kolker S, Schulze A, Kohlmuller D, Olgemoller K, Lindner M, Hoffmann GF, Wanders RJ, Mayatepek E. A method for quantitative acylcarnitine profiling in human skin fibroblasts using unlabelled palmitic acid: diagnosis of fatty acid oxidation disorders and differentiation between biochemical phenotypes of MCAD deficiency. Biochim Biophys Acta. 2002;1584:91–8. doi: 10.1016/s1388-1981(02)00296-2. [DOI] [PubMed] [Google Scholar]
- 29.Novak M, Monkus EF, Chung D, Buch M. Carnitine in the perinatal metabolism of lipids. I. Relationship between maternal and fetal plasma levels of carnitine and acylcarnitines. Pediatrics. 1981;67:95–100. [PubMed] [Google Scholar]
- 30.Novak M, Monkus EF, Buch M, Silverio J, Clouston OM, Cassady JC. L-carnitine supplementation of a soybean-based formula in early infancy: plasma and urine levels of carnitine and acylcarnitines. J Pediatr Gastroenterol Nutr. 1988;7:220–4. doi: 10.1097/00005176-198803000-00011. [DOI] [PubMed] [Google Scholar]
- 31.Minkler PE, Hoppel CL. Quantification of carnitine and specific acylcarnitines by high-performance liquid chromatography: application to normal human urine and urine from patients with methylmalonic aciduria, isovaleric acidemia or medium-chain acyl-CoA dehydrogenase deficiency. J Chromatogr. 1993;613:203–21. doi: 10.1016/0378-4347(93)80135-q. [DOI] [PubMed] [Google Scholar]
- 32.Matsumoto K, Takahashi M, Takiyama N, Misaki H, Matsuo N, Murano S, Yuki H. Enzyme reactor for urinary acylcarnitines assay by reversed-phase high-performance liquid chromatography. Clin Chim Acta. 1993;216:135–43. doi: 10.1016/0009-8981(93)90146-u. [DOI] [PubMed] [Google Scholar]
- 33.Lloyd-Still JD, Powers CA, Wessel HU. Carnitine metabolites in infants with cystic fibrosis: a prospective study. Acta Paediatr. 1993;82:145–9. doi: 10.1111/j.1651-2227.1993.tb12626.x. [DOI] [PubMed] [Google Scholar]
- 34.Kidouchi K, Sugiyama N, Morishita H, Kobayashi M, Wada Y, Nohara D. Identification of glutarylcarnitine in glutaric aciduria type 1 by carboxylic acid analyzer with an ODS reverse-phase column. Clin Chim Acta. 1987;164:261–6. doi: 10.1016/0009-8981(87)90300-7. [DOI] [PubMed] [Google Scholar]
- 35.Invernizzi F, Burlina AB, Donadio A, Giordano G, Taroni F, Garavaglia B. Lethal neonatal presentation of carnitine palmitoyltransferase I deficiency. J Inherit Metab Dis. 2001;24:601–2. doi: 10.1023/a:1012476029536. [DOI] [PubMed] [Google Scholar]
- 36.Hori T, Fukao T, Kobayashi H, Teramoto T, Takayanagi M, Hasegawa Y, Yasuno T, Yamaguchi S, Kondo N. Carnitine palmitoyltransferase 2 deficiency: the time- course of blood and urinary acylcarnitine levels during initial L-carnitine supplementation. Tohoku J Exp Med. 2010;221:191–5. doi: 10.1620/tjem.221.191. [DOI] [PubMed] [Google Scholar]
- 37.Xu S, Waddell J, Zhu W, Shi D, Marshall AD, McKenna MC, Gullapalli RP. In vivo longitudinal proton magnetic resonance spectroscopy on neonatal hypoxic-ischemic rat brain injury: Neuroprotective effects of acetyl-L-carnitine. Magn Reson Med. 2015;74:1530–1542. doi: 10.1002/mrm.25537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Demarest TG, Schuh RA, Waddell J, McKenna MC, Fiskum G. Sex dependent mitochondrial respiratory impairment and oxidative stress in a rat model of neonatal hypoxic-ischemic encephalopathy. J Neurochem. 2016;137:714–729. doi: 10.1111/jnc.13590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tang S, Xu S, Lu X, Gullapalli RP, McKenna MC, Waddell J. Neuroprotective Effects of Acetyl-L-Carnitine on Neonatal Hypoxia Ischemia-Induced Brain Injury in Rats. Dev Neurosci. 2016;38:384–396. doi: 10.1159/000455041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vaz FM, Wanders RJ. Carnitine biosynthesis in mammals. Biochem J. 2002;361:417–29. doi: 10.1042/0264-6021:3610417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.El-Hattab AW, Scaglia F. Disorders of carnitine biosynthesis and transport. Mol Genet Metab. 2015;116:107–12. doi: 10.1016/j.ymgme.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 42.Longo N, Amat di San Filippo C, Pasquali M. Disorders of carnitine transport and the carnitine cycle. Am J Med Genet C Semin Med Genet. 2006;142C:77–85. doi: 10.1002/ajmg.c.30087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Szabo K, Nagy Z, Juhasz V, Zolnerciks JK, Csorba A, Timar Z, Molnar E, Padar P, Johnson W, Beery E, Krajcsi P. Species specificity profiling of rat and human organic cation/carnitine transporter Slc22a5/SLC22A5 (Octn2/OCTN2) Drug Metab Pharmacokinet. 2016 doi: 10.1016/j.dmpk.2016.08.005. [DOI] [PubMed] [Google Scholar]
- 44.Nalecz KA, Miecz D, Berezowski V, Cecchelli R. Carnitine: transport and physiological functions in the brain. Mol Aspects Med. 2004;25:551–67. doi: 10.1016/j.mam.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 45.Nalecz KA, Nalecz MJ. Carnitine--a known compound, a novel function in neural cells. Acta Neurobiol Exp (Wars) 1996;56:597–609. doi: 10.55782/ane-1996-1165. [DOI] [PubMed] [Google Scholar]
- 46.Inazu M, Matsumiya T. [Physiological functions of carnitine and carnitine transporters in the central nervous system] Nihon Shinkei Seishin Yakurigaku Zasshi. 2008;28:113–20. [PubMed] [Google Scholar]
- 47.Inazu M, Takeda H, Maehara K, Miyashita K, Tomoda A, Matsumiya T. Functional expression of the organic cation/carnitine transporter 2 in rat astrocytes. J Neurochem. 2006;97:424–34. doi: 10.1111/j.1471-4159.2006.03757.x. [DOI] [PubMed] [Google Scholar]
- 48.Miecz D, Januszewicz E, Czeredys M, Hinton BT, Berezowski V, Cecchelli R, Nalecz KA. Localization of organic cation/carnitine transporter (OCTN2) in cells forming the blood-brain barrier. J Neurochem. 2008;104:113–23. doi: 10.1111/j.1471-4159.2007.05024.x. [DOI] [PubMed] [Google Scholar]
- 49.Czeredys M, Mysiorek C, Kulikova N, Samluk L, Berezowski V, Cecchelli R, Nalecz KA. A polarized localization of amino acid/carnitine transporter B(0,+) (ATB(0,+)) in the blood-brain barrier. Biochem Biophys Res Commun. 2008;376:267–70. doi: 10.1016/j.bbrc.2008.08.122. [DOI] [PubMed] [Google Scholar]
- 50.Czeredys M, Samluk L, Michalec K, Tulodziecka K, Skowronek K, Nalecz KA. Caveolin-1--a novel interacting partner of organic cation/carnitine transporter (Octn2): effect of protein kinase C on this interaction in rat astrocytes. PLoS One. 2013;8:e82105. doi: 10.1371/journal.pone.0082105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lamhonwah AM, Hawkins CE, Tam C, Wong J, Mai L, Tein I. Expression patterns of the organic cation/carnitine transporter family in adult murine brain. Brain Dev. 2008;30:31–42. doi: 10.1016/j.braindev.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 52.Januszewicz E, Bekisz M, Mozrzymas JW, Nalecz KA. High affinity carnitine transporters from OCTN family in neural cells. Neurochem Res. 2010;35:743–8. doi: 10.1007/s11064-010-0131-5. [DOI] [PubMed] [Google Scholar]
- 53.Reuter SE, Evans AM. Carnitine and acylcarnitines: pharmacokinetic, pharmacological and clinical aspects. Clin Pharmacokinet. 2012;51:553–72. doi: 10.1007/BF03261931. [DOI] [PubMed] [Google Scholar]
- 54.Rau TF, Lu Q, Sharma S, Sun X, Leary G, Beckman ML, Hou Y, Wainwright MS, Kavanaugh M, Poulsen DJ, Black SM. Oxygen glucose deprivation in rat hippocampal slice cultures results in alterations in carnitine homeostasis and mitochondrial dysfunction. PLoS One. 2012;7:e40881. doi: 10.1371/journal.pone.0040881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Muoio DM, Noland RC, Kovalik JP, Seiler SE, Davies MN, DeBalsi KL, Ilkayeva OR, Stevens RD, Kheterpal I, Zhang J, Covington JD, Bajpeyi S, Ravussin E, Kraus W, Koves TR, Mynatt RL. Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility. Cell Metab. 2012;15:764–77. doi: 10.1016/j.cmet.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kawamura N. Regulation of fatty acid oxidation in rat brain mitochondria: inhibition of high rates of palmitate oxidation by ADP. Arch Biochem Biophys. 1988;264:546–52. doi: 10.1016/0003-9861(88)90320-7. [DOI] [PubMed] [Google Scholar]
- 57.Scafidi S, Fiskum G, Lindauer SL, Bamford P, Shi D, Hopkins I, McKenna MC. Metabolism of acetyl-L-carnitine for energy and neurotransmitter synthesis in the immature rat brain. J Neurochem. 2010;114:820–831. doi: 10.1111/j.1471-4159.2010.06807.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shinohara M, Saitoh M, Takanashi J, Yamanouchi H, Kubota M, Goto T, Kikuchi M, Shiihara T, Yamanaka G, Mizuguchi M. Carnitine palmitoyl transferase II polymorphism is associated with multiple syndromes of acute encephalopathy with various infectious diseases. Brain Dev. 2011;33:512–7. doi: 10.1016/j.braindev.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 59.Sakai E, Yamanaka G, Kawashima H, Morishima Y, Ishida Y, Oana S, Miyajima T, Shinohara M, Saitoh M, Mizuguchi M. A case of recurrent acute encephalopathy with febrile convulsive status epilepticus with carnitine palmitoyltransferase II variation. Neuropediatrics. 2013;44:218–21. doi: 10.1055/s-0032-1333439. [DOI] [PubMed] [Google Scholar]
- 60.Celestino-Soper PB, Violante S, Crawford EL, Luo R, Lionel AC, Delaby E, Cai G, Sadikovic B, Lee K, Lo C, Gao K, Person RE, Moss TJ, German JR, Huang N, Shinawi M, Treadwell-Deering D, Szatmari P, Roberts W, Fernandez B, Schroer RJ, Stevenson RE, Buxbaum JD, Betancur C, Scherer SW, Sanders SJ, Geschwind DH, Sutcliffe JS, Hurles ME, Wanders RJ, Shaw CA, Leal SM, Cook EH, Jr, Goin-Kochel RP, Vaz FM, Beaudet AL, et al. A common X-linked inborn error of carnitine biosynthesis may be a risk factor for nondysmorphic autism. Proc Natl Acad Sci U S A. 2012;109:7974–81. doi: 10.1073/pnas.1120210109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rashidi-Nezhad A, Talebi S, Saebnouri H, Akrami SM, Reymond A. The effect of homozygous deletion of the BBOX1 and Fibin genes on carnitine level and acyl carnitine profile. BMC Med Genet. 2014;15:75. doi: 10.1186/1471-2350-15-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Longo N. Primary Carnitine Deficiency and Newborn Screening for Disorders of the Carnitine Cycle. Ann Nutr Metab. 2016;(68 Suppl 3):5–9. doi: 10.1159/000448321. [DOI] [PubMed] [Google Scholar]
- 63.Stanley CA, Palmieri F, MJ B. Disorders of the Mitochondrial Carnitine Shuttle. In: Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Antonarakis SE, Ballabio A, Gibson KMG, et al., editors. The Online Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill: New York: 2014. [Google Scholar]
- 64.Stanley CA, Bennett MJNL. Plasma Membrane Carnitine Transporter Defect. In: Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Antonarakis SE, Ballabio A, Gibson KMG, et al., editors. The Online Metabolic and Molecular Bases of Inherited Disease New York. McGraw-Hill: New York: 2014. [Google Scholar]
- 65.Angelini C, Trevisan C, Isaya G, Pegolo G, Vergani L. Clinical varieties of carnitine and carnitine palmitoyltransferase deficiency. Clin Biochem. 1987;20:1–7. doi: 10.1016/s0009-9120(87)80090-5. [DOI] [PubMed] [Google Scholar]
- 66.Schuck PF, Ferreira Gda C, Tahara EB, Klamt F, Kowaltowski AJ, Wajner M. cis-4-decenoic acid provokes mitochondrial bioenergetic dysfunction in rat brain. Life Sci. 2010;87:139–46. doi: 10.1016/j.lfs.2010.05.019. [DOI] [PubMed] [Google Scholar]
- 67.Ferreira GC, Tonin A, Schuck PF, Viegas CM, Ceolato PC, Latini A, Perry ML, Wyse AT, Dutra-Filho CS, Wannmacher CM, Vargas CR, Wajner M. Evidence for a synergistic action of glutaric and 3-hydroxyglutaric acids disturbing rat brain energy metabolism. Int J Dev Neurosci. 2007;25:391–8. doi: 10.1016/j.ijdevneu.2007.05.009. [DOI] [PubMed] [Google Scholar]
- 68.Schuck PF, Milanez AP, Felisberto F, Galant LS, Machado JL, Furlanetto CB, Petronilho F, Dal-Pizzol F, Streck EL, Ferreira GC. Brain and muscle redox imbalance elicited by acute ethylmalonic acid administration. PLoS One. 2015;10:e0126606. doi: 10.1371/journal.pone.0126606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Spiekerkoetter U, Wood PA. Mitochondrial fatty acid oxidation disorders: pathophysiological studies in mouse models. J Inherit Metab Dis. 2010;33:539–46. doi: 10.1007/s10545-010-9121-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Stanley CA. Carnitine disorders. Adv Pediatr. 1995;42:209–42. [PubMed] [Google Scholar]
- 71.Badve MS, Bhuta S, McGill J. Rare presentation of a treatable disorder: glutaric aciduria type 1. N Z Med J. 2015;128:61–4. [PubMed] [Google Scholar]
- 72.Davies SE, Iles RA, Stacey TE, de Sousa C, Chalmers RA. Carnitine therapy and metabolism in the disorders of propionyl-CoA metabolism studied using 1H-NMR spectroscopy. Clin Chim Acta. 1991;204:263–77. doi: 10.1016/0009-8981(91)90237-7. [DOI] [PubMed] [Google Scholar]
- 73.Hoffmann GF, Athanassopoulos S, Burlina AB, Duran M, de Klerk JB, Lehnert W, Leonard JV, Monavari AA, Muller E, Muntau AC, Naughten ER, Plecko-Starting B, Superti-Furga A, Zschocke J, Christensen E. Clinical course, early diagnosis, treatment, and prevention of disease in glutaryl-CoA dehydrogenase deficiency. Neuropediatrics. 1996;27:115–23. doi: 10.1055/s-2007-973761. [DOI] [PubMed] [Google Scholar]
- 74.Kolker S, Christensen E, Leonard JV, Greenberg CR, Boneh A, Burlina AB, Burlina AP, Dixon M, Duran M, Garcia Cazorla A, Goodman SI, Koeller DM, Kyllerman M, Muhlhausen C, Muller E, Okun JG, Wilcken B, Hoffmann GF, Burgard P. Diagnosis and management of glutaric aciduria type I--revised recommendations. J Inherit Metab Dis. 2011;34:677–94. doi: 10.1007/s10545-011-9289-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wolff JA, Carroll JE, Le Phuc T, Prodanos C, Haas R, Nyhan WL. Carnitine reduces fasting ketogenesis in patients with disorders of propionate metabolism. Lancet. 1986;1:289–91. doi: 10.1016/s0140-6736(86)90826-3. [DOI] [PubMed] [Google Scholar]
- 76.Sitta A, Vanzin CS, Biancini GB, Manfredini V, de Oliveira AB, Wayhs CA, Ribas GO, Giugliani L, Schwartz IV, Bohrer D, Garcia SC, Wajner M, Vargas CR. Evidence that L-carnitine and selenium supplementation reduces oxidative stress in phenylketonuric patients. Cell Mol Neurobiol. 2011;31:429–36. doi: 10.1007/s10571-010-9636-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Mescka CP, Wayhs CA, Vanzin CS, Biancini GB, Guerreiro G, Manfredini V, Souza C, Wajner M, Dutra-Filho CS, Vargas CR. Protein and lipid damage in maple syrup urine disease patients: l-carnitine effect. Int J Dev Neurosci. 2013;31:21–4. doi: 10.1016/j.ijdevneu.2012.10.109. [DOI] [PubMed] [Google Scholar]
- 78.Ribas GS, Manfredini V, de Mari JF, Wayhs CY, Vanzin CS, Biancini GB, Sitta A, Deon M, Wajner M, Vargas CR. Reduction of lipid and protein damage in patients with disorders of propionate metabolism under treatment: a possible protective role of L-carnitine supplementation. Int J Dev Neurosci. 2010;28:127–32. doi: 10.1016/j.ijdevneu.2010.01.002. [DOI] [PubMed] [Google Scholar]
- 79.Al-sharefi A, Bilous R. Reversible weakness and encephalopathy while on long-term valproate treatment due to carnitine deficiency. BMJ Case Rep. 2015 doi: 10.1136/bcr-2015-210727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kim H, Chu K, Jung KH, Lee ST, Kim JM, Lee SK. Acquired encephalopathy associated with carnitine deficiency after cefditoren pivoxil administration. Neurol Sci. 2012;33:1393–6. doi: 10.1007/s10072-012-0939-7. [DOI] [PubMed] [Google Scholar]
- 81.Stanley CA. Carnitine deficiency disorders in children. Ann N Y Acad Sci. 2004;1033:42–51. doi: 10.1196/annals.1320.004. [DOI] [PubMed] [Google Scholar]
- 82.Clark RH, Kelleher AS, Chace DH, Spitzer AR. Gestational age and age at sampling influence metabolic profiles in premature infants. Pediatrics. 2014;134:e37–46. doi: 10.1542/peds.2014-0329. [DOI] [PubMed] [Google Scholar]
- 83.Limketkai BN, Zucker SD. Hyperammonemic encephalopathy caused by carnitine deficiency. J Gen Intern Med. 2008;23:210–3. doi: 10.1007/s11606-007-0473-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Karakoc E, Erdem S, Sokmensuer C, Kansu T. Encephalopathy due to carnitine deficiency in an adult patient with gluten enteropathy. Clin Neurol Neurosurg. 2006;108:794–7. doi: 10.1016/j.clineuro.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 85.Schols L, Zange J, Abele M, Schillings M, Skipka G, Kuntz-Hehner S, van Beekvelt MC, Colier WN, Muller K, Klockgether T, Przuntek H, Vorgerd M. L-carnitine and creatine in Friedreich’s ataxia. A randomized, placebo-controlled crossover trial. J Neural Transm (Vienna) 2005;112:789–96. doi: 10.1007/s00702-004-0216-x. [DOI] [PubMed] [Google Scholar]
- 86.Ueno Y, Koike M, Shimada Y, Shimura H, Hira K, Tanaka R, Uchiyama Y, Hattori N, Urabe T. L-carnitine enhances axonal plasticity and improves white-matter lesions after chronic hypoperfusion in rat brain. J Cereb Blood Flow Metab. 2015;35:382–91. doi: 10.1038/jcbfm.2014.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Binienda ZK, Ali SF, Virmani A, Amato A, Salem N, Przybyla BD. Co-regulation of dopamine D1 receptor and uncoupling protein-2 expression in 3-nitropropionic acid-induced neurotoxicity: neuroprotective role of L-carnitine. Neurosci Lett. 2006;410:62–5. doi: 10.1016/j.neulet.2006.09.070. [DOI] [PubMed] [Google Scholar]
- 88.Yu Z, Iryo Y, Matsuoka M, Igisu H, Ikeda M. Suppression of pentylenetetrazol-induced seizures by carnitine in mice. Naunyn Schmiedebergs Arch Pharmacol. 1997;355:545–9. doi: 10.1007/pl00004981. [DOI] [PubMed] [Google Scholar]
- 89.Pande SV, Blanchaer MC. Reversible inhibition of mitochondrial adenosine diphosphate phosphorylation by long chain acyl coenzyme A esters. J Biol Chem. 1971;246:402–11. [PubMed] [Google Scholar]
- 90.Shug AL, Shrago E, Bittar N, Folts JD, Koke JR. Acyl-CoA inhibition of adenine nucleotide translocation in ischemic myocardium. Am J Physiol. 1975;228:689–92. doi: 10.1152/ajplegacy.1975.228.3.689. [DOI] [PubMed] [Google Scholar]
- 91.Stumpf DA, McAfee J, Parks JK, Eguren L. Propionate inhibition of succinate:CoA ligase (GDP) and the citric acid cycle in mitochondria. Pediatr Res. 1980;14:1127–31. doi: 10.1203/00006450-198010000-00008. [DOI] [PubMed] [Google Scholar]
- 92.Matsuishi T, Stumpf DA, Seliem M, Eguren LA, Chrislip K. Propionate mitochondrial toxicity in liver and skeletal muscle: acyl CoA levels. Biochem Med Metab Biol. 1991;45:244–53. doi: 10.1016/0885-4505(91)90027-i. [DOI] [PubMed] [Google Scholar]
- 93.Chalmers RA, Roe CR, Stacey TE, Hoppel CL. Urinary excretion of l-carnitine and acylcarnitines by patients with disorders of organic acid metabolism: evidence for secondary insufficiency of l-carnitine. Pediatr Res. 1984;18:1325–8. doi: 10.1203/00006450-198412000-00021. [DOI] [PubMed] [Google Scholar]
- 94.Roe CR, Millington DS, Maltby DA, Bohan TP, Kahler SG, Chalmers RA. Diagnostic and therapeutic implications of medium-chain acylcarnitines in the medium-chain acyl-coA dehydrogenase deficiency. Pediatr Res. 1985;19:459–66. doi: 10.1203/00006450-198505000-00011. [DOI] [PubMed] [Google Scholar]
- 95.Frye RE, Melnyk S, Macfabe DF. Unique acyl-carnitine profiles are potential biomarkers for acquired mitochondrial disease in autism spectrum disorder. Transl Psychiatry. 2013;3:e220. doi: 10.1038/tp.2012.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Thomas RH, Foley KA, Mepham JR, Tichenoff LJ, Possmayer F, MacFabe DF. Altered brain phospholipid and acylcarnitine profiles in propionic acid infused rodents: further development of a potential model of autism spectrum disorders. J Neurochem. 2010;113:515–29. doi: 10.1111/j.1471-4159.2010.06614.x. [DOI] [PubMed] [Google Scholar]
- 97.Rosenthal RE, Williams R, Bogaert YE, Getson PR, Fiskum G. Prevention of postischemic canine neurological injury through potentiation of brain energy metabolism by acetyl-L-carnitine. Stroke. 1992;23:1312–1317. doi: 10.1161/01.str.23.9.1312. [DOI] [PubMed] [Google Scholar]
- 98.Virmani MA, Caso V, Spadoni A, Rossi S, Russo F, Gaetani F. The action of acetyl-L-carnitine on the neurotoxicity evoked by amyloid fragments and peroxide on primary rat cortical neurones. Ann N Y Acad Sci. 2001;939:162–178. doi: 10.1111/j.1749-6632.2001.tb03623.x. [DOI] [PubMed] [Google Scholar]
- 99.Calabrese V, Ravagna A, Colombrita C, Scapagnini G, Guagliano E, Calvani M, Butterfield DA, Giuffrida Stella AM. Acetylcarnitine induces heme oxygenase in rat astrocytes and protects against oxidative stress: involvement of the transcription factor Nrf2. J Neurosci Res. 2005;79:509–521. doi: 10.1002/jnr.20386. [DOI] [PubMed] [Google Scholar]
- 100.Aureli T, Miccheli A, Di Cocco ME, Ghirardi O, Giuliani A, Ramacci MT, Conti F. Effect of acetyl-L-carnitine on recovery of brain phosphorus metabolites and lactic acid level during reperfusion after cerebral ischemia in the rat--study by 13P- and 1H–NMR spectroscopy. Brain Res. 1994;643:92–99. doi: 10.1016/0006-8993(94)90013-2. [DOI] [PubMed] [Google Scholar]
- 101.White HL, Scates PW. Acetyl-L-carnitine as a precursor of acetylcholine. Neurochem Res. 1990;15:597–601. doi: 10.1007/BF00973749. [DOI] [PubMed] [Google Scholar]
- 102.Ricciolini R, Scalibastri M, Kelleher JK, Carminati P, Calvani M, Arduini A. Role of acetyl-L-carnitine in rat brain lipogenesis: implications for polyunsaturated fatty acid biosynthesis. J Neurochem. 1998;71:2510–7. doi: 10.1046/j.1471-4159.1998.71062510.x. [DOI] [PubMed] [Google Scholar]
- 103.Bogaert YE, Rosenthal RE, Fiskum G. Postischemic inhibition of cerebral cortex pyruvate dehydrogenase. Free Radic Biol Med. 1994;16:811–820. doi: 10.1016/0891-5849(94)90197-x. [DOI] [PubMed] [Google Scholar]
- 104.Liu Y, Rosenthal RE, Starke-Reed P, Fiskum G. Inhibition of postcardiac arrest brain protein oxidation by acetyl-L-carnitine. Free Radic Biol Med. 1993;15:667–670. doi: 10.1016/0891-5849(93)90171-p. [DOI] [PubMed] [Google Scholar]
- 105.Chiechio S, Copani A, Nicoletti F, Gereau RWt. L-acetylcarnitine: a proposed therapeutic agent for painful peripheral neuropathies. Curr Neuropharmacol. 2006;4:233–237. doi: 10.2174/157015906778019509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Janiri L, Falcone M, Persico A, Tempesta E. Activity of L-carnitine and L-acetylcarnitine on cholinoceptive neocortical neurons of the rat in vivo. J Neural Transm Gen Sect. 1991;86:135–46. doi: 10.1007/BF01250574. [DOI] [PubMed] [Google Scholar]
- 107.Smeland OB, Meisingset TW, Borges K, Sonnewald U. Chronic acetyl-L-carnitine alters brain energy metabolism and increases noradrenaline and serotonin content in healthy mice. Neurochem Int. 2012;61:100–7. doi: 10.1016/j.neuint.2012.04.008. [DOI] [PubMed] [Google Scholar]
- 108.Nasca C, Xenos D, Barone Y, Caruso A, Scaccianoce S, Matrisciano F, Battaglia G, Mathe AA, Pittaluga A, Lionetto L, Simmaco M, Nicoletti F. L-acetylcarnitine causes rapid antidepressant effects through the epigenetic induction of mGlu2 receptors. Proc Natl Acad Sci U S A. 2013;110:4804–9. doi: 10.1073/pnas.1216100110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Madiraju P, Pande SV, Prentki M, Madiraju SR. Mitochondrial acetylcarnitine provides acetyl groups for nuclear histone acetylation. Epigenetics. 2009;4:399–403. doi: 10.4161/epi.4.6.9767. [DOI] [PubMed] [Google Scholar]
- 110.McKenna MC, Ferreira GC. Enzyme Complexes Important for the Glutamate-Glutamine Cycle. Adv Neurobiol. 2016;13:59–98. doi: 10.1007/978-3-319-45096-4_4. [DOI] [PubMed] [Google Scholar]
- 111.McKenna MC, Rae CD. A new role for alpha-ketoglutarate dehydrogenase complex: regulating metabolism through post-translational modification of other enzymes. J Neurochem. 2015;134:3–6. doi: 10.1111/jnc.13150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Gibson GE, Xu H, Chen HL, Chen W, Denton TT, Zhang S. Alpha-ketoglutarate dehydrogenase complex-dependent succinylation of proteins in neurons and neuronal cell lines. J Neurochem. 2015;134:86–96. doi: 10.1111/jnc.13096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kuratsune H, Watanabe Y, Yamaguti K, Jacobsson G, Takahashi M, Machii T, Onoe H, Onoe K, Matsumura K, Valind S, Kitani T, Langstrom B. High uptake of [2-11C]acetyl-L-carnitine into the brain: a PET study. Biochem Biophys Res Commun. 1997;231:488–93. doi: 10.1006/bbrc.1996.5919. [DOI] [PubMed] [Google Scholar]
- 114.Aureli T, Puccetti C, Di Cocco ME, Arduini A, Ricciolini R, Scalibastri M, Manetti C, Conti F. Entry of [(1,2-13C2)acetyl]-L-carnitine in liver tricarboxylic acid cycle and lipogenesis: a study by 13C NMR spectroscopy in conscious, freely moving rats. Eur J Biochem. 1999;263:287–93. doi: 10.1046/j.1432-1327.1999.00524.x. [DOI] [PubMed] [Google Scholar]
- 115.Cruz F, Scott SR, Barroso I, Santisteban P, Cerdan S. Ontogeny and cellular localization of the pyruvate recycling system in rat brain. J Neurochem. 1998;70:2613–9. doi: 10.1046/j.1471-4159.1998.70062613.x. [DOI] [PubMed] [Google Scholar]
- 116.Kunnecke B, Cerdan S, Seelig J. Cerebral metabolism of [1,2-13C2]glucose and [U-13C4]3-hydroxybutyrate in rat brain as detected by 13C NMR spectroscopy. NMR Biomed. 1993;6:264–77. doi: 10.1002/nbm.1940060406. [DOI] [PubMed] [Google Scholar]
- 117.Cerdan S, Kunnecke B, Seelig J. Cerebral metabolism of [1,2-13C2]acetate as detected by in vivo and in vitro 13C NMR. J Biol Chem. 1990;265:12916–26. [PubMed] [Google Scholar]
- 118.Richards EM, Rosenthal RE, Kristian T, Fiskum G. Postischemic hyperoxia reduces hippocampal pyruvate dehydrogenase activity. Free Radic Biol Med. 2006;40:1960–70. doi: 10.1016/j.freeradbiomed.2006.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Martin E, Rosenthal RE, Fiskum G. Pyruvate dehydrogenase complex: metabolic link to ischemic brain injury and target of oxidative stress. J Neurosci Res. 2005;79:240–7. doi: 10.1002/jnr.20293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bogaert YE, Sheu KF, Hof PR, Brown AM, Blass JP, Rosenthal RE, Fiskum G. Neuronal subclass-selective loss of pyruvate dehydrogenase immunoreactivity following canine cardiac arrest and resuscitation. Exp Neurol. 2000;161:115–26. doi: 10.1006/exnr.1999.7250. [DOI] [PubMed] [Google Scholar]
- 121.Vereczki V, Martin E, Rosenthal RE, Hof PR, Hoffman GE, Fiskum G. Normoxic resuscitation after cardiac arrest protects against hippocampal oxidative stress, metabolic dysfunction, and neuronal death. J Cereb Blood Flow Metab. 2006;26:821–35. doi: 10.1038/sj.jcbfm.9600234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Scafidi S, O’Brien J, Hopkins I, Robertson C, Fiskum G, McKenna M. Delayed cerebral oxidative glucose metabolism after traumatic brain injury in young rats. J Neurochem. 2009;(109 Suppl 1):189–97. doi: 10.1111/j.1471-4159.2009.05896.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hassel B, Sonnewald U, Fonnum F. Glial-neuronal interactions as studied by cerebral metabolism of [2-13C]acetate and [1-13C]glucose: an ex vivo 13C NMR spectroscopic study. J Neurochem. 1995;64:2773–82. doi: 10.1046/j.1471-4159.1995.64062773.x. [DOI] [PubMed] [Google Scholar]
- 124.Ebert D, Haller RG, Walton ME. Energy contribution of octanoate to intact rat brain metabolism measured by 13C nuclear magnetic resonance spectroscopy. J Neurosci. 2003;23:5928–35. doi: 10.1523/JNEUROSCI.23-13-05928.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Aureli T, Di Cocco ME, Puccetti C, Ricciolini R, Scalibastri M, Miccheli A, Manetti C, Conti F. Acetyl-L-carnitine modulates glucose metabolism and stimulates glycogen synthesis in rat brain. Brain Res. 1998;796:75–81. doi: 10.1016/s0006-8993(98)00319-9. [DOI] [PubMed] [Google Scholar]
- 126.Ori C, Freo U, Pizzolato G, Dam M. Effects of acetyl-L-carnitine on regional cerebral glucose metabolism in awake rats. Brain Res. 2002;951:330–5. doi: 10.1016/s0006-8993(02)03290-0. [DOI] [PubMed] [Google Scholar]
- 127.Waniewski RA, Martin DL. Preferential utilization of acetate by astrocytes is attributable to transport. J Neurosci. 1998;18:5225–33. doi: 10.1523/JNEUROSCI.18-14-05225.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Bigford GE, Del Rossi G. Supplemental substances derived from foods as adjunctive therapeutic agents for treatment of neurodegenerative diseases and disorders. Adv Nutr. 2014;5:394–403. doi: 10.3945/an.113.005264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Calabrese V, Scapagnini G, Ravagna A, Bella R, Butterfield DA, Calvani M, Pennisi G, Giuffrida Stella AM. Disruption of thiol homeostasis and nitrosative stress in the cerebrospinal fluid of patients with active multiple sclerosis: evidence for a protective role of acetylcarnitine. Neurochem Res. 2003;28:1321–8. doi: 10.1023/a:1024984013069. [DOI] [PubMed] [Google Scholar]
- 130.Markowska AL, Ingram DK, Barnes CA, Spangler EL, Lemken VJ, Kametani H, Yee W, Olton DS. Acetyl-1-carnitine. 1: Effects on mortality, pathology and sensory-motor performance in aging rats. Neurobiol Aging. 1990;11:491–8. doi: 10.1016/0197-4580(90)90109-d. [DOI] [PubMed] [Google Scholar]
- 131.Barnes CA, Markowska AL, Ingram DK, Kametani H, Spangler EL, Lemken VJ, Olton DS. Acetyl-1-carnitine. 2: Effects on learning and memory performance of aged rats in simple and complex mazes. Neurobiol Aging. 1990;11:499–506. doi: 10.1016/0197-4580(90)90110-l. [DOI] [PubMed] [Google Scholar]
- 132.Kobayashi S, Iwamoto M, Kon K, Waki H, Ando S, Tanaka Y. Acetyl-L-carnitine improves aged brain function. Geriatr Gerontol Int. 2010;(10 Suppl 1):S99–106. doi: 10.1111/j.1447-0594.2010.00595.x. [DOI] [PubMed] [Google Scholar]
- 133.Al-Majed AA, Sayed-Ahmed MM, Al-Omar FA, Al-Yahya AA, Aleisa AM, Al-Shabanah OA. Carnitine esters prevent oxidative stress damage and energy depletion following transient forebrain ischaemia in the rat hippocampus. Clin Exp Pharmacol Physiol. 2006;33:725–33. doi: 10.1111/j.1440-1681.2006.04425.x. [DOI] [PubMed] [Google Scholar]
- 134.McKenna MC, Scafidi S, Robertson CL. Metabolic alterations in developing brain after injury: Knowns and unknowns. Neurochem Res. 2015;40:2527–2543. doi: 10.1007/s11064-015-1600-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Higgins RD, Raju T, Edwards AD, Azzopardi DV, Bose CL, Clark RH, Ferriero DM, Guillet R, Gunn AJ, Hagberg H, Hirtz D, Inder TE, Jacobs SE, Jenkins D, Juul S, Laptook AR, Lucey JF, Maze M, Palmer C, Papile L, Pfister RH, Robertson NJ, Rutherford M, Shankaran S, Silverstein FS, Soll RF, Thoresen M, Walsh WF. Hypothermia and other treatment options for neonatal encephalopathy: an executive summary of the Eunice Kennedy Shriver NICHD workshop. J Pediatr. 2011;159:851–858. doi: 10.1016/j.jpeds.2011.08.004. e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Fatemi A, Wilson MA, Johnston MV. Hypoxic-ischemic encephalopathy in the term infant. Clin Perinatol. 2009;36:835–58. doi: 10.1016/j.clp.2009.07.011. vii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Graham EM, Ruis KA, Hartman AL, Northington FJ, Fox HE. A systematic review of the role of intrapartum hypoxia-ischemia in the causation of neonatal encephalopathy. Am J Obstet Gynecol. 2008;199:587–95. doi: 10.1016/j.ajog.2008.06.094. [DOI] [PubMed] [Google Scholar]
- 138.Rice JE, 3rd, Vannucci RC, Brierley JB. The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol. 1981;9:131–41. doi: 10.1002/ana.410090206. [DOI] [PubMed] [Google Scholar]
- 139.Smith AL, Rosenkrantz TS, Fitch RH. Effects of Sex and Mild Intrainsult Hypothermia on Neuropathology and Neural Reorganization following Neonatal Hypoxic Ischemic Brain Injury in Rats. Neural Plast. 2016;2016:2585230. doi: 10.1155/2016/2585230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Morken TS, Brekke E, Haberg A, Wideroe M, Brubakk AM, Sonnewald U. Altered astrocyte-neuronal interactions after hypoxia-ischemia in the neonatal brain in female and male rats. Stroke. 2014;45:2777–2785. doi: 10.1161/STROKEAHA.114.005341. [DOI] [PubMed] [Google Scholar]
- 141.Chavez-Valdez R, Martin LJ, Razdan S, Gauda EB, Northington FJ. Sexual dimorphism in BDNF signaling after neonatal hypoxia-ischemia and treatment with necrostatin-1. Neuroscience. 2014;260:106–19. doi: 10.1016/j.neuroscience.2013.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Brekke EM, Morken TS, Wideroe M, Haberg AK, Brubakk AM, Sonnewald U. The pentose phosphate pathway and pyruvate carboxylation after neonatal hypoxic-ischemic brain injury. J Cereb Blood Flow Metab. 2014;34:724–34. doi: 10.1038/jcbfm.2014.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Osredkar D, Sall JW, Bickler PE, Ferriero DM. Erythropoietin promotes hippocampal neurogenesis in in vitro models of neonatal stroke. Neurobiol Dis. 2010;38:259–65. doi: 10.1016/j.nbd.2010.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.de Paula S, Vitola AS, Greggio S, de Paula D, Mello PB, Lubianca JM, Xavier LL, Fiori HH, Dacosta JC. Hemispheric brain injury and behavioral deficits induced by severe neonatal hypoxia-ischemia in rats are not attenuated by intravenous administration of human umbilical cord blood cells. Pediatr Res. 2009;65:631–5. doi: 10.1203/PDR.0b013e31819ed5c8. [DOI] [PubMed] [Google Scholar]
- 145.Yang J, Liu J, Niu G, Liu Y, Wu EX. Magnetic resonance imaging of migrating neuronal precursors in normal and hypoxic-ischemic neonatal rat brains by intraventricular MPIO labeling. Conf Proc IEEE Eng Med Biol Soc. 2008;2008:363–6. doi: 10.1109/IEMBS.2008.4649165. [DOI] [PubMed] [Google Scholar]
- 146.Demarest TG, Schuh RA, Waite EL, Waddell J, McKenna MC, Fiskum G. Sex dependent alterations in mitochondrial electron transport chain proteins following neonatal rat cerebral hypoxic-ischemia. J Bioenerg Biomembr. 2016;48:591–598. doi: 10.1007/s10863-016-9678-4. [DOI] [PubMed] [Google Scholar]
- 147.Demarest TG, Schuh RA, Waddell J, McKenna MC, Fiskum G. Sex-dependent mitochondrial respiratory impairment and oxidative stress in a rat model of neonatal hypoxic-ischemic encephalopathy. J Neurochem. 2016;137:714–29. doi: 10.1111/jnc.13590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Faul M, Xu L, Wald MM, Coronado VG. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations and Deaths 2002–2006. Atlanta: Centers for Disease Control and Prevention, National Center for Injury Prevention and Control; 2010. [Google Scholar]
- 149.Yeates KO, Swift E, Taylor HG, Wade SL, Drotar D, Stancin T, Minich N. Short-and long-term social outcomes following pediatric traumatic brain injury. J Int Neuropsychol Soc. 2004;10:412–26. doi: 10.1017/S1355617704103093. [DOI] [PubMed] [Google Scholar]
- 150.Wechsler B, Kim H, Gallagher PR, DiScala C, Stineman MG. Functional status after childhood traumatic brain injury. J Trauma. 2005;58:940–9. doi: 10.1097/01.ta.0000162630.78386.98. discussion 950. [DOI] [PubMed] [Google Scholar]
- 151.Ryan NP, Anderson V, Godfrey C, Beauchamp MH, Coleman L, Eren S, Rosema S, Taylor K, Catroppa C. Predictors of very-long-term sociocognitive function after pediatric traumatic brain injury: evidence for the vulnerability of the immature “social brain”. J Neurotrauma. 2014;31:649–57. doi: 10.1089/neu.2013.3153. [DOI] [PubMed] [Google Scholar]
- 152.Catroppa C, Godfrey C, Rosenfeld JV, Hearps SS, Anderson VA. Functional recovery ten years after pediatric traumatic brain injury: outcomes and predictors. J Neurotrauma. 2012;29:2539–47. doi: 10.1089/neu.2012.2403. [DOI] [PubMed] [Google Scholar]
- 153.Babikian T, Asarnow R. Neurocognitive outcomes and recovery after pediatric TBI: meta-analytic review of the literature. Neuropsychology. 2009;23:283–96. doi: 10.1037/a0015268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Zhang X, Liu S, Newport GD, Paule MG, Callicott R, Thompson J, Liu F, Patterson TA, Berridge MS, Apana SM, Brown CC, Maisha MP, Hanig JP, Slikker W, Jr, Wang C. In Vivo Monitoring of Sevoflurane-induced Adverse Effects in Neonatal Nonhuman Primates Using Small-animal Positron Emission Tomography. Anesthesiology. 2016;125:133–46. doi: 10.1097/ALN.0000000000001154. [DOI] [PubMed] [Google Scholar]
- 155.Zhang X, Liu S, MG P, Newport GD, Callicott R, Berridge MS, Apana SMSWCW., Jr Protective Effects of Acetyl L-Carnitine on Inhalation Anesthetic-Induced Neuronal Damage in the Nonhuman Primate. J Mol Pharm Org Process Res. 2013;1 doi: 10.4172/2329-9029.1000102. [DOI] [Google Scholar]
- 156.Zou X, Sadovova N, Patterson TA, Divine RL, Hotchkiss CE, Ali SF, Hanig JP, Paule MG, Slikker W, Jr, Wang C. The effects of L-carnitine on the combination of, inhalation anesthetic-induced developmental, neuronal apoptosis in the rat frontal cortex. Neuroscience. 2008;151:1053–65. doi: 10.1016/j.neuroscience.2007.12.013. [DOI] [PubMed] [Google Scholar]
- 157.Walters JL, Paule MG. Review of preclinical studies on pediatric general anesthesia-induced developmental neurotoxicity. Neurotoxicol Teratol. 2017;60:2–23. doi: 10.1016/j.ntt.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 158.De Simone R, Ramacci MT, Aloe L. Effect of acetyl-L-carnitine on forebrain cholinergic neurons of developing rats. Int J Dev Neurosci. 1991;9:39–46. doi: 10.1016/0736-5748(91)90071-s. [DOI] [PubMed] [Google Scholar]
- 159.Piovesan P, Quatrini G, Pacifici L, Taglialatela G, Angelucci L. Acetyl-L-carnitine restores choline acetyltransferase activity in the hippocampus of rats with partial unilateral fimbria-fornix transection. Int J Dev Neurosci. 1995;13:13–9. doi: 10.1016/0736-5748(94)00070-j. [DOI] [PubMed] [Google Scholar]
- 160.Picconi B, Barone I, Pisani A, Nicolai R, Benatti P, Bernardi G, Calvani M, Calabresi P. Acetyl-L-carnitine protects striatal neurons against in vitro ischemia: the role of endogenous acetylcholine. Neuropharmacology. 2006;50:917–23. doi: 10.1016/j.neuropharm.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 161.Imperato A, Ramacci MT, Angelucci L. Acetyl-L-carnitine enhances acetylcholine release in the striatum and hippocampus of awake freely moving rats. Neurosci Lett. 1989;107:251–5. doi: 10.1016/0304-3940(89)90826-4. [DOI] [PubMed] [Google Scholar]
- 162.Mitsushima D, Sano A, Takahashi T. A cholinergic trigger drives learning-induced plasticity at hippocampal synapses. Nat Commun. 2013;4:2760. doi: 10.1038/ncomms3760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Gold PE. Acetylcholine modulation of neural systems involved in learning and memory. Neurobiol Learn Mem. 2003;80:194–210. doi: 10.1016/j.nlm.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 164.Sarter M, Bruno JP, Givens B. Attentional functions of cortical cholinergic inputs: what does it mean for learning and memory? Neurobiol Learn Mem. 2003;80:245–56. doi: 10.1016/s1074-7427(03)00070-4. [DOI] [PubMed] [Google Scholar]
- 165.Phillis JW. Acetylcholine release from the central nervous system: a 50-year retrospective. Crit Rev Neurobiol. 2005;17:161–217. doi: 10.1615/critrevneurobiol.v17.i3-4.30. [DOI] [PubMed] [Google Scholar]
- 166.Furukawa S, Yang L, Sameshima H. Galantamine, an acetylcholinesterase inhibitor, reduces brain damage induced by hypoxia-ischemia in newborn rats. Int J Dev Neurosci. 2014;37:52–7. doi: 10.1016/j.ijdevneu.2014.06.011. [DOI] [PubMed] [Google Scholar]
- 167.Furukawa S, Yang L, Sameshima H, Ikenoue T. Repetitive administration of acetylcholine receptor agonist rescues brain inflammation and brain damage after hypoxia-ischemia in newborn rat. J Perinat Med. 2014;42:379–84. doi: 10.1515/jpm-2013-0175. [DOI] [PubMed] [Google Scholar]
- 168.Furukawa S, Sameshima H, Yang L, Harishkumar M, Ikenoue T. Regional differences of microglial accumulation within 72 hours of hypoxia-ischemia and the effect of acetylcholine receptor agonist on brain damage and microglial activation in newborn rats. Brain Res. 2014;1562:52–8. doi: 10.1016/j.brainres.2014.03.028. [DOI] [PubMed] [Google Scholar]
- 169.Juul SE, Ferriero DM. Pharmacologic neuroprotective strategies in neonatal brain injury. Clin Perinatol. 2014;41:119–31. doi: 10.1016/j.clp.2013.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Kochanek PM, Bell MJ, Bayir H. Quo vadis 2010? - carpe diem: challenges and opportunities in pediatric traumatic brain injury. Dev Neurosci. 2010;32:335–42. doi: 10.1159/000323016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Kochanek PM, Carney N, Adelson PD, Ashwal S, Bell MJ, Bratton S, Carson S, Chesnut RM, Ghajar J, Goldstein B, Grant GA, Kissoon N, Peterson K, Selden NR, Tasker RC, Tong KA, Vavilala MS, Wainwright MS, Warden CR. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents--second edition. Pediatr Crit Care Med. 2012;(13 Suppl 1):S1–82. doi: 10.1097/PCC.0b013e31823f435c. [DOI] [PubMed] [Google Scholar]
- 172.Kochanek PM, Jackson TC, Ferguson NM, Carlson SW, Simon DW, Brockman EC, Ji J, Bayir H, Poloyac SM, Wagner AK, Kline AE, Empey PE, Clark RS, Jackson EK, Dixon CE. Emerging therapies in traumatic brain injury. Semin Neurol. 2015;35:83–100. doi: 10.1055/s-0035-1544237. [DOI] [PMC free article] [PubMed] [Google Scholar]