Abstract
Scleroderma is a complex connective tissue disease characterized by fibrosis, vasculopathy, and immune system dysfunction. The heterogeneity of disease presentation and poorly understood etiology has made the management of scleroderma difficult. The available treatment options like immunosuppressive agents are associated with potentially hazardous side effects and physiotherapy, which to a certain degree helps to minimize the loss of function in digits and limbs, has only limited success. Also, studies investigating antifibrotic therapies have failed to report any significant improvement. Hence, there is currently no effective therapy for scleroderma. Recently, phototherapy has been extensively studied and found to be effective in treating scleroderma. Initially psoralen + ultraviolet A (PUVA) significantly enriched the therapeutic panel, but more recently ultraviolet A1 (UVA1) is seen to replace PUVA therapy. This might be because of UVA1 therapy being free of side effects seen with psoralens such as nausea, vomiting or photokeratitis. In addition, UVA1 is seen to lower risk of phototoxic reactions with deeper penetration of radiation. The present review will put some light on the use of UVA1 for treating cutaneous lesion in scleroderma and we aim to find the most benefitted group of patients and most effective dose of UVA1 for different types of scleroderma.
Keywords: Ultraviolet A1, phototherapy, localized scleroderma, systemic sclerosis
Introduction
Scleroderma is a chronic connective tissue disease characterized by production and deposition of large amount of collagen leading to fibrosis as well as vascular alterations. Two types of scleroderma exist: localized scleroderma (LS) (also called circumscribed scleroderma or morphea), which involves only skin and systemic scleroderma, which involves skin and internal organs. LS is further classified into plaque, linear, deep (en coup de sabre), and generalized forms. Similarly, systemic scleroderma includes CREST syndrome and progressive systemic sclerosis. The skin lesions in all types of scleroderma are histologically identical [1]. The pathogenesis of disease involves injury to endothelial cells leading to their apoptosis and a lowered vascular density, which are associated with inflammatory cells infiltration [2]. The currently accepted therapy includes drugs like corticosteroids, penicillamine, vitamin D derivatives, tacrolimus and methotrexate all with unproven efficacy and some with undesirable side effects. In more severe cases, combination therapy such as corticosteroids with MTX has been used but has not been established as consistently effective [3]. Hence, there is currently no effective therapy for scleroderma. This necessitates the need to develop new treatment modality that would be universally applicable and free of adverse effects.
During the last few decades, UVA1 has emerged as a phototherapeutic modality for treating cutaneous lesions in scleroderma and it is evolving as a new phototherapeutic modality with distinct modes of action. Many studies are being conducted in a hope to establish safety, efficacy and doses profile. Plewig and colleagues [4] and Muthzas and associates [5] were the first to report on a new device equipped with a super pressure mercury lamp that delivered selective high-dose radiation energy in the longer wavelength region of the ultraviolet A (UVA) spectrum, i.e. UVA1 (340-400 nm). The UVA1 device developed by these investigators was predominantly used for diagnostic purposes, such as photoprovocation tests of photodermatoses and patch testing [4,5]. Soon UVA1 was recognized as a beneficial phototherapeutic modality for some skin conditions including scleroderma. The devices used for therapeutic purpose were the fluorescent lamps that permit low dose to medium dose UVA1 phototherapy and high-output metal halide bulbs that permit high dose UVA1 phototherapy [6]. There are no internationally agreed definitions of different treatment doses but < 10 J/cm2 is generally considered very low dose, 10-29 J/cm2 low dose (LD), 30-59 J/cm2 medium dose (MD) and > 60 J/cm2 high dose (HD) [7]. UVA1 phototherapy is a method affecting various stages of the sclerodermal process. The therapy inhibits the inflammatory process, thus preventing the progression of disease as well as altering fibrosis, an effect of the disease. Acute adverse effects of UVA1 are minimal. Skin pigmentation (tanning), which can persist for months, is the most common problem. Uncommon acute adverse effects include reactivation of herpes simplex, cholinergic urticaria, and transient and reversible changes in the appearance of moles [7]. The risk of long-term adverse effects, particularly skin cancer, is unknown. This might be because studies of chronic UVA1 effects in humans are limited. However, information on UVA1 from animal models have shown that it can induce squamous cell carcinomas and melanomas [7]. The studies done on UVA1 for various sclerodermal skin lesions are discussed below:
Localized scleroderma (or morphea)
In 1995, since Kerscher et al [8] for the first time reported successful use of UVA1 to treat LS, different doses of UVA1 (low, medium and high) in different types of morphea were studied, most of them achieving a successful result (Table 1). Kerscher et al [8] treated ten consecutive patients with severe LS with low dose UVA1 phototherapy. After 24 irradiations, more than 80% of the lesions completely cleared and no side effects were observed. In 1998, Kerscher et al reported another study [9] of twenty consecutive patients with plaque, linear or subcutaneous (SC) morphea. All the patients received 30 treatments with low dose UVA1 phototherapy and clearance of > 80% of lesions were seen in 18 of 20 patients. 2 patients with SC morphea did not respond to the treatment. In 2001, Camacho et al [10] reported a study of 7 patients with plaque or linear type morphea treated with medium dose UVA1 phototherapy. All patients showed significant decrease in induration of the skin. In addition, 1 patient with decrease mobility of elbow joint restored the mobility and function of joint to almost normal levels. Yet another study in the same year by Gruss et al [11] reported the successful use of UVA-1 on different clinical stages of severe plaque type morphea. 3 patients with late-stage lesions, active inflammatory lesions and associated overlying lichen sclerosusetatrophicus (LSA) respectively were treated with low dose UVA1 phototherapy. Following therapy, the sclerotic plaques resolved, leaving smooth and soft tanned skin with normal structure, consistency and folding capability. In 2003, de Rie et al [12] performed a controlled pilot study with medium-dose UVA1 on 8 patients with LS. All patients responded to therapy and treated lesional skin showed a significantly increased elasticity at weeks 3, 5 and 7 when compared to control skin. In 2006, kreuter et al reported 2 studies, one with low dose [13] and another with medium dose [14] UVA1 phototherapy on 14 and 8 patients respectively. The patients were classified to have either plaque or generalized morphea. All the patients received 40 irradiations and showed marked improvement in skin status resulting in significant reduction of the clinical score from baseline to the end of the treatment. In 2011, Su et al [15] confirmed the effectiveness of UVA1 with comparatively large sample sized study recruiting 35 patients with different type morphea at different clinical stages. In all patients, medium-dose UVA1 therapy softened sclerotic plaques, and marked clinical improvement was observed in 29 of 35 patients.
Table 1.
UVA1 phototherapy for Localized scleroderma
| Scleroderma type | No. of patients | Treatment regimen | Dose per treatment (J/cm2) | No. of irradiations | Outcome | Referemce/first author |
|---|---|---|---|---|---|---|
| Severe LS | 10 | 4 s/w for 6 w | 20 | 24 | In all patients sclerosis regressed greatly after fewer than 15 treatment sessions. After 24 treatments, more than 80% of the lesions were completely cleared. | [8]/Martina Kerscher |
| Plaque, linear or deep morphea | 20 | 4 s/w for 6 w, further 1 s/w for 6 w | 20 | 30 | After 24 treatments,clearance of > 80% of lesions in 18 of 20 patients. 2 patients with SC morphea did not improve. | [9]/Martina Kerscher |
| Plaque or linear morphea | 7 | 3 s/w for 10 w | 30 | 30 | All patients showed significant decrease in induration of skin. In 1 patient, reduced joint mobility of elbow was restored to almost normal levels. | [10] Nydia R. Camacho |
| Severe plaque type morphea in different stages, 1 with overlying LSA | 3 | 4 s/w for 6 w, further 1 s/w for 6 w | 20 | 30 | After less than 15 sessions, clinical palpation for tethering or thickening of the skin revealed remarkable softening of sclerotic lesions. In morphea with overlying LSA the elastic fibers did not completely return to the superficial papillary dermis despite the clinical clearance. | [11]/Claus J. Gruss |
| LS | 8 | 4 s/w for 5 w | 48 | 20 | Treated skin showed a significantly increased elasticity at weeks 3, 5 and 7 when compared to control skin. No patients showed complete regression of sclerotic plaques. | [12]/M.A. de Rie |
| Plaque or generalized type LS | 14 | 5 s/w for 8 w | 20 | 40 | Skin status markedly improved in all 14 patients, resulting in a significant reduction of clinical score from baseline to the end of treatment. | [13]/A. Kreuter |
| Plaque or generalized type LS | 8 | 5 s/w for 8 w | 50 | 40 | Skin status markedly improved in all patients, resulting in a significant reduction of clinical score from baseline to the end of treatment. | [14]/Alexander Kreuter |
| Plaque, linear, generalized, deep or pansclerotic morphea. Generalized associated with LSA | 35 | 3-5 s/w for 10-15 w | 30 | 30-45 | In all patients, sclerotic plaques were softened and marked clinical improvement was observed in 29 of 35 patients. | [15]/Ozlem Su |
LS: localosed scleroderma, LSA: lichen sclerosusatrophicus, s/w: session per week, w: week.
In addition to studies mentioned above, there are few other studies [16-18] that compared the effect of different doses of UVA1, also compared its effect with narrow band UVB (NBUVB). Stege et al [16] performed a study on HD (n=10) and LD (n=7) UVA1 and found that HD UVA1 was superior to LD UVA1 therapy. All patients undergoing high-dose UVA1 therapy reported softening of sclerotic plaques, and complete clearance was observed in 4 of 10 patients. Whereas, 5 of 7 patients undergoing low-dose UVA1 treatment reported lack of reduction or softening of sclerotic lesions, and none reported complete clearance of skin lesions. Sator et al [17] sought to compare the immediate and long-term efficacy of low-versus medium-dose UVA1 phototherapy for plaque-type LS and found that medium-dose provides for better long-term results than low-dose UVA1 in LS. Kreuter et al [18] reported a prospective, open, randomized controlled 3 arm study that recruited 64 patients to compare the safety and efficacy of LD UVA1, MD UVA1, and NBUVB phototherapy in the treatment of different types LS. The study concluded that UVA1 phototherapy should be considered among the first approaches in the management of LS. Also reported that MD UVA1 was significantly more effective than NBUVB.
Andres et al [19] also in their retrospective and prospective study proved the efficacy of MD UVA1 in LS. This open study showed a positive short- and long-term efficacy of UVA1 with a reduction in sclerotic plaques, an increase in skin elasticity and a reduction of lesional skin thickness.
Systemic sclerosis (SS)
The studies demonstrating successful use of UVA1 in treating LS has more recently drawn its attention towards systemic sclerosis. Though very few, the studies have reported exciting result of UVA1 in treating acrosclerosis, limited and diffuse type systemic sclerosis and microstomia (Table 2). Two subsequent reports of successful use of UVA1 to treat acrosclerosis in patients with SS were published in the year 2004 [20,21]. Kreuter et al [20] reported an open, nonrandomized study of 18 patients receiving low-dose UVA1 phototherapy. All patients had SS associated with diffuse cutaneous sclerosis, pronounced acral edema and sclerosis, and 4 patients even had piecemeal necrosis. Breuckmann et al [21] reported a study of 4 patients exhibiting SS associated with severe diffuse acral cutaneous sclerosis. The patients were treated with low-dose UVA1 phototherapy of the hands. In exception to these successful results, one study in 2007 [22] reported the in efficacy of UVA1 for acrosclerosis. Draund et al [22] performed a randomized, investigator-blinded, controlled study on 9 patients to determine whether UVA1 phototherapy is effective for acrosclerosis in SS. The results suggested that UVA1 phototherapy does not improve cutaneous thickness in acrosclerosis.
Table 2.
UVA1 phototherapy for systemic sclerosis
| Skin lesion in SS | No. of patients | Treatment regimen | Dose per treatment (J/cm2) | No. of irradiations | Outcome | Referemce/first author |
|---|---|---|---|---|---|---|
| Diffuse cutaneous sclerosis, pronounced acral edema and sclerosis, 4 patients even had piecemeal necrosis | 18 | 4 s/w for 8 w, further 3 s/w for 6 w | 30 | 50 | 16 patients showed marked softening of affected skin, an improvement of finger mobility and 4 patients with piecemeal necrosis showed almost complete healing. Only 2 patients showed no or just a slight improvement. | [20]/Alexander Kreuter |
| Severe diffuse acral cutaneous sclerosis | 4 | 4 s/w for 8 w, further 3 s/w for 6 w | 30 | 50 | UVA1 irradiation exerted its positive effects by a modulation of endothelial regulation/transformation. | [21]/Frank Breuckmann |
| Acrosclerosis | 9 | 3 s/w for 14 w | 40 | 42 | 2 patients improved functions of treated hand but no modification of the index flexion or extension was seen. Cutaneous thickness was not improved. | [22]/F. Durand |
| Diffuse and limited type SS | 4 | 5 times/week from mon to fri | 60 | 9-29 | In all patients, treated skin lesions were markedly softened after 9 to 29 exposures. Clinical improvement was associated with increase in joint passive range of motion, skin temperature and cutaneous elasticity. | [23]/Akimichi Morita |
| Diffuse and limited type SS with involvement of proximal and acral sites | 8 | 3 s/w | 30-40 | 26-32 | In all patients skin lesions improved, demonstrated by a fall in the modified Rodnan skin score. | [24]/Rebecca F. Rose |
| Sclerodactyly and microstomia | 1 | 2-3 s/w | 50 | 40 | After 10 treatments all sclerotic areas of skin including perioral area were less indurated. After 21 treatments she could articulate words normally and there was reduced furrowing around mouth. After 40 treatments, she could open her mouth more normally. | [25]/Angela Tewari |
s/w: session per week, w: week, mon: Monday, fri: Friday.
Morita et al [23] reported positive effect of UVA1 phototherapy in limited and diffuse type SS. 4 patients with SS (1 limited type, 3 diffuse type) were treated with MD UVA1 and treated skin lesions were markedly softened after 9 to 29 exposures in all the patients. In 2009, Rebecca et al [24] for the first time reported the use of UVA1 for proximal skin lesions in patients with SS. 8 patients with scleroderma affecting acral and proximal sites were retrospectively studied. Skin lesions improved in all patients following LD UVA1 therapy. In 2011, Tiwari et al [25] for the first time reported a case of successful treatment of microstomia with UVA1 phototherapy in a patient with SS. Till date; this is the only study reporting the use of UVA1 for microstomia.
Conclusion
Based on the data retrieved from literature, UVA1 exposure seems to be effective treatment modality for treating sclerodermal skin lesions. But treatment regimen and doses vary from study to study, making it difficult, at this stage to tell the most effective dose. Even, the dose range for low, medium and high dose is not consistent. According to the British Photodermatology group workshop report published in the year 2011, 10-29 J/cm2 is low dose and 30-59 J/cm2 is medium dose [7]. Whereas, some studies are found to be consistent with this doses regimen, others are not. Kreuter et al [20] and Breuckmann et al [21] used UVA1 in the dose of 30 J/cm2, and Durand et al [22] used 40 J/cm2 and said it a low dose, which according to the British Photodermatology group workshop report is a medium dose. Similarly, Morita et al [23], in their study mentioned 60 J/cm2 as medium dose, which is considered as high dose by others. This points towards the necessity of establishment of standard protocol for UVA1 doses. Stege et al [16] compared LD (20 J/cm2) with HD (130 J/cm2) for LS and found that HD UVA1 was superior to LD UVA1 therapy. Similarly, Sator et al [17] compared LD (20 J/cm2) with MD (70 J/cm2) UVA1 phototherapy for plaque-type LS and found that MD provides for better long-term results than LD UVA1 in LS. According to these two studies [16,17], LD seems comparatively less effective than MD or HD. Now, studies comparing MD with HD is needed to determine the most effective dose for LS.
Studies demonstrating the action mechanism of UVA1 for scleroderma have presented its distinct mode of action (Figure 1). Immunohistochemical test for matrix-metalloproteinase-1 showed that even low-dose UVA1 irradiation may suppress collagen synthesis, initiate collagenase activity, and cause a reduction of the cellular infiltrate resulting in clinically observed softening of former sclerotic skin lesions [20]. Kreuter et al [14] proposed that UVA1 phototherapy decreases inhibitory SMAD7 gene expression in LS. Kreuter et al [13] also proposed that UVA1 induces downregulation of humanβ-defensins and interleukin-6 and interleukin-8. Camacho et al [10] proposed that the effectiveness of UVA1 phototherpy is associated with an increase in the number of CD34 positive dendritic cells in the dermis. Sawada et al [26] suggested that decreased and normalized levels of accumulated decorin might relate to the efficacy of sclerotic lesions in UVA1 phototherapy. Breuckmann et al [21] suggested that UVA1 induces concomitant modulation of endothelial dysfunction and apoptosis, which may substantially contribute to an improvement in acrosclerosis. Breuckmann et al [27] demonstrated UVA1-induced reduction in dermal neuron-specific enolase levels correlating with a softening of sclerotic lesions in acrosclerosis. These limited small studies proposing distinct mode of action warrants future studies on a larger group of patients to confirm their preliminary results and to elucidate in more detail their pathogenetic role in sclerodermal skin lesions. Moreover, the pathogenetic relevance of the levels of SMAD7 gene, humanβ-defensins, interleukin-6, interleukin-8, CD34 positive dendritic cells, decorin, and neuron-specific enolase with respect to clinical improvement needs further investigation.
Figure 1.
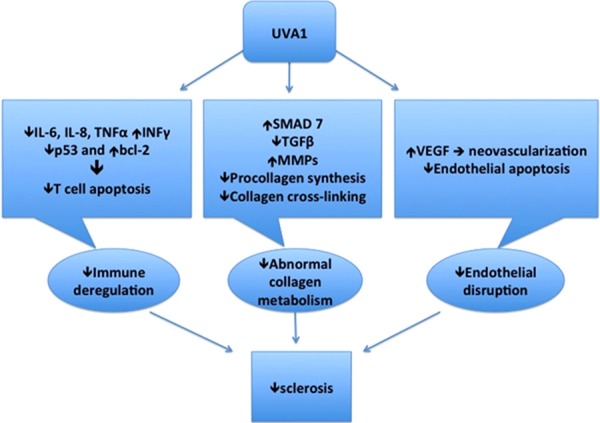
Distinct mode of action of UVA1 for scleroderma.
Pereira et al [28] compared the effect of UVA1 in various subgroups (including plaque-type morphea, linear morphea, generalized morphea, deep morphea, pansclerotic morphea of children, and systemic scleroderma) and concluded that plaque-type morphea obtained the best result. This study however, cannot set a standard protocol because patients in different subgroups received different treatment regimen and dose of UVA1. Other studies too, that used same dose of UVA1 for different subgroups found comparatively better result in plaque type morphea [9,15]. In contrast to this, poor response was seen in deep morphea. 2 out of 2 patients with deep morphea showed no response [9] and 1 out of 2 showed fair to poor response [15] to UVA1 phototherapy. In one study [22], all the 9 patients with acrosclerosis showed no improvement with UVA1. With these results, again it would be unfair to conclude that plaque type is the best responder to UVA1 therapy because plaque type, being the most common type, is most frequently studied, comparing to rare types. Also, it cannot be judged at this moment, which skin type patient would be most benefitted because very few studies mentioned the skin type of patients enrolled. Studies that reported the skin type include Fitzpatrick skin type II or III [13,14,17,18,24] and Fitzpatrick skin type II, III or IV [15]. So studies in future need to report skin type of patients and studies on Fitzpatrick skin type I, IV and V are highly desirable.
There is always a question about safety, is UVA1 phototherapy safe, is it free of side effects? Like other phototherapy, UVA1 is also seen to have some adverse effects. However, serious systemic effects are not yet reported. Except for mild tanning or skin pigmentation no acute or subacute side effects were observed. Only one patient complained of occasional headaches during therapies [10] and one patient reported pruritus at the beginning of therapy [15]. Even the study that used maximum UVA1 dose (130 J/cm2 30 times) did not report any serious side effects, only one patient felt uncomfortable during the last 10 minutes of high-dose UVA1 exposure because of the associated heat load [16].
In conclusion, UVA1 phototherapy seems to offer a promising approach to treatment for LS and skin involvement in systemic sclerosis. However, the number of studies or case series so far is low, partially due to the rarity of disease and perhaps also due to accessibility restricted to specialized centers.
So, multicenter studies recruiting large number of patients with similar symptoms and stages of the disease are required to more accurately define treatment schedules. On the basis of published studies in scleroderma so far, it is clear that UVA1 could stop or inhibit the fibrotic processes in scleroderma. These reports indicate that phototherapy could be an effective therapeutic option for treatment of sclerotic skin in patients with Scleroderma.
Disclosure of conflict of interest
None.
References
- 1.Agnieszka OM, Aleksandra DP, Karoline OH, Wojciech S. Role of UVA1 phototherapy in treatment of scleroderma and sclerodermalike disorders. Centr Eur J Immunol. 2012;37:391–398. [Google Scholar]
- 2.Katsumoto TR, Whitfield ML, Connolly MK. “The pathogenesis of systemic sclerosis”. Annual Review of Pathology. 2011;6:509–37. doi: 10.1146/annurev-pathol-011110-130312. [DOI] [PubMed] [Google Scholar]
- 3.Badea I, Taylor M, Rosenberg A, Foldvari M. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology. 2009;48:213–221. doi: 10.1093/rheumatology/ken405. [DOI] [PubMed] [Google Scholar]
- 4.Plewig G, Hofmann C, Braun-Falco O, Nath G, Kreitmair A. A new apparatus for the delivery of high intensity UVA and UVA+UVB irradiation, and some dermatological applications. Br J Dermatol. 1978;98:15–24. doi: 10.1111/j.1365-2133.1978.tb07328.x. [DOI] [PubMed] [Google Scholar]
- 5.Mutzhas MF, Holzle E, Hofmann C, Plewig G. A new apparatus with high radiation energy between 320-460 nm: physical description and dermatological applications. J Invest Dermatol. 1981;76:42–47. doi: 10.1111/1523-1747.ep12524813. [DOI] [PubMed] [Google Scholar]
- 6.Gambichler T, Terras S, Kreuter A. Treatment regimens, protocols, dosage, and indications for UVA1 phototherapy: facts and controversies. Clin Dermatol. 2013;31:438–454. doi: 10.1016/j.clindermatol.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 7.Kerr AC, Ferguson J, Attili SK, Beattie PE, Coleman AJ, Dawe RS, Eberlein B, Goulden V, Ibbotson SH, Menage Hdu P, Moseley H, Novakovic L, Walker SL, Woods JA, Young AR, Sarkany RP. Ultraviolet A1 phototherapy: a British Photodermatology Group workshop report. Clin Exp Dermatol. 2012;37:219–226. doi: 10.1111/j.1365-2230.2011.04256.x. [DOI] [PubMed] [Google Scholar]
- 8.Kerscher M, Dirschka T, Volkenandt M. Treatment of localized scleroderma by UVA1 phototherapy. Lancet. 1995;346:1166. doi: 10.1016/s0140-6736(95)91843-4. [DOI] [PubMed] [Google Scholar]
- 9.Kerscher M, Volkenandt M, Gruss C, Reuther T, von Kobyletzki G, Freitag M, Dirschka T, Altmeyer P. Low-dose UVA phototherapy for treatment of localized scleroderma. J Am Acad Dermatol. 1998;38:21–26. doi: 10.1016/s0190-9622(98)70533-x. [DOI] [PubMed] [Google Scholar]
- 10.Camacho NR, Sánchez JE, Martin RF, González JR, Sánchez JL. Medium-dose UVA1 phototherapy in localized scleroderma and its effect in CD34-positive dendritic cells. J Am Acad Dermatol. 2001;45:697–699. doi: 10.1067/mjd.2001.117735. [DOI] [PubMed] [Google Scholar]
- 11.Gruss CJ, Von Kobyletzki G, Behrens-Williams SC, Lininger J, Reuther T, Kerscher M, Altmeyer P. Effects of low dose ultraviolet A-1 phototherapy on morphea. Photodermatol Photoimmunol Photomed. 2001;17:149–155. doi: 10.1034/j.1600-0781.2001.170401.x. [DOI] [PubMed] [Google Scholar]
- 12.de Rie MA, Enomoto DN, de Vries HJ, Bos JD. Evaluation of medium-dose UVA1 phototherapy in localized scleroderma with the cutometer and fast Fourier transform method. Dermatology. 2003;207:298–301. doi: 10.1159/000073093. [DOI] [PubMed] [Google Scholar]
- 13.Kreuter A, Hyun J, Skrygan M, Sommer A, Bastian A, Altmeyer P, Gambichler T. Ultraviolet A1-induced downregulation of human beta-defensins and interleukin-6 and interleukin-8 correlates with clinical improvement in localized scleroderma. Br J Dermatol. 2006;155:600–607. doi: 10.1111/j.1365-2133.2006.07391.x. [DOI] [PubMed] [Google Scholar]
- 14.Kreuter A, Hyun J, Skrygan M, Sommer A, Tomi NS, Breuckmann F, Altmeyer P, Gambichler T. Ultraviolet A1 phototherapy decreases inhibitory SMAD7 gene expression in localized scleroderma. Arch Dermatol Res. 2006;298:265–272. doi: 10.1007/s00403-006-0695-8. [DOI] [PubMed] [Google Scholar]
- 15.Su O, Onsun N, Onay HK, Erdemoglu Y, Ozkaya DB, Cebeci F, Somay A. Effectiveness of medium-dose ultraviolet A1 phototherapy in localized scleroderma. Int J Dermatol. 2011;50:1006–1013. doi: 10.1111/j.1365-4632.2010.04843.x. [DOI] [PubMed] [Google Scholar]
- 16.Stege H, Berneburg M, Humke S, Klammer M, Grewe M, Grether-Beck S, Boedeker R, Diepgen T, Dierks K, Goerz G, Ruzicka T, Krutmann J. High-dose UVA1 radiation therapy for localized scleroderma. J Am AcadDermatol. 1997;36:938–944. doi: 10.1016/s0190-9622(97)80277-0. [DOI] [PubMed] [Google Scholar]
- 17.Sator PG, Radakovic S, Schulmeister K, Hönigsmann H, Tanew A. Medium-dose is more effective than low-dose ultraviolet A1 phototherapy for localized scleroderma as shown by 20-MHz ultrasound assessment. J Am Acad Dermatol. 2009;60:786–791. doi: 10.1016/j.jaad.2008.12.013. [DOI] [PubMed] [Google Scholar]
- 18.Kreuter A, Hyun J, Stücker M, Sommer A, Altmeyer P, Gambichler T. A randomized controlled study of low-dose UVA1, medium-dose UVA1, and narrowband UVB phototherapy in the treatment of localized scleroderma. J Am Acad Dermatol. 2006;54:440–447. doi: 10.1016/j.jaad.2005.11.1063. [DOI] [PubMed] [Google Scholar]
- 19.Andres C, Kollmar A, Mempel M, Hein R, Ring J, Eberlein B. Successful ultraviolet A1 phototherapy in the treatment of localized scleroderma: a retrospective and prospective study. Br J Dermatol. 2010;162:445–447. doi: 10.1111/j.1365-2133.2009.09438.x. [DOI] [PubMed] [Google Scholar]
- 20.Kreuter A, Breuckmann F, Uhle A, Brockmeyer N, Von Kobyletzki G, Freitag M, Stuecker M, Hoffmann K, Gambichler T, Altmeyer P. Lowdose UVA1 phototherapy in systemic sclerosis: effects on acrosclerosis. J Am Acad Dermatol. 2004;50:740–747. doi: 10.1016/j.jaad.2003.08.026. [DOI] [PubMed] [Google Scholar]
- 21.Breuckmann F, Stuecker M, Altmeyer P, Kreuter A. Modulation of endothelial dysfunction and apoptosis: UVA1-mediated skin improvement in systemic sclerosis. Arch Dermatol Res. 2004;296:235–239. doi: 10.1007/s00403-004-0513-0. [DOI] [PubMed] [Google Scholar]
- 22.Durand F, Staumont D, Bonnevalle A, Hachulla E, Hatron PY, Thomas P. Ultraviolet A1 phototherapy for treatment of acrosclerosis in systemic sclerosis: controlled study with half-side comparison analysis. Photodermatol Photoimmunol Photomed. 2007;23:215–221. doi: 10.1111/j.1600-0781.2007.00308.x. [DOI] [PubMed] [Google Scholar]
- 23.Morita A, Kobayashi K, Isomura I, Tsuji T, Krutmann J. Ultraviolet A1 (340-400 nm) phototherapy for scleroderma in systemic sclerosis. J Am Acad Dermatol. 2000;43:670–674. doi: 10.1067/mjd.2000.105165. [DOI] [PubMed] [Google Scholar]
- 24.Rose RF, Turner D, Goodfield MJ, Goulden V. Low-dose UVA1 phototherapy for proximal and acral scleroderma in systemic sclerosis. Photodermatol Photoimmunol Photomed. 2009;25:153–155. doi: 10.1111/j.1600-0781.2009.00422.x. [DOI] [PubMed] [Google Scholar]
- 25.Tewari A, Garibaldinos T, Lai-Cheong J, Groves R, Sarkany R, Branislav Novakovic L. Successful treatment of microstomia with UVA1 phototherapy in systemic sclerosis. Photodermatol Photoimmunol Photomed. 2011;27:113–114. doi: 10.1111/j.1600-0781.2011.00570.x. [DOI] [PubMed] [Google Scholar]
- 26.Sawada H, Isogai Z, Morita A. Altered decorin expression of systemic sclerosis by UVA1 (340-400 nm) phototherapy: immunohistochemical analysis of 3 cases. BMC Dermatol. 2003;3:2. doi: 10.1186/1471-5945-3-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Breuckmann F, Appelhans C, Bastian A, Stuecker M, Altmeyer P, Kreuter A. UVA1-induced decrease in dermal neuron-specific enolase (NSE) in acrosclerosis. Arch Dermatol Res. 2004;296:182–184. doi: 10.1007/s00403-004-0495-y. [DOI] [PubMed] [Google Scholar]
- 28.Pereira N, Santiago F, Oliveira H, Figueiredo A. Low-dose UVA1 phototherapy for scleroderma: what benefit can we expect? J Eur Acad Dermatol Venereol. 2012;26:619–626. doi: 10.1111/j.1468-3083.2011.04137.x. [DOI] [PubMed] [Google Scholar]


