Abstract
Spatial variation in mercury (Hg) and methylmercury (MeHg) bioaccumulation in urban coastal watersheds reflects complex interactions between Hg sources, land use, and environmental gradients. We examined MeHg concentrations in fauna from the Delaware River estuary, and related these measurements to environmental parameters and human impacts on the waterway. The sampling sites followed a north to south gradient of increasing salinity, decreasing urban influence, and increasing marsh cover. Although mean total Hg in surface sediments (top 4cm) peaked in the urban estuarine turbidity maximum and generally decreased downstream, surface sediment MeHg concentrations showed no spatial patterns consistent with the examined environmental gradients, indicating urban influence on Hg loading to the sediment but not subsequent methylation. Surface water particulate MeHg concentration showed a positive correlation with marsh cover whereas dissolved MeHg concentrations were slightly elevated in the estuarine turbidity maximum region. Spatial patterns of MeHg bioaccumulation in resident fauna varied across taxa. Small fish showed increased MeHg concentrations in the more urban/industrial sites upstream, with concentrations generally decreasing farther downstream. Invertebrates either showed no clear spatial patterns in MeHg concentrations (blue crabs, fiddler crabs) or increasing concentrations further downstream (grass shrimp). Best-supported linear mixed models relating tissue concentration to environmental variables reflected these complex patterns, with species specific model results dominated by random site effects with a combination of particulate MeHg and landscape variables influencing bioaccumulation in some species. The data strengthen accumulating evidence that bioaccumulation in estuaries can be decoupled from sediment MeHg concentration, and that drivers of MeHg production and fate may vary within a small region.
Keywords: methylmercury, bioaccumulation, estuary, landscape, sediment, fish
Introduction
Throughout the last century, urban rivers and waterways in the northeastern US have faced numerous challenges to ecosystem health; yet they have also experienced substantial improvements in water quality, with documented reductions in sulfate, phosphorus, organic loading, and metal pollutants (Burns et al. 2006; Eshleman et al. 2013; Sanudo-Wilhelmy and Gill 1999; Skjelkvale et al. 2005) across water bodies. Coastal rivers and estuaries continue to be affected by current stressors and both organic and inorganic legacy contamination. Accurate assessments of the current condition of urban aquatic ecosystems are necessary for their continued use and improvement.
Given the history of urban waters, wildlife and human exposures to contaminants are particular concerns. Mercury (Hg) is a contaminant of global concern (UNEP 2013), and in the US, ranks third on the Agency for Toxic Substances and Disease Registry's 2013 priority list of hazardous substances (ATSDR 2013). Methylmercury (MeHg) is a potent neurotoxin that biomagnifies through food webs, affecting humans and wildlife alike. The main route of exposure to humans is through seafood (Chen et al. 2008; Sunderland 2007). Fish consumption advisories exist for MeHg in fresh and coastal waters throughout the US (USEPA 2013), including urban rivers, which, due to the complexity of multiple historic and current contaminant inputs may also have advisories for other pollutants.
Drivers of MeHg bioavailability and fate are complex. Our previous studies focused on regional variation of MeHg bioaccumulation in estuaries from Maine to New Jersey, finding strong relationships between organic carbon and Hg concentrations in sediments across systems ranging in latitude, anthropogenic influence, and tidal flushing characteristics (Chen et al. 2009; Schartup et al. 2013, Balcom et al. 2015). Numerous other studies have found similar relationships in sediments (Conaway et al. 2003; Lambertsson and Nilsson 2006; Mason and Lawrence 1999). Strong relationships have been observed between forage fish tissue MeHg concentrations and dissolved and particulate MeHg concentrations in the water column, but not with sediment MeHg concentrations (Chen et al. 2014). This may result from the lack of a strong correlation between MeHg in sediment and water column MeHg across ecosystems (Balcom et al. 2015).
Previous studies suggest that under many conditions, Hg loading to the sediment does not determine sediment MeHg concentrations or MeHg levels in biota (e.g. Chalmers et al. 2014). Food web characteristics also relate to MeHg in fish tissue; higher concentrations of MeHg have been observed in pelagic feeding fauna and higher trophic level taxa have a higher percent of total Hg as MeHg (%MeHg) (Chen et al. 2014; Chen et al. 2009). In addition, urban development influences Hg distribution (Chalmers et al. 2014; Sunderland et al. 2012). Factors affecting production and trophic transfer of MeHg, such as sediment carbon concentration, particulate composition, and food chain length, can be related to water quality and ecosystem challenges that face urban waterways. For example, eutrophication has the potential to change Hg and MeHg bioavailability through biomass dilution and/or by changing carbon loading (Driscoll et al. 2012). Environmental gradients such as salinity may also affect bioaccumulation (Dutton and Fisher 2011; Fry and Chumchal 2012; Mason et al. 1996).
Our study ecosystem, the Delaware River estuary, has a long history of anthropogenic impacts including loadings of nutrients and organic and metal contaminants (Ashley et al. 2009; Church et al. 2006). Areas of intense industrialization have occurred around Philadelphia PA and Wilmington DE (Ashley et al. 2009). PCB and mercury concentrations, particularly in the industrialized corridor, have lead in part to the establishment of fish consumption advisories throughout the estuary (DE Department of Natural Resources and Environmental Control 2016; NJ Department of Environmental Protection 2016). Our recent study in the estuarine turbidity maximum (ETM) of the river examined the relative importance of watershed and sediment sources of MeHg to the upper estuary (Gosnell et al. 2015).
The current study was undertaken to examine MeHg concentrations in fauna from sites within an urban estuary, and relate these measurements to environmental parameters such as organic carbon, salinity and urban development as well as water and sediment (abiotic) Hg and MeHg concentrations. We aimed to determine if within-system variation in MeHg bioaccumulation was related to environmental factors similar to our findings across systems; in particular, whether landscape influenced MeHg bioavailability and fate. This approach linking biotic and abiotic Hg and MeHg measurements along a salinity and human-use gradient enhances the current body of knowledge regarding MeHg fate in the Delaware River estuary.
Methods
In July 2012, we sampled 10 sites on the Delaware River/Bay from Philadelphia, PA to southern NJ, chosen to span a range of salinity and contaminant exposures (Figure 1, Table S1). Sediment cores and grabs, and both aqueous and biotic samples were taken at each site to investigate the relationship between abiotic exposures and bioaccumulation in lower trophic level organisms.
Fig. 1.
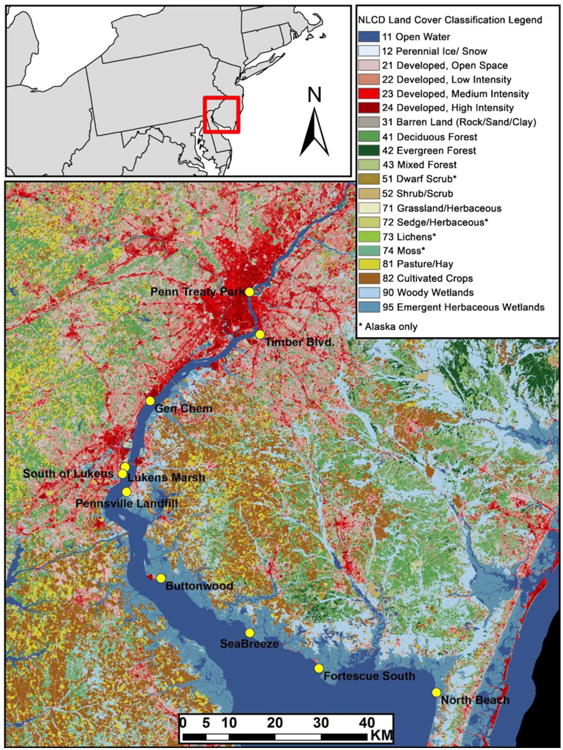
Sampling locations (yellow circles) marked on National Land Cover Database land cover classification map.
Field samples
Two replicate sediment cores were collected from each site and sectioned in 2 cm intervals up to 10 cm for MeHg and total Hg (THg) analysis (Balcom et al. 2015). In addition, grab samples of the top 2 cm of sediment were taken from 18-20 locations at each site for analysis to determine the range of organic carbon (measured as percent loss on ignition (%LOI)) and THg. A single surface water grab sample was taken at high tide at each site using trace-metal clean technique and filtered (0.45μm quartz fiber filter) in a laminar flow hood within 12h for dissolved and particulate MeHg and THg, as well as dissolved organic carbon (DOC) and chlorophyll (chla) (Balcom et al. 2015; Gosnell et al. 2015). Surface water temperature, salinity, dissolved oxygen, and pH was measured on site using a hand-held sensor (Hydrolab MiniSonde MS5, Hach).
Fauna were sampled using a seine, minnow traps, or hand collected. Killifish (Fundulus heteroclitus (mummichog) or F. diaphanous (banded killifish)) were sampled at every site, and juvenile white perch (Morone americana), juvenile striped bass (Morone saxatalis), fiddler crabs (Uca pugnax), grass shrimp (Palaemonetes sp.), and blue crabs (Callinectes sapidus) were present at five or more locations. Similarly sized individuals were chosen based on total length or carapace width to minimize the influence of size on MeHg concentration (Table S4), freeze-dried (Labconco Freezone) for a minimum of 48hr and homogenized prior to analysis (Chen et al. 2014). For white perch and striped bass greater than 10cm in total length, a small portion of filet was analyzed, rather than the whole individual as for all other samples.
THg and MeHg analyses
Sediment and water samples were analyzed at the University of Connecticut Department of Marine Sciences. Sediment %LOI was determined by combustion at 550°C for 8-12 hours. Sediment THg was analyzed using a Milestone DMA80 pyrolytic analyzer (U.S. Environmental Protection Agency 1998). THg in dissolved and particulate fractions (digestion matrix included 2% bromine monochloride) were measured using purge and trap (dual gold-amalgamation) cold vapor atomic fluorescence spectrometry (Tekran 2500 CVAFS detector) (Bloom and Fitzgerald 1988; Bloom and Crecelius 1983; Fitzgerald and Gill 1979). Particulate THg filters were acid leached (4N HNO3) prior to the above analyses (Hammerschmidt and Fitzgerald 2001). MeHg in filtered water, the particulate fraction, and sediment was analyzed using purge and trap (tenax) CVAFS (Tekran 2700 methylmercury analyzer) following distillation and ethylation (Hammerschmidt and Fitzgerald 2001; Tseng et al. 2004). Sample results were corrected for field and preparation blanks as appropriate.
Biotic samples were analyzed for MeHg and inorganic Hg (iHg) tissue concentrations at the Dartmouth Trace Element Analysis Core by species-specific isotope dilution using an automated MeHg system MERX-M (Brooks Rand Labs, Seattle, WA) interfaced with an Element 2 ICP-MS (Thermo, Bremen, Germany) using an adaptation of EPA method 1630 (Taylor et al. 2011; Taylor et al. 2008). Total mercury was calculated as the sum of MeHg and iHg concentrations. Teleost sample iHg concentrations were typically below sample specific detection limits, as they were generally greater than 90% MeHg. These calculations were corroborated by DMA analysis when sample mass permitted. QA/QC for abiotic and biotic analyses are detailed in the supporting information.
Statistical analysis
All sediment, biota, and particulate Hg concentration data are presented on a dry weight basis. Statistics were carried out using JMP10 (SAS). ANOVA was used to assess significant differences among sites in tissue MeHg and %MeHg for species present at a minimum of five sites. Assumptions for ANOVA were assessed with Levene's test and a normal quantile plot. Fiddler crab, banded killifish, and grass shrimp MeHg concentrations were log10 transformed. Post-hoc Tukey's Honestly Significant Difference (HSD) test was used to assign differences between sites. Percent MeHg for mummichogs, banded killifish, juvenile white perch, blue crab, and sediment grab THg did not meet the assumption of homoscedasticity and were analyzed using Welch's ANOVA. A Games-Howell post-hoc test was used to assess among site differences for sediment THg. Relationships within sediment cores were analyzed using scatterplot matrices and Spearman's rank correlation.
Akaike's Information Criterion adjusted for small sample sizes (AICc) was used to assess the best a priori model for describing patterns in bioaccumulation among the different species. National Land Cover Database land cover classifications (Homer et al. 2015) were used to generate estimates of the level of development, forest, and marsh within a 1km radius buffer surrounding each sampling site (Table S1). A scatterplot matrix and Spearman correlations were used to determine variables that were highly correlated and should not be included in the same model (ρ > 0.7).
For each species, a 2-part model selection procedure was employed where: 1) the best of six MeHg source models (combinations of dissolved MeHg, particulate MeHg, sediment MeHg and sediment %LOI) was chosen followed by 2) combinations of environmental parameters which were combined with the best model from step one, resulting in a final comparison of 14 models. Environmental parameters hypothesized to influence MeHg production and fate included salinity, % marsh, % forest, % medium to high development, and total suspended solids (TSS). All model inputs were chosen a priori based on variables of interest that are predicted to influence MeHg fate. Only abiotic MeHg concentrations were included as these are the “pools” with direct potential to bioaccumulate in the food web. Sample site was included as a random intercept term in all models to account for potential correlation among samples from the same site while allowing all samples to be used without pseudoreplication (Hurlbert 1984; Pinherio and Bates 2000). When there was insufficient support for a single model (ΔAICc < 2 better top models), the simplest model was selected. Models were fit with the R statistical software (R Core Team 2015) package lme4 (Bates et al. 2014) using Maximum Likelihood (ML) estimation for model comparison and the best model was refit using Restricted Maximum Likelihood (REML) estimation for coefficient interpretation. Unlike ML, REML provides unbiased estimates of fixed effect coefficients in mixed models but it uses a correction for the number of model parameters making it inappropriate for AIC model comparison (Zuur et al. 2009). Comparisons of model AICc were made using the package AICcmodavg (Mazerolle 2014). The proportion of the variance explained by the fixed effects and random effects were calculated using the package MuMIn (Barton 2015) based on the method of Nakagawa and Schielzeth (2013).
Results
Landscape gradients
Land cover estimations indicated a north to south pattern of decreasing development (57% at Penn Treaty and <1% at North Beach) and increasing marsh cover (0% at Penn Treaty and 50% at North Beach) with relatively little occurrence of forest at all sites (Table S1). This north to south trend corresponds to an increase in salinity along the transect of sample sites (Table 1).
Table 1.
Dissolved (ng/L) and particulate (ng/g DW) Hg concentrations and ancillary surface water measurements from a single sample per site.
| dissolved (ng/L) | particulate (ng/g DW) | ancillary | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||
| MeHg (% of total) | THg | MeHg (% of total) | THg | TSS (mg/L) | salinity | DOC (mg/L) | Chla (ug/L) | nitrate (ug/L) | pH | DO (mg/L) | |
| PT | 0.010 (5.0) | 0.2 | 0.315* (0.20) | 155 | 7.0 | 0.12 | 5.57 | 7.1 | 4.7 | ||
| TB | 0.006* (2.1) | 0.29 | 0.573* (0.25) | 232 | 6.3 | 0.06 | 2.76 | 7.23 | 717 | 6.9 | 6.8 |
| GC | 0.114 (22.4) | 0.51 | 0.313 (0.54) | 58.4 | 32.7 | 0.6 | 2.55 | 10.6 | 807 | 7.6 | 7.0 |
| LM | 0.092 (17.7) | 0.52 | 2.75 (0.75) | 366 | 14.0 | 2.5 | 2.89 | 15.8 | 647 | 7.6 | 7.1 |
| SL | 0.044 (9.4) | 0.47 | 1.03 (0.78) | 132 | 28.4 | 2.5 | 2.92 | 17.1 | 815 | 7.7 | 7.2 |
| PL | 0.006* (0.9) | 0.69 | 0.697 (0.55) | 126 | 30.5 | 1.5 | 2.90 | 27.8 | 494 | 7.7 | 7.8 |
| BW | 0.056 (10.0) | 0.56 | 0.954 (0.85) | 113 | 26.9 | 5.3 | 3.07 | 18.6 | 365 | 7.7 | 7.1 |
| SB | 0.024 (4.1) | 0.59 | 0.551 (1.52) | 36.2 | 114 | 16.8 | 4.34 | 14.8 | 670 | 6.9 | 6.3 |
| FS | 0.076 (17.0) | 0.45 | 1.62 (2.24) | 72.4 | 72.2 | 19.3 | 3.60 | 71.0 | 612 | 7.6 | 7.2 |
| NB | 0.013 (1.4) | 0.92 | 1.89 (1.99) | 95.0 | 106 | 20.8 | 6.08 | 74.4 | 1113 | 7.8 | 7.9 |
indicates below detection limit
PT=Penn Treaty, TB= Timber Blvd., GC= Gen Chem, LM=Luken's Marsh, SL=South of Lukens, PL=Pennsville Landfill, BW=Buttonwood, SB=Sea Breeze, FS=Fortescue, and NB=North Beach. MeHg concentrations are presented graphically in the supporting information Figure S1.
Sediment
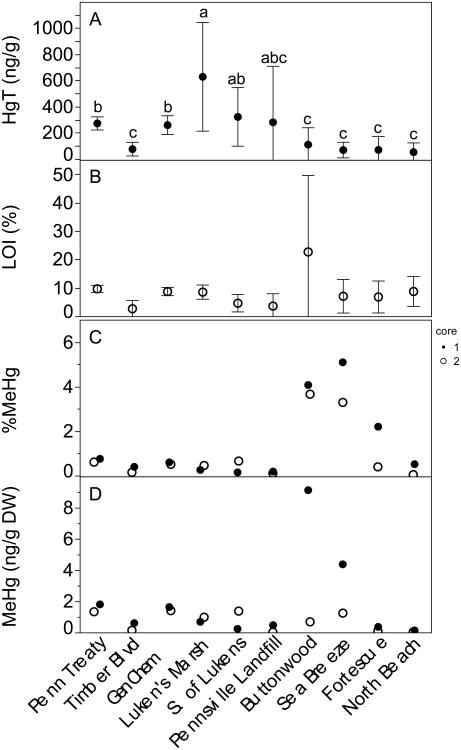
Total Hg in sediment grabs ranged from an average (± st. dev.) of 628 ± 415 ng/g dry weight (DW) at industrially impacted Luken's Marsh and S. Lukens (322 ± 224 ng/g DW) to 53 ± 70 ng/g DW at rural North Beach, with higher spatial variability in sediment THg concentration within the most contaminated sites (Figure 2, Table S2). Organic carbon (%LOI) showed variability both between and within sites for the grab samples with a low of 2.6 ± 3.0% at Timber Blvd and a high of 23 ± 26% at Buttonwood where there were large mats of peat present (Figure 2). Across all sites combined, there was a weak but significant positive correlation between %LOI and THg in surface sediment grab samples (Spearman's ρ=0.52 p<0.0001), however, on a site by site basis the relationship was stronger, with all sites except Penn Treaty, South of Luken's and North Beach exhibiting a significant positive correlation between %LOI and THg (Table S3). Total Hg in the grab samples was highest at the industrial sites near Wilmington (Luken's Marsh) and decreased further south (F9,74.71=33.14, p<0.0001), a pattern that was qualitatively similar to samples taken in the top 4cm average of the two replicate sediment cores taken at each site.
Fig. 2.
Sediment concentrations across sites from north to south. A) THg (ng/g DW) mean +/- st. dev. in 18-20 grab samples at each site. Different letters indicate significant differences as assessed by a Games-Howell post-hoc test. B) carbon content (% loss on ignition) mean +/- st. dev. of across 18-20 grab samples per site. C) %MeHg duplicate cores (top 4cm avg.). D) MeHg (ng/g DW) in top 4 cm of duplicate cores.
In contrast, average MeHg in the duplicate sediment cores (top 4cm) was lowest at North Beach and highest at Buttonwood (both undeveloped), with all sites except marsh-dominated Buttonwood and Sea Breeze below 1.6 ng/g DW (Figure 2). Both Buttonwood and Sea Breeze exhibited large variation in MeHg concentration between duplicate cores. Core top sediment MeHg did not correlate with sediment THg (ρ = 0.43) across sites. At most sites, the vertical distribution of THg, MeHg and %LOI in sediments did not vary significantly, and the concentrations in duplicate cores were consistent. Buttonwood and Fortescue displayed much higher %MeHg in upper sediment than the deeper sections, while at all other sites %MeHg was consistent downcore. Average sediment %MeHg in the top 4cm (a proxy for methylation efficiency (Drott et al. 2008) ranged from 0.18% at Pennsville Landfill to 4.6% at Sea Breeze (Figure 2) and was lowest in the upper estuary and at North Beach.
Water
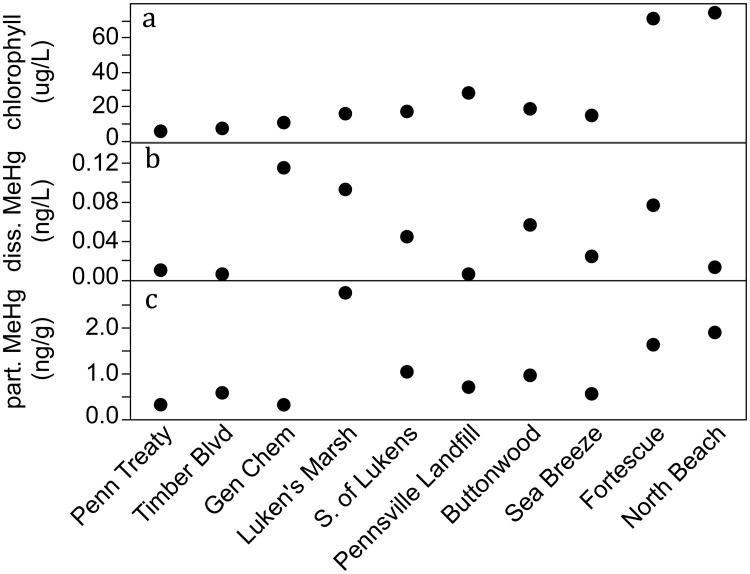
THg dissolved in surface water increased from north to south along the increasing salinity gradient, with a low of 0.2ng/L at Penn Treaty (0 psu) and a high of 0.92 ng/L at North Beach (21 psu). Dissolved MeHg ranged from below detection limits (Timber Blvd, Pennsville Landfill) to 0.1 ng/L (Gen Chem & Luken's Marsh), and was variable for sites in the lower estuary (Figure 3). Percent MeHg in the dissolved fraction followed a spatial pattern similar to dissolved MeHg concentration and was highest in the ETM near Gen Chem and Luken's Marsh, and variable in the lower estuary. DOC and chla concentration also increased from north to south (Table 1). Particulate THg showed a general trend of higher concentration in the north (highest 366 ng/g at Luken's Marsh) and lower concentration in the southern sites (lowest 36.2 ng/g at Sea Breeze), though Gen Chem was an exception to this pattern (Table 1). In contrast, particulate MeHg concentration increased from north to south (0.3 ng/g at Penn Treaty and 1.9 ng/g at North Beach) with the exception of a spike in concentration near Luken's Marsh (2.8 ng/g) (Figure 3). TSS and particulate %MeHg also generally increased along the salinity gradient (Table 1).
Fig. 3.
Surface water concentration across sites from a single grab sample per site. a) chlorophyll a (μg/L), b) dissolved MeHg (ng/L), c) particulate MeHg (ng/g DW)
Biota
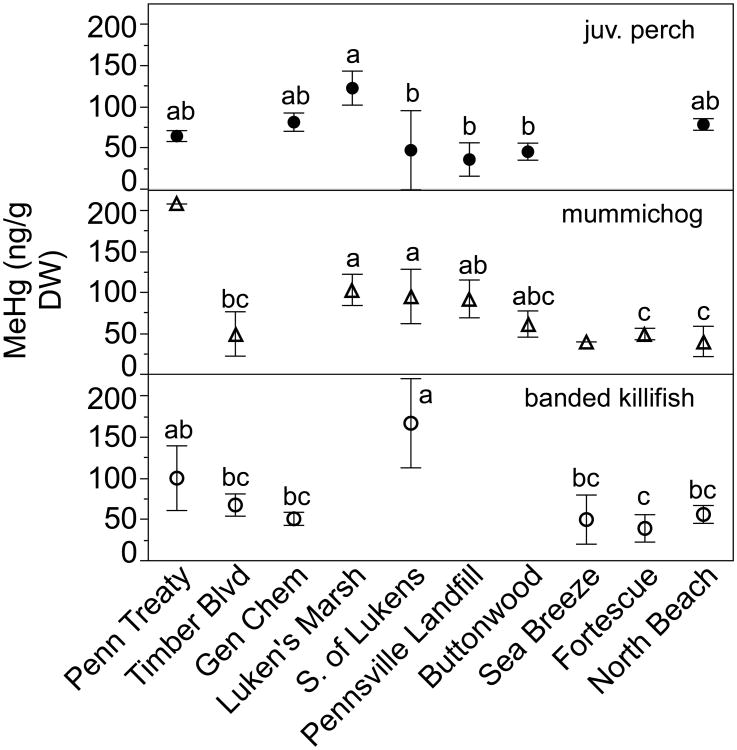
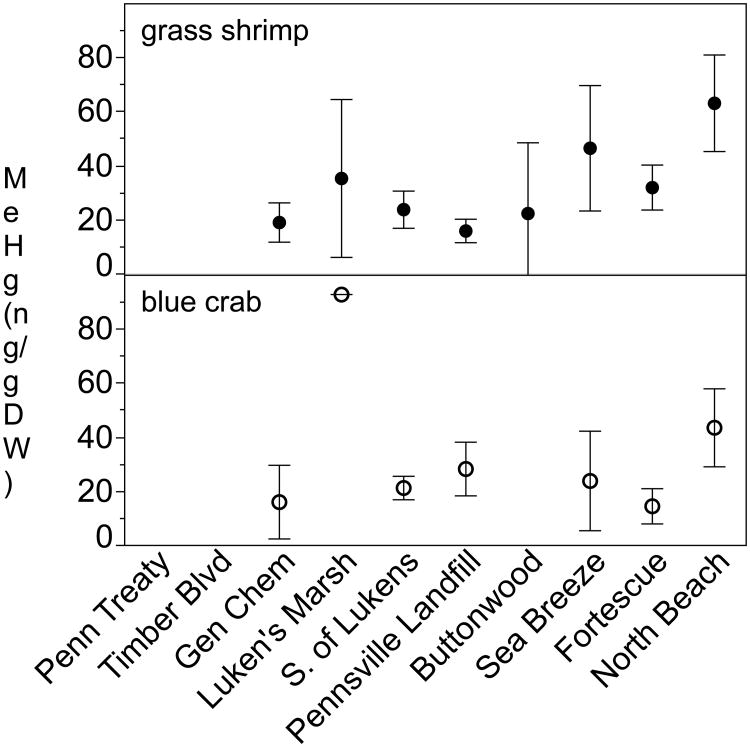
MeHg and THg tissue concentrations varied between species, with no consistent cross-species spatial patterns of bioaccumulation (Figures 4 & 5, Table S4). ANOVA between site comparisons indicated that mummichogs (R2=0.60, F6,32=7.87, p<0.0001), banded killifish (R2=0.69, F6,27=9.34, p<0.0001; one NB individual removed as an outlier) and whole juvenile white perch (R2=0.69, F6,13=4.83, p=0.0084) all had significantly higher MeHg tissue concentrations in the ETM region near Luken's Marsh/S. of Lukens than at other sites (Figure 4). Striped bass filet subsamples also showed significantly higher MeHg concentrations near Luken's Marsh (R2=0.75, F3,9=8.95, p=0.0046) than at other sites. Grass shrimp, fiddler crabs, and blue crabs showed no significant differences in MeHg concentration across sites, but shrimp were qualitatively higher at the southern sites (Figure 5). Percent MeHg in grass shrimp increased along the transect from a mean of 40% at Gen Chem (37% at Pennsville Landfill) to 83% at North Beach (F=21.53, p=0.0006; Figure S5). Juvenile white perch also exhibited a difference in %MeHg between sites, driven by two individuals at S. Lukens with less than 50% MeHg while all others were greater than 88% MeHg.
Fig. 4.
Mean +/- st. dev. of MeHg tissue concentration (ng/g DW) in small fish. Different letters indicate significant differences as indicated by Tukey's HSD. Mummichog n=3-7 per site except for PT and SB where n=1. Juvenile perch n=3 per site except BW where n=2. Banded killifish n=3-7 per site except LM where n=1.
Fig. 5.
Mean +/- st. dev. of MeHg concentrations (ng/g DW) in tissue from invertebrates. Blue crab n=3 per site except LM where n=1. Grass shrimp n=3 per site.
AICc regression analysis and model comparison
The scatterplot matrix revealed strong correlations with surface salinity to sediment THg (r=-0.89), % LOI, (r=0.71) % marsh (r=0.72), and TSS (r=0.92) indicating that salinity should not be included in the same model with these variables. Similarly, development was correlated with sediment THg (r=0.69), salinity (r=-0.65), pH (r=-0.75), and TSS (r=-0.66), while forest cover and marsh cover were moderately correlated (r=-0.63), as were particulate MeHg and marsh cover (r=0.63). As a result of this analysis and the a priori variables of interest, only marsh cover, forest cover, TSS, and development were included as landscape variables in part two of the AICc analysis.
The best model for describing mummichog MeHg bioaccumulation was particulate MeHg + %forest + %development, with a generally positive effect of development (Table 2). The standard deviation in the random-effects intercept across sites was 22.9 (unexplained random variation among sites with a mean of zero). Overall, 41% of the variance was explained by the covariates (R2cov=0.41) and 20% was explained by random site effects (R2random=0.20). Blue crabs were best described by particulate MeHg alone (Table 2), with a positive correlation between particulate MeHg and blue crab tissue MeHg (sd=14.4, R2cov=0.43, R2random=0.34). Juvenile whole white perch were also best described by particulate MeHg alone (Table 2), with a positive correlation between particulate MeHg and perch whole body tissue MeHg (sd=19.4, R2cov=0.30, R2random=0.29).
Table 2.
Mixed effects model regression results with mean coefficient estimates (standard error).
| mummichog | banded killifish | shrimp | blue crab | fiddler | striped bass | juvenile perch | |
|---|---|---|---|---|---|---|---|
| (Intercept) | 79.2*** (9.7) | 91.2*** (21.3) | 32.0*** (5.6) | 29.4*** (6.3) | 17.3*** (4.2) | 356.7*** (71.0) | 67.7*** (9.0) |
| part_MeHg | 12.2 (10.3) | 16.4** (5.7) | 19.7* (9.3) | ||||
| pctforest | 16.5 (10.2) | ||||||
| pct_med_high_developed | 30.2*** (8.1) | ||||||
|
| |||||||
| Num. obs. | 42 | 35 | 24 | 19 | 15 | 14 | 20 |
| Num. groups: site | 9 | 7 | 8 | 7 | 5 | 5 | 7 |
| Variance: site.(Intercept) | 524.1 | 1921.6 | 146.6 | 208.3 | 57.9 | 20706.3 | 377.7 |
| Variance: Residual | 1028.5 | 6014.4 | 323.2 | 153.2 | 87.2 | 10321.9 | 524.3 |
p < 0.001,
p < 0.01,
p < 0.05
The null model (random variability) best described banded killifish (sd = 43.8, R2random=0.24), grass shrimp (sd=12.1, R2random=0.31), fiddler crabs (sd=7.6, R2random=0.40), juvenile striped bass filets, and species with little data. Striped bass AICc results indicated support for additional models including salinity + dissolved MeHg, and dissolved MeHg + %forest + %development (Table 2), but these models were not statistically distinguishable from the null model.
Discussion
The current study of MeHg bioaccumulation along a gradient of urban development and salinity in the Delaware River estuary indicated that of the MeHg source and environmental variables examined, there was no one single variable or consistent combination of abiotic MeHg concentrations and/or landscape variables that best predicted MeHg bioaccumulation across the sampled taxa within this estuarine ecosystem. Instead, there was large within system site-to-site variation, indicating small scale, local processes may be driving MeHg production and fate. In addition, there was evidence that landscape factors and marsh inputs exert increasing influence on MeHg in biota at downstream sites, while sediment sources and diffusive flux of dissolved MeHg from sediment pore water to the overlying water column may be influencing tissue MeHg concentrations of small fish in the ETM region. Our observations lead us to hypothesize that drivers of bioaccumulation are locally influenced, and can change in relative importance in different regions of the estuary. This within system variability has also been observed for PCB bioaccumulation in white perch, channel catfish, small prey fish, and amphipods in the Delaware River estuary, and similarly attributed to within-zone factors such as heterogeneity of contamination or individual dietary preference (Ashley et al. 2009). In the current study, local drivers including presence/absence of marsh and level of urbanization, watershed versus local sources of Hg and MeHg, as well as whether the site was inside of or outside of the ETM all influenced MeHg fate.
Urban areas have long been associated with impairment of surrounding waterways, being sources of contaminants as well as contributing to degradation of habitat (Rodriguez et al. 2007). Pinedo et al (2014) found that coastal metal pollution in surface sediments was closely linked with the presence of urban areas and river mouths in the Mediterranean Sea. Atmospheric deposition of Hg has been strongly correlated with proximity to urban areas in freshwater lakes and streams in the US (Chalmers et al. 2014; Van Metre 2012), yet higher Hg loading to a system doesn't necessarily result in increased MeHg bioaccumulation in resident fauna. The Delaware River estuary formerly had numerous known sources of Hg contamination to the river including a chlor-alkali facility, an industrial wastewater treatment plant, a coal-fired power plant, and numerous industrial facilities (Church et al. 2006), and the influence of legacy contamination as well as current sources are reflected in the sediments. Sediment THg (often used as an indicator of Hg loading) was highest near the industrial Wilmington sites and the urban area of Philadelphia and decreased as development decreased downstream, although within site variability was high at the most contaminated sites, indicating an uneven distribution of Hg contamination in the sediments. Similar patterns of loading were observed in past studies conducted on THg in sediments and striped bass on the Delaware River (Greene 2007; Reinfelder and Totten 2006; Gosnell et al. 2015), which indicate that Hg in sediment and in surface water follow the same general pattern; Hg is more elevated nearer to the shoreline, and in the upper portion of zone 5 as defined by the Delaware River Basin Commission (Greene 2011). This zone begins at the DE/NJ/PA border and corresponds with the highly industrialized region near Wilmington, DE, and is also a transitional zone between the more freshwater upper portion of the estuary and the more saline lower bay, encompassing the ETM where we also observed comparatively higher MeHg concentrations in small fish.
Despite the observed influence of development on THg loading to the sediment, the effect on resulting MeHg bioaccumulation is complex and less well defined. At first glance, small fish bioaccumulation appeared highest where sediment THg concentration was also higher, in particular near Luken's Marsh, a relationship that is not commonly observed. However, higher particulate or dissolved MeHg concentration at some of these sites confound this observation, as water column MeHg “pools” are more directly available to enter the pelagic food web than sediment THg (Figure 3). In addition, small whole perch and mummichogs have tissue concentrations at North Beach and Buttonwood respectively that are similar to those at Luken's Marsh, but sediment THg at these sites are significantly lower, illustrating the diversity of influences on MeHg bioaccumulation. While sediment THg was sampled more intensively, MeHg concentrations in sediment and surface water across all sites were not correlated with sediment THg, indicating again that sediment loading was not the only control on production and bioavailability of MeHg within this ecosystem.
Sediment THg concentrations at more developed sites (Penn Treaty, Gen Chem, Luken's Marsh, South Luken's and Pennsville Landfill) were within the range of concentrations found in other urban estuaries including Long Island Sound (181-395 ng/g DW), NY/NJ Harbor (281-2006 ng/g DW), Hackensack NJ (42-2668 ng/g DW) and portions of Great Bay near Portsmouth NH (207-572 ng/g DW)(Balcom et al. 2015, Hammerschmidt et al. 2004, Hammerschmidt et al. 2008, Gosnell et al. 2015). Meanwhile, the undeveloped lower estuary sites (Buttonwood, SeaBreeze, Fortescue, and North Beach) and the residential Timber Blvd had sediment THg concentrations more closely matching less developed estuarine sites such as Wells ME (9.4 ng/g DW), Waquoit Bay MA (3.2 ng/g DW), Barn Island CT (74 ng/g DW), and portions of the Chesapeake Bay (60-200 ng/g DW) (Balcom et al. 2015; Chen unpublished data; Chen et al. 2014; Hollweg et al. 2009; Mason et al. 2014).
While the urban/industrial sites had similar MeHg sediment concentrations to other urban estuaries, two non-urban sites (Buttonwood and SeaBreeze) had sediment MeHg concentrations that are more similar to highly impacted portions of the Great Bay and Hackensack than non-developed sites with similar sediment THg concentrations (e.g. Chesapeake Bay). Yet Fundulus mean MeHg concentrations at these two sites were within the range of both urban (Providence RI, Jamaica Bay NY) and relatively undeveloped (Barn Island CT, Waquoit Bay MA) sites in our previous work (Chen et al. 2014), indicating again a disconnect between sediment MeHg concentrations and bioaccumulation. We hypothesize that this site-to-site variation in bioaccumulation is less related to sediment Hg concentration than to local influences such as productivity gradients, marsh presence, and dietary shifts.
In a dynamic and variable environment like an estuary, the relative importance of environmental factors influencing MeHg production and subsequent bioaccumulation can be difficult to determine. Chalmers et al (2014) suggest that rural environments are more conducive to methylation, whereas urban areas are often limited in methylation efficiency by ecosystem variables such as organic carbon or alkalinity, and in turn experience less relative bioaccumulation in resident fauna. The increase in marsh presence and concurrent increase in surface sediment %MeHg and particulate MeHg concentration, as well as particulate %MeHg in the less urbanized areas along the Delaware support this hypothesis. The relatively high bioaccumulation in small fish at Luken's Marsh and S. Lukens, may be due in part to the greater presence of Phragmites marsh at these sites relative to the other highly developed sites, providing a location for methylation, contributing to increased particulate MeHg concentrations, and, in combination with other factors discussed below, creating a localized “hotspot” of bioaccumulation in the resident fish. In addition, MeHg in surface water does not follow the same spatial pattern as in the sediment, indicating the influence of MeHg inputs other than diffusive flux from sediment to the overlying water, particularly outside of the ETM.
Concentrations of dissolved MeHg in the water column were highest at Gen. Chem, whereas particulate MeHg peaked at Lukens' Marsh; both particulate and dissolved MeHg decreased downstream of Luken's, then increased in the most saline southern sites. (Figure 3). There was no overall relationship between MeHg in the sediment and the water column, but concentration of THg in the sediment at Luken's Marsh coincided with increased particulate MeHg in the water column. While a direct link between these parameters at this site cannot be made without locally elevated sediment MeHg, there is likely another source of methylation (e.g. the marsh), and Hg loading may extend upstream into the watershed, providing additional fluvial inputs of MeHg to the water column. Other studies of watershed impacts on Hg and MeHg levels in tributaries to Lake Michigan and the Chesapeake Bay (Hurley et al. 1995; Lawson et al. 2001) similarly found a correlation between MeHg concentration, %MeHg, and the extent of marsh area and/or agricultural activity within the watershed. Recently, Schartup et al. (2015) showed the decoupling of MeHg in sediment and water compartments, and the relatively greater importance of water column Hg methylation to MeHg inputs at sites of large freshwater inflow into a stratified shallow Arctic estuary.
The lack of correlation between sediment MeHg and water column MeHg in the current study indicates that resuspension of sediment and dissolved flux to the surface water may have less influence on particulate and dissolved MeHg concentrations than fluvial inputs, particularly in wetland-dominated systems like those in the more saline portions of the Delaware estuary. Gosnell et al. (2015) concluded that in the ETM region, inputs of MeHg from the sediment were an important source under low flow summer conditions (when current sampling occurred) but not at other times of the year. The data suggest that the Delaware River estuary shifts from dominantly sediment sources of increased dissolved MeHg in the ETM (observed in forage fish) to a relatively greater influence of wetland sources of water column MeHg in the lower portion (reflected in shrimp tissue concentrations). These water column dynamics, including interactions and the influence of complexation with changing DOC character and salinity on MeHg bioavailability (Schartup et al. 2015), will impact uptake and transfer through the food chain.
Within the Delaware River Estuary, real-world complexity and diversity of parameters influencing both methylation efficiency and subsequent transfer to biota are reflected in the results of analyses linking abiotic MeHg concentrations and land use patterns to bioaccumulation in low trophic level fauna. Comparison of mixed-effects regression models using AICc indicated random site effects as the most robust model to explain site-to-site differences in bioaccumulation for most species. While the analysis would have been improved with larger sample sizes, the results lend further support to the importance of within system variation to Hg and MeHg fate and bioaccumulation. Our previous work has shown a relationship between water column MeHg concentration and that in resident biota across ten estuarine systems (Chen et al. 2014), and we had anticipated observing similar relationships within the Delaware. While particulate MeHg concentration (but not the dissolved fraction) appeared to exert some influence on tissue concentrations for killifish and blue crabs in the Delaware, it is not the only influence. Within the particulate fraction alone, we hypothesize that there is large variability in the character and quality of the particles, affecting not only particulate MeHg concentration, but also the extent to which this particulate material is preferentially consumed. Gosnell et al (2015) conclude that sediment resuspension supplies particulates to the water column within the ETM. Previous studies also suggest that suspended particulates tend to get trapped in the upper estuary and not exported out of the ETM (Cook et al. 2007; Sommerfield and Wong 2011).
Due to particle trapping within the ETM and the increased presence of marshes outside of this zone, a difference in the character of particulates available for consumption may in part drive differences in bioaccumulation between the upper and lower estuary. The high particulate MeHg concentration within the ETM is most likely of sediment origin, with localized marsh influence as discussed above, and thus a lower quality food (e.g. detrital particles) than at the southern sites. In contrast, the slightly higher particulate MeHg concentrations at Fortescue and North Beach co-occur with increasing chla concentration, suggesting that particulate MeHg at the southern sites is more associated with algal cells than detrital or non-organic particles. This hypothesized difference in particulate composition would most affect bioaccumulation in pelagic herbivores.
Differing spatial patterns of bioaccumulation between grass shrimp and small fish highlight the influence of benthic versus pelagic foraging strategies (and thus sources of MeHg) for these two omnivores. The general N-S increase in MeHg and %MeHg within grass shrimp suggests differences in food availability/quality for the shrimp at the different sampling sites. Grass shrimp have been observed to graze on epiphytic algae on salt marsh grasses as well as phytoplankton and small invertebrates that would accumulate MeHg from pelagic sources (Fleeger et al. 1999; Kneib 1985; Morgan 1980). In estuaries with a large salinity gradient like the Delaware, the relative contribution of terrestrial versus marine organic matter will also vary, impacting availability and uptake of MeHg from the dissolved phase to algae. There is increasing evidence that the type of DOC has a substantial impact on uptake at the base of the pelagic food chain (Luengen et al. 2012; Schartup et al. 2015b), with MeHg bound to marine DOC being more bioavailable than that bound to terrestrial DOC, potentially helping in part to explain the apparent increase in MeHg concentration in both particulates and the shrimp consuming them in the higher salinity sites. In addition, Gosnell et al. (2017) found in their study of differences in MeHg bioaccumulation in Long Island Sound (LIS) that concentrations were lower in zooplankton in the more impacted western location (WLIS) than those of the eastern end of the estuary (ELIS) and the coastal shelf, despite higher water column concentrations of MeHg in WLIS. Overall, zooplankton in WLIS had higher δ15N, indicating the importance of terrestrial and human inputs to the pelagic food web and thus bioaccumulation in this region. These results suggest that differences in bioaccumulation in pelagic-feeding organisms in the Delaware could be driven by both differences in MeHg bioavailability over the salinity gradient due to changes in DOC composition, as well as from differences in the MeHg concentration of their algal and plankton prey.
In contrast, killifish and juvenile perch tissue concentrations are higher within the ETM, and in particular near Luken's Marsh, which could indicate greater dependence on benthic and/or detrital food sources at these sites, as well as a sediment source of MeHg within the ETM. Mummichogs have been found to rely heavily on detritus as their main food source depending on their location (McMahon et al. 2005). This reliance on detrital food sources would contribute to the increase in bioaccumulation near Luken's Marsh in fish, but not in shrimp if the fish are preferentially consuming detritus rather than algae or invertebrates at these sites. In addition, if the particulate matter is mainly composed of sediment or detritus at Luken's Marsh, it is possible that the particulate fraction itself is a substrate for methylation (Ortiz et al. 2015), contributing to the higher MeHg concentrations of both the particulates themselves, and of the small fish consuming them. The lower fish tissue concentrations at the southern sites are indicative of a more varied diet due to greater productivity and health of the ecosystem or greater reliance on a pelagic food web than in the ETM.
The patterns of bioaccumulation within the current study strengthen previous observations that sediment MeHg concentrations are not a good predictor of MeHg tissue concentrations in biota. Rather, bioaccumulation of MeHg is driven by a combination of water column MeHg concentration and bioavailability, and the influence of local landscape characteristics in driving methylation efficiency and MeHg bioavailability. In addition the major drivers of bioaccumulation may change in relative influence in different portions of the estuary. The comparison with our previous work also speaks to the influence of scale on patterns driving MeHg bioaccumulation. While the across-estuary comparison in Chen et al (2014) is indicative of more global processes, the current within-estuary comparison emphasizes the importance of local drivers such as carbon sources and bioavailability, mixing, marsh presence, and differences in productivity on the net formation and bioaccumulation of MeHg.
Past studies within the Delaware River estuary have shown decreases in fish tissue concentrations over time (Greene 2013), yet mercury bioaccumulation is still a concern. While many land-based sources of Hg to the estuary have been controlled, legacy contamination and long-range transport of Hg to the watershed may impede further improvement. Additional data are therefore needed to determine whether recent improvements in the upper estuary have translated to improvements in MeHg concentration in biota inhabiting the Bay. Data from the current study suggest there are still hotspots of bioaccumulation due to legacy contamination associated with urban and industrial areas in the upper estuary, and especially in the ETM, that may affect sport fish management policies and consumption advisories. The current data set establishes a useful base line for future comparisons of both small fish and invertebrates throughout the estuary, allowing managers to assess the success of future policy and remediation. In particular, Fundulus sp., due to their widespread availability, ease of capture, relative site fidelity, and similar spatial patterns of MeHg bioaccumulation to the juvenile perch and striped bass, may be useful as a bio-indicator species for future comparisons (Evers et al. 2008). The data set also emphasizes the importance of examining the complex site-to-site variation within a system in order to understand estuary-wide contaminant dynamics, and demonstrates that processes driving MeHg fate may be different in different regions of the same system. As other estuaries also exhibit similar transitions from urban to less developed landscapes (e.g. Chesapeake Bay or Narragansett Bay), and similar challenges from contaminants and development, these findings are relevant to understanding processes affecting MeHg fate in other estuaries as well.
Supplementary Material
Figure S1: Mean ± st, dev of %MeHg in grass shrimp.
Table S1: location of sampling sites and percent land cover estimates.
Table S2: Range, mean ± st. dev., and (n) of sediment THg, MeHg, and %LOI by site. Top 4cm averages consist of the top two sections of two replicate cores.
Table S3: Significant sediment correlations within grab samples as assessed by Spearman's rank.
Table S4: Mean ± st. dev. of biota MeHg tissue concentration (ng/g DW) and length (mm) data for all species. MeHg, length, (n).
Acknowledgments
We wish to thank A. Schartup for assistance with field collections and C. McCleery, V. Ortiz and N. Mazrui for assistance with sample processing. R. Greene provided valuable suggestions to improve the manuscript, as did three anonymous reviewers. We acknowledge help from the Delaware Department of Natural Resources and Environmental Control (DNREC). This work was funded by grants from The USDA Forest Service (Nislow, 09-JV-11242307-124), NIEHS (Chen, R01 ES021950), and NIH/NIEHS (Stanton P42 ES007373).
References
- Ashley JTF, Webster M, Horwitz R, Velinsky D, Baker J. Polychlorinated biphenyls in sediment and biota from the Delaware River Estuary. Proceedings of the Academy of Natural Sciences of Philadelphis. 2009;158:89–105. [Google Scholar]
- ATSDR. Substance Priority List. [Accessed 14 Nov 2015];2013 http://www.atsdr.cdc.gov/spl/
- Balcom P, Schartup A, Mason R, Chen CY. Sources of water column methylmercury across multiple estuaries in the Northeast U.S. Marine Chemistry. 2015;177:721–730. doi: 10.1016/j.marchem.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barton K. MuMIn: Multi-Model Inference. R Package version 1.13.4 2015 [Google Scholar]
- Bates D, Maechler M, Bolker B, Walker S. Lme4: Linear mixed-effects models using Eigen and S4. R package version 1.1-7 2014 [Google Scholar]
- Bloom N, Fitzgerald WF. Determination of volatile mercury species at the picogram level by low-temperature gas chromatography with cold-vapour atomic fluorescence detection. Analytica Chimica Acta. 1988;208:151–161. [Google Scholar]
- Bloom NS, Crecelius EA. Determination of mercury in seawater at sub-nanogram per liter levels. Marine Chemistry. 1983;14:49–59. [Google Scholar]
- Burns DA, McHale MR, Driscoll CT, Roy KM. Response of surface water chemistry to reduced levels of acid precipitation: comparison of trends in two regions of New York, USA. Hydrological Processes. 2006;20:1611–1627. [Google Scholar]
- Chalmers AT, Krabbenhoft DP, Van Metre PC, Nilles MA. Effects of urbanization on mercury deposition and New England. Environmental Pollution. 2014;192:104–112. doi: 10.1016/j.envpol.2014.05.003. [DOI] [PubMed] [Google Scholar]
- Chen CY, Borsuk M, Bugge D, Hollweg T, Balcom P, Ward DM, Williams J, Mason R. Benthic and pelagic pathways of methylmercury bioaccumulation in estuarine food webs of the northeast United States. PLoS ONE. 2014;9:e89305. doi: 10.1371/journal.pone.0089305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CY, Dionne M, Mayes BM, Ward DM, Sturup S, Jackson BP. Mercury bioavailability and bioaccumulation in estuarine food webs in the Gulf of Maine. Environmental Science & Technology. 2009;43:1804–1810. doi: 10.1021/es8017122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CY, Serrell N, Evers DC, Fleishman BJ, Lambert KF, Weiss J, Mason RP, Bank MS. Meeting Report: Methylmercury in marine ecosystems-from sources to seafood consumers. Environmental Health Perspectives. 2008;116:1706–1712. doi: 10.1289/ehp.11211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Church, Sommerfield CK, Velinsky D, Point D, Benoit C, Amouroux D, Plaa D, Donard O. Marsh sediments as records of sedimentation, eutrophication and metal pollution in the urban Delaware Estuary. Marine Chemistry. 2006;102:72–95. [Google Scholar]
- Conaway CH, Squire S, Mason RP, Flegal AR. Mercury speciation in the San Francisco Bay estuary. Marine Chemistry. 2003;80:199–225. [Google Scholar]
- Cook TL, Sommerfield CK, Wong KC. Observations of tidal and springtime sediment transport in the upper Delaware Estuary. Estuarine Coastal and Shelf Science. 2007;72:235–246. [Google Scholar]
- DE Department of Natural Resources and Environmental Control. Delaware Fish Consumption Advisories. [Accessed 16 Dec 2016];2016 PDF available at http://www.dnrec.delaware.gov/fw/Fisheries/pages/advisories.aspx.
- Driscoll CT, Chen CY, Hammerschmidt CR, Mason RP, Gilmour CC, Sunderland EM, Greenfield BK, Buckman KL, Lamborg CH. Nutrient supply and mercury dynamics in marine ecosystems: A conceptual model. Environmental Research. 2012;119:118–131. doi: 10.1016/j.envres.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drott A, Lambertsson L, Bjorn E, Skyllberg U. Do potential methylation rates reflect accumulated methyl mercury in contaminated sediments? Environmental Science & Technology. 2008;42:153–158. doi: 10.1021/es0715851. [DOI] [PubMed] [Google Scholar]
- Dutton J, Fisher NS. Salinity effects on the bioavailability of aqueous metals for the estuarine killifish Fundulus heteroclitus. Environmental Toxicology and Chemistry. 2011;30:2107–2114. doi: 10.1002/etc.600. [DOI] [PubMed] [Google Scholar]
- Eshleman KN, Sabo RD, Kline KM. Surface Water Quality Is Improving due to Declining Atmospheric N Deposition. Environmental Science & Technology. 2013;47:12193–12200. doi: 10.1021/es4028748. [DOI] [PubMed] [Google Scholar]
- Evers DC, Mason RP, Kamman NC, Chen CY, Bogomolni AL, Taylor DL, Hammerschmidt CR, Jones SH, Burgess NM, Munney K, Parsons KC. Integrated mercury monitoring program for temperate estuarine and marine ecosystems on the North American Atlantic coast. EcoHealth. 2008;5(4):426–441. doi: 10.1007/s10393-008-0205-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitzgerald WF, Gill GA. Subnanogram determination of mercury by two-stage gold amalgamation and gas phase detection applied to atmospheric analysis. Analytical Chemistry. 1979;51:1714–1720. [Google Scholar]
- Fleeger JW, Carman KR, Webb S, Hilbun N, Pace MC. Consumption of microalgae by the grass shrimp Palaemonetes pugio. Journal of Crustacean Biology. 1999;19:324–336. [Google Scholar]
- Fry B, Chumchal MM. Mercury bioaccumulation in estuarine food webs. Ecological Applications. 2012;22:606–623. doi: 10.1890/11-0921.1. [DOI] [PubMed] [Google Scholar]
- Gosnell K, Balcom P, Ortiz V, DiMento B, Schartup A, Greene R, Mason R. Seasonal cycling and transport of mercury and methylmercury in the estuarine turbididty maximum of the Delaware Estuary. Aquatic Geochemistry 2015 [Google Scholar]
- Gosnell K, Balcom PH, Tobias C, Gilhooly W, Mason RP. Spatial and temporal trophic transfer dynamics of mercury and methylmercury into zooplankton and phytoplankton of Long Island Sound. Limnology and Oceanography. 2017 doi: 10.1002/lno.10490. [DOI] [Google Scholar]
- Greene R. Mercury in Delaware surface water. Powerpoint presentation given at the 2007 Delaware Water Science Seminar; Dover DE. 2007. [Google Scholar]
- Greene R. An assessment of sediment metals data from the Delaware Estuary Benthic Inventory (DEBI) 2011 Powerpoint presentation given Feb 1, 2011. [Google Scholar]
- Greene R. Assessment of 2012 Delaware Toxics in Biota Data. Dover, DE: Delaware DNREC, Division of Watershed Stewardship; 2013. [Google Scholar]
- Hammerschmidt CR, Fitzgerald WF. Formation of artifact methylmercury during extraction from a sediment reference material. Analytical Chemistry. 2001;73:5930–5936. doi: 10.1021/ac010721w. [DOI] [PubMed] [Google Scholar]
- Hammerschmidt CR, Fitzgerald WF, Balcom PH, Visscher PT. Organic matter and sulfide inhibit methylmercury production in sediments of New York/New Jersey Harbor. Marine Chemistry. 2008;109:165–182. [Google Scholar]
- Hammerschmidt CR, Fitzgerald WF, Lamborg CH, Balcom PH, Visscher PT. Biogeochemistry of methylmercury in sediments of Long Island Sound. Marine Chemistry. 2004;90:31–52. [Google Scholar]
- Hollweg TA, Gilmour CC, Mason RP. Methylmercury production in sediments of Chesapeake Bay and the mid-Atlantic continental margin. Marine Chemistry. 2009;114:86–101. [Google Scholar]
- Homer C, Dewitz J, Yang LM, Jin S, Danielson P, Xian G, Coulston J, Herold N, Wickham J, Megown K. Completion of the 2011 National Land Cover Database for the Conterminous United States - Representing a Decade of Land Cover Change Information. Photogrammetric Engineering and Remote Sensing. 2015;81:345–354. [Google Scholar]
- Hurlbert SH. Pseudoreplication and the design of ecological field experiments. Ecological Monographs. 1984;54:197–211. [Google Scholar]
- Hurley JP, Benoit JM, Babiarz CL, Shafer MM, Andren AW, Sullivan JR, Hammond R, Webb DA. Influences of watershed characteristics on mercury levels in Wisconsin rivers. Environmental Science & Technology. 1995;29(7):1867–1875. doi: 10.1021/es00007a026. [DOI] [PubMed] [Google Scholar]
- Kneib R. Predation and disturbance by grass shrimp, Palaemonetes pugio Holthuis, in soft-substratum benthic invertebrate assemblages. Journal of Experimental Marine Biology and Ecology. 1985;93:91–102. [Google Scholar]
- Lambertsson L, Nilsson M. Organic material: the primary control on mercury methylation and ambient methyl mercury concentrations in estuarine sediments. Environmental Science & Technology. 2006;40:1822–1829. doi: 10.1021/es051785h. [DOI] [PubMed] [Google Scholar]
- Lawson NM, Mason RP, Laporte JM. The fate and transport of mercury, methylmercury, and other trace metals in Chesapeake Bay tributaries. Water Research. 2001;35(2):501–515. doi: 10.1016/s0043-1354(00)00267-0. [DOI] [PubMed] [Google Scholar]
- Luengen AC, Fisher NS, Bergamaschi BA. Dissolved organic matter reduces algal accumulation of methylmercury. Environmental Toxicology and Chemistry. 2012;31:1712–1719. doi: 10.1002/etc.1885. [DOI] [PubMed] [Google Scholar]
- Mason R, Balcom P, Gosnell K, Ortiz V, Dimento B, Schartup A. Final Report: Sources and biogeochemical cycling of methylmercury and mercury in the sub-tidal reaches of the Delaware River Estuary 2014 [Google Scholar]
- Mason RP, Lawrence AL. Concentration, distribution, and bioavailability of mercury and methylmercury in sediments of Baltimore Harbor and Chesapeake Bay, Maryland, USA. Environmental Toxicology and Chemistry. 1999;18:2438–2447. [Google Scholar]
- Mason RP, Reinfelder JR, Morel FM. Uptake, toxicity, and trophic transfer of mercury in a coastal diatom. Environmental Science & Technology. 1996;30:1835–1845. [Google Scholar]
- Mazerolle MJ. AICcmodavg: Model selection and mulitmodel inference based on (Q)AIC(c) R package version 2.0-1 2014 [Google Scholar]
- McMahon KW, Johnson BJ, Ambrose WG. Diet and movement of the killifish, Fundulus heteroclitus, in a Maine salt marsh assessed using gut contents and stable isotope analyses. Estuaries. 2005;28:966–973. [Google Scholar]
- Morgan MD. Grazing and predation of the grass shrimp Palaemonetes pugio. Limnology and Oceanography. 1980;25:896–902. [Google Scholar]
- Nakagawa S, Schielzeth H. A general and simple method for obtaining R2 from Generalized Linear Mixed-effects Models. Methods in Ecology and Evolution. 2013;4 [Google Scholar]
- NJ Department of Environmental Protection. Fish Smart, Eat Smart A guide to health advisories foreating fish and crabs caught in New Jersey waters. [Accessed 16 Dec 2016];2016 PDF available at http://www.state.nj.us/dep/dsr/njmainfish.htm.
- Pinedo S, Jordana E, Monia Flagella M, Ballesteros E. Relationships between heavy metals contamination in shallow marine sediments with industrial and urban development in Catalonia (Northwestern Mediterranean Sea) Water Air and Soil Pollution. 2014;225 [Google Scholar]
- Ortiz VL, Mason RP, Ward JE. An examination of the factors influencing mercury and methylmercury particulate distributions, methylation and demethylation rates in laboratory-generated marine snow. Marine chemistry. 2015;177:753–762. doi: 10.1016/j.marchem.2015.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinherio JC, Bates DM. Mixed-effects models in S and S-Plus. New York: Springer; 2000. [Google Scholar]
- R Core Team. Version 3.1.3. Vienna, Austria: Foundation for Statistical Computing; 2015. R: A language and environment for statistical computing. [Google Scholar]
- Reinfelder J, Totten L. Mercury in the Delaware River Estuary: Past and present. Report to the Delaware River Basin Commission 2006 [Google Scholar]
- Rodriguez W, August PV, Wang Y, Paul JF, Gold A, Rubinstein N. Empirical relationships between land use/cover and estuarine condition in the Northeastern United States. Landscape Ecology. 2007;22:403–417. [Google Scholar]
- Sanudo-Wilhelmy SA, Gill GA. Impact of the Clean Water Act on the levels of toxic metals in urban estuaries: The Hudson River estuary revisited. Environmental Science & Technology. 1999;33:3477–3481. [Google Scholar]
- Schartup AT, Balcom PH, Soerensen AL, Gosnell KJ, Calder RSD, Mason RP, Sunderland EM. Freshwater discharges drive high levels of methylmercury in Arctic marine biota. Proceedings of the National Academy of Sciences of the United States of America. 2015a;112:11789–11794. doi: 10.1073/pnas.1505541112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schartup AT, Mason RP, Balcom PH, Hollweg TA, Chen CY. Methylmercury production in estuarine sediments: Role of organic matter. Environmental Science & Technology. 2013;47:695–700. doi: 10.1021/es302566w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schartup AT, Ndu U, Balcom PH, Mason RP, Sunderland EM. Contrasting effects of marine and terrestrially derived dissolved organic matter on mercury speciation and bioavailability in seawater. Environmental Science & Technology. 2015b;49:5965–5972. doi: 10.1021/es506274x. [DOI] [PubMed] [Google Scholar]
- Skjelkvale BL, Stoddard JL, Jeffries DS, Torseth K, Hogasen T, Bowman J, Mannio J, Monteith DT, Mosello R, Rogora M, Rzychon D, Vesely J, Wieting J, Wilander A, Worsztynowicz A. Regional scale evidence for improvements in surface water chemistry 1990-2001. Environmental Pollution. 2005;137:165–176. doi: 10.1016/j.envpol.2004.12.023. [DOI] [PubMed] [Google Scholar]
- Sommerfield CK, Wong KC. Mechanisms of sediment flux and turbidity maintenance in the Delaware Estuary. Journal of Geophysical Research-Oceans. 2011;116 [Google Scholar]
- Sunderland EM. Mercury exposure from domestic and imported estuarine and marine fish in the US seafood market. Environmental Health Perspectives. 2007;115:235–242. doi: 10.1289/ehp.9377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunderland EM, Amirbahman A, Burgess NM, Dalziel J, Harding G, Jones SH, Kamai E, Karagas MR, Shi X, Chen CY. Mercury sources and fate in the Gulf of Maine. Environmental Research. 2012;119:27–41. doi: 10.1016/j.envres.2012.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor VF, Carter A, Davies C, Jackson BP. Trace-level automated mercury speciation analysis. Analytical Methods. 2011;3:1143–1148. doi: 10.1039/C0AY00528B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor VF, Jackson BP, Chen CY. Mercury speciation and total trace element determination of low-biomass biological samples. Analytical and Bioanalytical Chemistry. 2008;392:1283–1290. doi: 10.1007/s00216-008-2403-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng CM, Hammerschmidt CR, Fitzgerald WF. Determination of methylmercury in environmental matrixes by on-line flow injection and atomic fluorescence spectrometry. Analytical Chemistry. 2004;76:7131–7136. doi: 10.1021/ac049118e. [DOI] [PubMed] [Google Scholar]
- United Nations Environmental Programme. Global Mercury Assessment 2013: Sources, Emissions, Releases and Environmental Transport. UNEP Chemicals Branch; Geneva, Switzerland: 2013. p. 44. [Google Scholar]
- U.S. Environmental Protection Agency. Method 7473: Mercury in solids and solutions by thermal decomposition, amalgamation, and atomic absorption spectrophotometry. Washington, D.C: 1998. [Google Scholar]
- U.S. Environmental Protection Agency. 2011 National Listing of Fish Advisories. 2013 EPA-820-F-13-058. [Google Scholar]
- Van Metre PC. Increased atmospheric deposition of mercury in reference lakes near major urban areas. Environmental Pollution. 2012;162:209–215. doi: 10.1016/j.envpol.2011.11.003. [DOI] [PubMed] [Google Scholar]
- Zuur A, Ieno E, Walker N, Saveliev A, Smith G. Mixed effects models and extensions in ecology with R. New York: Springer; 2009. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1: Mean ± st, dev of %MeHg in grass shrimp.
Table S1: location of sampling sites and percent land cover estimates.
Table S2: Range, mean ± st. dev., and (n) of sediment THg, MeHg, and %LOI by site. Top 4cm averages consist of the top two sections of two replicate cores.
Table S3: Significant sediment correlations within grab samples as assessed by Spearman's rank.
Table S4: Mean ± st. dev. of biota MeHg tissue concentration (ng/g DW) and length (mm) data for all species. MeHg, length, (n).