Abstract
Context:
It has been well established that the human gut microbiome plays a critical role in the regulation of important biological processes and the mechanisms underlying numerous complex diseases. Although researchers have only recently begun to study the relationship between the gut microbiota and bone metabolism, early efforts have provided increased evidence to suggest an important association.
Evidence Acquisition:
In this study, we attempt to comprehensively summarize the relationship between the gut microbiota and bone metabolism by detailing the regulatory effects of the microbiome on various biological processes, including nutrient absorption and the intestinal mucosal barrier, immune system functionality, the gut–brain axis, and excretion of functional byproducts. In this review, we incorporate evidence from various types of studies, including observational, in vitro and in vivo animal experiments, as well as small efficacy clinic trails.
Evidence Synthesis:
We review the various potential mechanisms of influence for the gut microbiota on the regulation of bone metabolism and discuss the importance of further examining the potential effects of the gut microbiota on the risk of osteoporosis in humans. Furthermore, we outline some useful tools/approaches for metagenomics research and present some prominent examples of metagenomics association studies in humans.
Conclusion:
Current research efforts, although limited, clearly indicate that the gut microbiota may be implicated in bone metabolism, and therefore, further exploration of this relationship is a promising area of focus in bone health and osteoporosis research. Although most existing studies investigate this relationship using animal models, human studies are both needed and on the horizon.
We summarized potential mechanisms of gut microbiota on regulation of bone mass, outlined some useful tools for metagenomics, and presented examples of metagenome-wide association studies.
The human body is colonized with rich and diverse microbial communities consisting of bacteria, viruses, fungi, and protozoa, all of which taken together compose the human microbiome (1). The majority of these microorganisms reside in the gastrointestinal tract (gut microbiota), typically lining the mucosal surfaces of the host. These microbes begin to interact with the human body beginning from the earliest stages of life, as it is believed the human fetus may be exposed to the maternal microbiome during gestation and development (2). After birth, the gut microbiota quickly colonize in the digestive tract, and the microbiome is established within the first few years of life (1, 3). Although the composition of an individual’s gut microbiota usually remains relatively stable in adulthood (1), it may be altered by various factors, including host genetics (4), diet (5), age (6), geography (6), host immune status (7), travel (8), and use of certain medications (9).
It is estimated that >1000 different microbial species exist in the human gut (10). The combination of the unique genomes corresponding to each of these species contributes to the gut metagenome, which is estimated to contain >450 times the number of genes as the human genome (11). Previous association studies have shown that the gut microbiome is involved in the regulation of a wide variety of biological processes, including gut physiology (12), nutrient production and absorption (13), host growth (1), energy balancing (14), metabolic functions (15), immune-system functions (16), brain–behavior systems (17), and inflammatory processes (12). Additionally, differences in the composition of the gut microbiota have been found to be associated with the pathogenesis of several complex human diseases, including obesity (18), irritable bowel syndrome (19), type 1 (20) and type 2 diabetes (21), colorectal cancer (22), Parkinson’s disease (23), transient ischemic attack (24), and rheumatoid arthritis (25).
Although the number of studies assessing the impact of the microbiota composition on bone metabolism is still very limited, the current findings suggest a potentially intriguing and complex relationship that warrants further examination to determine the specific mechanisms by which the microbiome may modulate bone physiology. In this review, we comprehensively summarize the existing evidence of the connections between the gut microbiota and bone processes and discuss the potential pathways of functional influence. Whereas some aspects of the microbiota–bone relationship have previously been reviewed (26), we also contribute some useful bioinformatics methodology and approaches for metagenomics research and provide perspectives for future explorations of the role of the gut microbiota in the etiology of osteoporosis.
Evidence of the Association Between Gut Microbiota and Bone
Intestinal bacterial overgrowth and bone loss
The earliest evidence of a relationship between the gut microbiota and bone metabolism reported that bone mineral density (BMD) was associated with intestinal bacterial overgrowth. In a prospective cohort study, Di Stefano et al. (27) found that bone loss at the site of the lumbar spine and femoral neck was associated with intestinal bacterial overgrowth, indicating overgrowth of the gut microbiota may be an important risk factor in osteopenia/osteoporosis. These findings were further validated by Stotzer et al. (28), who observed that individuals with intestinal bacterial overgrowth had significantly lower BMD in the lumbar spine and the femoral neck. In contrast to these findings, a separate study in a small elderly cohort (∼80 years old) found no differences in femoral BMD between intestinal bacterial overgrowth subjects and controls (29). However, we hypothesize that the negative results may be attributed to the overwhelming effects of other bone-related factors (e.g., extremely low physical activity) and comorbidities that are common to elderly populations.
We note that intestinal bacterial overgrowth has also been shown to be associated with malabsorption, which can influence the metabolism of essential elements for bone processes such as calcium, carbohydrates, vitamin B, and vitamin K (30). Although malabsorption is known to be an important risk factor for bone-related diseases, the effect of bacterial overgrowth on bone loss is not solely accounted for by this deficiency. In the Stotzer et al. study (28), serum-ionized calcium and 1,25-dihydroxyvitamin D3 levels in the bacterial overgrowth individuals were measured to be at normal levels, suggesting that the observed osteoporosis/osteopenia is most likely mediated by several different mechanisms.
Experiments with germfree animals
Germfree mice, born and raised under sterile conditions and therefore largely free of microbiota, are useful tools to study the effects of the gut microbial communities on host physiology. Sjogren et al. (31) reported that 7-week-old female germfree mice had increased BMD and a reduced number of osteoclasts compared with conventionally raised mice. More importantly, colonization of the germfree mice with a normal gut microbiota reduced bone mass, suggesting that the absence of gut microbiota may be responsible for the elevated BMD in the germfree mice. In contrast, Schwarzer et al. (32) showed that 8-week-old male germfree mice had significantly decreased bone growth characteristics, including femur length, cortical thickness, and cortical/trabecular bone fraction of the femur compared with wild-type mice that had normal gut colonization.
We hypothesize that the conflicting findings from these studies may be attributed to the different genetic profiles of the C57BL/6J mice used in Sjogren et al. (31) and the BALB/c mice used in Schwarzer et al. (32). It has been well documented that the immune reactions of these two mouse strains to gut bacteria are very different. For example, innate secretory immunoglobulin A, a core fundamental molecule for intestinal immune homeostasis, has been shown to be elevated in BALB/c mice compared with C57BL6/J mice in both feces and serum samples (33). In the same study, it was shown that, when challenged by Typhimurium aroA, the level of Salmonella-specific IgA in the feces was similar for both strains, although the level in the serum was much less in BALB/c mice (33). Collectively, these differences in immune responses may result in differences in the levels of cytokines critical for bone metabolism such as tumor necrosis factor (TNF)-α and interleukin (IL)-6, potentially leading to differences in BMD.
Evidence from prebiotics, probiotics, and antibiotics studies
Prebiotics are nondigestible food ingredients that benefit host health by modification of the composition and activities of the gut microbiota (34). Essentially, prebiotics include certain types of plant fiber that reside inside the gastrointestinal tract and provide nourishment for the healthy bacteria of the gut. They are mainly known for their role in digestive processes; however, common prebiotics such as galactooligosaccharide, inulin, and resistant starch have also been shown to promote mineral (calcium, magnesium, and zinc) absorption (35, 36), a process that has an important impact on the regulation of BMD and the prevention of bone loss (37).
In contrast, probiotics are living microorganisms that have important positive health effects on the host, particularly by acting on the digestive system (38). The effects of several important probiotics, mainly Lactobacillus and Bifidobacteria, on the regulation of BMD have been studied in both animal models and humans. Treating mice with the probiotic Lactobacillus reuteri was shown to significantly decrease osteoclastogenesis and bone resorption, preventing bone loss in a mouse model (39). Similar results have also been observed for other Lactobacillus strains, such as Lactobacillus rhamnosus and Lactobacillus paracasei, among others (40–42).
Narva et al. (42) identified Lactobacillus helveticus fermented milk to have an acute positive effect on calcium metabolism. Although milk has long been known to contain nutritional elements that are beneficial for bone health, there have not been any current studies exploring whether milk consumption benefits bone metabolism by altering the composition of the gut microbiota. However, it has been shown that some types of gut bacteria may aid in the breakdown of proteins contained in milk to biologically active peptides (43), suggesting that the gut bacteria may regulate the beneficial effects from milk consumption on bone metabolism. Taken together, all the evidence suggests that improved bone metabolism may be included among the many protective health benefits associated with probiotics.
It is also well known that antibiotic treatments have the ability to perturb the composition of the gut microbiota. Cho et al. (9) treated mice with four types of antibiotic regimens (penicillin, vancomycin, penicillin plus vancomycin, and chlortetracycline) and demonstrated that altering the composition of the microbiome by antibiotic treatment can significantly affect bone growth during early life development. Several additional antibiotics studies have supported these findings, indicating that the gut microbiota may impact bone growth through changes in metabolic hormones as well as the regulation of hepatic gene expression (44, 45).
Potential Mechanisms for Gut Microbiota Affecting Bone Metabolism
There are several potential mechanisms by which the gut microbiota may influence biological processes important for human health, and in the subsequent sections we aim to outline the current findings as they relate to bone metabolism.
Influence on nutrient absorption and the intestinal mucosal barrier
Among the extensive variety of bacterial species in the gut microbiota, many can influence the processes of nutrient absorption. For instance, an elevated concentration of the probiotics L. reuteri and Bifidobacterium longum in the gut may increase BMD by promoting mineral (calcium, magnesium, and phosphate) absorption (46). It has also been shown that the composition of the gut microbiota can influence the pH level of the gut (47), an important factor for nutrient absorption, especially calcium (48). Other studies have shown that the gut microbiota aid in the breakdown of macromolecules to smaller components that can be more easily absorbed, an important feature for both bone health and human metabolism more generally (49). Moreover, these microorganisms play an essential role in the synthesis of vitamin B and vitamin K as well as the metabolism of bile acids (50). It is well known that vitamins B and K are critical for the regulation of bone health (51, 52), and that various bile acids may play key roles in the control of calcium absorption. For example, it has been shown that ursodeoxycholic acid promotes calcium absorption, whereas deoxycholic acid inhibits calcium absorption (53).
Nutrient absorption may also be influenced by host diet, which can in turn impact the composition of the microbial profile. The intake of carbohydrates and other nutrients provides energy for the survival of the gut bacteria; however, the composition of the diet can have important effects on the microbial community. High-calorie diets are associated with a reduction in the Bacteriodetes/Firmicutes ratio (54), which can lead to metabolic disturbance of the host. In contrast, low-calorie diets increase the concentration of harmful substances in the intestinal tract (55), which may also have negative consequences for host health. Although adequate protein intake provides necessary elements for bone growth, an excess of protein in the diet may also lead to an elevated level of toxins in the intestinal tract, such as hydrogen sulfide and methane (56). Therefore, it is crucial to maintain a balanced diet and adequate carbohydrate/protein ratio because dietary intake can lead to meaningful alterations in the gut microbiota, thereby influencing bone metabolic processes.
The relationship between the gut microbiota and the intestinal mucosal barrier is quite complex, as they are known to codevelop and work together to engage foreign pathogens (57). Hamilton et al. (58) demonstrated that a change in the composition of the gut microbiota induced an increase in intestinal permeability, which could result in metabolic disorders. The dysfunction of the intestinal mucosal barrier may lead to an increase in serum levels of lipopolysaccharide (LPS), which could in turn increase membrane permeability, resulting in metabolic endotoxemia (59). Early studies have suggested that LPS promotes the survival of osteoclasts in vitro (60, 61); however, most studies have used much higher dosages of LPS than what is necessary to induce metabolic endotoxemia, and therefore it is unclear whether endotoxemia would influence bone mass in vivo.
Influence on immune system
In order for the immune system to adequately function, the human body must be able to distinguish between the molecules that are normally present in the host environment and the foreign microorganisms that are not. Because the gut microbiota are acquired from the environment, they may elicit immune responses at either the local site of the gut or systemically throughout the body. Therefore, the presence of certain species may lead to an increase in a number of cytokines, including many that are associated with bone metabolism, such as TNF-α (31). In support of these findings, Sjogren et al. (31) found that germfree mice had reduced expression of proinflammatory cytokines TNF-α and IL-6. TNF-α is known to stimulate the receptor activator for nuclear factor κB ligand signaling pathway, which may promote bone loss (62), as well as suppress the differentiation of mesenchymal stem cells into osteoblasts, inhibiting bone formation (63).
It has been noted that there is an interesting relationship between bone loss and bariatric surgeries (64). The mechanism of the bone loss induced by bariatric surgeries is not fully understood, although it is believed to include malabsorption and certain immune system factors along with multiple other unknown components (65). In one recent study, it was reported that gut microbiota and proinflammatory cytokines (including TNF-α and IL-6) were altered after sleeve gastrectomy (66). In particular, after treatment by sleeve gastrectomy, the levels of TNF-α and IL-6 were reduced (66). However, bone loss is known to be associated with increased levels of TNF-α and IL-6 rather than decreased levels. Therefore, although it is worth noting, these findings are inconsistent, and it is unclear exactly how bariatric surgeries may impact the microbiome relevant to bone.
Influence on gut–brain axis
In recent years, it has been discovered that the gut microbiota may have important effects on the nervous system through regulation of the synthesis of hormones and neurotransmitters such as serotonin (5-HT) (17). The 5-HT signal transduction system is regarded as an important factor for the regulation of bone development and maintenance. Bliziotes et al. (67) reported that both osteoblast and osteocyte cells contain 5-HT receptors, and that increased 5-HT levels are associated with decreased bone mass in mice. In accordance with this discovery, another study found that decreasing the 5-HT levels with a synthesized molecular inhibitor was able to prevent ovariectomized-induced bone loss in mice (68). Additionally, Sjogren et al. (31) showed that germfree mice had decreased 5-HT levels and increased trabecular bone volume/tissue volume. Therefore, the evidence suggests that the gut microbiota may influence bone processes by affecting the levels of metabolic hormones within the body.
Effects by gut microbial excretion byproducts
Microbial byproducts not only help digestion and absorption of nutrients, but also have their own potential function in the regulation of BMD. For example, some short chain fatty acids produced by the gut microbiota (e.g., butyrate) play an important role in bone formation and bone mineralization by influencing the Runx and osteoprotegerin signaling pathways (69, 70). In addition, butyrate reduces osteoclastogenesis by suppressing the receptor activator for nuclear factor κB ligand signaling pathway (71). It is also known that the gut microbiota may influence intestinally derived estrogen such as flavonoids and diethylstilbestrol (72, 73). The decrease of estrogen levels is a major factor contributing to postmenopausal osteoporosis risk, and therefore, the gut microbiota may influence the regulation of bone health by altering levels of nonovarian estrogens.
Additional studies have discovered that short chain fatty acids also might indirectly affect BMD by significantly influencing the function of host endocrine factors that are related to bone metabolism, such as peptide YY and glucagon-like peptide 1 (74). Peptide YY is a gastrointestinal hormone secreted from the endocrine L cells and has been shown to be negatively associated with total body and hip BMD in premenopausal women (75). Glucagon-like peptide 1, an amino acid hormone that is also secreted from the endocrine L cells, has been shown to act as a regulator of bone metabolism by altering the balance between osteoblast and adipocyte differentiation from bone mesenchymal stem cells (76).
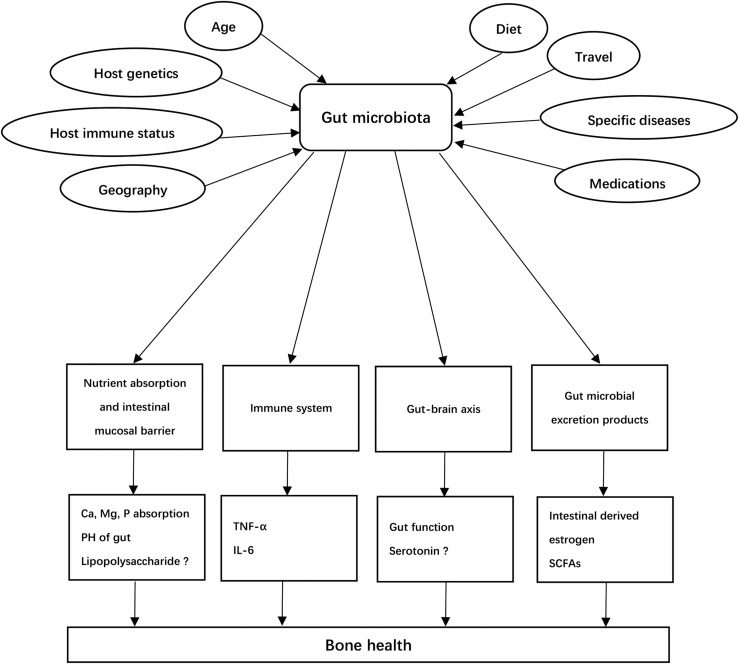
The overall relationship between the gut microbiota and bone metabolism is summarized in Fig. 1.
Figure 1.
Potential etiology of osteoporosis attributed to gut microbiota. Gut microbiota may be altered by various factors, including host genetics, diet, age, geography, host immune status, travel, and use of certain medications. Gut microbiota play important roles on the regulation of bone via nutrition absorbing, changing the permeability of intestinal mucosal barrier, immune system, gut–brain axis, and excrete products. ?, evidence for the effects of 5-HT and LPS on bone is unconfirmed as more studies are needed for validation. PH, potential of hydrogen; SCFA, short chain fatty acid.
Approaches for Metagenomics Analysis
Given the importance of the gut microbiota for bone metabolism and potentially the risk of osteoporosis development, we anticipate a growing number of metagenomic studies in this field throughout the near future. In the following section, we outline some prevailing technical and analytical approaches that are appropriate for gut microbiome studies of complex diseases such as osteoporosis.
Profiling techniques: 16S ribosomal RNA gene sequencing, metagenomic shotgun sequencing, and DNA microarrays
The 16S ribosomal RNA (rRNA) gene sequencing is a highly efficient and cost-effective method for microbiota profiling (77). Bacterial DNA is extracted after sample collection, and either the variable regions or full-length (78) of the 16S rRNA gene are selected for sequencing. Bacterial identification is accomplished using sequence alignment to cluster the microbes into operational taxonomic units (OTUs) based on genetic similarity, which can then be incorporated into statistical association analysis. Although it has been shown that the 16S sequencing approach is fairly powerful to identify microbiota taxa (9), there are some important technical limitations, such as poor taxonomic resolution, low sensitivity making it difficult to detect the rare taxa, and it does not provide information about genomic functional information of the microbes (79).
Recent improvements in next-generation sequencing technology have stimulated the development and application of metagenomic shotgun sequencing for gut microbiome analysis (21, 22, 25). In this approach, DNA is extracted from all microbes in a community, but instead of targeting a specific marker gene for amplification, the entire nucleotide pool is separated into small fragments that are subsequently sequenced. Because this approach does not rely on the single-copy and evolutionary assumptions of marker genes, it can produce a less biased community profile than does 16S rRNA sequencing (80). In addition, metagenomic shotgun sequencing can generally provide higher-resolution descriptions of the microbial compositions, making it possible to identify particular species and even strains of microbes (81). More importantly, sequencing reads sampled from coding sequences can provide insight into the genes and biological functions encoded by specific microbes. Despite these benefits, we note that the necessary bioinformatics tools for metagenomic shotgun sequencing may be considered more challenging compared with that for the 16S rRNA sequencing data, although computational and bioinformatics tools are currently being developed (79–82).
In addition to the aforementioned sequencing-based approaches, the DNA microarray technique is also commonly used. There are many advantages to DNA microarray–based analysis, including high-throughput capabilities, cost effectiveness, as well as relative ease and quickness. DNA microarrays for the analysis of the gut microbiota, such as Human Intestinal Tract Chip (HITChip), are composed of oligonucleotide probes contained in known gene catalogs (20). Although both simple and convenient, the major disadvantage of the microarray approach is that it is based on complementary DNA hybridization, and therefore it is not possible to study uncultured/unknown strains of bacteria.
The strengths and weaknesses of the aforementioned three approaches are outlined in Table 1.
Table 1.
Strengths and Weaknesses of the Three Main Approaches in Metagenome-Wide Association Study
| Features | 16S rRNA Sequencing | Metagenomic Shotgun Sequencing | DNA Microarrays |
|---|---|---|---|
| Strengths | 1. Identify speciesa/genusb of bacteria, including known and novel speciesa/genusb of bacteria | 1. Detect very low-abundance microbes | 1. High throughput |
| 2. Highly efficient and cost effective | 2. Provide information at all taxonomic levels | 2. Quickness | |
| 3. Provide potential functional information of gut microbiota | 3. Direct phylogenetic identification | ||
| 4. Cost effective | |||
| Weaknesses | 1. Poor taxonomic resolution | 1. Expensive | 1. Can’t find novel species/strains |
| 2. Can’t detect very low-abundance microbes | 2. Need complex analysis tools to generate all the data | 2. Cross-hybridization and hybridization | |
| 3. Can’t provide biological functional information of gut microbiota |
Full-length 16S rRNA gene sequencing can pinpoint to species.
16S rRNA amplicon sequencing can pinpoint to genus, not species.
Data analysis workflow
The data analysis workflow for 16S rRNA sequencing and metagenomic shotgun sequencing data have been extensively reviewed (21, 79, 83, 84), and we summarize several commonly used computational tools for metagenomic data analysis in Table 2.
Table 2.
Bioinformatics Tools for 16S rRNA Gene–Sequencing and Metagenomic Shotgun–Sequencing Studies
| Tools | Main Function | Reference | Web Site/Note |
|---|---|---|---|
| DADAa | Denoising | 107 | sites.google.com.site/dadadenoiser |
| Denoisera | Denoising | 108 | qimme.org |
| ChimeraSlayera | Chimera detection | 109 | microbiomeutil.sourceforge.net |
| DECIPHERa | Chimera detection | 110 | decipher.cee.wisr.edu |
| UCLUSTa | OTU clustering | 111 | www.drive5.com/usearch |
| CD-HIT-OTUa | OTU clustering | 112 | weizhing-laboratory.ucsd.edu/cd-hit/otu |
| Mothura | ALL three stepsc | 113 | Mothur.org |
| QIIMEa | ALL three stepsc | 114 | qimme.org |
| MetaIDBAb | Assembly | 115 | For short reads (75–150 bp) |
| MEGANb | Binning | 116 | Compositional-based |
| PhyloPythiaSb | Binning | 117 | Similarity-based |
| MetaClusterb | Binning | 118 | Both compositional-based and similarity-based |
| MetaGeneAnnotatorb | Functional annotation | 119 | metagene.nig.ac.jp/ |
| METAREPb | Functional annotation | 120 | jcvi.org/metarep/ |
Bioinformatics tools for 16S rRNA gene–sequencing studies.
Bioinformatics tools for Metagenomic shotgun–sequencing studies.
All three steps included in the general workflow of analysis for 16S rRNA gene–sequencing data.
Briefly, the bacterial identification and classification of 16S rRNA sequencing data involve three main steps. The first step includes raw sequencing data processing, quality filtering, de-noising, removing artificial chimera sequences, and data normalization. The second step involves global alignment for sequence taxonomy, clustering by OTUs, and building phylogenetic trees. The final step involves statistical analyses of the OTU taxa to measure α- and β-diversity, the microbial diversity within and between samples, respectively.
Similarly, in metagenomic shotgun sequencing, the process begins with raw sequencing data processing, quality filtering, sequence alignment, removal of human sequencing reads, and de novo assembly of the metagenome. This is followed by taxonomic assignment, which is typically accomplished by comparing metagenomic reads with a database of taxonomically informative marker genes and using sequence or phylogenetic similarity to taxonomically characterize metagenomic homologs. Finally, functional information of the microbes is provided by annotating predicted genes or sequencing reads into orthologous gene families and metabolic pathways using various databases such as the Kyoto Encyclopedia of Genes and Genomes, Clusters of Orthologous Groups (85), eggNOG (86), Pfam (87), and TIGRFAMs (88).
Although the current bioinformatics tools for metagenomic data certainly have utility, there are still several crucial limitations that leave much room for improvement. Although new approaches that can accommodate the types of analytical challenges common to metagenomic data are rapidly evolving, there is a necessity for an increased focus on methodology development.
Perspective
In this section, we offer some suggestions for research areas that may provide further insight into the relationship between the gut microbiome and bone metabolism. Although to date there are no current metagenome-wide association studies (MGWASs), multiomics, or translational studies specific to osteoporosis, there are promising examples for other complex diseases that may serve as a guide for future bone research.
Metagenomics studies for osteoporosis
With increasingly reduced costs of high-throughput sequencing, MGWASs, which aim to identify the association between the relative abundances of taxonomic units in a metagenome (e.g., gut microbiome) and a phenotypic trait, are quickly becoming more popular. Recent MGWASs have not only shed insights into the pathophysiological mechanisms of a number of complex human diseases, but have also shown the effective power to distinguish between cases and healthy controls based on the composition of the gut microbiota. Qin et al. (21) conducted a two-stage MGWAS using metagenomic shotgun sequencing and identified several type 2 diabetes–associated biomarkers. In another study, Zhang et al. (25) used a metagenomic shotgun-sequencing approach to show that the composition of the gut microbiota in rheumatoid arthritis cases was significantly different from the microbial profile of healthy controls.
MGWAS for osteoporosis holds great promise in providing findings regarding the specific microbial features that are involved in the underlying biological mechanisms of osteoporosis. There is a necessity for studies in humans to further examine the impact of the gut microbiota composition on the risk of osteoporosis. We caution that the incidence and mechanisms of osteoporosis in females are largely different from that in males, and thus, sex specificity should be taken into consideration when designing future MGWASs for osteoporosis.
Multiomics studies for gut microbiota
Although metagenomic sequencing can provide information about the genetic makeup of the bacteria present in the microbiome, it cannot identify the particular genes that are actively expressed within the metagenome. Recently, there has been an increased focus to develop assays that analyze the RNA, protein expression, and small-molecule metabolites of the gut microbiota to provide useful information regarding meta-transcriptomics, meta-proteomics, and meta-metabolomics (89–91). Integrating multiple layers of omics from the gut microbiota will provide a more comprehensive and systematic understanding of how the microbiota may affect certain aspects of host physiology such as bone metabolism (92).The simultaneous advances in multiomics studies for both humans and the gut microbiota offer opportunities for combining studies of host omics with that of the gut microbiome (93).
Several recent studies have demonstrated that the gut microbiota composition may be influenced by the host genome (94, 95), and there is also a growing appreciation for the role of epigenetic regulation of the host–microbiota interactions (96). Gut-microbial byproducts may affect the methylation status of the host genome and in turn lead to transcriptomic alterations that can modify the risk of phenotypic trait expression (97, 98). Therefore, the gut microbiota may act as an important mediator of host gene environment interactions. Further exploration through integrative multiomics and network analysis studies is needed to elucidate the full range of these interactions as they relate to bone health and other complex human diseases.
Translational potential of gut microbiota
The apparent association between the gut microbiota and bone metabolic processes suggests that the characterization and identification of important gut microbiota features may have great clinical potential. Remarkably, several recent studies (21, 22, 25, 99) have shown that distinctions in the gut microbiota composition may be used to distinguish between individuals having differential disease status with high statistical power based on the area under the curve metric (area under the receiver operating characteristic curve), a common indicator to evaluate sensitivity and specificity (Table 3). Therefore, it is conceivable that the gut microbiota may furnish effective biomarkers in the diagnosis/prognosis of bone diseases and other phenotypic traits.
Table 3.
Examples of the Area Under the Curve of Using Gut Microbiota to Distinguish Patients From Healthy Samples for Complex Diseases
| Disease | AUC | 95% Confidence Interval |
|---|---|---|
| Type 2 diabetes (21) | 0.81 | 0.76–0.85 |
| Obesity (99) | 0.78 | /a |
| Colorectal carcinoma (22) | 0.96 | 0.8788–1.00 |
| Rheumatoid arthritis (25) | 0.9396 | /a |
An AUC of 1 represents a perfect test; an AUC of 0.5 represents a meaningless test.
Abbreviation: AUC, area under the curve of receiver operating characteristic.
No information could be found.
Currently, the majority of therapeutic efforts targeting the gut microbiota have been focused around the preventative/protective effects provided by probiotics and prebiotics (100, 101). These types of treatments have been successfully tested in clinical interventions for dozens of human diseases, including obesity (102), ulcerative colitis (103), atopic diseases of children (104), hypercholesterolemia (105), and autism (106). Narrow-spectrum antibiotics directed toward the gut microbiota and their byproducts are believed to hold great promise as a microbiome-based therapy (100, 101). Although there has not yet been much focus to assess the success of microbiome-based therapies in the treatment of bone-related diseases, it is possible that in the future the identification of those microbiota important for the regulation of bone metabolism may serve as therapeutic targets.
Acknowledgments
Financial Support: This work was partially supported by National Institutes of Health Grants P50AR055081, R01AR057049, R01AR059781, D43TW009107, P20GM109036, R01MH107354, R01MH104680, and R01GM109068 and the Edward G. Schlieder Endowment fund from Tulane University. Y.-C.C. was also partially supported by the National Natural Science Foundation of China (81302228), the Foundation for P Pearl River Nova program of Guangzhou (2014J2200034), and the Foundation from China Scholarship Council (20150322).
Author Contributions: All authors contributed to drafting and revision of the manuscript.
Acknowledgments
Disclosure Summary: The authors have nothing to disclose.
Footnotes
- 5-HT
- serotonin
- BMD
- bone mineral density
- IL
- interleukin
- LPS
- lipopolysaccharide
- MGWAS
- metagenome-wide association study
- OTU
- operational taxonomic unit
- rRNA
- ribosomal RNA
- TNF
- tumor necrosis factor.
References
- 1.Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013;11(4):227–238. [DOI] [PubMed] [Google Scholar]
- 2.Aagaard K, Ma J, Antony KM, Ganu R, Petrosino J, Versalovic J. The placenta harbors a unique microbiome. Sci Transl Med. 2014;6(237):237ra65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Faderl M, Noti M, Corazza N, Mueller C. Keeping bugs in check: the mucus layer as a critical component in maintaining intestinal homeostasis. IUBMB Life. 2015;67(4):275–285. [DOI] [PubMed] [Google Scholar]
- 4.Blekhman R, Goodrich JK, Huang K, Sun Q, Bukowski R, Bell JT, Spector TD, Keinan A, Ley RE, Gevers D, Clark AG. Host genetic variation impacts microbiome composition across human body sites. Genome Biol. 2015;16:191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Graf D, Di Cagno R, Fåk F, Flint HJ, Nyman M, Saarela M, Watzl B. Contribution of diet to the composition of the human gut microbiota. Microb Ecol Health Dis. 2015;26:26164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI. Human gut microbiome viewed across age and geography. Nature. 2012;486(7402):222–227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fiebiger U, Bereswill S, Heimesaat MM. Dissecting the interplay between intestinal microbiota and host immunity in health and disease: lessons learned from fermfree and gnotobiotic animal models. Eur J Microbiol Immunol (Bp). 2016;6(4):253–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Riddle MS, Connor BA. The traveling microbiome. Curr Infect Dis Rep. 2016;18(9):29. [DOI] [PubMed] [Google Scholar]
- 9.Cho I, Yamanishi S, Cox L, Methé BA, Zavadil J, Li K, Gao Z, Mahana D, Raju K, Teitler I, Li H, Alekseyenko AV, Blaser MJ. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature. 2012;488(7413):621–626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nielsen HB, Almeida M, Juncker AS, Rasmussen S, Li J, Sunagawa S, Plichta DR, Gautier L, Pedersen AG, Le Chatelier E, Pelletier E, Bonde I, Nielsen T, Manichanh C, Arumugam M, Batto JM, Quintanilha Dos Santos MB, Blom N, Borruel N, Burgdorf KS, Boumezbeur F, Casellas F, Doré J, Dworzynski P, Guarner F, Hansen T, Hildebrand F, Kaas RS, Kennedy S, Kristiansen K, Kultima JR, Léonard P, Levenez F, Lund O, Moumen B, Le Paslier D, Pons N, Pedersen O, Prifti E, Qin J, Raes J, Sørensen S, Tap J, Tims S, Ussery DW, Yamada T, Renault P, Sicheritz-Ponten T, Bork P, Wang J, Brunak S, Ehrlich SD; MetaHIT Consortium. Identification and assembly of genomes and genetic elements in complex metagenomic samples without using reference genomes. Nat Biotechnol. 2014;32(8):822–828. [DOI] [PubMed] [Google Scholar]
- 11.Li J, Jia H, Cai X, Zhong H, Feng Q, Sunagawa S, Arumugam M, Kultima JR, Prifti E, Nielsen T, Juncker AS, Manichanh C, Chen B, Zhang W, Levenez F, Wang J, Xu X, Xiao L, Liang S, Zhang D, Zhang Z, Chen W, Zhao H, Al-Aama JY, Edris S, Yang H, Wang J, Hansen T, Nielsen HB, Brunak S, Kristiansen K, Guarner F, Pedersen O, Doré J, Ehrlich SD, Bork P, Wang J; MetaHIT Consortium. An integrated catalog of reference genes in the human gut microbiome. Nat Biotechnol. 2014;32(8):834–841. [DOI] [PubMed] [Google Scholar]
- 12.Collins SM. A role for the gut microbiota in IBS. Nat Rev Gastroenterol Hepatol. 2014;11(8):497–505. [DOI] [PubMed] [Google Scholar]
- 13.O’Connor EM. The role of gut microbiota in nutritional status. Curr Opin Clin Nutr Metab Care. 2013;16(5):509–516. [DOI] [PubMed] [Google Scholar]
- 14.Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027–1031. [DOI] [PubMed] [Google Scholar]
- 15.Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489(7415):242–249. [DOI] [PubMed] [Google Scholar]
- 16.Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kennedy PJ, Cryan JF, Dinan TG, Clarke G. Kynurenine pathway metabolism and the microbiota-gut-brain axis. Neuropharmacology. 2017;112(Pt B):399–412. [DOI] [PubMed] [Google Scholar]
- 18.Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, Sogin ML, Jones WJ, Roe BA, Affourtit JP, Egholm M, Henrissat B, Heath AC, Knight R, Gordon JI. A core gut microbiome in obese and lean twins. Nature. 2009;457(7228):480–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lyra A, Rinttilä T, Nikkilä J, Krogius-Kurikka L, Kajander K, Malinen E, Mättö J, Mäkelä L, Palva A. Diarrhoea-predominant irritable bowel syndrome distinguishable by 16S rRNA gene phylotype quantification. World J Gastroenterol. 2009;15(47):5936–5945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Goffau MC, Fuentes S, van den Bogert B, Honkanen H, de Vos WM, Welling GW, Hyöty H, Harmsen HJ. Aberrant gut microbiota composition at the onset of type 1 diabetes in young children. Diabetologia. 2014;57(8):1569–1577. [DOI] [PubMed] [Google Scholar]
- 21.Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, Peng Y, Zhang D, Jie Z, Wu W, Qin Y, Xue W, Li J, Han L, Lu D, Wu P, Dai Y, Sun X, Li Z, Tang A, Zhong S, Li X, Chen W, Xu R, Wang M, Feng Q, Gong M, Yu J, Zhang Y, Zhang M, Hansen T, Sanchez G, Raes J, Falony G, Okuda S, Almeida M, LeChatelier E, Renault P, Pons N, Batto JM, Zhang Z, Chen H, Yang R, Zheng W, Li S, Yang H, Wang J, Ehrlich SD, Nielsen R, Pedersen O, Kristiansen K, Wang J. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55–60. [DOI] [PubMed] [Google Scholar]
- 22.Feng Q, Liang S, Jia H, Stadlmayr A, Tang L, Lan Z, Zhang D, Xia H, Xu X, Jie Z, Su L, Li X, Li X, Li J, Xiao L, Huber-Schönauer U, Niederseer D, Xu X, Al-Aama JY, Yang H, Wang J, Kristiansen K, Arumugam M, Tilg H, Datz C, Wang J. Gut microbiome development along the colorectal adenoma-carcinoma sequence. Nat Commun. 2015;6:6528. [DOI] [PubMed] [Google Scholar]
- 23.Unger MM, Spiegel J, Dillmann KU, Grundmann D, Philippeit H, Bürmann J, Faßbender K, Schwiertz A, Schäfer KH. Short chain fatty acids and gut microbiota differ between patients with Parkinson’s disease and age-matched controls. Parkinsonism Relat Disord. 2016;32:66–72. [DOI] [PubMed] [Google Scholar]
- 24.Yin J, Liao SX, He Y, Wang S, Xia GH, Liu FT, Zhu JJ, You C, Chen Q, Zhou L, Pan SY, Zhou HW. Dysbiosis of gut microbiota with reduced trimethylamine-N-oxide level in patients with large-artery atherosclerotic stroke or transient ischemic attack. J Am Heart Assoc. 2015;4(11):e002699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang X, Zhang D, Jia H, Feng Q, Wang D, Liang D, Wu X, Li J, Tang L, Li Y, Lan Z, Chen B, Li Y, Zhong H, Xie H, Jie Z, Chen W, Tang S, Xu X, Wang X, Cai X, Liu S, Xia Y, Li J, Qiao X, Al-Aama JY, Chen H, Wang L, Wu QJ, Zhang F, Zheng W, Li Y, Zhang M, Luo G, Xue W, Xiao L, Li J, Chen W, Xu X, Yin Y, Yang H, Wang J, Kristiansen K, Liu L, Li T, Huang Q, Li Y, Wang J. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat Med. 2015;21(8):895–905. [DOI] [PubMed] [Google Scholar]
- 26.Hernandez CJ, Guss JD, Luna M, Goldring SR.. Links between the microbiome and bone. J Bone Miner Res. 2016;31(9):1638–1646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Di Stefano M, Veneto G, Malservisi S, Corazza GR. Small intestine bacterial overgrowth and metabolic bone disease. Dig Dis Sci. 2001;46(5):1077–1082. [DOI] [PubMed] [Google Scholar]
- 28.Stotzer PO, Johansson C, Mellström D, Lindstedt G, Kilander AF. Bone mineral density in patients with small intestinal bacterial overgrowth. Hepatogastroenterology. 2003;50(53):1415–1418. [PubMed] [Google Scholar]
- 29.Mitsui T, Shimaoka K, Takagi C, Goto Y, Kagami H, Ito A. Small bowel bacterial overgrowth may not affect bone mineral density in older people. Clin Nutr. 2005;24(6):920–924. [DOI] [PubMed] [Google Scholar]
- 30.Saltzman JR, Russell RM. The aging gut: nutritional issues. Gastroenterol Clin North Am. 1998;27(2):309–324. [DOI] [PubMed] [Google Scholar]
- 31.Sjogren K, Engdahl C, Henning P, Lerner UH, Tremaroli V, Lagerquist MK, Backhed F, Ohlsson C.. The gut microbiota regulates bone mass in mice. J Bone Miner Res. 2012;27(6):1357–1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Schwarzer M, Makki K, Storelli G, Machuca-Gayet I, Srutkova D, Hermanova P, Martino ME, Balmand S, Hudcovic T, Heddi A, Rieusset J, Kozakova H, Vidal H, Leulier F. Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition. Science. 2016;351(6275):854–857. [DOI] [PubMed] [Google Scholar]
- 33.Fransen F, Zagato E, Mazzini E, Fosso B, Manzari C, El Aidy S, Chiavelli A, D’Erchia AM, Sethi MK, Pabst O, Marzano M, Moretti S, Romani L, Penna G, Pesole G, Rescigno M. BALB/c and C57BL/6 mice differ in polyreactive IgA abundance, which impacts the generation of antigen-specific IgA and microbiota diversity. Immunity. 2015;43(3):527–540. [DOI] [PubMed] [Google Scholar]
- 34.Hutkins RW, Krumbeck JA, Bindels LB, Cani PD, Fahey G Jr, Goh YJ, Hamaker B, Martens EC, Mills DA, Rastal RA, Vaughan E, Sanders ME. Prebiotics: why definitions matter. Curr Opin Biotechnol. 2016;37:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Raschka L, Daniel H. Diet composition and age determine the effects of inulin-type fructans on intestinal calcium absorption in rat. Eur J Nutr. 2005;44(6):360–364. [DOI] [PubMed] [Google Scholar]
- 36.Younes H, Coudray C, Bellanger J, Demigné C, Rayssiguier Y, Rémésy C. Effects of two fermentable carbohydrates (inulin and resistant starch) and their combination on calcium and magnesium balance in rats. Br J Nutr. 2001;86(4):479–485. [DOI] [PubMed] [Google Scholar]
- 37.Chonan O, Matsumoto K, Watanuki M. Effect of galactooligosaccharides on calcium absorption and preventing bone loss in ovariectomized rats. Biosci Biotechnol Biochem. 1995;59(2):236–239. [DOI] [PubMed] [Google Scholar]
- 38.Brown AC, Valiere A. Probiotics and medical nutrition therapy. Nutr Clin Care. 2004;7(2):56–68. [PMC free article] [PubMed] [Google Scholar]
- 39.Britton RA, Irwin R, Quach D, Schaefer L, Zhang J, Lee T, Parameswaran N, McCabe LR. Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J Cell Physiol. 2014;229(11):1822–1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li JY, Chassaing B, Tyagi AM, Vaccaro C, Luo T, Adams J, Darby TM, Weitzmann MN, Mulle JG, Gewirtz AT, Jones RM, Pacifici R. Sex steroid deficiency-associated bone loss is microbiota dependent and prevented by probiotics. J Clin Invest. 2016;126(6):2049–2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ohlsson C, Engdahl C, Fåk F, Andersson A, Windahl SH, Farman HH, Movérare-Skrtic S, Islander U, Sjögren K. Probiotics protect mice from ovariectomy-induced cortical bone loss. PLoS One. 2014;9(3):e92368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Narva M, Nevala R, Poussa T, Korpela R. The effect of Lactobacillus helveticus fermented milk on acute changes in calcium metabolism in postmenopausal women. Eur J Nutr. 2004;43(2):61–68. [DOI] [PubMed] [Google Scholar]
- 43.Mohanty DP, Mohapatra S, Misra S, Sahu PS. Milk derived bioactive peptides and their impact on human health: a review. Saudi J Biol Sci. 2016;23(5):577–583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cox LM, Yamanishi S, Sohn J, Alekseyenko AV, Leung JM, Cho I, Kim SG, Li H, Gao Z, Mahana D, Zárate Rodriguez JG, Rogers AB, Robine N, Loke P, Blaser MJ. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell. 2014;158(4):705–721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nobel YR, Cox LM, Kirigin FF, Bokulich NA, Yamanishi S, Teitler I, Chung J, Sohn J, Barber CM, Goldfarb DS, Raju K, Abubucker S, Zhou Y, Ruiz VE, Li H, Mitreva M, Alekseyenko AV, Weinstock GM, Sodergren E, Blaser MJ. Metabolic and metagenomic outcomes from early-life pulsed antibiotic treatment. Nat Commun. 2015;6:7486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rodrigues FC, Castro AS, Rodrigues VC, Fernandes SA, Fontes EA, de Oliveira TT, Martino HS, de Luces Fortes Ferreira CL. Yacon flour and Bifidobacterium longum modulate bone health in rats. J Med Food. 2012;15(7):664–670. [DOI] [PubMed] [Google Scholar]
- 47.Yang LC, Wu JB, Lu TJ, Lin WC. The prebiotic effect of Anoectochilus formosanus and its consequences on bone health. Br J Nutr. 2013;109(10):1779–1788. [DOI] [PubMed] [Google Scholar]
- 48.Palmer MF, Rolls BA. The absorption and secretion of calcium in the gastrointestinal tract of germ-free and conventional chicks. Br J Nutr. 1981;46(3):549–558. [DOI] [PubMed] [Google Scholar]
- 49.Quigley EM. Gut bacteria in health and disease. Gastroenterol Hepatol (N Y). 2013;9(9):560–569. [PMC free article] [PubMed] [Google Scholar]
- 50.Clarke G, Stilling RM, Kennedy PJ, Stanton C, Cryan JF, Dinan TG. Minireview: gut microbiota: the neglected endocrine organ. Mol Endocrinol. 2014;28(8):1221–1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.van Wijngaarden JP, Doets EL, Szczecińska A, Souverein OW, Duffy ME, Dullemeijer C, Cavelaars AE, Pietruszka B, Van’t Veer P, Brzozowska A, Dhonukshe-Rutten RA, de Groot CP. Vitamin B12, folate, homocysteine, and bone health in adults and elderly people: a systematic review with meta-analyses. J Nutr Metab. 2013;2013:486186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Villa JK, Diaz MA, Pizziolo VR, Martino HS. Effect of vitamin K in bone metabolism and vascular calcification: a review of mechanisms of action and evidences [published online ahead of print July 20, 2016]. Crit Rev Food Sci Nutr. doi: 10.1080/10408398.2016.1211616. [DOI] [PubMed]
- 53.Rodríguez V, Rivoira M, Marchionatti A, Pérez A, Tolosa de Talamoni N. Ursodeoxycholic and deoxycholic acids: a good and a bad bile acid for intestinal calcium absorption. Arch Biochem Biophys. 2013;540(1-2):19–25. [DOI] [PubMed] [Google Scholar]
- 54.Vaughn AC, Cooper EM, DiLorenzo PM, O’Loughlin LJ, Konkel ME, Peters JH, Hajnal A, Sen T, Lee SH, de La Serre CB, Czaja K. Energy-dense diet triggers changes in gut microbiota, reorganization of gut‑brain vagal communication and increases body fat accumulation. Acta Neurobiol Exp (Warsz). 2017;77(1):18–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, Tuohy K. Gut microbiota functions: metabolism of nutrients and other food components [published online ahead of print April 9, 2017]. Eur J Nutr. doi: 10.1007/s00394-017-1445-8. [DOI] [PMC free article] [PubMed]
- 56.Li Q, Lauber CL, Czarnecki-Maulden G, Pan Y, Hannah SS. Effects of the dietary protein and carbohydrate ratio on gut microbiomes in dogs of different body conditions. MBio. 2017;8(1):e01703-16.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Guo Y, Zhou G, He C, Yang W, He Z, Liu Z. Serum levels of lipopolysaccharide and 1,3-β-D-glucan refer to the severity in patients with Crohn's disease. Mediators Inflamm. 2015;2015:843089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hamilton MK, Boudry G, Lemay DG, Raybould HE. Changes in intestinal barrier function and gut microbiota in high-fat diet-fed rats are dynamic and region dependent. Am J Physiol Gastrointest Liver Physiol. 2015;308(10):G840–G851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, Bastelica D, Neyrinck AM, Fava F, Tuohy KM, Chabo C, Waget A, Delmée E, Cousin B, Sulpice T, Chamontin B, Ferrières J, Tanti JF, Gibson GR, Casteilla L, Delzenne NM, Alessi MC, Burcelin R. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772. [DOI] [PubMed] [Google Scholar]
- 60.Hou GQ, Guo C, Song GH, Fang N, Fan WJ, Chen XD, Yuan L, Wang ZQ. Lipopolysaccharide (LPS) promotes osteoclast differentiation and activation by enhancing the MAPK pathway and COX-2 expression in RAW264.7 cells. Int J Mol Med. 2013;32(2):503–510. [DOI] [PubMed] [Google Scholar]
- 61.Itoh K, Udagawa N, Kobayashi K, Suda K, Li X, Takami M, Okahashi N, Nishihara T, Takahashi N. Lipopolysaccharide promotes the survival of osteoclasts via Toll-like receptor 4, but cytokine production of osteoclasts in response to lipopolysaccharide is different from that of macrophages. J Immunol. 2003;170(7):3688–3695. [DOI] [PubMed] [Google Scholar]
- 62.Kitaura H, Kimura K, Ishida M, Kohara H, Yoshimatsu M, Takano-Yamamoto T. Immunological reaction in TNF-α-mediated osteoclast formation and bone resorption in vitro and in vivo. Clin Dev Immunol. 2013;2013:181849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kotake S, Nanke Y. Effect of TNFα on osteoblastogenesis from mesenchymal stem cells. Biochim Biophys Acta. 2014;1840(3):1209–1213. [DOI] [PubMed] [Google Scholar]
- 64.Liu C, Wu D, Zhang JF, Xu D, Xu WF, Chen Y, Liu BY, Li P, Li L. Changes in bone metabolism in morbidly obese patients after bariatric surgery: a meta-analysis. Obes Surg. 2016;26(1):91–97. [DOI] [PubMed] [Google Scholar]
- 65.Hage MP, El-Hajj Fuleihan G.. Bone and mineral metabolism in patients undergoing Roux-en-Y gastric bypass. Osteoporosis Int. 2014;25(2):423–439. [DOI] [PubMed] [Google Scholar]
- 66.Liu R, Hong J, Xu X, Feng Q, Zhang D, Gu Y, Shi J, Zhao S, Liu W, Wang X, Xia H, Liu Z, Cui B, Liang P, Xi L, Jin J, Ying X, Wang X, Zhao X, Li W, Jia H, Lan Z, Li F, Wang R, Sun Y, Yang M, Shen Y, Jie Z, Li J, Chen X, Zhong H, Xie H, Zhang Y, Gu W, Deng X, Shen B, Xu X, Yang H, Xu G, Bi Y, Lai S, Wang J, Qi L, Madsen L, Wang J, Ning G, Kristiansen K, Wang W. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23(7):859–868. [DOI] [PubMed] [Google Scholar]
- 67.Bliziotes M, Eshleman A, Burt-Pichat B, Zhang XW, Hashimoto J, Wiren K, Chenu C. Serotonin transporter and receptor expression in osteocytic MLO-Y4 cells. Bone. 2006;39(6):1313–1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yadav VK, Balaji S, Suresh PS, Liu XS, Lu X, Li Z, Guo XE, Mann JJ, Balapure AK, Gershon MD, Medhamurthy R, Vidal M, Karsenty G, Ducy P. Pharmacological inhibition of gut-derived serotonin synthesis is a potential bone anabolic treatment for osteoporosis. Nat Med. 2010;16(3):308–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lee HW, Suh JH, Kim AY, Lee YS, Park SY, Kim JB. Histone deacetylase 1-mediated histone modification regulates osteoblast differentiation. Mol Endocrinol. 2006;20(10):2432–2443. [DOI] [PubMed] [Google Scholar]
- 70.Katono T, Kawato T, Tanabe N, Suzuki N, Iida T, Morozumi A, Ochiai K, Maeno M. Sodium butyrate stimulates mineralized nodule formation and osteoprotegerin expression by human osteoblasts. Arch Oral Biol. 2008;53(10):903–909. [DOI] [PubMed] [Google Scholar]
- 71.Rahman MM, Kukita A, Kukita T, Shobuike T, Nakamura T, Kohashi O. Two histone deacetylase inhibitors, trichostatin A and sodium butyrate, suppress differentiation into osteoclasts but not into macrophages. Blood. 2003;101(9):3451–3459. [DOI] [PubMed] [Google Scholar]
- 72.Chiang SS, Pan TM. Beneficial effects of phytoestrogens and their metabolites produced by intestinal microflora on bone health. Appl Microbiol Biotechnol. 2013;97(4):1489–1500. [DOI] [PubMed] [Google Scholar]
- 73.Flores R, Shi J, Fuhrman B, Xu X, Veenstra TD, Gail MH, Gajer P, Ravel J, Goedert JJ. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites: a cross-sectional study. J Transl Med. 2012;10:253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tang WH, Kitai T, Hazen SL. Gut microbiota in cardiovascular health and disease. Circ Res. 2017;120(7):1183–1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Scheid JL, Toombs RJ, Ducher G, Gibbs JC, Williams NI, De Souza MJ. Estrogen and peptide YY are associated with bone mineral density in premenopausal exercising women. Bone. 2011;49(2):194–201. [DOI] [PubMed] [Google Scholar]
- 76.Luo G, Liu H, Lu H. Glucagon-like peptide-1(GLP-1) receptor agonists: potential to reduce fracture risk in diabetic patients? Br J Clin Pharmacol. 2016;81(1):78–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, Nielsen T, Pons N, Levenez F, Yamada T, Mende DR, Li J, Xu J, Li S, Li D, Cao J, Wang B, Liang H, Zheng H, Xie Y, Tap J, Lepage P, Bertalan M, Batto JM, Hansen T, Le Paslier D, Linneberg A, Nielsen HB, Pelletier E, Renault P, Sicheritz-Ponten T, Turner K, Zhu H, Yu C, Li S, Jian M, Zhou Y, Li Y, Zhang X, Li S, Qin N, Yang H, Wang J, Brunak S, Doré J, Guarner F, Kristiansen K, Pedersen O, Parkhill J, Weissenbach J, Bork P, Ehrlich SD, Wang J; MetaHIT Consortium . A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464(7285):59–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wagner J, Coupland P, Browne HP, Lawley TD, Francis SC, Parkhill J. Evaluation of PacBio sequencing for full-length bacterial 16S rRNA gene classification. BMC Microbiol. 2016;16(1):274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sharpton TJ. An introduction to the analysis of shotgun metagenomic data. Front Plant Sci. 2014;5:209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Weinstock GM. Genomic approaches to studying the human microbiota. Nature. 2012;489(7415):250–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Morgan XC, Huttenhower C. Meta’omic analytic techniques for studying the intestinal microbiome. Gastroenterology. 2014;146(6):1437–1448.e1. [DOI] [PubMed] [Google Scholar]
- 82.Ma J, Prince A, Aagaard KM. Use of whole genome shotgun metagenomics: a practical guide for the microbiome-minded physician scientist. Semin Reprod Med. 2014;32(1):5–13. [DOI] [PubMed] [Google Scholar]
- 83.Andoh A, Nishida A, Takahashi K, Inatomi O, Imaeda H, Bamba S, Kito K, Sugimoto M, Kobayashi T. Comparison of the gut microbial community between obese and lean peoples using 16S gene sequencing in a Japanese population. J Clin Biochem Nutr. 2016;59(1):65–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Brown CT, Davis-Richardson AG, Giongo A, Gano KA, Crabb DB, Mukherjee N, Casella G, Drew JC, Ilonen J, Knip M, Hyöty H, Veijola R, Simell T, Simell O, Neu J, Wasserfall CH, Schatz D, Atkinson MA, Triplett EW. Gut microbiome metagenomics analysis suggests a functional model for the development of autoimmunity for type 1 diabetes. PLoS One. 2011;6(10):e25792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tatusov RL, Fedorova ND, Jackson JD, Jacobs AR, Kiryutin B, Koonin EV, Krylov DM, Mazumder R, Mekhedov SL, Nikolskaya AN, Rao BS, Smirnov S, Sverdlov AV, Vasudevan S, Wolf YI, Yin JJ, Natale DA. The COG database: an updated version includes eukaryotes. BMC Bioinformatics. 2003;4:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Powell S, Szklarczyk D, Trachana K, Roth A, Kuhn M, Muller J, Arnold R, Rattei T, Letunic I, Doerks T, Jensen LJ, von Mering C, Bork P. eggNOG v3.0: orthologous groups covering 1133 organisms at 41 different taxonomic ranges. Nucleic Acids Res. 2012;40(Database issue):D284–D289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, Eddy SR, Heger A, Hetherington K, Holm L, Mistry J, Sonnhammer EL, Tate J, Punta M. Pfam: the protein families database. Nucleic Acids Res. 2014;42(Database issue):D222–D230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Haft DH, Selengut JD, Richter RA, Harkins D, Basu MK, Beck E. TIGRFAMs and genome properties in 2013. Nucleic Acids Res. 2013;41(Database issue):D387–D395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gosalbes MJ, Abellan JJ, Durbán A, Pérez-Cobas AE, Latorre A, Moya A. Metagenomics of human microbiome: beyond 16s rDNA. Clin Microbiol Infect. 2012;18(Suppl 4):47–49. [DOI] [PubMed] [Google Scholar]
- 90.Young JC, Pan C, Adams RM, Brooks B, Banfield JF, Morowitz MJ, Hettich RL. Metaproteomics reveals functional shifts in microbial and human proteins during a preterm infant gut colonization case. Proteomics. 2015;15(20):3463–3473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Vernocchi P, Del Chierico F, Putignani L. Gut microbiota profiling: metabolomics based approach to unravel compounds affecting human health. Front Microbiol. 2016;7:1144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mondot S, Lepage P. The human gut microbiome and its dysfunctions through the meta-omics prism. Ann N Y Acad Sci. 2016;1372(1):9–19. [DOI] [PubMed] [Google Scholar]
- 93.Spor A, Koren O, Ley R. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat Rev Microbiol. 2011;9(4):279–290. [DOI] [PubMed] [Google Scholar]
- 94.Goodrich JK, Waters JL, Poole AC, Sutter JL, Koren O, Blekhman R, Beaumont M, Van Treuren W, Knight R, Bell JT, Spector TD, Clark AG, Ley RE. Human genetics shape the gut microbiome. Cell. 2014;159(4):789–799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang J, Thingholm LB, Skieceviciene J, Rausch P, Kummen M, Hov JR, Degenhardt F, Heinsen FA, Rühlemann MC, Szymczak S, Holm K, Esko T, Sun J, Pricop-Jeckstadt M, Al-Dury S, Bohov P, Bethune J, Sommer F, Ellinghaus D, Berge RK, Hübenthal M, Koch M, Schwarz K, Rimbach G, Hübbe P, Pan WH, Sheibani-Tezerji R, Hasler R, Rosenstiel P, D’Amato M, Cloppenborg-Schmidt K, Künzel S, Laudes M, Marschall HU, Lieb W, Nöthlings U, Karlsen TH, Baines JF, Franke A. Genome-wide association analysis identifies variation in vitamin D receptor and other host factors influencing the gut microbiota. Nat Genet 2016;48(11):1396–1406. [DOI] [PMC free article] [PubMed]
- 96.Alenghat T, Artis D. Epigenomic regulation of host-microbiota interactions. Trends Immunol. 2014;35(11):518–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Putignani L, Dallapiccola B. Foodomics as part of the host-microbiota-exposome interplay. J Proteomics. 2016;147:3–20. [DOI] [PubMed] [Google Scholar]
- 98.Cortese R, Lu L, Yu Y, Ruden D, Claud EC. Epigenome-Microbiome crosstalk: a potential new paradigm influencing neonatal susceptibility to disease. Epigenetics. 2016;11(3):205–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, Grarup N, Jørgensen T, Brandslund I, Nielsen HB, Juncker AS, Bertalan M, Levenez F, Pons N, Rasmussen S, Sunagawa S, Tap J, Tims S, Zoetendal EG, Brunak S, Clément K, Doré J, Kleerebezem M, Kristiansen K, Renault P, Sicheritz-Ponten T, de Vos WM, Zucker JD, Raes J, Hansen T, Bork P, Wang J, Ehrlich SD, Pedersen O; MetaHIT Consortium . Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500(7464):541–546. [DOI] [PubMed] [Google Scholar]
- 100.Sonnenburg JL, Fischbach MA. Community health care: therapeutic opportunities in the human microbiome. Sci Transl Med. 2011;3(78):78ps12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Olle B. Medicines from microbiota. Nat Biotechnol. 2013;31(4):309–315. [DOI] [PubMed] [Google Scholar]
- 102.Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, Okano M, Kagoshima M, Tsuchida T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur J Clin Nutr. 2010;64(6):636–643. [DOI] [PubMed] [Google Scholar]
- 103.Bennet JD, Brinkman M. Treatment of ulcerative colitis by implantation of normal colonic flora. Lancet. 1989;1(8630):164. [DOI] [PubMed] [Google Scholar]
- 104.Kalliomäki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet. 2001;357(9262):1076–1079. [DOI] [PubMed] [Google Scholar]
- 105.Nguyen TD, Kang JH, Lee MS. Characterization of Lactobacillus plantarum PH04, a potential probiotic bacterium with cholesterol-lowering effects. Int J Food Microbiol. 2007;113(3):358–361. [DOI] [PubMed] [Google Scholar]
- 106.Finegold SM, Molitoris D, Song Y, Liu C, Vaisanen ML, Bolte E, McTeague M, Sandler R, Wexler H, Marlowe EM, Collins MD, Lawson PA, Summanen P, Baysallar M, Tomzynski TJ, Read E, Johnson E, Rolfe R, Nasir P, Shah H, Haake DA, Manning P, Kaul A. Gastrointestinal microflora studies in late-onset autism. Clin Infect Dis. 2002;35(Suppl 1):S6–S16. [DOI] [PubMed] [Google Scholar]
- 107.Erten S, Bebek G, Ewing RM, Koyutürk M. DADA: Degree-Aware Algorithms for Network-Based Disease Gene Prioritization. BioData Min. 2011;4:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Reeder J, Knight R. Rapidly denoising pyrosequencing amplicon reads by exploiting rank-abundance distributions. Nat Methods. 2010;7(9):668–669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Haas BJ, Gevers D, Earl AM, Feldgarden M, Ward DV, Giannoukos G, Ciulla D, Tabbaa D, Highlander SK, Sodergren E, Methé B, DeSantis TZ, Petrosino JF, Knight R, Birren BW; Human Microbiome Consortium . Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res. 2011;21(3):494–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wright ES, Yilmaz LS, Noguera DR. DECIPHER, a search-based approach to chimera identification for 16S rRNA sequences. Appl Environ Microbiol. 2012;78(3):717–725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Edgar RC. Search and clustering orders of magnitude faster than BLAST. Bioinformatics. 2010;26(19):2460–2461. [DOI] [PubMed] [Google Scholar]
- 112.Fu L, Niu B, Zhu Z, Wu S, Li W. CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics. 2012;28(23):3150–3152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol. 2009;75(23):7537–7541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kuczynski J, Stombaugh J, Walters WA, Gonzalez A, Caporaso JG, Knight R. Using QIIME to analyze 16S rRNA gene sequences from microbial communities. Curr Protoc Microbiol. 2012;Chapter 1:Unit 1E.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Peng Y, Leung HC, Yiu SM, Chin FY. Meta-IDBA: a de Novo assembler for metagenomic data. Bioinformatics. 2011;27(13):i94–i101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Huson DH, Weber N. Microbial community analysis using MEGAN. Methods Enzymol. 2013;531:465–485. [DOI] [PubMed] [Google Scholar]
- 117.Patil KR, Roune L, McHardy AC. The PhyloPythiaS web server for taxonomic assignment of metagenome sequences. PLoS One. 2012;7(6):e38581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Leung HC, Yiu SM, Yang B, Peng Y, Wang Y, Liu Z, Chen J, Qin J, Li R, Chin FY. A robust and accurate binning algorithm for metagenomic sequences with arbitrary species abundance ratio. Bioinformatics. 2011;27(11):1489–1495. [DOI] [PubMed] [Google Scholar]
- 119.Noguchi H, Taniguchi T, Itoh T. MetaGeneAnnotator: detecting species-specific patterns of ribosomal binding site for precise gene prediction in anonymous prokaryotic and phage genomes. DNA Res. 2008;15(6):387–396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Goll J, Rusch DB, Tanenbaum DM, Thiagarajan M, Li K, Methé BA, Yooseph S. METAREP: JCVI metagenomics reports--an open source tool for high-performance comparative metagenomics. Bioinformatics. 2010;26(20):2631–2632. [DOI] [PMC free article] [PubMed] [Google Scholar]