Abstract
Background
Interferon-beta (IFN-beta) is one of the most widely prescribed medications for relapsing-remitting multiple sclerosis (RRMS). IFN-related thrombotic microangiopathy (TMA) is a rare but severe complication, with a fulminant clinical onset and a possibly life-threatening outcome that may occur years after a well-tolerated treatment with IFN. Most patients evolve rapidly to advanced chronic kidney disease and eventually to renal failure.
Methods
We performed a retrospective analysis of TMA cases diagnosed and managed in our Nephrology Department from 2010 to 2015, and performed a literature review of IFN-beta-induced TMA.
Results
Three cases of TMA among patients treated with IFN-beta were identified who did not show any renal improvement following conventional therapy: IFN withdrawal and plasma exchange (PE, range 8–18) sessions. All of them responded favourably to eculizumab, with progressive clinical and renal improvement, allowing dialysis discontinuation, without recurrence of TMA during a long-term follow-up (range 1–5 years).
Conclusions
TMA is a recognized severe complication in RRMS patients treated with IFN-beta. Withdrawal of IFN and treatment with PE, steroids or rituximab did not improve the poor renal prognosis in our three patients and in all the previously described cases in the literature. In our experience, eculizumab had a strikingly favourable effect on renal recovery, suggesting a role of IFN-beta as a trigger in complement-mediated TMA. Neurologists and nephrologists should be vigilant to this complication to prevent possibly irreversible renal damage.
Keywords: eculizumab, interferon-beta, malignant hypertension, multiple sclerosis, thrombotic microangiopathy
Introduction
Interferon-beta (IFN-beta) is a first-line treatment for relapsing-remitting multiple sclerosis (RRMS). Multiple sclerosis (MS) is the most frequent inflammatory demyelinating disease of the central nervous system, usually affecting young adults, most frequently females (F:M ratio 2–3:1). The aetiology of the disease remains unknown, whereas the pathogenesis is most probably autoimmune. Despite the well-known efficacy and long-term tolerability of IFN-beta, a growing number of reports have recently associated long-term IFN-beta therapy with the development of systemic adverse effects.
Common side effects of IFN-based therapy are acute (influenza-like symptoms, fever, chills, myalgias, arthralgias, headache), sub-acute and chronic (fatigue, anorexia, general wasting). Complications associated with the administration of IFNs can affect nervous, haematopoietic, cardiovascular, endocrine and gastrointestinal systems. Clinical symptoms of autoimmune involvement and renal toxicity have also been described.
In preclinical surveys and in clinical trials, IFN-beta renal side effects were not infrequently reported but usually limited to mild proteinuria (rarely exceeding 1 g/24 h) in 15–20% of patients, increased enzymuria (indicating tubular injury), mild abnormalities in urinalysis, mild azotaemia and transient increase in serum creatinine [1, 2].
Different types of renal toxicity have been associated with IFN therapy: podocytopathies, immune-mediated glomerulonephritis, acute tubular necrosis, chronic tubulo-interstitial nephritis (acute and chronic) or thrombotic microangiopathy (TMA) [3]. The incidence of renal complications remains undetermined, and the causal link between IFN and TMA is still underestimated.
TMA is a rare but severe disorder characterized by thrombotic occlusion of the microcirculation that causes consumptive thrombocytopenia, microangiopathic haemolytic anaemia and signs of ischaemic damage in different organs. Two processes dominate the sequence of pathological events: (i) damage of the endothelium and its activation, resulting in intravascular thrombosis; and (ii) platelet aggregation. Both of these events cause vascular obstruction and vasoconstriction and ultimately precipitation of distal ischaemia. Clinically, there may be different entities indicated as TMA-associated syndromes. Thrombotic thrombocytopenic purpura (TTP) and haemolytic uremic syndrome (HUS) represent the two most frequent causes of TMA. Secondary forms of TMA can be observed in association with drugs, connective tissue diseases, malignancies, transplantation, pregnancy, infections and other factors.
An extensive list of drugs has been associated with TMA [4, 5]. The first case of TMA associated with IFN-alpha was reported in 1993 [6], and the first case associated with IFN-beta was reported in 1998 [7]. Since then, 24 cases of IFN-beta related TMA have been reported in the literature, mostly as individual case reports. Recently, a direct causal effect has been suggested linking TMA with IFN-alpha [8] and IFN-beta [9].
The prognosis of drug-related TMA and particularly IFN-related TMA is particularly severe. It is generally characterized by fulminant presentation, even after years of well-tolerated treatment with IFN-beta, with severe clinical course, poor response to therapy and bad renal prognosis. Treatment has usually been IFN withdrawal and then plasma exchange (PE) and corticosteroids.
Eculizumab is a humanized monoclonal antibody inhibiting C5 activation, a critical effector mechanism in several types of TMA. Approved as first-line treatment for atypical HUS (aHUS), eculizumab was initially reported efficacious in cases that were refractory to plasma therapy and/or relapsing.
Here we report three cases of TMA in patients on IFN-beta, who did not respond to conventional therapy for TMA and who were successfully managed with eculizumab-based treatments.
Materials and methods
We describe three cases of IFN-beta-induced TMA diagnosed by our Nephrology Department between 2010 and 2015 in MS patients. The patients were regularly followed up at the Multiple Sclerosis Centre of the Department of Neurosciences, at the Careggi University Hospital, Florence.
Information on MS, including age at onset, duration of the disease, type of IFN treatment, concomitant medications, physical examination and disability levels at the time of the diagnosis of TMA, laboratory and clinical data were retrieved from medical records.
We also reviewed the IFN-related TMA cases reported in the literature.
A PubMed search was conducted using the medical subject headings terms: ‘thrombotic microangiopathy’, ‘multiple sclerosis’, ‘haemolytic uremic syndrome’, ‘thrombotic thrombocytopenic purpura’ and ‘beta-interferon’.
Articles describing renal TMA cases in patients exposed to IFN-beta treatment were included. We excluded cases in which other possible aetiologic factors or triggers of TMA were present.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Results
Between 2010 and 2015, three cases of IFN-beta-induced TMA were diagnosed and followed by our Nephrology Department. All of the them were part of a cohort of 1988 patients regularly followed by the Multiple Sclerosis Centre of our hospital, of whom 913 were treated with IFN-beta: 441 with subcutaneous IFN-beta 1a (Rebif®), 322 with intramuscular IFN-beta 1a (Avonex®) and 150 with subcutaneous IFN-beta 1b (Betaferon®).
Our three cases are young patients with MS who developed TMA after a prolonged treatment with subcutaneous IFN-beta-1a (44 µg thrice weekly), which was effective in controlling neurological symptoms. Anti-IFN antibodies were negative in all of them.
Patients had normal renal function until a few weeks before admission, with silent medical history except for the RRMS. Immunological tests [cryoglobulins, anti-nuclear antibody (ANA), anti-dsDNA, ANCA, anti-phospholipid antibodies, complement levels] as well as infectious disease tests [hepatitis B virus, hepatitis C virus, human immunodeficiency virus, cytomegalovirus, Epstein–Barr virus, parvovirus B19, Escherichia coli serotype O157:H7) and cancer markers were normal for all the patients.
All the patients presented with marked asthenia, severe high blood pressure (BP), acute kidney injury (AKI) and proteinuria in the absence of purpura, microscopic haematuria or leucocyturia.
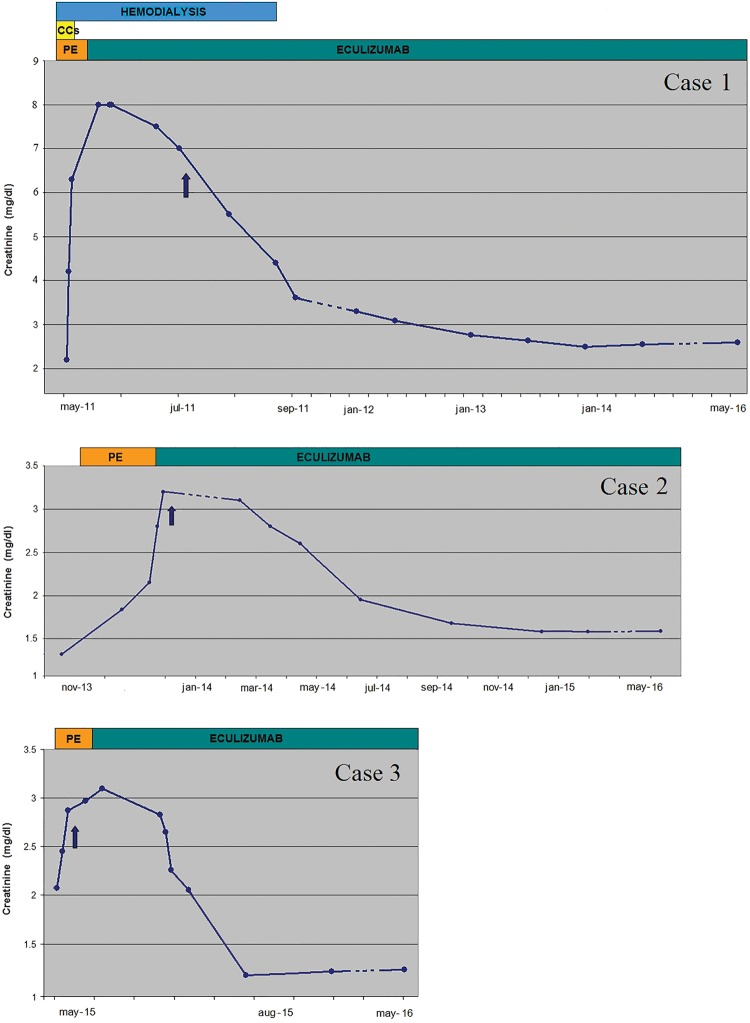
Case 1 is a 46-year-old Caucasian female diagnosed with RRMS in 1994, and treated with IFN-beta-1a (Rebif®) since 1996 with good response and no complications, except for mild leukopenia and skin nodules in the injection sites. In May 2011, she referred to the Emergency Department due to the onset of severe headache, epigastric discomfort and profound weakness. On admission, severe arterial hypertension (250/130 mmHg) was detected and a laboratory workup compatible with TMA was evident (Table 1). The patient was transferred to the Nephrology ward where haemodialysis was initiated due to the severe AKI and anuria. Echocardiogram showed second-degree diastolic dysfunction, mild to moderate pulmonary hypertension and a good systolic function. Subsequent tests, carried out for suspected TMA, showed no mutations in complement genes and normal ADAMTS-13 activity. Despite IFN withdrawal, intravenous corticosteroids and 18 sessions of PE, the severe renal failure persisted with only a transient haematological response (Figure 1). PE was discontinued and eculizumab was started at a dose of 900 mg intravenously for 4 weeks. At discharge the patient was still anuric, on dialysis with suboptimal BP control, but normal haematological values, despite eculizumab maintenance dosing of 1200 mg infused every 2 weeks. Not suspecting a pathogenic effect IFN-beta was reintroduced, but after 1 month it was suspected to be the causative factor for the TMA and was definitively withdrawn. The patient continued with eculizumab, with slow and progressive improvement of renal function, and after 5 months dialysis was permanently discontinued (Figure 1). Four and a half years after the clinical onset, the patient is still under eculizumab therapy and she reports good health and has had no further recurrences of MS. She regained kidney function and remains out of dialysis [chronic kidney disease (CKD) stage 4], her BP is in good control with three anti-hypertensive drugs, there is no proteinuria and no signs of TMA.
Table 1.
Laboratory findings in the three cases at admission
| Case 1 | Case 2 | Case 3 | |
|---|---|---|---|
| Age (years) | 46 | 32 | 34 |
| Sex | F | F | M |
| Creatinine (mg/dL) | 2.2 | 2.15 | 2.97 |
| Urea (g/L) | 1.01 | 1.28 | |
| Urinalysis | No microhaematuria | No microhaematuria | Microhaematuria, glycosuria |
| 24 h proteinuria (g) | 1.5 | 4.9 | 1.32 |
| Hb (g/dL) | 6.9 | 8.0 | 6.3 |
| Platelet (per mm3) | 43 000 | 19 000 | 73 000 |
| PT, APTT, antithrombin, D-dimer | Normal range | Normal range | Normal range |
| Lactate dehydrogenase (U/L) | 1230 | 1929 | 1840 |
| Coombs test | Negative | Negative | Negative |
| Haptoglobine | Undetectable | Undetectable | Undetectable |
| Schistocytes on peripheral blood smear (%) | 7 | 5 | 3 |
| ADAMTS-13 (%) (ref. 50 – 150%) | 90 | 52 | 89 |
| Anti-CFH antibodies | Negative | Negative | Negative |
| Complement gene mutation analysis | Negative (CFH, CFHR1-R3, MCP, CFI, CFB) |
|
|
PT, prothrombin time; APTT, activated partial thromboplastin time.
Fig. 1.
Effect of eculizumab and IFN withdrawal on kidney function in three patients with TMA. Changes in renal function in the three cases since presentation of TMA. Blue arrow: definite discontinuation of IFN-beta from this timepoint; orange bar: period of PE; green bar: period of eculizumab therapy; yellow bar: corticosteroids.
Case 2 is a 32-year-old Caucasian female diagnosed with RRMS in 2002 and treated with IFN-beta-1a (Rebif®) since then. IFN-beta had generally allowed good clinical control of the disease activity. In December 2013, she was hospitalized for dyspnoea associated with tachycardia and hypertension (170/100 mmHg). Echocardiogram showed hypertensive heart disease (interventricular septum 13 mm) with preserved systolic function (ejection fraction 60%). Her laboratory workup on admission (Table 1) showed severe thrombocytopenia, microangiopathic haemolytic anaemia, AKI and nephrotic-range proteinuria. The clinical and laboratory findings were compatible with the diagnosis of TMA. Complement gene mutation analysis showed a heterozygous deletion of CFHR1-R3, an alteration described as non-pathogenic in aHUS. PE with cryo-poor fresh frozen plasma was initiated immediately on a daily basis and then on an alternate-day regime, without any appreciable effect on intravascular haemolysis. She received a total of nine sessions of PE with poor response to treatment, as evidenced by progressive thrombocytopenia with lactate dehydrogenase and creatinine values persistently elevated. Moreover, during the first week, daily blood transfusions were required in order to maintain haemoglobin levels above 8 g/dL. Suspecting an IFN-related TMA, IFN-beta was discontinued and eculizumab therapy was then started. The patient was given intravenous (IV) eculizumab 900 mg weekly for 4 weeks, followed by 1200 mg 1 week later as the fifth dose and then every 2 weeks thereafter. The underlying microangiopathic process has gradually turned off, with slow but progressive improvement in kidney function (Figure 1).
Two months later the patient was hospitalized for left leg weakness, and prescribed three pulses of methylprednisolone and oral prednisone, with rapid tapering. She has since been neurologically stable for over 1 year without steroids. Two and a half years after eculizumab initiation, the patient remains well, with persistent CKD (plasma creatinine 1.65 mg/dL; CKD stage 3), proteinuria 0.85 g/24 h, well-controlled BP with four anti-hypertensive drugs, and no signs of TMA.
Case 3 is a 34-year-old Caucasian man who developed RRMS in 1999 following the appearance of diplopia and right upper limb paraesthesias. In 2001, treatment with IFN-beta-1a (Rebif®) was started with good clinical control of the disease. In May 2015, he developed blurred vision, severe hypertension (205/115 mmHg), palpitations and tinnitus. Laboratory tests performed on arrival at the emergency unit were compatible with TMA (Table 1). TTP was ruled out by measuring ADAMTS 13 activity. Genetic analysis identified a heterozygous MCP mutation in the exon 11, p.Ala353Val(c.1058C > T), previously reported in aHUS but with unknown pathogenic role. IFN treatment was stopped, and the patient was treated with daily PE for a total of eight sessions, until complete normalization of the haematological parameters. However, there was no complete renal function recovery, and a kidney biopsy was performed. Histological evaluation showed endothelial swelling and thickening of the capillary wall due to sub-endothelial expansion. Eculizumab treatment was then started. After about 10 months of treatment, laboratory tests showed a gradual improvement of renal function without clinical or laboratory signs of TMA recurrence (Figure 1). The patient is still on therapy with eculizumab, maintaining stable renal function (CKD stage 2) and good BP control.
Discussion
The case reports we present, together with the 24 reported in the literature (Table 2), suggest a causal relationship between IFN-beta treatment and TMA, hindering the hypothesis of a fortuitous association. This study enables us to delineate several peculiarities of this association.
Table 2.
Cases of TMA occurring in MS patients treated with IFN-beta reported in the literature
| Author | Patient (sex, age in years) | Exposure to IFN-beta | Clinical presentation | Laboratory analysis | Complement mutations/ADAMTS13 activity | Therapy in addition to withdrawal of IFN-beta and anti-hypertensive drugs | Outcome |
|---|---|---|---|---|---|---|---|
| Nerrant [10] | F, 38 | 7 months |
|
AKI | no complement mutations, normal ADAMTS13 | PE, steroids per os | ESRD |
| Broughton [11] | F, 53 | 8 years Betaseron® |
mild/severe hypertension, headache | AKI, proteinuria, haematuria | – | – | CKD |
| Mahe [12] | F, 38 | 5 years | BP 180/130 mmHg | AKI | no complement mutations | – | stage 4 CKD |
| Mahe (FPD) | NA, 58 | 5 years | severe hypertension | AKI | – | PE, steroids | CKD |
| Mahe (FPD) | NA, 66 | 1 year | hypertension | – | – | – | remission |
| Mahe (FPD) | NA, 52 | >3 years | hypertension | AKI | ADAMTS13 deficit | PE | CKD |
| Mahe (FPD) | NA, 55 | 6 years | – | – | Ab-anti-ADAMTS13 | PE, steroids, RTX | remission |
| Mahe (FPD) | NA, 38 | 10 years | – | – | – | PE, steroids | NA |
| Orvain [13] | M, 52 | 4 years |
|
|
|
PE, steroids 1 mg/kg/day, RTX | ESRD |
| Vosoughi [9] | M, 52 | 14 years Betaseron® |
|
AKI, no proteinuria | – | PE | ESRD |
| Vosoughi [9] | F, 41 | 12 years Rebif® |
|
|
– | – | recovered |
| Rubin [14] | F, 41 | 10 years |
|
AKI, haematuria, proteinuria 1.35 g/24 h | – | – | remission |
| Larochelle [15] | F, 34 | 14 months |
|
|
– | PE, IV steroids, vincristine, RTX | Death |
| Larochelle [15] | F, 47 | 11 years |
|
AKI, mild proteinuria | – | PE, IV steroids | stage 4 CKD |
| Larochelle [15] | F, 41 | 5 years |
|
AKI | – | PE, steroids per os, RTX | ESRD |
| Azkune Calle [16] | M, 36 | 9 years IFN-beta-1a |
|
no AKI | – | PE, steroids | recovered |
| Capobianco [17] | F, 22 | 13 years IFN-beta-1a |
|
|
– | – | remission |
| Li Cavoli [18] | F, 36 | 3 months | severe hypertension | AKI | – | PE, steroids (IV and per os) | ESRD |
| Olea [19] | F, 37 | 5 months | BP 205/110 mmHg |
|
no complement mutations | steroids 1 mg/kg/day | recovered |
| Hunt [20] | NA | 8 years |
|
AKI | no complement mutations, normal ADAMTS13 | NA | ESRD |
| Hunt [20] | NA | 6 years |
|
AKI | no complement mutations, normal ADAMTS13 | NA | ESRD |
| Hunt [20] | NA | 10 years |
|
AKI | no complement mutations, normal ADAMTS13 | NA | recovered |
| Hunt [20] | NA | 6 years | BP 185/133 mmHg | AKI | no complement mutations, normal ADAMTS13 | NA | ESRD |
| Serrano [21] | M, 50 | 9 years IFN-beta-1b |
headache, lipothymia, visual loss | AKI | – | PE, steroids | remission |
| Case 1 | F, 46 | 15 years |
|
AKI, subnephrotic-range proteinuria | no complement mutations, normal ADAMTS13 | PE, steroids, eculizumab | stage 4 CKD |
| Case 2 | F, 32 | 11 years |
|
AKI, nephrotic-range proteinuria | no complement mutations, heterozygous CFHR1-R3 deletion, normal ADAMTS13 | PE, eculizumab | stage 3 CKD |
| Case 3 | M, 35 | 14 years |
|
AKI, subnephrotic-range proteinuria, microhaematuria | heterozygous MCP mutation, normal ADAMTS13 | PE, eculizumab | stage 2 CKD |
FPD, French Pharmacovigilance Database; NA, not available; RTX, rituximab.
The onset of TMA occurred after a mean of 7 years of IFN-beta therapy. Only 6 out of 27 patients had a drug exposure <4 years, while 10 out of 27 patients had an exposure ≥ 10 years. Our three cases, as well as those described in the literature, indicate that long-term IFN therapy can induce late-onset TMA, suggesting that the development of renal lesions may be the result of a cumulative effect in renal and systemic microvessels.
In all the eight published cases reporting the results of genetic analysis, no mutations of complement factors were identified. In two cases, anti-ADAMTS13 IgG, the synthesis of which may have been favoured by IFN-beta treatment, were described. In support of this theory and in support of the immunogenicity of IFN [22], there are several case reports describing acquired anti-ADAMTS13 IgG after IFN-alpha therapy.
In our cases, case 1 had no identified mutations, case 2 showed a heterozygous deletion CFHR1-R3, which is a polymorphism often associated with anti-CFH antibodies in aHUS, that were absent in this patient, and case 3 showed a heterozygous MCP mutation p.Ala353Val(c.1058C > T). This mutation has been previously described in three children with aHUS, but the pathological role is still unclear, because all those patients had other risk factors or triggers of aHUS. The 3-year-old boy described by Davin et al. [23] had several at-risk polymorphisms of MCP and CFH, which may have enhanced the pathogenic role of the MCP mutation. The child described by Bouts et al. [24] had methylmalonic aciduria and homocystinuria secondary to cobalamin CblC disorder, which was considered the real cause of aHUS. The 9-year-old girl described by Banks et al. [25] was diagnosed with a focal segmental glomerulosclerosis 1 year before developing aHUS. It is well known that different types of glomerulopathies may trigger aHUS with different mechanisms, frequently when associated with mutations or risk haplotypes for regulators of the alternative complement pathway [26]. In the three mentioned cases, as in our patient, the mutation of MCP could predispose to aHUS, but a trigger is probably necessary, which in this case was the treatment with IFN-beta.
Genetic analysis of complement factors, as well as ADAMTS-13 levels and ADAMTS-13 inhibitors, should be performed in patients with IFN-related TMA. This may prove useful in the interpretation of the response to therapy and in driving the maintenance regimen.
The clinical presentation of IFN-related TMA is usually very severe and the renal prognosis is often poor.
When considering all the cases of IFN-beta-induced TMA, in published reports and herein, 85% presented with AKI, generally associated with sub-nephrotic proteinuria and mild or absent microhaematuria. Of these, 42% evolved towards end-stage renal disease (ESRD), 37% to advanced degree CKD, while only 21% recovered, often with no restitutio ad integrum and with persistent hypertension or proteinuria.
All published cases of IFN-beta-induced TMA had high BP, reported as severe hypertension (65%) or even malignant hypertension (MHT) (25%). This is particularly interesting since the US Food and Drug Administration reported arterial hypertension to occur rarely (0.4–5%) in patients treated with Rebif® or Betaseron®. All the patients had fulminant presentation of the disease characterized by severe or MHT developed after years of well-tolerated treatment with IFN-beta. Moreover, hypertension often persisted after TMA resolution.
Malignant hypertension is defined as severe hypertension accompanied by ischaemic failure of one or more organs (kidney, cardiac hypertrophy, hypertensive encephalopathy) [27].
Our three patients had severe hypertension, ischaemic damage of the kidneys, cardiac hypertrophy and hypertensive encephalopathy, which can be redefined as MHT-induced TMA secondary to IFN-beta.
Neurological involvement has been described in the acute phase in 17–55% of aHUS and in 100% of TTP cases. In patients with IFN-related-TMA, neurological involvement was frequent (70% of cases). The most common presenting symptom was severe headache, but seizures, confusion, focal deficits (paresis, hemiparesis), psychosis and visual loss were also described.
In about 25% of patients with neurological symptoms described in the literature, magnetic resonance imaging (MRI) documented cerebral lesions compatible with posterior reversible encephalopathy syndrome (PRES). The presence of PRES in the course of TMA has often been described, generally with uncontrolled hypertension and other factors like severe hypoproteinaemia, hypocalcaemia, high haematocrit or pulse methylprednisolone; characteristic presenting symptoms were headache, dimness of vision or cortical blindness, seizures and altered mentation. Unfortunately, cerebral MRI has not been performed in our patients at onset.
Despite treatment with PE, steroids or rituximab, renal dysfunction progressed in some patients, and the prognosis was generally poor.
Our patients and almost all the cases reviewed in the literature had normal renal function and urinalyses prior to IFN-beta therapy. Pre-existing hypertension was reported only in a few patients.
Conventional approaches to managing this entity, such as withdrawal of the suspected offending agent and/or PE, obtained limited improvement, with high rates of renal failure.
Eculizumab has been registered for the treatment of aHUS due to mutations in complement factors of the alternative pathway, but it also appears to be effective in the treatment of several secondary forms of TMA, e.g. pregnancy, haematopoietic stem cell transplantation, systemic lupus erythematosus and catastrophic antiphospholipid syndrome.
From a review of the literature, there are several described cases in which eculizumab was prescribed for drug-induced TMA that were refractory to conventional therapies. Eculizumab has been used with remarkable effectiveness for TMA secondary to therapy with gemcitabine [28, 29], cisplatin [30] and forms of de novo TMA after kidney transplantation associated with calcineurin inhibitors [31, 32].
In MHT-induced TMA, it was demonstrated in rats that activation of renin–angiotensin system leads to angiotensin II-induced damage and complement activation [33]. We could speculate that this may be the pathophysiological rationale regarding the effectiveness of eculizumab in our patients.
To our knowledge, we report the first three cases of IFN-related TMA treated with eculizumab showing a strikingly favourable effect on renal recovery. All of them responded with progressive clinical and renal improvement, allowing dialysis discontinuation, without recurrence of TMA during a long-term follow-up.
Despite the favourable adverse event profile of eculizumab, the very high cost of the drug and the scarceness of comparative studies on its long-term effectiveness and safety do not allow us to recommend the use of eculizumab as first-line strategy for IFN-beta induced TMA. We decided to start eculizumab after a mean of 12 PE (range 8–18) sessions, due to the severity of renal involvement despite partial haematological improvement.
There is also a need for a better understanding on the strategy to taper or withdraw eculizumab once it has been initiated. In our experience, we decided not to stop eculizumab considering the severity of renal involvement, the genetic abnormalities potentially predisposing to a recurrence of disease and the extremely favourable effect as a single agent on the course of MS, which is now object of further analysis.
In conclusion, TMA can occur in MS patients treated with IFN-beta, and these observations should prompt the neurologist to vigilance on this rare complication. Even after years of well-tolerated therapy, new onset of headache, hypertension or renal disease should be carefully evaluated since early diagnosis may improve clinical outcomes. The clinical presentation of IFN-related TMA is usually severe and the renal prognosis is often poor. Once it is suspected, interruption of IFN treatment is warranted while PE or eculizumab therapy is still a matter of debate. In our experience, after a run of unsuccessful PE, all three patients had a very favourable effect on TMA response and renal function with eculizumab therapy.
Conflict of interest statement
M.A., C.L.C., L.C., G.A., S.F., M.P.A. and E.E.M. declare no conflicts of interest with respect to the research, authorship and/or publication of this article. The authors declare that the results presented in this paper have not been published previously, in whole or part.
References
- 1. Quesada JR, Talpaz M, Rios A. et al. Clinical toxicity of interferons in cancer patients: a review. J Clin Oncol 1986; 4: 234–243 [DOI] [PubMed] [Google Scholar]
- 2. Kurschel E, Metz-Kurschel U, Niederle N. et al. Investigations on the subclinical and clinical nephrotoxicity of interferon alpha-2B in patients with myeloproliferative syndromes. Ren Fail 1991; 13: 87–93 [DOI] [PubMed] [Google Scholar]
- 3. Markowitz GS, Bomback AS, Perazella MA.. Drug-induced glomerular disease: direct cellular injury. Clin J Am Soc Nephrol 2015; 10:1291–1299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Al-Nouri ZL, Reese JA, Terrell DR. et al. Drug-induced thrombotic microangiopathy: a systematic review of published reports. Blood 2015; 125: 616–618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Reese JA, Bougie DW, Curtis BR. et al. Drug-induced thrombotic microangiopathy: experience of the Oklahoma registry and the BloodCenter of Wisconsin. Am J Hematol 2015; 90: 406–410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Stratta P, Canavese C, Dogliani M. et al. Hemolytic-uremic syndrome during recombinant alpha-interferon treatment for hairy cell leukemia. Ren Fail 1993; 15: 559–561 [DOI] [PubMed] [Google Scholar]
- 7. Ubara Y, Hara S, Takedatu H. et al. Hemolytic uremic syndrome associated with beta-interferon therapy for chronic hepatitis C. Nephron 1998; 80: 107–108 [DOI] [PubMed] [Google Scholar]
- 8. Demoulin N, Jadoul M.. Interferon-α-induced thrombotic microangiopathy in patients with chronic myelogenous leukemia. Kidney Int 2014; 85: 213–214 [DOI] [PubMed] [Google Scholar]
- 9. Vosoughi R, Marriott JJ.. Thrombotic microangiopathy in Interferon Beta treated multiple sclerosis patients: review of literature and report of two new cases. Mult Scler Relat Disord 2014; 3: 321–325 [DOI] [PubMed] [Google Scholar]
- 10. Garg N, Weinstock-Guttman B, Bhasi K. et al. An association between autoreactive antibodies and anti-interferon-beta antibodies in multiple sclerosis. Mult Scler 2007; 13: 895–899 [DOI] [PubMed] [Google Scholar]
- 11. Davin JC, Buter N, Groothoff J. et al. Prophylactic plasma exchange in CD46-associated atypical haemolytic uremic syndrome. Pediatr Nephrol 2009; 24: 1757–1760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Bouts AH, Roofthooft MT, Salomons GS. et al. CD46-associated atypical hemolytic uremic syndrome with uncommon course caused by CblC deficiency. Pediatr Nephrol 2010; 25: 2547–2548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Banks A, Grisaru S, Wade A. et al. CD46-associated atypical hemolytic uremic syndrome with initial focal segmental glomerulosclerosis. CSN 2014. Abstracts. http://csn.qxmd.com/csn-2013-abstracts/ [Google Scholar]
- 14. Manenti L, Gnappi E, Vaglio A. et al. Atypical haemolytic uraemic syndrome with underlying glomerulopathies. A case series and a review of the literature. Nephrol Dial Transplant 2013; 28: 2246–2259 [DOI] [PubMed] [Google Scholar]
- 15. Mancia G, Fagard R, Narkiewicz K. et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 2013; 34: 2159–2219 [DOI] [PubMed] [Google Scholar]
- 16. Ustwani O, Lohr J, Dy G. et al. Eculizumab therapy for gemcitabine induced hemolytic uremic syndrome: case series and concise review. J Gastrointest Oncol 2014; 5: E30–E33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Rogier T, Gerfaud-Valentin M, Pouteil-Noble C. et al. Clinical efficacy of eculizumab as treatment of gemcitabine-induced thrombotic microangiopathy: a case report. Rev Med Interne 2016 [DOI] [PubMed] [Google Scholar]
- 18. Gilbert RD, Stanley LK, Fowler DJ. et al. Cisplatin-induced haemolytic uraemic syndrome associated with a novel intronic mutation of CD46 treated with eculizumab. Clin Kidney J 2013; 6: 421–425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chandran S, Baxter-Lowe L, Olson JL. et al. Eculizumab for the treatment of de novo thrombotic microangiopathy post simultaneous pancreas-kidney transplantation – a case report. Transplant Proc 2011; 43: 2097–2101 [DOI] [PubMed] [Google Scholar]
- 20. Safa K, Logan MS, Batal I. et al. Eculizumab for drug-induced de novo post-transplantation thrombotic microangiopathy: a case report. Clin Nephrol 2015; 83: 125–129 [DOI] [PubMed] [Google Scholar]
- 21. Shagdarsuren E, Wellner M, Braesen JH. et al. Complement activation in angiotensin II-induced organ damage. Circ Res 2005; 97: 716–724 [DOI] [PubMed] [Google Scholar]
- 22. Nerrant E, Charif M, Ramay AS. et al. Hemolytic uremic syndrome: an unusual complication of interferon-β treatment in a MS patient. J Neurol 2013; 260: 1915–1916 [DOI] [PubMed] [Google Scholar]
- 23. Broughton A, Cosyns JP, Jadoul M.. Thrombotic microangiopathy induced by long-term interferon-β therapy for multiple sclerosis: a case report. Clin Nephrol 2011; 76: 396–400 [DOI] [PubMed] [Google Scholar]
- 24. Mahe J, Meurette A, Moreau A. et al. Renal thrombotic microangiopathy caused by interferon beta-1a treatment for multiple sclerosis. Drug Des Devel Ther 2013; 7: 723–728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Orvain C, Augusto JF, Besson V. et al. Thrombotic microangiopathy due to acquired ADAMTS13 deficiency in a patient receiving interferon-beta treatment for multiple sclerosis. Int Urol Nephrol 2014; 46: 239–242 [DOI] [PubMed] [Google Scholar]
- 26. Rubin S, Lacraz A, Galantine V et al.. Malignant hypertension and interferon-beta: a case report. J Hum Hypertens 2014: 340–341 [DOI] [PubMed] [Google Scholar]
- 27. Larochelle C, Grand'maison F, Bernier GP. et al. Thrombotic thrombocytopenic purpura-hemolytic uremic syndrome in relapsing-remitting multiple sclerosis patients on high-dose interferon β. Mult Scler 2014; 20: 1783–1787 [DOI] [PubMed] [Google Scholar]
- 28. Azkune Calle I, Sánchez Menoyo JL, Ruiz Ojeda J. et al. Case report of thrombotic microangiopathy associated with subcutaneous interferon beta-1a: an emerging complication? Neurologia 2014; pii: S0213-4853(14)00204-7 [DOI] [PubMed] [Google Scholar]
- 29. Capobianco M, Piccoli G, Neve Vigotti F. et al. Interferon beta-related nephropathy and interstitial lung disease: a new association and a long-term warning. Mult Scler 2014; 20: 889–891 [DOI] [PubMed] [Google Scholar]
- 30. Li Cavoli G, Bono L, Tortorici C. et al. Renal thrombotic microangiopathy induced by b-interferon. NDT Plus 2011; 4: 79–80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Olea T, Díaz-Mancebo R, Picazo ML. et al. Thrombotic microangiopathy associated with use of interferon-beta. Int J Nephrol Renovasc Dis 2012; 5: 97–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hunt D, Kavanagh D, Drummond I. et al. Thrombotic microangiopathy associated with interferon beta. N Engl J Med 2014; 370: 1270–1271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Serrano A, Xicoy B, Grifols JR. et al. Thrombotic thrombocytopenic purpura during treatment with interpheron. Med Clin (Barc) 2007; 128: 276–277 [DOI] [PubMed] [Google Scholar]