Abstract
Background:
Polycystic ovarian syndrome (PCOS) and hypovitaminosis D are the two most common endocrine disorders in young women leading to many adverse metabolic consequences. We evaluated the correlation of 25-hydroxy Vitamin D (25OHD) with metabolic parameters and insulin resistance in PCOS.
Materials and Methods:
We included 100 PCOS patients (age 18–40 years, duration >6 months) serially, in this cross-sectional study. We excluded patients with past use of insulin sensitizers and hormone therapy. All patients underwent a physical examination, body fat estimation, and a single fasting blood sample was analyzed for the biochemical parameters. The patients were divided into 2 groups as per the 25OHD level: Group 1 (Deficient, <30 ng/mL) and Group 2 (normal). The data were analyzed using appropriate statistical methods, and a P < 0.05 was considered statistically significant.
Results:
The study population had a mean age of 28.6 ± 6.3 years, body mass index (BMI) 30.4 ± 6.1 kg/m2 and body fat of 39.1 ± 13%. A total of 90 women had 25OHD deficiency, and hypovitaminosis D was observed more in younger, obese patients. Patients with hypovitaminosis D had a higher BMI (P = 0.0124), low- high-density lipoprotein (P = 0.0094), calcium (P ≤ 0.0001), and elevated testosterone (P = 0.0412) in comparison with normal 25OHD patients. None of the metabolic parameters showed significant correlation with 25OHD (P > 0.05).
Conclusion:
Hypovitaminosis D is very common in PCOS patients and exacerbates the metabolic abnormalities. It is essential to screen all the PCOS patients for 25OHD deficiency, and further large-scale studies are required to confirm our findings.
Keywords: India, insulin resistance, obesity, polycystic ovarian syndrome, Vitamin D
Introduction
Polycystic ovarian syndrome (PCOS) is the most common endocrine disorder in women of reproductive age group. PCOS is characterized by hyperandrogenic chronic anovulation, dysfunctional uterine bleeding, and altered ovarian morphology.[1] The diagnosis of PCOS implies an increased risk of infertility, endometrial carcinoma, obesity, type 2 diabetes mellitus, dyslipidemia, hypertension, and cardiovascular disease (CVD) in women.[2] Insulin resistance (IR) occurs in approximately 60%–80% of women with PCOS and in 95% of obese women with PCOS.[3] Vitamin D deficiency is reported to be very common in our country across all age groups and is linked with many metabolic disorders.[4] Obesity is considered to be a risk factor for hypovitaminosis D and the association between obesity, hypovitaminosis D and PCOS cohorts has been well established.[5,6,7]
Vitamin D is endowed with pleiotropic effects on a spectrum of intracellular regulatory mechanisms, including insulin metabolism or intrinsic apoptotic pathway, in both classical and nonclassical tissues, such as ovary.[8] Few authors have hypothesized that Vitamin D supplementation might be an interesting, economical, and safe therapeutic approach in PCOS.[9] Vitamin D supplementation leads to an improvement in lean mass, regulation of insulin release, altered insulin receptor expression, and improves insulin sensitivity.[10] Obese PCOS patients have different metabolic consequences when compared with lean PCOS patients.[11] The studies relating to Vitamin D and body composition in PCOS are scanty in India.[12,13] Hence, we conducted this study to determine the correlation of Vitamin D with body composition, IR, metabolic, and androgenic parameters in women with PCOS.
Materials and Methods
Study population
We conducted this cross-sectional study in a tertiary care, teaching hospital located in North India (latitude: 18° 74’). The participants were recruited from the endocrinology/medicine/gynecology clinic of our hospital seen between September and December 2015. We included 100 patients serially with a diagnosis of PCOS (aged between 18–40 years, duration of at least 6 months, premenopausal, and normal thyroid function) in our study. We excluded patients with past use of insulin sensitizers, hormone therapy, calcium, Vitamin D, pregnancy, renal calculi, and use of other drugs that affect the insulin sensitivity (beta blockers, glucocorticoids, and thiazides). The patients were divided into 2 groups for the comparison as per the 25-hydroxy Vitamin D (25OHD) level: Group 1 (Deficient) and Group 2 (Normal).
Study measures
Clinical data were collected from all the participants, including demographic details such as age, menstrual history, dietary history (special reference to the intake of calcium-rich foods such as milk, cheese, paneer, and dark green leafy vegetables), and drug history (hormonal preparations, vitamin supplements, metformin, glucocorticoids, beta blockers, etc.). A detailed general examination was conducted with a special emphasis for identification of xanthelesma, lipodystrophy, acanthosis nigricans, and skin tags. Weight was recorded on a digital weighing scale using OMRON HN 286 (Omron Corporation, Kyoto, Japan with a sensitivity of 100 g), height using a SWWS05 stadiometer (Multicare Company, Delhi, India with a sensitivity of 0.1 cm), and body mass index (BMI) was calculated as weight in kilograms divided height in meters squared.
Body fat percentage was determined in the fasting state at the same time of the day using the Inbody 720 (Inbody, Seoul 06313, Korea) machine. All the subjects reported on an empty bladder without any prior morning exercise for the body fat estimation. The body composition analyzer is based on the principle of bioelectric impedance analysis. Anthropometric data such as height, weight, age, and gender were fed into the instrument. The patients were asked to firmly hold the paddles in both the arms and keep the legs slightly separated. Inbody 720 machine gives quantitative estimate value for various body compartments which equals the weight of each compartment, which is added together gives persons total weight. The fat percentage of the total body weight was calculated in this study using following formula: Percent body fat (%) = Body fat mass (kg)/body weight (kg) × 100.
Study interventions
Fasting venous blood samples were collected after an overnight fast for more than 12 h and analyzed for hematological and biochemical parameters. They include glucose, lipid profile, calcium, phosphorus, alkaline phosphatase, 25OHD, intact parathyroid hormone (iPTH), insulin, luteinizing hormone (LH), follicle stimulating hormone, prolactin, testosterone, dehydroepiandrosterone, and thyroid function tests. Plasma glucose was estimated by the glucose oxidase method, and 25OHD by the chemiluminescence method and the coefficients of variation for all biochemical tests were < 10% in our laboratory. The local Ethics Committee approved the study protocol, and all patients provided written informed consent.
Definitions
We used the Rotterdam criteria for the diagnosis of PCOS.[14] Endocrine society clinical practice guidelines define Vitamin D deficiency as levels of 25OHD <20 ng/mL, Vitamin D insufficiency as 25OHD between 21 and 29 ng/mL and normal as 25OHD level more than 30 ng/mL.[15] In our study, we considered any value, including and above 30 ng/mL as normal and <30 ng/mL as a deficiency. The Vitamin D deficiency is subdivided into mild (25OHD between 10 and 20), moderate (25OHD between 5 and 10), and severe (25OHD <5 ng/mL). All the participants with hypovitaminosis D were given weekly oral Vitamin D of 60,000 units for 12 weeks, followed by monthly once. Overweight, Grade I, Grade II, and Grade III obesity were defined as a BMI of 25–30, 30–35, 35–40 and >40 kg/m2 respectively.[16] In this study, we considered the normal percentage body fat as < 30% as given by the manufacturer's data.
Statistical analysis
Data are presented as mean, standard deviation, and descriptive statistics were used for the data analysis. Unpaired t-test and Chi-square test were used to compare the data between the groups. Spearman's correlation was used to find the association between 25OHD and other clinical parameters. We used nonparametric tests in our study because the sample was derived from a specialized group of PCOS patients. We did not calculate the power of the study and sample size as our study was purely observational in nature. A two-tailed P < 0.05 was considered statistically significant for all the tests. The statistical analysis and graph generation was done using the Graph Pad Prism Software, Version 6 (Graph Pad Software, San Deigo, CA, USA).
Results
The study population (n = 100) had a mean age of 28.6 ± 6.3 years, body weight 74.2 ± 13.2 kg, BMI 30.4 ± 6.1 kg/m2, waist circumference 77.1 ± 6.1 cm, hip circumference 91.1 ± 11 cm, waist: hip ratio 0.86 ± 0.1, and body fat of 39.1 ± 13%. A total of 90 women had 25OHD deficiency and the mean 25OHD in them was 19.1 ± 9.2 ng/mL. Hypovitaminosis D was observed more in younger patients (51 patients had an age of < 30 years). Subdivision of Vitamin D deficiency showed that 46 patients had Vitamin D insufficiency, 40 had mild deficiency, and 4 had a moderate 25OHD deficiency. None of the study participants had severe Vitamin D deficiency. The hypovitaminosis D was observed more in overweight and obese individuals as shown in Figure 1.
Figure 1.
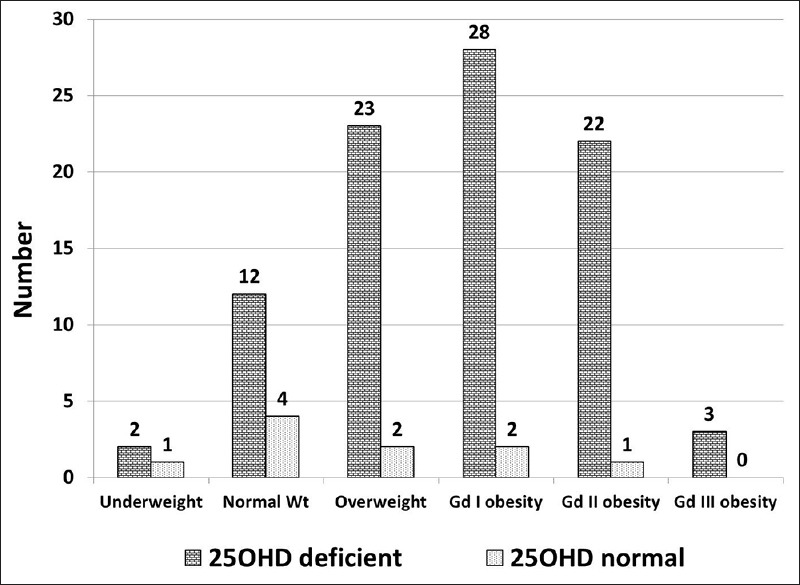
Prevalence of hypovitaminosis D according to the body weight
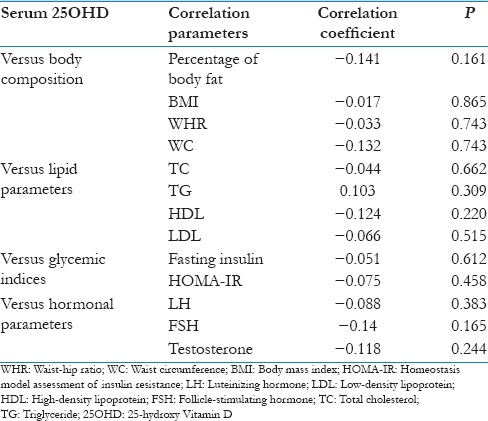
The comparison between the clinical and biochemical parameters between the two groups is given in Table 1. Briefly, the data suggest that the patients with hypovitaminosis D had higher BMI, low- high-density lipoprotein (HDL), calcium and elevated iPTH, testosterone and LH values. The distribution of important metabolic and glycemic parameters, according to the subdivisions of 25OHD value is given in Table 2. We did not compare the groups using analysis of variance for the small sample size in individual groups. Univariate correlation analysis between 25OHD and all the important parameters was given in Table 3. None of the parameters showed any significant correlation with Vitamin D.
Table 1.
Comparison between two groups regarding demographic and biochemical parameters
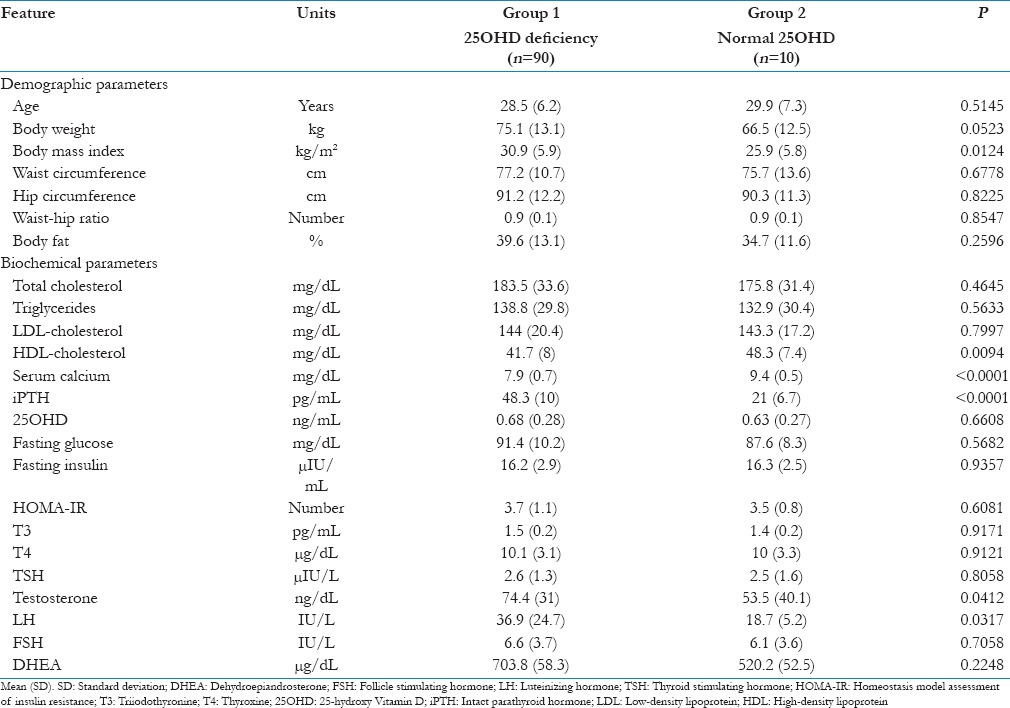
Table 2.
Comparison of metabolic parameters as per the 25-hydroxy Vitamin D level
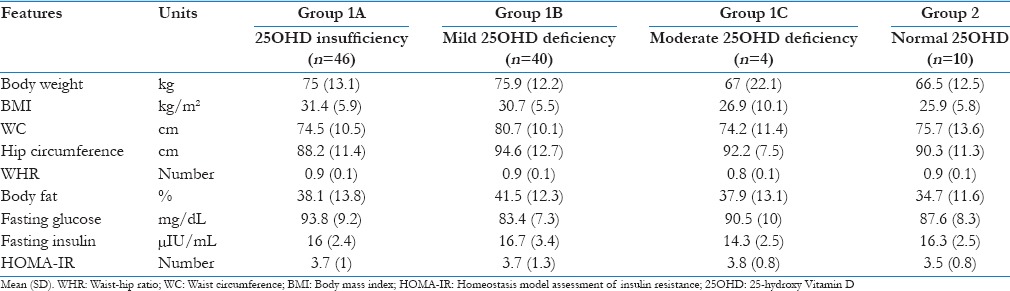
Table 3.
Correlation between 25-hydroxy Vitamin D and various metabolic and biochemical parameters
Discussion
Our study showed that hypovitaminosis D is very common in patients with PCOS and has certain implications. Previous studies have shown that Vitamin D deficiency is seen in 85% of patients with PCOS from developed countries.[17,18] Our data suggest that the Vitamin D replacement should be given to all patients with PCOS to improve their metabolic milieu. The increased prevalence of hypovitaminosis D in obese and overweight patients confirms the known fact about the relationship between them.[5,6,19] However, our study being cross-sectional in nature does not suggest a causal role of Vitamin D deficiency in obesity. Hypovitaminosis D was observed more in younger patients than in older patients. This finding has certain clinical implications in females younger than 30 years of age who are yet to achieve their peak bone mass.[20] The younger patients often overlook the intake of Vitamin D supplements, when compared to elderly patients. The need of taking adequate calcium and Vitamin D should be emphasized more to these vulnerable patients.
The alteration in calcium and iPTH could be explained by the Vitamin D deficiency induced secondary hyperparathyroidism. Previous research has reported the Vitamin D receptor polymorphisms and altered aromatase gene expression in hypovitaminosis D, leading to higher LH levels.[21,22] We did not account for the normal variation of LH during a menstrual cycle, which could be another contributing factor in the high values. An increase in the BMI could explain the associated low HDL and is a part of the metabolic syndrome. Many studies have shown a high prevalence of metabolic syndrome in PCOS patients and their relatives.[23,24]
Our data showed the presence of IR in almost all the study participants. The prevalence of IR is reported in 80% of PCOS patients from our country.[12] The coexisting hypovitaminosis D could exacerbate the metabolic abnormalities in the PCOS, leading to its increased CVD risk. The metabolic parameters did not show any change as per the increasing severity of the 25OHD deficiency. However, the heartening fact of our study is that the majority of the participants had mild forms of 25OHD deficiency and none of the participants had a severe deficiency. This could explain the lack of difference between the 25OHD deficiency and sufficient groups. Previous researchers have shown that a critical level of 25OHD is enough to carry out the metabolic functions without much disruption.[25]
In our study, 25OHD did not show any relation with the clinical, metabolic, and hormonal parameters. Previous research has shown that the 25OHD is inversely correlated with the total cholesterol, triglycerides, body weight, and other metabolic disturbances in PCOS patients.[26] The observed discrepancy in our study could be due to the small sample size and presence of Vitamin D insufficiency in the majority of the patients. The strengths of our study include a detailed evaluation of all the patients and close follow-up in a single center. The limitations of our study include small sample size, cross-sectional design, and lack of evaluation of body fat and IR using robust methods.
Conclusion
Hypovitaminosis D is very common in PCOS patients and exacerbates the metabolic abnormalities. It is essential to screen all the PCOS patients for 25OHD deficiency and institute appropriate replacement therapy to prevent the adverse consequences. Further large-scale studies with more number of patients are required to confirm the findings observed in our study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.March WA, Moore VM, Willson KJ, Phillips DI, Norman RJ, Davies MJ. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25:544–51. doi: 10.1093/humrep/dep399. [DOI] [PubMed] [Google Scholar]
- 2.Alexander CJ, Tangchitnob EP, Lepor NE. Polycystic ovary syndrome: A major unrecognized cardiovascular risk factor in women. Rev Obstet Gynecol. 2009;2:232–9. [PMC free article] [PubMed] [Google Scholar]
- 3.Dunaif A. Insulin resistance and the polycystic ovary syndrome: Mechanism and implications for pathogenesis. Endocr Rev. 1997;18:774–800. doi: 10.1210/edrv.18.6.0318. [DOI] [PubMed] [Google Scholar]
- 4.Ritu G, Gupta A. Vitamin D deficiency in India: Prevalence, causalities and interventions. Nutrients. 2014;6:729–75. doi: 10.3390/nu6020729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yildizhan R, Kurdoglu M, Adali E, Kolusari A, Yildizhan B, Sahin HG, et al. Serum 25-hydroxyvitamin D concentrations in obese and non-obese women with polycystic ovary syndrome. Arch Gynecol Obstet. 2009;280:559–63. doi: 10.1007/s00404-009-0958-7. [DOI] [PubMed] [Google Scholar]
- 6.Hahn S, Haselhorst U, Tan S, Quadbeck B, Schmidt M, Roesler S, et al. Low serum 25-hydroxyvitamin D concentrations are associated with insulin resistance and obesity in women with polycystic ovary syndrome. Exp Clin Endocrinol Diabetes. 2006;114:577–83. doi: 10.1055/s-2006-948308. [DOI] [PubMed] [Google Scholar]
- 7.Wehr E, Pilz S, Schweighofer N, Giuliani A, Kopera D, Pieber TR, et al. Association of hypovitaminosis D with metabolic disturbances in polycystic ovary syndrome. Eur J Endocrinol. 2009;161:575–82. doi: 10.1530/EJE-09-0432. [DOI] [PubMed] [Google Scholar]
- 8.Kinuta K, Tanaka H, Moriwake T, Aya K, Kato S, Seino Y. Vitamin D is an important factor in estrogen biosynthesis of both female and male gonads. Endocrinology. 2000;141:1317–24. doi: 10.1210/endo.141.4.7403. [DOI] [PubMed] [Google Scholar]
- 9.Rashidi B, Haghollahi F, Shariat M, Zayerii F. The effects of calcium-Vitamin D and metformin on polycystic ovary syndrome: A pilot study. Taiwan J Obstet Gynecol. 2009;48:142–7. doi: 10.1016/S1028-4559(09)60275-8. [DOI] [PubMed] [Google Scholar]
- 10.Teegarden D, Donkin SS. Vitamin D: Emerging new roles in insulin sensitivity. Nutr Res Rev. 2009;22:82–92. doi: 10.1017/S0954422409389301. [DOI] [PubMed] [Google Scholar]
- 11.LaZovic G, Radivojevic U, Milicevic S, Spremovic S. Influence of adiposity on leptin, LH and androgen levels in lean, overweight and obese PCOS patients. Int J Fertil Womens Med. 2007;52:82–8. [PubMed] [Google Scholar]
- 12.Kalra A, Nair S, Rai L. Association of obesity and insulin resistance with dyslipidemia in Indian women with polycystic ovarian syndrome. Indian J Med Sci. 2006;60:447–53. [PubMed] [Google Scholar]
- 13.Ramanand SJ, Ramanand JB, Ghongane BB, Patwardhan MH, Patwardhan VM, Ghanghas R, et al. Correlation between serum adiponectin and clinical characteristics, biochemical parameters in Indian women with polycystic ovary syndrome. Indian J Endocrinol Metab. 2014;18:221–5. doi: 10.4103/2230-8210.129116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shroff R, Syrop CH, Davis W, Van Voorhis BJ, Dokras A. Risk of metabolic complications in the new PCOS phenotypes based on the Rotterdam criteria. Fertil Steril. 2007;88:1389–95. doi: 10.1016/j.fertnstert.2007.01.032. [DOI] [PubMed] [Google Scholar]
- 15.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Evaluation, treatment, and prevention of Vitamin D deficiency: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–30. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 16.Seidell JC, Flegal KM. Assessing obesity: Classification and epidemiology. Br Med Bull. 1997;53:238–52. doi: 10.1093/oxfordjournals.bmb.a011611. [DOI] [PubMed] [Google Scholar]
- 17.Selimoglu H, Duran C, Kiyici S, Ersoy C, Guclu M, Ozkaya G, et al. The effect of Vitamin D replacement therapy on insulin resistance and androgen levels in women with polycystic ovary syndrome. J Endocrinol Invest. 2010;33:234–8. doi: 10.1007/BF03345785. [DOI] [PubMed] [Google Scholar]
- 18.Kotsa K, Yavropoulou MP, Anastasiou O, Yovos JG. Role of Vitamin D treatment in glucose metabolism in polycystic ovary syndrome. Fertil Steril. 2009;92:1053–8. doi: 10.1016/j.fertnstert.2008.07.1757. [DOI] [PubMed] [Google Scholar]
- 19.Wehr E, Pieber TR, Obermayer-Pietsch B. Effect of Vitamin D3 treatment on glucose metabolism and menstrual frequency in polycystic ovary syndrome women: A pilot study. J Endocrinol Invest. 2011;34:757–63. doi: 10.3275/7748. [DOI] [PubMed] [Google Scholar]
- 20.Weaver CM, Gordon CM, Janz KF, Kalkwarf HJ, Lappe JM, Lewis R, et al. The National Osteoporosis Foundation's position statement on peak bone mass development and lifestyle factors: A systematic review and implementation recommendations. Osteoporos Int. 2016;27:1281–386. doi: 10.1007/s00198-015-3440-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wehr E, Trummer O, Giuliani A, Gruber HJ, Pieber TR, Obermayer-Pietsch B. Vitamin D-associated polymorphisms are related to insulin resistance and Vitamin D deficiency in polycystic ovary syndrome. Eur J Endocrinol. 2011;164:741–9. doi: 10.1530/EJE-11-0134. [DOI] [PubMed] [Google Scholar]
- 22.Sander VA, Hapon MB, Sícaro L, Lombardi EP, Jahn GA, Motta AB. Alterations of folliculogenesis in women with polycystic ovary syndrome. J Steroid Biochem Mol Biol. 2011;124:58–64. doi: 10.1016/j.jsbmb.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 23.Ranasinha S, Joham AE, Norman RJ, Shaw JE, Zoungas S, Boyle J, et al. The association between polycystic ovary syndrome (PCOS) and metabolic syndrome: A statistical modelling approach. Clin Endocrinol (Oxf) 2015;83:879–87. doi: 10.1111/cen.12830. [DOI] [PubMed] [Google Scholar]
- 24.Cirik DA, Dilbaz B. What do we know about metabolic syndrome in adolescents with PCOS? J Turk Ger Gynecol Assoc. 2014;15:49–55. doi: 10.5152/jtgga.2014.95776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jones G, Strugnell SA, DeLuca HF. Current understanding of the molecular actions of Vitamin D. Physiol Rev. 1998;78:1193–231. doi: 10.1152/physrev.1998.78.4.1193. [DOI] [PubMed] [Google Scholar]
- 26.Krul-Poel YH, Snackey C, Louwers Y, Lips P, Lambalk CB, Laven JS, et al. The role of Vitamin D in metabolic disturbances in polycystic ovary syndrome: A systematic review. Eur J Endocrinol. 2013;169:853–65. doi: 10.1530/EJE-13-0617. [DOI] [PubMed] [Google Scholar]


