Abstract
Objective:
To evaluate the implementation of early screening for critical congenital heart defects (CCHDs) in the neonatal intensive care unit (NICU) and potential exclusion of sub-populations from universal screening.
Study Design:
Prospective evaluation of CCHD screening at multiple time intervals was conducted in 21 NICUs across five states (n=4556 infants).
Results:
Of the 4120 infants with complete screens, 92% did not have prenatal CHD diagnosis or echocardiography before screening, 72% were not receiving oxygen at 24 to 48 h and 56% were born ⩾2500 g. Thirty-seven infants failed screening (0.9%); none with an unsuspected CCHD. False positive rates were low for infants not receiving oxygen (0.5%) and those screened after weaning (0.6%), yet higher among infants born at <28 weeks (3.8%). Unnecessary echocardiograms were minimal (0.2%).
Conclusion:
Given the majority of NICU infants were ⩾2500 g, not on oxygen and not preidentified for CCHD, systematic screening at 24 to 48 h may be of benefit for early detection of CCHD with minimal burden.
Introduction
Delayed detection of critical congenital heart defects (CCHD) may result in significant morbidity or mortality, and thus early recognition is crucial.1, 2 Screening using pulse oximetry has become a near-universal tool to aid in early identification of CCHD.3, 4 Many states require screening of all infants irrespective of clinical status or setting, posing unique considerations for implementation in the neonatal intensive care unit (NICU).4
Established CCHD screening algorithms include specified saturation thresholds with recommended timing between 24 to 48 h of age; parameters intended for application to well infants.5, 6 Feasibility of screening for the NICU population has been questioned given complexities such as oxygen requirements and extreme prematurity. Approximately 8% of all newborns receive care in a NICU where frequent physical examination, extended hospitalization and continuous pulse oximetry monitoring are common.7, 8 In this setting, abnormal findings may lead to a diagnosis of CCHD before screening. Lack of specifications for NICU screening and concerns of higher false positive rates (FPRs) have led some units to exclude NICU infants from CCHD screening with the assumption that routine monitoring is sufficient to detect unsuspected CCHD.9 Yet, generalizing a lower risk of undetected CCHD to all NICU infants may be problematic given different levels of NICU care and the sizable population of infants born at ⩾2500 g or ⩾37 weeks who approximate the well-baby population.7, 9
Existing literature on CCHD screening in the NICU in US populations is limited.10, 11, 12, 13 Faced with mandated screening and minimal empiric evidence to guide implementation in the NICU, salient questions have been raised regarding appropriate exclusion criteria, timing, influence of supplemental oxygen and resource utilization. We performed a prospective, multicenter evaluation of CCHD screening in the NICU. Our objectives were to evaluate the feasibility and burden associated with (1) early timing options for screening and (2) exclusion of infants with a prenatal CHD diagnosis, echocardiography conducted before screening or those born at <28 weeks from universal CCHD screening in the NICU.
Methods
Screening protocol
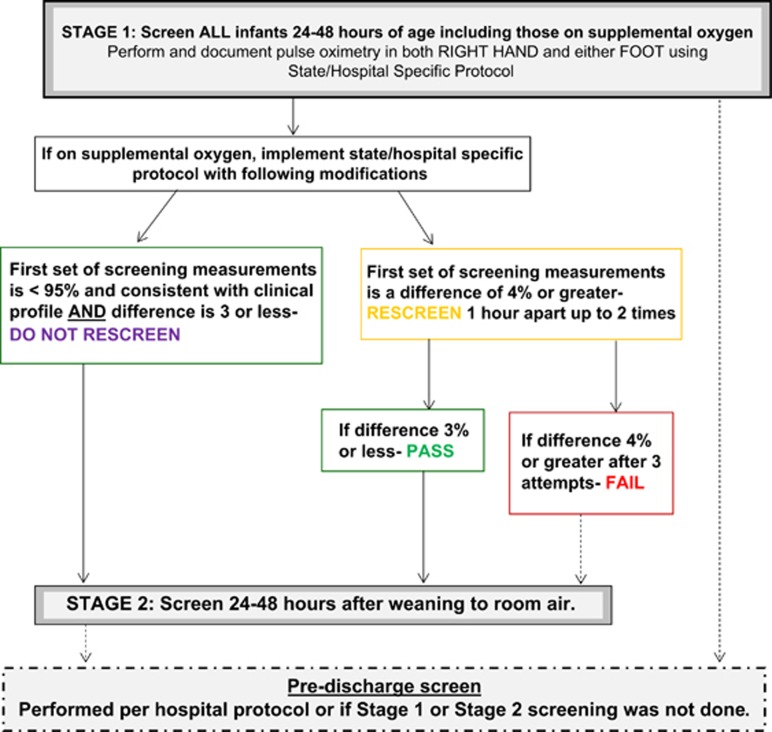
The screening protocol consisted of two stages (Figure 1). Stage 1 was targeted for all infants at 24 to 48 h after admission. Stage 2 was conducted on infants who were receiving supplemental oxygen (any mode of delivery) at 24 to 48 h and was optimally performed within 24 to 48 h after weaning. Two widely referenced CCHD screening algorithms were used in the evaluation. These included the New Jersey (NJ)-recommended algorithm (fail or rescreen criteria: <95% in one extremity (right hand or either foot) and/or >3% differential in saturations)5 and the American Academy of Pediatrics (AAP)-endorsed algorithm (fail or rescreen criteria: <95% in both extremities (right hand and either foot) and/or >3% differential in saturations).6 Participating NICUs used either the NJ recommended (n=18) or AAP (n=3) algorithm for the evaluation. For this evaluation, both the NJ- and AAP-endorsed algorithms were modified at stage 1 for infants receiving oxygen. This modification removed the rescreening requirement for saturations <95% if results were consistent with clinical management (for example, lower target saturations for premature infants). The modification was intended to avoid unnecessary repeat screening attempts while aiding in early detection of aortic arch anomalies. Therefore, for infants on oxygen at Stage 1, rescreening was only required for >3% differential between pre- and post-ductal readings. Screening results of <95% that did not necessitate rescreening were considered conditional passes. Conversely, saturations of <95% without a corresponding clinical explanation were regarded as fails. NJ- or AAP-endorsed algorithms were used without modification at Stage 2. Infants with a complete screen performed upon discharge with no Stage 1 or Stage 2 results were categorized as having a predischarge screen. For analysis of screening results, we universally applied the NJ thresholds because all failed screens per the AAP algorithm would also fail the NJ algorithm. As the majority of sites implemented the NJ algorithm and these thresholds are inclusive of the fail and rescreen criteria as defined by the AAP, the NJ thresholds were analytically applied to all screening results. Deidentified infant data were collected via a secure, web-based instrument and included information on birth weight, gestational age, prenatal CHD diagnosis, echocardiogram before the screen, age at time of screen(s), pulse oximetry screening values, receipt of supplemental oxygen at 24 to 48 h and/or at time of discharge, if on oxygen and results of <95% whether this was consistent with their clinical profile and final diagnosis.
Figure 1.
Multistage algorithm for evaluation of critical congenital heart defects screening in the neonatal intensive care unita. aImplementation of this screening algorithm should not take the place of clinical judgment and intervention.
Study population
This evaluation included infants admitted to 21 level III and IV NICUs14 in five states: California (1), Illinois (1), New Jersey (9), New York (5) and Minnesota (5). Participation was voluntary with no funding provided. Infants born between 1 February 2015 and 30 September 2015 with a NICU stay of >23 h and at least one set of screening results were eligible for inclusion. Infants with a prenatal CHD diagnosis and/or echocardiography before screening (preidentifying factors) were included to evaluate universal screening in the NICU. Transfers to the NICU after failed screening on mother–baby units or well-baby nurseries were excluded.
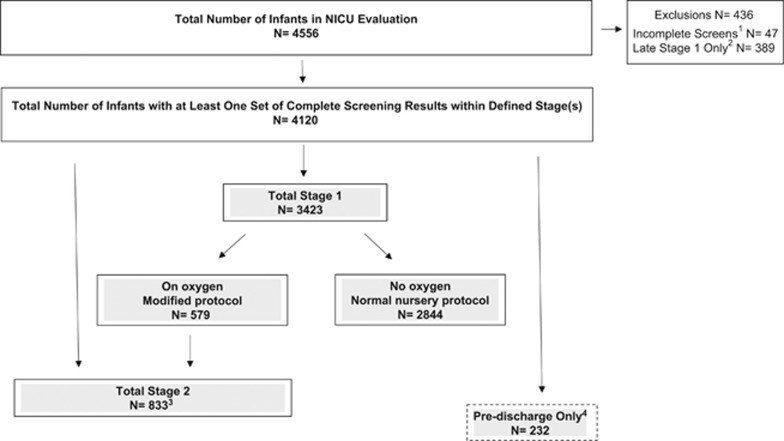
Of the 4556 enrolled infants, a total of 436 infants were excluded. Forty-seven infants (1%) were excluded for not having a complete set of screening results; initial pre-and post-ductal results required a rescreen that was not performed (Figure 2). Infants with a Stage 1 screen performed ⩾56.5 h after NICU admission (referenced as late Stage 1) were also excluded (n=389 infants, 8.5% Figure 2). In practice, the strict cutoff of 48 h did not allow for minor variation because of practical implementation factors (for example, time of day) that may have resulted in a calculated screening time of slightly >48 h. To accommodate reasonable, real-world application with the goal of early screening, we used the s.d. (7.5 h) of the 24 to 48 h stage 1 target timing window to analytically define Stage 1 screens as >23 to <56.6 h (n=160). Infants with incomplete and late Stage 1 screens were more likely to have been on oxygen and born at <28 weeks than infants with complete screening results.
Figure 2.
Flowchart of participants in critical congenital heart defects screening neonatal intensive care unit (NICU) evaluation, February 2015 to September 2015. 1Infants for whom one pre-and post-ductal result was obtained, the results required a re-screen to be performed, and this re-screen was not done. 2Infants with only a Stage 1 screen which was performed after defined Stage 1 time window (greater than 56.7 hours). 3Includes infants on oxygen at 24 to 48 h after birth screened at Stage 1 and Stage 2 (n=368), and infants on oxygen at 24 to 48 h after birth with only a Stage 2 screen (n=465) because: a) NICU did not screen infants on oxygen or b) Stage 1 screen was missed or incomplete. 4Infants only screened within 4 days of discharge (N=112 not on oxygen at Stage 1).
The final sample consisted of 4120 infants with at least one set of complete screening results within a defined stage(s): 83.1% were screened at stage 1 (n=3423), 20.2% at stage 2 (n=833) and 5.6% predischarge only (n=232) (Figure 2). Quality assurance was conducted by each site on 29% of the final sample. This evaluation was deemed public health practice by each site’s institutional review board.
Statistical analyses
FPRs for CCHD were calculated as the number of infants without CCHD who failed on the first set of screening results divided by the number of infants without CCHD who were screened multiplied by 100 using the NJ-recommended algorithm5 with modification for infants on oxygen. In addition, FPRs were calculated for CCHD and CHD combined. The χ2 tests of significance at P<0.05 were used.
Implementation survey
An 11-question survey was administered to qualitatively assess implementation of the multistage screening protocol. This survey was completed by 258 nurses with representation from each NICU. A 10-point Likert scale was used to assess burden of implementation with 1=no burden and 10=extremely burdensome.15 The mean score was interpreted as: low burden=1 to 3.9, moderate burden=4.0 to 6.9, and high burden=7 to 10.15
Results
Of the 4120 infants with complete, appropriately timed screens, 43.8% were discharged from the NICU before 5 days (<144 h), 71.7% were not receiving supplemental oxygen at 24 to 48 h, 56.3% were born ⩾2500 g and 48.1% were born at ⩾37 weeks (Table 1). In addition, 91.5% of infants did not have a prenatal CHD diagnosis or echocardiogram before the screen. The majority of our NICU population (67.7%, n=2791) had no preidentifying factor and were not on oxygen, comprising the subgroup of infants who may benefit from early screening at 24 to 48 h.
Table 1. Characteristics of infants in evaluation of critical congenital heart defects (CCHD) screening in the neonatal intensive care unit, February to September 2015 (N = 4556).
| Total, N=4556 | Complete screensa, N=4120 |
Stage 1b, N=3423 |
|||
|---|---|---|---|---|---|
| On O2, n=579 | Not on O2, n=2844 | Stage 2c, N=833 | |||
| Length of stay (hours) | % | % | % | % | % |
| Short stay (<144) | 43.9 | 43.8 | 13.3 | 56.1 | 18.6 |
| Long stay (⩾144) | 56.1 | 56.2 | 86.7 | 43.9 | 81.4 |
| Prenatal CHD diagnosis and/or echo before screen | 9.0 | 8.5 | 15.9 | 5.5 | 13.7 |
| Prenatal CHD diagnosis only | 0.5 | 0.5 | 0.2 | 0.6 | 0.1 |
| Echo before screen only | 7.2 | 6.7 | 13.5 | 3.7 | 12.7 |
| Prenatal CHD diagnosis and echo before screen | 1.3 | 1.3 | 2.2 | 1.2 | 0.8 |
| Neither prenatal CHD diagnosis nor echo before screen | 91.0 | 91.5 | 84.1 | 94.5 | 86.3 |
| Gestational age (weeks) | |||||
| <28 | 4.3 | 4.2 | 14.7 | 0.7 | 9.7 |
| 28–31 | 8.9 | 9.0 | 20.9 | 4.3 | 21.5 |
| 32–36 | 38.2 | 38.8 | 35.4 | 39.5 | 39.1 |
| ⩾37 | 48.6 | 48.1 | 29.0 | 55.4 | 29.7 |
| Median | 36 | 36 | 34 | 37 | 34.0 |
| Range | 23–42 | 23–42 | 23–41 | 23–42 | 23–42 |
| Birth weight (grams) | |||||
| <1000 | 4.3 | 4.1 | 13.6 | 0.9 | 9.5 |
| 1000–1499 | 7.1 | 7.2 | 17.6 | 3.7 | 16.1 |
| 1500–2499 | 32.0 | 32.4 | 30.7 | 31.9 | 33.6 |
| ⩾2500 | 56.7 | 56.3 | 38.0 | 63.4 | 40.8 |
| Median | 2700 | 2690 | 2090 | 2891 | 2180 |
| Range | 325–6100 | 325–6100 | 325–6100 | 510–5639 | 410–6100 |
Complete screens within specific stage time definitions.
Stage 1: screening targeted for 24–48 h after admission.
Stage 2: screening targeted for 24–48 h after weaning from supplemental oxygen.
Multistage screening results
Of the 3423 infants screened at Stage 1, 0.9% failed (n=32) and 99.1% passed (n=3391), inclusive of 138 conditional passes. Of Stage 2 screens, 0.6% failed (n=5) and 99.4% passed (n=828). Of infants with only a predischarge screen (n=232), 100% passed.
Use of supplemental oxygen
Of the 579 infants screened on oxygen at Stage 1, 97.9% passed and 2.1% failed using the modified algorithm. Without modification, the fail rate would increase to 25.9% that is, if conditional passes are considered fails) (Table 2). Despite the modification, screening results were significantly less favorable for infants on oxygen than for their counterparts on room air (P<0.002).
Table 2. Final Stage 1 screening outcomes by potential exclusion criteria, evaluation of critical congenital heart defects (CCHD) screening in the neonatal intensive care unit, February to September 2015 (N=3423).
|
On oxygen N=579 |
Not on oxygen N=2844 |
|||||||
|---|---|---|---|---|---|---|---|---|
| Total pass | Pass | Conditional passa | Failb | Pass | Failb | |||
| N | % | % | % | % | N | % | % | |
| Total infants screened at Stage 1 | 579 | 97.9 | 74.1 | 23.8 | 2.1 | 2844 | 99.3 | 0.7 |
| Prenatal CHD diagnosis and/or echo before screen | 92 | 95.7 | 68.5 | 27.2 | 4.3 | 157 | 95.5 | 4.5 |
| Neither prenatal diagnosis nor echo before screen | 487 | 98.4 | 75.2 | 23.2 | 1.6 | 2687 | 99.5 | 0.5 |
| Gestational age (weeks) | ||||||||
| <28 | 85 | 97.6 | 56.5 | 41.2 | 2.4 | 21 | 90.5 | 9.5 |
| 28–31 | 121 | 99.2 | 74.4 | 24.8 | 0.8 | 123 | 100.0 | – |
| 32–36 | 205 | 99.0 | 74.6 | 24.4 | 1.0 | 1124 | 99.8 | 0.2 |
| ⩾37 | 168 | 95.8 | 82.1 | 13.7 | 4.2 | 1576 | 98.4 | 1.0 |
| Birth weight (g) | ||||||||
| <1000 | 79 | 97.5 | 64.6 | 32.9 | 2.5 | 27 | 92.6 | 7.4 |
| 1000–1499 | 102 | 100.0 | 67.6 | 32.4 | — | 106 | 100.0 | – |
| 1500–2499 | 178 | 98.9 | 75.3 | 23.6 | 1.1 | 907 | 100.0 | – |
| ⩾2500 | 220 | 96.4 | 79.5 | 16.8 | 3.6 | 1804 | 99.0 | 1.0 |
Infants on supplemental oxygen were deemed conditional passes if saturations <95%, yet differential <4% and saturations were consistent with clinical profile. Rescreening only performed as per the modified arm of the algorithm. Percentage is of all infants on oxygen who passed the screen.
Fail as per the New Jersey-recommended algorithm.
Exclusion criteria
Prenatal CHD diagnosis and/or echocardiogram before the screen
Of the 32 infants who failed Stage 1, the majority (66%, n=21) did not have a potential exclusion criteria of a prenatal CHD diagnosis and/or echocardiogram before the screen. One of these infants with previously unsuspected CHD was identified by screening (Supplementary Material I). Infants without a preidentifying factor on room air had significantly lower fail rates (0.5%) than those on oxygen (1.6%, P<0.004). Among infants who failed the Stage 1 screen without a preidentifying factor, 38% (n=8) were on oxygen, 5 of whom had an echocardiogram in response to their screen that yielded a patent foramen ovale or patent ductus arteriosis. As anticipated, overall, infants with a prenatal CHD diagnosis and/or echocardiogram before the screen had higher fail rates at Stage 1 than those without a preidentifying factor (P<0.0001) (Table 2). For these preidentified infants, receipt of supplemental oxygen had no significant influence on Stage 1 pass and fail rates.
Gestational age and birth weight
Stage 1 fail rates were significantly higher among infants born at <28 weeks and <1000 g compared with those born at higher birth weights and older gestational ages (P<0.0003 and P<0.0001, respectively) (Table 2). The proportion of conditional passes increased with decreasing gestational age; most notably for infants born at <28 weeks for whom 41.2% of passes were conditional and would have qualified as rescreens or fails using a nonmodified protocol. Infants born at ⩾37weeks or ⩾2500 g and receiving oxygen had higher fail rates (4.2% and 3.6%, respectively) than comparable infants not on oxygen (1.0% and 1.0%, respectively).
Discharge diagnoses
Of the 4556 total infants, 24 had CCHD yielding a prevalence of 5.3 per 1000 (95% confidence interval (CI): 3.5, 7.9) in our NICU population. In all, 17 infants with CCHD had complete screening results, 9 passed the screen (52.9%), 3 were conditional passes (17.6%) and 5 failed (29.4%) (Supplementary Material II). No infants with CCHD were identified because of the screen. Of the 12 false negative screens (including the 3 conditional passes), 5 infants had coarctation of the aorta. Although 2 of 12 infants did not have a preidentifying factor, all were symptomatic before screening (for example, murmur, tachypnea, desaturations). Of the 47 infants who were excluded because of incomplete screening results, 6 had CCHD and were symptomatic before the screen. In the absence of complete screening results, we cannot conclusively categorize these infants as either true positive or false negative screens.
The remaining infants with complete screens had noncardiac final diagnoses (86.6%), an unresolved patent foramen ovale or patent ductus arteriosis (10.1%), noncritical CHDs (2.8%) or expired after screening (0.2%). Only 0.3% of infants with noncardiac diagnoses failed at Stage 1 or Stage 2. Approximately 3.9% of infants with an unresolved patent foramen ovale and/or patent ductus arteriosis confirmed by echocardiogram failed either the Stage 1 or Stage 2 screen. The proportion of infants with CHD with a failed screen was 3.5%. One infant with a ventricular septal defect (VSD) was identified due to screening (Supplementary Material I).
Assessment of burden
Applying the NJ-recommended algorithm with modification, the overall FPR of NICU screening was 0.8% (95% CI: 0.6, 1.1; Table 3). The overall FPR increased to 4.0% (95% CI: 3.5, 4.7) when conditional passes were treated as fails. FPRs were highest among infants screened on oxygen (2.1%, 95% CI: 1.2, 3.7) and those born at <28 weeks (3.8%, 95% CI: 1.4, 10.2). Compared with Stage 1 screening on oxygen (2.1%), FPRs were significantly lower for Stage 1 not on oxygen (0.5%) and Stage 2 (0.6%) (P<0.0001 and P<0.01, respectively). Given our finding of one infant with an unsuspected VSD identified by screening, FPRs were examined for the broader group of CHD and were remarkably comparable to FPRs for detection of CCHD (Table 3).
Table 3. False positive rates (FPRs)a applying New Jersey-recommended critical congenital heart defects (CCHD) screening algorithm, b.
| Overall FPR | FPR among infants without a prenatal CHD diagnosis or echocardiogram before screen | |
|---|---|---|
| % (95% CI) | % (95% CI) | |
| FPR for CCHD | ||
| Overall | 0.8 (0.6, 1.1) | 0.6 (0.4, 0.9) |
| Stage 1 | ||
| On oxygen | 2.1 (1.2, 3.7) | 1.6 (0.8, 3.3) |
| No oxygen | 0.5 (0.3, 0.9) | 0.6 (0.4, 0.9) |
| Extremely premature (<28 weeks) | 3.8 (1.4, 10.2) | 4.0 (1.5, 10.8) |
| Stage 2 | 0.6 (0.3, 1.4) | 0.3 (0.1, 1.1) |
| FPR for CCHD+noncritical CHD | ||
| Overall | 0.7 (0.5, 1.0) | 0.6 (0.4, 0.9) |
| Stage 1 | ||
| On oxygen | 2.1 (1.1, 3.7) | 1.7 (0.9, 3.5) |
| No oxygen | 0.5 (0.3, 0.9) | 0.6 (0.4, 0.9) |
| Extremely premature (<28 weeks) | 4.0 (1.5, 10.6) | 4.2 (1.6, 11.2) |
| Stage 2 | 0.4 (0.1, 1.2) | 0.3 (0.1, 1.2) |
Abbreviations: CHD, congenital heart defects; CI, confidence interval.
Fail/rescreen=<95% either or >3%.
Conditional passes are considered passes in all FPR calculations.
When restricted to infants without a prenatal CHD diagnosis or echocardiogram before the screen, the FPR using the modified algorithm decreased to 0.6% overall (95% CI: 0.4, 0.9); 1.6% and 0.6% for Stage 1 on and off oxygen respectively, and 0.3% for Stage 2. FPRs for this restricted group were not significantly different from those using the entire sample (P=0.367).
Overall, 13.1% of infants had an echocardiogram during hospitalization (n=540/4120), yet only 7 (0.2%) were performed in response to failed screens at any stage, one of which identified the CHD. Based on quantitative survey findings, nursing staff reported screening was of low burden during this evaluation (mean=3.5). Approximately 80% of survey respondents reported that having a NICU-specific CCHD screening protocol facilitated differentiation between screening and routine monitoring with pulse oximetry. In addition, 81% responded that utilization of the evaluation protocol increased awareness of unsuspected CHD in the NICU.
Discussion
Our findings demonstrate that the NICU population is heterogeneous; with a large percentage of infants born ⩾2500 g, not receiving supplemental oxygen and discharged within 5 days after birth, therefore similar to the well-baby population for which CCHD screening was initially intended.8 In the absence of systematic screening, approximately two-thirds of our NICU population (that is, those without a preidentifying factor and not receiving supplemental oxygen) were at risk for late detection of unsuspected CCHD and had the potential to benefit from early systematic screening at 24 to 48 h. This estimate of infants who may benefit from screening at 24 to 48 h is likely higher in communities with lower prenatal detection rates as the NICUs in our evaluation represented high resource settings that are likely to have a lower rate of undiagnosed CCHD. For all newborns, regardless of setting, clinical exam and pulse oximetry screening are used in tandem to evaluate the presence of CCHD. Despite more intensive clinical monitoring, admission to a NICU does not preclude the potential for unrecognized CCHD and/or CHD as evidenced by our finding that screening identified one infant with an unsuspected noncritical CHD. As data demonstrate that critical and noncritical CHD are associated with neurodevelopmental delays, early identification and intervention across all CHD is important.16 Consistent with our findings, a recent international, single-center study evaluated pulse oximetry screening upon admission in conjunction with clinical exam and supported the feasibility of screening in the NICU.17 Although this study screened infants with pulse oximetry at <24 h, their recommendation for universal screening was made despite a markedly higher overall FPR (55.8%) than found in our evaluation.
Concerns regarding timing and appropriateness of screening among NICU infants receiving supplemental oxygen have been noted.10, 11, 12, 13, 18, 19 Our protocol of screening infants at 24 to 48 h, including those receiving supplemental oxygen, aimed to address these issues. Aware that target saturations for infants receiving supplemental oxygen may be intentionally lower, our modification of only requiring a rescreen for >3% pre- and post-ductal differential or saturations <95% inconsistent with clinical profile, afforded the ability to minimize false positives and potentially aid in early detection of aortic arch anomalies. Our modification did significantly reduce the overall FPR (0.8%) compared with when no modification was implemented (4.1%). Of important note, the FPR was further reduced when infants on oxygen at 24 to 48 h were screened as soon as possible after weaning (0.6%) and strikingly comparable to those not on oxygen at 24 to 48 h (0.5%). Difficulties were observed in tracking the timing of multiple screens for infants receiving oxygen at 24 to 48 h highlighting challenges with implementing the modified multistage protocol. Additional complexities arose, including misinterpretation of the >3% differential criteria, although this challenge is not unique to the NICU.20
Application of exclusion criteria based on preidentifying factors and gestational age is not straightforward. The use of echocardiography alone for exclusion of infants from screening raised concerns regarding variability in echocardiogram timing and quality, and accessibility of pediatric specialty expertise for interpretation to rule out CCHD. A prenatal CHD diagnosis alone affected a very small proportion of the NICU population, resulting in negligible added burden. Furthermore, collection of pulse oximetry screening values for infants with confirmed CHD can provide useful insight into the predictive potential of alternative saturation thresholds for improving the sensitivity of CCHD screening. Although infants born at <28 weeks or <1000 g also comprised a small proportion of our NICU population, the higher incomplete screen rates and FPRs at 24 to 48 h for this subgroup compared with other gestational ages and birth weights emphasize the challenges for the timing of CCHD screening in this population. As the goal of universal screening is early detection, screening infants born at <28 weeks or <1000 g as soon as possible when medically stable is a reasonable goal.
Our qualitative survey findings provided additional endorsement for systematic NICU screening. Consistent with evidence of the effectiveness of cognitive forcing strategies to decrease diagnostic error, nursing staff reported that implementation of a systematic, NICU protocol on all infants instilled a heightened awareness among clinicians of the potential for underlying, unsuspected CCHD.21 Furthermore, clinicians reported that application of exclusion criteria within the NICU introduces practice variation that may increase the likelihood of missed screens for the remainder of the population.
We quantified the burden of our multistage protocol in several ways. First, the number of echocardiograms conducted in direct response to failed screens was extremely low and one resulted in early identification of a VSD. As the number of fails and unnecessary echocardiograms related to Stage 2 were also minimal, screening as soon as possible after weaning from oxygen is reasonable. This is particularly applicable for longer stay infants for whom screening closer to the time of discharge might be too late for early identification of CCHD. In addition, the majority of echocardiograms performed in response to failed screens were among infants on oxygen at 24 to 48 h with an unresolved patent foramen ovale or patent ductus arteriosis, providing further evidence for screening infants as soon as possible after weaning. Second, we quantified FPRs as a mechanism to evaluate burden of screening. Of the four US studies conducted, three reported no failed screens10, 11, 12 with the fourth reporting13 a FPR (0.7%) that was similar to our overall findings, yet higher than that of asymptomatic newborns.22 de-Wahl Granelli et al.23 reported a FPR of 0.2% in the well-baby nursery for infants screened using both extremities at >24 h excluding those with a prenatal CHD diagnosis. Although caution is needed in making direct comparisons between FPRs from our evaluation with those of well-baby populations, our overall FPR (0.8%) was more comparable when restricted to infants who were not on oxygen at 24 to 48 h (0.5%) and among infants after weaning from oxygen without a preidentifying factor (0.3%). Our FPR among infants born at <28 weeks was significantly higher than all other gestational age groups, emphasizing the complexities of screening this sub-population. As FPRs for CCHD categorize infants with a noncritical CHD identified by screening as a false positive, we also calculated FPRs for all CHD, critical and noncritical combined.24 These rates were quite comparable to those restricted to CCHD, further supporting the minimal added burden of screening all NICU infants with the potential benefit of identifying other unsuspected, noncritical conditions. Finally, the minimal burden reported by nursing staff during the evaluation demonstrated that CCHD screening is feasible in the NICU population. These findings coupled with the added benefits of increased awareness of unsuspected CHD and clarification of screening versus monitoring suggest that CCHD screening may have a meaningful role in the NICU without the significant added burden as previously perceived.
This evaluation was strengthened by its ability to prospectively implement a multi-stage protocol. As participating NICUs represented closely monitored infants of varying levels of acuity, we were able to assess the population for which CCHD screening was questionable. Our ability to collect individual-level information on preidentifying factors enabled us to further refine the population potentially benefiting from screening. Information on echocardiograms performed in response to failed screens was crucial for quantifying resource burden. Although the overall sensitivity of pulse oximetry screening in our NICU evaluation was low, it is essential to incorporate preidentification of CCHD when evaluating the unique contribution of pulse oximetry for early detection. As the majority of our infants with CCHD were identified before screening, we did not have sufficient power to conclude that screening with pulse oximetry in the NICU is not of benefit. Conversely, two of the nine infants with false negative screens were not preidentified, suggesting necessary caution against overreliance on either clinical assessment or screening alone for the detection of CCHD in the NICU. Further investigation is needed on the implications of applying different screening thresholds to specific NICU sub-populations as well as feasible options for early identification of aortic arch anomalies.
Given the majority of our population comprised infants ⩾2500 g, not on oxygen and without either preidentifying factor, CCHD screening targeted at 24 to 48 h may benefit NICU infants who approximate the well-baby population and are at potentially higher risk for undetected CCHD. Although challenges arose when early screening was conducted on the small population of infants born at <28 weeks and those receiving supplemental oxygen, exclusion of sub-populations within the NICU introduces practice variation that may potentially result in missed screens. Collectively, the evidence from our evaluation supports the feasibility, minimal burden and potential benefits of implementing systematic early screening in the NICU for undetected CCHD.
Acknowledgments
This project was partially supported by the Health Resources and Services Administration (HRSA) of the US Department of Health and Human Services (HHS) under Grant Number H46MC24059, Genetic Services Project (CCHD Screening). The content and conclusions are those of the authors and should not be construed as the official position or policy of, nor should any endorsements be inferred by HRSA, HHS or the US Government. We acknowledge the efforts from all participating centers to implement the evaluation as well as collect and enter data. Thanks to the key contributors and staffs from the following NICUs: Naheed Abedin, Capital Health Medical Center-Hopewell; Patricia Benedetto, Mary Keller-Olsen, Children’s Hospital of NJ at Newark Beth Israel Medical Center; Mayuri Shah, Krystyna Toczylowski, Hackensack Medical Center; Michael Carter, Jersey Shore Medical Center; Stephanie Salerno, Laura Padvarietis, Long Island Jewish (LIJ) Cohen Children’s Medical Center; Patricia Quinn, LIJ Lenox Hill; Jennifer Ruggiano, LIJ North Shore; John Ladino, Megan Shea, Morristown Medical Center; Bonnie Adler, Lisa D’Amico, Monmouth Medical Center; Laura Wenzel, Northwell Staten Island University Hospital, St Joseph’s Healthcare System; Pansy Haynes-Mayfield, Debbie Bowman, Sunita Shah, Edith Legaspi, Saint Peter’s University Hospital; Angela Huang, Wei-Fen Den, Santa Clara Valley Medical Center; Nithi Fernandes, Mike Sacilowski, University of Buffalo Medical Center; Kelsey Balcer, University of Illinois Medical Center; Debra Brendel, University of Medicine and Dentistry of NJ; Bev Rose, Melissa Engel, University of Minnesota Masonic Children’s Hospital (UMMCH); Juanita Royle, Fairview Ridges Hospital; Juanita Royle, Nanette Nys, Fairview Southdale Hospital; Katie Pfister, Maple Grove Hospital; Mary Pat Osborne, North Memorial Medical Center; Thomas George, NICU UMMCH, Fairview Ridges/Southdale Hospitals. We thank the NJ NICU Collaborative for their support of this project. We thank Emily Carey PerezdeAlejo, Office of Continuing Professional Education, Rutgers, The State University of New Jersey, for assistance with development of the data instrument and technical support throughout the project. We also show our gratitude to Fran Gallagher, Harriett Lazarus, and Lindsay Caporrino, NJ Chapter, American Academy of Pediatrics, and Mary Knapp, Marilyn Gorney-Daley, and Joy Rende, Early Identification and Monitoring Program, Special Child Health and Early Intervention Services, Division of Family Health Services, NJ Department of Health, for support of this evaluation.
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Footnotes
Supplementary Information accompanies the paper on the Journal of Perinatology website (http://www.nature.com/jp)
The authors declare no conflict of interest.
Supplementary Material
References
- Critical congenital heart disease. Genetics Home Reference. U.S. National Library of Medicine. May 2013, available at http://ghr.nlm.nih.gov/condition/critical-congenital-heart-disease.
- Mahle WT, Newburger JW, Matherne GP, Smith FC, Hoke TR, Koppel R et al. Role of pulse oximetry in examining newborns for congenital heart disease: a scientific statement from the AHA and AAP. Pediatrics 2009; 124(2): 823–836. [DOI] [PubMed] [Google Scholar]
- Mahle WT, Martin GR, Beekman RH, Morrow WR Section on Cardiology and Cardiac Surgery Executive Committee. Endorsement of Health and Human Services recommendation for pulse oximetry screening for critical congenital heart disease. Pediatrics 2012; 129(1): 190–192. [DOI] [PubMed] [Google Scholar]
- NewSTEPS. Association of Public Health Laboratories. CCHD Screening Status. Available at https://www.newsteps.org/cchd (Accessed on 12 March 2016).
- Garg LF, Van Naarden Braun K, Knapp M, Anderson TM, Koppel RI, Hirsch D et al. Results from the New Jersey statewide critical congenital heart defects screening program. Pediatrics 2013; 132(2): e314–e323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemper AR, Mahle WT, Martin GR, Cooley WC, Kumar P, Morrow WR et al. Strategies for implementing screening for critical congenital heart disease. Pediatrics 2011; 128(5): e1259–e1267. [DOI] [PubMed] [Google Scholar]
- Harrison W, Goodman D. Epidemiologic trends in neonatal intensive care, 2007-2012. JAMA Pediatr 2015; 169: 855–862. [DOI] [PubMed] [Google Scholar]
- Osterman MJK, Martin JA, Mathews TJ, Hamilton BE. Expanded Data from the New Birth Certificate. 2008. National Vital Statistics Reports; Vol 59, no.7. National Center for Health Statistics: Hyattsville, MD, 2011. [PubMed] [Google Scholar]
- Oster ME, Aucott SW, Glidewell J, Hackell J, Kochilas L, Martin GR et al. Lessons learned from newborn screening for critical congenital heart defects. Pediatrics 2016; 1(37): e2 0154573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakshminrusimha S, Turkovich S, Manja V, Nair J, Kumar VHS. Critical congenital heart disease screening with pulse oximetry in the neonatal intensive care unit. J Neonatal Res 2012; 2: 96–101. [Google Scholar]
- Iyengar H, Kumar P, Kumar P. Pulse-oximetry screening to detect critical congenital heart disease in the neonatal intensive care unit. Pediatr Cardiol 2014; 35(3): 406–410. [DOI] [PubMed] [Google Scholar]
- Goetz EM, Magnuson KM, Eickhoff JC, Porte MA, Hokanson JS. Pulse oximetry screening for critical congenital heart disease in the neonatal intensive care unit. J Perinatol 2016; 36(1): 52–56. [DOI] [PubMed] [Google Scholar]
- Manja V, Mathew B, Carrion V, Lakshminrusimha S. Critical congenital heart disease screening by pulse oximetry in a neonatal intensive care unit. J Perinatol 2015; 35(1): 67–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Academy of Pediatrics Committee on Fetus and Newborn. Levels of neonatal care. Pediatrics 2012; 130(3): 587–597. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. Rapid implementation of pulse oximetry newborn screening to detect critical congenital heart defects - New Jersey, 2011. MMWR Morb Mortal Wkly Rep 2013; 62(15): 292–294. [PMC free article] [PubMed] [Google Scholar]
- Riehle-Colarusso T, Autry A, Razzaghi H, Boyle CA, Mahle WT, Van Naarden Braun K et al. Congenital heart defects and receipt of special education services. Pediatrics 2015; 136(3): 496–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X, Zhao Q, Ma X, Yan W, Ge X, Jia B et al. Pulse oximetry could significantly enhance the early detection of critical congenital heart disease in neonatal intensive care units. Acta Paediatr 2016; 105: e499–e505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suresh GK. Pulse oximetry screening for critical congenital heart disease in neonatal intensive care units. J Perinatol 2013; 33(8): 586–588. [DOI] [PubMed] [Google Scholar]
- Lakshminrusimha S, Sambalingam D, Carrion V. Universal pulse oximetry screen for critical congenital heart disease in the NICU. J Perinatol 2014; 34(5): 343–344. [DOI] [PubMed] [Google Scholar]
- Oster ME, Kuo KW, Mahle WT. Quality improvement in screening for critical congenital heart disease. J Pediatr 2014; 164: 67–71. [DOI] [PubMed] [Google Scholar]
- Sherbino J, Dore KL, Siu E, Norman G. The effectiveness of cognitive forcing strategies to decrease diagnostic error: an exploratory study. Teach Learn Med 2011; 23(1): 78–84. [DOI] [PubMed] [Google Scholar]
- Thangaratinam S, Brown K, Zamora J, Khan KS, Ewer AK. Pulse oximetry screening for critical congenital heart defects in asymptomatic newborn babies: a systematic review and meta-analysis. Lancet 2012; 379(9835): 2459–2464. [DOI] [PubMed] [Google Scholar]
- de-Wahl Granelli A, Wennergren M, Sandberg K, Mellander M, Bejlum C, Inganäs L et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ 2009; 338: a3037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ewer AK, Martin GR. Newborn pulse oximetry screening: which algorithm is best? Pediatrics 2016; 138(5): e20161206. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.