Abstract
Patient: Female, 59
Final Diagnosis: Systemic mastocytosis in association with small lymphocytic lymphoma
Symptoms: Skin rash
Medication: —
Clinical Procedure: Bone marrow biopsy
Specialty: Hematology
Objective:
Rare disease
Background:
Systemic mastocytosis with an associated hematologic non-mast cell lineage disease is a rare entity, and the majority of systemic mastocytosis cases are associated with myeloid neoplasm. Lymphoproliferative disorders are less commonly associated with systemic mastocytosis and a few cases of systemic mastocytosis associated with chronic lymphocytic leukemia have been described in the literature.
Case Report:
We present a case of indolent systemic mastocytosis associated with small lymphocytic lymphoma. The bone marrow biopsy demonstrated mast cells in the form of clusters and perivascular distribution on immunohistochemistry for tryptase, CD2, and CD25 markers. In addition, 30% involvement by small lymphocytic lymphoma was discovered in the form of interstitial lymphoid aggregates composed of small lymphocytes. Flow cytometry showed B-cells positively stained for CD19, CD20, CD5, CD23, and kappa light chains, and the CD38 expression was <5%.
Conclusions:
In systemic mastocytosis with an associated hematologic non-mast cell lineage disease, the combination of systemic mastocytosis associated with small lymphocytic lymphoma is rare and the management strategy follows the principle of treating the two entities individually as if they are not related. Clinical surveillance is indicated for indolent systemic mastocytosis and low-risk small lymphocytic lymphoma to monitor for disease progression.
MeSH Keywords: Leukemia, Lymphocytic, Chronic, B-Cell; Mast Cells; Mastocytosis, Systemic
Background
Mast cells develop from the common myeloid progenitor cells and are widely distributed in the skin and mucosal surfaces [1]. Mastocytosis is a clonal mast-cell disease which could be limited to skin (cutaneous mastocytosis) or involve internal organs with or without skin infiltration (systemic mastocytosis) [2–5]. Mastocytosis has a reported prevalence of 1 in 10,000 inhabitants, and it is likely under diagnosed [6]. Systemic mastocytosis (SM) is the major subtype of mastocytosis in adults, and bone marrow is the most common extra-cutaneous organ involved [2–5].
SM is a heterogeneous group of mast cell disorders that is categorized into four entities by the 2008 World Health Organization (WHO) classification: indolent systemic mastocytosis (ISM), aggressive systemic mastocytosis (ASM), systemic mastocytosis with an associated hematologic non-mast cell lineage disease (SM-AHNMD), and mast cell leukemia (MCL) [7]. SM patients could present with B findings (organ enlargement without organ dysfunction) or C findings (organ dysfunction due to excessive mast cell infiltration). The C findings can include pancytopenia due to bone marrow infiltration, deranged liver function, impaired coagulation, portal hypertension due to liver involvement, splenomegaly or hypersplenism, weight loss or malabsorption secondary to GI tract infiltration, osteolytic lesions and pathological fractures due to underlying bone involvement [8].
SM-AHNMD comprises between 5–40% of cases of SM [9,10]. In SM-AHNMD, the associated hematological component could be diagnosed with, before, or after SM, and sometimes the mast cell aggregates may be obscured [11]. Associated myeloid neoplasia accounts for the majority of SM-AHNMD cases (90%) and includes myelodysplastic syndromes (MDS), myeloproliferative neoplasms (MPN), MDS/MPN, and acute myeloid leukemia (AML). Chronic myelomonocytic leukemia is the neoplasm most commonly associated with SM-AHNMD.
SM associated with lymphoproliferative disorders or plasma cell myeloma is rare [12–15] and a few cases of SM with associated chronic lymphocytic leukemia (SM-CLL) have been described in the literature [16–19], mainly concentrating on histopathologic features. Here, we report a case of SM associated with small lymphocytic lymphoma (SM-SLL), focusing on histopathologic features, prognosis, and management.
Case Report
A 59-year-old Caucasian female presented with constitutional symptoms and a skin rash associated with intractable itching. She noted small reddish-brown papules initially on her forearms which developed to whole-body distribution in two years. She had no fever, weight loss, lymphadenopathy, cutaneous flushing, dyspnea, abdominal pain, or diarrhea. She had a medical history of congestive heart failure, hypertension, obesity, and chronic obstructive pulmonary disease. She had a surgical history of aortic valve replacement and hence was on warfarin. The patient had a 30 pack a year smoking history. There was no known family history of mast-cell disorders. She had a history of drug allergies, in the form of skin reactions, to penicillins and sulfa drugs but reported no anaphylaxis. Exposure to Hymenoptera stings was unknown.
Physical examination showed numerous pink and pink-tan 2–5 mm macules and thin papules scattered on the chest, abdomen, trunk, arms, and thighs. Darier’s sign was not elicited. A grade 5 ejection systolic murmur was present. There was no hepatosplenomegaly, lymphadenopathy, bruising, or sign of infection.
On initial presentation, the hemoglobin concentration was 13.2 g/dL (12.3–15.5 g/dL), hematocrit 41% (35–44.5%), mean corpuscular volume 90 fL (80–96 fL), white blood cell count 7,100/µL (3,400–9,400/µL) and platelet count 187,000/µL (140,000–410,000/µL). Differential analysis showed 54% absolute neutrophils (40–60%), 38% absolute lymphocytes (20–40%), 3% absolute monocytes (2–8%), 4% absolute eosinophils (1–3%) and 1% absolute basophils (0–1%). Coagulation studies showed a prothrombin time of 28.4 seconds (11–13.5 seconds) and an international normalized ratio (INR) of 2.6 (0.8–1.1) while being on warfarin. There were no electrolyte abnormalities. Liver function tests showed alkaline phosphatase 79 IU/L (30–130 IU/L), aspartate aminotransferase 15 IU/L (0–41 IU/L), alanine aminotransferase 13 IU/L (0–45 IU/L), total bilirubin 0.7 mg/dL (0–1 mg/dL) and total protein of 6.9 g/dL (6–8 g/dL). Serum creatinine and BUN concentrations were 0.5 g/dL (0.7–1.4 mg/dL) and 13 mg/dL (6–22 mg/dL) respectively.
Differential diagnosis was broad and included urticaria pigmentosa, cutaneous T-cell lymphoma, cutaneous mastocytosis, or drug-eruption. She was initially diagnosed with urticaria pigmentosa and offered topical symptomatic treatment, but remained refractory after four months of therapy. Skin punch biopsies from two different locations showed mild hyperkeratosis with sparse superficial perivascular lymphocytic infiltrates. A screening serum tryptase level was 49.3 ng/mL (normal level ≤10.9 ng/mL). Further investigative work-up revealed detection of KIT (D816V) mutation by PCR analysis in peripheral blood. A peripheral smear was morphologically unremarkable.
Cytological and pathological findings
A bone marrow biopsy demonstrated normal erythropoiesis, mildly increased eosinophils, increased interstitial population of mast cells in the form of clusters, and perivascular distribution on immunohistochemistry for tryptase, CD2, and CD 25 markers (Figures 1, 2). PCR detected a positive KIT (D816V) point mutation. Repeat serum tryptase level was 56.6 ng/mL. In addition, 30% involvement by small lymphocytic lymphoma was discovered in the form of several intermediate-sized, interstitial lymphoid aggregates composed of small lymphocytes (Figure 3). Flow cytometry showed B-cells positively stained for CD19, CD20, CD5, CD23, and kappa light chains, and the CD38 expression was <5%.
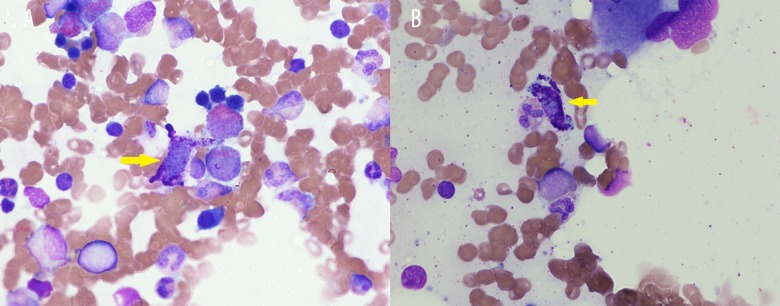
Figure 1.
(A) Bone marrow crush preparation shows mast cell with surrounding maturing granulocytes (Wright stain ×1,000). (B) A degranulating mast cell as indicated by an arrow.
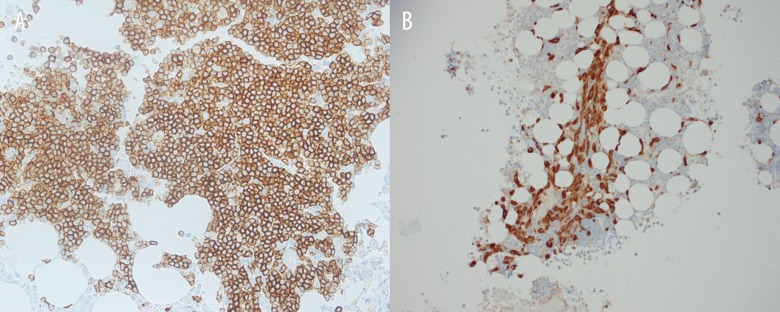
Figure 2.
Immunohistochemical stains show that the lymphoid infiltrate is composed of mainly B-cells (CD20+ A). There is increased interstitial population of mast cells with perivascular accentuation as highlighted by the tryptase stain (B).
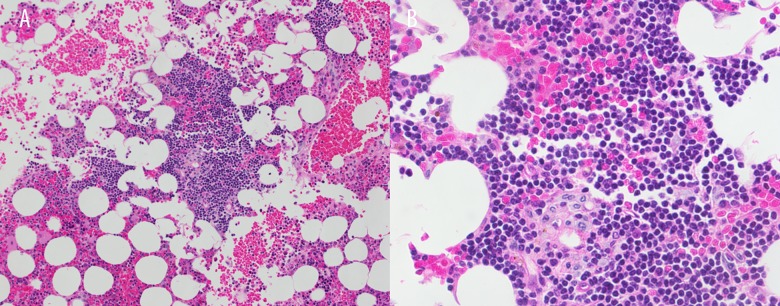
Figure 3.
(A, B) Hematoxylin and eosin sections of clot show bone marrow with several intermediate sized interstitial lymphoid aggregates composed of small lymphocytes. (200×, 500×).
The patient was diagnosed with systemic mastocytosis with an associated clonal hematological non-mast cell lineage disease (SM-AHNMD). She had no SM-related C symptoms at the time of diagnosis. Staging work-up did not show hepatomegaly, splenomegaly, or lymphadenopathy, so the SLL was classified as low-risk early stage SLL. The patient remained under active surveillance for signs of mast cell organ infiltration.
After 12 months following diagnosis, she complained of a blistering rash in the perineal area, and bilateral vulvar lesions were discovered on pelvic examination. A vulvar biopsy confirmed well-differentiated squamous cell carcinoma. A PET scan revealed bilateral inguinal and external iliac lymph node (LN) involvement. She underwent a LN biopsy which revealed involvement by small lymphocytic lymphoma and no evidence of metastatic carcinoma.
Discussion
SM-AHNMD is a rare entity and the majority of AHNMDs are of myeloid origin, the most common being chronic myelomonocytic leukemia [16]. A few cases of systemic mastocytosis with an associated lymphoproliferative disorder have been described in the literature. Among those cases, plasma cell myeloma (SM-PCM) is the most common, followed by monoclonal gammopathy of undetermined significance (SM-MGUS) [11,20]. To our knowledge there is one case report of SM associated with SLL and few case reports of SM-CLL, based on our literature review [16–19].
Lymphoproliferative disorders could be associated with increased number of mast cells; however, the diagnostic criteria for SM-AHNMD as defined by the WHO classification should be met for diagnosis. In our case, the patient fulfilled major SM criterion: multi-focal mast cell clusters (>15 mast cells/cluster) identified by tryptase immunohistochemistry in the bone marrow. She also exhibited several minor criteria: spindle-shaped mast cell morphology, KIT D816 mutation, abnormal CD25 and CD2 expression immunophenotypically, and increased basal serum tryptase level >20 ng/mL.
Immunohistochemistry revealed an aberrant expression of CD 5 and CD 23, thus fulfilling the criteria for B-cell lymphocytic lymphoma which involved 30% of the bone marrow.
KIT codon 816 mutations are variably present in SM-AHNMD patients, depending on the subtype of AHNMD. Sotlar et al. reported absence of KIT 816 mutations in eight patients with lymphoproliferative AHNMDs [21]. Du et al. reported that four known cases of SM-CLL were positive for KIT 816V mutation in mast cells [11]. The KIT D816V mutation is considered a promoter of mast cell differentiation and maturation, rather than proliferation, therefore, mast cells in ISM are non-proliferating and progression to the aggressive forms of SM is rare [9,22,23].
The current treatment recommendations for SM-AHNMD emphasize treating the two entities separately as if they are not associated. Treatment approach for indolent SM includes surveillance, symptom directed therapy in the form of systemic or topical therapy, and cytoreductive therapy [11]. In advanced SM, KIT D816V is considered a major therapeutic target, and cytoreductive therapy options for aggressive cases of SM include tyrosine kinase inhibitors [24]. However, the KIT D816V mutation confers intrinsic resistance to imatinib and masatinib [25–27]. Nilotinib may have efficacy in SM and an overall response rate of 22% was reported in 37 ASM patients, but complete response was not achieved [28]. In a phase II study of tyrosine kinase inhibitors, dasatinib reported an overall response rate of 33% and complete response was observed in two out of 33 patients [29]. More recently, in April 2017, the FDA approved midostaurin, a multikinase inhibitor/KIT inhibitor, based on response rates and duration in a single-arm, open-label study of midostaurin 100 mg orally twice daily. The study showed that the rates of confirmed complete remission (CR) plus incomplete remission (ICR) were 38% for ASM and 16% for SM-AHNMD with six cycles of midostaurin. One patient (5%) with mast cell leukemia achieved CR [30]. Ustun et al. analyzed data in a retrospective study of 57 SM patients to study the role of allogeneic hematopoietic stem-cell transplantation (allo-HCT). They showed a response rate of 70% in all study patients and 68% in the 38 SM-AHNMD patients. Complete remission was observed in 28% of all patients and 26% in SMAHNMD cases [31].
In our case of SM-AHNMD, the SM component was indolent systemic mastocytosis with minimal symptoms of urticaria, and the AHNMD component was low-risk early stage SLL. We offered our patient topical triamcinolone acetonide 0.1% and oral loratadine for symptomatic relief. For the SM component, she is undergoing surveillance for signs of disease progression. A serum chemistry panel, liver function tests, and complete blood count are obtained every three months. After 12 months of follow-up, her serum tryptase level was 65.1 ng/mL. Our SM case showed a stable clinical course without signs of progression during the follow-up period. Since the SM has remained stable with no C findings, our approach has been to wait and watch. The AHNMD component is low-risk SLL and asymptomatic, for which our patient is also under active surveillance. There is no internal organ involvement in the patient’s AHNMD component. She developed vulvar carcinoma and inguinal lymphadenopathy during surveillance, and was found to have involvement by small lymphocytic lymphoma. She is under clinical surveillance for signs of progression and has remained stable with no B symptoms of lymphoma. Median survival in SM-AHNMD largely depends on the associated AHNMD component.
Conclusions
In SM-AHNMD, the combination of systemic mastocytosis associated with small lymphocytic lymphoma is rare and the management strategy follows the principle of treating the two entities individually as if they are not related. Clinical surveillance is indicated for indolent systemic mastocytosis and low-risk small lymphocytic lymphoma to monitor for disease progression.
Acknowledgments
We thank Lydia Howes MSI of Southern Illinois University School of Medicine Library for proofreading the manuscript and organizing the references.
Footnotes
Conflict of interest
None.
References:
- 1.Metcalfe DD. Mast cells and mastocytosis. Blood. 2008;112(4):946–56. doi: 10.1182/blood-2007-11-078097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Golkar L, Bernhard JD. Mastocytosis. Lancet. 1997;349(9062):1379–85. doi: 10.1016/S0140-6736(96)07056-0. [DOI] [PubMed] [Google Scholar]
- 3.Horny HP, Sotlar K, Valent P. Mastocytosis: State of the art. Pathobiology. 2007;74(2):121–32. doi: 10.1159/000101711. [DOI] [PubMed] [Google Scholar]
- 4.Austen KF. Systemic mastocytosis. N Engl J Med. 1992;326(9):639–40. doi: 10.1056/NEJM199202273260912. [DOI] [PubMed] [Google Scholar]
- 5.Castells MC. Mastocytosis: Classification, diagnosis, and clinical presentation. Allergy Asthma Proc. 2004;25(1):33–36. [PubMed] [Google Scholar]
- 6.Brockow K. Epidemiology, prognosis, and risk factors in mastocytosis. Immunol Allergy Clin North Am. 2014;34(2):283–95. doi: 10.1016/j.iac.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 7.Tefferi A, Vardiman JW. Classification and diagnosis of myeloproliferative neoplasms: The 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia. 2008;22(1):14–22. doi: 10.1038/sj.leu.2404955. [DOI] [PubMed] [Google Scholar]
- 8.Valent P, Akin C, Escribano L, et al. Standards and standardization in mastocytosis: Consensus statements on diagnostics, treatment recommendations and response criteria. Eur J Clin Invest. 2007;37(6):435–53. doi: 10.1111/j.1365-2362.2007.01807.x. [DOI] [PubMed] [Google Scholar]
- 9.Lim KH, Tefferi A, Lasho TL, et al. Systemic mastocytosis in 342 consecutive adults: survival studies and prognostic factors. Blood. 2009;113(23):5727–36. doi: 10.1182/blood-2009-02-205237. [DOI] [PubMed] [Google Scholar]
- 10.Wang SA, Hutchinson L, Tang G, et al. Systemic mastocytosis with associated clonal hematological non-mast cell lineage disease: Clinical significance and comparison of chomosomal abnormalities in SM and AHNMD components. Am J Hematol. 2013;88(3):219–24. doi: 10.1002/ajh.23380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Du S, Rashidi HH, Le DT, et al. Systemic mastocytosis in association with chronic lymphocytic leukemia and plasma cell myeloma. Int J Clin Exp Pathol. 2010;3(4):448–57. [PMC free article] [PubMed] [Google Scholar]
- 12.Garcia-Montero AC, Jara-Acevedo M, Teodosio C, et al. KIT mutation in mast cells and other bone marrow hematopoietic cell lineages in systemic mast cell disorders: A prospective study of the Spanish Network on Mastocytosis (REMA) in a series of 113 patients. Blood. 2006;108(7):2366–72. doi: 10.1182/blood-2006-04-015545. [DOI] [PubMed] [Google Scholar]
- 13.Horny HP, Sotlar K, Sperr WR, Valent P. Systemic mastocytosis with associated clonal haematological non-mast cell lineage diseases: A histopathological challenge. J Clin Pathol. 2004;57(6):604–8. doi: 10.1136/jcp.2003.014860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sperr WR, Horny HP, Lechner K, Valent P. Clinical and biologic diversity of leukemias occurring in patients with mastocytosis. Leuk Lymphoma. 2000;37(5–6):473–86. doi: 10.3109/10428190009058500. [DOI] [PubMed] [Google Scholar]
- 15.Sperr WR, Horny HP, Valent P. Spectrum of associated clonal hematologic non-mast cell lineage disorders occurring in patients with systemic mastocytosis. Int Arch Allergy Immunol. 2002;127(2):140–42. doi: 10.1159/000048186. [DOI] [PubMed] [Google Scholar]
- 16.Horny HP, Sotlar K, Stellmacher F, et al. An unusual case of systemic mastocytosis associated with chronic lymphocytic leukaemia (SM-CLL) J Clin Pathol. 2006;59(3):264–68. doi: 10.1136/jcp.2005.026989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sanz MA, Valcarcel D, Sureda A, et al. Systemic mast cell disease associated with B-chronic lymphocytic leukemia. Haematologica. 2001;86(10):1106–7. [PubMed] [Google Scholar]
- 18.Hauswirth AW, Fodinger M, Fritz M, et al. Indolent systemic mastocytosis associated with atypical small lymphocytic lymphoma: A rare form of concomitant lymphoproliferative disease. Hum Pathol. 2008;39(6):917–24. doi: 10.1016/j.humpath.2007.10.022. [DOI] [PubMed] [Google Scholar]
- 19.Ault P, Lynn A, Tam CS, et al. Systemic mastocytosis in association with chronic lymphocytic leukemia: A rare diagnosis. Leuk Res. 2007;31(12):1755–58. doi: 10.1016/j.leukres.2007.04.002. [DOI] [PubMed] [Google Scholar]
- 20.Pardanani A, Lim KH, Lasho TL, et al. Prognostically relevant breakdown of 123 patients with systemic mastocytosis associated with other myeloid malignancies. Blood. 2009;114(18):3769–72. doi: 10.1182/blood-2009-05-220145. [DOI] [PubMed] [Google Scholar]
- 21.Sotlar K, Colak S, Bache A, et al. Variable presence of KITD816V in clonal haematological non-mast cell lineage diseases associated with systemic mastocytosis (SM-AHNMD) J Pathol. 2010;220(5):586–95. doi: 10.1002/path.2677. [DOI] [PubMed] [Google Scholar]
- 22.Mayerhofer M, Gleixner KV, Hoelbl A, et al. Unique effects of KIT D816V in BaF3 cells: Induction of cluster formation, histamine synthesis, and early mast cell differentiation antigens. J Immunol. 2008;180(8):5466–76. doi: 10.4049/jimmunol.180.8.5466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Valent P, Horny HP, Escribano L, et al. Diagnostic criteria and classification of mastocytosis: A consensus proposal. Leuk Res. 2001;25(7):603–25. doi: 10.1016/s0145-2126(01)00038-8. [DOI] [PubMed] [Google Scholar]
- 24.Arock M, Sotlar K, Akin C, et al. KIT mutation analysis in mast cell neoplasms: Recommendations of the European Competence Network on Mastocytosis. Leukemia. 2015;29(6):1223–32. doi: 10.1038/leu.2015.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ma YS, Zeng S, Metcalfe DD, et al. The c-KIT mutation causing human mastocytosis is resistant to STI571 and other KIT kinase inhibitors; kinases with enzymatic site mutations show different inhibitor sensitivity profiles than wild-type kinases and those with regulatory-type mutations. Blood. 2002;99(5):1741–44. doi: 10.1182/blood.v99.5.1741. [DOI] [PubMed] [Google Scholar]
- 26.Akin C, Brockow K, D’Ambrosio C, et al. Effects of tyrosine kinase inhibitor STI571 on human mast cells bearing wild-type or mutated c-kit. Exp Hematol. 2003;31(8):686–92. doi: 10.1016/s0301-472x(03)00112-7. [DOI] [PubMed] [Google Scholar]
- 27.Valent P, Akin C, Sperr WR, et al. Mastocytosis: pathology, genetics, and current options for therapy. Leuk Lymphoma. 2005;46(1):35–48. doi: 10.1080/10428190400010775. [DOI] [PubMed] [Google Scholar]
- 28.Hochhaus A, Baccarani M, Giles FJ, et al. Nilotinib in patients with systemic mastocytosis: Analysis of the phase 2, open-label, single-arm nilotinib registration study. J Cancer Res Clin Oncol. 2015;141(11):2047–60. doi: 10.1007/s00432-015-1988-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Verstovsek S, Tefferi A, Cortes J, et al. Phase II study of dasatinib in Philadelphia chromosome-negative acute and chronic myeloid diseases, including systemic mastocytosis. Clin Cancer Res. 2008;14(12):3906–15. doi: 10.1158/1078-0432.CCR-08-0366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gotlib J, Kluin-Nelemans HC, George TI, et al. Efficacy and safety of midostaurin in advanced systemic mastocytosis. N Engl J Med. 2016;374(26):2530–41. doi: 10.1056/NEJMoa1513098. [DOI] [PubMed] [Google Scholar]
- 31.Ustun C, Reiter A, Scott BL, et al. Hematopoietic stem-cell transplantation for advanced systemic mastocytosis. J Clin Oncol. 2014;32(29):3264–74. doi: 10.1200/JCO.2014.55.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]