Abstract
Selective serotonin reuptake inhibitors (SSRIs) are the most commonly used medications for mood and anxiety disorders, and adult neurogenesis in the dentate gyrus has been shown to be involved in the behavioral effects of SSRIs in mice. Studies have shown the varied effects of chronic treatment with SSRIs on adult neurogenesis. One such effect is the acceleration of neuronal maturation, which affects the functional integration of new neurons into existing neuronal circuitry. In this study, we labeled new neurons by using GFP-expressing retroviral vectors in mice and investigated the effect of an SSRI, fluoxetine, on these neurons at different time points after neuronal birth. Chronic treatment with fluoxetine accelerated the dendritic development of the newborn neurons and shifted the timing of the expression of the maturational marker proteins, doublecortin and calbindin. This accelerated maturation was observed even after sub-chronic treatment, only when fluoxetine was administered during the second week of neuronal birth. These results suggest the existence of a ‘critical period’ for the fluoxetine-induced maturation of new neurons. We propose that the modified functional integration of new neurons in the critical period may underlie the behavioral effects of fluoxetine by regulating anxiety-related decision-making processes.
Introduction
Anxiety and mood disorders are among the most prevalent mental disorders worldwide, with a lifetime prevalence of 16% and 12%, respectively.1 In addition, depression has recently been ranked as the leading cause of burden of disease globally.2 One commonly used type of medication for the treatment of both anxiety and mood disorders is selective serotonin reuptake inhibitors (SSRIs), which exhibit a therapeutic effect only after chronic treatment over multiple weeks.3 The behavioral effects of the SSRIs in mice are dependent on adult neurogenesis in the dentate gyrus,4, 5 but the mechanism by which newborn neurons contribute to the behavioral effects is not well understood.
Newborn neurons are generated from neuronal precursor cells located in the subgranular zone, and those neurons that survive the first few weeks are incorporated into the existing circuitry as granule cells, which are excitatory principal neurons in the dentate gyrus.6 Chronic SSRI treatment has been shown to increase the proliferation of neuronal precursor cells and the survival of newborn neurons in rodents, although these effects vary depending on mouse strains, age, stress and corticosterone levels.4, 7, 8, 9, 10, 11, 12, 13
Soon after their birth, the newborn neurons begin to expand their dendritic trees, which continue to increase their size for a month.14 The dendritic growth has been shown to be regulated by neuronal activity15, 16, 17, 18 and enhanced by hippocampus-dependent learning.19 These activity- and experience-dependent regulations occur during a period of early neuronal maturation, in which new neurons play a role in hippocampus-dependent learning.20, 21, 22 Therefore, the dendritic development and resulting formation of new circuits may be a closely regulated process that determines how new neurons contribute to brain functions.
Chronic treatment with a commonly prescribed SSRI, fluoxetine, has been found to affect the dendritic arborization of immature neurons that express doublecortin (DCX).13 The exact age of affected new neurons is unclear, because the age range of DCX-expressing cell populations is broad and may be also shifted by accelerated neuronal maturation. Considering that altered dendritic development may mediate the behavioral effects, we examined how chronic fluoxetine treatment affects the dendritic arborization of new neurons at different time points during their maturation in mice.
Materials and methods
Mice
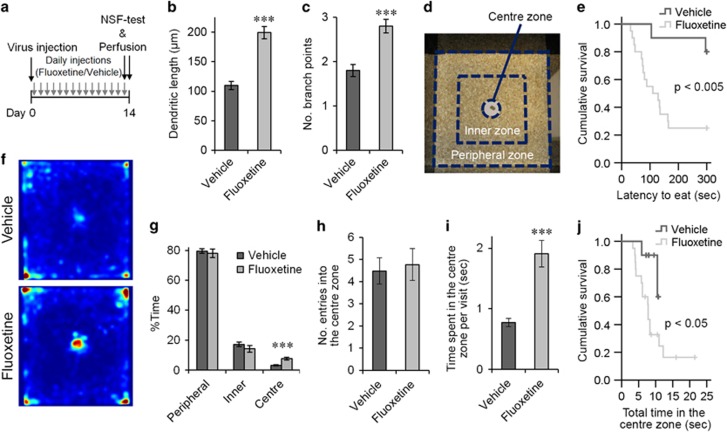
The experimental and caring procedures for mice were approved by the Norwegian Animal Research Authority or the Institutional Animal Care and Use Committee of the Biological Resource Centre at Biopolis, Singapore. We used both female (F) and male (M) C57BL/6J mice aged 6–7 weeks (Figures 1 and 3, 14 days, fluoxetine treatment (flx): 6F, vehicle treatment (veh): 6F, 21 days, flx: 6 M, veh: 4 M+2F, 28 days, flx: 3F+3 M, veh: 4F+2 M; Figure 2, flx: 3F+3 M, veh: 3F+4 M; Figure 4, treatment day 7–14, flx: 3F+3 M, veh: 4F+2 M, day 0–7, flx: 6 M, veh: 6 M; Figure 5, flx: 20 M, veh: 10 M). The mice were housed in acrylic cages with ad libitum access to food and water under 12-h light/12-h dark cycle conditions, with the exception of 12 h of food deprivation before the novelty-suppressed feeding test, as described below. No randomization and blinding were done.
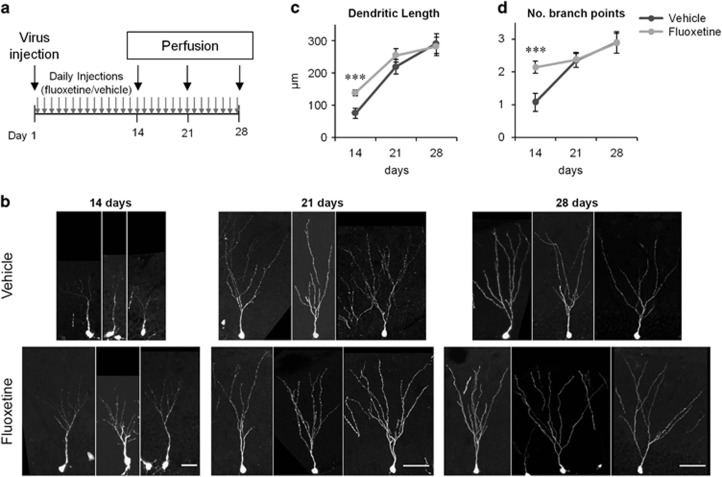
Figure 1.
Fluoxetine-induced transient increase in the dendritic arborization of new neurons. (a) Experimental time line. (b) Examples of GFP+ granule cells from the fluoxetine- or vehicle-treated mice 14, 21 and 28 days after virus injection. Scale bars, 25 μm (day 14), 50 μm (day 21 and 28). (c and d) Total dendritic length (c) and number of branch points (d) of GFP+ neurons on days 14, 21 and 28. ***P<0.005, two-tailed t-test with Bonaferroni correction.
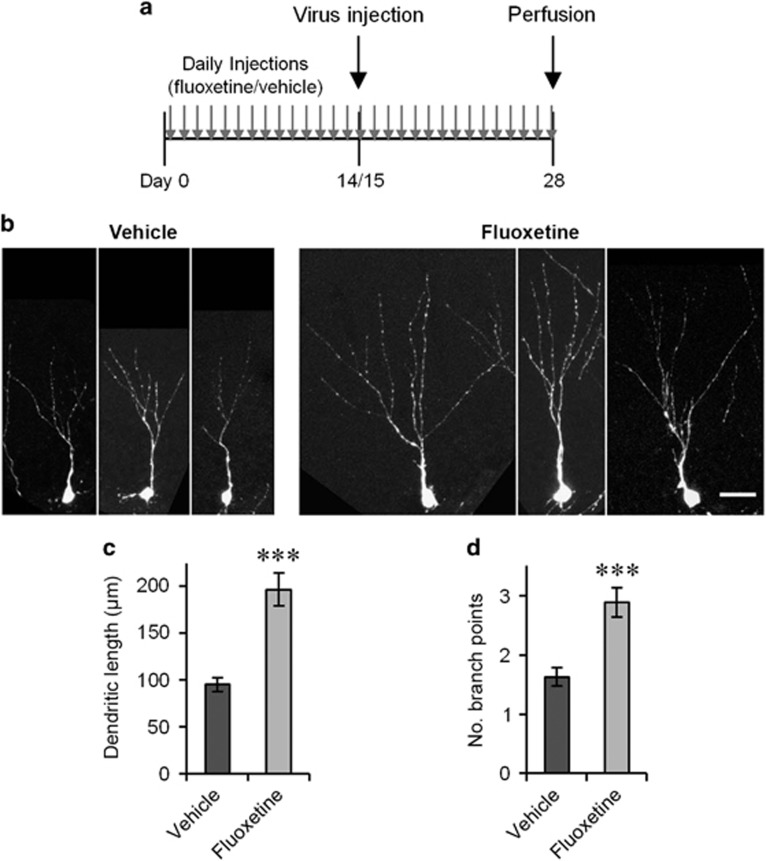
Figure 2.
The morphological effect on immature neurons is maintained after 4 weeks of fluoxetine treatment. (a) Experimental time line. (b) Example images of GFP+/Prox1+ neurons 2 weeks after virus injection. Scale bar, 25 μm. (c and d) Total dendritic length (c) and number of branch points (d) of GFP+/Prox1+ neurons on day 14. ***P<0.005, two-tailed t-test.
Production and injection of viral vectors
The procedures for the production and injection of viral vectors have been previously described in detail.23 We used a retroviral construct containing the green fluorescent protein gene under the control of the CAG promoter, which contains the CMV early enhancer, chicken beta actin promoter and splice acceptor of the rabbit beta-globin gene (RV-CAG-GFP).14 The mice were anesthetized with isoflurane gas, and the viral vector was bilaterally injected into the dentate gyrus using a stereotactic coordinate of 2.0 mm posterior and 1.5 mm lateral (each hemisphere) to the bregma and 2.3 mm ventral from the skull surface.
Fluoxetine injections
The mice were given intraperitoneal injections with fluoxetine (methyl[3-phenyl–3-[4-(trifluormethyl)phenoxy]propyl]ammoniumchlorid, Sequoia Research Products, Pangbourne, UK) at a dose of 18 mg kg−1 body weight in 0.9% saline with 0.3% Tween 20 (Cat#P1379, Sigma-Aldrich, St. Louis, MO, USA) or vehicle (0.9% saline with 0.3% tween 20) daily at ~1300 hours for the number of days specified in the Results section for each experiment.
Novelty-suppressed feeding test
The novelty-suppressed feeding test4 was performed 14 (control: n=8, fluoxetine: n=16) or 15 (control: n=2, fluoxetine: n=4) days after viral injection. The mice were deprived of food for 12 h before the test. The test was then performed in a novel environment, which was an open field (60 cm × 60 cm square with black walls). A food pellet was placed on a small white paper platform in the center of the open field. The room had dim lighting, and the floor was covered with the same type of bedding as used in the mice’s home cage. New bedding was used for each mouse. The test was started when a mouse was placed in the corner closest to the position of the experimenter, which was the same corner for all mice. The test was stopped when the mouse started to eat the pellet or when the time limit of 5 min had been reached. The time from start to eating was recorded as 300 s when the time limit was passed. After the test, the mice were transferred back to their home cage, and the amount of food consumed in 10 min was measured. The test was captured with an overhead mounted camera into video files, which were used to track the positions of the mouse head (not the center of the body) using the ANY-maze software (Stoelting, Wood Dale, IL, USA). The floor of the open field was divided into 25 (5 × 5) squares. In total 16 squares along walls were defined as a peripheral zone. The remaining nine squares except the area of the paper platform were defined as an inner zone. A circular area around the paper platform was defined as the central zone (Figure 5d). The time spent in each zone was measured. For occupancy plots, the tracked position data from the ANY-maze software were smoothened with two-dimensional Gaussian filter and used to calculate the time spent in each position of the open field using Matlab software (Mathworks, USA).
Histology
Preparation of 40-μm-thick brain sections and immunostaining were performed, as previously described.24, 25 The primary antibodies used were goat anti-DCX (1:500 dilution, SC-8066, Santa Cruz Biotechnology, Dallas, TX, USA), rat anti-GFP (1:1000, Cat# 04404-84, Nacalai Tesque, Japan), rabbit anti-Prox1 (1:1000, Cat# AB5475, Millipore, Germany, and Cat# PRB-238C, Covance, Princeton, NJ, USA), goat anti-Prox1 (1:40, Cat# AF 2727, R&D Systems, Minneapolis, MN, USA), and rabbit anti-calbindin (1:1000, Cat#ab11426, Abcam, UK). The secondary antibodies used were anti-rat-AlexaFluor488 (Cat# 712–545–153), anti-rabbit-Cy3 (Cat# 712–165–152), anti-rabbit-DyLight549 (Cat# 711–505–152), anti-rabbit-DyLight649 (Cat# 711–495–152), anti-goat-Cy3 (Cat #705–165–003), and anti-goat-Cy5 (Cat # 705–175–147). All secondary antibodies were raised in donkey, purchased from Jackson Immuno (West Grove, PA, USA) and used at 1:600 dilutions. The brain sections were mounted on glass slides with anti-fading mounting medium.
Confocal microscopy
Confocal images were taken using LSM710 confocal microscopes (Zeiss, Germany) equipped with 488-nm, 543-βnm and 633-nm laser lines and the ZEN image-acquisition software. A 40x objective lens (NA 1.3) was used for the co-localization analysis, and a 20x objective lens (NA 0.8) was used for the neuronal morphology analysis. For neuronal morphology analyses, neurons in z-stacks from 12 brain sections distributed over the full anterior-posterior axis containing the granule cell layer (every sixth section) were analyzed for each mouse. From each z-stack, we selected either a few GFP-positive (GFP+) cell bodies randomly or all GFP+ cell bodies which were in the infra- or suprapyramidal blade (not in the hinge area) and analyzed the dendrites connected to the cell bodies by measuring their lengths and numbers of branch points. For all the analyses at 2-week time points, Prox1 immunoreactivity was used to determine the granule cell identity of GFP+ cells. Prox1-negative(Prox1−)/GFP+ cells were removed from the analyses. For 3-week and 4-week time points, the identification of granule cells was easily performed based on their morphology. Therefore, Prox1+ immunoreactivity was used only for the half of mice distributed equally between control and fluoxetine treated mice. For the alternative method used for Supplementary Figure 1, we imaged all GFP+ dendrites in the entire dentate gyrus in either 6 sections (every 12th section) or 36 sections (every second section). For each mouse, the total length of all the GFP+ dendrites from all sections was measured whether the GFP+ dendrites were connected to cell bodies or not. Then this total dendritic length was divided by the number of cell bodies found in the same set of sections to calculate dendritic length per cell (Supplementary Figure 1). For the co-localization analysis of GFP/Prox1/DCX or GFP/Prox1/calbindin, we imaged and analyzed all GFP+ cells in the granule cell layer of both hemispheres from 6 to 12 sections (every 12th or 6th section). Percentage of DCX+ or calbindin+ cells in Prox1+ cells was analyzed from three images from three different sections (every 24th section), each of which represents one focal plane. All image analyses were performed using the ImageJ software (National Institutes of Health, USA), with the NeuronJ plugin26 for the neuronal morphology analysis.
Statistics
Statistical analyses were performed using SPSS Statistics software (IBM, Armonk, NY, USA) and Excel 2010 (Microsoft, Redmond, WA, USA). For independent t-tests, Levene’s test for equal variance was performed, and depending on the P-value, a t-test with (P>0.05) or without (P<0.05) equal variance assumption (Student’s or Welch’s t-test, respectively) was used. When sample numbers were low (n=4–6 for each group), Mann–Whitney U tests were used to compare two groups. For the analysis of latency to eat the food pellet (Figure 5e) and total time spent in the central zone (Figure 5j), we performed a Kaplan–Meier survival analysis with the log-rank test.27 Eating the food pellet was treated as an event, while mice were censored if their test duration reached the time limit of 300 s. All data are presented as the mean±s.e.m.
Results
Chronic fluoxetine treatment induces a transient increase in the dendritic arborization of new neurons
To examine the effect of chronic fluoxetine treatment on the morphology of new neurons, we injected mice with a GFP-expressing retroviral vector. Then, we administered daily fluoxetine or vehicle treatment until 14, 21 or 28 days after viral injection, at which point we collected mouse brains and prepared sections (Figure 1a). We quantified the extent of dendritic arborization of GFP+ cells by two parameters, total dendritic length and the number of branch points. We observed a significant increase in both parameters in fluoxetine-treated mice compared with vehicle-injected mice on day 14 (Figures 1c and d; flx: n=77 cells, veh: n=78 cells, P<0.005 for each, two-tailed t-test with Bonaferroni correction). In contrast, we did not observe any difference between the fluoxetine- and vehicle-treated mice on day 21 or 28 (Figures 1c and d; day 21, flx: n=149 cells, veh: n=105 cells, day 28, flx: n=65 cells, veh: n=78 cells, P>0.05 for both parameters at both time points, two-tailed t-tests with Bonaferroni correction). Thus, chronic fluoxetine treatment transiently increased dendritic arborization on day 14, but this increase disappeared at later time points despite continued fluoxetine treatment. As expected, dendritic arborization is increased during the maturation from days 14 to 28 in both the vehicle- and fluoxetine-treated mice (P<0.001 for total dendritic length for both treatments, two-tailed t-tests), indicating that the new neurons have not reached full maturity in terms of dendritic morphogenesis on day 14, with or without fluoxetine treatment.
The effect on dendritic arborization is transient along neuronal maturation
In the previous experiment, we observed the transient effect of chronic fluoxetine treatment on day 14. This observation suggests two possibilities. The first possibility is that the effect is transient in terms of treatment duration such that the effect observed with 2-week treatment dissipates after additional days of treatment. The other possibility is that the effect is transient along the maturation of individual neurons such that the effect observed in 2-week-old neurons disappears when new neurons become more mature. To separate these two possibilities, we provided chronic fluoxetine or vehicle treatment for 28 days starting on day 0 and injected the GFP-expressing retroviral vector on day 14 or 15 (Figure 2a). In this manner, we examined the effect on 2-week-old GFP+/Prox1+ (positive for both GFP and Prox1) cells after 28 days of treatment. We observed a significant increase in total dendritic length and the number of branch points in the fluoxetine-treated mice compared with the vehicle-treated mice (Figures 2b–d; flx: n=89 cells, veh: n=105 cells, P<0.001 for both, two-tailed t-tests). This result indicates that the effect on 2-week-old neurons is not transient in terms of treatment duration but is transient along the maturation of individual neurons.
Chronic fluoxetine treatment accelerates the maturation of new neurons
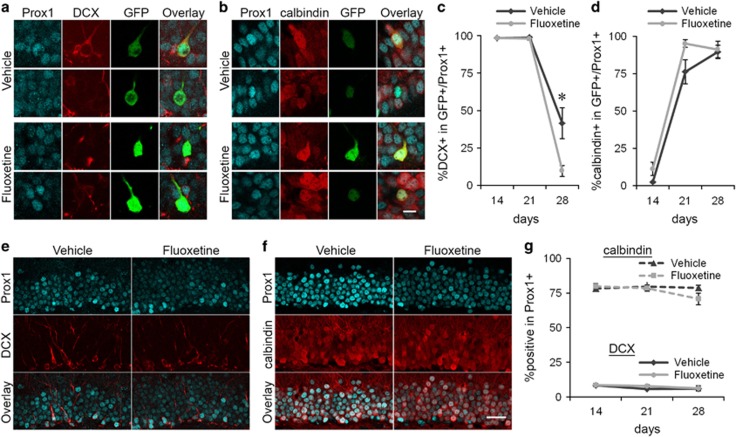
Next, we examined the expression of DCX and calbindin (Figures 3a and b). These marker proteins are down- and up-regulated, respectively, along the maturation of new neurons and are thus used as indicators for neuronal maturity. Consistent with previous studies,28, 29 the percentage of GFP+/Prox1+ cells expressing DCX decreased from days 14 to 28 in vehicle-treated mice, whereas the percentage of GFP+/Prox1+ cells expressing calbindin increased during the same period (Figures 3c and d). These changes were also observed in fluoxetine-treated mice along neuronal maturation. The percentages of GFP+/Prox1+ cells expressing DCX were comparable between fluoxetine- and vehicle-treated mice on days 14 and 21 (Figure 3c, day 14, flx: n=6 mice (913 cells), veh: n=6 mice (851 cells), day 21, flx: n=6 mice (689 cells), veh: n=5 mice (316 cells), P>0.05 for both, Mann-Whitney U tests with Bonferroni correction), with nearly all cells expressing DCX. In contrast, the percentage on day 28 was significantly lower in the fluoxetine-treated mice than the vehicle-treated mice (Figure 3c, flx: n=6 mice (519 cells), veh: n=6 mice (228 cells), P<0.05, Mann-Whitney U tests with Bonferroni correction), indicating that chronic fluoxetine treatment shut off the DCX expression in new neurons earlier. The proportion of DCX-expressing cells in the vehicle-treated mice after 28 days is consistent with previous studies.18, 30 The comparable percentages of GFP+/Prox1+ cells were positive for calbindin between the fluoxetine- and vehicle-treated mice on days 14 and 28 (Figure 3d, day 14, flx: n=6 mice (1220 cells), veh: n=6 mice (1147 cells), day 28, flx: n=4 mice (528 cells), veh: n=4 mice (191 cells), P>0.05 for both, Mann-Whitney U tests with Bonferroni correction), with nearly none and all of them expressing calbindin, respectively. On day 21, the percentage showed a trend of increase in the fluoxetine-treated mice compared with the vehicle-treated mice while it is not significant (Figure 3d, flx: n=6 mice (478 cells), veh: n=6 mice (262 cells), P>0.05, Mann-Whitney U test with Bonferroni correction). The effects on DCX expression indicate that chronic fluoxetine treatment accelerates the maturation of new neurons.
Figure 3.
Fluoxetine treatment shifts maturational changes in the expression of DCX and calbindin in new neurons. The experimental time line is described in Figure 1a. (a) Representative images of two GFP+/Prox1+ cells each from the vehicle- and fluoxetine-treated mice on day 28. One of them expresses DCX. Red: DCX, Cyan: Prox1, Green: GFP. (b) Representative images of two GFP+/Prox1+ cells each from the vehicle- and fluoxetine-treated mice on day 28. Three of them express calbindin. Red: calbindin, Cyan: Prox1, Green: GFP. (c, d) Percentages of DCX+ cells in GFP+/Prox1+ cells (c) and calbindin+ cells in GFP+/Prox1+ cells (d) on days 14, 21, and 28. *P<0.05, Mann–Whitney U test with Bonaferroni correction. (e) Representative images of the granule cell layer from vehicle- and fluoxetine-treated mice. Red: DCX, Cyan: Prox1. (f) Representative images of the granule cell layer from the vehicle- and fluoxetine-treated mice. Red: calbindin, Cyan: Prox1. (g) Percentage of DCX+ cells and calbindin+ cells in Prox1+ cells on days 14, 21 and 28. Scale bars, 10 μm (a and b), 30 μm (e and f).
We also quantified the percentages of Prox1+ cells expressing DCX or calbindin (Figures 3e and f) to evaluate the effect on the entire granule cell population. We did not find any significant difference between the fluoxetine- and vehicle-treated mice at either analyzed time point for DCX (Figure 3g, day 14, flx: n=6 mice (16484 cells), veh: n=6 mice (15377 cells), day 21, flx: n=5 mice (15087 cells), veh: n=5 mice (17343 cells), day 28, flx: n=6 mice (11562 cells), veh: n=6 mice (6038 cells), P>0.05 for all, Mann–Whitney U tests with Bonferroni correction], or for calbindin (Figure 3g, day 14, flx: n=6 mice (16281 cells), veh: n=6 mice (19247 cells), day 21, flx: n=6 mice (14555 cells), veh: n=6 mice (14371 cells), day 28, flx: n=4 mice (10801 cells), veh: n=4 mice (11378 cells), P>0.05 for all, Mann–Whitney U tests with Bonferroni correction). It has been previously described that chronic fluoxetine treatment causes the ‘dematuration’ in the dentate gyrus, which is defined as alteration of neuronal properties to more immature states, such as the reduction of calbindin expression.31 There was a decreasing trend in the percentage of Prox1+ cells expressing calbindin on day 28 in the fluoxetine-treated mice. Although this trend may be reminiscent of dematuration, we did not detect a significant difference supporting it.
Fluoxetine-induced dendritic arborization requires treatment during the second week after neuronal birth
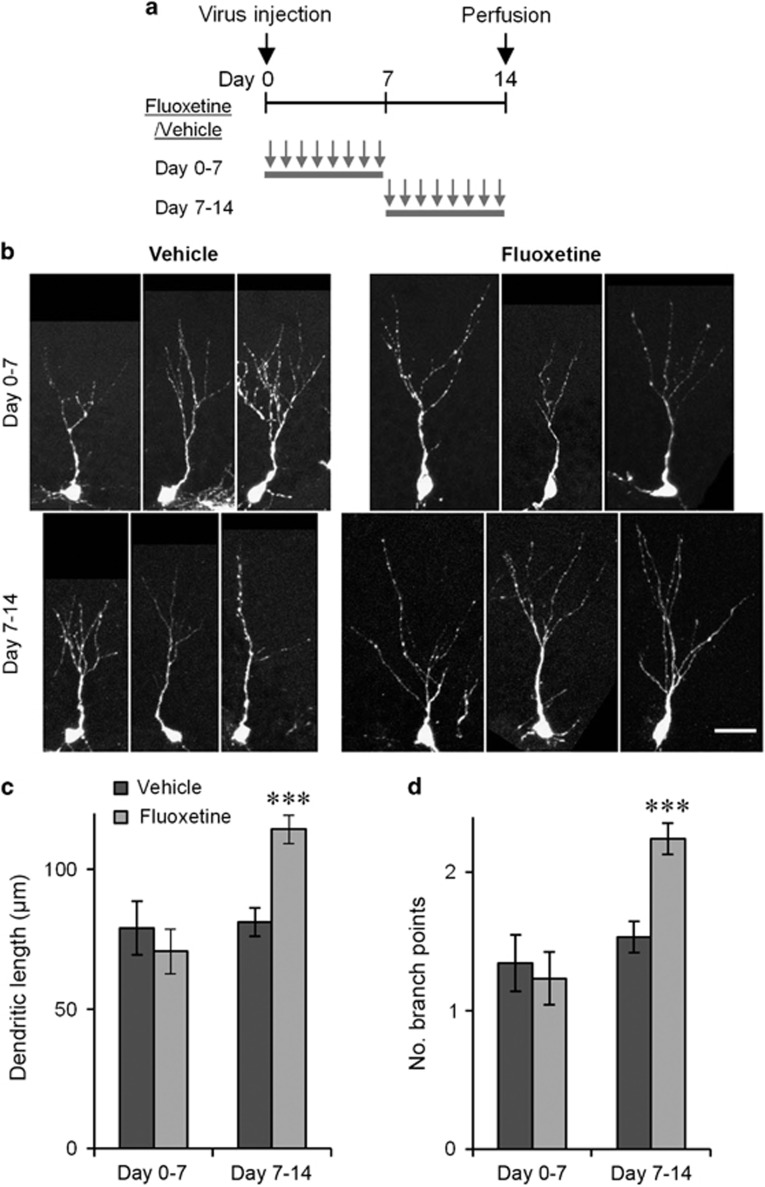
Our results thus far demonstrated that 2-week fluoxetine treatment starting from the day of virus injection increased the extent of dendritic arborization of new neurons measured on the last day of fluoxetine treatment. Next, we examined whether a shorter treatment is sufficient to cause the same effect and, if so, which maturational period is liable. Thus, we injected the viral vector on day 0 and administered the fluoxetine or vehicle treatment daily from days 0 to 7 or from days 7 to 14 and analyzed the morphology of GFP-expressing neurons on day 14 (Figure 4a). For the treatment from day 0 to 7, we did not find a significant difference in total dendritic length or the number of branch points between the fluoxetine- and vehicle-treated mice (Figures 4c and d; flx: n=90 cells, veh: n=90 cells, P>0.05 for both, two-tailed t-tests with Bonaferroni correction). In contrast, for the treatment from days 7 to 14, the total dendritic length and number of branch points of new neurons were significantly higher in the fluoxetine-treated mice than in the vehicle-treated mice (Figures 4c and d; flx: n=484 cells, veh: n=365 cells P<0.001 for both, two-tailed t-tests with Bonaferroni correction). These results indicate that seven days of fluoxetine treatment is sufficient to increase the dendritic arborization and that the effect requires fluoxetine treatment during the second week after neuronal birth.
Figure 4.
Fluoxetine-induced dendritic arborization requires treatment during the second week after neuronal birth. (a) Experimental time lines. (b) Example images of GFP+/Prox1+ neurons on day 14 with treatment on days 0–7 or days 7–14. Scale bar, 25 μm. (c) Total dendritic length (c) and number of branch points (d) of GFP+/Prox1+ neurons with treatment on days 0–7 or days 7–14. ***P<0.005, two-tailed t-test with Bonaferroni correction.
Association between the dendritic complexity of immature neurons and anxiety-related behavior
To examine whether this morphological effect is associated with a behavioral effect, we injected the GFP-expressing retroviral vector and administered chronic fluoxetine or vehicle treatment for two weeks before the mice were tested in the novelty-suppressed feeding test (Figure 5a). Consistent with previous experiments, GFP+/Prox1+ neurons in the fluoxetine-treated mice had increased dendritic arborization compared with those in the vehicle-treated mice, as quantified by the total dendritic length and number of branch points (Figures 5b and c; flx: n=299 cells, veh: 150 cells, P<0.001 for both, two-tailed t-tests). For the novelty-suppressed feeding test, a food-deprived mouse was placed in a novel environment, which was an open field with a food pellet at its center (Figure 5d), and the latency to initiating the consumption of the food pellet was measured as an indication of the mouse’s anxiety level. The fluoxetine-treated mice had a significant reduction in latency compared with the vehicle-treated mice (Figure 5e, flx: n=20 mice, veh: n=10 mice, P<0.005, Kaplan–Meier survival analysis, Log-rank test), indicating a reduced anxiety level in the fluoxetine-treated mice. After the test, the amount of pellet consumed during the first 10 min in the home cage was measured for each mouse. The amount was significantly less in the fluoxetine-treated mice than the vehicle-treated mice (flx: 0.10±0.01 g, n=19 mice, veh: 0.16±0.02 g, n=10 mice, P<0.05, two-tailed t-test), indicating that the reduced latency in the novelty-suppressed feeding test was not caused by an increased appetite in the fluoxetine-treated mice.
Figure 5.
The morphological effect of fluoxetine treatment is associated with the behavioral effect in the novelty-suppressed feeding test. (a) Experimental time line. NSF-test: novelty-suppressed feeding test. (b and c) Total dendritic length (b) and number of branch points (c) of GFP+/Prox1+ neurons from the vehicle- and fluoxetine-treated mice. (d) Open field used for the novelty-suppressed feeding test and subdivision into the central, inner and peripheral zones. (e) Latency to eat the food pellet. The crosses indicate the time points at which data were censored because of the maximum time limit. (f) Percentage time spent in the central, inner and peripheral zones. (g) Color-coded average occupancy plots of the time that animals spent in individual positions. Warmer colors indicate a higher percentage of time spent in that position. There was no significant difference between the vehicle- and fluoxetine-treated mice in terms of the percentage time spent in the corners (P>0.05, two tailed t-test). (h) Number of entries into the central zone per 100 s. (i) Time spent in the central zone per visit. (j) Total time spent in the central zone. The crosses indicate the time points at which data were censored because the tests reached the maximum time limit. ***P<0.005, two-tailed t-test.
We further analyzed the behavior of the mice by tracking their movements in the open field, which had been divided into central, inner and peripheral zones (Figure 5d). The fluoxetine-treated mice spent a significantly higher proportion of time in the central zone compared with the vehicle-treated mice (Figures 5f and g, flx: n=20 mice, veh: n=10 mice, P=0.001, two-tailed t-test). In contrast, we did not observe a difference between the vehicle- and fluoxetine-treated mice in terms of the proportion of time spent in the peripheral and inner zones (Figures 5f and g, P>0.05 for both, two-tailed t-tests). No significant differences were observed in the number of entries to the central zone per unit time between the two groups of mice (Figure 5h, P>0.05, two-tailed t-test). However, each visit to the central zone was longer for the fluoxetine- compared with vehicle-treated mice on average (Figures 5i, P<0.001, two-tailed t-test). To examine how long mice explored the food pellet and its vicinity before eating the pellet, we analyzed the total time spent in the central zone (Figure 5j). The fluoxetine-treated mice stayed in the central zone for a significantly shorter amount of time before eating the pellet than the vehicle-treated mice (P<0.05, Kaplan–Meier survival analysis, log-rank test).
Discussion
Critical period for the fluoxetine-induced accelerated maturation of new neurons
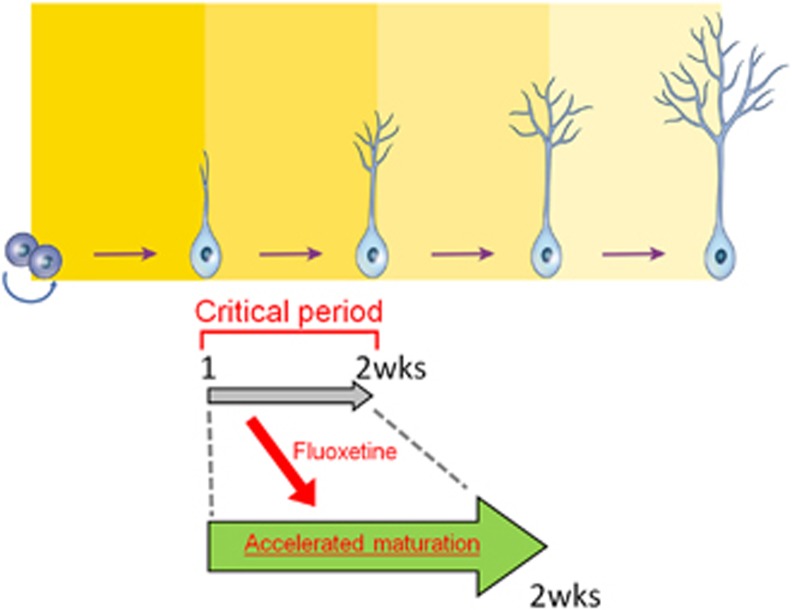
In this study, we labeled newborn neurons in the dentate gyrus using a GFP-expressing retroviral vector and found that chronic fluoxetine treatment increased the dendritic complexity of new neurons when they were examined 14 days after retroviral vector injection (Figures 1 and 2). These effects occurred in both female and male mice (Supplementary Table 1). In addition, chronic fluoxetine treatment shifted the timing of maturational changes in marker protein expression, DCX and calbindin to earlier after the retroviral injection (Figure 3). Altogether, these findings suggest that chronic fluoxetine treatment results in an accelerated maturation of the new neurons, which is consistent with a previous study.13 Our results further indicated the existence of a critical period for this fluoxetine-induced maturation. Fluoxetine treatment increased the dendritic complexity of GFP-expressing new neurons 14 days after retroviral vector injection, whereas this effect was not found at later time points (Figure 1). An alternative method for dendritic length measurement indicated that this disappearance of the effect at later time points is not due to a technical limitation caused by the use of brain sections (Supplementary Figure 1). Further, the morphological effects were observed only when the fluoxetine treatment was given during the second, but not the first, week after retroviral injection (Figure 4), indicating the unique property of the second week after neuronal birth (Figure 6). Thus, this time window can be regarded as a ‘critical period’ for fluoxetine-induced maturation of new neurons. By two weeks of age, the newly generated neurons integrate into existing neural circuits by receiving synaptic inputs16, 32, 33 and forming synaptic outputs to hilar and CA3 targets under physiological conditions.34, 35, 36 The fluoxetine-induced maturation of these neurons may facilitate their integration into the local circuitry and thereby strengthen their contribution to brain functions, which also occurs with exposure to an enriched environment.37
Figure 6.
Schematic showing that fluoxetine treatment during the critical period of the second week after neuronal birth results in the accelerated maturation of new neurons.
The same maturational period has been suggested as a critical period for the experience-specific functional modification of new neurons.25 Exposure to an enriched environment during the second week after neuronal birth has been shown to maximize the survival of new neurons and to make more new neurons responsive to re-exposure to the same environment at a later time point. Furthermore, spatial learning during this critical period increased the dendritic arborization,19 enhanced the survival of adult born neurons and made the neurons more responsive to the same task at a later time point.38 Our finding further strengthens the idea that the second week after neuronal birth is a critical period for determining the morphological and functional integration of new neurons in the dentate gyrus.39 Studies have provided evidence for the contribution of new neurons around this critical period to brain functions.20, 21, 22 In particular, a series of studies by Deng et al.22, 40 used a genetic approach to transiently suppress adult neurogenesis and showed that the absence of the 1- to 3-week-old neurons resulted in impaired long-term retention of spatial memory and delayed extinction of contextual fear conditioning22 as well as longer latency to eat in the novelty-suppressed feeding test.40
Sub-chronic fluoxetine treatment is sufficient for accelerated maturation
The behavioral effects of fluoxetine require chronic treatment over multiple weeks both in human patients3 and animals.41 Furthermore, 14 days of treatment consistently affected the performance of mice in the novelty-suppressed feeding test (Figure 5). In contrast, sub-chronic treatment over 5–7 days has been shown to fail to cause detectable behavioral changes in the novelty-suppressed feeding test.41 In parallel, studies have shown that chronic, but not sub-chronic, treatment increases cell proliferation in the dentate gyrus,13, 42 which suggests that increased neurogenesis may be a mechanism underlying the behavioral effects. Increased cell proliferation is an attractive potential mechanism underlying the behavioral effects of fluoxetine. However, because the newborn cells generated by cell proliferation are not immediately functional, the network requires additional weeks before gaining increased numbers of neurons become mature and functional. Moreover, other studies did not observe increased cell proliferation in animals showing the behavioral effects,10, 11 indicating that increased cell proliferation may not be an absolute requirement for the behavioral effects.
An alternative possibility is that the primary effects of fluoxetine start to appear after sub-chronic treatment, and the accumulation of these effects over multiple weeks leads to the behavioral changes. In this study, sub-chronic fluoxetine treatment over 7 days increased the dendritic complexity of new neurons (Figure 4). Because 7 days of fluoxetine treatment is not sufficient for behavioral effects in the novelty-suppressed feeding test, the morphological/maturational effects observed here precede the appearance of the behavioral effects. A 7-day treatment period affects the dendritic complexity of new neurons that are in the critical period. However, this effect by itself would not be sufficient to generate robust behavioral changes. Younger neurons that were not in the critical period are not affected during the first 7 days of treatment. However, with an additional 7 days of treatment, these younger neurons come into the critical period and are thereby affected by the treatment. Then, after 14 days of fluoxetine treatment, more new neurons undergo the morphological/maturational effects and integrate into the neural circuit differently. This accumulation of larger numbers of new neurons affected during their individual critical periods may be the basis of fluoxetine-induced behavioral changes.
A recent study showed that the direct target of serotonin signaling modulated by fluoxetine is mature granule cells but not new neurons.29 The same study suggested that new neurons are also required as a downstream mediator through brain-derived neurotrophic factor (BDNF) signaling from mature granule cells. These findings raise the possibility that our observation of fluoxetine-induced accelerated maturation of new neurons may be mediated indirectly through mature granule cells. In line with this possibility, a study suggested that TrkB, a high-affinity receptor of BDNF, in new neurons is involved in the dendritic development of new neurons in the dentate gyrus,43 supporting the hypothesis that BDNF is a mediator of the accelerated maturation during the critical period. Moreover, BDNF and the TrkB receptor have been shown to play a role in the action of fluoxetine.44
What anxiety-related process is affected by chronic fluoxetine treatment?
In addition to the accelerated maturation of new neurons, 14 days of fluoxetine treatment was observed to affect behavior in the novelty-suppressed feeding test, consistent with previous studies.4 In the novelty-suppressed feeding test, food-deprived mice undergo a conflict between a drive to approach and consume food and an aversion from eating in a novel unprotected environment. Their behavior under this conflict is thought to be modulated by the anxiety level of mice,45 and the latency to start eating a food pellet is used as an index for anxiety level.4 This latency appears to be affected by at least four decision-making processes of the mice. First, mice must choose to run away from the walls and into an open field. Otherwise, in a novel environment, mice have a tendency to stay in protective areas, such as near walls, a behavior called thigmotaxis.46, 47 Second, in an open field, mice must decide to stop near the food pellet to explore it. Third, even when mice stay close to the food pellet, they require a certain amount of time to explore the food and its surroundings before consuming it. During this period, mice may decide to run away from the food pellet. Fourth, otherwise mice may decide to eat the food pellet.
Our results indicate that chronic fluoxetine treatment does not affect the first two decisions but does affect the latter two. Neither proportions of time in the peripheral or inner zone were different between the vehicle- and fluoxetine-treated groups (Figure 5g), indicating that the first decision is not affected by chronic fluoxetine treatment. The frequencies of entry into the central zone per unit time were not different between the two groups either (Figure 5h), which suggests that the second decision is not affected. In contrast, time spent in the central zone during individual visits was significantly increased in the fluoxetine-treated group (Figure 5i), which suggests that chronic fluoxetine treatment shifted the third decision toward not running away from the food pellet. The total time spent in the central zone before eating the food pellet was reduced in fluoxetine-treated mice (Figure 5j), indicating that chronic fluoxetine treatment facilitates mice to make a final decision regarding whether to eat the food pellet. These dual effects on the two separate decision-making processes may cooperate to make the novelty-suppressed feeding test robustly sensitive to detect the behavioral effects of chronic fluoxetine treatment.
In conclusion, our results identified a critical period for the fluoxetine-induced accelerated maturation of new granule cells and suggested a possibility that this accelerated maturation affects anxiety-related decision-making processes. Further investigation of down- and upstream mechanisms for the fluoxetine-induced accelerated maturation of new neurons would be required to fully understand the therapeutic actions of SSRIs and to facilitate the development of novel therapies of mood disorders.
Acknowledgments
We would like to thank Ms Chika Yoshii and Teruyo Tashiro for their technical assistance, Dr Menno Witter for the use of microscopes in his laboratory, and Drs Alexandre Surget, Paul Anderson and Stefan Blankvoort for their comments. This work was supported by FUGE Midt-Norge (to IÅ), Kontaktutvalget St Olavs-NTNU, James S. McDonnell foundation, Nanyang Technological University and the University of Warwick (to AT).
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
The authors declare no conflict of interest.
Supplementary Material
References
- Kessler RC, Aguilar-Gaxiola S, Alonso J, Chatterji S, Lee S, Ormel J et al. The global burden of mental disorders: an update from the WHO World Mental Health (WMH) surveys. Epidemiol Psichiatria Soc 2009; 18: 23–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith K. Mental health: a world of depression. Nature 2014; 515: 181. [DOI] [PubMed] [Google Scholar]
- Blier P. The pharmacology of putative early-onset antidepressant strategies. Eur Neuropsychopharmacol: J Eur Coll Neuropsychopharmacol 2003; 13: 57–66. [DOI] [PubMed] [Google Scholar]
- Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003; 301: 805–809. [DOI] [PubMed] [Google Scholar]
- Surget A, Saxe M, Leman S, Ibarguen-Vargas Y, Chalon S, Griebel G et al. Drug-dependent requirement of hippocampal neurogenesis in a model of depression and of antidepressant reversal. Biol Psychiatry 2008; 64: 293–301. [DOI] [PubMed] [Google Scholar]
- van Praag H, Schinder AF, Christie BR, Toni N, Palmer TD, Gage FH. Functional neurogenesis in the adult hippocampus. Nature 2002; 415: 1030–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David DJ, Samuels BA, Rainer Q, Wang JW, Marsteller D, Mendez I et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 2009; 62: 479–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David DJ, Wang J, Samuels BA, Rainer Q, David I, Gardier AM et al. Implications of the functional integration of adult-born hippocampal neurons in anxiety-depression disorders. Neuroscientist 2010; 16: 578–591. [DOI] [PubMed] [Google Scholar]
- Malberg JE, Eisch AJ, Nestler EJ, Duman RS. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J Neurosci 2000; 20: 9104–9110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holick KA, Lee DC, Hen R, Dulawa SC. Behavioral effects of chronic fluoxetine in BALB/cJ mice do not require adult hippocampal neurogenesis or the serotonin 1A receptor. Neuropsychopharmacol: Off Publ Am Coll Neuropsychopharmacol 2008; 33: 406–417. [DOI] [PubMed] [Google Scholar]
- Huang GJ, Bannerman D, Flint J. Chronic fluoxetine treatment alters behavior, but not adult hippocampal neurogenesis, in BALB/cJ mice. Mol Psychiatry 2008; 13: 119–121. [DOI] [PubMed] [Google Scholar]
- Navailles S, Hof PR, Schmauss C. Antidepressant drug-induced stimulation of mouse hippocampal neurogenesis is age-dependent and altered by early life stress. J Comp Neurol 2008; 509: 372–381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang JW, David DJ, Monckton JE, Battaglia F, Hen R. Chronic fluoxetine stimulates maturation and synaptic plasticity of adult-born hippocampal granule cells. J Neurosci 2008; 28: 1374–1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao C, Teng EM, Summers RG Jr., Ming GL, Gage FH. Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J Neurosci 2006; 26: 3–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overstreet-Wadiche LS, Bromberg DA, Bensen AL, Westbrook GL. Seizures accelerate functional integration of adult-generated granule cells. J Neurosci 2006; 26: 4095–4103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge S, Goh EL, Sailor KA, Kitabatake Y, Ming GL, Song H. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 2006; 439: 589–593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duveau V, Laustela S, Barth L, Gianolini F, Vogt KE, Keist R et al. Spatiotemporal specificity of GABAA receptor-mediated regulation of adult hippocampal neurogenesis. Eur J Neurosci 2011; 34: 362–373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jagasia R, Steib K, Englberger E, Herold S, Faus-Kessler T, Saxe M et al. GABA-cAMP response element-binding protein signaling regulates maturation and survival of newly generated neurons in the adult hippocampus. J Neurosci 2009; 29: 7966–7977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tronel S, Fabre A, Charrier V, Oliet SH, Gage FH, Abrous DN. Spatial learning sculpts the dendritic arbor of adult-born hippocampal neurons. Proc Natl Acad Sci USA 2010; 107: 7963–7968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shors TJ, Miesegaes G, Beylin A, Zhao M, Rydel T, Gould E. Neurogenesis in the adult is involved in the formation of trace memories. Nature 2001; 410: 372–376. [DOI] [PubMed] [Google Scholar]
- Snyder JS, Hong NS, McDonald RJ, Wojtowicz JM. A role for adult neurogenesis in spatial long-term memory. Neuroscience 2005; 130: 843–852. [DOI] [PubMed] [Google Scholar]
- Deng W, Saxe MD, Gallina IS, Gage FH. Adult-born hippocampal dentate granule cells undergoing maturation modulate learning and memory in the brain. J Neurosci 2009; 29: 13532–13542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tashiro A, Zhao C, Suh H, Gage FH. Purification and injection of retroviral vectors. Cold Spring Harbor Protocols 2015; 2015: 925–931. [DOI] [PubMed] [Google Scholar]
- Tashiro A, Zhao C, Suh H, Gage FH. Imaging newborn granule cells in fixed sections. Cold Spring Harbor Protocols 2015; 2015: 932–933. [DOI] [PubMed] [Google Scholar]
- Tashiro A, Makino H, Gage FH. Experience-specific functional modification of the dentate gyrus through adult neurogenesis: a critical period during an immature stage. J Neurosci 2007; 27: 3252–3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meijering E, Jacob M, Sarria JC, Steiner P, Hirling H, Unser M. Design and validation of a tool for neurite tracing and analysis in fluorescence microscopy images. Cytometry Part A: J Int Soc Anal Cytol 2004; 58: 167–176. [DOI] [PubMed] [Google Scholar]
- Rich JT, Neely JG, Paniello RC, Voelker CC, Nussenbaum B, Wang EW. A practical guide to understanding Kaplan-Meier curves. Otolaryngol—Head Neck Surg: Off J Am Acad 2010; 143: 331–336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown JP, Couillard-Despres S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG. Transient expression of doublecortin during adult neurogenesis. J Comp Neurol 2003; 467: 1–10. [DOI] [PubMed] [Google Scholar]
- Samuels BA, Anacker C, Hu A, Levinstein MR, Pickenhagen A, Tsetsenis T et al. 5-HT1A receptors on mature dentate gyrus granule cells are critical for the antidepressant response. Nat Neurosci 2015; 18: 1606–1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder JS, Choe JS, Clifford MA, Jeurling SI, Hurley P, Brown A et al. Adult-born hippocampal neurons are more numerous, faster maturing, and more involved in behavior in rats than in mice. J Neurosci 2009; 29: 14484–14495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi K, Ikeda Y, Sakai A, Yamasaki N, Haneda E, Miyakawa T et al. Reversal of hippocampal neuronal maturation by serotonergic antidepressants. Proc Natl Acad Sci USA 2010; 107: 8434–8439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esposito MS, Piatti VC, Laplagne DA, Morgenstern NA, Ferrari CC, Pitossi FJ et al. Neuronal differentiation in the adult hippocampus recapitulates embryonic development. J Neurosci 2005; 25: 10074–10086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heigele S, Sultan S, Toni N, Bischofberger J. Bidirectional GABAergic control of action potential firing in newborn hippocampal granule cells. Nat Neurosci 2016; 19: 263–270. [DOI] [PubMed] [Google Scholar]
- Ide Y, Fujiyama F, Okamoto-Furuta K, Tamamaki N, Kaneko T, Hisatsune T. Rapid integration of young newborn dentate gyrus granule cells in the adult hippocampal circuitry. Eur J Neurosci 2008; 28: 2381–2392. [DOI] [PubMed] [Google Scholar]
- Faulkner RL, Jang MH, Liu XB, Duan X, Sailor KA, Kim JY et al. Development of hippocampal mossy fiber synaptic outputs by new neurons in the adult brain. Proc Natl Acad Sci USA 2008; 105: 14157–14162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu Y, Arruda-Carvalho M, Wang J, Janoschka SR, Josselyn SA, Frankland PW et al. Optical controlling reveals time-dependent roles for adult-born dentate granule cells. Nat Neurosci 2012; 15: 1700–1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chancey JH, Adlaf EW, Sapp MC, Pugh PC, Wadiche JI, Overstreet-Wadiche LS. GABA depolarization is required for experience-dependent synapse unsilencing in adult-born neurons. J Neurosci: Off J Soc Neurosci 2013; 33: 6614–6622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trouche S, Bontempi B, Roullet P, Rampon C. Recruitment of adult-generated neurons into functional hippocampal networks contributes to updating and strengthening of spatial memory. Proc Natl Acad Sci USA 2009; 106: 5919–5924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aasebo IE, Blankvoort S, Tashiro A. Critical maturational period of new neurons in adult dentate gyrus for their involvement in memory formation. Eur J Neurosci 2011; 33: 1094–1100. [DOI] [PubMed] [Google Scholar]
- Deng W, Gage FH. The effect of immature adult-born dentate granule cells on hyponeophagial behavior is related to their roles in learning and memory. Front Syst Neurosci 2015; 9: 34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dulawa SC, Holick KA, Gundersen B, Hen R. Effects of chronic fluoxetine in animal models of anxiety and depression. Neuropsychopharmacol: Off Publ Am Coll Neuropsychopharmacol 2004; 29: 1321–1330. [DOI] [PubMed] [Google Scholar]
- Kodama M, Fujioka T, Duman RS. Chronic olanzapine or fluoxetine administration increases cell proliferation in hippocampus and prefrontal cortex of adult rat. Biol Psychiatry 2004; 56: 570–580. [DOI] [PubMed] [Google Scholar]
- Bergami M, Rimondini R, Santi S, Blum R, Gotz M, Canossa M. Deletion of TrkB in adult progenitors alters newborn neuron integration into hippocampal circuits and increases anxiety-like behavior. Proc Natl Acad Sci USA 2008; 105: 15570–15575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinnock SB, Blake AM, Platt NJ, Herbert J. The roles of BDNF, pCREB and Wnt3a in the latent period preceding activation of progenitor cell mitosis in the adult dentate gyrus by fluoxetine. PLoS ONE 2010; 5: e13652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pare WP. Hyponeophagia in Wistar Kyoto (WKY) rats. Physiol Behav 1994; 55: 975–978. [DOI] [PubMed] [Google Scholar]
- Treit D, Fundytus M. Thigmotaxis as a test for anxiolytic activity in rats. Pharmacol Biochem Behav 1988; 31: 959–962. [DOI] [PubMed] [Google Scholar]
- Simon P, Dupuis R, Costentin J. Thigmotaxis as an index of anxiety in mice. Influence of dopaminergic transmissions. Behav Brain Res 1994; 61: 59–64. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.