Abstract
Patients with mutations of the THRA gene exhibit classical features of hypothyroidism, including erythroid disorders. We previously created a mutant mouse expressing a mutated TRα1 (denoted as PV; Thra1PV/+ mouse) that faithfully reproduces the classical hypothyroidism seen in patients. Using Thra1PV/+ mice, we explored how the TRα1PV mutant acted to cause abnormalities in erythropoiesis. Thra1PV/+ mice exhibited abnormal red blood cell indices similarly as reported for patients. The total bone marrow cells and erythrocytic progenitors were markedly reduced in the bone marrow of Thra1PV/+ mice. In vitro terminal differentiation assays showed a significant reduction of mature erythrocytes in Thra1PV/+ mice. In wild-type mice, the clonogenic potential of progenitors in the erythrocytic lineage was stimulated by thyroid hormone (T3), suggesting that T3 could directly accelerate the differentiation of progenitors to mature erythrocytes. Analysis of gene expression profiles showed that the key regulator of erythropoiesis, the Gata-1 gene, and its regulated genes, such as the Klf1, β-globin, dematin genes, CAII, band3 and eALAS genes, involved in the maturation of erythrocytes, was decreased in the bone marrow cells of Thra1PV/+ mice. We further elucidated that the Gata-1 gene was a T3-directly regulated gene and that TRα1PV could impair erythropoiesis via repression of the Gata-1 gene and its regulated genes. These results provide new insights into how TRα1 mutants acted to cause erythroid abnormalities in patients with mutations of the THRA gene. Importantly, the Thra1PV/+ mouse could serve as a preclinical mouse model to identify novel molecular targets for treatment of erythroid disorders.
Author summary
Patients with mutations of the THRA gene exhibit erythroid disorders. The molecular pathogenesis underlying erythroid abnormalities is poorly understood. In Thra1PV/+ mice expressing a dominant negative mutant TRα1PV, we found abnormal red blood cell indices similar to patients. Total bone marrow cells, the clonogenic potential of erythrocytic progenitors, and terminal differentiation of erythrocytes were markedly decreased in Thra1PV/+ mice. We elucidated that Gata-1, a key erythroid gene, was directly positively regulated by TRα1. The erythroid defects in Thra1PV/+ mice were due, at least partly, to the TRα1PV-mediated suppression of the Gata-1 gene and its down-stream target genes. Over-expression of Gata-1 rescued impaired terminal differentiation. Our studies elucidated molecular mechanisms by which TRα1 mutants caused erythroid disorders in patients. The present study suggests that therapies aimed at GATA1 could be tested as a potential target in treating erythroid abnormalities in patients.
Introduction
Thyroid hormones have long been known to play an important role in erythropoiesis. Early in vitro studies demonstrated that L-thyroxine (T4) stimulates the biosynthesis of hemoglobin [1]. Experimental animal models also showed that T4 enhances red blood cell formation and stimulates hemoglobin synthesis [2]. Studies in humans have shown a causal association between hypothyroidism and anemia [3]. Persons with subclinical hypothyroidism had lower hemoglobin levels [4, 5] and a higher prevalence of anemia than euthyroid persons. Treatment of patients with subclinical hypothyroidism with thyroid hormone resulted in a significant increase in hemoglobin content [6] or erythropoietin levels [7]. However, how hypothyroidism results in erythroid disorders at the molecular level remains largely unknown.
Recently, patients with mutations of the thyroid hormone receptor α gene (THRA) have been reported to exhibit some of the classical symptoms and signs of hypothyroidism with impaired growth and delayed bone development [8–11]. These patients also display anemia. These findings suggested that erythropoietic disorders in humans are mediated by TRα1 mutants and support the critical role of TRα1 in erythropoiesis. The availability of a mouse model (Thra1PV/+ mice), harboring a mutated TRα1 (designated as TRα1PV) [12], has made it possible for us to elucidate the role of TRα1 mutants in erythroid disorders. TRα1PV has a C-terminal mutated sequence (398-PPFVLGSVRGLD- 409) [12], similar to the truncated C-terminal sequence in two patients (398-PPTLPRGL -405) [9]. The PV mutation was first identified from a patient with severe resistance to thyroid hormone (RTHβ), characterized by elevated thyroid hormone levels accompanied by normal TSH, short stature, goiter, and tachycardia [13]. The PV mutated sequence was targeted to the Thra gene at the corresponding position as in THRB gene to assess the functional consequence of TRα1 mutations at the time when no patients with the mutations of the THRA gene was discovered. Thra1PV/+ mice exhibit displayed phenotype of hypothyroidism as in the patients with severe growth retardation [12] and delayed bone development [12, 14, 15]. The Thra1PV/+ mouse has been used as a preclinical model to test the effectiveness of T4 treatment for the correction of impaired bone development due the actions of mutated TRα1 [16]. These studies further validate the usefulness of the Thra1PV/+ mouse to understand how mutations of the THRA gene result in deleterious abnormalities in patients.
In the present study, we first characterized the erythroid phenotypes in Thra1PV/+ mice and showed that Thra1PV/+ mice exhibit anemia with decreased red blood cells and reduced hemoglobin content similar to patients with mutations of the THRA gene [11]. We further identified the genes that were abnormally regulated by TRα1PV, resulting in defective erythropoiesis. Thus, our study has provided direct molecular evidence to show that mutations of the THRA gene could impair erythropoiesis and has uncovered novel molecular actions of TRα1 in the erythroid differentiation and development.
Results
Alterations of peripheral blood composition in Thra1PV/+ mice
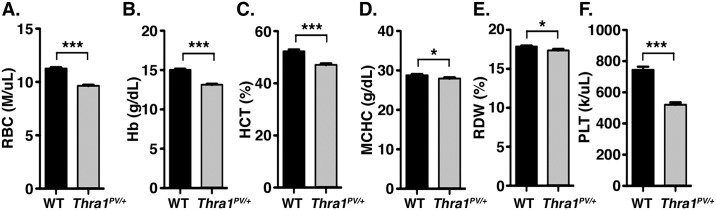
We first analyzed peripheral blood composition to characterize erythropoietic phenotypes in Thra1PV/+ mice. Complete blood count revealed that major indices for erythrocytes, such as red blood cell count (RBC), hemoglobin levels (Hb) and hematocrit (HCT), were significantly lower in Thra1PV/+ mice than in wild-type (WT) mice. As shown in Fig 1, RBC, Hb, and HCT were decreased by 14% (panel A), 13% (panel B), and 10% (panel C), respectively. Other erythrocyte peripheral indices, namely, the mean corpuscular hemoglobin concentration (MCHC), red cell distribution width (RDW), and platelets (PLT), were 3% (panel D), 3% (panel E), and 30% (panel F) lower in Thra1PV/+ mice than in WT mice. These results indicated that mutations of the Thra gene could affect different lineages leading to defects.
Fig 1. Alterations in the red blood cell indices of Thra1PV/+ mice.
Comparison of complete blood counts between wild-type (WT) and Thra1PV/+ mice: panel A, the number of red blood cells (RBC); B, hemoglobin (Hb); C, hematocrit (HCT); D, mean corpuscular hemoglobin concentration (MCHC); E, red cell distribution width (RDW); and F, platelets (PLT) (wild type, n = 32; Thra1PV/+ mice, n = 31). Values are means ± SEM. Any statistical difference by t test versus control is indicated by p values. * denotes p<0.05; ** denotes p<0.01, *** p<0.001.
Defective cellularity in the bone marrow of Thra1PV/+ mice
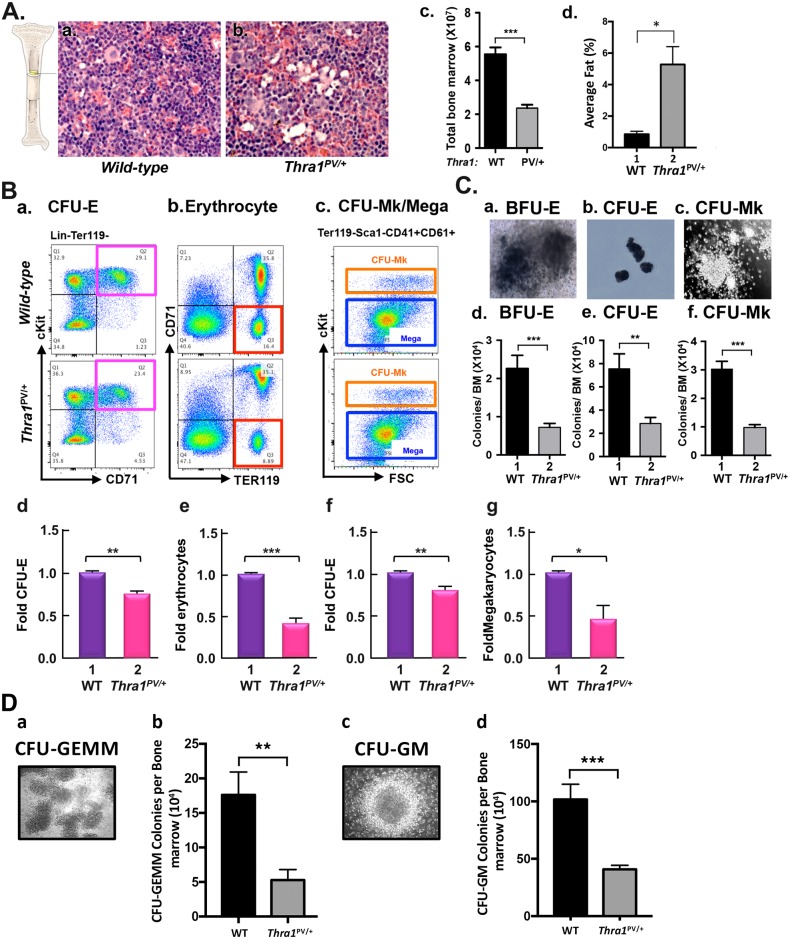
After birth and throughout adult life, the bone marrow remains the major hematopoietic organ in mice [17]. We therefore examined the histology of H & E stained femur sections to assess the cellularity of the bone marrow. As shown in Fig 2A, fewer bone marrow cells with a higher fat deposit were apparent in Thra1PV/+ mice than wild-type mice (Fig 2A, compare panel b with panel a). By counting cell numbers, we found that total bone marrow cells were decreased 57% in Thra1PV/+ mice (Fig 2A-c). We have measured the fat areas in the bone marrow of wild-type mice (n = 3) and Thra1PV/+ mice (n = 3). The quantitative data is now shown as Fig 2A-d, indicating that the fat (% of total area) was 6.2-fold higher in the bone marrow of Thra1PV/+ mice than in wild-type mice.
Fig 2. Decreased bone marrow cellularity and progenitors in Thra1PV/+ mice.
(A) Hematoxylin and eosin-stained femurs sections from wild-type and Thra1PV/+ mice (a and b). Magnification = X330. (c). The numbers of total bone marrow cells were counted from wild-type (panel a; n = 20) and Thra1PV/+ mice (panel b; n = 18). (d). The fat areas in the bone marrow of wild-type mice (n = 3) and Thra1PV/+ mice (n = 3) shown in Figure A-a and A-b were measured by morphometry. Values are means ± SEM (n = 3). (B) The distribution of respective erythroid and megakaryocytic populations in the bone marrow of wild type and Thra1PV/+ mice. Erythroid and megakaryocytic progenitors and mature cells were analyzed by FACS using cell surface markers. CFU-E erythrocyte progenitors identified in the bone marrow cells that had characteristics of Lin-Ter119-cKit+CD71+ (the pink box in panel a). Erythrocytes identified as CD71-Ter119+ bone marrow cells (the red box in panel b). Colony-forming units-megakaryocytes (CFU-Mk) megakaryocytic progenitors identified as Ter119-Sca1-CD41+CD61+cKit+ bone marrow cells (the orange box in panel c). Megakaryocytes were identified as Ter119-Sca1-CD41+CD61+cKit- bone marrow cells (the blue box in panel c). The numbers of CFU-E cells (panel d), erythrocytes (panel e), CFU-Mk cells (panel f), and megakaryocytes (panel g) were counted and the data calculated as fold changes in Thra1PV/+ versus wild-type mice. The representative data from three experiments are shown. Values are means ± SEM (n = 3). The p values are shown. (C) In vitro colony forming assays of bone marrow cells from wild-type and Thra1PV/+ mice (panels a-c). Morphological characteristics of burst-forming units-erythroid (BFU-E) progenitors (panel a), CFU-E progenitors (panel b), and CFU-Mk progenitor (panel c) colonies by phase contrast microscopy. Panels d, e, and f show the scored colonies of the BFU-E, CFU-E, and CFU-Mk. BFU-E was counted after 14 days. CFU-E was counted after 2 days. CFU-Mk was counted after 7 days in vitro culturing. The data are presented as ratios of total number of colonies versus total bone marrow cells. Values are means ± SEM (duplicates in each assay; WT mice, n = 7; Thra1PV/+ mice, n = 6). * denotes p<0.05; ** denotes p<0.01, *** p<0.001. (D). Morphological characteristics of multi-potential (CFU-GEMM) progenitor colonies (panel a), granulocyte/macrophage progenitor cells (CFU-GM) (panel c) by phase contrast microscopy (x100). Panels b and d show the number of colonies of CFU-GEMM and CFU-GM, respectively. The colonies were counted after 8 days in vitro culturing as described in Methods. The data are presented as number of colonies versus total bone marrow cells. Values are means ± SEM (WT mice, n = 4; Thra1PV/+ mice, n = 4; quadruplicate in each assay).
We next assessed which abnormal population of progenitors in the bone marrow cells of Thra1PV/+ mice contributed to anemia. It is known that a small population of hematopoietic stem cells (HSC) gives rise to multipotent progenitors (MPP), which subsequently differentiate into common myeloid population cells (CMP population cells) and common lymphoid progenitors (CLP). CMP is differentiated into megakaryocytic/erythroid (MEP) and granulocyte-myeloid (GMP) progenitors. Because patients with mutations of the THRA gene exhibit anemia, we focused on the analysis of the lineage derived from MEP, and its subsequent downstream progenitors: burst-forming unit-erythroid (BFU-E) to colony-forming unit-erythroid (CFU-E), then to erythroblasts, and ultimately to mature erythrocytes [18–20]. To assess the effect of TRα1PV mutation on the number of progenitors in the MEP lineage, we analyzed the progenitors by specific cell surface markers in the bone marrow using flow cytometry. Erythroid cells at different developmental stages can be identified by different cell surface markers: Ter119 (erythroid specific glycophorin) and CD71 (the transferrin receptor). Ter119 expression increases as maturation of erythrocyte progresses [21, 22]. CD71 is the transferrin receptor that expresses at high levels in early erythroid precursors, but its levels decrease toward erythroid maturation [21, 22]. cKit (CD117) is a cytokine receptor expressed on the surface of hematopoietic stem cells, MPP, and CMP. Stem cells antigen 1 (Sca1) is expressed in HSC [19, 23]. Using these specific cell markers, we identified which sub-populations were altered in the bone marrow cells of Thra1PV/+ mice.
CFU-E progenitors reside in the lineage negative (Lin-)Ter119-CD71+cKit+ populations of the bone marrow (Fig 2B, panel a). CFU-E progenitors were decreased 24% in Thra1PV/+ mice compared with wild-type mice (Fig 2B, panel d). Mature red blood cells and reticulocyte precursors exhibited characteristic Ter119+CD71- expression on the cell surface (Fig 2B, panel b). We found that erythrocytes were decreased 56% in the bone marrow of Thra1PV/+ mice (Fig 2B, panel e). CFU-Mk progenitors and megakaryocytes reside in Ter119-Sca1-CD41+CD61+ population (Fig 2B panel c). The only difference between CFU-Mk and megakaryocyte is that CFU-Mk expresses cKit as a marker in the progenitors. We found that CFU-Mk progenitors and megakaryocytes were decreased 19% and 52%, respectively, in the bone marrow of Thra1PV/+ mice (Fig 2B, panels f and g, respectively). Notably, the extent of differences between WT and Thra1PV/+ mice was greater in the CFU-E and erythrocytes than in CFU-Mk and megakaryocytes.
We further carried out in vitro colony forming unit assays. BFU-E progenitors were defined as cells that colonize after 14 days under defined-in vitro culture conditions (Fig 2C, panel a). CFU-E progenitors were colonized cells after 2 days in defined culture conditions (Fig 2C, panel b). CFU-Mk progenitors were counted after 7 days in defined culture conditions (Fig 2C, panel c). BFU-E, CFU-E, and CFU-Mk (expressed as relative colonies versus total bone marrow cells) were decreased 68% (Fig 2C-d), 62% (Fig 2C-e), and 68% (Fig 2C-f), respectively, in Thra1PV/+ mice as compared with WT mice. We further carried out colony assays for CFU-GEMM and CFU-GM. The number of CFU-GEMM and CFU-GM colonies as shown in Fig 2D-a and 2D-c, respectively, was 70% and 60% lower, respectively, in Thra1PV/+ mice than in WT mice (Fig 2D-b and 2D-d). These results imply that the capacity of progenitor cells to differentiate from MEP to erythroblasts as well as to megakaryocytes was impaired, leading to erythroid disorders in Thra1PV/+ mice.
Stimulation of the erythroid lineage differentiation by T3
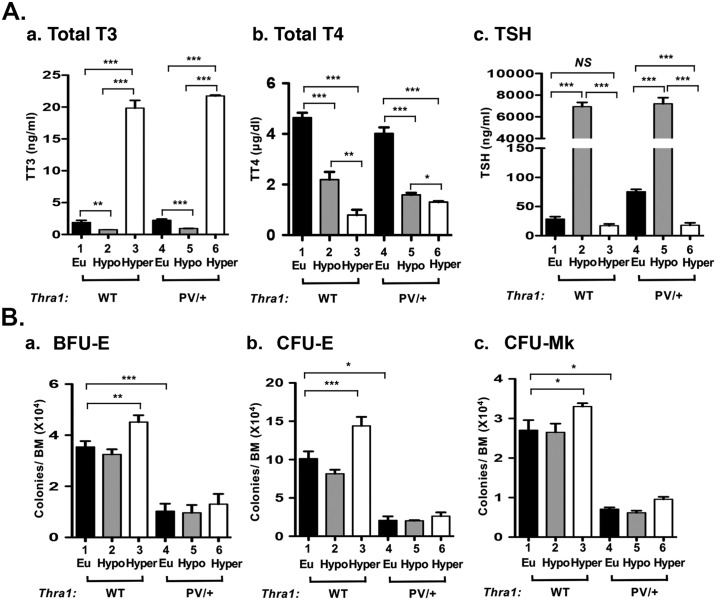
TRα1PV is a dominantly negative mutant and cannot bind T3 [12]. That the capacity of the progenitors to form BFU-E, CFU-E, and CFU-Mk was impaired in the bone marrow of Thra1PV/+ mice prompted us to ascertain whether the differentiation from MEP to downstream BFU and CFU was regulated by thyroid hormones. We therefore rendered Thra1PV/+ mice hypothyroid by treating them with PTU and rendered the PTU-treated mice hyperthyroid by T3 treatment. As shown in Fig 3A, PTU treatment was effective in lowering serum total T3 and T4 levels (bar 2 in panels a and b, respectively) in WT mice. In line with the lowering of T3 and T4, TSH was highly elevated (bar 2 in Fig 3A-c). T3 treatment of PTU-treated WT mice led to highly elevated T3 (Fig 3A-a, bar 3) and so suppressed TSH levels (Fig 3A-c, bar 3). Treatment of Thra1PV/+ mice with PTU followed by T3 injection led to similar changes as shown for WT mice (Fig 3A-a, 3A-b and 3A-c, bars 4–6). These findings are consistent with the earlier reports that the feedback loop in the pituitary-thyroid axis was not affected by expressing TRα1PV mutant in Thra1PV/+ mice [12].
Fig 3. Comparison of effects of thyroid hormone on the progenitors in the megakaryocyte/erythroid lineage of wild-type and Thra1PV/+ mice.
(A) Serum total T3 (panel a), total T4 (panel b), and TSH (panel c) were determined from wild type and Thra1PV/+ mice (each n = 3) under the euthyoid, hypothyroid and hyperthyroid condition as described in Methods. (B) Colonies of erythroid progenitors (BFU-E, panel a, and CFU-E, panel b) and megakaryocytic progenitors (CFU-Mk, panel c) were counted from total bone marrow of euthyroid (filled bars), hypothyroid (grey bars), and hyperthyroid (open bars) mice. Values are means ± SEM (duplicates in each assay from WT mice, n = 3; Thra1PV/+ mice, n = 3). * denotes p<0.05; ** denotes p<0.01; *** p<0.001.
We next isolated bone marrow cells from hypothyroid (PTU-treated) and hyperthyroid (T3-treated) mice and carried out colony forming assays. No significant differences in BFU-E (Fig 3B-a, bars 1–2), CFU-E (Fig 3B-b, bars 1–2), and CFU-Mk (Fig 3B-c, bars 1–2) were detected between hypothyroid and euthyroid mice. However, T3 treatment increased the numbers of BFU-E (Fig 3B-a, bar 3), CFU-E (Fig 3B-b, bar 3), and CFU-Mk (Fig 3B-c, bar 3) by 42%, 77% and 25%, respectively from hypothyroid to hyperthyroid mice. In contrast, in Thra1PV/+ mice, compared to WT mice, there was markedly decreased BFU-E (Fig 3B-a), CFU-E (Fig 3B-b), and CFU-Mk (Fig 3B-c) (compare bar 4–6 to bars 1–3). Moreover, the extent of decreases in Thra1PV/+ mice were not affected by T3 treatment (bars 4–6). These results indicated that colony forming units derived from MEP were stimulated by T3 in WT mice, but were not affected by T3 in Thra1PV/+ mice due to the actions of dominant negative TRα1PV mutant.
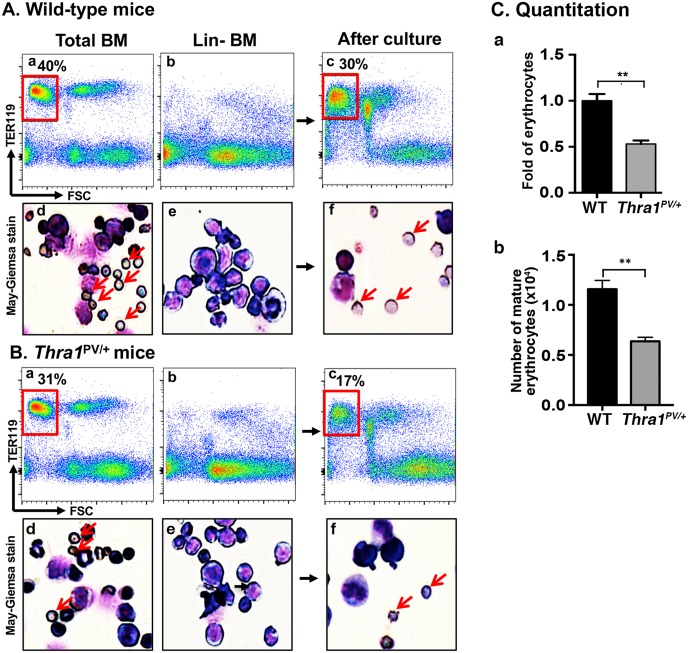
To further confirm that TRα1PV mutant acted to inhibit the differentiation in the erythroid lineage, we isolated lineage depleted bone marrow cells (Lin-BM) by eliminating mature lineage cells including T cells, B cells, macrophages, granulocytes, and erythrocytes. Using Lin- BM, we compared the maturation of erythrocytes in WT and Thra1PV/+ mice using an in vitro terminal erythropoiesis system [24]. Using an equal number of total bone marrow cells from WT (Fig 4A-a) and Thra1PV/+ mice (Fig 4B-a), we found 40% and 31%, respectively, of Ter119+ with low FSC population (boxed in red). As shown by May-GrÜnwald—Giemsa staining (Fig 4A-d and 4B-d), these populations were enriched with mature erythrocytes (marked by red arrows). After depletion of mature lineage cells as evidenced by markedly reduction of Ter119-positive cells (panel b in Fig 4A and 4B) and enucleated erythrocytes by May-GrÜnwald—Giemsa staining (panel e in Fig 4A and 4B), progenitor cells were stimulated by erythyropoietin to undergo terminal erythropoiesis [24]. After culturing for 3 days, Ter119-positive cells associated with low FSC fraction were detected (panel c in Fig 4A and 4B). The enucleated cells were visualized by May-GrÜnwald—Giemsa staining (panel f in Fig 4A and 4B). The gated Ter119-positive cells associated with low FSC fraction (boxed in red) in panel c were quantified: 30% and 17% of mature erythrocytes were detected for WT and Thra1PV/+ mice, respectively. These results represent a 47% reduction in the capacity of Lin- MB cells from Thra1PV/+ mice to mature into erythrocytes (Fig 4C).
Fig 4. Decreased terminal erythroid differentiation in the lineage depleted bone marrow cells (Lin- BM) of Thra1PV/+ mice.
(A and B) Total bone marrow profile from wild-type (A-a) and Thra1PV/+ mice (B-a) as determined by flow cytometry using a late erythroid surface phenotype (Ter119+FSClow) population is boxed in red. The corresponding May-GrÜnwald-Giemsa stained cells are shown (A-d for wild-type mice and B-d for Thra1PV/+ mice). Population of Lin- BM cells from wild-type (A-b) and Thra1PV/+ (B-b) mice. After 3-day culturing, Ter119+FSClow population is shown boxed in red (A-c for wild-type mice and B-c for Thra1PV/+ mice). An arrow indicates an enucleated erythrocyte after May-GrÜnwald-Giemsa staining (A-f for wild-type mice and B-f for Thra1PV/+ mice). (C-a) Quantitative analysis shows the fold changes of erythrocytes after terminal erythroid differentiation of Lin- BM cells of wild-type and Thra1PV/+ mice. (C-b). The number of matured erythrocytes from Lin-bone marrow cells of WT mice and Thra1PV/+ mice were also compared. Values are means ± SEM (n = 3). ** denotes p<0.01.
The expression of the Gata1 and its regulated genes is repressed in the bone marrow of Thra1PV/+ mice
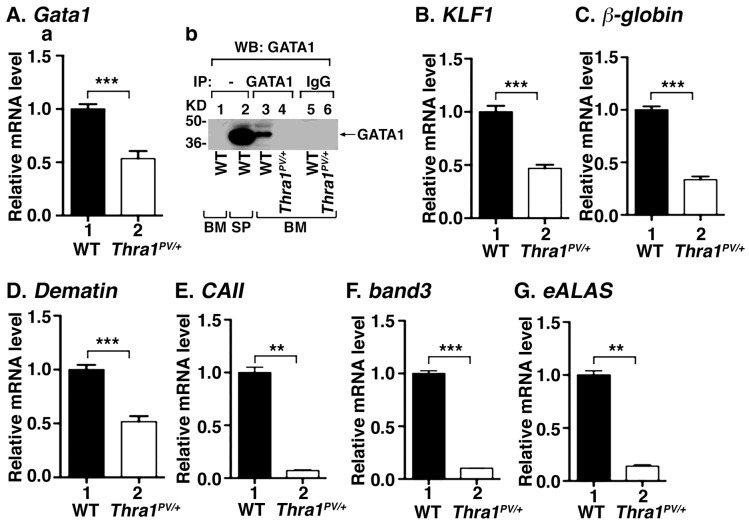
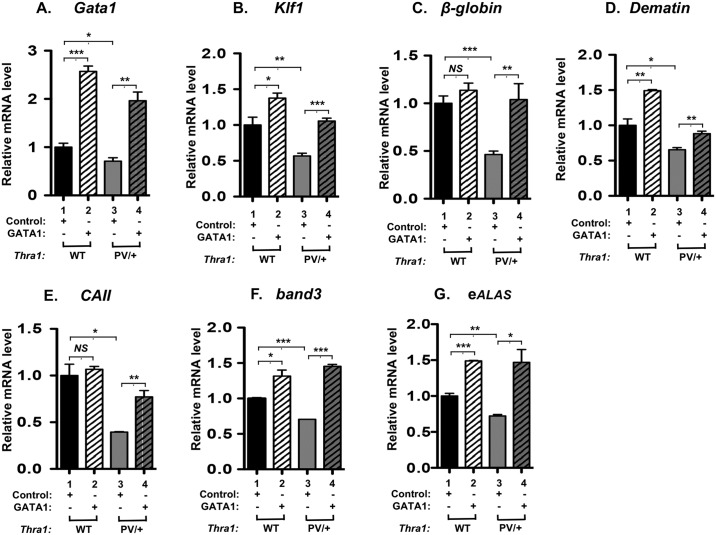
To investigate the molecular mechanisms by which TRα1PV mutant induced erythropoietic disorders, we first analyzed the expression of a major regulator of erythropoiesis, the Gata1 gene (erythroid transcription factor; GATA-binding factor 1). GATA1 is a member of the GATA transcription factor family and is essential for erythroid development by regulating a large ensemble of genes that mediate both the development and function of red blood cells [25, 26]. The loss of Gata1 expression leads to erythroid maturation arrest and embryonic lethality due to anemia [27–31]. We found the expression of the Gata1 gene was ~50% lower in the bone marrow of Thra1PV/+ mice than in WT mice (Fig 5A-a, compare bar 2 with bar 1). Direct western blot analysis shows that the GATA1 protein level in the bone marrow was lower than that in the spleen of WT mice (compare lane 2 with lane 1, Fig 5A-b). However, when the bone marrow cell lysates of WT mice were first enriched by immunoprecipitation followed by western blot analysis (Co-IP), GATA1 proteins were detected (lane 3, Fig 5A-b). In contrast, under identical experimental conditions, GATA1 proteins were not detected (lane 4), indicating that the GATA1 protein level in the bone marrow of Thra1PV/+ mice was lower than that of WT mice. Lanes 5 and 6 were the corresponding negative controls in that an irrelevant IgG was used in the immunoprecipitation step. That no signals were detected in lanes 5 and 6 indicated that the protein detected by Co-IP was specific. GATA1 regulates the expression of the Klf1 gene, which drives erythropoiesis by affecting its downstream target genes critical for maturation of erythrocytes [25, 32, 33]. Consistent with decreased expression of the Gata1 gene, the expression of the Klf1 gene was 53% lower in the bone marrow of Thra1PV/+ mice (Fig 5B, bar 2 versus bar 1). We also analyzed the expression of KLF1 and GATA1 target genes, such as the β-globin (beta major globin) and the dematin genes (erythroid membrane and cytoskeleton related gene). The β-globin protein along with α-globin makes up the most common form of hemoglobin in adult humans. Dematin is a relatively low abundance actin binding and bundling protein associated with the spectrin—actin junctions of mature erythrocytes. Dematin binds to spectrin and dynamically regulates red cell membrane mechanical function [34]. Consistent with the reduced expression of the Klf1 gene, the expressions of the β-globin and dematin genes were also repressed by 70% and 50%, respectively, in the bone marrow of Thra1PV/+ mice (Fig 5C and 5D). We further analyzed the expression of CAII, band 3 and eALAS genes, which are regulated by GATA-1 during erythropoiesis. Moreover, these three genes are known to be directly regulated by TRα1 and T3 in birds [35–37]. The expression of these three genes was inhibited 93%, 90% and 86%, respectively, in the bone marrow of Thra1PV/+ mice (Fig 5E, 5F and 5G, respectively). Taken together, these data indicate that suppression of Gata-1 gene and the known T3- target genes led to impaired erythropoiesis.
Fig 5. Decreased erythropoiesis related gene expression in the bone marrow of Thra1PV/+ mice.
The mRNA levels of Gata1 (panel A-a), Klf1 (panel B), β-globin (panel C), dematin (panel D), CA II (panel E), band 3 (panel F) and eALAS (panel G) in bone marrow cells were determined by quantitative real-time PCR. Values are means ± SEM (n = 2–4). * denotes p<0.05; ** denotes p<0.01; *** p<0.001. (A-b). Direct western blot analysis of GATA1 protein levels in the bone marrow and spleen of WT mice (lanes 1, and 2, respectively). (A-b). Analysis of the GATA1 protein levels in the bone marrow by Co-IP as described in Methods (lanes 3 and 5, WT mice; lanes 4 and 6, Thra1PV/+ mice). Lanes 5 and 6 were the negative controls using mouse IgG in the immunoprecipitation. BM: bone marrow; SP: spleen.
Repression of the Gata1 gene by TRα1PV mediates erythroid disorders in Thra1PV/+ mice
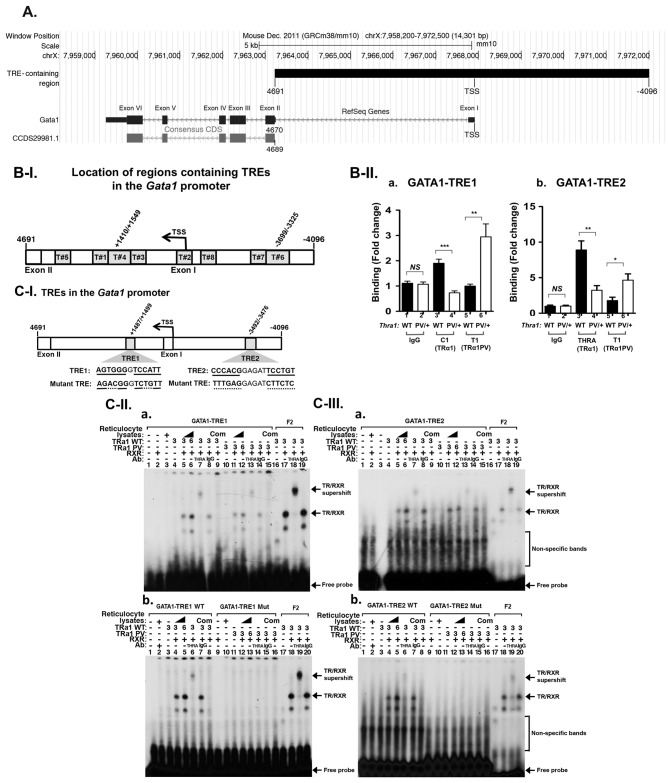
That the expression of the Gata1 gene was suppressed by TRα1PV prompted us to ascertain whether the Gata1 gene was directly regulated by TR/T3. We searched for putative thyroid hormone response elements (TREs) in the GATA1 hematopoietic regulatory domain [38], located upstream of the transcription starting site (TSS) of the Gata1 gene (Fig 6A). We also searched for putative TREs in the +4691 intronic sequence between exon I and exon II (Fig 6A). In this -4096 to +4691 region, we found putative TREs-containing regions with the core consensus sequences of the hexa-nucleotide “half-site” (A/G)GGT(C/A/G)A (T#1 –T#8, Fig 6B-I). To identify relevant TREs, we used chromatin immunoprecipitation (ChIP) to ascertain the binding of TRα1 to these eight putative TREs in the bone marrow of WT and Thra1PV/+ mice. When anti-TRα1 antibodies were used, a significantly higher binding of TRα1 to TRE1 (AGTGGGGTCCATT in the region +1410 to +1549 bp) were found in the bone marrow of WT mice than when anti-IgG antibodies (negative controls) were used (bar 3 versus bar 1, Fig 6B-II-a). In the bone marrow of Thra1PV/+ mice, when specific anti-TRα1PV antibodies (rabbit polyclonal antibody T1) were used, a significantly higher binding of TRα1PV to TRE1 than when anti-IgG antibodies (negative controls) were used (bar 6 versus bar 2, Fig 6B-II-a). Similar significantly higher binding of TRα1 (bar 3 versus bar 1, Fig 6B-II-b) and TRα1PV (bar 6 versus bar 2, Fig 6B-II-b) to TRE2 (CCCACGGAGATTCCTGT, in the region -3699 to -3325 bp) were also detected. However, we did not find specific binding of TRα1 and TRα1PV to other regions containing putative TREs (i.e., T#1, T#2, T#3, T#5, T#7, and T#8, Fig 6B-I). These ChIP results suggested that TRα1 and TRα1PV could bind directly to these TRE-containing regions in TRE1 (region T#4) and TRE2 (region T#6) on the intronic region and the proximal promoter, respectively, of the Gata1 gene.
Fig 6. Identification of thyroid hormone response elements in the Gata1 promoter.
(A) Schematic representation of the mouse Gata1 gene locus. The promoter region encompassed -4096 upstream from the transcription starting site (TSS) and the intronic sequences between exon I and exon II. The ATG was from 4689 to 4691. This figure was prepared with UCSC Genome Browser <http://genome.ucsc.edu>. (B-I) The putative TRE with consensus binding half sites are marked by numbers. (B-II) ChIP analysis in the binding of TRα1 (bars 3–4) or TRα1PV (bars 5–6) to TRE1 and TRE2 in panel a and b, respectively. IgG was used as a background control. Values are means ± SEM (n = 3). (C-I) The locations and sequences of wild-type and mutated TRE1 and TRE2 in the Gata1 gene promoter. (C-II) EMSA analysis in the binding of TRα1 and TRα1PV to TRE1 (panel a) and to mutated TRE1 (panel b). (C-III) EMSA analysis in the binding of TRα1 and TRα1PV to TRE2 (panel a) and to mutated TRE2 (panel b). Lanes are marked in (C-II) and (C-III). The bands representing binding of TRE as TR/RXR heterodimers are marked by arrows.
We next used electrophoretic mobility shift assay (EMSA) to demonstrate directly the binding of TRα1 and TRα1PV to TREs in TRE1 and TRE2 regions (Fig 6C-I). As shown in Fig 6C-II-a, TRα1 prepared by in vitro transcription/translation system, bound to TRE1 (the TRE sequence is shown in Fig 6C-I) as heterodimers with the retinoid X receptor (RXR) (lanes 5 and 6 show the increasing concentrations of TRα1). The TRE1-bound TRα1 was supershifted by anti-TRα1 antibodies (lane 7), but not by control anti-IgG antibodies (lane 8). In the presence of unlabeled TRE, no binding to 32P-labeled TRE1 was detected (lane 9). These data indicate that TRE1 bound specifically to TRα1 as heterodimers with RXR. Similarly, TRα1PV also bound to TRE1 as heterodimers with RXR (lanes 11–12), which was supershifted by anti-TRα1PV (lane 13). No specific binding to labeled TRE in the presence of unlabeled TRE was observed (lane 15). Lanes 16–19 were positive controls using a labeled F2 (TRE with inverted two half-sites). Moreover, when TRE1 from AGTGGGGTCCATT was mutated to AGACGGGTCTGTT, no binding was detected by EMSA (Fig 6C-II-b; compare lanes 11–16 with lanes 3–8 in which wild-type TRE was used in EMSA). These EMSA results indicate that TRE1 bound directly and specifically to TRα1 and TRα1PV. Using similar EMSA, we also found specific binding of TRα1 (Fig 6C-III-a, lanes 5–9) and TRα1PV (Fig 6C-III-a, lanes 11–15) to TRE2. The binding was further confirmed by mutational analysis in which, when TRE2 was mutated from CCCACGGAGATTCCTGT to TTTGAGGAGATCTTCTC, no binding was detected by EMSA (Fig 6C-III-b; compare lanes 11–16 with lanes 3–8 in which wild-type TRE was used in EMSA). Taken together, these results indicate that we have uncovered two specific TRα1 and TRα1PV binding TREs in the promoter of the Gata1 gene.
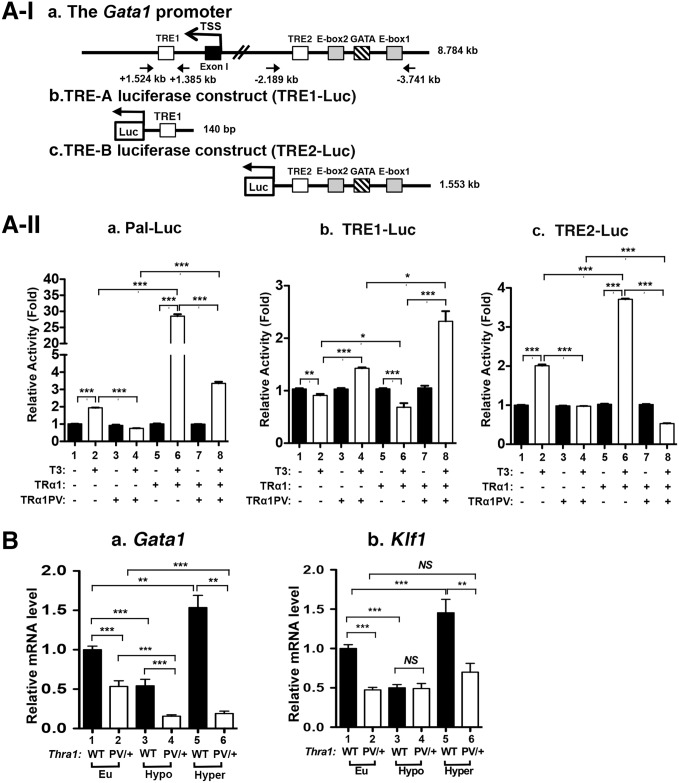
To assess whether these two TREs mediated the TRα1-dependent regulation of the transcription of the Gata1 gene, we constructed reporters in which the expression of luciferase is mediated by TRE1 or TRE2. We cloned the 0.140 kb fragment containing the TRE1 (Fig 7A-I-a and 7A-I-b) and 1.553 kb fragment containing the TRE2 into the pGL4.23 luciferase plasmid (Fig 7A-I-b and 7A-I-c, respectively). It is of interest to note that in the 1.553 kb fragment, one GATA box and two E-boxes, enhancers to regulate the transcription of the Gata1 gene, were also present [39]. These two Luc-reporters were transfected into human erythroleukemia 562 (K562) cells, which express endogenous TRα1 [40]. Indeed, using a Luc-reporter containing palindromic TRE (Pal-Luc), the reporter activity mediated by the endogenous TRα1 was detected in the presence of T3 (Fig 7A-II-a, bar 2 versus bar 1). When the TRα1PV expression vector was transfected into K562 cells, the Pal-Luc reporter activity was suppressed by dominant negative activity of TRα1PV (bar 4 versus bar 2). Transfection of the TRα1 expression plasmid led to an additional 15-fold activation of Pal-Luc reporter activity (bar 6 versus bar 2). This TRα1/T3-mediated activation was repressed by the transfection of the TRα1PV expression plasmid (bar 8 versus bar 6). These results indicate that K562 cells are a good model cell line to evaluate the activity of TRE1 and TRE2 in the regulation of the Gata1 gene by TRα1.
Fig 7. Thyroid hormone response elements in the Gata1 promoter mediates the transcription of TRα1.
Schematic representation of the Gata1 promoter with TRE1 downstream of TSS site and TRE2 together with GATA box and two E-boxes upstream of the TSS (A-I-a), the luciferase reporter construct containing TRE1 (A-I-b), the luciferase reporter construct containing TRE2 (A-I-c). Reporter activity mediated by a Palindromic TRE (Pal-Luc) (A-II-a), TRE1 (A-II-b) and TRE2 (A-II-c) in K562 cells expressing endogenous TR (bars 1–2) and with transfected TRα1PV(bars 3–4), with cells transfected with TRα1 without TRα1PV (bars 5–6) or with TRα1PV (bars 7–8), in the absence or presence of T3. Values are means ± SEM (n = 3). (B) Effects of thyroid hormone on the expression of the Gata1 (panel a, n = 4) and the Klf1 gene (panel b; n = 3). The treatment of mice with PTU with or without T3, to make mice hypothyroid or hyperthyroid, was the same as described in Fig 3. Values are means ± SEM (n = 3). * denotes p<0.05; ** denotes p<0.01; *** p<0.001.
In contrast to Pal-Luc, in the presence of T3, a small, but significant repression of TRE1-Luc activity was observed (Fig 7A-II-b, bar 2 versus 1). Interestingly, transfection of TRα1PV led to a 1.3-fold activation of TRE1-Luc activity (bar 4 versus 2). When TRα1 expression plasmid was transfected into K562 cells, more suppression of TRE1-Luc activity than by endogenous TRα1 was observed (bar 6 versus 2). Remarkably, co-transfection of the TRα1PV expression plasmid with TRα1 expression plasmid into K562 cells resulted in a 2.5-fold activation of TRE1-Luc activity (bar 8 versus bar 6). These results demonstrated that TRE1 mediated the negative regulation activity of TRα1. These findings are reminiscent of the regulation of the Tshα common subunit (α-SU) gene by TR/T3 in the pituitary of Thra1PV/+ mice [12].
In contrast to TRE1-Luc activity, the luciferase activity mediated by TRE2 was activated two-fold by T3 in K562 cells (Fig 7A-II-c, bar 2 versus bar 1). This T3-stimuated reporter activity was totally abolished by transfection of the TRα1PV expression plasmid (bar 4 versus bar 2). Transfection of the TRα1 expression plasmid led to an additional 1.9-fold activation of the reporter activity (bar 6 versus bar 2). This T3-activated reporter activity mediated by TRα1 was totally abolished by co-transfection of the TRα1PV expression plasmid (bar 8 versus bar 6). These results indicate that TRE2, via T3-TRα1, mediates the positive regulation of the Gata1 gene transcription.
The identification of one positive TRE and one negative TRE in the promoter prompted us to assess the overall regulation by TRα1of the Gata1 gene in the bone marrow. We rendered WT and Thra1PV/+ mice hypothyroid by treating them with PTU and then some PTU-treated mice with T3 to make them hyperthyroid (see also Fig 3). The expression of the Gata1 mRNA was ~50% lower in hypothyroid WT mice than in euthyroid WT mice (Fig 7B-a, bar 3 versus bar 1). The expression of the Gata1 mRNA was ~3-fold higher in hyperthyroid WT mice than in hypothyroid WT mice (Fig 7B-a, bar 5 versus bar 3). These results indicate that the Gata1 gene was positively regulated by T3. In Thra1PV/+ mice, Fig 7B-a also shows that the expression of the Gata1 mRNA in the bone marrow of euthyroid Thra1PV/+ mice was ~50% lower than that in euthyroid WT mice (bar 2 versus 1). The Gata1 mRNA expression in hypothyroid Thra1PV/+ mice was also lower than that in hypothyroid WT mice (bar 4 versus 3). However, the expression of the Gata1 mRNA in the hyperthyroid in Thra1PV/+ mice was not significantly increased as compared with that in hypothyroid mice (Fig 7B-a, bar 6 versus bar 4), indicating that TRα1PV expressed in the bone marrow of Thra1PV/+ mice has lost transcription capacity due to its inability to bind T3.
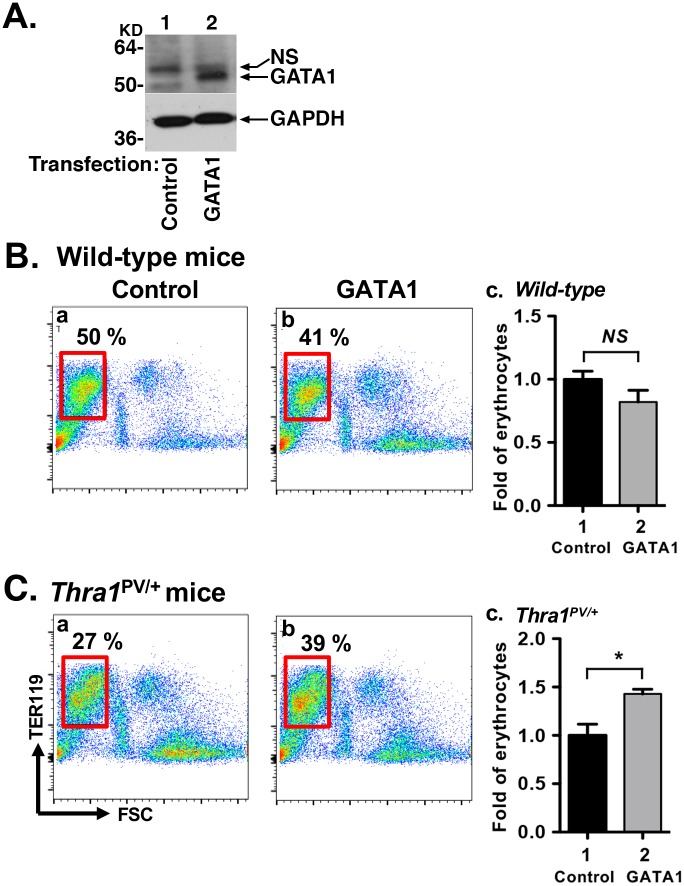
Consistent with the regulation of the Gata1 gene by T3, similar T3-regulatory patterns in the expression of the Klf1 gene was found in the WT mice (Fig 7B-b). The Klf1 mRNA level was lower in the hypothyroid mice than in euthyroid mice (bar 3 versus bar 1), but was higher in hyperthyroid mice than in hypothyroid mice (Fig 7B-b, bar 5 versus bar 3). The Klf1 mRNA level was lower in euthyroid Thra1PV/+ mice than in euthyroid WT mice (bar 2 versus 1). No apparent increase in the expression of the Klf1 mRNA level was found in hyperthyroid Thra1PV/+ mice as compared with hypothyroid Thra1PV/+ mice (Fig 7B-b, bar 6 versus bar 4) due to the loss of T3 binding activity and transcriptional activity of TRα1PV. Taken together, these results indicate that the Gata1 gene is directly positively regulated by T3/TRα1. Mutations of TRα1, such as TRα1PV, suppressed the expression of the Gata1 gene to impair erythropoiesis in Thra1PV/+ mice. To further support this notion, we carried out rescue experiments. We exogenously expressed the GATA1 gene in the Lin-bone marrow cells followed by terminal differentiation assay. As shown in Fig 8A, GATA1-tagged with V5 protein was detected by anti-V5 antibodies (lane 2), whereas it was not detected in the bone marrow cells transfected with on the control plasmid (lane 1). FACS analysis shows that exogenously expressed GATA1 did not further increase mature erythrocytes in WT mice, as indicated by Ter119+ with low FSC population (Fig 8B-a versus 8B-b, areas boxed in red). In contrast, exogenously expressed GATA1 led to increase in mature erythrocytes (39% in the Fig 8C-b versus 27% in the Fig 8C-a, areas boxed in red) in Thra1PV/+ mice. Quantitation graph represent a 43% increase in mature erythrocytes from Thra1PV/+ mice (Fig 8C, panel c). These data demonstrated that the defective terminal erythropoiesis from Lin-BM in Thra1PV/+ mice was partially corrected by exogenous expression of GATA1 protein.
Fig 8. Defective terminal erythropoiesis was rescued by overexpression of the GATA1 gene.
(A) Expression of GATA1 tagged with V5 was determined in the bone marrow cells by western blot analysis using anti-V5 antibodies. Anti-GAPDH antibody served as the loading control. (B). Terminal erythropoiesis was determined by flow cytometry using a late erythroid surface phenotype (Ter119+FSClow, boxed in red) after transfection of control (panel a) or GATA1 expression plasmids (panel b) in wild-type (B) and Thra1PV/+ mice (C). The Fold changes of matured erythrocytes after transfection of GATA1 gene in WT mice (B, panel c) and Thra1PV/+ mice (C, panel c) are indicated. Values are means ± SEM (n = 3). * denotes p<0.05.
We further evaluated whether the exogenously transfected GATA1 as described above could affect the erythroid genes critical for erythropoiesis. Fig 9 shows the expression of Gata1 (panel A), Klf1 (panel B), β-globin (panel C), dematin (panel D), CAII (panel E), band3 (panel F), and eALAS (panel G) were all increased by the exogenous expression of GATA1 (compare lanes 4 with lanes 3 in all panels). These results indicated that the elevated expression of these erythroid genes contributed to the partial rescue of the defective erythropoiesis and further support the essential role of GATA1 in mediating the impaired erythropoiesis in Thra1PV/+ mice.
Fig 9. Expression of erythroid genes was increased by overexpression of the GATA1 gene in the bone marrow of Thra1PV/+ mice.
After transfection of V5-tagged GATA1 plasmids into bone marrow cells, mRNA expression of erythroid genes was analyzed by quantitative real-time PCR (panel A-G). mRNA expression of Gata1 (panel A), Klf1 (panel B), β-globin (panel C), dematin (panel D), CAII (panel E), band 3 (panel F) and eALAS (panel G) was increased in the bone marrow of Thra1PV/+ mice (lanes 4) compared with the empty vector control (lanes 3). Values are means ± SEM (n = 3). NS: not significant; * denotes p<0.05; ** denotes p<0.01; *** p<0.001.
Discussion
In 2002, we created the Thra1PV/+ mouse that displayed aspects of the phenotype of hypothyroidism with retarded growth, delayed bone development, and marginally abnormal thyroid function tests [12]. These phenotypic manifestations are distinct from those of the ThrbPV/+ mice that faithfully reproduce resistance to thyroid hormone (RTH) in patients caused by mutations of the THRB gene [41]. The distinct phenotypic manifestations by Thra1PV/+ mice and ThrbPV/+ mice suggested TR isoform-dependent actions of TR mutants in vivo. However, because of the lack of patients with mutations of the THRA gene at the time, it was not clear whether this postulate held true. The discovery of patients with mutations of the THRA gene in 2012 [8], exhibiting similar hypothyroidism as in the Thra1PV/+ mouse, has validated that the Thra1PV/+ mouse is a valuable model to elucidate the molecular basis of hypothyroidism caused by mutated TRα1. Indeed, Thra1PV/+ mice have been used to predict the outcome of prolonged treatment with a supraphysiologic dose of T4 aimed at ameliorating the skeletal abnormalities in patients [16]. The studies provided valuable information showing that patients with different THRA mutations would display responses to T4 treatment that vary depending on the severity of the causative mutation.
In the present studies, we used Thra1PV/+ mice to understand the molecular actions of TRα1 mutants that lead to erythroid disorders in patients. We found that Thra1PV/+ mice exhibited abnormal red blood cell indices, decreased erythroid lineage progenitors in the bone marrow, and reduction in the terminal differentiation of progenitors in the erythroid lineage. Moreover, for the first time, we have identified a key erythropoietic gene, the Gata1 gene, as a direct TR/T3 target gene by the discovery of thyroid hormone response elements in the promoter region of that gene. Thus, using the Thra1PV/+ mouse, we have provided direct evidence to indicate that mutations of TRα1 could lead to erythroid disorders and that the disorders are mediated, at least in part, by the suppressed expression of a key erythropoietic regulator, the Gata1 gene, by TRα1 mutants. The present studies have shed new light on the molecular basis of the erythroid disorders found in patients with mutations of the THRA gene.
The critical role of TRα1 was also demonstrated in another mouse model, the Thra-/- mouse [42]. Deficiency of TRα1 led to defective fetal and adult erythropoiesis in that erythroid progenitor numbers were decreased in fetal livers; moreover, terminal maturation of erythrocytes was impaired [42]. While the erythroid defects observed in Thra-/- mice were similar to those of Thra1PV/+ mice, the underlying mechanisms would be different. The defective erythropoiesis in Thra-/- mice would be mediated by the lack of TRα1 in regulating the transcription of erythroid-related TRα1 target genes. In contrast, in Thra1PV/+ mice, the impaired erythropoiesis was due to the dominant negative action of TRα1 mutants on erythroid-related TRα1 target genes, such as the suppression of the T3-positively regulated Gata1 gene demonstrated in the present studies. At present, in Thra-/- mice, the erythroid-related TRα1 target genes affected by TRα1 deficiency have not been elucidated. It is difficult to compare the extent and the scope of erythroid abnormalities from the TRα1 deficiency or mutated TRα1 until a comprehensive analysis of global gene expression profiles becomes available. Still, the similar erythroid defects observed in Thra-/- mice and Thra1PV/+ mice clearly highlight the important role of TRα1 in erythropoiesis.
The loss of normal functions of TRα1 due to mutations as in Thra1PV/+ mice or due to TRα1 deficiency as in Thra-/- mice could lead to erythroid disorders as shown in the present studies and in Kendrick et al. [42], respectively. However, that wild-type TRα1 plays a critical role in erythropoiesis was also demonstrated by the findings that thyroid hormones promoted the clonogenic forming ability of BFU-E, CFU-E, and CFU-Mk in hyperthyroid mice shown in the present studies (Fig 3B). These findings are consistent with the observations from clinical studies, indicating that thyroid hormones play a critical role in erythropoiesis. Qualitative and quantitative studies of erythropoiesis in patients with hyperthyroidism exhibit mild erythrocytosis with erythyroid hyperplasia and increased erythropoietic activity in the bone marrow [43]. In addition, erythrocyte counts, serum erythropoietin, and hypoxia-inducible factor 1α levels patients with untreated Graves' hyperthyroidism were significantly higher than those in the age- and sex-matched healthy controls. Methimazole or subsequent radioiodine therapy of patients with hyperthyroidism reduced erythrocytosis and thyroid function returned to normal, suggesting that thyroid hormone promotes erythrocytosis [44]. Moreover, recent population-based studies on euthyroid subjects revealed a significant positive association between thyroid hormones and erythrocyte indices, such as erythrocyte counts, and hemoglobin levels [45]; [46]. These association studies support the notion that thyroid hormones stimulate erythropoiesis. Still, the molecular mechanisms by which thyroid hormones promote erythropoiesis in hyperthyroidism are not clear. The models of Thra1PV/+ and Thra-/- mice would provide valuable tools for such studies.
Early studies have shown that v-erbA is one of the two oncogenes of the avian erythroblastosis virus (AEV) [47], an acute chicken retrovirus that induces lethal erythroleukemia and sarcoma in vivo. V-erbA is a mutated TRα1, which acts in neoplasia by blocking erythroid differentiation and by altering the growth properties of fibroblasts [47]. Similar to TRα1PV, v-erbA functions as a transcription repressor by dominant negative interference with the transcription activity of its normal cellular homolog, c-erbA (TRα1). While the v-erbB locus alone is sufficient to induce erythroleukemia and sarcoma independent of the v-erbA gene, the v-erbA by itself is not capable of independently causing transformation in either erythroid cells or fibroblasts [47, 48], The expression of the v-erbA gene in erythroid cells blocks terminal differentiation and keeps the cells in very immature and highly proliferative stages [47]. Similar to v-erbA, TRα1PV is a potent dominant negative mutant of TRα1. These observations on the functional characteristics of v-erbA raised an important question about whether TRα1PV could act as an oncogene in Thra1PV/+ mice. Up to now, we have not observed any transformed phenotypes in erythroid cells of Thra1PV/+ mice. It is likely that another oncogene such as v-erbB (a mutated version of epidermal growth factor receptor; EGFR) would be needed, as in AEV-induced erythroleukemia and sarcoma, to collaborate with TRα1PV to bring out the transformed phenotypes in erythroid cells of Thra1PV/+ mice. This possibility is testable and awaits future studies.
The present studies have shown that Thra1PV/+ mice exhibited erythroid disorders with abnormal red blood cell indices, decreased total bone marrow cells, and reduced clonogenic potential of erythroid progenitors. The defective erythropoiesis was mediated by TRα1PV-mediated suppression of a key erythropoietic gene (the Gata1 gene), resulting in concurrent repression of other genes involved in the maturation of erythrocytes. Two TREs were identified on the Gata1 gene that responded to T3 differently in the reporter assay. TRα1/T3 via interacting with TRE1 mediated suppression of the transcription whereas TRα1/T3 via interacting with TRE2 activated transcription. It is of interest to point out that located upstream of TRE2 are two E-boxes and one GATA box (GATA1 gene hematopoietic enhancers) critical to the GATA1 gene transcription (see Fig 7A-I). In view of the findings that in vivo, T3 activated the overall transcription of the Gata1 gene as shown in the hyperthyroid WT mice (Fig 7B-a, makes it tempting to speculate that TRE1, though shown to be negatively regulated by T3 in the reporter assay (Fig 7A-II-b), could conceivably be affected by the GATA1 gene hematopoietic enhancers via a long-range looping mechanism, functionally acting as a positive TRE. However, interestingly, in the chicken GATA1 promoter, a negative TRE was identified [49]. TRα1 binds to this TRE as heterodimers with the chicken ovalbumin upstream promoter transcription factor [49], suggesting that the regulatory role of TRα1 in the GATA1 gene transcription is conserved between chicken and mouse.
The identification of the Gata1 gene as a key regulator would suggest that the Gata1 gene or the genes it regulates could potentially be targets for treatment. Moreover, novel TR isoform-specific thyroid hormone analogs are being developed. The Thra1PV/+ mouse would be a valuable model to test the effectiveness of these potential targets to correct the erythroid disorders.
Materials and methods
Mice and treatment
All animal studies were performed according to the approved protocols of the National Cancer Institute Animal Care and Use Committee. The animal study protocol is NCI LMB-036. Mice harboring the mutated Thra1PV gene (Thra1PV mice) were prepared and genotyped by PCR as described earlier [12]. Wild-type (Thra1+/+) and Thra1PV/+ female siblings were used in this study. To induce hypothyroidism, mice were fed a low-iodine diet supplemented with 0.15% propylthiouracil (LoI/PTU) (Cat# TD 95125, Harlan Teklad, Madison, WI) for 10 days. To induce hyperthyroidism, T3 (5 μg; Cat# T2752, Sigma-Aldrich, St. Louis, MO) was injected intraperitoneally to each mouse for 6 days while they were being fed with LoI/PTU diet. The same volume of vehicle (phosphate-buffered saline) was injected in the control group.
Cells
Erythroleukemia K562 cell line was maintained in RPMI1650 (Thermo Fisher Scientific, Waltham, MA) with 10% fetal bovine serum (FBS; GE Healthcare life science, Marlborough, MA) with 50 units/ml penicillin G and 50 μg/ml streptomycin (Thermo Fisher Scientific, Waltham, MA). Bone marrow cells were isolated from femurs and tibiae of wild-type and Thra1PV mice (age: 3–5 months). Single cell suspensions were prepared by passing bone marrow through a 70 μM cell strainer.
Peripheral blood profile analysis
For analysis of complete blood counts, peripheral blood was collected in a heparinized microtube and analyzed by hematology analyzer (HEMAVET HV950FS, Drew Scientific, Miami Lakes, FL).
Serum thyroid stimulating hormone (TSH), total T3, and T4 assays
The level of TSH in serum was measured as described [50]. Total T4 (TT4) and T3 (TT3) levels were determined by using Gamma Coat T4 and T3 assay radioimmunoassay (RIA) kits according to the manufacturer’s instruction (Cat# 06B256447 and 06B254029, MP Biomedical, LLC, Solon, OH).
Colony assays
To detect burst-forming units-erythroid (BFU-E) colonies, 5 X 104 bone marrow cells were seeded in duplicates in semisolid medium (Methocult M3434; STEMCELL Technologies, Vancouver, BC). To detect colony forming units-erythroid (CFU-E) colonies, 8 X 104 bone marrow cells were seeded in duplicates in semisolid medium (Methocult M3334; STEMCELL Technologies, Vancouver, BC). To detect colony- forming units-megakaryocytes (CFU-Mk) colonies, 1 X 105 bone marrow cells were seeded in duplicates in semisolid medium (Methocult-c, 04974; STEMCELL Technologies, Vancouver, BC) supplemented with 10 ng/ml Interleukin (IL)-3, 20 ng/ml Interleukin (IL)-6, 50 ng/ml thrombopoietin (TPO) (STEMCELL Technologies, Vancouver, BC). To analyze the colonies of multi-potential progenitor cells (CFU-GEMM) and granulocyte/macrophage progenitor (CFU-GM), 4 x 104 bone marrow cells were mixed with semisolid medium (Methocult GF M3434; STEMCELL Technologies, Vancouver, BC) by vortexing. Bone marrow cells (4 x 104 cells) from WT mice (n = 4) and Thra1PV/+ mice (n = 4) were seeded in 6 wells plate (DENVILLE, SCIENTIFIC INC., quadruplicates) which was cultured in 5% CO2 humidified incubator at 37°C. The numbers of colonies were counted under inverted microscope (Primo Vert, Ziess) by morphologic criteria 8 days after plating.
RNA extraction and quantitative RT-PCR
Total RNA was isolated from bone marrow cells using Trizol (Thermo Fisher Scientific, Waltham, MA). RT—qPCR was performed with one step SYBR Green RT-qPCR Master Mix (Qiagen, Valencia, CA). The mRNA level of each gene was normalized to the GAPDH (glyceraldehyde-3-phosphate dehydrogenase) mRNA level. The primer sequences are listed in S1 Table.
Chromatin immunoprecipitation assays (ChIP)
ChIP assay with bone marrow cells was performed as described previously [51]. Quantitative PCR was performed to detect the upstream fragment in Gata1 genes (primer sequences are listed in S1 Table). The fold of changes in binding was relative to the control of IgG level as 1.
Electrophoretic mobility-shift assay (EMSA)
Oligonucleotide probe containing mouse Gata1 TREs or F2 TRE (positive control) was labeled with [α-32p] dCTP by Klenow fill-in reaction. Assays were performed as described previously [52].
Plasmid constructs, and transient transfection
The Gata1 TRE2 and Gata1 TRE1 luciferase constructs were generated by cloning upstream Gata1 promoter fragments into the pGL4.23 luciferase plasmid. The Gata1- TRE2 luciferase construct was made by insertion of a 1.553 kb XhoI-HindIII fragment representing the sequences between -3.741 kb and -2.189 kb. The Gata1-TRE1 luciferase construct was made by insertion of a 140 bp XhoI-HindIII fragment representing the sequences between +1.385 kb and +1.524 kb. Insert sequence validated by DNA sequencing. K562 cells were transfected with the TRE-luc reporters with Genepulse X cell electrophorators (Biorad). Bone marrow cells (2X106 cells) were transfected with the GATA1-pLenti6/V5 plasmid (3 μg) provided by Dr. GP Rodgers (NHLBI; [53]) using 4D nucleofector (Lonza) in accordance with the manufacturer’s instructions. The Gata1 reporter plasmids cloned in pGL3 basic (5 μg), and the expressing plasmid for TRα1 (pcDNA3.1-TRα1; 10 μg) with or without the expression plasmid for TRα1PV (pcDNA3.1-TRα1PV; 80 μg) were transfected into K562 cells according to Guigon et al [54]. Luciferase activity was measured using Victor 3 (PerkinElmer Life and Analytical Sciences, Waltham, MA). Luciferase values were standardized to the ratio of β-galactosidase activity and protein concentration. The fold of changes in activity was based on using the values of negative control (no plasmid transfected cell without T3) as 1.
In vitro terminal erythropoiesis assay
For lineage depleted bone marrow cell preparation, linage marker positive cells were depleted using the biotin based selection kit (cat# 19856, STEMCELL Technologies, Vancouver, BC) according to the manufacturer's instructions. Lin- BM cells were seeded in fibronectin-coated wells (Corning Inc, Corning, NY). To induce erythropoiesis, Lin- BM cells were cultured as described [24].
Cytology and histochemistry
For May-GrÜnwald Giemsa stain, cytocentrifuged cells were stained with May-GrÜnwald solution (Cat# MG500, Sigma-Aldrich, St. Louis, MO) for 5 minutes and in Giemsa (Cat# GS500, Sigma-Aldrich, St. Louis, MO) for 20 minutes. For whole bone marrow sections, femurs were fixed in 10% (vol/vol) neutral buffered formalin solution (NBF, approximately 4% formaldehyde) (Sigma-Aldrich, St. Louis, MO). The embedded sections were stained with hematoxylin and eosin (HistoServ, Germantown, MD).
Flow cytometry analysis
All antibodies used in flow cytometry were from eBiosciences (Thermo Fisher Scientific, Waltham, MA). The sources of antibodies and fluorophore-labeled antibodies used in FACS analyses are listed in S2 Table. The flow cytometry analyses were performed on a BD LSR II flow cytometer (BD bioscience, San Jose, CA) and analyzed with FloJo, LLC (Tree Star Inc, Ashland, OR).
Western blot analysis and co-immunoprecipitation
The western blot analysis of bone marrow lysates was carried as described previously [50]. To determine the GATA1 tagged with V5 (The V5 tag is derived from a small epitope found on the P and V proteins of the paramyxovirus of simian virus 5 (SV5). after transfection with GATA1-V5 expression plasmid, anti-V5 antibodies (1:2000 dilution; Thermo Fisher Scientific) was used to detect the expressed GATA1-V5. GAPDH (1:4000 dilution; Cell Signaling Technology (Danvers, MA) was used as a loading control.
For the detection of GATA1 proteins in the bone marrow of WT and Thra1PV/+ mice, bone marrow lysates (600 μg each) were first immunoprecipitated with rat anti-GATA1 antibody (4 μg; Santa Crus Biotecholology, Cat.# Sc-265) or mouse IgG (4 μg; negative controls) followed by pulling down the enriched GATA1-anti-GATA1 antibody-complex with protein G-agarose beads. GATA1 proteins were subsequently detected by western blot analysis as described above using rabbit anti-GATA1 antibody (1:1000 dilution; abcam, Cat.# ab28839).
Statistical analysis
All statistical analyses and the graphs were performed and generated using GraphPad Prism version 6.0 (GraphPad Software, La Jolla, CA). P < 0.05 is considered statistically significant. All data are expressed as mean ± SEM.
Supporting information
(DOCX)
(DOCX)
Acknowledgments
We thank Chin-Hsien Tai for mapping the genomic regions of TREs and preparing of Fig 6A for us, Dr. Yangjo Chung for the gift of K562 cells and technical advice on the in vitro erythropoietic culture and cytology of bone marrow cells, and Dr. Kye-yoon Park for valuable discussion on plasmid transfection into bone marrow cells.
We are grateful to Dr. GP Rodgers for the GATA1-pLenti6/V5 plasmid.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This research was supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, National Institutes of Health. SP was supported by a grant from the NIH and the Korean Visiting Scientist Training Award (KVSTA) through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI14C1192). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Krause RL, Sokoloff L. Effects of thyroxine on initiation and completion of protein chains of hemoglobin in vitro. J Biol Chem. 1967;242(7):1431–8. . [PubMed] [Google Scholar]
- 2.Fein HG, Rivlin RS. Anemia in thyroid diseases. Med Clin North Am. 1975;59(5):1133–45. . [DOI] [PubMed] [Google Scholar]
- 3.Erdogan M, Kosenli A, Ganidagli S, Kulaksizoglu M. Characteristics of anemia in subclinical and overt hypothyroid patients. Endocr J. 2012;59(3):213–20. . [DOI] [PubMed] [Google Scholar]
- 4.Bremner AP, Feddema P, Joske DJ, Leedman PJ, O'Leary PC, Olynyk JK, et al. Significant association between thyroid hormones and erythrocyte indices in euthyroid subjects. Clin Endocrinol (Oxf). 2012;76(2):304–11. doi: 10.1111/j.1365-2265.2011.04228.x . [DOI] [PubMed] [Google Scholar]
- 5.Bashir H, Bhat MH, Farooq R, Majid S, Shoib S, Hamid R, et al. Comparison of hematological parameters in untreated and treated subclinical hypothyroidism and primary hypothyroidism patients. Med J Islam Repub Iran. 2012;26(4):172–8. . [PMC free article] [PubMed] [Google Scholar]
- 6.Vitale G, Fatti LM, Prolo S, Girola A, Caraglia M, Marra M, et al. Screening for hypothyroidism in older hospitalized patients with anemia: a new insight into an old disease. J Am Geriatr Soc. 2010;58(9):1825–7. doi: 10.1111/j.1532-5415.2010.03040.x . [DOI] [PubMed] [Google Scholar]
- 7.Christ-Crain M, Meier C, Huber P, Zulewski H, Staub JJ, Muller B. Effect of restoration of euthyroidism on peripheral blood cells and erythropoietin in women with subclinical hypothyroidism. Hormones (Athens). 2003;2(4):237–42. . [DOI] [PubMed] [Google Scholar]
- 8.Bochukova E, Schoenmakers N, Agostini M, Schoenmakers E, Rajanayagam O, Keogh JM, et al. A mutation in the thyroid hormone receptor alpha gene. N Engl J Med. 2012;366(3):243–9. doi: 10.1056/NEJMoa1110296 . [DOI] [PubMed] [Google Scholar]
- 9.van Mullem AA, Chrysis D, Eythimiadou A, Chroni E, Tsatsoulis A, de Rijke YB, et al. Clinical phenotype of a new type of thyroid hormone resistance caused by a mutation of the TRalpha1 receptor: consequences of LT4 treatment. J Clin Endocrinol Metab. 2013;98(7):3029–38. doi: 10.1210/jc.2013-1050 . [DOI] [PubMed] [Google Scholar]
- 10.Moran C, Schoenmakers N, Agostini M, Schoenmakers E, Offiah A, Kydd A, et al. An adult female with resistance to thyroid hormone mediated by defective thyroid hormone receptor alpha. J Clin Endocrinol Metab. 2013;98(11):4254–61. doi: 10.1210/jc.2013-2215 . [DOI] [PubMed] [Google Scholar]
- 11.Tylki-Szymanska A, Acuna-Hidalgo R, Krajewska-Walasek M, Lecka-Ambroziak A, Steehouwer M, Gilissen C, et al. Thyroid hormone resistance syndrome due to mutations in the thyroid hormone receptor alpha gene (THRA). J Med Genet. 2015;52(5):312–6. doi: 10.1136/jmedgenet-2014-102936 . [DOI] [PubMed] [Google Scholar]
- 12.Kaneshige M, Suzuki H, Kaneshige K, Cheng J, Wimbrow H, Barlow C, et al. A targeted dominant negative mutation of the thyroid hormone alpha 1 receptor causes increased mortality, infertility, and dwarfism in mice. Proc Natl Acad Sci U S A. 2001;98(26):15095–100. doi: 10.1073/pnas.261565798 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Parrilla R, Mixson AJ, McPherson JA, McClaskey JH, Weintraub BD. Characterization of seven novel mutations of the c-erbA beta gene in unrelated kindreds with generalized thyroid hormone resistance. Evidence for two "hot spot" regions of the ligand binding domain. J Clin Invest. 1991;88(6):2123–30. doi: 10.1172/JCI115542 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.O'Shea PJ, Bassett JH, Sriskantharajah S, Ying H, Cheng SY, Williams GR. Contrasting skeletal phenotypes in mice with an identical mutation targeted to thyroid hormone receptor alpha1 or beta. Mol Endocrinol. 2005;19(12):3045–59. doi: 10.1210/me.2005-0224 . [DOI] [PubMed] [Google Scholar]
- 15.O'Shea PJ, Bassett JH, Cheng SY, Williams GR. Characterization of skeletal phenotypes of TRalpha1 and TRbeta mutant mice: implications for tissue thyroid status and T3 target gene expression. Nucl Recept Signal. 2006;4:e011 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bassett JH, Boyde A, Zikmund T, Evans H, Croucher PI, Zhu X, et al. Thyroid hormone receptor alpha mutation causes a severe and thyroxine-resistant skeletal dysplasia in female mice. Endocrinology. 2014;155(9):3699–712. doi: 10.1210/en.2013-2156 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Medvinsky A, Rybtsov S, Taoudi S. Embryonic origin of the adult hematopoietic system: advances and questions. Development. 2011;138(6):1017–31. doi: 10.1242/dev.040998 . [DOI] [PubMed] [Google Scholar]
- 18.Akashi K, Traver D, Miyamoto T, Weissman IL. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature. 2000;404(6774):193–7. doi: 10.1038/35004599 . [DOI] [PubMed] [Google Scholar]
- 19.Pronk CJ, Rossi DJ, Mansson R, Attema JL, Norddahl GL, Chan CK, et al. Elucidation of the phenotypic, functional, and molecular topography of a myeloerythroid progenitor cell hierarchy. Cell Stem Cell. 2007;1(4):428–42. doi: 10.1016/j.stem.2007.07.005 . [DOI] [PubMed] [Google Scholar]
- 20.Elliott S, Pham E, Macdougall IC. Erythropoietins: a common mechanism of action. Exp Hematol. 2008;36(12):1573–84. doi: 10.1016/j.exphem.2008.08.003 . [DOI] [PubMed] [Google Scholar]
- 21.Pilon AM, Arcasoy MO, Dressman HK, Vayda SE, Maksimova YD, Sangerman JI, et al. Failure of terminal erythroid differentiation in EKLF-deficient mice is associated with cell cycle perturbation and reduced expression of E2F2. Mol Cell Biol. 2008;28(24):7394–401. doi: 10.1128/MCB.01087-08 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang J, Socolovsky M, Gross AW, Lodish HF. Role of Ras signaling in erythroid differentiation of mouse fetal liver cells: functional analysis by a flow cytometry-based novel culture system. Blood. 2003;102(12):3938–46. doi: 10.1182/blood-2003-05-1479 . [DOI] [PubMed] [Google Scholar]
- 23.Adolfsson J, Borge OJ, Bryder D, Theilgaard-Monch K, Astrand-Grundstrom I, Sitnicka E, et al. Upregulation of Flt3 expression within the bone marrow Lin(-)Sca1(+)c-kit(+) stem cell compartment is accompanied by loss of self-renewal capacity. Immunity. 2001;15(4):659–69. . [DOI] [PubMed] [Google Scholar]
- 24.Shuga J, Zhang J, Samson LD, Lodish HF, Griffith LG. In vitro erythropoiesis from bone marrow-derived progenitors provides a physiological assay for toxic and mutagenic compounds. Proc Natl Acad Sci U S A. 2007;104(21):8737–42. doi: 10.1073/pnas.0701829104 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cantor AB, Orkin SH. Transcriptional regulation of erythropoiesis: an affair involving multiple partners. Oncogene. 2002;21(21):3368–76. doi: 10.1038/sj.onc.1205326 . [DOI] [PubMed] [Google Scholar]
- 26.Hattangadi SM, Wong P, Zhang L, Flygare J, Lodish HF. From stem cell to red cell: regulation of erythropoiesis at multiple levels by multiple proteins, RNAs, and chromatin modifications. Blood. 2011;118(24):6258–68. doi: 10.1182/blood-2011-07-356006 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McDevitt MA, Shivdasani RA, Fujiwara Y, Yang H, Orkin SH. A "knockdown" mutation created by cis-element gene targeting reveals the dependence of erythroid cell maturation on the level of transcription factor GATA-1. Proc Natl Acad Sci U S A. 1997;94(13):6781–5. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fujiwara Y, Browne CP, Cunniff K, Goff SC, Orkin SH. Arrested development of embryonic red cell precursors in mouse embryos lacking transcription factor GATA-1. Proc Natl Acad Sci U S A. 1996;93(22):12355–8. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pevny L, Lin CS, D'Agati V, Simon MC, Orkin SH, Costantini F. Development of hematopoietic cells lacking transcription factor GATA-1. Development. 1995;121(1):163–72. . [DOI] [PubMed] [Google Scholar]
- 30.Pevny L, Simon MC, Robertson E, Klein WH, Tsai SF, D'Agati V, et al. Erythroid differentiation in chimaeric mice blocked by a targeted mutation in the gene for transcription factor GATA-1. Nature. 1991;349(6306):257–60. doi: 10.1038/349257a0 . [DOI] [PubMed] [Google Scholar]
- 31.Gutierrez L, Tsukamoto S, Suzuki M, Yamamoto-Mukai H, Yamamoto M, Philipsen S, et al. Ablation of Gata1 in adult mice results in aplastic crisis, revealing its essential role in steady-state and stress erythropoiesis. Blood. 2008;111(8):4375–85. doi: 10.1182/blood-2007-09-115121 . [DOI] [PubMed] [Google Scholar]
- 32.Yien YY, Bieker JJ. EKLF/KLF1, a tissue-restricted integrator of transcriptional control, chromatin remodeling, and lineage determination. Mol Cell Biol. 2013;33(1):4–13. doi: 10.1128/MCB.01058-12 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Forand A, Beck L, Leroy C, Rousseau A, Boitez V, Cohen I, et al. EKLF-driven PIT1 expression is critical for mouse erythroid maturation in vivo and in vitro. Blood. 2013;121(4):666–78. doi: 10.1182/blood-2012-05-427302 . [DOI] [PubMed] [Google Scholar]
- 34.Koshino I, Mohandas N, Takakuwa Y. Identification of a novel role for dematin in regulating red cell membrane function by modulating spectrin-actin interaction. J Biol Chem. 2012;287(42):35244–50. doi: 10.1074/jbc.M111.305441 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zenke M, Munoz A, Sap J, Vennstrom B, Beug H. v-erbA oncogene activation entails the loss of hormone-dependent regulator activity of c-erbA. Cell. 1990;61(6):1035–49. . [DOI] [PubMed] [Google Scholar]
- 36.Bauer A, Mikulits W, Lagger G, Stengl G, Brosch G, Beug H. The thyroid hormone receptor functions as a ligand-operated developmental switch between proliferation and differentiation of erythroid progenitors. EMBO J. 1998;17(15):4291–303. doi: 10.1093/emboj/17.15.4291 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Disela C, Glineur C, Bugge T, Sap J, Stengl G, Dodgson J, et al. v-erbA overexpression is required to extinguish c-erbA function in erythroid cell differentiation and regulation of the erbA target gene CAII. Genes Dev. 1991;5(11):2033–47. . [DOI] [PubMed] [Google Scholar]
- 38.Ohneda K, Shimizu R, Nishimura S, Muraosa Y, Takahashi S, Engel JD, et al. A minigene containing four discrete cis elements recapitulates GATA-1 gene expression in vivo. Genes Cells. 2002;7(12):1243–54. . [DOI] [PubMed] [Google Scholar]
- 39.Nishimura S, Takahashi S, Kuroha T, Suwabe N, Nagasawa T, Trainor C, et al. A GATA box in the GATA-1 gene hematopoietic enhancer is a critical element in the network of GATA factors and sites that regulate this gene. Mol Cell Biol. 2000;20(2):713–23. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yamamoto Y, Shiraishi M, Fujita M, Kojima I, Tanaka Y, Tachibana S. Triiodothyronine suppresses activin-induced differentiation of erythroleukemia K562 cells under hypoxic conditions. Molecular and Cellular Biochemistry. 2014;391(1–2):217–24. doi: 10.1007/s11010-014-2005-7 [DOI] [PubMed] [Google Scholar]
- 41.Kaneshige M, Kaneshige K, Zhu X, Dace A, Garrett L, Carter TA, et al. Mice with a targeted mutation in the thyroid hormone beta receptor gene exhibit impaired growth and resistance to thyroid hormone. Proc Natl Acad Sci U S A. 2000;97(24):13209–14. doi: 10.1073/pnas.230285997 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kendrick TS, Payne CJ, Epis MR, Schneider JR, Leedman PJ, Klinken SP, et al. Erythroid defects in TRalpha-/- mice. Blood. 2008;111(6):3245–8. doi: 10.1182/blood-2007-07-101105 . [DOI] [PubMed] [Google Scholar]
- 43.Das KC, Mukherjee M, Sarkar TK, Dash RJ, Rastogi GK. Erythropoiesis and erythropoietin in hypo- and hyperthyroidism. J Clin Endocrinol Metab. 1975;40(2):211–20. doi: 10.1210/jcem-40-2-211 . [DOI] [PubMed] [Google Scholar]
- 44.Liu X, Liu J, Fan L, Shi B. Erythrocytosis associated with hyperthyroidism: a rare case report and clinical study of possible mechanism. Endocr Res. 2015;40(4):177–80. doi: 10.3109/07435800.2014.952017 . [DOI] [PubMed] [Google Scholar]
- 45.Schindhelm RK, Ten Boekel E, Heima NE, van Schoor NM, Simsek S. Thyroid hormones and erythropoiesis: a complex relation? Eur J Intern Med. 2014;25(1):e6 doi: 10.1016/j.ejim.2013.03.018 . [DOI] [PubMed] [Google Scholar]
- 46.Bremner AP, Feddema P, Leedman PJ, Brown SJ, Beilby JP, Lim EM, et al. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012;97(5):1554–62. doi: 10.1210/jc.2011-3020 . [DOI] [PubMed] [Google Scholar]
- 47.Graf T, Beug H. Role of the v-erbA and v-erbB oncogenes of avian erythroblastosis virus in erythroid cell transformation. Cell. 1983;34(1):7–9. . [DOI] [PubMed] [Google Scholar]
- 48.Frykberg L, Palmieri S, Beug H, Graf T, Hayman MJ, Vennstrom B. Transforming capacities of avian erythroblastosis virus mutants deleted in the erbA or erbB oncogenes. Cell. 1983;32(1):227–38. . [DOI] [PubMed] [Google Scholar]
- 49.Trainor CD, Evans T, Felsenfeld G. Negative regulation of chicken GATA-1 promoter activity mediated by a hormone response element. Mol Endocrinol. 1995;9(9):1135–46. doi: 10.1210/mend.9.9.7491106 . [DOI] [PubMed] [Google Scholar]
- 50.Furumoto H, Ying H, Chandramouli GV, Zhao L, Walker RL, Meltzer PS, et al. An unliganded thyroid hormone beta receptor activates the cyclin D1/cyclin-dependent kinase/retinoblastoma/E2F pathway and induces pituitary tumorigenesis. Mol Cell Biol. 2005;25(1):124–35. doi: 10.1128/MCB.25.1.124-135.2005 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fozzatti L, Park JW, Zhao L, Willingham MC, Cheng SY. Oncogenic Actions of the Nuclear Receptor Corepressor (NCOR1) in a Mouse Model of Thyroid Cancer. PLoS One. 2013;8(6):e67954 doi: 10.1371/journal.pone.0067954 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ying H, Suzuki H, Zhao L, Willingham MC, Meltzer P, Cheng SY. Mutant thyroid hormone receptor beta represses the expression and transcriptional activity of peroxisome proliferator-activated receptor gamma during thyroid carcinogenesis. Cancer Res. 2003;63(17):5274–80. . [PubMed] [Google Scholar]
- 53.Zhu J, Chin K, Aerbajinai W, Trainor C, Gao P, Rodgers GP. Recombinant erythroid Kruppel-like factor fused to GATA1 up-regulates delta- and gamma-globin expression in erythroid cells. Blood. 2011;117(11):3045–52. doi: 10.1182/blood-2010-07-294751 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Guigon CJ, Kim DW, Zhu X, Zhao L, Cheng SY. Tumor suppressor action of liganded thyroid hormone receptor beta by direct repression of beta-catenin gene expression. Endocrinology. 2010;151(11):5528–36. doi: 10.1210/en.2010-0475 . [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.