Functional repair of injured tissue in the adult central nervous system (CNS) still remains a big challenge for current biomedical research and its upcoming clinical translation. The axonal regeneration of the adult CNS is generally low, and it is additionally restricted after injury by the presence of inhibitory molecules, generated by the glial scar.
Current preclinical treatment strategies after CNS injury include cell-based therapies, gene therapy and drug and growth factor delivery, and there is already a number of studies which are evaluating the safety and efficiency of these methods in clinical settings. To achieve functional replacement of the damaged or injured tissue, tissue engineering approaches are focused on the application of scaffolding materials, which are optimized to fill the tissue defect and provide a supportive and stimulatory niche that could contribute to replacement of the lost tissue, and re-establish the damaged connections after injury.
The best approach for the development of materials suitable for tissue repair is to mimic exactly the biological and mechanical properties of the cell microenvironment seen in vivo. In native tissue, the cells reside in the extracellular matrix (ECM), which is a complex fibrillary network mostly composed of collagen, elastin, proteoglycans, glycoproteins and various cytokines and growth factors. ECM creates not only a structural framework to maintain tissue integrity, but also provides spatial and biochemical information for the cells, which is essential for the regulation of intracellular signaling and cell behavior.
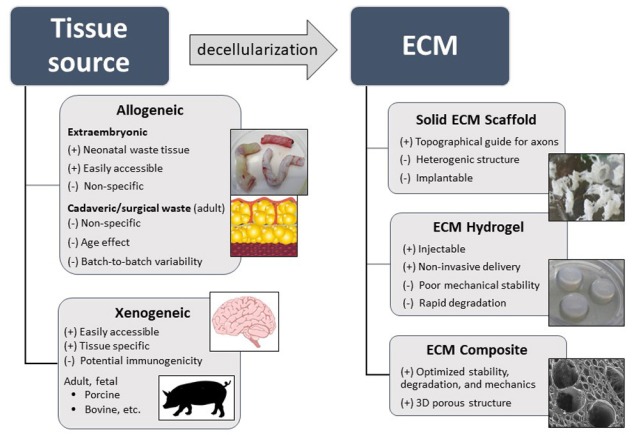
In neural tissue engineering, efforts are being made via various synthetic as well as natural materials, either alone or in combination with cells, growth factors or drugs. Since chemically-synthetized materials fail to mimic the complex morphological structure and chemistry of the native ECM, and require further modifications to enhance their bioactivity and cell adhesiveness, natural materials are generally preferable. Natural-based materials used for CNS tissue repair include purified ECM-molecules, such as collagen, fibronectin, hyaluronic acid, and other naturally occurring polysaccharides (alginate, agarose, chitosan) or their combinations. Along with this, a promising technique of how to produce a natural scaffold that resembles structural and biochemical cues of the native tissue is tissue decellularization, in which the ECM is obtained by cell removal (Figure 1).
Figure 1.
Scheme of potential tissue sources for preparation of extracellular matrix (ECM) based biomaterials for central nervous system (CNS) tissue repair.
Biologic scaffolds composed of native ECM represent materials with many advantages, such as a three-dimensional structure, low immunogenicity, complex biomolecular composition, or retention of growth factors and other chemical cues. The therapeutic effect of ECM scaffolds is based on their ability to facilitate constructive tissue remodelling to the tissue-appropriate structures, by modulation of the immune response and stimulation of endogenous regenerative processes (Badylak et al., 2009). In addition, ECM materials may also serve as a supportive and stimulatory niche for the transplantation of exogenous cells.
Generally, various techniques of decellularization are being used for cell removal, such as physical and mechanical methods, the use of detergents, organic solvents or enzymatic digestion. The resulting ECM scaffolds can then be prepared in the form of a solid fibrillary matrix, sheet, powder or solubilized into the hydrogel (Badylak et al., 2009).
ECM scaffolds have been used for the reconstruction or replacement of many tissues, which include peripheral nerve, myocardium, muscles, blood vessels, valves, oesophagus, bones, skin or whole organs such as the heart, lungs, liver, kidneys, etc. The numbers of such allogenic as well as xenogeneic materials are currently commercially available and have already been utilized for the treatment of many patients with tolerable tissue responses and positive clinical outcomes (Badylak et al., 2009). However, despite extensive research into various tissue engineering applications, there are currently few studies evaluating the therapeutic potential of ECM scaffolds for CNS repair (Meng et al., 2014; Wang et al., 2015).
An attempt has been made using an acellular muscle scaffold with naturally multiple linear channels as a topographical guide for the sprouting axons to treat spinal cord injury (SCI). After implantation into a spinal cord hemisection, the acellular muscle scaffold integrated with the host tissue and promoted linear axonal sprouting throughout the lesion, vascularization and neuronal surviving (Zhang et al., 2012). In another report, the acellular sciatic nerve scaffold stimulated axonal regeneration after spinal cord transection, which was further potentiated when the scaffold was loaded with brain-derived neurotrophic factor. However, no significant improvement was found in locomotor recovery in this study (Li et al., 2012). Significant locomotor recovery improvement was found after implantation of acellular spinal cord scaffold alone or seeded with human umbilical cord blood-derived mesenchymal stem cells into the acute hemisected SCI lesion (Liu et al., 2013).
Solid acellular scaffolds, such as those prepared from muscle tissue and spinal cord, or peripheral nerves, may offer an advantage of structures with naturally multiple linear channels, which can provide a topographical guide for sprouting axons. On the other hand, the heterogeneous shape and conformation of such fibrillary scaffolds limit their non-invasive application to the CNS defect. For these reasons, injectable in situ gelling ECM hydrogels are more appropriate, as these materials can easily conform to the lesion irregularity with minimal tissue damage during delivery.
Injectable ECM hydrogels are commonly prepared by pepsin digestion of ECM (pH < 2) which cleaves amine bonds preferentially at the N-terminal side of aromatic amino acids. When the pH of this viscous pre-gel is adjusted to physiological value, a physically cross-linked gel based on collagen fiber self-assembly is formed at 37°C. Of note, the natural organization of the ECM is commonly lost by decellularization and reduction to a soluble powder. After self-assembly, ECM hydrogels reveal a nanofiber network topology with randomly oriented fibers of ~50–100 nm average diameters (Kočí et al., 2017). In general, the ECM materials are composed predominantly of collagen, glycosaminoglycans (GAGs), laminin, fibronectin, and other proteins and their degradation products, with unique biomolecular components depending on the source tissue and the decellularization process (Marçal et al., 2012).
At present, injectable ECM hydrogels have been successfully prepared from a range of tissues and used in vitro as well as in vivo for the repair of soft tissues, such as skeletal muscle, fat, heart, liver, lungs, as well as the spinal cord or brain (Saldin et al., 2017). Both homologous and heterologous ECM hydrogels have been shown to provide an excellent supportive environment for the in vitro cell growth and migration, as well as good tissue integration with no adverse immune response when grafted in vivo.
To prepare ECM hydrogels for CNS repair, both tissues of neural origin (porcine brain and spinal cord) and non-neuronal tissues (porcine urinary bladder, human umbilical cord) were prepared and evaluated in the in vitro and in vivo studies (Medberry et al., 2013; Kočí et al., 2017).
The general concept of tissue-specific effects of ECM on the cellular functions and cell fate commitment suggests that CNS-derived ECMs should have enhanced site-specific potential to stimulate growth and differentiation of neural tissue-derived cell populations. But unlike other tissues, the adult CNS tissue is enriched with myelin-derived molecules and negatively charged proteoglycans, such as chondroitin sulphate proteoglycans, which play a role in retaining growth factors but also have an adverse effect on the axonal outgrowth.
Indeed, a tissue specific response was approved in vitro, when much stronger neuronal differentiation and neurite extension was found for ECM hydrogels derived from porcine brain and spinal cord, than was observed for non-CNS ECM derived from the urinary bladder (Crapo et al., 2014). In contrast, in our recent report, we did not observe any significant differences in chemotactic or neurotrophic properties between the CNS- and non-CNS-derived porcine ECMs and the ECM derived from human umbilical cord, while significant differences were found between these matrices in nano-scale topography, GAGs content, mechanical properties and the speed of gelation (Kočí et al., 2017).
Notably, apart from the in vitro studies, an advantage of tissue specificity for ECM scaffolds in neural tissue repair has not yet been demonstrated in vivo. In our recent study, we compared ECM hydrogels prepared by decellularization of porcine spinal cord or porcine urinary bladder injected into an acute spinal cord hemisection. Both ECM hydrogels bridged the lesion cavity, modulated the innate immune response and provided the benefit of a stimulatory substrate for in vivo neural tissue regeneration. But no significant differences were found between CNS-derived spinal cord-ECM and non-CNS urinary bladder-ECM hydrogels with regards to tissue repair within the 8 weeks after the hydrogel injection into the SCI lesion. Moreover, the fast ECM degradation appeared to be a limiting factor which disabled the complex neural tissue remodeling (Tukmachev et al., 2016).
It is evident that tissue processing and methods of ECM decellularization have a significant influence on the properties, growth factor retention and consequently on the bioactivity and tissue remodeling ability of the resultant ECM. To achieve effective cell removal from myelinated CNS tissue, the decellularization methods are generally more complex compared with non-CNS tissues. As a result, the harsh tissue processing may effectively remove axonal growth-inhibitory molecules, but on the other hand, also eliminate neurosupportive proteins and growth factors, which may lead to a loss of tissue-specific bioactivity. Optimizing the decellularization protocol to effectively remove the cellular component, while retaining the growth factors and biochemical composition balanced towards axonal growth-promoting effects, is therefore crucial to achieve ECM scaffold appropriate for CNS repair.
It was shown previously that the degradation of ECM scaffolds is essential for the constructive tissue remodeling process; while degrading biomaterial is gradually replaced by endogenous cells, which can deposit new equivalent and functional ECM as opposed to the scar tissue (Badylak et al., 2009). As the speed of the ECM scaffold degradation has a significant impact on the character of the tissue replacement, it is therefore desirable that the degradation match with the new tissue formation.
However, after placement into the CNS lesion, the ECM hydrogels are immediately populated with endogenous cells, mostly microglia and macrophage which onset the degradative processes. Since ECM hydrogels are mostly formed by physically crosslinked collagen, their in vivo degradation is generally fast and might be further sped up by the hostility of the lesion due to the acute inflammatory response. In effect, rapid hydrogel degradation might burden the neuroregenerative potential of the ECM hydrogels and therefore significantly influence the character of the tissue replacement. Covalent crosslinking of the ECM hydrogels can be used to slow down the ECM degradation and provide more time to complete the tissue remodelling within the lesion. On the other hand, crosslinking of the ECM may reduce the amount of cellular infiltration into the implant or even cause a foreign body reaction.
Apart from degradation rate, an important concern that needs to be addressed in the development of ECMs to CNS repair are the age and type of the tissue source. In contrast to adult tissue, the immature ECM from fetal or neonatal tissue promotes more effective tissue remodelling. Besides xenogeneic ECMs, human derived allografts are more desirable as they avoid concerns related to potential immune responses as well as xenogeneic disease transfer. Among them, acellular ECM derived from umbilical cord represent an easily accessible neonatal tissue source without age-related impediments such as ECM-crosslinking due to non-enzymatic protein glycation, adipose tissue deposition, fibrosis, oxidative stress and other negative changes (Kočí et al., 2017).
A further essential step in the development of ECM materials into clinical setting is characterization of their composition using advanced methods, such as mass spectroscopy or other methods that would comprehensively specify their molecular profile. Appropriate acquisition of qualitative as well as quantitative criteria is necessary for the optimization of the effectivity of decellularization methods, to achieve stable biological activity and minimal heterogeneity between the batches.
In conclusion, both solid and solubilized ECM biologic scaffolds from CNS and non-CNS tissue sources are undoubtedly advantageous for CNS repair, in terms of their biocompatibility and degradability, as well as their biologic activity, which is able to modulate the immune response and stimulate the endogenous repair process and axonal sprouting. In SCI repair, a combination of ECM hydrogels with more mechanically stable synthetic or natural scaffolds which can provide topographic guiding cues for oriented axonal regrowth might be another possibility of how to take advantage of the bioactive properties of ECM materials.
However, while promising results on ECM scaffolds in CNS repair have been reported, the safety of standardized ECM hydrogels must be thoroughly verified in vivo in various CNS injury preclinical models before ECM materials can be considered for clinical applications.
This work was supported by MEYS of the Czech Republic, No. LO1309.
Footnotes
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer review report:
Reviewer: Sérgio Moura, Universidade Federal do Rio Grande do Norte, Brazil.
Comments to author: The article deals with a topic that is not yet fully understood and clarified in the area of neural regeneration studies. The strengths of the article refer to the contents presented clearly and referenced by the appropriate literature.
References
- Badylak SF, Freytes DO, Gilbert TW. Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 2009;5:1–13. doi: 10.1016/j.actbio.2008.09.013. [DOI] [PubMed] [Google Scholar]
- Crapo PM, Tottey S, Slivka PF, Badylak SF. Effects of biologic scaffolds on human stem cells and implications for CNS tissue engineering. Tissue Eng Part A. 2014;20:313–323. doi: 10.1089/ten.tea.2013.0186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kočí Z, Výborný K, Dubišová J, Vacková I, Jäger A, Lunov O, Jiráková K, Kubinová Š. Extracellular matrix hydrogel derived from human umbilical cord as a scaffold for neural tissue repair and its comparison with extracellular matrix from porcine tissues. Tissue Eng Part C Methods. 2017;23:333–345. doi: 10.1089/ten.TEC.2017.0089. [DOI] [PubMed] [Google Scholar]
- Li C, Zhang X, Cao R, Yu B, Liang H, Zhou M, Li D, Wang Y, Liu E. Allografts of the acellular sciatic nerve and brain-derived neurotrophic factor repair spinal cord injury in adult rats. PLoS One. 2012;7:e42813. doi: 10.1371/journal.pone.0042813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Chen J, Liu B, Yang C, Xie D, Zheng X, Xu S, Chen T, Wang L, Zhang Z, Bai X, Jin D. Acellular spinal cord scaffold seeded with mesenchymal stem cells promotes long-distance axon regeneration and functional recovery in spinal cord injured rats. J Neurol Sci. 2013;325:127–136. doi: 10.1016/j.jns.2012.11.022. [DOI] [PubMed] [Google Scholar]
- Marçal H, Ahmed T, Badylak SF, Tottey S, Foster LJ. A comprehensive protein expression profile of extracellular matrix biomaterial derived from porcine urinary bladder. Regen Med. 2012;7:159–166. doi: 10.2217/rme.12.6. [DOI] [PubMed] [Google Scholar]
- Medberry CJ, Crapo PM, Siu BF, Carruthers CA, Wolf MT, Nagarkar SP, Agrawal V, Jones KE, Kelly J, Johnson SA, Velankar SS, Watkins SC, Modo M, Badylak SF. Hydrogels derived from central nervous system extracellular matrix. Biomaterials. 2013;34:1033–1040. doi: 10.1016/j.biomaterials.2012.10.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng F, Modo M, Badylak SF. Biologic scaffold for CNS repair. Regen Med. 2014;9:367–383. doi: 10.2217/rme.14.9. [DOI] [PubMed] [Google Scholar]
- Saldin LT, Cramer MC, Velankar SS, White LJ, Badylak SF. Extracellular matrix hydrogels from decellularized tissues: Structure and function. Acta Biomater. 2017;49:1–15. doi: 10.1016/j.actbio.2016.11.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tukmachev D, Forostyak S, Koci Z, Zaviskova K, Vackova I, Vyborny K, Sandvig I, Sandvig A, Medberry CJ, Badylak SF, Sykova E, Kubinova S. Injectable extracellular matrix hydrogels as scaffolds for spinal cord injury repair. Tissue Eng Part A. 2016;22:306–317. doi: 10.1089/ten.tea.2015.0422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Lin XF, Wang LR, Lin YQ, Wang JT, Liu WY, Zhu GQ, Braddock M, Zhong M, Zheng MH. Decellularization technology in CNS tissue repair. Expert Rev Neurother. 2015;15:493–500. doi: 10.1586/14737175.2015.1030735. [DOI] [PubMed] [Google Scholar]
- Zhang XY, Xue H, Liu JM, Chen D. Chemically extracted acellular muscle: a new potential scaffold for spinal cord injury repair. J Biomed Mater Res A. 2012;100:578–587. doi: 10.1002/jbm.a.33237. [DOI] [PubMed] [Google Scholar]