Abstract
Introduction
Down syndrome results in neuromotor impairment that affects selective motor control, compromising the acquisition of motor skills and functional independence. The aim of the proposed study is to evaluate and compare the effects of multiple-monopolar anodal transcranial direct current stimulation and sham stimulation over the primary motor cortex during upper limb motor training involving virtual reality on motor control, muscle activity, cerebral activity and functional independence.
Methods and analysis
A randomised, controlled, double-blind, clinical trial is proposed. The calculation of the sample size will be defined based on the results of a pilot study involving the same methods. The participants will be randomly allocated to two groups. Evaluations will be conducted before and after the intervention as well as 1 month after the end of the intervention process. At each evaluation, three-dimensional analysis of upper limb movement muscle activity will be measured using electromyography, cerebral activity will be measured using an electroencephalogram system and intellectual capacity will be assessed using the Wechsler Intelligence Scale for Children. Virtual reality training will be performed three times a week (one 20 min session per day) for a total of 10 sessions. During the protocol, transcranial stimulation will be administered concomitantly to upper limb motor training. The results will be analysed statistically, with a p value≤0.05 considered indicative of statistical significance.
Ethical aspects and publicity
The present study received approval from the Institutional Review Board of Universidade Nove de Julho (Sao Paulo,Brazil) under process number 1.540.113 and is registered with the Brazilian Registry of Clinical Trials (N° RBR3PHPXB). The participating institutions have presented a declaration of participation. The volunteers will be permitted to drop out of the study at any time with no negative repercussions. The results will be published and will contribute evidence regarding the use of this type of intervention on children.
Keywords: down syndrome, transcranial direct current stimulation, upper limb
Strengths and limitations of this study.
The proposed project involves the combination of virtual reality (VR) activities for upper limb motor training and multiple-monopolar anodal transcranial direct current stimulation (tDCS) over the primary motor cortex with the aim of optimising motor control and upper limb function in children with Down syndrome (DS).
Adequate upper limb motor control enables individuals to perform daily, functional and academic activities in an independent fashion.
The use of VR activities to improve motor control is a promising therapeutic resource that has demonstrated satisfactory results in the scientific literature, including for individuals with DS.
Non-invasive brain stimulation techniques, specifically tDCS, are currently considered effective means to facilitate motor cortical excitability of brain regions underlying the stimulation electrode, leading to improvements in motor control and motor learning. Despite the lack of reports on the effects of transcranial stimulation in children with DS, studies involving paediatric patients have demonstrated that the technique is safe, with little or no adverse effects.
We believe that the administration of multiple-monopolar anodal transcranial direct current stimulation over the primary motor cortex, specifically the areas that correspond to upper limb motor control (C3 and C4 of the 10–20 electroencephalogram system) during upper limb motor training with the use of VR activities will enhance the cortical excitability of motor regions and optimise cerebral activity, thereby potentiating the effects of upper limb motor therapy.
The literature reports positive effects with the use of tDCS on upper limb movements in children with cerebral palsy. Optimising such movements has a direct impact on improving one’s performance of activities of daily living and functional independence. However, no scientific data were found regarding the use of tDCS during upper limb training in the population of the proposed study (children with DS).
The literature also reports promising results with the use of VR regarding improvements in cognitive aspects of the population in question, as this intervention constitutes multisensory therapy that optimises one’s concentration and assists in the anticipation of movements, thereby exerting an impact on learning aspects in children submitted to this intervention.
The limitations of the proposed study regard the lack of scientific data from previous studies involving children with DS for the purposes of comparison with the findings obtained in the proposed study. However, this aspect also demonstrates the importance of the data that will be generated in the proposed study.
Introduction
Down syndrome (DS) is a highly prevalent genetic disease caused by the inheritance of an additional chromosome 21 and is one of the most frequent causes of mental impairment, affecting approximately 20% of the total number of individuals with mental disability.1 The incidence in the USA is one out of every 700 births, and it is estimated that at least 100 thousand individuals in Brazil are diagnosed with the syndrome.2–4
Structural and functional abnormalities are found in the nervous system of children with DS. Diffuse brain damage and peculiar electrical functioning during cognitive development result in poor analysis, synthesis and speech skills. Moreover, such children demonstrate difficulties in selecting and directing a stimulus due to the fatigue of the connections. These abnormalities result in neurological disorders that vary in terms of manifestation and intensity.5
According to Flórez and Troncoso, the brain of individuals with DS is smaller in volume in comparison to individuals without this condition. Hypoplasia of the frontal and occipital lobes is a common finding. A unilateral or bilateral reduction in the temporal lobe occurs in up to 50% of cases and reductions in the corpus callosum, anterior commissure and hippocampus are found.6 7 Such individuals also have a smaller number of secondary sulci in comparison to individuals without this syndrome, the temporal gyri are underdeveloped and differences in nerve cells are also reported. For instance, Pandilla reports differences in the axons and dendrites of pyramidal neurons in the motor cortex.8 Such differences are highly correlated with fragmentation problems and necrosis of these branches as well as differences in the electrical activity of the brain.9 This problem leads to limitations with regard to synaptic connections and the neural transmission of nerve impulses.
The literature also reports atrophied nerve cells, which are likely associated with lags during the integration of visual and spatial information. According to Block, individuals with DS also have a smaller cerebellum and base ganglia, which are related to the control of coordination, timing and balance. Such problems imply limitations with regard to the acquisition of motor skills.10 According to Bomono and Rosetti, neuromotor abnormalities in DS include hypotonia, diminished primitive reflexes, delayed motor and cognitive development and lower levels of learning.11
Seaman and DePauw9 propose a model in which reaching phases of fundamental movements and culturally determined movements is conditioned by the achievement of previous development phases.9 As this population exhibits problems with regard to systems of early-onset and late-onset maturation, children with DS could encounter difficulties reaching the phase of sensorimotor responses and even acquiring motor skills. According to Connolly, the mechanisms or systems that offer support to development and the acquisition of motor skills can be understood using the concepts of ‘hardware’ and ‘software’, in which changes in ‘hardware’ regard structure, such as the myelinisation that occurs in axons, whereas changes in ‘software’ regard function, such as a gain in information processing speed as a result of myelinisation; thus, individuals with DS have problems with their ‘hardware’ that have repercussions on their ‘software’.12 ‘Hardware’ problems lead to limitations with regard to physical and motor aspects, which is an important problem, as both physical proficiency and perceptive-motor proficiency contribute to the acquisition and performance of motor skills. In other words, it is possible that problems with balance, timing and agility constitute a hindrance to the acquisition of fundamental patterns or specialised skills.13
The population with DS exhibits abnormal muscle coordination, difficulty processing sensory information and functional limitations. The upper limb dysfunctions in this population (muscle weakness and hypotonus, slow reflexes, abnormal biomechanics, sensory deficiency) exert a negative impact on the performance of activities of daily living, independence and quality of life.14
Studies have been conducted to understand why individuals with DS have slow, unharmonious movements.15–25 The investigation of electromyographic activity and muscle torque demonstrates this deficiency, which can be corrected by the repetition of a given movement during motor training activities. Motor control strategies used in the execution of complex activities, such as a reaching task, have been investigated in this population.15–25
The positive results achieved with virtual reality (VR) are believed to be related to training in an interactive environment that provides a broad range of activities and scenarios with multiple sensory channels, enabling the creation of exercises at an intensity that is promising for the needs of individuals with DS.26–28 VR can be used as an auxiliary tool involving a playful, motivational objective that can facilitate the development of perceptions and motor skills through the training of planning skills and motor control as well as stimulation of the plasticity of the central nervous system.27 28
Non-invasive brain stimulation methods have been employed in physical rehabilitation protocols due to the promising results achieved with regard to motor learning in the paediatric population with cerebral palsy since it was never used in DS.29 30 Transcranial direct current stimulation (tDCS) is a relatively low-cost, non-invasive brain stimulation technique that is easy to administer and offers minimal adverse effects. This method is known to produce lasting changes in motor cortical excitability.31 Cortical modulation depends on the polarity of the current: anodal stimulation increases cortical excitability, favouring the depolarisation of the neuronal membrane, whereas cathodal stimulation has an inhibitory effect due to the hyperpolarisation of the neuronal membrane.31–36
TDCS has advantages over other transcranial stimulation techniques, such as providing a long-lasting modulating effect on cortical function as well as its ease of use because its device is portable, so it is possible to because simultaneously with rehabilitation techniques and has lower cost. The results of clinical trials have demonstrated its considerable potential in the treatment of neurological disorders and the investigation of processes of cortical excitability modulation.37–42 Moreover, this type of intervention offers a better condition for sham stimulation, which confers greater specificity to the findings.39 40 In the rehabilitation process, the aim of neuromodulating techniques is to enhance local synaptic efficiency and alter the maladaptive plasticity pattern that emerges after a cortical injury.41–45
Although DS is one of the most prevalent diseases in the paediatric population, no studies were found on the effects of tDCS on children with this syndrome. Thus, the lack of investigations on anodal tDCS over the primary motor cortex during motor training for children with DS constitutes a gap in the scientific literature.46–48 Considering the high prevalence of DS, the motor limitations stemming from this disease, which exert a negative impact on functionality and independence and the fact that tDCS is not contraindicated in most cases of this syndrome, the investigation of the effects of this non-invasive brain stimulation technique on children with DS is relevant.43–45
The proposed study could be used as the basis for the development of further projects conducted to broaden knowledge on this technique, enabling a novel intervention option for the optimisation of motor training in individuals with DS.
Objectives
Primary objective
The aim of the proposed study is to evaluate and compare the effect of multiple-monopolar anodal tDCS and sham stimulation over the primary motor cortex during upper limb motor training involving VR on motor control (spatiotemporal variables and kinematics of a reaching task), activity of the elbow flexors and extensors, cerebral activity and functional independence in children with DS.
Hypotheses
Null hypothesis
Ten sessions of tDCS over the motor cortex concomitantly to upper limb motor training involving the use of VR activities will result in the same effects as motor training with the use of virtual reality combined with sham transcranial stimulation in children with DS
Alternative hypothesis
Ten sessions of tDCS over the motor cortex concomitantly to upper limb motor training involving the use of virtual reality activities will result in the better effects than motor training with the use of VR combined with sham tDCS in children with DS.
Secondary objectives
Determine possible correlations between upper limb motor control (movement velocity and total duration of movement) and muscle activity (elbow flexors and extensors), cerebral activity (activity of the parietal lobe, specifically regions C3 and C4) and functional independence with regard to self-care.
Identify possible prediction factors for the response of upper limb motor control (movement velocity and total duration of movement) in children with DS. Muscle activity of elbow flexors and extensors, cerebral activity (areas C3 and C4 of the 10–20 electroencephalogram system) and tDCS (active and sham) will be the factors investigated.
Methods and analysis
The sample will be composed of children with DS recruited from the physical therapy clinics of Universidade Nove de Julho, São Paulo, Brazil. Letters and emails will be sent to paediatricians, physiotherapists and paediatric neurologists to divulge the study. The following will be the inclusion criteria: (1) a diagnosis of DS; (2) adequate comprehension and cooperation during the procedures; (3) age six to 12 years; (4) compromised upper limb motor coordination and (5) statement of informed consent signed by a legal guardian. The exclusion criteria will be (1) having undergone surgical procedures in the 12 months prior to the onset of the training sessions, (2) orthopaedic deformity of the lower limbs or spinal column with an indication for surgery, (3) epilepsy, (4) metal implant in skull or hearing aids, (5) associated neurological disorder and (6) use of a pacemaker.
Study design
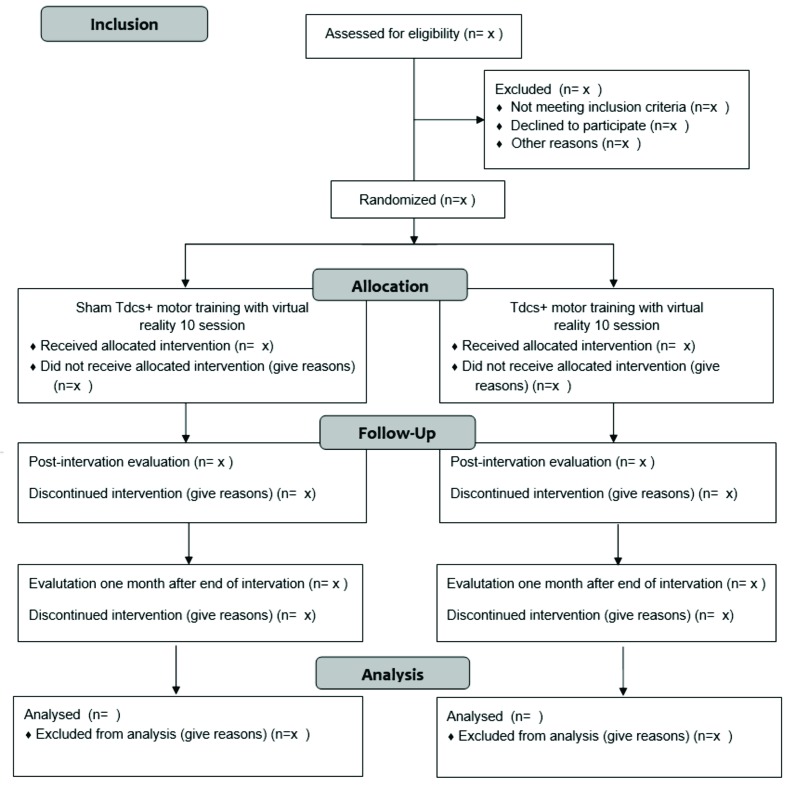
A phase I–II study will be conducted (figure 1): analytical, paired, randomised, controlled, double-blind, clinical trial.
Figure 1.
Flowchart of study based on the Consolidated Standards for Reporting of Trials
Sample size
The sample size will be calculated based on the results of a pilot study with the same methods as those of the main study. The pilot study will involve 10 children randomly allocated to the experimental and control groups (five children in each group). The sample size will be calculated based on the mean of both groups considering total duration of movement as the primary outcome, with a unidirectional alpha of 0.05 and an 80% power. The sample will be increased by 20% to compensate for possible dropouts.
Randomisation
Patients with DS who meet the eligibility criteria and agree to participate in the study will be submitted to an initial evaluation and will then be randomly allocated to two groups using a randomisation method available at the site www.randomizacion.com. This process will be performed by a member of the research team who is not involved in the recruitment or development of the study. During the protocol, the blinding of the main researcher will be ensured with the use of the DC-Stimulator (NeuroConn, Germany), which has active and sham modes that function based on encrypted code, with three configurations to choose so that the more complex conditions of the study can be achieved. The parameters are adjusted individually and the activated mode can only be altered by the programmer.
Experimental group: multiple-monopolar anodal tDCS over the primary motor cortex bilaterally combined with upper limb motor training involving the use of VR.
Control group: sham tDCS over the primary motor cortex bilaterally combined with upper limb motor training involving the use of VR.
Evaluations
The participants will be submitted to three evaluations: preintervention, postevaluation (after 10 training sessions) and follow-up (1 month after last training session).
Three-dimensional movement analysis
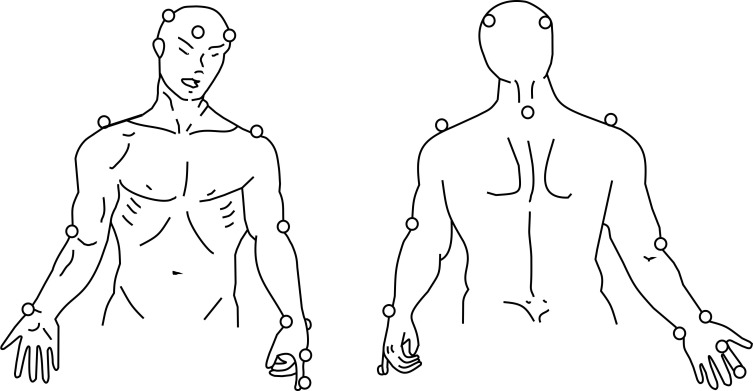
Three-dimensional analysis of upper limb movement: the kinematics of upper limb movement will be evaluated using the SMART-D 140 system (BTS, Milan, Italy), with eight cameras sensitive to infrared light, a sampling frequency of 100 Hz and video system synchronised with the SMART-D system. Passive markers will be positioned at anatomic references points directly on the skin with specific adhesive tape, following the protocol of the SMARTup: the experimental setup (figure 2).49–51 A total of 18 markers measuring 15 mm in diameter will be used to identify the position of the head, trunk and upper limbs (upper arm, forearm and hand).
Figure 2.
Placement of markers for three-dimensional analysis using SMARTup: the experimental setup.49
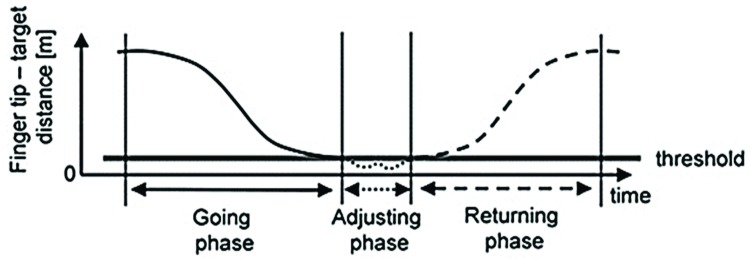
The movement will be divided into three phases: going phase (upper limb moving towards the target), adjusting phase (adjustment of arm to locate target precisely) and returning phase (return to initial position). At least six complete movements will be performed to obtain three adequate cycles for analysis (figure 3). The biomechanical model, filtering of the data and processing of the variables will be performed using the SMART analyser software (BTS, Milan, Italy). The variables will be identified and calculated for each movement cycle to evaluate any changes that occur after the intervention. The following variables will be considered, with the mean of the results used in the statistical analyses:
Figure 3.
Phases of reaching cycle.49
Total duration of movement: total time required to perform the complete reaching task;
Mean movement velocity: computed during the going phase and determined using the marker positioned on the index finger;
Adjusting sway index: Defined as the length of the three-dimensional path described by the marker on the index finger during the adjusting phase;
Range of motion of elbow and shoulder: calculated as the difference between the maximum and minimum angles of the elbow and shoulder on the sagittal (elbow and shoulder) and frontal (shoulder) planes during the going phase, as described in the literature.49–51
Electromyographic analysis
Muscle activity during the reaching movement will be determined using electromyographic (EMG). The electrical activity resulting from the activation of the elbow flexors and extensors will be collected using an eight-channel electromyograph (FREEEMG, BTS Engineering) with a bioelectrical signal amplifier, wireless data transmission and bipolar electrodes with a total gain of 2000-fold and frequency ranging from 20 to 450 Hz. Impedance and the common rejection mode ratio of the equipment are >1015 Ω/0.2 pF and 60/10 Hz 92 dB, respectively. The motor point of the muscles will be identified for the placement of the electrodes and the skin will be cleaned with 70% alcohol to reduce bioimpedance, following the recommendations of Surface Electromyography for the Non-Invasive Assessment of Muscles.52 All EMG data will be digitised at 1000 frames per second using the BTS MYOLAB software program. The data will be collected simultaneously to the kinematic data and both will be managed using the BTS system and Smart Capture software program.52 53
Electroencephalographic analysis
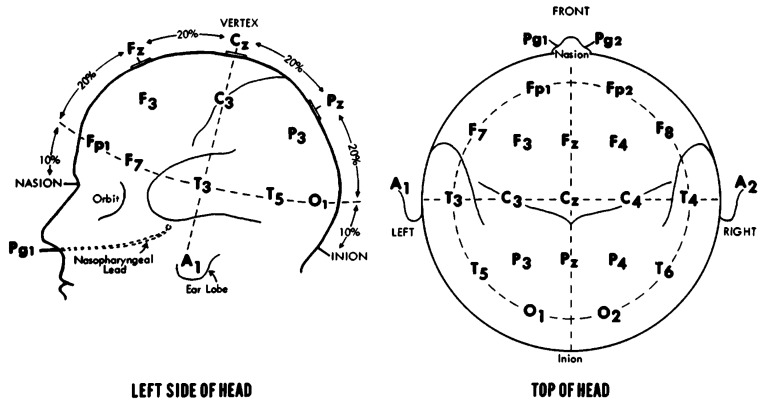
Brain activity will be investigated using electroencephalography (EEG), which will be performed during both the three-dimensional analysis of the reaching task and the evaluation of muscle activation using EMG. For such, the volunteer will be seated in an erect position on a chair in front of the table on which the reaching task will be performed. The BrainNet BNT36 device with 36 configurable channels (32 AC and four DC) and a 16-bit analogue–digital converter will be used for the acquisition of the EEG signal (figure 4). The analysis of the signal will be performed with the aid of the EEGLab tool implemented on Matlab, which is also capable of furnishing a topographic map of cerebral activity as a function of time. The electrodes will be positioned following the guidelines of the 10/20 EEG system (figure 5).54 55
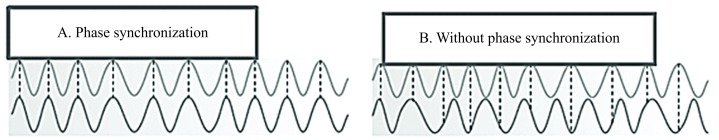
Figure 4.
Phase relationships. (A) Synchronised signals—differences in phases between both signals are stable (constant); (B) non-synchronised signals— differences in phases are variable.
Figure 5.
Positioning of electroencephalography electrodes based on 10–20 standard.
Paediatric evaluation of disability inventory
The children’s functional performance will be assessed quantitatively using the Paediatric Evaluation of Disability Inventory (PEDI), which is a questionnaire administered in interview format to a caregiver who can provide information regarding the child’s performance on typical activities and routine tasks. The PEDI is composed of three parts, the first of which is used to evaluate skills grouped into three functional domains: self-care (73 items), mobility (59 items) and social function (65 items). Each item is scored either zero (not part of the child’s repertoire) or 1 (part of the child’s repertoire). The scores are then totalled per domain.56–58
Wechsler Intelligence Scale for Children
The Wechsler Intelligence Scale (WISC) was developed for the assessment of the intellectual performance of adults. The WISC was developed as a version for children, which was followed by the revised version, WISC-R. The WISC III is the third version of the scale for children and is used to assess intellectual capacity using 13 subtests, 12 from earlier versions and one additional subtest. The subtests are organised into two groups (verbal and perceptive-motor or execution) and are administrated in alternating order. The verbal subtests are Information, Similarities, Arithmetic, Vocabulary, Comprehension and Digits. The execution group is composed of Matrix Reasoning, Coding, Figure Weights, Block Design, Picture Concepts, Symbol Search and Mazes. Many studies have been conducted and, although improvements have been made with the addition of new items, the fundamental characteristics of the WISC and WISC-R remained the same in WISC III.59
Procedures
Intervention protocol
The therapeutic intervention will consist of a combination of tDCS and VR during reaching movements. The protocol will follow safety procedures described in the literature for the use of tDCS on the paediatric population.29 51 60 Three 20 min sessions of combined therapy (tDCS concomitantly to upper limb motor training) will be held for a total of ten sessions.29 30 39 40
Transcranial direct current stimulation
Stimulation will be administered using a tDCS device (DC-Stimulator NeuroConn, Germany), with three sponge (non-metallic) surface electrodes measuring 25 cm2 (5×5 cm) soaked in saline solution.61 62 The children will be randomly allocated to two types of treatment: (1) active anodal stimulation over the primary cortex bilaterally and (2) sham transcranial stimulation. The two anodal electrodes will be positioned over C3 and C4 of the 10–20 international electroencephalogram system62 and the cathode will be positioned over the right deltoid muscle. This montage will enable the child to receive multiple-monopolar anodal tDCS over the primary motor cortex, specifically the area that manages upper limb motor control, while minimising the effect of cathodal stimulation in the brain.61–63 A current of 1 mA (current density: 0.029 mA/cm2) will be administered over the primary motor cortex for 20 min during upper limb training.29 30 39 41 The stimulator has a button that allows the operator to control the intensity of the current. At the beginning of the session, stimulation will be increased gradually until reaching 1 mA and gradually diminished during the final 10 s of the session. Sham stimulation will consist of the same electrode montage and the stimulator will be switched on for 30 s, giving the child the initial sensation of stimulation, but no current will be administered during the remainder of the session. This is considered a valid control procedure in studies involving tDCS.64 65
Adverse effects
Potential adverse effects of tDCS will be evaluated at the end of each session using a questionnaire administered to the child. The questionnaire will address the perception of symptoms having occurred during the session, such as tingling, a burning sensation, headache, pain at the electrode sites, sleepiness and altered mood. The children will be instructed to answer using a three-point scale. The caregivers and children will also be asked open-ended questions at the beginning of each session regarding the occurrence of headache, scalp pain, burning sensations, redness of the skin, sleepiness, difficulty concentrating and mood swings during periods between sessions.
Virtual reality training protocol
Training sessions will be held three times per week on non-consecutive days. Each session will last 20 min and will involve the use of the XBOX 360TM with the KinectTM motion detector.66 The game entitled ‘Bursting Bubbles’ of the Adventure set of games was chosen based on the potential to stimulate cognitive skills and enhance execution time, motor coordination, attention, concentration, reasoning, memory, persistence and precise movement. The activity will be held in a specific room of the Integrated Human Movement Analysis Laboratory measuring 2.5×4.0 m, with a projection screen (200×150 cm) attached to the wall and stereo speakers to provide adequate visual and auditory stimuli. Initially, the child will be instructed to remain standing at a distance of two to three metres in front of the motion detector to capture the movements better as well as for the estimation of height and calculation of the body mass index. Two mobility training sessions with the use of the Xbox 360 exercises will be performed prior to the onset of the intervention protocol. Records will be made of the number of sessions attended and duration of each session.66–68
Analysis of results
The Shapiro-Wilk test will be used to determine whether the data adhere to the Gaussian curve. Parametric variables will be expressed as mean and SD. Non-parametric variables will be expressed as median and IQR. Effect sizes will be calculated from the differences in means between the preintervention and post-intervention evaluations. The effect size values will be expressed with respective 95% CIs. Either two-way analysis of variance (parametric variables) or the Kruskal- Wallis test (non-parametric variables) will be used for the analysis of the effects of the upper limb motor training activity with active and sham tDCS. Logistic regression models will be created to determine factors predictive of the response to the intervention. For such, movement velocity and total duration of movement will be considered. The response capacity will be defined as a clinically significant increase in performance in comparison to baseline. The independent variables will be age (years), sex (male/female), activity of elbow flexors and extensors, cerebral activity (C3 and C4) and functional independence (aspects of self-care). Univariate regressions will be performed for each variable. Based on the initial analyses, the predictors associated with the outcome with a p value≤0.05 will be incorporated into the multivariate model. Moreover, Pearson’s correlation coefficients will be calculated to determine correlations among the variables analysed. A p value<0.05 will be considered indicative of statistical significance. The data will be organised and tabulated with the aid of the Statistical Package for the Social Sciences (SPSS V.19.0).
Discussion
Upper limb motor control enables individuals to perform functional activities. VR will be used as a therapeutic tool to enhance motor control.29 30 Moreover, a non-invasive brain stimulation method (tDCS) will be employed to facilitate motor cortical excitability in the areas subjacent to stimulation to enhance the effects of motor control and learning.37–42 Lazzari et al 37 demonstrated the efficacy of the combination of tDCS and VR in potentiating motor effects on balance and functional mobility in children with cerebral palsy.37
This document offers a detailed description of a randomised, controlled, double-blind, clinical trial designed to determine the effectiveness of VR training combined with tDCS on upper limb movements in individuals with DS.
Ethical aspects and divulgation
The present study is in compliance with the guidelines regulating studies involving human subjects established by the Brazilian National Board of Health in October 1996 and updated in Resolution 466 in 2012. The study will be developed at the Integrated Human Movement Analysis Laboratory of University Nove de Julho (Sao Paulo, Brazil) and has received approval from the Human Research Ethics Committee of the university under process number 1.517.470 (see online supplementary appendix 1). The protocol has been registered with clinical trials. All legal guardians will receive clarifications regarding the procedures and will be aware that participation is voluntary, free of cost and experimental. Those who agree to their child’s participation will sign a statement of informed consent (see online supplementary appendix 2). The guardians will be assured of access to all information and will be informed of the possibility of dropping out of the study or withdrawing consent at any time with no negative consequences. The anonymity of the children and the confidentiality of their information will be ensured, following the ethical principles of privacy. The findings will be published and will contribute evidence regarding the use of transcranial direct current stimulation combined with upper limb motor training in this population.
bmjopen-2017-016260supp001.pdf (293.4KB, pdf)
bmjopen-2017-016260supp002.pdf (113.5KB, pdf)
Supplementary Material
Acknowledgments
The authors gratefully acknowledge financial support from the Brazilian fostering agencies Foundation for Research Support (FAPESP - 2016 / 11156-0), Coordination for the Improvement of Higher Education Personnel (CAPES) and the National Council forS cientific and Technological Development (CNPq).
Footnotes
Funding: The authors gratefully acknowledge financial support from the Brazilian fostering agencies Foundation for Research Support (FAPESP - 2016 / 11156-0).
Competing interests: None declared.
Patient consent: Guardian consent obtained.
Ethics approval: Ethics committee of University Nove de Julho.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1. Moreira LMA, El-Hani CN, Gusmão FAF. A síndrome de down e sua patogênese: considerações sobre o determinismo genético. Revista Brasileira de Psiquiatria 2000;22:96–9. 10.1590/S1516-44462000000200011 [DOI] [Google Scholar]
- 2. Lewanda AF, Matisoff A, Revenis M, et al. . Preoperative evaluation and comprehensive risk assessment for children with down syndrome. Paediatr Anaesth 2016;26:356–62. 10.1111/pan.12841 [DOI] [PubMed] [Google Scholar]
- 3. Moreira LMA, Gusmão FAF. Aspectos genéticos e sociais da sexualidade em pessoas com síndrome de down. Revista Brasileira de Psiquiatria 2002;24:94–9. 10.1590/S1516-44462002000200011 [DOI] [Google Scholar]
- 4. Silva M, Kleinahas ACS. Cognitive processes and brain plasticity in down syndrome. Revis. Bras. Edc. Esp 2006;12:123–38. [Google Scholar]
- 5. Luria AR, Tsvetkova LS. The programming of constructive activity in local brain injuries. Neuropsychologia 1964;2:95–107. 10.1016/0028-3932(64)90015-6 [DOI] [Google Scholar]
- 6. Flórez BJ, Troncoso VM. Síndrome De Down y educacíon. 3. reimp. Barcelona: Masson – Salvat Medicina y Santander 1997. [Google Scholar]
- 7. Santos APM, Weiss LI, Almeida GMF. Assessment and intervention in the motor development of a child with down syndrome. Rev Bras Ed Esp Marília 2010;16:19–30. [Google Scholar]
- 8. Pandilla MM. Pyramidal cell abnormalities in the motor cortex of a child with Down's syndrome: a golgi study. Journal of Comparative Neurology;197:63–81. [DOI] [PubMed] [Google Scholar]
- 9. Seaman J, Depauw KP. The new adapted physical education. California, Mayfield 1982. [Google Scholar]
- 10. Block ME. Motor development in children with Down syndrome: a review of the literature. Adapted Physical Activity Quarterly 1991;8:179–209. 10.1123/apaq.8.3.179 [DOI] [Google Scholar]
- 11. Bomono LMM, Rosseti CB. Aspects in perceptual-motor development and sensory-motor intelligence in down syndrome. Rev bras crescimento desenvolv hum 2010;3:723–34. [Google Scholar]
- 12. Connolly KJ. Skill development: problems and plan : CONNOLLY K.J, Mechanisms of motor skill development. London: Academic Press, 1970. [Google Scholar]
- 13. Gimenez R, Stefanoni FF, Farias PB. Relação entre a capacidade de sincronização temporal e as habilidades motoras rebater e receber em indivíduos portadores da síndrome de Down. Revista Brasileira de Ciência e Movimento. [Google Scholar]
- 14. Schwartzman JS. Síndrome De Down. São Paulo: Editora Mackenzie, 1999. [Google Scholar]
- 15. Marconi NF, Almeida GL. Principles for learning horizontal-planar arm movements with reversal. J Electromyogr Kinesiol 2008;18:771–9. 10.1016/j.jelekin.2007.09.005 [DOI] [PubMed] [Google Scholar]
- 16. Marconi NF, Almeida GL, Gottlieb GL. Electromyographic and kinetic strategies to control movements. Rev Bras Fisioter 2006;10:1–8. 10.1590/S1413-35552006000100002 [DOI] [Google Scholar]
- 17. Latash ML, Corcos DM. Kinematic and electromyographic characteristics of single-joint movements of individuals with Down syndrome. Am J Ment Retard 1991;96:189–201. [PubMed] [Google Scholar]
- 18. Almeida GL, Corcos DM, Latash ML. Practice and transfer effects during fast single-joint elbow movements in individuals with Down syndrome. Phys Ther 1994;74:1000–12. 10.1093/ptj/74.11.1000 [DOI] [PubMed] [Google Scholar]
- 19. Almeida GL, Corcos DM, Hasan Z. Horizontal-plane arm movements with direction reversals performed by normal individuals and individuals with down syndrome. J Neurophysiol 2000;84:1949–60. [DOI] [PubMed] [Google Scholar]
- 20. Aruin AS, Almeida GL, Latash ML. Organization of a simple two-joint synergy in individuals with down syndrome. Am J Ment Retard 1996;101:256–68. [PubMed] [Google Scholar]
- 21. Aruin AS, Almeida GL. A coactivation strategy in anticipatory postural adjustments in persons with Down syndrome. Motor Control 1997;1:178–91. 10.1123/mcj.1.2.178 [DOI] [Google Scholar]
- 22. Latash ML, Anson JG. What are “normal movements” in atypical populations? Behav Brain Sci 1996;19:55–68. 10.1017/S0140525X00041467 [DOI] [Google Scholar]
- 23. Marconi NF. Controle motor de movimentos de reversão em indivíduos neurologicamente normais e portadores da síndrome de down: O efeito do feedback intrínseco [dissertação]. Campinas (SP): Universidade Estadual de Campinas, 2000. [Google Scholar]
- 24. Ferreira SMS. Modulação da Latência da Musculatura Antagonista Em Indivíduos "neurologicamente normais" e portadores da Síndrome de Down [dissertação]. Rio Claro (SP): Universidade Estadual Paulista, 2000. [Google Scholar]
- 25. Lorenzo SM, Braccialli LMP, Araújo RCT. Realidade virtual como intervenção na Síndrome de down: uma perpectiva de ação na interface educação e saúde. Rev Bras Educ Espc 2015;21:392–6. [Google Scholar]
- 26. Wuang YP, Chiang CS, Su CY, et al. . Effectiveness of virtual reality using Wii gaming technology in children with down syndrome. Res Dev Disabil 2011;32:312–21. 10.1016/j.ridd.2010.10.002 [DOI] [PubMed] [Google Scholar]
- 27. Lin HC, Wuang YP. Strength and agility training in adolescents with Down syndrome: a randomized controlled trial. Res Dev Disabil 2012;33:2236–44. 10.1016/j.ridd.2012.06.017 [DOI] [PubMed] [Google Scholar]
- 28. Mello B, Ramalho T. Use of virtual reality in the physical therapeutic treatment of individuals with down syndrome. Revista Neurociências 2015;23:143–9. 10.4181/RNC.2015.23.01.985.7p [DOI] [Google Scholar]
- 29. Moura RCF, Santos C, Collange Grecco L, et al. . Effects of a single session of transcranial direct current stimulation on upper limb movements in children with cerebral palsy: A randomized, sham-controlled study. Dev Neurorehabil 2017;19:1–8. 10.1080/17518423.2017.1282050 [DOI] [PubMed] [Google Scholar]
- 30. Lazzari RD, Politti F, Santos CA, et al. . Effect of a single session of transcranial direct-current stimulation combined with virtual reality training on the balance of children with cerebral palsy: a randomized, controlled, double-blind trial. J Phys Ther Sci 2015;27:763–8. 10.1589/jpts.27.763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Stagg CJ, Bachtiar V, O'Shea J, et al. . Cortical activation changes underlying stimulation-induced behavioural gains in chronic stroke. Brain 2012;135:276–84. 10.1093/brain/awr313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Miranda PC, Lomarev M, Hallett M. Modeling the current distribution during transcranial direct current stimulation. Clin Neurophysiol 2006;117:1623–9. 10.1016/j.clinph.2006.04.009 [DOI] [PubMed] [Google Scholar]
- 33. Wagner T, Fregni F, Fecteau S, et al. . Transcranial direct current stimulation: a computer-based human model study. Neuroimage 2007;35:1113–24. 10.1016/j.neuroimage.2007.01.027 [DOI] [PubMed] [Google Scholar]
- 34. Liebetanz D, Nitsche MA, Tergau F, et al. . Pharmacological approach to the mechanisms of transcranial DC-stimulation-induced after-effects of human motor cortex excitability. Brain 2002;125:2238–47. 10.1093/brain/awf238 [DOI] [PubMed] [Google Scholar]
- 35. Kuo MF, Unger M, Liebetanz D, et al. . Limited impact of homeostatic plasticity on motor learning in humans. Neuropsychologia 2008;46:2122–8. 10.1016/j.neuropsychologia.2008.02.023 [DOI] [PubMed] [Google Scholar]
- 36. Monte-Silva K, Kuo MF, Thirugnanasambandam N, et al. . Dose-dependent inverted U-shaped effect of dopamine (D2-like) receptor activation on focal and nonfocal plasticity in humans. J Neurosci 2009;29:6124–31. 10.1523/JNEUROSCI.0728-09.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Lazzari RD, Politti F, Belina SF, et al. . Effect of transcranial direct current stimulation combined with virtual reality training on balance in children with cerebral palsy: a randomized, controlled, double-blind, clinical trial. J Mot Behav 2017;49:329–36. 10.1080/00222895.2016.1204266 [DOI] [PubMed] [Google Scholar]
- 38. Grecco LA, Duarte NA, de Mendonça ME, et al. . Effect of transcranial direct current stimulation combined with gait and mobility training on functionality in children with cerebral palsy: study protocol for a double-blind randomized controlled clinical trial. BMC Pediatr 2013;13:168 10.1186/1471-2431-13-168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Dumont AJ, Araujo MC, Lazzari RD, et al. . Effects of a single session of transcranial direct current stimulation on static balance in a patient with hemiparesis: a case study. J Phys Ther Sci 2015;27:955–8. 10.1589/jpts.27.955 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Nitsche MA, Liebetanz D. Schlitterlau a, Henschke U, Friche K, Frommann K, et al. GABAergic modulation of DC stimulation-induced motor cortex excitability shifts in humans . Eur J Neurosco 2004;19:2720–6. [DOI] [PubMed] [Google Scholar]
- 41. Duarte NA, Grecco LA, Galli M, et al. . Effect of transcranial direct-current stimulation combined with treadmill training on balance and functional performance in children with cerebral palsy: a double-blind randomized controlled trial. PLoS One 2014;9:8:e105777 10.1371/journal.pone.0105777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Grecco LA, Duarte NA, Zanon N, et al. . Effect of a single session of transcranial direct-current stimulation on balance and spatiotemporal gait variables in children with cerebral palsy: a randomized sham-controlled study. Braz J Phys Ther 2014;18:419–27. 10.1590/bjpt-rbf.2014.0053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Mendonça ME, Fregni F. Neuromodulação com estimulação cerebral não invasiva: aplicação no acidente vascular encefálico, doença de Parkinson e dor crônica ASSIS RD, Condutas práticas em fisioterapia neurológica. SãoPaulo: Manole, 2012:307–39. [Google Scholar]
- 44. Fregni F, Gimenes R, Valle AC, et al. . A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum 2006;54:3988–98. 10.1002/art.22195 [DOI] [PubMed] [Google Scholar]
- 45. Fregni F, Bossio PS, Brunoni AR. Neuromodulação terapêutica: princípios e avanços da estimulação cerebral não invasiva em Neurologia, reabilitação, psiquiatria e neuropsicologia. São Paulo: Sarvier, 2012. [Google Scholar]
- 46. Fritsch B, Reis J, Martinowich K, et al. . Direct current stimulation promotes BDNF-dependent synaptic plasticity: potential implications for motor learning. Neuron 2010;66:198–204. 10.1016/j.neuron.2010.03.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Reis J, Robertson EM, Krakauer JW, et al. . Consensus: can transcranial direct current stimulation and transcranial magnetic stimulation enhance motor learning and memory formation? Brain Stimul 2008;1:363–9. 10.1016/j.brs.2008.08.001 [DOI] [PubMed] [Google Scholar]
- 48. Antal A, Lang N, Boros K, et al. . Homeostatic metaplasticity of the motor cortex is altered during headache-free intervals in migraine with aura. Cereb Cortex 2008;18:2701–5. 10.1093/cercor/bhn032 [DOI] [PubMed] [Google Scholar]
- 49. Cimolin V, Beretta E, Piccinini L, et al. . Constraint-induced movement therapy for children with hemiplegia after traumatic brain injury: a quantitative study. J Head Trauma Rehabil 2012;27:177–87. 10.1097/HTR.0b013e3182172276 [DOI] [PubMed] [Google Scholar]
- 50. Menegoni F, Milano E, Trotti C, et al. . Quantitative evaluation of functional limitation of upper limb movements in subjects affected by ataxia. Eur J Neurol 2009;16:232–9. 10.1111/j.1468-1331.2008.02396.x [DOI] [PubMed] [Google Scholar]
- 51. Petuskey K, Bagley A, Abdala E, et al. . Upper extremity kinematics during functional activities: three-dimensional studies in a normal pediatric population. Gait Posture 2007;25:573–9. 10.1016/j.gaitpost.2006.06.006 [DOI] [PubMed] [Google Scholar]
- 52. Hermes JH, Freriks B, Merletti R, et al. . Hagg G: SENIAM 8: surface electromyography for the non-invasive assessment of muscles. Roessingh Research and Development 1999. [Google Scholar]
- 53. Rab G, Petuskey K, Bagley A. A method for determination of upper extremity kinematics. Gait Posture 2002;15:113–9. 10.1016/S0966-6362(01)00155-2 [DOI] [PubMed] [Google Scholar]
- 54. Jasper HH. The ten-twenty electrode system of the International Federation Electroencephalogria. Clin Neurophysiol 1958;10:371–5. [PubMed] [Google Scholar]
- 55. Homan RW, Herman J, Purdy P. Cerebral location of international 10-20 system electrode placement. Electroencephalogr Clin Neurophysiol 1987;66:376–82. 10.1016/0013-4694(87)90206-9 [DOI] [PubMed] [Google Scholar]
- 56. Fell J, Axmacher N. The role of phase synchronization in memory processes. Nat Rev Neurosci 2011;12:105–18. 10.1038/nrn2979 [DOI] [PubMed] [Google Scholar]
- 57. Haley S, Coster W, Ludlow L. Inventário de avaliação pediátrica de disfunção: versão brasileira. Tradução e adaptação Cultural. mancini MC. Belo Horizonte:Laboratórios de Atividade e Desenvolvimento Infantil, Departamento de Terapia Ocupacional, Universidade Federal de Minas Gerais, 2000. [Google Scholar]
- 58. Feldman AB, Haley SM, Coryell J. Concurrent and construct validity of the Pediatric Evaluation of Disability Inventory. Phys Ther 1990;70:602–10. 10.1093/ptj/70.10.602 [DOI] [PubMed] [Google Scholar]
- 59. Cruz MBZ. WISC III: Escala De Inteligência Wechsler para crianças: manual. Periodicos eletrônicos em psicologia 2005;4:309. [Google Scholar]
- 60. Grecco LA, de Almeida Carvalho Duarte N, Mendonça ME, et al. . Transcranial direct current stimulation during treadmill training in children with cerebral palsy: a randomized controlled double-blind clinical trial. Res Dev Disabil 2014;35:2840–8. 10.1016/j.ridd.2014.07.030 [DOI] [PubMed] [Google Scholar]
- 61. Gillick BT, Feyma T, Menk J, et al. . Safety and feasibility of transcranial direct current stimulation in pediatric hemiparesis: randomized controlled preliminary study. Phys Ther 2015;95:337–49. 10.2522/ptj.20130565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Grecco LA, Oliveira CS, Duarte NA, et al. . Cerebellar transcranial direct current stimulation in children with ataxic cerebral palsy: A sham-controlled, crossover, pilot study. Dev Neurorehabil 2017;20:142–8. 10.3109/17518423.2016.1139639 [DOI] [PubMed] [Google Scholar]
- 63. Nasseri P, Nitsche MA, Ekhtiari H. A framework for categorizing electrode montages in transcranial direct current stimulation. Front Hum Neurosci 2015;9:54 10.3389/fnhum.2015.00054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Boggio PS, Ferrucci R, Mameli F, et al. . Prolonged visual memory enhancement after direct current stimulation in Alzheimer's disease. Brain Stimul 2012;5:223–30. 10.1016/j.brs.2011.06.006 [DOI] [PubMed] [Google Scholar]
- 65. Lapenta OM, Fregni F, Oberman LM, et al. . Bilateral temporal cortex transcranial direct current stimulation worsens male performance in a multisensory integration task. Neurosci Lett 2012;527:105–9. 10.1016/j.neulet.2012.08.076 [DOI] [PubMed] [Google Scholar]
- 66. Yalon-Chamovitz S, Weiss PL. Virtual reality as a leisure activity for young adults with physical and intellectual disabilities. Res Dev Disabil 2008;29:273–87. 10.1016/j.ridd.2007.05.004 [DOI] [PubMed] [Google Scholar]
- 67. Wuang YP, Chiang CS, Su CY, et al. . Effectiveness of virtual reality using Wii gaming technology in children with down syndrome. Res Dev Disabil 2011;32:312–21. 10.1016/j.ridd.2010.10.002 [DOI] [PubMed] [Google Scholar]
- 68. Lin HC, Wuang YP. Strength and agility training in adolescents with Down syndrome: a randomized controlled trial. Res Dev Disabil 2012;33:2236–44. 10.1016/j.ridd.2012.06.017 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2017-016260supp001.pdf (293.4KB, pdf)
bmjopen-2017-016260supp002.pdf (113.5KB, pdf)