Abstract
Metabolic syndrome (MetS) is a risk factor for erectile dysfunction (ED), but the underlying mechanisms are unclear. The aims of this study were to determine the underlying mechanisms of metabolic syndrome-related ED (MED). Sprague Dawley (SD) rats were fed a high-fat diet for 6 months, and metabolic parameters were then assessed. An apomorphine test was conducted to confirm MED. Only rats with MED were administered an intracavernosal injection of either epidermal growth factor (EGF) or vehicle for 4 weeks. Erectile responses were evaluated by determining the mean arterial blood pressure (MAP) and intracavernosal pressure (ICP). Levels of protein expression were examined by western blotting and immunohistochemistry. Body weight, fasting blood glucose, plasma insulin and plasma total cholesterol were increased in the MetS rats compared with those in control rats (each p < 0.05). The maximum ICP/MAP, total ICP/MAP and concentration of cyclic guanosine mono-phosphate (cGMP) were significantly decreased in MED rats (each p < 0.05). The expression levels of p110α, p-Akt1 (Tyr308)/Akt1 and p-eNOS (Ser1177)/eNOS were reduced in MED rats (each p < 0.05). Activation of the PI3K/Akt/eNOS signaling cascade (intracavernosal injection of EGF) reversed these changes (each p < 0.05). The present study demonstrates that downregulation of the PI3K/Akt/eNOS signaling pathway is involved in MED.
Introduction
Erectile dysfunction (ED), which is the inability to achieve and sustain an erection sufficient for satisfactory vaginal intercourse, is a common clinical condition that affects quality of life1. ED affects 5–20% of men worldwide, and metabolic syndrome (MetS) has been proven to be a risk factor2–4. MetS, a disease arising from the worldwide obesity epidemic, manifests as insulin resistance, hypertension, hyperlipidemia, and diabetes. The definition of MetS varies somewhat in the literature, but the presence of three or more of the aforementioned components is typically defined as MetS5. MetS has a high prevalence around the world and has been reported to be as high as 9.5% in Europe and 35–39% in the US6. Furthermore, the prevalence of ED in MetS patients has been reported to be twofold greater than in control patients7. Phosphodiesterase type 5 inhibitor (PDE5i) therapy, which is currently the first-line treatment for ED, remains markedly inefficient in this population, and therefore, new therapeutic methods are needed8. Hence, the aims of this study were to determine the underlying mechanisms of metabolic syndrome-related ED (MED).
The phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PKB/Akt) signaling pathway is one of the main regulatory networks in the cell and influences almost all cellular activities, including replication, growth, metabolism, movement and differentiation. PI3K comprises three classes of enzymes (classes I–III). Class IA PI3K is a heterodimeric kinase that consists of a p110 catalytic subunit and a p85 regulatory subunit and plays an important role in human cell biology. The class IA p110 isoform is a complex composed of p110α, p110β and p110γ, while p85 isoforms contain p85α and p85β9. PI3K is activated by various extracellular signals, such as epidermal growth factor receptor (EGFR) or insulin-like growth factor1receptor (IGF1R). Akt is a downstream serine/threonine protein kinase that acts as a core signaling input to regulate downstream effectors and preserve cell homeostasis. Akt comprises three highly conserved homologous isoforms: Akt1 (PKBα), Akt2 (PKBβ), and Akt3 (PKBγ)10. Three Akt isoforms all contain a C-terminal hydrophobic domain and a N-terminal regulatory pleckstrin homology (PH) domain, a central kinase domain with serine/threonine specificity11,12. Studies have shown that Akt isoforms are similar in structure but distinct in function in physiological and pathological processes, partly due to different tissue-specific expression of Akt isoforms. For example, although Akt1 expression is ubiquitous, whereas Akt2 is highly expressed in adipocytes and muscle and plays an important role in glucose homeostasis. The distribution of Akt3 is more restricted and is primarily expressed in the testes and brain13–16. Upon activation, Akt phosphorylates many downstream substrates, which regulate protein synthesis and cell growth. Activated Akt can directly activate endothelial nitric oxide synthase (eNOS) via phosphorylation at Ser1177, leading to augmented nitric oxide (NO) synthesis17. Previous work revealed that the Akt-dependent phosphorylation of eNOS mediates penile erection18. Other studies revealed that Akt-dependent phosphorylation and activation of eNOS leads to sustained NO production and maximal erection19,20. Similarly, Wen et al. demonstrated that the A2B adenosine receptor contributes to penile erection via PI3K/Akt signaling-mediated eNOS activation21. However, the role of the PI3K/Akt/eNOS signaling pathway in MED remains to be elucidated.
The aims of the present study were to determine the effect of the PI3K/Akt/eNOS signaling pathway on erectile function in a rat in vivo model of MED and to suggest a potential novel treatment strategy for MED.
Results
Metabolic Parameters
Body weight, fasting blood glucose, plasma total cholesterol and insulin levels in rats fed the high-fat diet for 6 months were significantly higher than those in the age-matched controls. No significant differences in blood pressure, triglycerides, low-density-lipoprotein (LDL) and high-density-lipoprotein (HDL) were found among the groups (Table 1).
Table 1.
Effect of the high-fat diet on general parameters.
| CO | MED | EGF | |
|---|---|---|---|
| Body weight (g) | 577.0 ± 27.0 | 640.0 ± 39.0* | 634.0 ± 48.0* |
| Fasting blood glucose (mmol/L) | 6.4 ± 0.5 | 7.6 ± 0.8* | 7.5 ± 1.1* |
| Blood pressure (mmHg) | 121 ± 18 | 128 ± 15 | 124 ± 17 |
| Insulin level (mIU/L) | 16.0 ± 1.1 | 20.6 ± 1.6* | 20.5 ± 1.7* |
| Total cholesterol (mmol/L) | 1.95 ± 0.08 | 4.32 ± 1.05* | 2.68 ± 0.26# |
| Triglycerides (mmol/L) | 0.64 ± 0.08 | 0.59 ± 0.10 | 0.68 ± 0.07 |
| High density lipoprotein (mmol/L) | 2.81 ± 0.61 | 3.11 ± 1.15 | 3.35 ± 1.47 |
| Low density lipoprotein (mmol/L) | 2.27 ± 0.12 | 2.35 ± 0.16 | 2.21 ± 0.13 |
Data are shown as the means ± SD of 5~7 rats. *p < 0.05 compared with the CO group, #p < 0.05 compared with the MED group. CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; SD, standard deviation.
Upregulation of the PI3K/Akt/eNOS Signaling Pathway Improves Erectile Function in MED Rats
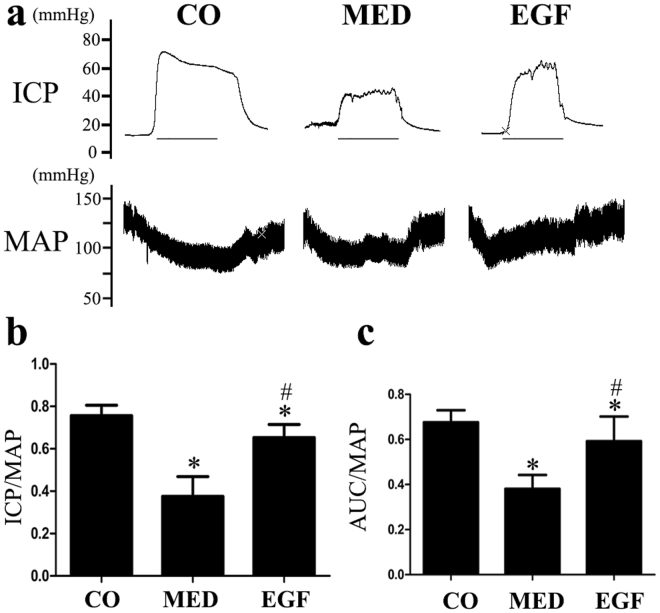
The effect of the PI3K/Akt/eNOS signaling pathway upregulation on the recovery of erectile function is illustrated in Fig. 1. The control group exhibited normal maximum (max) intracavernous pressure (ICP)/mean arterial blood pressure (MAP) and total ICP/MAP, whereas MetS consistently caused ED. The max ICP/MAP and total ICP/MAP ratios were lower in the MED group than in the control group. Partial but significant recovery of erectile function was observed in the epidermal growth factor (EGF)-treated groups compared with the MED group, as reflected by significantly higher max ICP/MAP and total ICP/MAP ratios in response to cavernous nerve electrical stimulation (p < 0.05).
Figure 1.
(a) Evaluation of erectile function via electrical stimulation of the cavernous nerve. Representative carotid artery pressure and intracavernous pressure tracings obtained following cavernous nerve stimulation at 5 V for 1 min, respectively, in the CO, MED and EGF rats. (b,c) Erectile function presented as the maximum ICP/MAP and AUC/MAP in the three groups. Data are expressed as the means ± SD of 5~7 rats. *p < 0.05 vs the CO group, #p < 0.05 vs the MED group. CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; ICP, intracavernosal pressure; MAP, mean arterial pressure; AUC, total intracavernosal pressure; SD, standard deviation.
EGF Treatment Upregulates EGF Expression in MED Rats
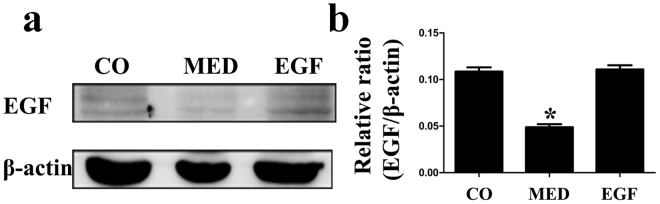
To clarify the mechanism of PI3K/Akt/eNOS activation, we examined EGF expression by western blotting. In the penis, EGF protein expression levels were significantly reduced in MED rats. EGF administration completely reversed the decrease. Statistically significant differences in EGF expression were observed in the three groups (Fig 2a,b).
Figure 2.
(a,b) Western blot analysis of epidermal growth factor (EGF) expression normalized to β-actin levels in the CO, MED and EGF groups. Data in the bar graphs are expressed as the means ± SD of 5~7 rats. *p < 0.05 vs the CO group. CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; SD, standard deviation.
EGF Treatment Upregulates PI3K Expression in MED Rats
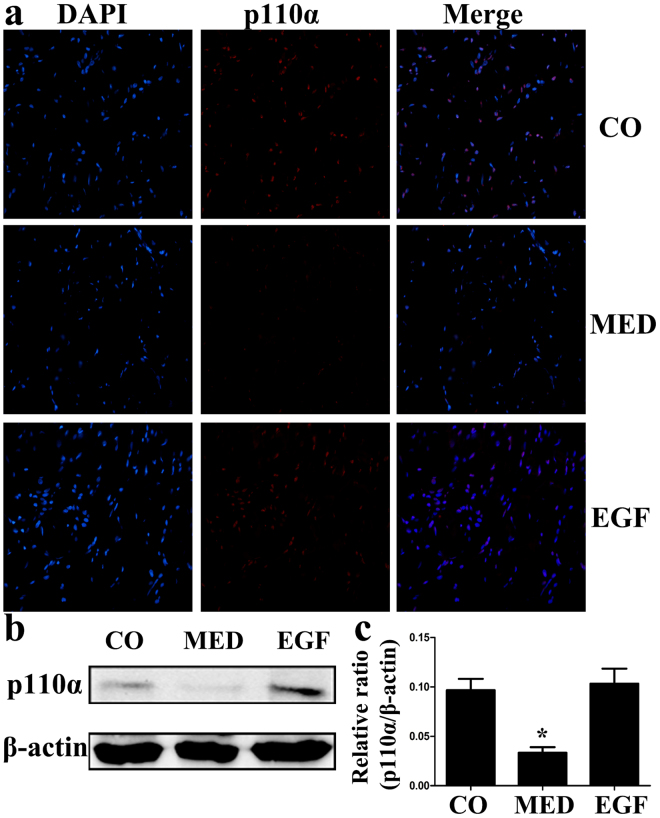
The expression levels of PI3K subunits were determined in all three groups. Baseline p110α levels were reduced in the MED group compared with those in the control group, and EGF treatment increased p110α protein expression (Fig. 3b,c). The expression levels of other PI3K subunits were similar in all three groups. We also performed immunofluorescence staining for p110α. Only faint staining was visible in the MED group, indicating little p110α protein expression in the MED rats. In EGF-treated rats, p110α staining revealed strong expression (as assessed by five high-powered fields per slide; Fig. 3a).
Figure 3.
(a) Immunofluorescence staining of cavernous tissue using an antibody against p110α in the CO, MED and EGF groups. (b,c) Western blot analysis of p110α expression normalized to β-actin levels in the three groups. Data in the bar graphs are expressed as the means ± SD of 5~7 rats. *p < 0.05 vs the CO group. CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; SD, standard deviation.
EGF Treatment Upregulates Akt1 Phosphorylation in MED Rats
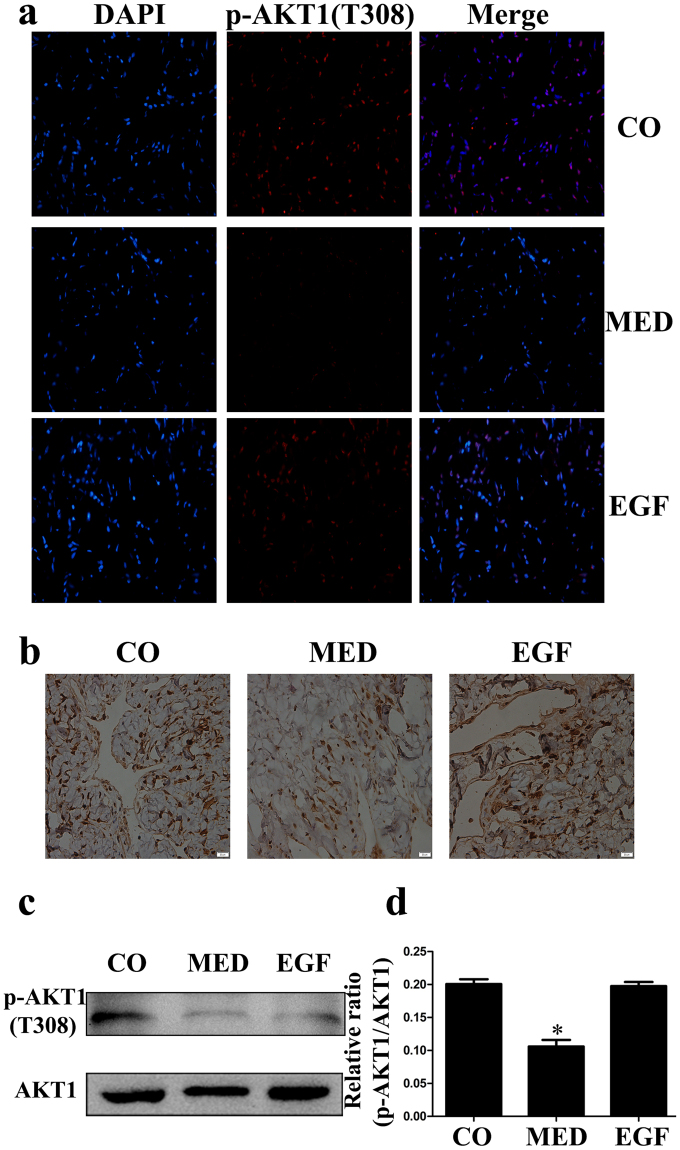
We performed immunohistochemical staining and western blotting of cavernous tissue with antibodies against Akt1 and p-Akt1 (Tyr 308) in all three groups. Representative photomicrographs of each group are shown in Fig. 4. We detected significantly lower Akt1 (Tyr 308) phosphorylation in the MED rats than in the control rats. After intracavernous administration of EGF, Akt1 (Tyr308) phosphorylation showed a clear increase compared with that in the MED group, as determined by immunohistochemical and western blot analyses (Fig. 4a,b,c).
Figure 4.
(a) Immunofluorescence staining of cavernous tissue using an antibody against p-Akt1(Tyr308) in the CO, MED and EGF groups. (b) Immunohistochemistry staining of cavernous tissue was performed with an antibody against Akt1 in three groups (magnification: ×400 scale bar: 20 μm). (c,d) Western blot analysis of p-Akt1 (Tyr308) and Akt1 expression in the three groups. Data in the bar graphs are expressed as the means ± SD of 5~7 rats. *p < 0.05 vs the CO group. p-Akt, phosphor-protein kinase B; CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; SD, standard deviation.
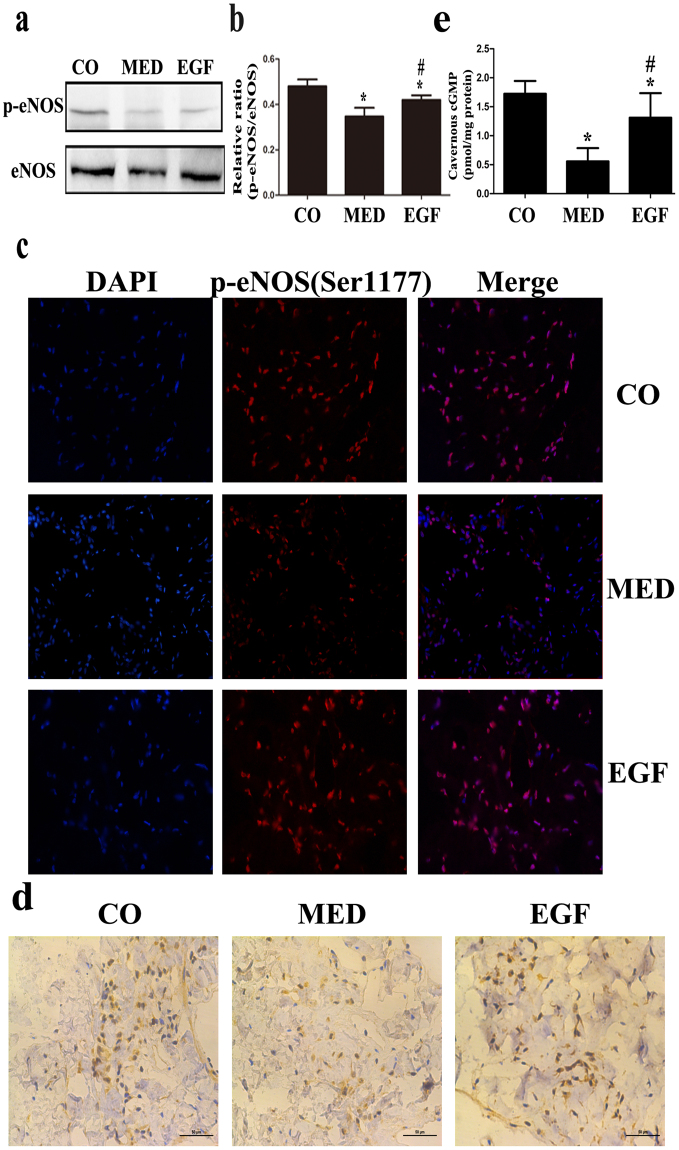
EGF Treatment Upregulates eNOS Phosphorylation in MED Rats
We next evaluated the expression of phospho-eNOS (p-eNOS, Ser1177) and eNOS in the corpus cavernosum. In MED rats, the basal p-eNOS (Ser1177)/eNOS ratio was significantly reduced compared with that in the age-matched controls. At 4 weeks after intracavernous EGF treatment, eNOS phosphorylation increased compared with that in the MED rats, as determined by western blotting and immunohistochemistry (Fig. 5a–d). We also measured cavernous tissue cyclic guanosine mono-phosphate (cGMP) concentrations in all three groups. Cavernous cGMP levels decreased significantly in MED rats compared with those in controls. Cavernous cGMP concentrations were markedly increased in MED rats treated with EGF but were still lower than those in the controls (Fig. 5e).
Figure 5.
(a,b) Western blot analysis of eNOS and p-eNOS (Ser1177) expression in the CO, MED and EGF groups. (c) Immunofluorescence staining of cavernous tissue using an antibody against p-eNOS (Ser1177) in the three groups. (magnification: ×400). (d) Immunohistochemistry staining of cavernous tissue was performed with an antibody against eNOS in three groups. (magnification: × 400). (e) cGMP concentrations in the three groups. Data in the bar graphs are expressed as the means ± SD of 5~7 rats. *p < 0.05 vs the CO group, #p < 0.05 vs the MED group. eNOS, endothelial nitric oxide synthase; CO, normal control rats; MED, metabolic syndrome-related erectile dysfunction rats; EGF, MED rats treated with epithelial growth factor; SD, standard deviation.
Discussion
In this study, we demonstrated that EGF contributes to penile erection via eNOS activation in a PI3K/Akt-dependent manner in MED rats. Significantly reduced erectile function was observed following downregulation of the PI3K/Akt/eNOS signaling pathway in MED rats. Mechanistically, we demonstrated that upregulation of the PI3K/Akt/eNOS signaling pathway is a dual mechanism that ameliorates erectile function in MED rats. Thus, our study reveals a previously unrecognized role for PI3K/Akt/eNOS signaling pathway in penile erection in MED rats and offers a potential novel therapeutic strategy for MED rats.
ED is known to be more common in men with obesity, type 2 diabetes mellitus, and/or MetS22. Due to the popularity of western diets, an increasing number of people suffer from obesity, high blood pressure, diabetes, insulin resistance, high blood lipids and/or MetS, and subsequently, the prevalence of ED caused by the above symptoms is also increasing. Although MetS animals are commercially available, these transgenic mice are not representative of the natural course of MetS. To mimic the natural processes of MED in the general population, an MED model was established by feeding rats a high-fat diet. A recent study using an animal model of type 1 diabetes-associated ED revealed that impairment of the PI3K/Akt/eNOS signaling pathway in the penis is associated with reduced erectile capability23. Another study reported that metabolic disorders lead to abnormal endothelial function that reduces eNOS generation and decreases eNOS phosphorylation at Ser1177 and that activation of the PI3K/Akt/eNOS signaling pathway reverses endothelial dysfunction18. Hence, the PI3K/Akt/eNOS signaling pathway may be the mechanism responsible for MED. In the current study, we established an MED model and found that erectile function in MED rats was lower than that in control rats. We then investigated the molecular mechanisms and found downregulation of the PI3K/Akt/eNOS signaling pathway, decreased expression of PI3K subunit p110α, decreased levels of phosphorylated Akt1 and eNOS, and decreased corpus cavernosum cGMP concentrations in MED rats. These data demonstrate that downregulation of the PI3K/Akt/eNOS signaling pathway is involved in MED. Therefore, the PI3K/Akt/eNOS signaling pathway might be an appropriate therapeutic option for MED.
EGF is a member of the growth factor family with many biological effects, including regulating cell replication, cell movement and cell survival. PI3K transduces signals from various cytokines and growth factors into intracellular messages by generating phospholipids, which activate downstream effectors including Akt and eNOS. The PI3K/Akt pathway regulates the phosphorylation of eNOS at Ser1177, which causes an increase in NO, leading to the accumulation of cGMP24–26. Studies have reported that insulin-like growth factor-1 (IGF-1) improves erectile function in diabetic and aging rats through increased eNOS expression27,28. However, how elevated eNOS is induced by IGF-1 remains unclear. In the current study, we used an intracavernous injection of EGF to investigate erectile responses. According to our ICP data, erectile function markedly recovered after EGF treatment, indicating a therapeutic effect. We found that the recovery of erectile function in MED rats by EGF administration was triggered at the molecular level by activation of the PI3K/Akt/eNOS signaling pathway. eNOS activation caused the sustained production of NO, which promotes cavernosal smooth muscle relaxation and the maintenance of an erection. In our study, we demonstrated that p110α and phosphorylated Akt1 (Tyr308) and eNOS (Ser1177) were reduced in MED rats. Treatment with EGF, which actives the PI3K/Akt/eNOS signaling pathway, increased the expression of p110α and the phosphorylation of Akt1 (Tyr308) and eNOS (Ser1177). This finding strongly suggests that EGF activates the PI3K/Akt pathway, promoting increased phosphorylation and, thus, activation of eNOS. The latter increases NO availability and produces larger quantities of cGMP, which maintain penile erection.
However, our work has some limitations. First, we did not investigate how EGF regulates the PI3K/Akt/eNOS signaling pathway. Second, the recovery of erectile function was relatively short-lived. Further studies are needed to test whether repeated intracavernous injections of EGF can induce more durable recovery of erectile function.
In summary, the PI3K/Akt/eNOS signaling pathway is involved in MED. We offer strong evidence demonstrating that EGF successfully actives the PI3K/Akt/eNOS signaling pathway, leading to an increase in the cGMP concentration and improvement of erectile function in MED rats. Therefore, our study offers novel findings and thereby insights into the molecular mechanisms of MED. Further studies that elucidate the precise mechanisms are warranted.
Materials and Methods
Experimental Outline
All animal studies were approved by the Committees on Animal Experiments at Tongji Hospital (Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, China; IRB ID:TJ-A20150701), and all procedures complied with the Chinese Council on Animal Care Regulations for the care and use of laboratory animals.
A total of 70 male Sprague Dawley rats (3 weeks old) were randomly assigned to a control group (n=8) or the MetS group (n=62). The control group was fed a regular diet, and the MetS group was fed a high-fat diet for 6 months (Table 2). After 6 months, body weight, blood pressure, plasma insulin, fasting blood glucose and plasma lipid were assessed. MetS rats were chosen based on the results. Then, an apomorphine (APO) test was conducted to confirm MED29–32. The rats were habituated in a hanging cage for 10 min and received subcutaneous injection of APO in saline (80 mg/kg) into the loose skin of the neck. The erectile response of the tested rats was observed for 30 min. Rats that did not present an erectile response were considered MED rats. Only rats with MED were administered a signal intracavernosal injection of either EGF (1 µg/kg, Prospec, Ness Ziona, Israel) or vehicle for 4 weeks. Age-matched rats were given saline for 4 weeks. A tourniquet was applied at the base of the penis, and the needle was left in place for 3 min to allow the medication to diffuse throughout the cavernous space during intracavernosal injection.
Table 2.
Diet Composition.
| Control Diet | High-fat Diet | |||
|---|---|---|---|---|
| Ingredients | % | Ingredients | gm | kcal |
| Corn | 40 | Casein, 80 Mesh | 200 | 800 |
| Soybean Meal | 20 | L-Cystine | 3 | 12 |
| Fish Meal | 7 | Corn Starch | 0 | 0 |
| Four | 22 | Maltodextrin 10 | 125 | 500 |
| Yeast | 4 | Sucrose | 68.8 | 275.2 |
| Grease | 1.5 | Cellulose, BW200 | 50 | 0 |
| Calcium Hydrogen Phosphate | 2 | Soybean Oil | 25 | 225 |
| Stone Powder | 1.5 | Lard | 245 | 2205 |
| Mineral Salts | 0.7 | Mineral Mix, S10026 | 10 | 0 |
| Vitamins | 0.3 | DiCalcium Phosphate | 13 | 0 |
| Amino Acids | 0.8 | Calcium Carbonate | 5.5 | 0 |
| Choline | 0.2 | Potassium Citrate, 1 H2O | 16.5 | 0 |
| Nutrient Content | % | Vitamin Mix, V10001 | 10 | 40 |
| Crude Protein | 20.7 | Choline Bitartrate | 2 | 0 |
| Crude Fiber | 2.31 | FD&C Blue Dye #1 | 0.05 | 0 |
| Crude Fat | 4.15 | Nutrient content | gm% | kcal% |
| Calcium | 1.24 | Protein | 26.2 | 20 |
| Phosphorus | 0.83 | Carbohydrate | 26.3 | 20 |
| Fat | 34.9 | 60 | ||
Measurement of Metabolic Parameters
After 6 months, blood was collected through the tail vein to determine plasma insulin and plasma lipid levels. Whole blood was centrifuged at 1580 g for 15 min at 4 °C. Plasma lipid and insulin levels were detected using a kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) according to the manufacturer’s instructions. Blood pressure was measured using a photoelectric tail-cuff system (AD Instruments PowerLab, Bella Vista, NSW, Australia) as described previously33, and fasting blood glucose was measured by obtaining a blood sample from the tail vein using a blood glucose meter (Johnson & Johnson, New Brunswick, NJ, USA).
Measurement of Erectile Function
After 4 weeks of EGF treatment, erectile function was assessed in all rats. The assessments were performed as described previously34. First, the major pelvic ganglion and cavernous nerves were exposed and mounted onto stainless steel bipolar wire electrodes, which were connected to an electrical stimulator. The electrical stimulation parameters were as follows: electrical field stimulation at 5 V, a frequency of 15 Hz, a pulse width of 1.2 ms, and a duration of 1 min. Then, a PE-50 cannula (Becton Dickinson & Co., Sparks, MD, USA) was inserted into the carotid artery to monitor systemic MAP. Finally, a 25-gauge needle was inserted into one side of the corpus cavernosum to monitor ICP, connected to PE-50 tubing, and filled with 250 U/mL of a heparin solution. Both blood pressure and ICP were measured continuously using a data acquisition system (AD Instruments PowerLab/4SP, Bella Vista, NSW, Australia). The max ICP/MAP and total ICP/MAP were recorded for each rat. The animals were sacrificed via injection of 20 mL air, and the corporeal tissue was immediately collected from each rat for subsequent studies.
Western Blotting
The assessments were performed as described by Li34. Lysates containing 40 mg protein were separated via SDS-PAGE and transferred to polyvinylidene fluoride membranes (Millipore Corp, Bedford, MA, USA). The primary antibodies used were anti-EGF (1:1000, Affinity, Zhenjiang, Jiangsu, China), anti-p110α (1:1000, Cell Signal Technology, Beverly, MA, USA), anti-Akt1 (1:500, Boster, Wuhan, Hubei, China), anti-phospho-Akt1 (Tyr308, 1:1000, Abcam, Cambridge, MA, USA), eNOS (1:1000, Abcam, Cambridge, MA, USA), p-eNOS (1:1000, Abcam, Cambridge, MA, USA) and β-actin (1:500, Multisciences, Hangzhou, Zhejiang, China). After hybridization with secondary antibodies, the samples were analyzed using a chemiluminescence detection system (Pierce, Thermo Fisher Scientific, Rockford, IL, USA).
Immunohistochemistry and Immunofluorescence
Freshly dissected tissue was fixed in cold 4% formaldehyde and immersed overnight in buffer containing 30% sucrose. Tissues were then paraffin embedded until use. Sections were cut at 4 µm, adhered to charged slides, dewaxed with xylene, hydrated with an ethanol gradient, and treated with hydrogen peroxide to quench endogenous peroxidase activity (not required for immunofluorescence). After being rinsed, sections were washed three times in PBS for 5 min, followed by antigen repair. After excess fluid was drained, sections were incubated overnight at 4 °C with anti-p110α (1:50, Cell Signal Technology, Beverly, MA, USA), anti-Akt1 (1:50, Boster, Wuhan, Hubei, China), anti-phospho-Akt1 (Tyr308, 1:50, Abcam, Cambridge, MA, USA), eNOS (1:100, Abcam, Cambridge, MA, USA), or p-eNOS (1:100, Abcam, Cambridge, MA, USA). After several washes with PBS, the sections were incubated with Cy3-conjugated rabbit anti-IgG or Cy3-conjugated mouse anti-IgG for 60 min at room temperature. Finally, antigen-antibody reactions were developed using diaminobenzidine (DAB). For immunofluorescence studies, the sections were incubated with Cy3-conjugated rabbit anti-IgG or Cy3-conjugated mouse anti-IgG for 60 min at room temperature and then counterstained with DAPI (Beyotime Institute of Biotechnology, Haimen, Jiangsu, China). Signals were visualized on a microscope, and image analysis was performed using computerized densitometry in Image-Pro Plus (Media Cybernetics Inc., Bethesda, MD, USA).
Measurement of Cavernous Tissue cGMP Levels
Corpus cavernosum cGMP concentrations were detected using an ELISA kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) according to the manufacturer’s instructions. The assays were conducted in duplicate, and the protein concentrations were measured to normalize the data.
Statistical Analysis
The results were analyzed using SPSS 22.0 software (SPSS, Inc., Chicago, IL, USA) and are expressed as the means ± standard deviation (SD). All statistical analyses were performed using one-way analysis of variance (ANOVA) followed by Bonferroni’s multiple-comparison post-test to determine statistical significance (p < 0.05).
Acknowledgements
This work was supported by a grant from the National Natural Sciences Foundation of China (No. 81200435). The funders had no role in the study design, data collection, data analysis, decision to publish, or preparation of the manuscript.
Author Contributions
Rui Li conceived and designed the experiments, performed the experiments, wrote the paper, prepared figures and/or tables, and reviewed drafts of the paper. Kai Cui, Kang Liu, Hao Li and Yan Zhang performed the experiments, prepared figures and/or tables, and reviewed drafts of the paper. Xiaming Liu, Ruibao Chen and Mingchao Li analyzed the data and reviewed drafts of the paper. Tao Wang and Shaogang Wang contributed reagents/materials/analysis tools and reviewed drafts of the paper. Jihong Liu and Ke Rao conceived and designed the experiments and reviewed drafts of the paper.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jihong Liu, Email: jhliu@tjh.tjmu.edu.cn.
Ke Rao, Email: raokeke2009@163.com.
References
- 1.Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013;381:153–165. doi: 10.1016/S0140-6736(12)60520-0. [DOI] [PubMed] [Google Scholar]
- 2.Kubin M, Wanger G, Fugl-Meyer AR. Epidemiology of erectile dysfunction. International Journal of Impotence Research. 2003;15:63–71. doi: 10.1038/sj.ijir.3900949. [DOI] [PubMed] [Google Scholar]
- 3.Jackson G. The metabolic syndrome and erectile dysfunction: multiple vascular risk factors and hypogonadism. European Urology. 2006;50:426–427. doi: 10.1016/j.eururo.2006.03.035. [DOI] [PubMed] [Google Scholar]
- 4.Heidler S, et al. Is the metabolic syndrome an independent risk factor for erectile dysfunction? Journal of Urology. 2007;177:651–654. doi: 10.1016/j.juro.2006.09.043. [DOI] [PubMed] [Google Scholar]
- 5.Expert Panel on Detection, E. and Treatment & Adults, o. H. B. C. i Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) The Journal of the American Medical Association. 2001;285:2486–2497. doi: 10.1001/jama.285.19.2486. [DOI] [PubMed] [Google Scholar]
- 6.Golden SH, Robinson KA, Saldanha I, Anton B, Ladenson PW. Clinical review: Prevalence and incidence of endocrine and metabolic disorders in the United States: a comprehensive review. The Journal of Clinical Endocrinology and Metabolism. 2009;94:1853–1878. doi: 10.1210/jc.2008-2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kaya E, Sikka SC, Gur S. A comprehensive review of metabolic syndrome affecting erectile dysfunction. The Journal of Sexual Medicine. 2015;12:856–875. doi: 10.1111/jsm.12828. [DOI] [PubMed] [Google Scholar]
- 8.Shabsigh R, Mattern A, Group RS. REVITALISE: a large observational study assessing the safety and effectiveness of vardenafil in men with erectile dysfunction and metabolic syndrome. The Journal of Sexual Medicine. 2016;4:e135–144. doi: 10.1016/j.esxm.2016.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mabuchi S, Kuroda H, Takahashi R, Sasano T. The PI3K/AKT/mTOR pathway as a therapeutic target in ovarian cancer. Gynecologic Oncology. 2015;137:173–179. doi: 10.1016/j.ygyno.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 10.Carmona FJ, et al. AKT signaling in ERBB2-amplified breast cancer. Pharmacology & Therapeutics. 2016;158:63–70. doi: 10.1016/j.pharmthera.2015.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brazil DP, Hemminqs BA. Ten years of protein kinase B signalling: a hard Akt to follow. Thends in Biometrical Sciences. 2001;26:657–664. doi: 10.1016/s0968-0004(01)01958-2. [DOI] [PubMed] [Google Scholar]
- 12.Hanada M, Feng J, Hemminqs BA. Structure, regulation and function of PKB/AKT—a major therapeutic target. Biochimica et Biophysica Acta. 2004;1697:3–16. doi: 10.1016/j.bbapap.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 13.Dummler B, et al. Life with a single isoform of Akt: mice lacking Akt2 and Akt3 are viable but display impaired glucose homeostasis and growth deficiencies. Molecular and Cell Biology. 2006;26:8042–8051. doi: 10.1128/MCB.00722-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fayard E, Tintignac LA, Baudry A, Hemminqs BA. Protein kinase B/Akt at a glance. Journal of Cell Science. 2005;118(Pt 24):5675–5678. doi: 10.1242/jcs.02724. [DOI] [PubMed] [Google Scholar]
- 15.Gonzalez E, McGraw TE. The Akt kinases: isoform specificity in metabolism and cancer. Cell Cycle. 2009;8:2502–2508. doi: 10.4161/cc.8.16.9335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang ZZ, et al. Protein kinase B alpha/Akt1 regulates placental development and fetal growth. The Journal of Biological Chemistry. 2003;278:32124–32131. doi: 10.1074/jbc.M302847200. [DOI] [PubMed] [Google Scholar]
- 17.Garcia-Prieto CF, et al. High-fat diet induces endothelial dysfunction through a down-regulation of the endothelial AMPK-PI3K-Akt-eNOS pathway. Molecular Nutrition &Food Research. 2015;59:520–532. doi: 10.1002/mnfr.201400539. [DOI] [PubMed] [Google Scholar]
- 18.El Assar M, et al. FM19G11 reverses endothelial dysfunction in rat and human arteries through stimulation of the PI3K/Akt/eNOS pathway, independently of mTOR/HIF-1α activation. British Journal of Pharmacology. 2015;172:1277–1291. doi: 10.1111/bph.12993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hurt KJ, et al. Akt-dependent phosphorylation of endothelial nitric-oxide synthase mediates penile erection. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:4061–4066. doi: 10.1073/pnas.052712499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yin GN, et al. Inhibition of Ninjurin 1 restores erectile function through dual angiogenic and neurotrophic effects in the diabetic mouse. Proceedings of the National Academy of Sciences of the United States of America. 2014;111:E2731–E2740. doi: 10.1073/pnas.1403471111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wen J, et al. A2B adenosine receptor contributes to penile erection via PI3K/AKT signaling cascade-mediated eNOS activation. FASEB journal: Official Publication of the Federation of American Societies for Experimental Biology. 2011;25:2823–2830. doi: 10.1096/fj.11-181057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Esposito K, Giugliano D. Obesity, the metabolic syndrome, and sexual dysfunction. International Journal of Impotence Research. 2005;17:391–398. doi: 10.1038/sj.ijir.3901333. [DOI] [PubMed] [Google Scholar]
- 23.Musicki B, Kramer MF, Becker RE, Burnett AL. Inactivation of phosphorylated endothelial nitric oxide synthase (Ser-1177) by O-GlcNAc in diabetes-associated erectile dysfunction. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:11870–11875. doi: 10.1073/pnas.0502488102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kobayashi T, Taguchi K, Yasuhiro T, Matsumoto T, Kamata K. Impairment of PI3-K/Akt pathway underlies attenuated endothelial function in aorta of type 2 diabetic mouse model. Hypertension. 2004;44:956–962. doi: 10.1161/01.HYP.0000147559.10261.a7. [DOI] [PubMed] [Google Scholar]
- 25.Li R, et al. Vascular insulin resistance in prehypertensive rats: role of PI3-kinase/Akt/eNOS signaling. European Journal of Pharmacology. 2010;628:140–147. doi: 10.1016/j.ejphar.2009.11.038. [DOI] [PubMed] [Google Scholar]
- 26.Zhang QJ, et al. Ceramide mediates vascular dysfunction in diet-induced obesity by PP2A-mediated dephosphorylation of the eNOS-Akt complex. Diabetes. 2012;61:1848–1859. doi: 10.2337/db11-1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pu XY, et al. Insulin-like growth factor-1 restores erectile function in aged rats: modulation the integrity of smooth muscle and nitric oxide-cyclic guanosine monophosphate signaling activity. The Journal of Sexual Medicine. 2008;5:1345–1354. doi: 10.1111/j.1743-6109.2008.00817.x. [DOI] [PubMed] [Google Scholar]
- 28.Pu XY, Hu LQ, Wang HP, Luo YX, Wang XH. Improvement in erectile dysfunction after insulin-like growth factor-1 gene therapy in diabetic rats. Asian Journal of Andrology. 2007;9:83–91. doi: 10.1111/j.1745-7262.2007.00215.x. [DOI] [PubMed] [Google Scholar]
- 29.Heaton JP, Varrin SJ, Morales A. The characterization of a bio-assay of erectile function in a rat model. Journal of Urology. 1991;145:1099–1102. doi: 10.1016/S0022-5347(17)38543-9. [DOI] [PubMed] [Google Scholar]
- 30.Brien SE, Wilson E, Heaton JP, Adams MA. The conditioned response erection (CRE)—A new approach to modelling erectile responses in the rat. International Journal of Impotence Research. 2000;12:91–96. doi: 10.1038/sj.ijir.3900488. [DOI] [PubMed] [Google Scholar]
- 31.Hale TM, Okabe H, Bushfield TL, Heaton JP, Adams MA. Recovery of erectile function after brief aggressive antihypertensive therapy. Journal of Urology. 2002;168:348–354. doi: 10.1016/S0022-5347(05)64919-1. [DOI] [PubMed] [Google Scholar]
- 32.Zhang Y, et al. Decreased endogenous hydrogen sulfide generation in penile tissues of diabetic rats with erectile dysfunction. The Journal of Sexual Medicine. 2016;13:350–360. doi: 10.1016/j.jsxm.2016.01.002. [DOI] [PubMed] [Google Scholar]
- 33.Xiao B, et al. Overexpression of cytochrome P450 epoxygenases prevents development of hypertension in spontaneously hypertensive rats by enhancing atrial natriuretic peptide. The Journal of Pharmacology and Experimental Therapeutics. 2010;334:784–794. doi: 10.1124/jpet.110.167510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Li R, et al. Testosterone improves erectile function through inhibition of reactive oxygen species generation in castrated rats. Peerj. 2016;4:e2000. doi: 10.7717/peerj.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]