Abstract
Glucose tolerance after meal ingestion in vivo is the result of multiple processes that occur in parallel. Insulin secretion together with reciprocal inhibition of glucagon secretion contributes to glucose tolerance. However, other factors beyond glucose effectiveness and insulin action require consideration. The absorption of ingested nutrients and their subsequent systemic rate of appearance largely depend on the rate of delivery of nutrients to the proximal small intestine. This is determined by the integrated response of the upper gastrointestinal tract to a meal. While gastric emptying is probably the most significant component, other factors need to be considered. This review will examine all processes that could potentially alter the fraction and rate of appearance of ingested nutrients in the peripheral circulation. Several of these processes may be potential therapeutic targets for the prevention and treatment of diabetes. Indeed, there is increased interest in gastrointestinal contributions to nutritional homeostasis, as demonstrated by the advent of antidiabetes therapies that alter gastrointestinal motility, the effect of bariatric surgery on diabetes remission, and the potential of the intestinal microbiome as a modulator of human metabolism. The overall goal of this review is to examine current knowledge of the gastrointestinal contributions to metabolic control.
Introduction
Our collective understanding of the physiological response to meal ingestion has centered around the pancreatic islet response to nutrient ingestion. However, while insulin secretion together with reciprocal inhibition of glucagon secretion is important for the maintenance of glucose tolerance, other factors require consideration. These include the ability of glucose and of insulin to stimulate glucose uptake and suppress glucose production (glucose effectiveness and insulin action, respectively) (1). Given that the gastrointestinal tract is the first organ system to make contact with ingested nutrients, it is necessary to consider its role in determining the systemic rate of appearance of ingested nutrients and the direct and indirect contributions of the gastrointestinal tract to postprandial metabolism. The systemic rate of appearance of ingested nutrients is largely determined by the rate of delivery of nutrients to the proximal small intestine through the rate of gastric emptying. While gastric emptying is arguably the most significant of the myriad processes occurring within the gastrointestinal tract, many other factors are either overlooked or misunderstood. This article provides a systematic overview of the mechanisms that can alter the fraction and rate of appearance of ingested nutrients in the peripheral circulation.
The role of the upper gastrointestinal tract in the maintenance of glucose tolerance has been highlighted by the advent of antidiabetes therapies that can either exclusively alter gastrointestinal motility (with secondary effects on satiation and weight) or have a combined effect on gastrointestinal motility and β-cell function (2). In addition, the past decade has witnessed renewed interest in bariatric surgery given its effects on type 2 diabetes, suggesting that the gastrointestinal tract produces diabetogenic and/or diabetogenic mediators whose secretion is respectively inhibited or enhanced by surgery (3). The overall goal of this review is to examine current knowledge and provide an overview of the gastrointestinal contributions to metabolic control.
Meal Composition and Its Effects on Appetite, Glycemia, and Upper Gastrointestinal Function
Dietary fiber is composed predominantly of indigestible carbohydrate polymers, and its consumption is thought to confer benefits such as the prevention of ischemic heart disease, colorectal cancer, and type 2 diabetes. It decreases energy density of ingested foods, likely impairs absorption of some nutrients through physical interactions, and stimulates satiety. In epidemiological studies, dietary fiber intake is inversely correlated with adiposity and BMI (4). These effects may not be unique to dietary fiber; indeed, a low-energy density foam has been shown to have some effect on satiety and food intake that is sustained for a few hours after ingestion. Multiple short-term studies suggest that increased dietary consumption of fiber has short-term effects that are positive in terms of postprandial glycemia and appetite (5). However, not all interventional studies utilizing dietary fiber are associated with beneficial effects on weight, and their efficacy over the long term remains untested.
Effects of fiber have been reviewed in detail elsewhere (6). In summary, insoluble fibers demonstrate the strongest associations with decreased risk of diabetes, whereas soluble dietary fiber exerts physiological effects on the stomach and small intestine that modulate postprandial glycemic responses through delayed gastric emptying, modification of gastrointestinal myoelectrical activity and delaying small bowel transit, reduced glucose diffusion through the unstirred water layer, and reduced accessibility of α-amylase to its substrates due to increased viscosity of gut contents (6). Table 1 shows a summary of dietary fiber effects on gastric emptying, satiety, glucose homeostasis, intestinal hormones, and body weight regulation (6).
Table 1.
Summary of dietary fiber effects on gastric emptying, satiety, glucose homeostasis, intestinal hormones, and body weight regulation
| Dietary fiber type | Gastric emptying | Satiety | Glucose homeostasis | Intestinal hormones | Body weight/regulation of energy balance |
|---|---|---|---|---|---|
| Guar gum | Delayed in most studies; possible threshold at 5 g | Enhanced in most studies; effect is viscosity dependent, abolished by partial hydrolysis of guar, and modulated by meal fat content | Decreased postprandial glucose levels in most studies; GE delay is main factor; delayed absorption contributes | ↓GIP, ↑GLP-1, ↑CCK postprandially | WMD: −0.04 kg (95% CI −2.2, 2.1); gastrointestinal adverse effects limit guar use for weight loss |
| Psyllium | Minor effect | Enhanced in most studies; threshold in the range 5.2–8.5 g | Variable | ↔GLP-1 | BMI reduction of –2.0 ± 0.3 kg/m2 at 6 months; no effect |
| Pectin | Delayed with >10 g | Enhanced possibly through direct gastric effect | Decreased postprandial glucose when >10 g; possible dose-response relationship | ↔CCK, PP ↔CCK, GIP | No effect when supplemented to ad libitum diet; reduced energy intake (alginate-pectin combination) |
| Alginate | Unaffected in healthy normal-weight individuals; delayed in those with stable diabetes | Enhanced only by strong-gelling form independent to GE | Decrease in correlation to GE effect | Not reported | Strong-gelling form: 135 kcal (7%) reduction in mean daily energy intake over 7 weeks; reduced energy intake (alginate-pectin combination) |
| Glucomannan | No effect | Enhanced satiety, combination with psyllium | No effect | ↔GIP | WMD: −0.79 (95 % CI −1.53, −0.05); weight loss 2.5 kg more than with placebo in 8 weeks; 3.8 ± 0.9 kg weight loss more than with hypocaloric diet alone over 5 weeks in healthy overweight individuals |
| CM3 | No effect | No effect | Not reported | Not reported | 3–4 kg weight loss more than with placebo |
| Cellulose | Minor effects (unmodified); delayed (water-soluble) | Enhanced (EHEC) | “Second meal effect” in combination with amylopectin/amylose | ↔PP, CCK (EHEC) | No effect (methylcellulose) on ad libitum diet |
| Wheat fiber | Unaltered in most studies; delayed by undiluted and coarse bran | Enhanced in most studies; inverse correlation with degree of refinement | Variable effects | ↑GIP, ↔GLP-1 | Modest reductions; interpretation of results difficult as wheat grain coadministered with other dietary fiber sources in most studies |
Literature is limited for glucomannan, CM3, and cellulose. EHEC: ethyl-hydroxyethyl-cellulose (“liquid fiber”); GE, gastric emptying; PP, pancreatic polypeptide; WMD, weighted mean difference relative to placebo in meta-analysis. See also Papathanasopoulos and Camilleri (6).
The effect of volume of ingesta on postprandial satiety seems to be greater than that of calories ingested, and indeed a gastric balloon inflated to 400–800 mL rapidly induces satiety without altering gastric emptying. Subsequent studies have suggested that maximal stomach capacity affects the volume of (stomach) contents necessary to decrease spontaneous food consumption by half (7). Indeed, Geliebter et al. (7) suggested a significant difference in maximal stomach capacity between obese and lean subjects. Postprandial gastric volume seems to predict satiation across a wide range of BMI (8).
Gastric Emptying and Nutrient Composition
The nutrient content of the suspension traversing the pylorus influences the rate of emptying to the extent that caloric delivery is nearly constant and ∼200 kcal/h are delivered to the duodenum. This is based on the studies of Hunt et al. (9) who first suggested that the pressure differences between the stomach and the small intestine as well as the volume of ingested meal govern the emptying half-time. The volume of the meal, its energy density (kcal/mL), and the proportions of fat, carbohydrate, and protein in the meal have minor effects on the rate of gastric emptying of energy (10). Regulation is achieved through the osmotic effect (including calorie content) and calcium binding of the products of digestion in the duodenum.
Increased caloric content (increasing sucrose concentrations) delayed emptying irrespective of the volume of test meal ingested (9). Subsequently, the slowing of gastric emptying by disaccharides was shown to be consistent with the stimulation of duodenal osmoreceptors after hydrolysis to monosaccharides (11). A later series of studies suggested similar slowing of gastric emptying by isocaloric amounts of fat, protein, and carbohydrate. Hunt (10) ultimately suggested that the osmotic properties of the stomach contents reaching the duodenum as well as the saponification of partially hydrolyzed triglycerides determine the rate of gastric emptying. A similar pattern for small bowel motor activity (i.e., it is dependent on the caloric value of the liquid meal ingested) has also been observed (12). The presence of fat such as oleate in the duodenum stimulates cholecystokinin (CCK) secretion, which in turn inhibits antral motility, stimulates pyloric tone, and therefore delays gastric emptying (13). However, there is some adaptation to diet so that a high-fat diet may not always delay gastric emptying in response to a test meal.
In addition to the trituration of solid food, the stomach facilitates nutritional absorption through denaturation by its acidic milieu as well as through secretion of gastric lipase and pepsins. Pepsins are secreted by the gastric mucosa and are typically activated by the acidic environment within the stomach. Gastric lipase is secreted by the chief cells in the gastric fundus in response to stimuli such as gastrin and acetylcholine that are elicited by food intake. Gastric lipase initiates digestion of lipids and triglycerides—free fatty acids liberated by its actions in the duodenum stimulate CCK secretion, which, together with glucagon-like peptide 1 (GLP-1), inhibits lipase secretion. In contrast, carbohydrate digestion commences with salivary amylase (which is inactivated by a pH <4—conditions typically encountered within the stomach). Further digestion occurs at the surface of the intestinal mucosa and in the presence of pancreatic amylase in the duodenum and proximal small intestine (14).
It is controversial whether macronutrient composition independently affects upper gastrointestinal function and appetite. The classic studies by Hunt et al. (9,10) suggested only minor effects, and Park et al. (15) reported that ingestion for 2 weeks of four different classes of macronutrients (protein, carbohydrate, fat, or a mixture) in excess of required calories by 500 kcal did not significantly change gastric function or aggregate gastric symptoms after ingestion of a challenge meal. The maximum tolerated volume of Ensure was higher only in a subset of participants with high baseline maximum tolerated volume who were randomized to fat supplementation. Satiety (calories ingested) or food choices at an ad libitum buffet meal did not differ between groups (15).
A caloric “preload” prior to the ingestion of the “main” meal can alter postprandial glycemic excursion through modulation of upper gastrointestinal function. Fat added to carbohydrate-containing meals stimulates incretin hormones and delays gastric emptying (16). Similar results have been observed with a whey protein preload (17).
Gastric Accommodation and Vagal Function
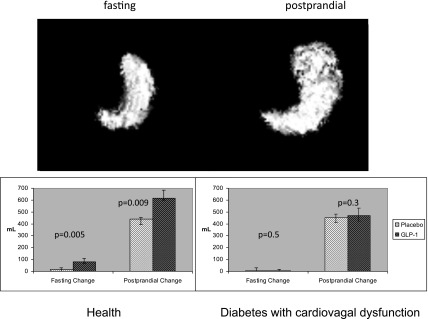
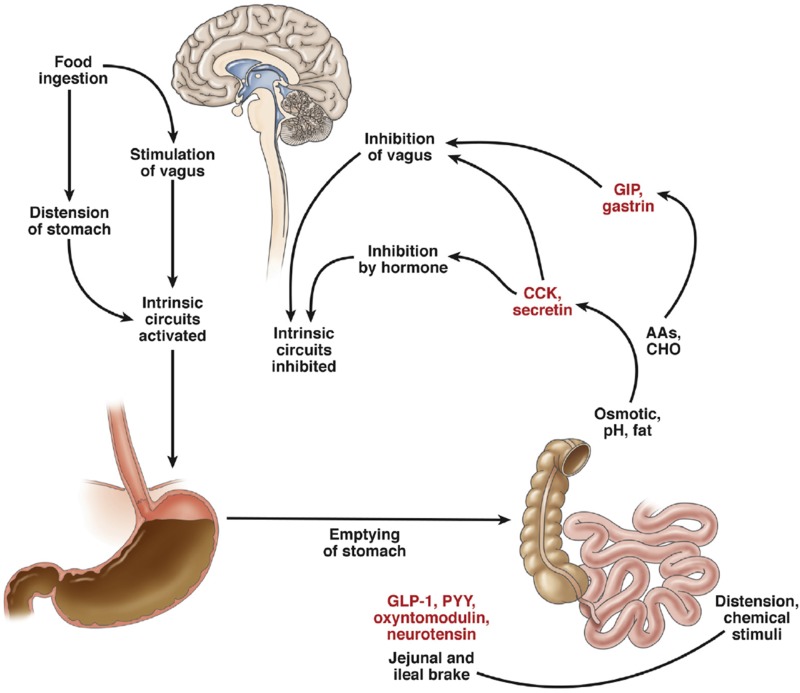
Meal ingestion stimulates gastric accommodation—the active process whereby gastric volume (proximal > distal) increases to accommodate the ingested meal without an increase in wall tension or intragastric pressure. This permits food ingestion without accompanying discomfort that would limit the ingestion of nutrients. Although gastric accommodation is thought to be mediated through the vagal nerve and denervation is associated with early satiety and decreased caloric intake, there is increasing evidence that the gastric relaxation responses recover with the passage of time after vagal denervation (18). Of note, pharmacological concentrations of the enteroendocrine hormone GLP-1 increase gastric volume (Fig. 1) (and delay solid emptying) but require an intact vagus, as this effect is not observed in people with cardiovagal neuropathy (who have otherwise normal gastric accommodation) (19). Inhibition of endogenous GLP-1 by exendin(9,39) (a competitive antagonist of GLP-1 at its cognate receptor) also produces a decrease in gastric compliance as measured by an intragastric device (20). These data suggest that, at least in the presence of an intact vagus nerve, endogenous GLP-1 contributes to gastric compliance (Fig. 2).
Figure 1.
GLP-1 increases gastric fasting and postprandial volume. However, in the presence of cardiovagal dysfunction, GLP-1 does not alter fasting or postprandial gastric volume. Reprinted with permission from Delgado-Aros et al. Effects of glucagon-like peptide-1 and feeding on gastric volumes in diabetes mellitus with cardio-vagal dysfunction. Neurogastroenterol Motil 2003;15:435–443.
Figure 2.
Interaction of nutrient intake and neurohormonal responses. AAs, amino acids; CHO, carbohydrates. Reprinted with permission from Camilleri. Peripheral mechanisms in appetite regulation. Gastroenterology 2015;148:1219–1233.
Gastric Emptying of Solids and Liquids, Satiation, Satiety, and Vagal Function
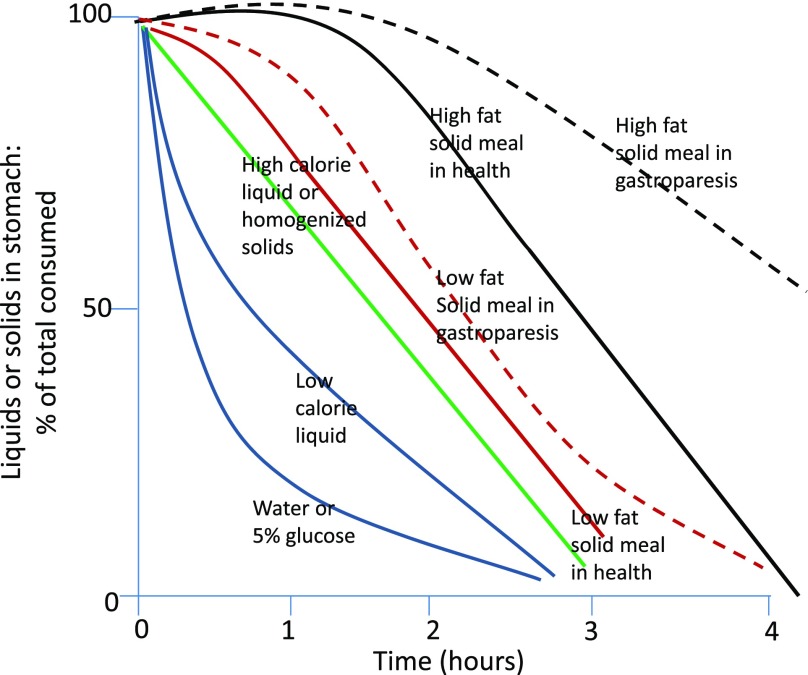
Unlike solid foods, ingested liquids tend to distribute passively and uniformly throughout the stomach. In contrast, solids are retained within the proximal stomach. The rate of liquid emptying differs significantly from that of solids and is much more rapid—the emptying half-time (time taken to empty 50%) being of the order of 20 min as opposed to ∼120 min (14) (Fig. 3). Nonnutrient liquids empty exponentially but as caloric content increases the emptying rate decreases and becomes more linear. This prompts three observations; it is important to appreciate that the nature of a test meal might affect the rate at which calories appear in the duodenum. In addition, the result of scintigraphic measurement of gastric emptying depends to a large extent on whether it is bound to the liquid or solid component (if any) of a test meal. For example, the use of a labeled egg meal measures the rate of emptying of a different phase than that measured by the systemic appearance of ingested acetaminophen solution. The final observation to make is that liquid emptying is not completely passive and the emptying rate is decreased by increasing caloric content, suggesting an active ability to regulate caloric delivery that is not entirely dependent on particle size and the ability to pass through the pylorus (10).
Figure 3.
Patterns of gastric emptying of liquids and solids in health and in gastroparesis (reprinted with permission from Camilleri and Shin [59]).
Solids food particles are subject to trituration by the circular contractions of the gastric antrum that propel food toward the closed pylorus. These forces together with acidic and peptic digestion (which commences in the stomach) breaks food into particles of sufficiently small size (∼2 mm) that they can traverse the pylorus. The corollary of this is that solid emptying is preceded by a lag phase where no emptying occurs followed by linear, postlag emptying. Solids typically empty over a period of 3–4 h (14). However, the volume, consistency and fat content will affect emptying rate so that large, fatty meals may empty over periods longer than 4 h. Gastric emptying rather than gastric accommodation appears to be the major gastric function determining postprandial satiation and satiety.
Bilateral truncal vagotomy, previously used to treat peptic ulcer, results in delayed gastric emptying, early satiety and weight loss (18). This is believed to be, at least in part, due to a decrease in gastric accommodation although symptoms decrease in severity over time. This may be due to the formation of collateral innervation or adaptation of the intrinsic enteric nervous system over time. While studies in animals suggest that vagal innervation modulates insulin secretion, insulin action and hepatic glucose metabolism (21), it is uncertain if these effects make significant contributions to the regulation of glucose metabolism in humans (22). The use of chronic, but reversible, electrical vagal blockade in humans produced a significant, but temporary, decrease in weight and caloric intake (23). No direct effects on glucose metabolism have been demonstrated with electrical vagal blockade (22) although in 26 subjects with type 2 diabetes electrical vagal blockade achieved sustained weight loss over a 1-year period, together with improved HbA1c, fasting glucose and blood pressure (24). These effects are likely to have resulted from the effect of vagal blockade on weight loss.
Gastric Emptying and Interaction With Glycemia
The rate of gastric emptying is subject to modulation by several circulating factors—blood glucose concentrations alter gastric emptying; hyperglycemia delays gastric emptying while hypoglycemia accelerates gastric emptying. However, the effect of raising blood glucose concentrations within the typical range observed in most people with diabetes has relatively minor effects on gastric emptying. For example, raising blood glucose from 4 to 8 mmol/L delayed emptying half-time of a liquid meal by about 10 min (25) or about 12 min in patients with type 1 diabetes (26). There was also 9% greater retention of solids at 100 min in the patients with type 1 diabetes between blood glucose 4 and 8 mmol/L (26). These studies utilized a hyperglycemic clamp with sustained glucose concentrations somewhat greater than those encountered in typical type 2 diabetes. Therefore the contribution of hyperglycemia per se to changes in gastric emptying is likely to be small.
Enteric Hormone Secretion and Its Effects on Upper Gastrointestinal Function
Gastrin and CCK
Gastrin is secreted by the G cells in the gastric antrum and by the parietal cells of the fundus and body. It is responsible for a significant proportion of postprandial acid release through direct activation of CCK2 receptors on parietal cells and through release of histamine from enterochromaffin-like cells (27). CCK is released from the duodenal mucosa in response to nutrients, particularly fatty acids of at least 12 carbon chain length. It directly activates vagal afferent fibers leading to relaxation of the proximal stomach (increasing its capacitance) and inhibition of gastric emptying (28). CCK also stimulates gallbladder contraction, and exocrine pancreatic secretion. Central and peripheral CCK receptors mediate satiety and fullness after meal ingestion. Of note, although CCK and gastrin share structural similarities as well as an affinity for CCK2 receptors, CCK induces somatostatin secretion which in turn inhibits gastrin secretion, gastrointestinal motility and gastric acid secretion (29).
Gastric Inhibitory Polypeptide/Glucose-Dependent Insulinotropic Polypeptide
Gastric inhibitory polypeptide (GIP), also called glucose-dependent insulinotropic polypeptide, is a peptide hormone secreted by K cells in the duodenum and proximal jejunum. It signals through a specific receptor (GIPR). In the absence of a competitive antagonist of GIP, most of its effects on physiology are inferred from transgenic mice lacking the receptor (30). GIP levels rise immediately after nutrient ingestion, inhibiting gastric acid secretion and emptying. In humans these effects are observed at pharmacological (supraphysiological) concentrations of GIP, and thus the contribution of GIP to gastric acid secretion and gastric emptying in humans is uncertain. GIP also stimulates insulin secretion in the setting of hyperglycemia; however, the secretory response to infused GIP is impaired in diabetes.
GLP-1
GLP-1 is secreted from L cells and is arguably the most important incretin hormone. It arises from posttranslational processing of proglucagon (which also give rise to GLP-2, a trophic factor for intestinal mucosa) and stimulates insulin secretion while inhibiting glucagon secretion in a glucose-dependent fashion. GLP-1–based therapy has been used successfully for the treatment of type 2 diabetes and obesity (2). Its effects on gastrointestinal function have also been alluded to earlier in this review. The physiological contributions of endogenous GLP-1 have been mainly studied using the GLP-1 receptor antagonist exendin(9,39) in humans and through studies of Glp1r−/− mice.
Ghrelin
Ghrelin functions as an orexigenic hormone in humans—rising concentrations during fasting decrease after food ingestion and correlate with increasing appetite. Sleeve gastrectomy (SG) decreases acyl-ghrelin concentrations, presumably due to excision of a large part of the ghrelin-secreting stomach, which should decrease appetite. Indeed, fasting after SG is not associated with a rise in the (low) ghrelin concentrations observed in these patients, in contrast to what occurs after Roux-en-Y gastric bypass (RYGB) (31). It is unclear whether restoration of fasting presurgical acyl-ghrelin concentrations will alter GI function and restore appetite after SG.
Beyond its direct central effects on appetite regulation, ghrelin can also accelerate gastric emptying of liquids and solids (32). However, in a dose used to stimulate physiological growth hormone secretion, synthetic human ghrelin does not seem to alter gastric motor functions (emptying or postprandial accommodation), suggesting that at physiological concentrations it does not contribute to gastric function in humans (33).
Amylin
Amylin is a peptide hormone cosecreted with insulin by the β-cell. Consequently, amylin is deficient in type 1 diabetes, while plasma levels are increased in obesity, impaired glucose tolerance, and type 2 diabetes. A synthetic analog, pramlintide, delays gastric emptying through vagal signaling in a dose-dependent fashion (34). Pramlintide is approved for the treatment of postprandial hyperglycemia in patients using intensive insulin therapy. Again, despite its pharmacological effects, the physiological contribution of amylin to the regulation of glucose metabolism is uncertain.
Bile Acids and Their Contribution to the Regulation of Metabolism
Bile acids are synthesized from cholesterol within the hepatocytes and excreted in the bile. They are subsequently reabsorbed in the distal small intestine so that 5% are lost in the feces. Conjugation with glycine or taurine makes bile acids impermeable to cell membranes and confers detergent-like properties that help solubilize lipid micelles and facilitate the absorption of fat in the small intestine (35). Bile acids are stored in the gallbladder during indigestive periods, but activation of CCK by fat emptying into duodenum stimulates gallbladder contraction and relaxation of the sphincter of Oddi, delivering bile to the small bowel. Bile acids act as natural ligands for the transcription factor farnesoid X receptor (FXR). Activation of this nuclear receptor stimulates the expression of genes encoding proteins involved in bile acid synthesis, transport, and metabolism. Activation by ligand binding causes heterodimerization with the retinoic X receptor α and subsequent binding to the promoter regions of target DNA (36). Bile acids differ in their ability to activate FXR, suggesting that changes in bile acid composition may alter the metabolic effects of FXR activation (37).
In addition, bile acids can also influence metabolism through the membrane-bound, G-protein–coupled bile acid receptor 1 (also known as TGR5). TGR5 is expressed in adipose tissue, the enteric nervous system, and the enteroendocrine L cells that produce GLP-1. Activation of the receptor leads to activation of protein kinase A and phosphorylation of target proteins. Increased GLP-1 secretion has been attributed to increased bile acid delivery to the small intestine after bariatric surgery (35).
FGF19 production is almost exclusively restricted to the terminal ileum, which corresponds to the site where bile acids are actively taken up by the ileal sodium/bile acid cotransporter (38). Its expression is increased by FXR signaling, and it is secreted into the portal circulation where it suppresses the rate-controlling enzyme of bile acid synthesis. It also stimulates hepatic glucose uptake and glycogen synthesis in an insulin-independent manner (39).
Changes in bile acid composition (and in the enterohepatic circulation) may contribute to the metabolic benefits of bariatric surgery. Certainly, differences among the various bariatric procedures might shed some light on the contribution of bile acids to metabolic regulation in humans. Plasma bile acid concentrations are higher after RYGB compared with those of weight-matched control subjects. However, these changes are not immediately apparent after surgery (at the time when most of the metabolic changes are occurring) and are only evident ∼1 year after the procedure (40).
In contrast, bile acid sequestration using bile acid binding resins decreases bile acid reabsorption by the terminal ileum, leading to increased fecal loss of bile acids. This is accompanied by decreased FGF19 concentrations and increased bile acid synthesis (41). However, in humans the decrease in FGF19 is accompanied by decreased fasting glucose concentrations and decreased meal ce of increased splanchnic extraction of the meal (42). Changes in GLP-1 concentrations have been variable (increase or no change) (41,42).
Bariatric Surgery and Mechanistic Insights Into Upper Gastrointestinal Function
Bariatric surgery is the most effective intervention for weight loss. Nonrandomized retrospective studies suggest that the remission rate is associated with length of bypassed intestine (∼85% for standard RYGB and ∼98% with duodenal switch [43]). However, prospective, randomized controlled trials have reported lower remission rates for diabetes with RYGB (44). Of note, caloric restriction acutely improves β-cell function, suggesting that altered caloric intake in addition to altered intestinal anatomy and/or function is a key contributor to improved glucose tolerance (45).
On the other hand, remission of diabetes after bariatric surgery seems to depend on the duration and severity of diabetes (as quantified by the number of oral medications and/or use of insulin) prior to the procedure (46). This would suggest that the effects of bariatric surgery on the ability to synthesize and secrete insulin are limited. Indeed, insulin secretion in response to hyperglycemia with GLP-1 or GIP—a test of supramaximal β-cell function—did not change after surgical invention (47). This effect is less apparent after oral challenges where quantification of islet function as a disposition index demonstrates improvements in β-cell function that are mainly due to improved insulin action (48). Acute caloric restriction improves insulin action prior to any discernable weight loss (45), and indeed direct comparison of caloric restriction to the early changes after bariatric surgery might suggest equivalence.
Procedures that result in more rapid appearance of calories in the proximal intestine result in increased GLP-1 secretion. The extent to which this contributes to the remission of diabetes has been debated with some divergence in the literature. Acute blockade of the GLP-1 receptor decreases β-cell function after RYGB (49). However, in subjects with intact glucose effectiveness, the effect on glucose tolerance is marginal. GLP-1 also delays gastrointestinal motility after RYGB (49).
GLP-1 also has direct effects on hypothalamic nuclei outside of the blood-brain barrier, and GLP-1 or GLP-1 receptor agonists decrease food intake and cause weight loss (2). More recently, activation of the GLP-1 receptor decreased food intake and food-related brain responses in patients with type 2 diabetes and in obese subjects as measured by functional MRI—an action blocked by exendin(9,39) (50). However, the effect of postprandial GLP-1 concentrations on satiety after RYGB or SG is at present uncertain (51).
There is some evidence that intestinal absorption of nutrients is more rapid and efficient in obese individuals than it is in lean humans, predisposing them to further weight gain (52). Active glucose transport measured using 3-O-methylglucose (3OMG)—which is taken up by SGLT-1 and GLUT2 but is not metabolized, being excreted unchanged in the urine—is increased in subjects post-RYGB compared with lean subjects. In addition, intestinal glucose absorption is increased in obese subjects (53). While intriguing, the methods used in those studies were qualitative because they measured differences in area under the curve of plasma 3OMG concentrations (reflecting the sum of intestinal absorption and renal clearance) after a 30 min bolus of 3OMG. Moreover, they provided no information as to how glucose absorption changes after RYGB.
The Intestinal Barrier and Nutrient Absorption
The gastrointestinal epithelium provides a selective barrier limiting permeation of toxins while allowing passage of nutrients and water. Disruption of the barrier may play a role in the pathogenesis of multiple gastrointestinal tract disorders. Selectivity is achieved by the tight junctions that respond to extracellular stimuli and alter paracellular permeability through changes in the multiple proteins that comprise the complex (54). Factors that influence permeability include fatty acids in the intestinal lumen (whether ingested directly or as products of bacterial fermentation). In addition, bile acids such as deoxycholic acid and chenodeoxycholic acid can increase paracellular permeability (55). Intriguingly, bile acids alter both permeability and GLP-1 secretion (56) by signaling through the same receptor—TGR5. The intestinal barrier has been assessed in vivo using oral administration of inert, water-soluble probe molecules such as sugars and radiolabeled EDTA that passively traverse the intestinal mucosa into the bloodstream and are recovered unchanged in the urine. Such techniques suggest that intestinal permeability is increased in diabetes, contributing to postprandial hyperglycemia and systemic inflammation (57) through mediators such as lipopolysaccharide (58).
Conclusions
The upper gastrointestinal tract is not a passive conduit of nutrients that subsequently traverse the gut wall and stimulate an endocrine response. Instead, it serves to integrate intraluminal nutrients and neural, mechanical, and hormonal mechanisms to modulate the response to caloric ingestion. We hope that this brief overview helps crystallize current knowledge and suggest future areas of research that might lead to novel therapies for the prevention and treatment of obesity and type 2 diabetes in humans.
Article Information
Acknowledgments. The authors thank Monica M. Davis from the Endocrine Research Unit, Mayo Clinic, Rochester, MN, for secretarial assistance.
Funding. A.V. and M.C. are supported by the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases (DK78646 and DK92179, respectively).
Duality of Interest. A.V. has received research grants from Novo Nordisk and has served as an advisory board member for Sanofi. No other potential conflicts of interest relevant to this article were reported.
References
- 1.Vella A, Shah P, Basu R, Basu A, Holst JJ, Rizza RA. Effect of glucagon-like peptide 1(7-36) amide on glucose effectiveness and insulin action in people with type 2 diabetes. Diabetes 2000;49:611–617 [DOI] [PubMed] [Google Scholar]
- 2.Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006;368:1696–1705 [DOI] [PubMed] [Google Scholar]
- 3.Rubino F, Forgione A, Cummings DE, et al. The mechanism of diabetes control after gastrointestinal bypass surgery reveals a role of the proximal small intestine in the pathophysiology of type 2 diabetes. Ann Surg 2006;244:741–749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McKeown NM, Meigs JB, Liu S, Wilson PW, Jacques PF. Whole-grain intake is favorably associated with metabolic risk factors for type 2 diabetes and cardiovascular disease in the Framingham Offspring Study. Am J Clin Nutr 2002;76:390–398 [DOI] [PubMed] [Google Scholar]
- 5.Silva FM, Kramer CK, Crispim D, Azevedo MJ. A high-glycemic index, low-fiber breakfast affects the postprandial plasma glucose, insulin, and ghrelin responses of patients with type 2 diabetes in a randomized clinical trial. J Nutr 2015;145:736–741 [DOI] [PubMed] [Google Scholar]
- 6.Papathanasopoulos A, Camilleri M. Dietary fiber supplements: effects in obesity and metabolic syndrome and relationship to gastrointestinal functions. Gastroenterology 2010;138:65–72.e1–e2 [DOI] [PMC free article] [PubMed]
- 7.Geliebter A. Gastric distension and gastric capacity in relation to food intake in humans. Physiol Behav 1988;44:665–668 [DOI] [PubMed] [Google Scholar]
- 8.Vazquez Roque MI, Camilleri M, Stephens DA, et al. Gastric sensorimotor functions and hormone profile in normal weight, overweight, and obese people. Gastroenterology 2006;131:1717–1724 [DOI] [PubMed] [Google Scholar]
- 9.Hunt JN, MacDonald I. The influence of volume on gastric emptying. J Physiol 1954;126:459–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hunt JN. Mechanisms and disorders of gastric emptying. Annu Rev Med 1983;34:219–229 [DOI] [PubMed] [Google Scholar]
- 11.Elias E, Gibson GJ, Greenwood LF, Hunt JN, Tripp JH. The slowing of gastric emptying by monosaccharides and disaccharides in test meals. J Physiol 1968;194:317–326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.von Schönfeld J, Evans DF, Renzing K, Castillo FD, Wingate DL. Human small bowel motor activity in response to liquid meals of different caloric value and different chemical composition. Dig Dis Sci 1998;43:265–269 [DOI] [PubMed] [Google Scholar]
- 13.Heddle R, Dent J, Read NW, et al. Antropyloroduodenal motor responses to intraduodenal lipid infusion in healthy volunteers. Am J Physiol 1988;254:G671–G679 [DOI] [PubMed] [Google Scholar]
- 14.Camilleri M. Integrated upper gastrointestinal response to food intake. Gastroenterology 2006;131:640–658 [DOI] [PubMed] [Google Scholar]
- 15.Park MI, Camilleri M, O’Connor H, et al. Effect of different macronutrients in excess on gastric sensory and motor functions and appetite in normal-weight, overweight, and obese humans. Am J Clin Nutr 2007;85:411–418 [DOI] [PubMed] [Google Scholar]
- 16.Feinle C, O’Donovan D, Doran S, et al. Effects of fat digestion on appetite, APD motility, and gut hormones in response to duodenal fat infusion in humans. Am J Physiol Gastrointest Liver Physiol 2003;284:G798–G807 [DOI] [PubMed] [Google Scholar]
- 17.Ma J, Stevens JE, Cukier K, et al. Effects of a protein preload on gastric emptying, glycemia, and gut hormones after a carbohydrate meal in diet-controlled type 2 diabetes. Diabetes Care 2009;32:1600–1602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smith DK, Sarfeh J, Howard L. Truncal vagotomy in hypothalamic obesity. Lancet 1983;1:1330–1331 [DOI] [PubMed] [Google Scholar]
- 19.Delgado-Aros S, Kim DY, Burton DD, et al. Effect of GLP-1 on gastric volume, emptying, maximum volume ingested, and postprandial symptoms in humans. Am J Physiol Gastrointest Liver Physiol 2002;282:G424–G431 [DOI] [PubMed] [Google Scholar]
- 20.Schirra J, Nicolaus M, Woerle HJ, Struckmeier C, Katschinski M, Goke B. GLP-1 regulates gastroduodenal motility involving cholinergic pathways. Neurogastroenterol Motil 2009;21:609–618, e21–e22 [DOI] [PubMed] [Google Scholar]
- 21.Nishi S, Seino Y, Ishida H, et al. Vagal regulation of insulin, glucagon, and somatostatin secretion in vitro in the rat. J Clin Invest 1987;79:1191–1196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sathananthan M, Ikramuddin S, Swain JM, et al. The effect of vagal nerve blockade using electrical impulses on glucose metabolism in nondiabetic subjects. Diabetes Metab Syndr Obes 2014;7:305–312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sarr MG, Billington CJ, Brancatisano R, et al.; EMPOWER Study Group . The EMPOWER study: randomized, prospective, double-blind, multicenter trial of vagal blockade to induce weight loss in morbid obesity. Obes Surg 2012;22:1771–1782 [DOI] [PubMed] [Google Scholar]
- 24.Shikora S, Toouli J, Herrera MF, et al. Vagal blocking improves glycemic control and elevated blood pressure in obese subjects with type 2 diabetes mellitus. J Obes 2013;2013:245683 [DOI] [PMC free article] [PubMed]
- 25.Hebbard GS, Samsom M, Sun WM, Dent J, Horowitz M. Hyperglycemia affects proximal gastric motor and sensory function during small intestinal triglyceride infusion. Am J Physiol 1996;271:G814–G819 [DOI] [PubMed] [Google Scholar]
- 26.Schvarcz E, Palmér M, Aman J, Horowitz M, Stridsberg M, Berne C. Physiological hyperglycemia slows gastric emptying in normal subjects and patients with insulin-dependent diabetes mellitus. Gastroenterology 1997;113:60–66 [DOI] [PubMed] [Google Scholar]
- 27.Schmidt WE, Schmitz F. Genetic dissection of the secretory machinery in the stomach. Gastroenterology 2004;126:606–609 [DOI] [PubMed] [Google Scholar]
- 28.Lal S, McLaughlin J, Barlow J, et al. Cholecystokinin pathways modulate sensations induced by gastric distension in humans. Am J Physiol Gastrointest Liver Physiol 2004;287:G72–G79 [DOI] [PubMed] [Google Scholar]
- 29.Beglinger C, Degen L. Fat in the intestine as a regulator of appetite--role of CCK. Physiol Behav 2004;83:617–621 [DOI] [PubMed] [Google Scholar]
- 30.Hansotia T, Maida A, Flock G, et al. Extrapancreatic incretin receptors modulate glucose homeostasis, body weight, and energy expenditure. J Clin Invest 2007;117:143–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee WJ, Chen CY, Chong K, Lee YC, Chen SC, Lee SD. Changes in postprandial gut hormones after metabolic surgery: a comparison of gastric bypass and sleeve gastrectomy. Surg Obes Relat Dis 2011;7:683–690 [DOI] [PubMed]
- 32.Murray CD, Martin NM, Patterson M, et al. Ghrelin enhances gastric emptying in diabetic gastroparesis: a double blind, placebo controlled, crossover study. Gut 2005;54:1693–1698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cremonini F, Camilleri M, Vazquez Roque M, et al. Obesity does not increase effects of synthetic ghrelin on human gastric motor functions. Gastroenterology 2006;131:1431–1439 [DOI] [PubMed] [Google Scholar]
- 34.Samsom M, Szarka LA, Camilleri M, Vella A, Zinsmeister AR, Rizza RA. Pramlintide, an amylin analog, selectively delays gastric emptying: potential role of vagal inhibition. Am J Physiol Gastrointest Liver Physiol 2000;278:G946–G951 [DOI] [PubMed] [Google Scholar]
- 35.Kuipers F, Bloks VW, Groen AK Beyond intestinal soap–bile acids in metabolic control. Nat Rev Endocrinol 2014;10:488-498 [DOI] [PubMed] [Google Scholar]
- 36.Staels B, Fonseca VA. Bile acids and metabolic regulation: mechanisms and clinical responses to bile acid sequestration. Diabetes Care 2009;32(Suppl. 2):S237–S245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Albaugh VL, Flynn CR, Cai S, Xiao Y, Tamboli RA, Abumrad NN. Early increases in bile acids post Roux-en-Y gastric bypass are driven by insulin-sensitizing, secondary bile acids. J Clin Endocrinol Metab 2015;100:E1225–E1233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Inagaki T, Choi M, Moschetta A, et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab 2005;2:217–225 [DOI] [PubMed] [Google Scholar]
- 39.Dong X, Park S, Lin X, Copps K, Yi X, White MF. Irs1 and Irs2 signaling is essential for hepatic glucose homeostasis and systemic growth. J Clin Invest 2006;116:101–114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Patti ME, Houten SM, Bianco AC, et al. Serum bile acids are higher in humans with prior gastric bypass: potential contribution to improved glucose and lipid metabolism. Obesity (Silver Spring) 2009;17:1671–1677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Beysen C, Murphy EJ, Deines K, et al. Effect of bile acid sequestrants on glucose metabolism, hepatic de novo lipogenesis, and cholesterol and bile acid kinetics in type 2 diabetes: a randomised controlled study. Diabetologia 2012;55:432–442 [DOI] [PubMed] [Google Scholar]
- 42.Smushkin G, Sathananthan M, Piccinini F, et al. The effect of a bile acid sequestrant on glucose metabolism in subjects with type 2 diabetes. Diabetes 2013;62:1094–1101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA 2004;292:1724–1737 [DOI] [PubMed] [Google Scholar]
- 44.Ikramuddin S, Korner J, Lee WJ, et al. Roux-en-Y gastric bypass vs intensive medical management for the control of type 2 diabetes, hypertension, and hyperlipidemia: the Diabetes Surgery Study randomized clinical trial. JAMA 2013;309:2240–2249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kelley DE, Wing R, Buonocore C, Sturis J, Polonsky K, Fitzsimmons M. Relative effects of calorie restriction and weight loss in noninsulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1993;77:1287–1293 [DOI] [PubMed] [Google Scholar]
- 46.Still CD, Wood GC, Benotti P, et al. Preoperative prediction of type 2 diabetes remission after Roux-en-Y gastric bypass surgery: a retrospective cohort study. Lancet Diabetes Endocrinol 2014;2:38–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dirksen C, Bojsen-Møller KN, Jørgensen NB, et al. Exaggerated release and preserved insulinotropic action of glucagon-like peptide-1 underlie insulin hypersecretion in glucose-tolerant individuals after Roux-en-Y gastric bypass. Diabetologia 2013;56:2679–2687 [DOI] [PubMed] [Google Scholar]
- 48.Nguyen KT, Billington CJ, Vella A, et al. preserved insulin secretory capacity and weight loss are the predominant predictors of glycemic control in patients with type 2 diabetes randomized to Roux-en-Y gastric bypass. Diabetes 2015;64:3104–3110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shah M, Law JH, Micheletto F, et al. Contribution of endogenous glucagon-like peptide 1 to glucose metabolism after Roux-en-Y gastric bypass. Diabetes 2014;63:483–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van Bloemendaal L, IJzerman RG, ten Kulve JS., et al. GLP-1 receptor activation modulates appetite- and reward-related brain areas in humans. Diabetes 2014;63:4186–4196 [DOI] [PubMed] [Google Scholar]
- 51.Lee W-J, Chong K, Ser K-H, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg 2011;146:143–148 [DOI] [PubMed] [Google Scholar]
- 52.Seimon RV, Brennan IM, Russo A, et al. Gastric emptying, mouth-to-cecum transit, and glycemic, insulin, incretin, and energy intake responses to a mixed-nutrient liquid in lean, overweight, and obese males. Am J Physiol Endocrinol Metab 2013;304:E294–E300 [DOI] [PubMed] [Google Scholar]
- 53.Nguyen NQ, Debreceni TL, Bambrick JE, et al. Upregulation of intestinal glucose transporters after Roux-en-Y gastric bypass to prevent carbohydrate malabsorption. Obesity (Silver Spring) 2014;22:2164–2171 [DOI] [PubMed] [Google Scholar]
- 54.Suzuki T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol Life Sci 2013;70:631–659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Camilleri M. Peripheral mechanisms in irritable bowel syndrome. N Engl J Med 2012;367:1626–1635 [DOI] [PubMed] [Google Scholar]
- 56.Katsuma S, Hirasawa A, Tsujimoto G. Bile acids promote glucagon-like peptide-1 secretion through TGR5 in a murine enteroendocrine cell line STC-1. Biochem Biophys Res Commun 2005;329:386–390 [DOI] [PubMed] [Google Scholar]
- 57.Damci T, Nuhoglu I, Devranoglu G, Osar Z, Demir M, Ilkova H. Increased intestinal permeability as a cause of fluctuating postprandial blood glucose levels in type 1 diabetic patients. Eur J Clin Invest 2003;33:397–401 [DOI] [PubMed] [Google Scholar]
- 58.Koeth RA, Wang Z, Levison BS, et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 2013;19:576–585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Camilleri M, Shin A. Novel and validated approaches for gastric emptying scintigraphy in patients with suspected gastroparesis. Dig Dis Sci 2013;58:1813–1815 [DOI] [PubMed] [Google Scholar]