Abstract
Children with cancer treated with cytotoxic drugs are frequently at risk of developing renal dysfunction. The cytotoxic drugs that are widely used for cancer treatment in children are cisplatin (CPL), ifosfamide (IFO), carboplatin, and methotrexate (MTX). Mechanisms of anticancer drug-induced renal disorders are different and include acute kidney injury (AKI), tubulointerstitial disease, vascular damage, hemolytic uremic syndrome (HUS), and intrarenal obstruction. CPL nephrotoxicity is dose-related and is often demonstrated with hypomagnesemia, hypokalemia, and impaired renal function with rising serum creatinine and blood urea nitrogen levels. CPL, mitomycin C, and gemcitabine treatment cause vascular injury and HUS. High-dose IFO, streptozocin, and azacitidine cause renal tubular dysfunction manifested by Fanconi syndrome, rickets, and osteomalacia. AKI is a common adverse effect of MTX, interferon-alpha, and nitrosourea compound treatment. These strategies to reduce the cytotoxic drug-induced nephrotoxicity should include adequate hydration, forced diuresis, and urinary alkalization. Amifostine, sodium thiosulfate, and diethyldithiocarbamate provide protection against CPL-induced renal toxicity.
Keywords: Acute kidney injury, anti-cancer drugs, chemotherapy, children, glomerular filtration rate, nephrotoxicity
Introduction
Renal injury may follow treatment with anticancer drugs and lead to glomerular, tubular dysfunctions, or any combination of these.[1,2] Acute kidney injury (AKI), tubulointerstitial disease, renal capillary endothelial injury and renal electrolytes, and acid-base disorders are the most frequent adverse effects of chemotherapy in children with cancer.[3]
Mechanisms of anticancer drug-induced renal disorders generally include a varying degree of prerenal hypoperfusion, intrinsic renal damage, renal tubular obstruction, and damage to the microvascular structure of the kidneys.[4,5,6]
Patients with malignancy often treated with nephrotoxic antibiotics such as aminoglycosides, vancomycin, and amphotericin B for concurrent bacterial and fungal infections. These patients may also undergo radiation therapy and various radiologic studies with iodine contrast media, which can cause nephrotoxicity.[7,8]
Other risk factors potentiating anticancer-induced nephrotoxicity are extracellular volume depletion, preexisting renal insufficiency, cardiovascular disease, diabetes mellitus, and concomitant use of nonsteroidal NSAIDs allopurinol or other nephrotoxic medications.[9,10]
Chemotherapy-induced nephrotoxicity is a major cause of morbidity and mortality among cancer patients. Therefore, assessing baseline renal function before initiation of therapy and during therapy, adjusting drug dosages, avoiding nephrotoxic drug combinations, and correcting the extracellular fluid volume depletion is essential in the cancer patients.[11]
This review highlights the potential preventive strategies to reduce nephrotoxicity induced by anticancer treatment in children. For this review, we searched current medical literature through PubMed/MEDLINE, EMBASE, Scopus, Cochrane Central Register of Controlled Trials, and Google Scholar search databases, and EBSCO for keywords such as AKI, anticancer drugs, chemotherapy, children, glomerular filtration rate (GFR), and nephrotoxicity as published in English over the last four decades.
Methods
We aimed to provide an organized practical approach for the pediatrician to preventing drug-related nephrotoxicity in children and adolescents to address the following key questions:
What is the prevalence of nephtotoxicity and its associated comorbidities in children and adolescents?
How should chemotherapy- related nephrotoxicity in children and adolescents be diagnosed, and what is the optimal approach to preventing nephrotoxicity in Pediatric patients?
What is the recommended workup for early detection of kidney involvement in patients treated with anticancer?
To address these key questions a systematic search and review of literature was performed using MEDLINE, EMBASE, and Chocrane of Database since 1984 and then the search was extended as linked citations indicated. The topics searched in included child, adolescent, intervention, anti cancer drugs, adverse effects, and outcome.
Results
A total of 36 selected original and review articles which, met the inclusion criteria included in this review. Chemotherapy-induced nephrotoxicity is a common finding among children and adolescent patients treated for cancer. It can be indicative of a significant renal disease. Evaluation of anticancer nephrotoxicity should begin with a careful history and thorough physical examination, urine microscopic examination, measurement of estimated creatinine clearance, and first morning urine for protein/creatinine ratio. Based on the physical findings and clinical judgment then further appropriate laboratory tests and radiologic evaluation may be indicated. Pediatric nephrologist referral is recommended when renal dysfunction persists beyond 2-3 months after completion of anticancer therapy.
Discussion
Assessment of glomerular filtration rate
GFR should be evaluated before and during the course of anticancer drug therapy for the early detection of renal toxicity. The renal clearance of inulin is the most reliable method for determination of a true GFR, but its use is limited because of expense, lack of availability, and problems with collecting timed urine samples in infants and small children. As a result, GFR estimation (eGFR) from the renal clearance of creatinine has been widely used in the pediatric population.
Creatinine-based eGFR without urine collection has been used frequently in children.
There are several ways to assess the eGFR in children. One of the easiest and more practical ones is Schwartz formula, which uses the concept of height as a measure of muscle mass divided by serum creatinine.[12,13]
eGFR (mL/min/1.73 m2) = (K) (height in cm)/serum creatinine (mg/dL). The value of K is 0.35 for low-birth-weight infants >1 year old, 0.45 for term infants throughout the 1st year of life, 0.55 for children and adolescent girls,[13] and 0.70 for adolescent boys.[13,14,15]
However, the use of Schwartz formula with nutritional compromise as is common in children with cancer undergoing radiation and chemotherapy may overestimate the eGFR values with subsequent overdosing of nephrotoxic medications.
Similarly, in patients with chronic kidney disease (CKD), falsely elevated creatinine clearance can occur due to the increase in tubular creatinine excretion.[16,17] Inhibition of tubular creatinine excretion by cimetidine may provide the accurate measurement of GFR in CKD patients.[18,19]
The overestimation of GFR using creatinine-based equations has resulted in higher than intended carboplatin doses with a potential for increased renal toxicity. This concern for patient's safety led to the development of Calvert formula to determine the optimal carboplatin dosages for cancer patients.[20] Calvert formula is used to cap the maximum carboplatin dose based on the target area under the curve (AUC). It is recommended that the maximum carboplatin dose should not exceed the target AUC [Table 1].
Table 1.
Maximum area under curve-based carboplatin dosage

Calvert formula
Total carboplatin dose (mg) = Target AUC (mg/mL/min) × creatinine clearance (mL/min) + 25 [Table 1].
Whereas the Schwartz formula is the widely used equation for the eGFR in children, the Modification of Diet in Renal Disease and Cockroft-Gault equations are the most popular GFR estimate for adults but are inaccurate for patients <18 years old.[21,22,23]
The plasma disappearance of (51) Cr-EDTA and iohexol are also used for GFR determinations; however, these are not ideal for use in children, especially for repeated studies.[24,25] Recently, Schwartz and Associates developed the several new equations to eGFR in children using patient's height, serum creatinine, cystatin C,[26,27] and blood urea nitrogen (BUN) of which the best equation with the highest accuracy and correlation and the narrowest 95% limits of agreement were as follows:
eGFR = 39.1 × (height [m2/SCr [mg/dL]])0.516 × (1.8/cysteine C [mg/L])0.294 × (30/BUN [mg/dL])0.169 × [1.099 male] × [height (m/1.4)]0.188.[18]
These authors, from this equation, also described a simple bedside equation of 0.413 × height (cm)/serum creatinine (mg/dL), which provides a good approximation of eGFR in children and adolescents with CKD based on serum creatinine determinations.[28]
Cisplatin (CPL) and ifosfamide (IFO) are the most widely used nephrotoxic drugs in children with cancer. Renal toxicity is less common with other cytotoxic drugs, such as carboplatin, methotrexate (MTX), and nitrosourea compounds [Table 2].
Table 2.
Nephrotoxicity associated with antineoplastic drugs
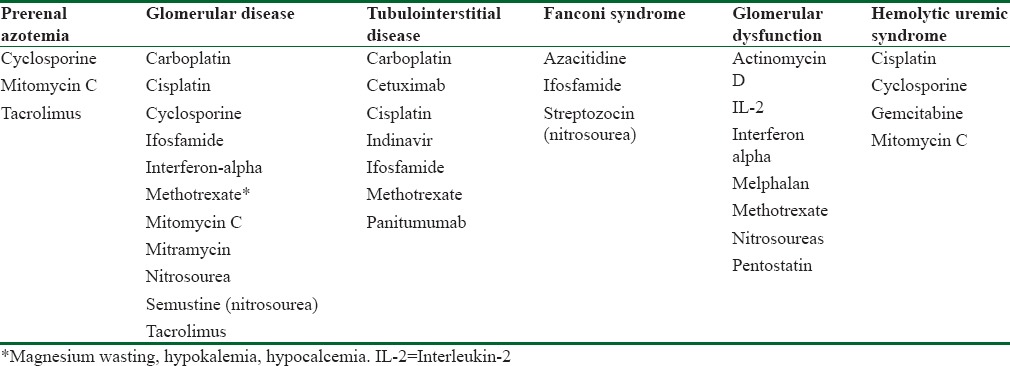
The treatment strategy to prevent drug-induced nephrotoxicity should include the use of intravenous hydration, forced diuresis, and elimination of known risk factors.
CPL can cause glomerular and tubular damage resulting in decreased GFR. The CPL-nephrotoxicity is known to be a risk factor and may also be a dose related.[29,30] Hypokalemia, hypomagnesemia, and hypocalcaemia are the most common CPL-related electrolyte disorders. Rising BUN and serum creatinine levels followed by AKI are generally noted after a bolus CPL administration >100 mg/m2/day. Forced diuresis, combing hydration with furosemide administration has proved clinically beneficial to prevent kidney injury.[31] The carbonic anhydrase inhibitor acetazolamide has also been considered useful for the prevention of contrast nephropathy and CPL nephrotoxicity.[32,33] Amifostine, sodium thiosulfate, and diethyldithiocarbamate also confer protection against CPL-induced renal dysfunction.[33]
The combination of CPL and carboplatin are associated with more renal toxicity than CPL and IFO alone or combined.[29,30]
Life-threatening carboplatin associated nephrotoxicity may occur from errors in calculating GFR. To avoid this, it is recommended to dose carboplatin by modified Calvert formula [Table 1].[20]
IFO can also cause glomerular and tubular toxicity. Proximal tubular dysfunction is a prominent complication of IFO therapy characterized by Fanconi syndrome and hypophosphatemic rickets.[34] IFO dose >100 g/m2, age younger than 5 years, and combination therapy with CPL and carboplatin increase the risk of IFO-associated nephrotoxicity.[34,35]
High-dose MTX can induce both glomerular and tubular dysfunction leading to delayed elimination of the drug.[36] The precipitation of drug may occur in concentrated and acid urine resulting in renal tubular obstruction and AKI. Adequate hydration and urine alkalization are the most important measures for the prevention of MTX-induced nephrotoxicity.
Monitoring the serum MTX level is necessary to prevent systemic toxicity including nephrotoxicity. Apart from vigorous hydration and alkalization of urine, leucovorin should be administered in high-dose MTX therapy not only on the absolute drug serum level >10−7 mol/L but also on the kinetics of these levels.[36]
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Denker B, Robles-Osorio ML, Sabath E. Recent advances in diagnosis and treatment of acute kidney injury in patients with cancer. Eur J Intern Med. 2011;22:348–54. doi: 10.1016/j.ejim.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 2.Lameire N, Van Biesen W, Vanholder R. Electrolyte disturbances and acute kidney injury in patients with cancer. Semin Nephrol. 2010;30:534–47. doi: 10.1016/j.semnephrol.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 3.Salahudeen AK, Doshi SM, Pawar T, Nowshad G, Lahoti A, Shah P. Incidence rate, clinical correlates, and outcomes of AKI in patients admitted to a comprehensive cancer center. Clin J Am Soc Nephrol. 2013;8:347–54. doi: 10.2215/CJN.03530412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lameire N, Kruse V, Rottey S. Nephrotoxicity of anticancer drugs – An underestimated problem? Acta Clin Belg. 2011;66:337–45. doi: 10.2143/ACB.66.5.2062585. [DOI] [PubMed] [Google Scholar]
- 5.Salahudeen AK, Bonventre JV. Onconephrology: The latest frontier in the war against kidney disease. J Am Soc Nephrol. 2013;24:26–30. doi: 10.1681/ASN.2012070690. [DOI] [PubMed] [Google Scholar]
- 6.Perazella MA. Onco-nephrology: Renal toxicities of chemotherapeutic agents. Clin J Am Soc Nephrol. 2012;7:1713–21. doi: 10.2215/CJN.02780312. [DOI] [PubMed] [Google Scholar]
- 7.Lameire N. The kidney in oncology. Acta Clin Belg. 2007;62:141–54. doi: 10.1179/acb.2007.025. [DOI] [PubMed] [Google Scholar]
- 8.Cummings BS, Schnellmann RG. Pathophysiology of nephrotoxic cell injury. In: Coffman TM, Falk RJ, Molitoris BA, editors. Schrier's Diseases of the Kidney. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2013. pp. 868–900. [Google Scholar]
- 9.Lam AQ, Humphreys BD. Onco-nephrology: AKI in the cancer patient. Clin J Am Soc Nephrol. 2012;7:1692–700. doi: 10.2215/CJN.03140312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Safirstein RL. Renal diseases induced by antineoplastic agents. In: Schrier RW, editor. Diseases of the Kidney and Urinary Tract. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2007. pp. 1068–81. [Google Scholar]
- 11.Kleber M, Cybulla M, Bauchmüller K, Ihorst G, Koch B, Engelhardt M. Monitoring of renal function in cancer patients: An ongoing challenge for clinical practice. Ann Oncol. 2007;18:950–8. doi: 10.1093/annonc/mdm055. [DOI] [PubMed] [Google Scholar]
- 12.Schwartz GJ, Haycock GB, Edelmann CM, Jr, Spitzer A. A simple estimate of glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics. 1976;58:259–63. [PubMed] [Google Scholar]
- 13.Schwartz GJ, Brion LP, Spitzer A. The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children, and adolescents. Pediatr Clin North Am. 1987;34:571–90. doi: 10.1016/s0031-3955(16)36251-4. [DOI] [PubMed] [Google Scholar]
- 14.Schwartz GJ, Feld LG, Langford DJ. A simple estimate of glomerular filtration rate in full-term infants during the first year of life. J Pediatr. 1984;104:849–54. doi: 10.1016/s0022-3476(84)80479-5. [DOI] [PubMed] [Google Scholar]
- 15.Schwartz GJ, Gauthier B. A simple estimate of glomerular filtration rate in adolescent boys. J Pediatr. 1985;106:522–6. doi: 10.1016/s0022-3476(85)80697-1. [DOI] [PubMed] [Google Scholar]
- 16.Mitch WE, Walser M. A proposed mechanism for reduced creatinine excretion in severe chronic renal failure. Nephron. 1978;21:248–54. doi: 10.1159/000181400. [DOI] [PubMed] [Google Scholar]
- 17.Levey AS, Perrone RD, Madias NE. Serum creatinine and renal function. Annu Rev Med. 1988;39:465–90. doi: 10.1146/annurev.me.39.020188.002341. [DOI] [PubMed] [Google Scholar]
- 18.Hellerstein S, Berenbom M, Alon US, Warady BA. Creatinine clearance following cimetidine for estimation of glomerular filtration rate. Pediatr Nephrol. 1998;12:49–54. doi: 10.1007/s004670050402. [DOI] [PubMed] [Google Scholar]
- 19.Schwartz GJ, Work DF. Measurement and estimation of GFR in children and adolescents. Clin J Am Soc Nephrol. 2009;4:1832–43. doi: 10.2215/CJN.01640309. [DOI] [PubMed] [Google Scholar]
- 20.van Warmerdam LJ, Rodenhuis S, ten Bokkel Huinink WW, Maes RA, Beijnen JH. The use of the Calvert formula to determine the optimal carboplatin dosage. J Cancer Res Clin Oncol. 1995;121:478–86. doi: 10.1007/BF01218365. [DOI] [PubMed] [Google Scholar]
- 21.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130:461–70. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 22.Filler G, Foster J, Acker A, Lepage N, Akbari A, Ehrich JH. The Cockcroft-Gault formula should not be used in children. Kidney Int. 2005;67:2321–4. doi: 10.1111/j.1523-1755.2005.00336.x. [DOI] [PubMed] [Google Scholar]
- 23.Pierrat A, Gravier E, Saunders C, Caira MV, Aït-Djafer Z, Legras B, et al. Predicting GFR in children and adults: A comparison of the Cockcroft-Gault, Schwartz, and modification of diet in renal disease formulas. Kidney Int. 2003;64:1425–36. doi: 10.1046/j.1523-1755.2003.00208.x. [DOI] [PubMed] [Google Scholar]
- 24.Rehling M, Møller ML, Thamdrup B, Lund JO, Trap-Jensen J. Simultaneous measurement of renal clearance and plasma clearance of 99mTc-labelled diethylenetriaminepenta-acetate, 51Cr-labelled ethylenediaminetetra-acetate and inulin in man. Clin Sci (Lond) 1984;66:613–9. doi: 10.1042/cs0660613. [DOI] [PubMed] [Google Scholar]
- 25.Schwartz GJ, Furth S, Cole SR, Warady B, Muñoz A. Glomerular filtration rate via plasma iohexol disappearance: Pilot study for chronic kidney disease in children. Kidney Int. 2006;69:2070–7. doi: 10.1038/sj.ki.5000385. [DOI] [PubMed] [Google Scholar]
- 26.Coll E, Botey A, Alvarez L, Poch E, Quintó L, Saurina A, et al. Serum cystatin C as a new marker for noninvasive estimation of glomerular filtration rate and as a marker for early renal impairment. Am J Kidney Dis. 2000;36:29–34. doi: 10.1053/ajkd.2000.8237. [DOI] [PubMed] [Google Scholar]
- 27.Bökenkamp A, Domanetzki M, Zinck R, Schumann G, Byrd D, Brodehl J. Cystatin C serum concentrations underestimate glomerular filtration rate in renal transplant recipients. Clin Chem. 1999;45:1866–8. [PubMed] [Google Scholar]
- 28.Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, et al. New equations to estimate GFR in children with CKD. J Am Soc Nephrol. 2009;20:629–37. doi: 10.1681/ASN.2008030287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hartmann JT, Fels LM, Franzke A, Knop S, Renn M, Maess B, et al. Comparative study of the acute nephrotoxicity from standard dose cisplatin+/- ifosfamide and high-dose chemotherapy with carboplatin and ifosfamide. Anticancer Res. 2000;20:3767–73. [PubMed] [Google Scholar]
- 30.Skinner R, Parry A, Price L, Cole M, Craft AW, Pearson AD. Persistent nephrotoxicity during 10-year follow-up after cisplatin or carboplatin treatment in childhood: Relevance of age and dose as risk factors. Eur J Cancer. 2009;45:3213–9. doi: 10.1016/j.ejca.2009.06.032. [DOI] [PubMed] [Google Scholar]
- 31.Howell SB, Taetle R. Effect of sodium thiosulfate on cis-dichlorodiammineplatinum (II) toxicity and antitumor activity in L1210 leukemia. Cancer Treat Rep. 1980;64:611–6. [PubMed] [Google Scholar]
- 32.Assadi F. Acetazolamide for prevention of contrast-induced nephropathy: A new use for an old drug. Pediatr Cardiol. 2006;27:238–42. doi: 10.1007/s00246-005-1132-z. [DOI] [PubMed] [Google Scholar]
- 33.Osman NM, Copley MP, Litterst CL. Amelioration of cisplatin-induced nephrotoxicity by the diuretic acetazolamide in F344 rats. Cancer Treat Rep. 1984;68:999–1004. [PubMed] [Google Scholar]
- 34.Stöhr W, Paulides M, Bielack S, Jürgens H, Treuner J, Rossi R, et al. Ifosfamide-induced nephrotoxicity in 593 sarcoma patients: A report from the Late Effects Surveillance System. Pediatr Blood Cancer. 2007;48:447–52. doi: 10.1002/pbc.20858. [DOI] [PubMed] [Google Scholar]
- 35.Oberlin O, Fawaz O, Rey A, Niaudet P, Ridola V, Orbach D, et al. Long-term evaluation of Ifosfamide-related nephrotoxicity in children. J Clin Oncol. 2009;27:5350–5. doi: 10.1200/JCO.2008.17.5257. [DOI] [PubMed] [Google Scholar]
- 36.Widemann BC, Balis FM, Kim A, Boron M, Jayaprakash N, Shalabi A, et al. Glucarpidase, leucovorin, and thymidine for high-dose methotrexate-induced renal dysfunction: Clinical and pharmacologic factors affecting outcome. J Clin Oncol. 2010;28:3979–86. doi: 10.1200/JCO.2009.25.4540. [DOI] [PMC free article] [PubMed] [Google Scholar]


