Abstract
Purpose
This study was conducted to investigate the neurocognitive functioning of children with intracranial germ cell tumor (IGCT) prior to receiving proton beam therapy (PBT), and to identify differential characteristics of their neurocognitive functioning depending on tumor location. As a secondary object of this study, neurocognitive functions were followed up at 1-2 years after PBT to examine early post-treatment changes.
Materials and Methods
Between 2008 and 2014, 34 childrenwith IGCT treatedwho received PBT atNational Cancer Center, Korea were enrolled in this study. Standardized neurocognitive tests of intelligence, memory, and executive functioning were performed with baseline psychological assessments using the Child Behavior Checklist (CBCL). Follow-up assessments after PBT were conducted in 20 patients (T2). The results were analyzed based on the locations of tumors, which included the suprasellar, pineal gland, basal ganglia, and bifocal regions.
Results
The neurocognitive function of IGCT patients was significantly lower than that of the normal population in performance intelligence quotient (p=0.041), processing speed (p=0.007), memory (p < 0.001), and executive functioning (p=0.010). Patients with basal ganglia tumors had significantly lower scores for most domains of neurocognitive functioning and higher scores for CBCL than both the normal population and patients with IGCT in other locations. There was no significant change in neurocognitive function between T1 and T2 for all types of IGCT patients in first 1-2 years after PBT.
Conclusion
Tumor location significantly affects the neuropsychological functioning in patients with IGCT. Neuropsychological functioning should be closely monitored from the time of diagnosis in IGCT patients.
Keywords: Brain neoplasms, Germ cell tumor, Cognitive function
Introduction
Intracranial germ cell tumors (IGCT), which are the most common types of brain tumors in Asian children and adolescents [1,2], are typically found in the suprasellar area and pineal gland, and less commonly in the basal ganglia. Pure germinomas are very sensitive to radiotherapy and chemotherapy, and show excellent prognosis with 5-year overall survival rates of ≥ 90% following radiotherapy with or without chemotherapy [3-5]. Non-germinomatous germ cell tumors (NGGCT) have less favorable prognosis than pure germinoma; however, the 5-year survival rate has reached 60%-80%, even for NGGCT after aggressive multimodal therapy was adopted [6-8]. The improvement in survival rates of patients with IGCTs has raised questions regarding the impact of these tumors on neuropsychological functioning and the quality of life of long-term survivors.
Radiotherapy has generally been thought to contribute to poor neurocognitive functions and quality of life of IGCT patients [9,10]. However, most studies of the neurocognitive functioning of IGCT patients have analyzed the long-term effects of radiotherapy by measuring neurocognitive functions at a specific time after treatment without performing pre-treatment assessments [11-13]. Hence, it is difficult to conclude that radiation therapy is solely responsible for the neurocognitive decline in these patients.
Merchant et al. [4] evaluated the long-term changes in neurocognitive functioning relative to a baseline assessment and found that there were no significant differences in the intelligence quotient (IQ) of the patients before and after radiotherapy. However, they reported that the IQ of the patients was consistently in low to average range from the time of diagnosis to the follow-up assessment. In contrast, several studies have revealed that the pre-treatment neurocognitive functioning of children with brain tumors was not markedly different from that of an age-matched normal population [14,15]. These findings, together with those of a study by Merchant et al. [4] have led the authors to hypothesize that the changes in neurocognitive functioning before starting radiotherapy may be specific to IGCT. Therefore, we investigated whether tumor location causes differences in neuropsychological functioning in patients with IGCT patients treated with proton beam therapy (PBT).
Materials and Methods
1. Study design and patients
Neurocognitive function tests were performed since 2008 as a part of hospital work up for the patients who were going to have PBT. However, analysis of the accumulated result of the tests and also, expanded performance of the neurocognitive function test in a prospective manner as a research project was approved by Institutional Review Board (IRB) in 2014 (IRB number: NCC2014-0044). Since then, all patients with IGCT undergoing PBT were referred to a clinical psychologist after providing informed consent to evaluate their neuropsychological functions before starting PBT (T1, baseline evaluation) and yearly after completion of PBT (T2, T3, etc.). The primary objective of this study was to determine the factors affecting the baseline neuropsychological functions. Additionally, because early follow-up results were available in some of the patients, changes in neuropsychological functions at 1-2 years after PBT were evaluated as a secondary objective.
Between June 2008 and November 2014, 40 children with an IGCT underwent PBT at the National Cancer Center, South Korea, and were referred for baseline evaluation. Of the 40 patients who underwent PBT, three who refused to undergo the neurocognitive and psychological tests and three who had previously received radiotherapy were excluded. Therefore, baseline data were analyzed for 34 patients. Follow-up assessments (T2) were conducted in 20 patients at a median of 15.0 months (range, 6.0 to 28.8 months) after PBT. Fourteen patients were not included in follow-up assessments because three patients refused to undergo the follow-up tests, seven were lost to follow-up, and four were followed up for < 1 year.
2. Treatment
All patients received four cycles of chemotherapy before PBT according to the Korean Pediatric Neuro-Oncology protocol. According to this protocol, the radiation field and dose are determined by the tumor histology, initial tumor markers, tumor extent, and response to neoadjuvant chemotherapy. Patients with germinoma received four cycles of chemotherapy with alternating carboplatin/etoposide (course A) and cyclophosphamide/VP-16 (course B) before radiotherapy. Patients with NGGCT received four cycles of chemotherapy consisting of carboplatin, etoposide, and bleomycin alternating with cyclophosphamide, etoposide, and bleomycin. Patients with pure germinoma received a whole ventricle irradiation (WVI) dose of 19.8 Gy followed by a primary site boost of 10.8 Gy in patients with a complete response to chemotherapy, and a 19.8 Gy boost in patients with a partial response. Patients with disseminating germinoma received a craniospinal irradiation (CSI) dose of 25.2 Gy. Patients with NGGCT received CSI at a dose of 36 Gy for localized disease or 39.6 Gy for disseminated disease. A primary site boost was given up to a total dose of 55.8-59.4 Gy depending on the extent of the residual tumor and the limitations imposed by the organs at risk. A boost target volume was created with a 1.5 cm margin around the primary tumor volume, and in the case of bifocal tumors (involving both suprasellar and pineal glands), the whole ventricle was included in boost volume. In the case of basal ganglia tumors, the boost target volume was same as that of the tumors in other locations, but because the remnant white matter change after chemotherapy precluded clear response evaluations, most patients with basal ganglia tumor received high-end of dose ranges. In all patients, PBT was used in passive scattering (double scattering) mode.
3. Assessment of neurocognitive function and emotional and behavioral problems
To measure general intellectual functions, the Korean Wechsler Intelligence Scale for Children, third edition (KWISC-III) and The Korean Wechsler Adult Intelligence Scale (K-WAIS) were used for children of 6 to 15 years and children aged ≥ 16 years, respectively [16,17]. The KWISC-III can yield full-scale IQ (FIQ), verbal IQ (VIQ), performance IQ (PIQ) scores, verbal comprehension, perceptual organization (PO), freedom from distractibility (FD), and processing speed (PS), while K-WAIS only provides FIQ, VIQ, and PIQ. The standard scores of all measures are a mean of 100 and a standard deviation (SD) of 15. The Rey-Kim Memory Test and Kim’s Frontal Lobe Executive Neuropsychological Test were used to assess the children’s memory and frontal lobe executive functions, respectively [18,19]. These tests provide the memory quotient (MQ) and the executive IQ (EIQ), respectively.
The Korean version of the Child Behavior Checklist (K-CBCL) [20] was used to assess emotional and behavioral problems. The standard test scores are normalized T scores with a mean of 50 and a SD of 10. Higher scores indicate greater problems.
4. Statistical analyses
PASW ver. 18 (SPSS Inc., Chicago, IL) was used for all analyses. All test scores were compared to age-matched standardized normative values using t tests. The effects of clinical factors on neurocognitive and psychological functioning were analyzed using t tests or analysis of variance (ANOVA). Differences in neurocognitive and psychological functioning between tumor locations were examined using ANOVA and χ2 tests. The changes in neurocognitive functioning between T1 and T2 were examined using repeated-measures ANOVA and the differences between T1 and T2 were analyzed using pairwise comparison tests according to the tumor location. Statistical significance was defined as p-values of < 0.05.
Results
1. Patient, treatment, and tumor characteristics
The clinical and tumor characteristics of the patients are summarized in Table 1. Overall, 66% of the patients were male, and the median age at T1 was 12.3 years (range, 7.3 to 18.4 years). There were eight patients with suprasellar tumors (22.9%), 10 with pineal gland tumors (28.6%), six with basal ganglia tumors (17.1%), and 10 with bifocal tumors involving both the suprasellar and pineal gland (31.4%). About 60% of patients had hydrocephalus and underwent procedures for treatment. Twenty patients (58.8%) received CSI or whole brain irradiation and the other 14 patients received WVI. The median radiation dose applied to the primary tumor was 30.6 Gy (range, 30.0 to 45.0 Gy) for patients with pure germinoma and 55.8 Gy (range, 30.6 to 55.8 Gy) for patients with NGGCT. Two patients diagnosed with pure germinomas received 45 Gy of radiotherapy because one patient had residual disease after chemotherapy and the other had a high β human chorionic gonadotropin level at initial diagnosis. Three patients with NGGCT had mature teratoma as a nongerminoma component and received 30.6 to 39.6 Gy. Several clinical variables, including the presence of hydrocephalus, endocrine symptoms (i.e., polydipsia and polydipsia), visual symptoms (i.e., double vision, visual disturbance, and strabismus), and the duration of symptoms (time from initial signs to tumor diagnosis) were closely related to tumor locations. Suprasellar tumor had more endocrine symptoms (p < 0.001), while pineal tumor was associated with hydrocephalus (p=0.008) and visual symptoms (p=0.140). No significant differences in the duration of symptoms were found according to the tumor locations, but tumors in pineal gland had a shorter duration than the other tumors (mean±SD, 4.0±3.77 months in basal ganglia vs. 12.37±13.53 in suprasella, 7.67±8.2 in basal ganglia and 11.1±11.25 in bifocal tumor; p=0.273). Therefore, we did not conduct further analyses investigating the effects of these variables on neuropsychological functioning.
Table 1.
Patient characteristics at baseline
| Characteristic | No. (%) |
|---|---|
| Sex | |
| Male | 23 (67.6) |
| Female | 11 (32.4) |
| Age at diagnosis (yr) | |
| Mean±SD | 12.3±3.1 |
| Median (range) | 12.0 (7.0-18.1) |
| Age at first evaluation (yr) | |
| Mean±SD | 12.6±3.1 |
| Median (range) | 12.3 (7.3-18.4) |
| Interval between diagnosis and first evaluation (mo) | |
| Mean±SD | 3.9±1.3 |
| Median (range) | 3.7 (0.5-8.0) |
| Tumor location | |
| Suprasellar | 8 (23.5) |
| Pineal gland | 10 (29.4) |
| Basal ganglia | 6 (17.6) |
| Bifocala) | 10 (29.4) |
| Diagnosis | |
| Germinoma | 18 (52.9) |
| NGGCT or mixed IGCT | 16 (47.1) |
| Hydrocephalus | |
| Yes | 21 (61.8) |
| No | 13 (38.2) |
| Procedure for hydrocephalusb) | |
| Yes | 20 (58.8) |
| No | 14 (41.2) |
| Surgery extent | |
| GTR | 3 (8.8) |
| NTR or STR | 4 (11.8) |
| Biopsy only | 24 (70.6) |
| No surgery | 3 (8.8) |
| Radiation field | |
| CSI+primary site | 19 (55.9) |
| WBI+primary site | 1 (2.9) |
| WVI+primary site | 14 (41.2) |
| Radiation dose, median (range, Gy) | |
| Germinoma | 30.6 (30.0-45.0) |
| NGGCT | 54.6 (30.6-55.8) |
| Total patients | 39.6 (30.0-55.8) |
SD, standard deviation; NGGCT, non-germinomatous germ cell tumor; IGCT, intracranial germ cell tumor; GTR, gross total resection; NTR, near total resection; STR, subtotal resection; CSI, craniospinal irradiation; WBI, whole brain irradiation; WVI, whole ventricle irradiation.
Suprasellar and pineal gland involvement,
Ventriculoperitoneal shunt, external ventricular drainage, and ventriculostomy.
2. Neurocognitive and psychological functioning at baseline (T1) and effect of tumor location
Baseline neurocognitive and psychological functioning did not show any differences between patients with pure germinomas and those with NGGCTs. To evaluate the effects of tumor location, patients were grouped into four subgroups; suprasellar, pineal gland, basal ganglia, and bifocal. Tumor location was not associated with tumor histology.
Table 2 shows the baseline neurocognitive functioning of the patients. The mean scores for MQ and EIQ were lower than those for the normal population (p=0.041 and p < 0.001, respectively). Although the patients also had slightly lower scores for PIQ and PS than the normal population, the values were still within the normal range (range, 85 to 115). When analyzed by tumor location, patients with basal ganglia tumor had lower scores than the normal population for all domains. One-way ANOVA showed that all neurocognitive functioning domains, except for the FD and MQ, differed significantly by tumor location (p=0.016 to p=0.044). Post hoc comparison revealed significant differences in each domain between patients with basal ganglia tumors and those with suprasellar tumors. When the frequencies of scores lower than one SD within each tumor location were compared with the expected 16% of the general population, they were greatest for patients with basal ganglia tumors on all neurocognitive domains (Fig. 1A).
Table 2.
Baseline neurocognitive functioning according to tumor location
| Variable | No. | Total patients | p-valuea) | Suprasellar | p-valuea) | Pineal gland | p-valuea) | Basal ganglia | p-valuea) | Bifocal | p-valuea) | One-way ANOVA |
Post hoc comparison | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| df | p-value | |||||||||||||
| FIQ | 34 | 96.74±21.36 | 0.379 | 110.50±17.98 | 0.143 | 92.70±25.60 | 0.391 | 76.83±8.66 | 0.001 | 101.70±15.69 | 0.740 | 3,30 | 0.016 | BG < suprasellar |
| VIQ | 33 | 101.36±19.13 | 0.685 | 112.00±20.84 | 0.178 | 99.80±18.78 | 0.974 | 83.33±10.52 | 0.012 | 106.30±16.21 | 0.250 | 3,29 | 0.033 | BG < suprasellar |
| PIQ | 33 | 91.55±22.80 | 0.041 | 107.57±14.67 | 0.221 | 86.10±36.46 | 0.183 | 73.83±10.82 | 0.002 | 96.40±15.59 | 0.484 | 3,29 | 0.035 | BG < suprasellar |
| VC | 29 | 102.17±18.72 | 0.537 | 112.25±16.74 | 0.077 | 96.63±20.56 | 0.657 | 83.80±13.24 | 0.052 | 109.13±12.30 | 0.074 | 3,25 | 0.020 | BG < suprasellar |
| PO | 29 | 96.83±21.48 | 0.433 | 110.25±17.54 | 0.142 | 87.25±27.44 | 0.230 | 82.00±11.38 | 0.024 | 102.25±14.45 | 0.673 | 3,25 | 0.044 | BG, pineal < suprasellar |
| FD | 28 | 99.39±19.35 | 0.869 | 102.86±17.06 | 0.673 | 102.88±26.51 | 0.768 | 84.40±7.47 | 0.010 | 102.25±16.26 | 0.707 | 3,24 | 0.312 | - |
| PS | 29 | 86.59±24.61 | 0.007 | 104.88±14.95 | 0.387 | 82.63±25.48 | 0.095 | 67.00±17.85 | 0.014 | 84.50±26.24 | 0.139 | 3,25 | 0.039 | BG < suprasellar |
| MQ | 32 | 84.75±19.35 | 0.000 | 86.50±23.85 | 0.153 | 90.78±16.13 | 0.125 | 81.50±16.51 | 0.041 | 79.33±20.92 | 0.018 | 3,28 | 0.633 | - |
| EIQ | 23 | 84.52±26.18 | 0.010 | 86.83±19.93 | 0.167 | 104.14±14.65 | 0.482 | 60.60±24.80 | 0.024 | 78.20±29.82 | 0.177 | 3,19 | 0.024 | BG < pineal |
Values are presented as mean±standard deviation. FIQ, full-scale intelligence quotient (IQ); BG, basal ganglia; VIQ, verbal IQ; PIQ, performance IQ; VC, verbal comprehension; PO, perceptional organization; FD, freedom of distractibility; PS, processing speed; MQ, memory quotient; EIQ, executive IQ.
Comparison with normal population.
Fig. 1.
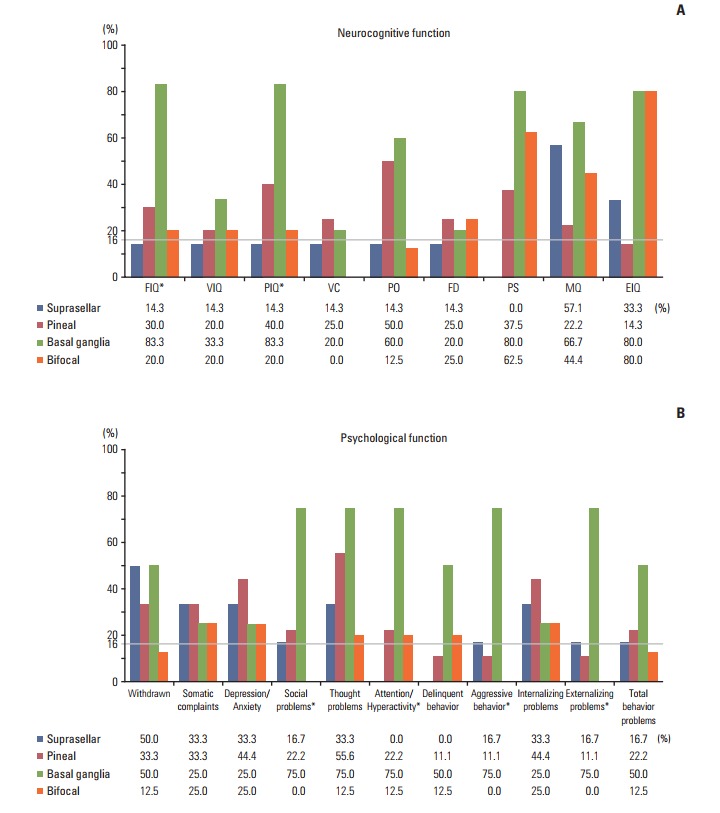
Proportions of patients with impairments in each neurocognitive (A) and psychological (B) domain. FIQ, full-scale intelligence quotient (IQ); VIQ, verbal IQ; PIQ, performance IQ; VC, verbal comprehension; PO, perceptional organization; FD, freedom of distractibility; PS, processing speed; MQ, memory quotient; EIQ, executive IQ. *p < 0.05.
Table 3 shows the results of the K-CBCL. In all patients combined, the mean scores for all domains were within the normal ranges for the general population (range, 40 to 60). Basal ganglia tumor patients had significantly higher scores for externalization problems, total behavioral problems, social problems, attention/hyperactivity, delinquent behaviors, and aggressive behaviors when compared with the normal population. Patients with a basal ganglia tumor more frequently had scores greater than one SD for most subscales of the K-CBCL when compared with the expected 16% of the normal population (Fig. 1B). Overall, 75% of the parents of patients with basal ganglia tumors reported that their children displayed attention/hyperactivity problems, aggressive behaviors, thought problems, social problems, and externalization problems.
Table 3.
Psychological functioning according to tumor location
| Variable | GCTs (n=27) | p-valuea) | Suprasellar (n=6) | p-valuea) | Pineal gland (n=9) | p-valuea) | Basal ganglia (n=4) | p-valuea) | Bifocal (n=8) | p-valuea) | ANOVA p-value | Post hoc comparison |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Internalizing problems | 54.89±10.26 | 0.020 | 58.00±8.25 | 0.063 | 55.11±13.68 | 0.295 | 57.75±3.78 | 0.026 | 50.88±9.55 | 0.803 | 0.579 | - |
| Externalizing problems | 48.74±10.62 | 0.543 | 49.67±9.14 | 0.932 | 47.11±9.44 | 0.385 | 63.25±5.85 | 0.020 | 42.63±8.70 | 0.048 | 0.008 | Pineal, bifocal < BG |
| Total behavior problems | 52.33±10.15 | 0.243 | 54.00±8.41 | 0.297 | 52.67±9.94 | 0.444 | 62.25±5.32 | 0.019 | 45.75±9.94 | 0.266 | 0.050 | Bifocal < BG |
| Withdrawn | 56.19±13.17 | 0.022 | 63.50±18.05 | 0.126 | 55.56±14.81 | 0.293 | 56.50±11.03 | 0.324 | 51.25±5.88 | 0.566 | 0.411 | - |
| Somatic complaints | 54.85±9.21 | 0.011 | 59.00±6.00 | 0.014 | 53.11±9.68 | 0.363 | 58.25±5.38 | 0.055 | 52.00±11.64 | 0.642 | 0.436 | - |
| Depression/Anxiety | 53.85±10.32 | 0.063 | 53.83±12.56 | 0.488 | 55.22±11.86 | 0.223 | 54.25±8.06 | 0.369 | 52.13±9.39 | 0.542 | 0.950 | - |
| Social problems | 56.07±9.52 | 0.003 | 53.33±6.51 | 0.265 | 56.11±9.33 | 0.085 | 68.75±7.18 | 0.014 | 51.75±8.07 | 0.559 | 0.016 | Bifocal < BG |
| Thought problems | 54.78±9.52 | 0.015 | 53.67±9.14 | 0.371 | 56.78±9.22 | 0.058 | 63.00±11.46 | 0.108 | 49.25±6.61 | 0.758 | 0.096 | - |
| Attention/Hyperactivity | 53.78±11.05 | 0.087 | 49.33±6.31 | 0.806 | 55.89±9.44 | 0.098 | 69.00±8.68 | 0.022 | 47.13±9.22 | 0.407 | 0.003 | Suprasellar, bifocal < BG |
| Delinquent behavior | 49.11±7.70 | 0.554 | 48.71±7.06 | 0.552 | 47.89±7.64 | 0.431 | 59.25±0.96 | 0.000 | 46.13±6.77 | 0.150 | 0.026 | Bifocal < BG |
| Aggressive behavior | 49.59±10.77 | 0.846 | 50.00±9.45 | 1.000 | 47.33±9.35 | 0.417 | 64.25±7.93 | 0.037 | 44.50±8.99 | 0.127 | 0.013 | Pineal, bifocal < BG |
Values are presented as mean±standard deviation. Higher scores mean greater problems. GCT, germ cell tumor; BG, basal ganglia.
Comparison with normal population.
3. Neurocognitive and psychological functioning at 1-2 years after PBT (T2)
At 1-2 years after PBT, most scores of the neurocognitive test and the CBCL were not significantly different from those of the normal population, except for the MQ (mean, 88.6; p=0.045). The T2 scores for all domains of neurocognitive functioning and the CBCL were not significantly different from those at T1. However, in patients with a suprasellar tumor, the test scores tended to decrease between T1 and T2, except for MQ and EIQ. In contrast, the scores for most domains increased between these times in patients with pineal, basal ganglia, or bifocal tumors. In pairwise comparisons between T1 and T2, differences were found as follows: suprasellar vs. basal ganglia for FIQ; suprasellar vs. pineal gland for VIQ; suprasellar vs. bifocal for PO; pineal gland vs. basal ganglia for PS; suprasellar vs. pineal gland and basal ganglia for MQ (Fig. 2). CBCL scores were not analyzed according to tumor location because of the small sample size of T2 (n=13).
Fig. 2.
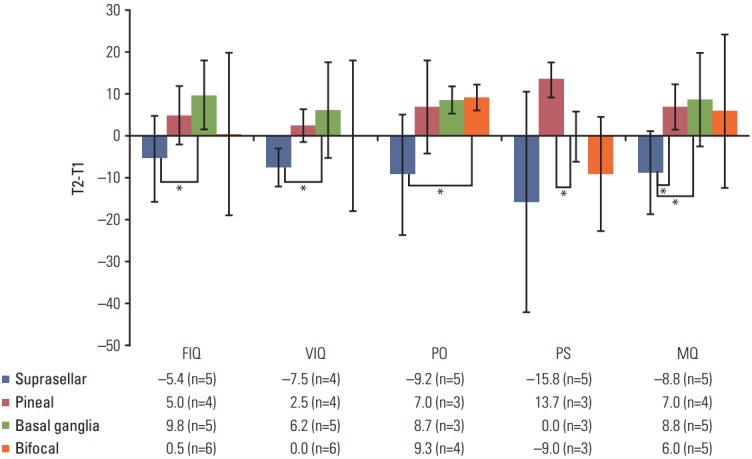
Changes in neurocognitive functioning from baseline (T1) to 1-2 years after proton beam therapy (T2) according to tumor location. The y axis represents the difference between T2 and T1; therefore, a negative value of T2-T1 indicates decreased functions. FIQ, full-scale intelligence quotient (IQ); VIQ, verbal IQ; PO, perceptional organization; PS, processing speed; MQ, memory quotient; T2-T1, difference between T1 and T2. *p < 0.05. Bars indicate standard deviation.
We also examined the potential effects of the radiation field and dose on neurocognitive functioning at T2 (Table 4). Patients who received CSI tended to have lower scores at T2 than at T1, whereas those who received WVI tended to have higher scores at T2 than at T1. However, these changes were not statistically significant. There were no significant differences in neurocognitive functioning scores between patients who received higher doses (> 39.6 Gy) and those who received lower doses (≤ 39.6 Gy).
Table 4.
Effect of radiation field and dose on cognitive functions
| Variable | Radiation field |
Radiation dose, Gy |
||||
|---|---|---|---|---|---|---|
| T2-T1 difference (mean±SD) |
p-value | T2-T1 difference (mean±SD) |
p-value | |||
| CSI | WVI | ≤ 39.6 | > 39.6 | |||
| FIQ | –0.80±17.79 (n=10) | 5.30±6.04 (n=10) | 0.327 | 4.41±9.14 (n=12) | –1.00±18.14 (n=8) | 0.387 |
| VIQ | –3.22±15.06 (n=9) | 4.00±8.38 (n=10) | 0.207 | 1.58±9.66 (n=12) | –1.14±16.50 (n=7) | 0.653 |
| PIQ | 0.22±23.60 (n=9) | 6.30±8.87 (n=10) | 0.458 | 7.33±14.37 (n=12) | –3.29±20.69 (n=7) | 0.203 |
| VC | 1.14±8.63 (n=7) | 2.63±9.23 (n=8) | 0.754 | 1.10±8.95 (n=10) | 3.60±8.79 (n=5) | 0.617 |
| PO | –4.00±14.85 (n=6) | 8.25±6.34 (n=8) | 0.078 | 4.80±11.30 (n=10) | –2.00±14.68 (n=5) | 0.336 |
| FD | –7.83±9.02 (n=6) | 5.00±14.44 (n=8) | 0.081 | 2.00±14.60 (n=10) | –6.75±9.91 (n=4) | 0.297 |
| PS | –12.67±24.30 (n=6) | 1.38±14.27 (n=8) | 0.199 | –3.78±20.43 (n=9) | –6.20±20.61 (n=5) | 0.836 |
| MQ | 1.80±16.31 (n=10) | 4.44±10.50 (n=9) | 0.684 | 5.09±10.11 (n=11) | 0.25±17.65 (n=8) | 0.458 |
| EIQ | 2.60±19.97 (n=5) | 4.38±14.88 (n=8) | 0.857 | 0.50±16.30 (n=10) | 14.33±12.66 (n=3) | 0.208 |
SD, standard deviation; CSI, craniospinal irradiation; WVI, whole ventricle irradiation; FIQ, full-scale intelligence quotient (IQ); VIQ, verbal IQ; PIQ, performance IQ; VC, verbal comprehension; PO, perceptional organization; FD, freedom of distractibility; PS, processing speed; MQ, memory quotient; EIQ, executive IQ.
Discussion
In this study, children with IGCT displayed significantly lower PIQ, PS, MQ, and EIQ scores than an age-matched normal population. Deficits in attention and memory, including slow speed processing, are the most commonly reported problems in childhood brain tumor survivors in general and are commonly attributed to cranial irradiation [21,22]. The deficits in memory function caused by cranial irradiation are now known to represent a component of diffuse global dysfunction of brain function associated with white matter changes [21,23] rather than isolated memory disturbances [24]. However, our data revealed disturbances in memory function were present even before radiotherapy was started. Wilkening et al. [25] specifically examined memory function in IGCT and observed amnesia in 38% of patients. They also reported that amnesia was not related to the presence of hydrocephalus or history of cranial radiotherapy, and that low memory function was not associated with global decreases in IQ, which are sometimes observed in younger children with other tumors, such as medulloblastoma [14,15]. Williams and Pennybacker [26] examined brain tumor patients with memory impairment and found that most of their tumors were located in a deep, midline position. They also reported that patients with tumors involving periventricular structures had more frequent memory loss than those with tumors in frontal locations .
The effects of tumor location on neurocognitive functioning observed in the present study were consistent with those reported by Liang et al. [11], who found that IGCT arising in the basal ganglia is associated with worse neuropsychological outcomes than tumors arising in other locations. We showed that poor neurocognitive functioning of patients with basal ganglia tumors persists after treatment, but tends to improve within the early follow-up period, although the mean scores at T2 still remained poor relative to the normal population. According to Liang et al. [11], the impaired neurocognitive function in the patients with basal ganglia tumors was likely to be life-long. In contrast to these authors, who only assessed neurocognitive aspects in children, we also examined psychological functioning in patients with basal ganglia tumors. We found that children with tumors located in the basal ganglia showed various behavioral issues, as reflected in the results of CBCL compared to patients with tumors in other locations. The basal ganglia have extensive connections with the cerebral cortex, especially the frontal cortex. In addition to motor function, the basal ganglia play various roles in cognition, memory, and emotion through connections with the limbic cortex [27,28], and disruption of these functions may cause externalization of symptoms of patients with basal ganglia tumors.
Notably, we observed tendencies for FIQ, VIQ, perceptional organization, and MQ to decrease between T1 and T2 in patients with suprasellar tumors. In contrast, these scores tended to increase between T1 and T2 in patients with tumors in the other sites. Although we only observed these changes over a relatively short time, the results appear to agree with those reported by Mabott et al. [13], who found a decline in the average PS and delayed visual memory, especially in patients with suprasellar tumors. Dennis et al. [29] reported impaired memory in children with hormone deficiencies. In the present study, patients with suprasellar tumors tended to have decreases in neurocognitive functioning, and the changes in memory function in suprasellar patients were significantly greater than those in children with pineal or basal ganglia tumors. All of the patients with suprasellar tumors had hormone deficiencies, which may have contributed to neurocognitive deficiencies. The boost field applied to suprasellar tumors must target a larger volume of the medial temporal lobes because of the anatomical proximity relative to tumors in the pineal gland [30]. A longer follow-up period is needed to determine if differences in the temporal dose–volume histograms have long-term effects on the neurocognitive functioning of children with IGCT.
It should be noted that our study was limited in that we analyzed a relatively small number of patients, and only 20/34 patients were assessed at T2. However, this study is a preliminary report of an ongoing prospective study and the first to report the short-term neurocognitive outcomes of PBT in children with IGCT. It should also be noted that we assessed the psychological status of children as well as neurocognitive functioning before and after treatment. Because the study included children with histories of chemotherapy and surgery, the results do not represent the neuropsychological state of the child at initial diagnosis. Nevertheless, the timing of the baseline examination before radiotherapy seems reasonable considering the practical limitations of performing neuropsychological evaluation upon diagnosis of IGCT. Further analysis of longitudinal changes in neurocognitive functions after PBT will be available after longer follow-up with more patients.
In conclusion, neuropsychological functioning in patients with IGCT may be primarily influenced by tumor location itself, and does not change in the short term after treatment. However, further study is needed to evaluate the long-term effects of PBT on neurocognitive function.
Acknowledgments
This study was supported by National Cancer Center Research Grant No. 1610590 and 1611460.
Footnotes
Conflict of interest relevant to this article was not reported.
References
- 1.Foo AS, Lim C, Chong DQ, Tan DY, Tham CK. Primary intracranial germ cell tumours: experience of a single South-East Asian institution. J Clin Neurosci. 2014;21:1761–6. doi: 10.1016/j.jocn.2014.01.021. [DOI] [PubMed] [Google Scholar]
- 2.Nomura K. Epidemiology of germ cell tumors in Asia of pineal region tumor. J Neurooncol. 2001;54:211–7. doi: 10.1023/a:1012771204732. [DOI] [PubMed] [Google Scholar]
- 3.Jinguji S, Yoshimura J, Nishiyama K, Aoki H, Nagasaki K, Natsumeda M, et al. Factors affecting functional outcomes in long-term survivors of intracranial germinomas: a 20-year experience in a single institution. J Neurosurg Pediatr. 2013;11:454–63. doi: 10.3171/2012.12.PEDS12336. [DOI] [PubMed] [Google Scholar]
- 4.Merchant TE, Sherwood SH, Mulhern RK, Rose SR, Thompson SJ, Sanford RA, et al. CNS germinoma: disease control and long-term functional outcome for 12 children treated with craniospinal irradiation. Int J Radiat Oncol Biol Phys. 2000;46:1171–6. doi: 10.1016/s0360-3016(99)00375-2. [DOI] [PubMed] [Google Scholar]
- 5.Schoenfeld A, Haas-Kogan DA, Molinaro A, Banerjee A, Nicolaides T, Tihan T, et al. Pure germinomas of the central nervous system: treatment strategies and outcomes. J Neurooncol. 2014;120:643–9. doi: 10.1007/s11060-014-1599-7. [DOI] [PubMed] [Google Scholar]
- 6.Kim JY, Baek HJ. The treatment result of intracranial germ cell tumors in Korea: KSPNO G081 multicenter trial, KSPNOG052/082 Trial. New horizon of the germ cell therapy. Seoul: The Korean Society for Pediatric Neuro-Oncology; 2013. [Google Scholar]
- 7.Kim JW, Kim WC, Cho JH, Kim DS, Shim KW, Lyu CJ, et al. A multimodal approach including craniospinal irradiation improves the treatment outcome of high-risk intracranial nongerminomatous germ cell tumors. Int J Radiat Oncol Biol Phys. 2012;84:625–31. doi: 10.1016/j.ijrobp.2011.12.077. [DOI] [PubMed] [Google Scholar]
- 8.Robertson PL, Jakacki R, Hukin J, Siffert J, Allen JC. Multimodality therapy for CNS mixed malignant germ cell tumors (MMGCT): results of a phase II multi-institutional study. J Neurooncol. 2014;118:93–100. doi: 10.1007/s11060-013-1306-0. [DOI] [PubMed] [Google Scholar]
- 9.Spiegler BJ, Bouffet E, Greenberg ML, Rutka JT, Mabbott DJ. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J Clin Oncol. 2004;22:706–13. doi: 10.1200/JCO.2004.05.186. [DOI] [PubMed] [Google Scholar]
- 10.Reimers TS, Ehrenfels S, Mortensen EL, Schmiegelow M, Sonderkaer S, Carstensen H, et al. Cognitive deficits in long-term survivors of childhood brain tumors: identification of predictive factors. Med Pediatr Oncol. 2003;40:26–34. doi: 10.1002/mpo.10211. [DOI] [PubMed] [Google Scholar]
- 11.Liang SY, Yang TF, Chen YW, Liang ML, Chen HH, Chang KP, et al. Neuropsychological functions and quality of life in survived patients with intracranial germ cell tumors after treatment. Neuro Oncol. 2013;15:1543–51. doi: 10.1093/neuonc/not127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wilkening GN, Madden JR. Memory disorders in children with central nervous system germ cell tumors. J Pediatr Oncol Nurs. 2012;29:161–70. doi: 10.1177/1043454212439938. [DOI] [PubMed] [Google Scholar]
- 13.Mabbott DJ, Monsalves E, Spiegler BJ, Bartels U, Janzen L, Guger S, et al. Longitudinal evaluation of neurocognitive function after treatment for central nervous system germ cell tumors in childhood. Cancer. 2011;117:5402–11. doi: 10.1002/cncr.26127. [DOI] [PubMed] [Google Scholar]
- 14.Iuvone L, Peruzzi L, Colosimo C, Tamburrini G, Caldarelli M, Di Rocco C, et al. Pretreatment neuropsychological deficits in children with brain tumors. Neuro Oncol. 2011;13:517–24. doi: 10.1093/neuonc/nor013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shortman RI, Lowis SP, Penn A, McCarter RJ, Hunt LP, Brown CC, et al. Cognitive function in children with brain tumors in the first year after diagnosis compared to healthy matched controls. Pediatr Blood Cancer. 2014;61:464–72. doi: 10.1002/pbc.24746. [DOI] [PubMed] [Google Scholar]
- 16.Kwak KJ, Park HW, Kim CT. Korean Wechsler Intelligence Scale for Children. 3rd ed. Seoul: Special Education Publishing Co.; 2001. [Google Scholar]
- 17.Yeum TH, Park YS, Oh KJ, Kim JK, Lee YH. The manual of Korean-Wechsler Adult Intelligence Scale. Seoul: Korea Guidance; 1992. [Google Scholar]
- 18.Kim HK. Rey-Kim Memory Test: guidance. Daegu: Neuropsychology Press; 2005. [Google Scholar]
- 19.Kim HK. Kims frontal-executive neuropsychological test. Daegu: Neuropsychology Press; 2001. [Google Scholar]
- 20.Oh K, Lee H. Development of Korean version of Child Behavior Checklist (K-CBCL) Seoul: Korean Research Foundation Report; 1990. [Google Scholar]
- 21.Nathan PC, Patel SK, Dilley K, Goldsby R, Harvey J, Jacobsen C, et al. Guidelines for identification of, advocacy for, and intervention in neurocognitive problems in survivors of childhood cancer: a report from the Children's Oncology Group. Arch Pediatr Adolesc Med. 2007;161:798–806. doi: 10.1001/archpedi.161.8.798. [DOI] [PubMed] [Google Scholar]
- 22.Nagel BJ, Delis DC, Palmer SL, Reeves C, Gajjar A, Mulhern RK. Early patterns of verbal memory impairment in children treated for medulloblastoma. Neuropsychology. 2006;20:105–12. doi: 10.1037/0894-4105.20.1.105. [DOI] [PubMed] [Google Scholar]
- 23.Jacola LM, Ashford JM, Reddick WE, Glass JO, Ogg RJ, Merchant TE, et al. The relationship between working memory and cerebral white matter volume in survivors of childhood brain tumors treated with conformal radiation therapy. J Neurooncol. 2014;119:197–205. doi: 10.1007/s11060-014-1476-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Reimers TS, Mortensen EL, Schmiegelow K. Memory deficits in long-term survivors of childhood brain tumors may primarily reflect general cognitive dysfunctions. Pediatr Blood Cancer. 2007;48:205–12. doi: 10.1002/pbc.20818. [DOI] [PubMed] [Google Scholar]
- 25.Wilkening GN, Madden JR, Barton VN, Roberts A, Foreman NK. Memory deficits in patients with pediatric CNS germ cell tumors. Pediatr Blood Cancer. 2011;57:486–91. doi: 10.1002/pbc.23096. [DOI] [PubMed] [Google Scholar]
- 26.Williams M, Pennybacker J. Memory disturbances in third ventricle tumours. J Neurol Neurosurg Psychiatry. 1954;17:115–23. doi: 10.1136/jnnp.17.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Helie S, Chakravarthy S, Moustafa AA. Exploring the cognitive and motor functions of the basal ganglia: an integrative review of computational cognitive neuroscience models. Front Comput Neurosci. 2013;7:174. doi: 10.3389/fncom.2013.00174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Leisman G, Braun-Benjamin O, Melillo R. Cognitive-motor interactions of the basal ganglia in development. Front Syst Neurosci. 2014;8:16. doi: 10.3389/fnsys.2014.00016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dennis M, Spiegler BJ, Obonsawin MC, Maria BL, Cowell C, Hoffman HJ, et al. Brain tumors in children and adolescents. III. Effects of radiation and hormone status on intelligence and on working, associative and serial-order memory. Neuropsychologia. 1992;30:257–75. doi: 10.1016/0028-3932(92)90004-6. [DOI] [PubMed] [Google Scholar]
- 30.Kim JY, Park J. Understanding the treatment strategies of intracranial germ cell tumors: focusing on radiotherapy. J Korean Neurosurg Soc. 2015;57:315–22. doi: 10.3340/jkns.2015.57.5.315. [DOI] [PMC free article] [PubMed] [Google Scholar]


