Abstract
Protein hydrolysates produced from different food sources exhibit therapeutic potential and can be used in the management of chronic diseases. This study was targeted to optimise the conditions for the hydrolysis of stone fish protein to produce antioxidant hydrolysates using central composite design (CCD) by response surface methodology (RSM). The stone fish protein was hydrolysed under the optimum predicted conditions defined by pH (6.5), temperature (54°C), E/S ratio (1.5%), and hydrolysis time (360 min). The hydrolysates were then evaluated for 2,2-diphenyl-1-picrylhydrazyl radical (DPPH•) scavenging activity and ferrous ion- (Fe2+-) chelating activity. Results validation showed no significant difference between the experimental values of DPPH• scavenging activity (48.94%) and Fe2+-chelating activity (25.12%) obtained at 54.62% degree of hydrolysis (DH) compared to their corresponding predicted values of 49.79% and 24.08% at 53.08% DH, respectively. The hydrolysates demonstrated non-Newtonian behavior (n < 1) with stronger shear-thinning effect and higher viscosities at increasing concentration. Thus, RSM can be considered as a promising strategy to optimise the production of stone fish protein hydrolysates containing antioxidant peptides. It is hoped that this finding will enhance the potential of stone fish protein hydrolysates (SHs) as therapeutic bioactive ingredient in functional foods development.
1. Introduction
The global prevalence of chronic diseases related to free radical generation has necessitated the search for alternative approaches to the use of synthetic agents that are often costly and associated with one or more life-threatening side effects. Alternatively, protein hydrolysates containing potent antioxidant peptides have been produced from locally available and easily accessible food sources. In addition to the management of chronic diseases, these naturally occurring hydrolysates are safe and can be incorporated as additives to halt lipid peroxidation so as to improve the quality and consumer acceptability of many food products. According to Benjakul et al. [1]; McCarthy et al. [2], and Bhaskar et al. [3], protein hydrolysates possess high nutritional and therapeutic potentials with diverse dietary applications. In this study, the enzymatic production of antioxidant hydrolysates from stone fish protein has been successfully optimised making it a chief source of bioactive peptides for various applications.
Stone fish (Actinopyga lecanora) as one of the noble sources of antioxidant hydrolysates is a marine invertebrate from the phylum echinoderm and class Holothuroidea. It is identified among the edible species of sea cucumber commonly found in Malaysia and other south Asian countries mostly collected as bycatch of fishery industry [4, 5].
Bordbar et al. [6] and Lakshmi and Ghosal [7] have recently reported the presence of antioxidants and antiamoebic compounds in stone fish extracts. Stone fish hydrolysates with antihypertensive, antibacterial [5, 8], and antioxidant/free radical-scavenging activities [6] have been enzymatically generated under different reaction conditions. The antioxidant activity of stone fish hydrolysates was found to be dependent on the enzyme specificity, DH, and peptide sequences generated [8]. Thus, the selection of appropriate condition for a particular enzyme is crucial for obtaining maximum antioxidant activity of the hydrolysates.
The hydrolysis conditions of many food protein derived antioxidant hydrolysates have been optimised using RSM. RSM refers to a collection of statistical techniques used for model building to determine a combination of factor levels that produce an optimum response under the influence of one or more independent factor/s such as temperature, pH, time, and E/S ratio [9, 10]. It uses polynomial equation to explore the type of relationship between the factors and their main and combined effects on the desired response [11–13].
Examples include bovine plasma protein hydrolysates [14], jellyfish (Rhopilema esculentum) umbrella collagen hydrolysates [15], sheep visceral protein hydrolysates [16], Whey protein hydrolysates [11, 17], cuttlefish (Sepia officinalis) protein hydrolysates [18], and hydrolysates of pumpkin seed protein isolates [19].
However, the optimum conditions for the hydrolysis of stone fish protein in the production of antioxidant hydrolysates have not been established. Therefore, the present study was aimed at hydrolysing stone fish protein with bromelain according to CCD by RSM and study the effect of the process conditions including pH, temperature, E/S ratio and time on DH, DPPH radical-scavenging activity, and Fe2+-chelating activity of the resulting hydrolysates. The optimum levels of these conditions for the generation of hydrolysates with maximum antioxidant activities were also determined. Furthermore, the effect of shear rate and different concentrations of SHs on its viscosity have been studied.
2. Materials and Methods
Fresh sample of stone fish was obtained from Kedah and Langkawi Breeding Centers (Malaysia), bromelain from pineapple stem tissue, 2.4 to 3 U/mg, was obtained from Acros Organics (Geel, Belgium), 2,2-diphenyl-1-picrylhydrazyl (DPPH) was purchased from Sigma-Aldrich (St. Louis, MO, USA), iron (II) chloride tetrahydrate was obtained from Merck KGaA (Darmstadt, Germany), and all other chemicals used were of analytical grade and obtained from Acros Organics (Geel, Belgium), Fisher Scientific (Loughborough, Leics, UK), and J.T Baker (Thailand).
2.1. Preparation of Antioxidant Stone Fish Protein Hydrolysates (SHs) with Bromelain
The freeze-dried stonefish tissue was hydrolysed using bromelain as previously described by Auwal et al. [20]. Briefly, ten grams of the stone fish powder was transferred into a 12–14 kDa molecular mass cut-off dialysis tube and dialysed for 4 hours using deionised water as the suspending medium at room temperature and for 20 hours by replacing the deionised water with either of the reaction buffers (0.05 M acetate buffer pH 4, 0.05 M acetate buffer pH 5.5, or 0.05 M phosphate buffer pH 7) at 4°C (Table 1). After dialysis, the samples were mixed with 50 mL of the same reaction buffer and then preheated to the required temperature before enzyme addition (Table 1). The hydrolysis reaction was carried out at 150 rpm in water bath shaker. The enzyme was inactivated to terminate the reaction by heating the mixture in boiling water at 100°C for 10 minutes. After centrifugation at 4°C and 10,000 ×g for 20 min, the hydrolysates consisting of the antioxidant peptides were collected as the supernatant, then freeze dried, and stored at −40°C prior to analysis.
Table 1.
Experimental and predicted values of response variables for central composite design.
| Run order | ∗Independent variables | ∗∗Dependent variables | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| X 1 | X 2 | X 3 | X 4 |
Y
1 (DH, %) |
Y
2 (DPPH• scavenging activity, %) |
Y
3 (Fe2+-chelating activity, %) |
||||
| Uncoded levels | Experimental | Predicted | Experimental | Predicted | Experimental | Predicted | ||||
| (1) | 4.0 | 70 | 2.00 | 30 | 34.49 | 36.34 | 13.76 | 13.26 | 13.86 | 16.04 |
| (2) | 5.5 | 55 | 1.25 | 195 | 56.66 | 54.64 | 47.26 | 45.99 | 16.96 | 18.57 |
| (3) | 5.5 | 55 | 0.50 | 195 | 42.10 | 43.68 | 47.62 | 49.57 | 7.48 | 7.44 |
| (4) | 4.0 | 70 | 0.50 | 360 | 29.96 | 31.17 | 22.98 | 24.67 | 10.05 | 8.91 |
| (5) | 5.5 | 55 | 1.25 | 360 | 57.82 | 55.52 | 54.98 | 56.22 | 19.37 | 18.40 |
| (6) | 7.0 | 55 | 1.25 | 195 | 43.00 | 44.19 | 30.56 | 32.69 | 29.77 | 30.17 |
| (7) | 7.0 | 40 | 0.50 | 360 | 32.25 | 32.41 | 33.18 | 34.97 | 14.80 | 13.13 |
| (8) | 7.0 | 40 | 0.50 | 30 | 23.96 | 24.35 | 32.92 | 30.72 | 15.54 | 13.48 |
| (9) | 4.0 | 70 | 0.50 | 30 | 28.25 | 28.03 | 21.55 | 20.41 | 11.26 | 9.25 |
| (10) | 4.0 | 40 | 2.00 | 360 | 51.99 | 53.34 | 23.52 | 22.74 | 17.75 | 18.18 |
| (11) | 5.5 | 55 | 1.25 | 195 | 54.81 | 54.64 | 44.98 | 45.99 | 19.02 | 18.57 |
| (12) | 7.0 | 70 | 0.50 | 30 | 29.25 | 28.43 | 26.82 | 25.49 | 8.76 | 10.99 |
| (13) | 4.0 | 40 | 0.50 | 360 | 31.89 | 32.00 | 32.55 | 29.90 | 10.98 | 11.39 |
| (14) | 5.5 | 40 | 1.25 | 195 | 51.86 | 53.83 | 33.65 | 34.47 | 14.32 | 12.84 |
| (15) | 5.5 | 70 | 1.25 | 195 | 57.56 | 55.45 | 27.82 | 29.24 | 8.64 | 10.35 |
| (16) | 5.5 | 55 | 1.25 | 195 | 53.56 | 54.64 | 47.26 | 45.99 | 18.84 | 18.57 |
| (17) | 5.5 | 55 | 2.00 | 195 | 58.64 | 56.20 | 39.95 | 42.41 | 13.95 | 14.23 |
| (18) | 5.5 | 55 | 1.25 | 195 | 53.95 | 54.64 | 46.65 | 45.99 | 18.14 | 18.57 |
| (19) | 5.5 | 55 | 1.25 | 195 | 54.05 | 54.64 | 46.86 | 45.99 | 16.40 | 18.57 |
| (20) | 7.0 | 40 | 2.00 | 360 | 50.62 | 49.13 | 29.05 | 27.82 | 18.43 | 19.92 |
| (21) | 5.5 | 55 | 1.25 | 30 | 41.98 | 43.41 | 50.96 | 51.96 | 23.20 | 18.75 |
| (22) | 4.0 | 40 | 0.50 | 30 | 25.03 | 23.95 | 24.50 | 25.64 | 9.91 | 11.74 |
| (23) | 7.0 | 70 | 0.50 | 360 | 32.90 | 31.57 | 27.91 | 29.75 | 8.21 | 10.65 |
| (24) | 4.0 | 70 | 2.00 | 360 | 51.25 | 52.50 | 17.20 | 17.52 | 18.01 | 15.69 |
| (25) | 7.0 | 40 | 2.00 | 30 | 26.69 | 28.06 | 22.26 | 23.56 | 20.00 | 20.26 |
| (26) | 7.0 | 70 | 2.00 | 30 | 32.65 | 32.14 | 18.42 | 18.34 | 19.37 | 17.78 |
| (27) | 4.0 | 40 | 2.00 | 30 | 34.68 | 32.26 | 16.68 | 18.48 | 17.75 | 18.52 |
| (28) | 4.0 | 55 | 1.25 | 195 | 48.14 | 46.09 | 27.49 | 27.61 | 28.60 | 28.43 |
| (29) | 5.5 | 55 | 1.25 | 195 | 53.82 | 54.64 | 49.18 | 45.99 | 16.16 | 18.57 |
| (30) | 5.5 | 55 | 1.25 | 195 | 53.79 | 54.64 | 48.65 | 45.99 | 19.56 | 18.57 |
| (31) | 7.0 | 70 | 2.00 | 360 | 47.25 | 48.30 | 24.81 | 22.59 | 18.96 | 17.44 |
∗Independent variables for hydrolysis of stone fish protein: X1; pH, X2; temperature (°C), X3; enzyme/substrate ratio and X4; time (min). ∗∗Dependent variables: Y1; degree of hydrolysis (DH, %), Y2; 2,2-diphenyl-1-picrylhydrazyl radical (DPPH•) scavenging activity (%) and Y3; Fe2+-chelating activity (%).
2.2. Measurement of Degree of Hydrolysis (DH)
The DH refers to the percentage of free amino terminal groups cleaved from proteins during hydrolysis and was determined using OPA (o-phthaldialdehyde) according to the method of Nielsen et al. [21] and Mirzaei et al. [22] with minor modifications. The content of the alpha-amino groups of the samples was determined as concentration of L-serine from a standard curve. The DH was then calculated as the ratio of alpha-amino nitrogen to total nitrogen content using the following equation:
| (1) |
where Li is the amount of released free amino groups resulting from hydrolysis at a time “i,” L0 is the amount of free amino groups in the original sample before the hydrolysis, and Ltotal is the total amount of free amino groups in the original sample following complete hydrolysis with 6 M HCl at 110°C for 24 h.
2.3. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Assay
The 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH•) scavenging activity was measured according to the method described by Hwang et al. [23] with minor modification. 100 μL aliquot of sample (1% w/v) was mixed with 100 μL of 0.25 mM DPPH that was dissolved in 100% methanol. After incubation in the dark at 25°C for 30 min, the absorbance of the mixture was determined at 517 nm in a 96-well microplate reader (Labomed, model UVD-2950, Culver City, CA, USA). The following equation was used to calculate the percentage DPPH• scavenging activity of the SHs:
| (2) |
where As is the absorbance of the tested stone fish hydrolysates (SHs), Ab is the absorbance of the blank, and Ac is the absorbance of the DPPH solution. All experiments were conducted in triplicate.
2.4. Ferrous Ion- (Fe2+-) Chelating Activity Assay
The Fe2+-chelating activity was determined as previously described by Carter [24] and Zarei et al. [25] with some modifications. A 10 μL aliquot of 1% w/v SHs was mixed with 250 μL of sodium acetate buffer (0.1 M, pH 4.9) and 30 μL of 0.01% w/v, FeCl2 solution. Afterwards, 12.5 μL of 0.04 M ferrozine solution was added following incubation for 30 min at 25°C and the absorbance was measured at 562 nm in a microplate reader. All determinations were carried in triplicate and the Fe2+-chelating effect was evaluated as follows:
| (3) |
where As is the absorbance of the tested stone fish hydrolysates (SHs), Ab is the absorbance of the blank, and Ac is the absorbance of the control. All experiments were conducted in triplicate.
2.5. Effect of Shear Rate and Concentration on the Viscosity of SHs
Stone fish protein hydrolysates solutions were produced by dispersion in deionised water at varying concentrations (1, 2, 5, and 10% w/v respectively) under constant stirring for 60 min at 250 rpm and 40°C. A dynamic controlled stress (RS 600) rheometer model was used to study the effect of shear rate and concentration on the viscosity of the hydrolysates. The samples were equilibrated for 120 seconds between cone and plate configured with a diameter: 35 mm, cone angle: 2° and gap size: 0.5 mm to allow for the relaxation of residual stresses prior to testing. The samples were continuously sheared at a rate of 0 to 100 s−1 in 150 s. All determinations were made at 25°C and the following Ostwald-de Waele power law model equation was used to analyse the flow behavior of the hydrolysates:
| (4) |
where τ is the shear stress (Pa), is the shear rate (1/s), K is the consistency coefficient (Pa sn), and n is the flow behavior index.
2.6. Experimental Design for Model Building and Statistical Analysis
Minitab version 16.0 was used for the model building and statistical analysis according to previous description by Auwal et al. [20]. The individual and interaction effects of the process variables including pH, X1; temperature, X2; E/S ratio, X3; and time, X4 on the outcome responses; DH (Y1), DPPH• scavenging activity (Y2), and Fe2+-chelating activity (Y3) were studied by RSM. Each response represents an average of experimental triplicates. A central composite design (CCD) was adapted with 31 experimental runs including 16 full factorial designs, 7 central points, and 8 axial points. The following second-order equation or its reduced form was used to fit the model:
| (5) |
where Y is the outcome or dependent variable; b0 stands for the intercept while bi, bii, and bij represent the coefficients of the linear, quadratic, and interaction terms whereas Xi and Xj are the independent or process variables, respectively. The statistical significance of the regression coefficients was evaluated using ANOVA. The fitted values predicted by the response regression equation were compared with the experimental values for validation of the model. Three-dimensional response surface plots were drawn using the Minitab version 16.0 to show the relationship between levels of the process variables and the outcome or response.
3. Results and Discussion
The observed effects due to the hydrolysis variables including pH (X1), temperature (X2), E/S ratio (X3), and time (X4) on the response values for DH (Y1), DPPH• scavenging activity (Y2), and Fe2+-chelating activity (Y3) are shown in Table 1. The results for the model's coefficients of variation are given in Table 2. A probability test of p < 0.05 was used to estimate the statistical significance of variation in the observed responses using ANOVA. Other statistical parameters including coefficient of determination R2 (R-sqd), adjusted coefficient of determination R2-adjusted (R2-adj), F-test probability, and lack of fit values are also given in Table 2. The results for the R-sqd values of Y1, Y2, and Y3 are 98.50%, 98.14%, and 90.17% while the R2-adj values include 97.74%, 97.57%, and 87.17% for Y1, Y2, and Y3. The F-values of Y1, Y2, and Y3 are 131.01, 173.28, and 30.12 while the results for the lack of fit values of Y1, Y2, and Y3 are 0.076, 0.176, and 0.146, respectively (Table 2).
Table 2.
Regression coefficients, R2 and F-test probability for DH, DPPH• scavenging activity, and Fe2+-chelating activity.
| Factors | Coefficients |
|||||
|---|---|---|---|---|---|---|
| Y 1 | p value | Y 2 | p value | Y 3 | p value | |
| Intercept | −116.530 | 0.000 | −341.992 | 0.000 | 43.4458 | 0.005 |
| X 1 | 47.132 | 0.000 | 79.148 | 0.000 | −51.8738 | 0.000 |
| X 2 | 0.151 | 0.003 | 6.735 | 0.000 | 3.3275 | 0.000 |
| X 3 | 29.774 | 0.000 | −4.769 | 0.000 | 38.9285 | 0.000 |
| X 4 | 0.105 | 0.000 | −0.103 | 0.000 | −0.0010 | 0.709 |
| X 1 ∗X 1 | 4.226 | 0.000 | −7.041 | 0.000 | 4.7686 | 0.000 |
| X 2 ∗X 2 | −0.001 | 0.800 | −0.063 | 0.000 | −0.0310 | 0.000 |
| X 3 ∗X 3 | −8.371 | 0.000 | −1.484 | 0.487 | −13.7613 | 0.000 |
| X 4 ∗X 4 | −0.000 | 0.000 | 0.000 | 0.000 | 0.0001 | 0.077 |
| X 1 ∗X 3 | −1.024 | 0.016 | 0.229 | 0.592 | 0.2378 | 0.573 |
| X 2 ∗X 4 | −0.000 | 0.010 | −0.000 | 0.222 | 0.0001 | 0.671 |
| X 3 ∗X 4 | 0.026 | 0.000 | 0.006 | 0.114 | 0.0018 | 0.635 |
| R 2 (R-sqd) | 98.50% | 98.14% | 90.170% | |||
| R 2-adj | 97.74% | 97.57% | 87.17% | |||
| Lack of fit | 0.076 | 0.176 | 0.146 | |||
| F-values | 131.01 | 0.000 | 173.28 | 0.000 | 30.12 | 0.000 |
The large coefficient of determination (R2) and nonsignificant lack of fit values (p > 0.05) of Y1, Y2, and Y3 responses demonstrated the significance of the models and fitness of the experimental values to the theoretical values predicted by the model's regression equation (Table 2). The adjusted coefficient of determination (R2-adj) showed that the observed data variation of 97.74%, 97.57%, and 87.17% for Y1, Y2, and Y3 occurred due to the effects of the process conditions. The Fisher test (F-test) revealed high F-values and low p values of p < 0.05, which further validated the suitability of the models to the experimental data.
3.1. Degree of Hydrolysis (DH)
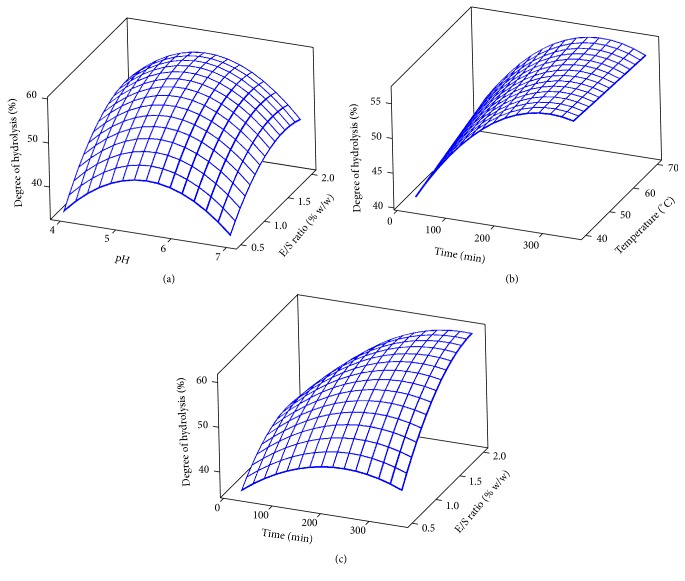
As shown in Table 2, a strong linear (p < 0.05) and quadratic (p < 0.05) effects of the hydrolysis variables in terms of pH, temperature, E/S ratio, and time were observed on DH as revealed by the regression coefficients. Hydrolysis temperature had no significant quadratic effect (p < 0.05) on DH. The three-dimensional (3D) response surface plots of DH are shown in Figure 1. The DH of stone fish protein was found to increase with the time of bromelain hydrolysis (Figure 1). As previously reported, the antioxidant activity is affected by the type of hydrolysis enzyme and the DH [14, 26, 27]. Ghanbari et al. [5] also observed higher DH of stone fish protein with increase in duration of hydrolysis for a total of 24 h.
Figure 1.
3D plots for degree of hydrolysis: (a) E/S ratio and pH; (b) temperature and time; (c) E/S ratio and time.
3.2. 2,2-Diphenyl-1-picrylhydrazyl Radical (DPPH•) Scavenging Activity
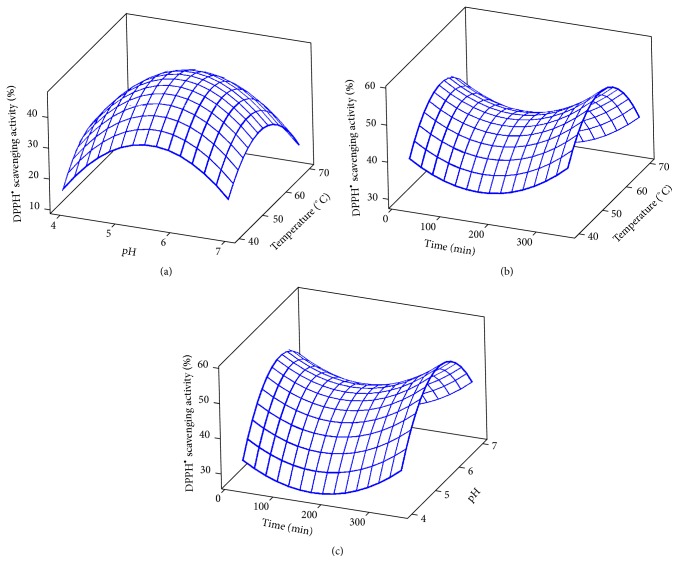
The results for the DPPH• scavenging activity are shown in Table 1 and Figure 2. The hydrolysis pH, temperature, and time indicated a strong linear (p < 0.05) and quadratic effects (p < 0.05) of the model with respect to DPPH• scavenging activity. However, the hydrolysis E/S ratio exerted only linear effect on DPPH• scavenging activity (Table 2). The 3D plots for the DPPH• scavenging activity are given in Figure 2. The ability of stone fish protein hydrolysates to scavenge free radicals could be due to their potential to donate hydrogen and neutralise or stabilise free radicals and terminate their propagation. The effect might also be attributed to specific amino acid sequences in the hydrolysates typically hydrophobic amino acids and histidine, whose imidazole ring can potentially chelate free radicals and trap lipid. This is possible by forming a physical barrier around fat droplets which delay lipid oxidation and prevents free radical chain reaction [27–29]. As earlier reported by Zarei et al. [26], the hydrolysates derived from palm kernel cake protein exhibited DPPH• scavenging activity that increased by increasing the protein hydrolysis time.
Figure 2.
3D plots for DPPH radical-scavenging activity: (a) temperature and pH; (b) temperature and time; (c) pH and time.
3.3. Ferrous Ion- (Fe2+-) Chelating Activity
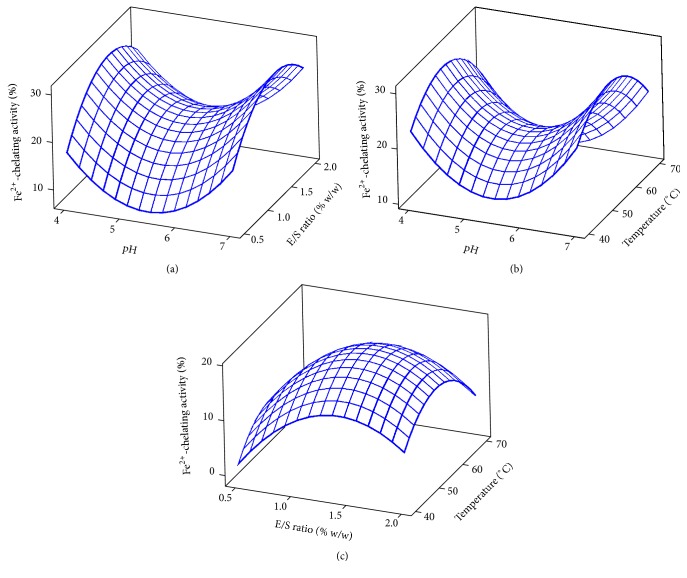
The results for the Fe2+-chelating activity of stone fish protein hydrolysates are shown in Table 1 and Figure 3. The coefficients of regression revealed strong effects for both linear (p < 0.05) and quadratic (p < 0.05) terms of the hydrolysis pH, temperature, and E/S ratio on Fe2+-chelating activity. The hydrolysis time indicated no significant (p < 0.05) effect on Fe2+-chelating activity of stone fish protein hydrolysates (Table 2). The 3D plots for the Fe2+-chelating activity are shown in Figure 3. The initiation of oxidative chain reactions may result from generation of the first few radicals through the catalytic action of transition metals. The availability of transition metals can be reduced by chelating agents; thereby inhibiting the free radical-induced lipid peroxidation in both food and living systems, thus enhancing animal health as well as food stability safety and quality [30, 31].
Figure 3.
3D plots for Fe2+-chelating activity: (a) E/S ratio and pH; (b) temperature and pH; (c) temperature and E/S ratio.
Transition metal ions acts as electron donors and react very quickly with peroxides resulting in alkoxyl radical formation [31].
The result obtained showed that the peptides in stone fish protein hydrolysates exhibited Fe2+-chelating activity and can potentially reduce lipid oxidation.
3.4. Effect of Shear Rate and Concentration on the Viscosity of SHs
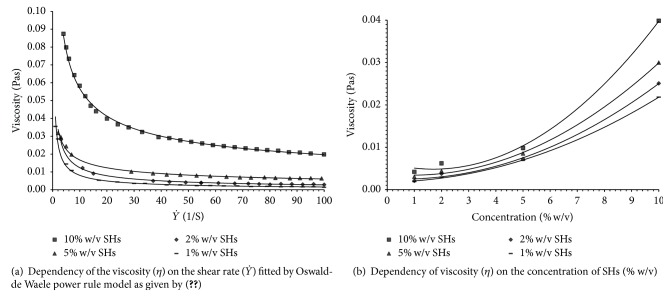
The flow properties of the hydrolysates (Figure 4(a)) revealed a decrease in apparent viscosity (η) of all concentrations with increasing shear rate (), which might be due to the deformation within the flow field [32]. The higher shear rate at increasing concentration was also associated with the greater intermolecular interaction between the amino acids composition of the hydrolysates.
Figure 4.
Dependency of viscosity on the shear rate (1/S) and concentration of SHs (% w/v).
The flow curve of each concentration was fitted using the power rule (see (4)) [33–36]. The SHs demonstrated non-Newtonian behavior (n < 1) or pseudoplasticity.
In addition, the effects of four different concentrations (1, 2, 5, and 10% w/v) were studied on the viscosity of SHs solutions at different speed using the same dynamic shear rheometer. As shown in Figure 4(b), the flow behavior of the hydrolysates at increasing concentration and fixed shear rates indicated their stronger shear-thinning effect and higher viscosities.
The curves are widely separated from each other at higher concentrations and closely related to one another at lower concentration where the viscosities lie much closer (Figure 4(b)). The figure has clearly showed that the SHs undergone conformational changes from being flexible to a more rigid structure with increase in concentration. Hence, the higher viscosities obtained at 10% w/v could be due to the formation of larger molecular weight assemblies or aggregates of the hydrolysates as a result ofinteraction between the constituent peptides fragments. However, the viscosity appears to be independent of the speed or shear rate at lower concentration. This is evident by the smaller change in viscosities at these concentrations under the different shear rates [33].
Shear induces the breakage of particles lumps or aggregates to promote their flow at a particular shear stress. Shear thinning is also mediated by the removal of solvent layers from dissolved molecules, thereby decreasing the intermolecular interactions, and reduces flow resistance [37]. The pseudoplastic property or shear-thinning flow behavior of the SHs was found to vary over the ranges of selected shear rates.
At low shear rate, the effect of the initial shear orientation does not influence the Brownian motion that set all the particles at random. The pseudoplastic SHs solutions exhibited zero shear that was independent of shear rate and behaves similar to Newtonian liquids.
As the shear rate increases, the shear induced particles orientation that exceeds the random effect of Brownian motion and the viscosity decreased drastically. The viscosity decreased with further shear rate until it reached an asymptotically finite constant level beyond which no more shear thinning occurs with higher shear rate and the optimum of perfect orientation is attained [37].
Thus, the viscosity of the non-Newtonic or the pseudoplastic hydrolysates was independent of shear rate at both low (first Newtonian) and high (second Newtonian) ranges.
3.5. Optimisation of Hydrolysis Conditions
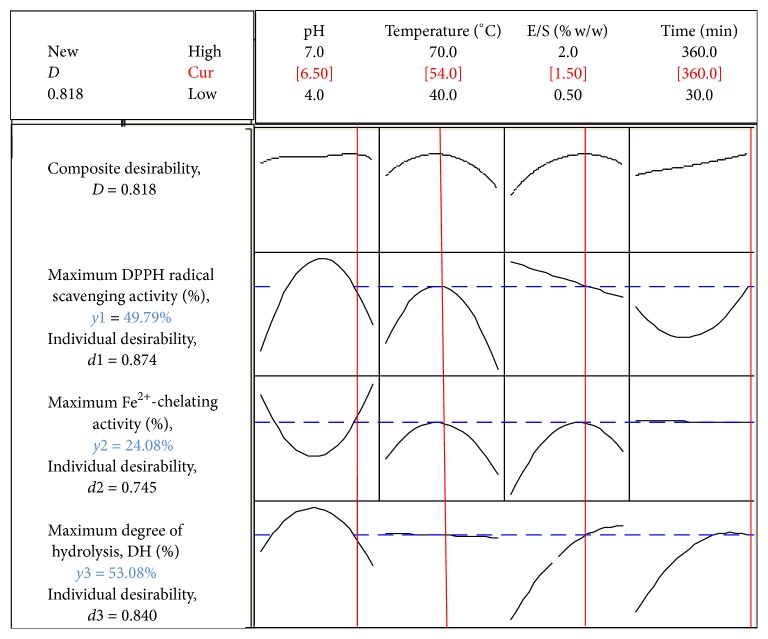
Response surface optimiser (Figure 5) was used to determine the optimum conditions for the hydrolysis of stone fish protein to produce hydrolysates with maximum responses in terms of DPPH• scavenging activity and Fe2+-chelating activity. The optimum levels obtained for the hydrolysis conditions were pH (6.5), temperature (54°C), E/S ratio (1.5% w/w), and time (360 min), respectively. The results for DH (54.62%) as well as DPPH• scavenging activity (48.94%) and Fe2+-chelating activity (25.12%) obtained under these conditions were not statistically different from the predicted values of DH 53.08%; DPPH• scavenging activity 49.79%; and Fe2+-chelating activity 24.08%, respectively, within 95% confidence interval.
Figure 5.
Response optimisation parameters, maximum predicted responses, and desirability levels.
Antioxidants act by absorbing free radicals and chelating metals to suppress lipid peroxidation [38]. Consequently, the antioxidant properties of stone fish protein hydrolysates can be exploited as a valuable functional ingredient in food formulation to improve dietary intake of antioxidants. Moreover, the hydrolysates can serve as a natural and safe additive to enhance the shelf-life of various food products that are susceptible to oxidation.
4. Conclusions
In this study, the optimal conditions of bromelain for the hydrolysis of stone fish protein in terms of pH, temperature, E/S ratio, and time were established for maximum responses of DPPH radical-scavenging activity and Fe2+-chelating activity using RSM. The optimised levels of these hydrolysis conditions were found to be pH 6.5, temperature 54°C, E/S ratio 1.5% w/w, and time 360 min. Under these conditions, the optimum values obtained for DH 54.62%, DPPH radical-scavenging activity 48.94%, and Fe2+-chelating activity 25.12% were not statistically different from the predicted values of DH 53.08%, DPPH radical-scavenging activity 49.79%, and Fe2+-chelating activity 24.08%, respectively, within 95% confidence interval.
Acknowledgments
This work was supported under Project no. 02-01-04-SF 2309 by the grant from the Malaysian Ministry of Science, Technology and Innovation.
Abbreviations
- SHs:
Stone fish protein hydrolysates
- RSM:
Response surface methodology
- CCD:
Central composite design
- OPA:
O-Phthaldialdehyde
- DH:
Degree of hydrolysis
- DPPH:
2,2-Diphenyl-1-picrylhydrazyl
- ANOVA:
Analysis of variance.
Conflicts of Interest
All the authors declare that they have no conflicts of interest.
Authors' Contributions
Nazamid Saari initiated the research and supervised its conduct from beginning to the final preparation of the manuscript; Shehu Muhammad Auwal designed and carried out the experiments as well as writing of the manuscript; Shehu Muhammad Auwal and Mohammad Zarei analysed the data; Azizah Abdul-Hamid contributed to the supervision process.
References
- 1.Benjakul S., Yarnpakdee S., Senphan T., Halldorsdottir S. M., Kristinsson H. G. Fish protein hydrolysates: production, bioactivities, and applications. In: Kristinsson G. H., editor. Antioxidants and Functional Components in Aquatic Foods. 1st. Chichester, West Sussex: John Wiley and Sons; 2014. pp. 237–281. [Google Scholar]
- 2.McCarthy A., O'Callaghan Y. C., O'Brien N. M. Protein Hydrolysates from Agricultural Crops—Bioactivity and Potential for Functional Food Development. Agriculture. 2013;3(1):112–130. doi: 10.3390/agriculture3010112. [DOI] [Google Scholar]
- 3.Bhaskar N., Modi V. K., Govindaraju K., Radha C., Lalitha R. G. Utilization of meat industry by products: Protein hydrolysate from sheep visceral mass. Bioresource Technology. 2007;98(2):388–394. doi: 10.1016/j.biortech.2005.12.017. [DOI] [PubMed] [Google Scholar]
- 4.Bordbar S., Anwar F., Saari N. High-value components and bioactives from sea cucumbers for functional foods—a review. Marine Drugs. 2011;9(10):1761–1805. doi: 10.3390/md9101761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ghanbari R., Ebrahimpour A., Abdul-Hamid A., Ismail A., Saari N. Actinopyga lecanora hydrolysates as natural antibacterial agents. International Journal of Molecular Sciences. 2012;13(12):16796–16811. doi: 10.3390/ijms131216796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bordbar S., Ebrahimpour A., Abdul Hamid A., Abdul Manap M. Y., Anwar F., Saari N. The improvement of the endogenous antioxidant property of stone fish (Actinopyga lecanora) tissue using enzymatic proteolysis. BioMed Research International. 2013;2013:9. doi: 10.1155/2013/849529.849529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lakshmi V., Ghosal S. In vitro and in vivo antiamoebic potential of actinopyga lecanora (jaeger) Bangladesh Pharmaceutical Journal. 2015;18(2):p. 118. doi: 10.3329/bpj.v18i2.24308. [DOI] [Google Scholar]
- 8.Ghanbari R., Zarei M., Ebrahimpour A., Abdul-Hamid A., Ismail A., Saari N. Angiotensin-I converting enzyme (ACE) inhibitory and anti-oxidant activities of sea cucumber (actinopyga lecanora) hydrolysates. International Journal of Molecular Sciences. 2015;16(12):28870–28885. doi: 10.3390/ijms161226140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dean A. M., Voss D. T. Design and analysis of experiments. New York, NY, USA: Springer-Verlag; 1999. (Springer Texts in Statistics). [DOI] [Google Scholar]
- 10.Majeed M., Hussain A. I., Chatha S. A. S., et al. Optimization protocol for the extraction of antioxidant components from Origanum vulgare leaves using response surface methodology. Saudi Journal of Biological Sciences. 2016;23(3):389–396. doi: 10.1016/j.sjbs.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tavares T. G., Contreras M. M., Amorim M. Optimisation, by response surface methodology, of degree of hydrolysis and antioxidant and ACE-inhibitory activities of whey protein hydrolysates obtained with cardoon extract. International Dairy Journal. 2011;21(12):926–933. doi: 10.1016/j.idairyj.2011.05.013. [DOI] [Google Scholar]
- 12.Ren J., Zhao M., Shi J., et al. Optimization of antioxidant peptide production from grass carp sarcoplasmic protein using response surface methodology. LWT - Food Science and Technology. 2008;41(9):1624–1632. doi: 10.1016/j.lwt.2007.11.005. [DOI] [Google Scholar]
- 13.Madamba P. S. The response surface methodology: An application to optimize dehydration operations of selected agricultural crops. LWT - Food Science and Technology. 2002;35(7):584–592. doi: 10.1016/S0023-6438(02)90914-X. doi: 10.1016/S0023-6438(02)90914-X. [DOI] [Google Scholar]
- 14.Seo H.-W., Jung E.-Y., Go G.-W., Kim G.-D., Joo S.-T., Yang H.-S. Optimization of hydrolysis conditions for bovine plasma protein using response surface methodology. Food Chemistry. 2015;185:106–111. doi: 10.1016/j.foodchem.2015.03.133. [DOI] [PubMed] [Google Scholar]
- 15.Zhuang Y.-L., Zhao X., Li B.-F. Optimization of antioxidant activity by response surface methodology in hydrolysates of jellyfish (Rhopilema esculentum) umbrella collagen. Journal of Zhejiang University: Science B. 2009;10(8):572–579. doi: 10.1631/jzus.B0920081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Meshginfar N., Sadeghi-Mahoonak A., Ziaiifar A. M., Ghorbani M., Kashaninejad M. Study of antioxidant activity of sheep visceral protein hydrolysate: Optimization using response surface methodology. ARYA Atherosclerosis. 2014;10(4):179–184. [PMC free article] [PubMed] [Google Scholar]
- 17.Goudarzi M., Madadlou A., Mousavi M. E., Emam-Djomeh Z. Optimized preparation of ACE-inhibitory and antioxidative whey protein hydrolysate using response surface method. Dairy Science and Technology. 2012;92(6):641–653. doi: 10.1007/s13594-012-0081-6. [DOI] [Google Scholar]
- 18.Kechaou E. S., Bergé J.-P., Jaouen P., Ben Amar R. Optimization of common cuttlefish (Sepia officinalis) protein hydrolysate using pepsin by response surface methodology. Journal of Aquatic Food Product Technology. 2015;24(3):270–282. doi: 10.1080/10498850.2013.773116. [DOI] [Google Scholar]
- 19.Fan S., Hu Y., Li C., Liu Y. Optimization of preparation of antioxidative peptides from pumpkin seeds using response surface method. PLoS ONE. 2014;9(3):p. e92335. doi: 10.1371/journal.pone.0092335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Auwal S. M., Zarei M., Abdul-Hamid A., Saari N. Optimization of bromelain-aided production of angiotensin I-converting enzyme inhibitory hydrolysates from stone fish using response surface methodology. Marine Drugs. 2017;15(4):p. 104. doi: 10.3390/md15040104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nielsen P. M., Petersen D., Dambmann C. Improved method for determining food protein degree of hydrolysis. Journal of Food Science. 2001;66(5):642–646. doi: 10.1111/j.1365-2621.2001.tb04614.x. [DOI] [Google Scholar]
- 22.Mirzaei M., Mirdamadi S., Ehsani M. R., Aminlari M., Hosseini E. Purification and identification of antioxidant and ACE-inhibitory peptide from Saccharomyces cerevisiae protein hydrolysate. Journal of Functional Foods. 2015;19:259–268. doi: 10.1016/j.jff.2015.09.031. [DOI] [Google Scholar]
- 23.Hwang J.-Y., Shyu Y.-S., Wang Y.-T., Hsu C.-K. Antioxidative properties of protein hydrolysate from defatted peanut kernels treated with esperase. LWT - Food Science and Technology. 2010;43(2):285–290. doi: 10.1016/j.lwt.2009.08.020. [DOI] [Google Scholar]
- 24.Carter P. Spectrophotometric determination of serum iron at the submicrogram level with a new reagent (ferrozine) Analytical Biochemistry. 1971;40(2):450–458. doi: 10.1016/0003-2697(71)90405-2. [DOI] [PubMed] [Google Scholar]
- 25.Zarei M., Ghanbari R., Tajabadi N., Abdul-Hamid A., Bakar F. A., Saari N. Generation, Fractionation, and Characterization of Iron-Chelating Protein Hydrolysate from Palm Kernel Cake Proteins. Journal of Food Science. 2016;81(2):C341–C347. doi: 10.1111/1750-3841.13200. [DOI] [PubMed] [Google Scholar]
- 26.Zarei M., Ebrahimpour A., Abdul-Hamid A., Anwar F., Saari N. Production of defatted palm kernel cake protein hydrolysate as a valuable source of natural antioxidants. International Journal of Molecular Sciences. 2012;13(7):8097–8111. doi: 10.3390/ijms13078097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kong B., Xiong Y. L. Antioxidant activity of zein hydrolysates in a liposome system and the possible mode of action. Journal of Agricultural and Food Chemistry. 2006;54(16):6059–6068. doi: 10.1021/jf060632q. [DOI] [PubMed] [Google Scholar]
- 28.Zhao L., Wang S., Huang Y. Antioxidant function of tea dregs protein hydrolysates in liposome-meat system and its possible action mechanism. International Journal of Food Science and Technology. 2014;49(10):2299–2306. doi: 10.1111/ijfs.12546. [DOI] [Google Scholar]
- 29.Pihlanto A. Antioxidative peptides derived from milk proteins. International Dairy Journal. 2006;16(11):1306–1314. doi: 10.1016/j.idairyj.2006.06.005. [DOI] [Google Scholar]
- 30.Liu Q., Kong B., Xiong Y. L., Xia X. Antioxidant activity and functional properties of porcine plasma protein hydrolysate as influenced by the degree of hydrolysis. Food Chemistry. 2010;118(2):403–410. doi: 10.1016/j.foodchem.2009.05.013. [DOI] [Google Scholar]
- 31.Pokorny J., Yanishlieva N., Gordon M. Antioxidants in food: practical applications. 1st. Abington Hall, Abington Cambridge CB1 6AH, England: Woodhead Publishing Limited; 2001. [DOI] [Google Scholar]
- 32.McClements D. J. D. J. McClements. Food Emulsion Principles. 2nd. Boca Raton, Florida 33431, USA: CRC Press; 2000. Food emulsion principles, practices, and techniques; pp. 361–364. [Google Scholar]
- 33.Thaiphanit S., Schleining G., Anprung P. Effects of coconut (Cocos nucifera L.) protein hydrolysates obtained from enzymatic hydrolysis on the stability and rheological properties of oil-in-water emulsions. Food Hydrocolloids. 2016;60:252–264. doi: 10.1016/j.foodhyd.2016.03.035. [DOI] [Google Scholar]
- 34.Kurt A., Kahyaoglu T. Rheological properties and structural characterization of salep improved by ethanol treatment. Carbohydrate Polymers. 2015;133:654–661. doi: 10.1016/j.carbpol.2015.07.028. [DOI] [PubMed] [Google Scholar]
- 35.Chen H., Wu F., Duan X., et al. Characterization of emulsions prepared by egg yolk phosvitin with pectin, glycerol and trehalose. Food Hydrocolloids. 2013;30(1):123–129. doi: 10.1016/j.foodhyd.2012.05.007. [DOI] [Google Scholar]
- 36.Wang B., Wang L.-J., Li D., Adhikari B., Shi J. Effect of gum Arabic on stability of oil-in-water emulsion stabilized by flaxseed and soybean protein. Carbohydrate Polymers. 2011;86(1):343–351. doi: 10.1016/j.carbpol.2011.04.059. [DOI] [Google Scholar]
- 37.Schramm G. A Practical Approach to Rheology and Rheometry. 2nd. D-76227 karlsruhe, Dieselstrasse 4 Federal Republic of Germany: Cambridge University Press; 2000. (Gebrueder HAAKE GmbH). [DOI] [Google Scholar]
- 38.Nimse S. B., Pal D. Free radicals, natural antioxidants, and their reaction mechanisms. Royal Society and Chemistry Advances. 2015;5(35):27986–28006. doi: 10.1039/c4ra13315c. [DOI] [Google Scholar]