Genome editing and glaucoma
Genome editing treats glaucoma in mouse model. Image courtesy of iStockphoto/Natali_Mis.
Mutations in the myocilin gene (MYOC) can lead to the misfolding and toxic buildup of the encoded protein in the trabecular meshwork (TM), a tissue that regulates pressure inside the eyes, triggering the death of TM cells, raising intraocular pressure, and leading to glaucoma. Ankur Jain et al. (pp. 11199–11204) used CRISPR-Cas9–mediated genome editing to disrupt the production of mutant myocilin in mouse and human TM cells, as well as in a mouse model of primary open glaucoma. Cells in which mutant MYOC was disrupted exhibited reduced levels of both myocilin and protein biomarkers of TM cell stress. Intravitreal injection of an adenovirus-based CRISPR construct that disrupted mutant MYOC lowered intraocular pressure and prevented progressive eye damage in the mouse model, compared with mice that received a control injection. The CRISPR construct also reversed elevated intraocular pressure in mice that exhibited elevated pressure for 9 months before treatment onset. Further, the authors demonstrated that treatment of perfusion-cultured human eyes with the CRISPR construct could disrupt secretion of myocilin into the culture medium, suggesting the translational potential of the approach. According to the authors, the findings provide a basis for the development of therapeutic genome editing for a common eye disorder with genetic underpinnings. — P.N.
Vision restoration after retinal degeneration
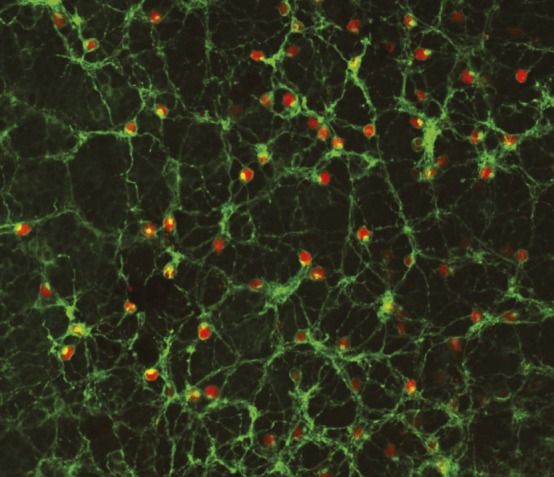
Long-term expression of human melanopsin (green) in residual cells in end-stage retinal degeneration.
The progressive loss of photoreceptor cells in inherited retinal degeneration can cause blindness. However, despite the loss of photoreceptors in end-stage disease, other retinal cells remain structurally intact. Expressing photosensitive proteins such as melanopsin, which is native to the human eye, in such cells through gene therapy could potentially rescue retinal function. Samantha De Silva et al. (pp. 11211–11216) report improved visual function following virus-mediated subretinal injection of the human melanopsin gene in a mouse model of retinal degeneration. Assessments in the mice revealed that human melanopsin was present in the mice retinas for up to 15 months after the injection. Additionally, tests of pupil light reflex and behavioral light avoidance were administered 13 months after the injection. In the pupil light reflex test, treated mice exhibited significantly greater pupil constriction than control mice; treated mice spent less time than control mice in a lighted chamber in the behavioral test. Moreover, treated mice exhibited an enhanced visual response upon recognizing objects in their environment. According to the authors, the findings may have implications for the treatment of retinal degeneration in humans. — C.S.
Assembly of complete HSV-1 genome
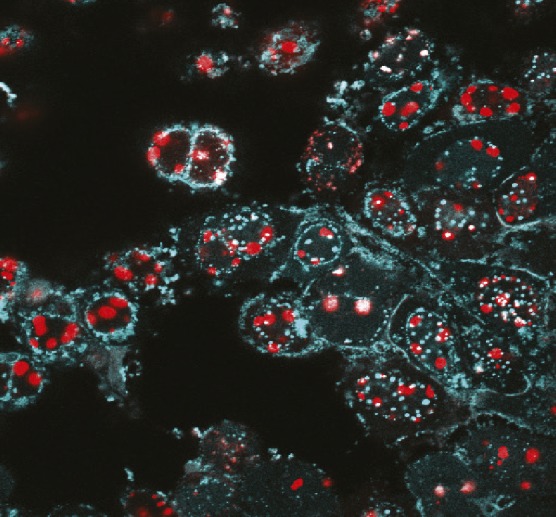
Engineered HSV-1 expressing fluorescent fusion proteins.
Genetically modified herpesviruses offer the promise of customizable therapeutics, including vaccines against a wide range of human pathogens. Although techniques such as bacterial artificial chromosome cloning have made inroads in the development of such therapeutics, they require multiple, time-consuming modifications. Focusing on herpes simplex type 1 (HSV-1), a large DNA virus, Lauren Oldfield et al. (pp. E8885–E8894) present a proof-of-concept approach to rapidly and efficiently introduce specific mutations into herpesvirus genomes. Using yeast transformation-associated recombination, the authors demonstrate that the HSV-1 genome can be cloned in yeast as a series of 11 DNA fragments, which can be reassembled in yeast into a complete genome that exhibits the same infectious properties in mammalian cells as the wild-type strain. In addition, the authors demonstrate how the technique can be applied to genome-wide virus engineering, generating modifications to single and multiple genes simultaneously. Although the findings are preliminary, the study offers a potential approach for viral engineering to rapidly develop vaccines and other therapeutic delivery systems. — T.J.



