Abstract
Introduction
Evaluation of accuracy, precision, and trending ability of cardiac index (CI) measurements using the Aesculon™ bioimpedance electrical cardiometry (Aesc) compared to the continuous pulmonary artery thermodilution catheter (PAC) technique before, during, and after cardiac surgery.
Methods
A prospective observational study with fifty patients with ASA 3-4. At six time points (T), measurements of CI simultaneously by continuous cardiac output pulmonary thermodilution and thoracic bioimpedance and standard hemodynamics were performed. Analysis was performed using Bland-Altman, four-quadrant plot, and polar plot methodology.
Results
CI obtained with pulmonary artery thermodilution and thoracic bioimpedance ranged from 1.00 to 6.75 L min−1 and 0.93 to 7.25 L min−1, respectively. Bland-Altman analysis showed a bias between CIBIO and CIPAC of 0.52 liters min−1 m−2, with LOA of [−2.2; 1.1] liters min−1 m−2. Percentage error between the two techniques was above 30% at every time point. Polar plot methodology and 4-quadrant analysis showed poor trending ability. Skin incision had no effect on the results.
Conclusion
CI obtained by continuous PAC and CI obtained by Aesculon bioimpedance are not interchangeable in cardiac surgical patients. No effects of skin incision were found. International clinical trial registration number is ISRCTN26732484.
1. Introduction
Measurement of cardiac output (CO) and cardiac index (CI) is commonly used in patients undergoing cardiac surgery. The continuous pulmonary artery thermodilution technique is well known as a method for advanced monitoring of cardiovascular function and is regularly used as a clinical reference technique in method comparison studies. However, a couple of studies during the past years showed that a true golden standard for measuring cardiac output does not exist.
Due to its invasive nature, the use of the PAC is associated with severe risk and there is still discussion on whether the positive effects of PAC outweigh the adverse effects [1–5].
Today, there are less invasive or even noninvasive monitoring devices available. The ideal technique should be reliable, noninvasive, continuous, cost-effective, and user independent and should have a fast response time enabling rapid detection of hemodynamic changes [6]. Considering the growing age of the surgical population with severe comorbidity, it is likely that monitoring of CO will be important, also in non-cardiac-surgery patients. Moreover, assessment of CO and stroke volume (SV) is a prerequisite to establish early goal-directed therapy during the perioperative period.
One of the most recent noninvasive techniques for assessment of CO is based on a modified thoracic bioimpedance algorithm. Briefly, thoracic bioimpedance is based on the theory that the thorax is a blood filled cylinder. According to Ohm's law (resistance = voltage/current) this model assumes that the impedance of thoracic tissue is parallel to that of blood. Blood related impedance changes repeat themselves with every heart beat and are linked to cardiac activity [7]. During systole, approximately 60 milliseconds after opening of the aortic valve, erythrocytes change their position from a random alignment to one parallel to the axial blood flow. This results in an increased conductivity allowing estimating the acceleration of flow through the aortic artery. However, early adoption of the technique showed diverging results with respect to precision and accuracy [8].
The basic equation was modified by Bernstein and Osypka, so that the maximum rate of change of impedance is related to the peak aortic blood acceleration [9]. The method used in the Aesculon was initially described as electrical cardiometry and it contributes the increase in conductivity to the orientation change of the red blood cells to determine the velocity of the blood flow, claiming to be a more accurate technique in a wide spectrum of patient conditions and patient populations including neonates and children [10, 11].
The bioimpedance method shows good results in clinical studies in young healthy volunteers. However, reliability in critically ill patients and in perioperative use is not proven and the available literature is inconclusive [12–15]. Moreover, until now it is unclear whether interruption of the skins integrity by a surgical incision could be a source of error in bioimpedance measurements. Recently Huang et al. found indications that skin incision can interfere with the bioimpedance technique [16].
Most available studies were performed in a neonatal and pediatric populations or during cardiac surgery, mainly in the postoperative period. Studies in adult patients undergoing high-risk cardiac surgery are lacking; therefore, we studied patient in the entire perioperative period [10, 11, 17].
The aim of the present study was to compare the accuracy, precision, and trending ability of a thoracic bioimpedance technique with continuous pulmonary artery thermodilution before, during, and after surgical intervention.
Secondary aim was to assess whether interruption of the skins integrity and opening of the thoracic cavity by a surgical incision could be a source of error in bioimpedance measurements.
2. Materials and Methods
2.1. Study Design
The study protocol was approved by the institutional review board of the Maastricht University Medical Center+ (MEC 08-4-075) and written informed consent was obtained from each patient. In this prospective observational study, 50 adult patients planned for elective cardiac surgery were included. Exclusion criteria were age < 18 years and a contraindication for placement of a Swan Ganz catheter. As placement of a Swan Ganz catheter was needed to retrieve CO measurements, we only included patients receiving the PAC in accordance with routine care.
2.2. Measurement Protocol
After arrival in the operating theatre, patients were connected to standard monitoring including heart rate, invasive blood pressure, and oxygen saturation. Anesthesia was induced according to local protocols. Thereafter, after rubbing and cleaning the skin with alcohol to achieve a skin-to-electrode impedance as low as possible, four standard electrocardiogram electrodes were placed according to the manual of the Aesculon (Osypka Medical, Berlin, Germany) on the left part of the neck and on the left part of the thorax at the level of the processus xiphoideus. Then the Aesculon monitor was connected to the electrodes for continuous display of bioimpedance cardiac index (CIBIO). A PAC (Edwards Life Sciences Corporation, Irvine, CA, USA, continuous cardiac output VIP catheter with SvO2, model 746F8) was inserted via the right or left internal jugular vein in order to continuously measure thermodilution cardiac index (CIPAC). To exclude the possibility of incorrect measurements during rapid fluid injections and hemodynamic instability we measured CIBIO and CIPAC at hemodynamic stability, in the absence of engraving events (e.g., profound bleeding, hypotension, and arrhythmias) and during normothermia.
We used the mean of two measurements of continuous CO. We used this as result per measurement and presented the results for each time point separately. There was no correction for repeated measurements applied because we analyzed the results per time point separately and assumed the measurements to be independent due to the extensive fluctuations.
Measurements were performed at 6 time points (T): after induction and prior to incision (T1), prior to cannulation of the aorta (T2), 10 minutes after protamine administration (T3), 30 minutes after arrival in the ICU (T4), 1 hour after extubation (T5), and 1 day postoperatively at 08.00 a.m. (T6). In addition to CI, heart rate, arterial blood pressure, and central and peripheral temperature were recorded at these time points.
2.3. Statistical Analysis
A sample size calculation was performed based on a previous study by Schmidt and coworkers [18]. Assuming a true difference between CIBIO and CIPAC of 0.5 liters/min/m2 and corresponding standard deviation (SD) of 1.0 liter/min/m2, a total number of 33 patients were needed to reject the null hypothesis that the difference is 0 with a power of 0.8 and type 1 error of 0.05. To correct for loss to follow-up an additional 17 patients were included, resulting in 50 patients in total.
Data were checked for normality using the Shapiro-Wilk test histograms (visually), including the difference between CIBIO and CIPAC. Descriptive analysis was performed using number (%) or mean ± SD. Differences between the absolute CI measurements were assessed using the paired t-test. Accuracy and precision of CIBIO against CIPAC at the various time points were assessed using Bland-Altman analysis and plots showing the bias, limits of agreement (LOA), and percentage error (PE) [19, 20]. CIBIO and CIPAC were considered interchangeable if the PE was <30%.
According to the literature, BMI might influence reliability of the bioimpedance measurements [18, 21]. Therefore additional analysis was performed comparing 16 patients with BMI > 30 with the remaining patients (BMI ≤ 30).
In the formulas for the LOA and PE, a t-statistic of 2.02 was used at the various time points (N = 50) and 1.97 for pooled data (N = 300). To evaluate trending ability, four-quadrant plot and polar plot methodology was applied to the change in CIBIO and CIPAC between the time points [22, 23]. Concerning polar plot analysis central zone data (<10% change) were excluded because they introduce statistical noise. Angular bias is defined as the mean polar angle to the 0° line. The radial LOA refer to the radial sector that contains 95% of the data points. Polar concordance represents the percentage data points that lie within ±30°. In case of good trending ability, most of the data points lie within this 30° sector [23, 24]. Trending ability of CIBIO was considered interchangeable with CIPAC if angular bias was between −5° and +5°, with radial LOA between −30° and +30°.
A P value < 0.05 was considered statistically significant and Bonferroni correction for multiple testing of the absolute differences at the six time points was applied (P < 0.05/6 measurements = 0.008).
Statistical analysis was carried out using SPSS software (SPSS Inc., Chicago, IL, USA) and Excel (Microsoft Corporation).
3. Results
Fifty patients undergoing cardiac surgery were included. The baseline characteristics are presented in Table 1. The hemodynamic variables and temperatures are presented in Table 2. CI varied between 1.00 and 6.75 (CIPAC) and 0.93 and 7.25 L min−1 (CIBIO). CIBIO and CIPAC were significantly different at each point except T6 (24 hours after surgery).
Table 1.
Patient and surgery characteristics.
| Mean (sd) | Range | |
|---|---|---|
| Age (years) | 64.2 (10.6) | 40 to 81 |
| Height (m) | 1.72 (0.1) | 1.55 to 1.94 |
| Weight (kg) | 83.3 (17.5) | 60 to 150 |
| BMI (kg/m2) | 28.2 (5.0) | 19.3 to 46.3 |
| n | ||
| Sex m/f | 35/15 | |
| CABG∗ | 31 | |
| AVR† | 10 | |
| MVR‡ | 2 | |
| CABG and AVR or MVR | 5 | |
| Other | 2 |
Table 2.
Haemodynamic data.
| T1 | T2 | T3 | T4 | T5 | T6 | |
|---|---|---|---|---|---|---|
| MAP | 71.7 (8.8) | 70.2 (10.0) | 70.9 (9.6) | 75.8 (12.0) | 74.7 (7.5) | 79.6 (11.4) |
| HR | 61 (11) | 68 (17) | 80 (11) | 80 (12) | 90 (14) | 86 (12) |
| CVP | 11 (4.6) | 9 (4.6) | 10 (4.0) | 7 (3.3) | 7 (4.6) | 7 (4.7) |
| CI Aesculon | 1.8 (0.5) | 1.9 (0.7) | 2.2 (0.8) | 2.2 (0.7) | 2.8 (1.1) | 2.6 (0.7) |
| CI Vigilance | 2.2 (0.7) | 2.3 (0.7) | 3.2 (0.7) | 2.6 (0.5) | 3.3 (0.9) | 2.9 (0.7) |
| Temp central | 36.1 (0.5) | 35.9 (0.5) | 36.3 (0.3) | 35.9 (0.5) | 37.7 (0.5) | 37.4 (0.5) |
| Temp peripheral | 30.9 (1.8) | 32.1 (1.6) | 33.3 (2.2) | 32.9 (1.7) | 34.7 (1.8) | 34.3 (1.9) |
Mean (sd). MAP: mean arterial pressure (mmHg), HR: heart rate (beats min−1), CVP: central venous pressure (mmHg), CI: cardiac index (liters min−1 m−2), and Temp: central and peripheral body temperature (°C). Time point 1 (T1) prior to surgery, after induction of anaesthesia; T2 prior to cannulation of the aorta; T3 10 minutes after protamine administration; T4 30 minutes after arrival at the ICU; T5 one hour after extubation at the ICU; T6 first postoperative day, 8:00 a.m. in the ICU ward.
Differences were present at open chest (T2, T3) and closed chest (T5, T6), respectively, indicating noninterchangeability between both techniques at both open and closed chest.
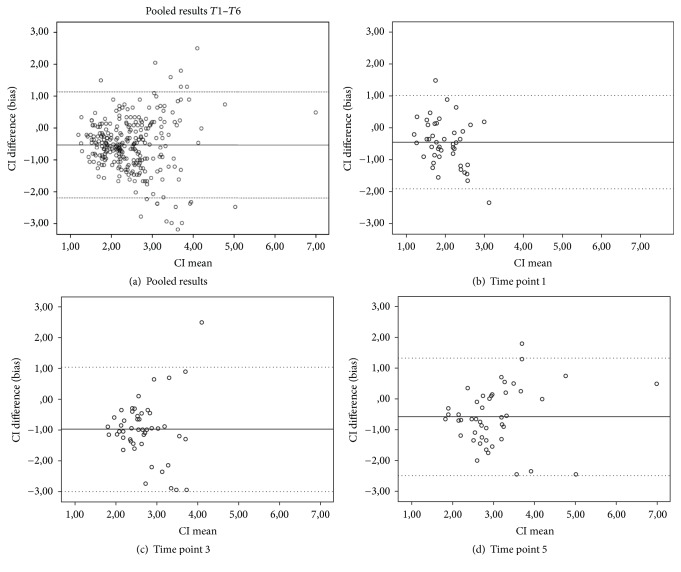
Bias between CIBIO and CIPAC was 0.52 liters min−1 m−2, with LOA of [−2.2; 1.1] liters min-1 m−2 (Table 3). Visual assessment of the Bland-Altman plots (Figure 1(a)) however shows that some agreement might be present at CI values between approximately 1.5 and 2.8 liters/min/m2. At higher CI values, the spread in the differences between CIBIO and CIPAC rapidly increases, especially at T1, T3, and T5 (Figures 1(a)–1(d)).
Table 3.
Agreement results.
| Pooled | T1 | T2 | T3 | T4 | T5 | T6 | |
|---|---|---|---|---|---|---|---|
| N | 284 | 47 | 50 | 48 | 46 | 46 | 47 |
| Bias (L min−1) | −0.52 | −0.44 | −0.38 | −0.98 | −0.45 | −0.57 | −0.28 |
| CI bias (L min−1) | −0.62 to −0.42 | −0.66 to −0.23 | −0.60 to −0.16 | −1.27 to −0.69 | −0.62 to −0.27 | −0.86 to −0.29 | −0.51 to −0.05 |
| LOA (L min−1) | −2.2 to 1.1 | −1.9 to 1.0 | −2.0 to 1.2 | −3.0 to 1.0 | −1.6 to 0.7 | −2.5 to 1.3 | −1.9 to 1.3 |
| CI lower LOA (L min−1) | −2.3 to −2.0 | −2.3 to −1.5 | −2.4 to −1.6 | −3.5 to −2.5 | −1.9 to −1.3 | −3.0 to −2.0 | −2.3 to −1.5 |
| CI upper LOA (L min−1) | 1.0 to 1.3 | 0.7 to 1.4 | 0.8 to 1.6 | 0.5 to 1.5 | 0.4 to 1.0 | 0.9 to 1.8 | 0.9 to 1.7 |
| Percentage error (95% CI) | 67 (60 to 73) | 74 (56 to 92) | 77 (58 to 96) | 75 (56 to 93) | 49 (37 to 62) | 63 (47 to 79) | 58 (43 to 72) |
Bias: difference between CI Aesculon and CI Vigilance. CI: 95% confidence interval. LOA: limits of agreement. Time point (T) 1 prior to surgery, after induction; T2 prior to cannulation of the aorta; T3 10 minutes after protamine administration; T4 30 minutes after arrival at the ICVU ward; T5 one hour after extubation at the ICU ward; T6 on day one postoperatively at 8:00 a.m. in the ICU ward.
Figure 1.
Bland and Altman plot. Fixed line indicates mean difference, and dotted lines indicate limits of agreement. (a) Pooled results, time point (T) 1 prior to surgery, after induction; T3 10 minutes after protamine administration; T5 one hour after extubation at the ICU ward.
The percentage error between CIBIO and CIPAC was above the 30% agreement limit at every time point, including the lower limit of the 95% confidence intervals (Table 3).
The percentage error in patients with BMI > 30 ranged from 30% to 62% versus 52% to 79% in the patients with BMI ≤ 30.
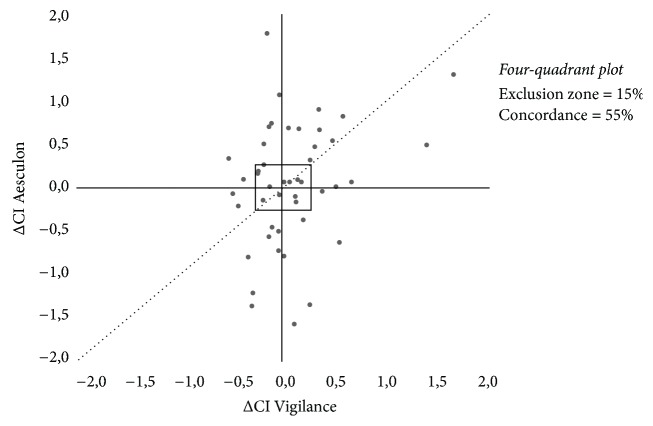
Trending ability was assessed in 89 pairs of changes in CI (Figure 2). Polar plot analysis in 77 data pairs outside the 10% exclusion zone showed an angular bias of −12°. The radial LOA were −55° to 51°. All values were outside the boundaries for acceptable trending ability. Polar concordance at 30° was 66%. These results were outside the boundaries for acceptable trending ability. Four-quadrant plot analysis also showed poor trending ability. Data pairs outside the 15% exclusion zone showed a concordance of only 55% (Figure 3).
Figure 2.
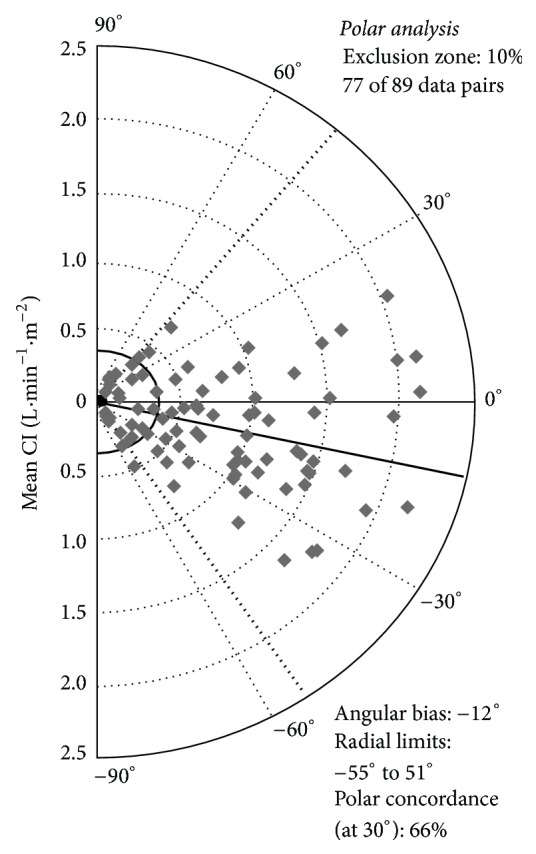
Polar plot analysis. In the polar plot, the distance from the centre represents the magnitude of the change in CI, whereas the angle with 0° line refers to its direction. Increases and decreases in CI are shown together in the so-called “half-moon” design. The mean change in CI (mean ΔCI) was used, with a 10% exclusion zone. Ideally, the mean angle of all data points is 0°, with at least 95% of all data points within the −30° to 30° sector (small dotted lines). Angular bias and radial LOA are depicted (solid line and thick dotted lines).
Figure 3.
Four-quadrant plot analysis. Assessment of trending ability for pooled data. In the four-quadrant plot, the change in CI Vigilance™ (x-axis) is plotted against the change in CI Aesculon (y-axis). Ideally, all data points lie along the line of identity “y 1/4 x” (dotted line). The central square refers to the 15% exclusion zone. The zero axes “y 1/4 0” and “x 1/4 0” cross in the centre of the plot, creating four quadrants. In the right upper and left lower quadrants, CI Aesculon and CI Vigilance agree, which means that CI Aesculon and CI Vigilance change in the same direction. The concordance refers to the percentage of data points in these quadrants.
4. Discussion
The present study investigated the accuracy, precision, and trending ability of a thoracic impedance CO monitor (Aesculon) versus pulmonary artery thermodilution in patients undergoing cardiac surgery. Our results do not support interchangeability of both devices in this patient group during surgery as well as during the early postoperative period, as both PE and trending ability exceeded the clinically acceptable, predefined limits.
Visual interpretation of the Bland-Altman plots revealed only moderate agreement at CI between 1.5 and 2.8 liters/min/m2. However, dispersion increased with higher CI. This so-called “heteroscedasticity” or proportional spread indicates that significant imprecision arises with increasing CO and that the LOA may even be underestimated in the high CO range. Several studies compared CO derived from bioimpedance with intermittent or continuous pulmonary artery thermodilution. The results of agreement and precision were inconclusive, demonstrating a need for additional studies in this field before drawing definitive conclusion on the validity and reliability of the Aesculon device technique [17, 25–27]. Patients undergoing cardiac surgery still represent a clinically challenging patient population, with relevant perioperative morbidity and mortality.
In comparable high-risk surgical patients, the use of invasive hemodynamic monitoring in combination with goal-directed therapy has been shown to improve postoperative outcome [28, 29]. In principle, thoracic bioimpedance represents a promising, noninvasive technique to be used in goal-directed strategies as the technique is noninvasive and independent from the observer and can be used also in patients at the ICU and medium care unit. Before implementation however, the reliability of the technology should be confirmed in the appropriate target patients. Apart from the clinical urgency, we decided to study patients undergoing cardiac surgery, because aortic cross clamping and clamp release induce profound changes in cardiac afterload followed by a profound ischemia-reperfusion injury. This implicates the necessity of profound observation and monitoring of these patients.
With the emergence of devices for continuous CO measurement, research increasingly focuses at trending ability rather than accuracy of individual measurements at a specific time point [30]. Especially during and immediately after cardiac surgery, the CO change in time may be more interesting than its absolute value, since the patients usually suffer from cardiovascular comorbidity and limited cardiac reserve [27, 28]. Therefore, 4-quadrant plot and polar plot methodology was used to objectify trending ability of the new technique. The results indicate poor trending ability, which was expected regarding the results from Bland and Altman analysis. Usually, high mean errors indicate that there is no fixed deviation between the experimental and reference technique, which impedes tracking of changes in CO in a reliable manner.
One of the strengths of the present study is the fact that most studies comparing the Aesculon with an established technique focus on neonatal, critically ill, and postoperative patient populations [10, 11, 15, 25–27].
Furthermore, there is lack of reliable published data on the trending abilities of the Aesculon [22]. In the present study, a relatively homogeneous population of cardiac surgical patients was studied during the intra- and postoperative period. As we were interested in the ability of the thoracic impedance under different conditions, we analyzed baseline values before surgery (T1) compared to open chest (T2, T3) and closed chest (T5, T6) conditions. However, the results demonstrated that the noninterchangeability between both techniques was present during both, open and closed thorax. In addition, we obtained CI measurements before and after weaning from mechanical ventilation and extubation up to 24 hours after surgery to simulate a medium care environment. We found the difference between both techniques independent from these conditions above the accepted range of 30%.
In our study population 16 patients had a BMI > 30. The percentage error was slightly lower in the BMI > 30 group but in both groups above the 30% limit. Clinical implication of these findings is questionable.
We included a substantial number of patients more than the calculated sample size to be sure we would have enough data. The concern was that we would have to deal with loss of data during observation at the ICU and possible failure of the Aesculon device. Accurate sampling at time points 5 and 6 also could be an expected problem.
Continuous pulmonary artery thermodilution was used as the reference technique. We used the mean of two measurements of continuous CO. Although thermodilution CO (TDCO) is considered as best acceptable clinical standard method for CO measurement, TDCO has an intrinsic variability, in particular during changing hemodynamic conditions and high CO [31]. Compared to the single bolus method, the continuous CO method has limited accuracy particularly during hemodynamic instability and hypothermia [31–33]. Being a combined measure of precision, the mean error may also be the result of variability of the reference technique [29, 34].
The pulmonary artery thermodilution catheter (PAC) has a precision of ±20% or less. The combination of two precisions of ±20% equates to a total error of ±28.3%, which is commonly rounded up to ±30% and is clinically acceptable [35, 36].
Moreover, the generally accepted range for bias (20%) and mean error (30%) are still a matter of discussion. The relatively large limit for bias was chosen to take possible hemodynamic changes into account. Depending on the clinical context, the limits can be defined more narrowly or even less narrowly. In general, it is advisable to use predefined criteria for acceptable bias and LOA in each method comparison study, since Bland and Altman analysis does not provide definitive answers. The same applies to the boundaries for trend parameters.
Our secondary aim was to assess whether the surgical incision, and therefore the interruption of the continuity of the skin of the thoracic cavity and opening of the cavity itself, could be an important factor in the reported discrepancy between the two instruments. Also, our study was not specifically designed to answer this question; we could not find any evidence for this hypothesis. The percentage error was always above 30%, being even above 70% in T1 and T3. Also the bias was very unpredictable, and the highest and the almost lowest value were measured during incision of the skin.
5. Conclusion
Analysis according to Bland-Altman shows that the Aesculon cannot be used interchangeably with the PAC in the operation theatre as a tool for beat to beat clinical decision making. We found no effect of skin interruption on accuracy, precision, and trending ability of the investigated technique.
Abbreviations
- PAC:
Pulmonary artery catheter
- EC:
Electrical cardiometry
- CO:
Cardiac output
- CI:
Cardiac index
- ICU:
Intensive care unit
- T:
Time point
- SPSS:
Statistical package for social science
- SD:
Standard deviation
- B-A:
Bland and Altman.
Additional Points
Key Messages. The bioimpedance cannot be used interchangeably with the PAC during cardiac surgery.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
P. B. W. Cox contributed to the design of the study, carried out the analyses, interpreted the data, and wrote the manuscript. M. Theunissen interpreted a part of the data and reviewed the manuscript. M. D. Lancé participated in its design and coordination. W. F. F. A. Buhre reviewed the manuscript critically. A. M. den Ouden helped in analyzing the data and contributed to the draft and improvement of the manuscript. M. A. E. Marcus contributed in initial design and reviewed the study. A. G. H. Kessels contributed in statistical analysis and data interpretation. L. J. Montenij advised substantially on statistical analysis and data interpretation.
References
- 1.Connors A. F., Jr., Speroff T., Dawson N. V., et al. The effectiveness of right heart catheterization in the initial care of critically ill patients. Journal of the American Medical Association. 1996;276(11):889–897. doi: 10.1001/jama.276.11.889. [DOI] [PubMed] [Google Scholar]
- 2.Rackow E. C. Pulmonary Artery Catheter Consensus Conference. Critical Care Medicine. 1997;25(6):p. 901. doi: 10.1097/00003246-199706000-00001. [DOI] [PubMed] [Google Scholar]
- 3.Afessa B., Spencer S., Khan W., LaGatta M., Bridges L., Freire A. X. Association of pulmonary artery catheter use with in-hospital mortality. Critical Care Medicine. 2001;29(6):1145–1148. doi: 10.1097/00003246-200106000-00010. [DOI] [PubMed] [Google Scholar]
- 4.Swan H. J. The pulmonary artery catheter in anesthesia practice. 1970. Anesthesiology. 2005;103(4):890–893. doi: 10.1097/00000542-200510000-00029. [DOI] [PubMed] [Google Scholar]
- 5.Rajaram S. S., Desai N. K., Kalra A., et al. Pulmonary artery catheters for adult patients in intensive care. Cochrane database of systematic reviews (Online) 2013;2:p. CD003408. doi: 10.1002/14651858.CD003408.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De Waal E. E. C., Wappler F., Buhre W. F. Cardiac output monitoring. Current Opinion in Anaesthesiology. 2009;22(1):71–77. doi: 10.1097/ACO.0b013e32831f44d0. [DOI] [PubMed] [Google Scholar]
- 7.Woltjer H. H., Bogaard H. J., De Vries P. M. J. M. The technique of impedance cardiography. European Heart Journal. 1997;18(9):1396–1403. doi: 10.1093/oxfordjournals.eurheartj.a015464. [DOI] [PubMed] [Google Scholar]
- 8.De Waal E. E. C., Konings M. K., Kalkman C. J., Buhre W. F. Assessment of stroke volume index with three different bioimpedance algorithms: Lack of agreement compared to thermodilution. Intensive Care Medicine. 2008;34(4):735–739. doi: 10.1007/s00134-007-0938-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bernstein D. O. Apparatus and method for determining an approximation of the stroke volume and the cardiac output of the heart. US patent 6,511,438 B2, 2003.
- 10.Rauch R., Welisch E., Lansdell N., et al. Non-invasive measurement of cardiac output in obese children and adolescents: Comparison of electrical cardiometry and transthoracic Doppler echocardiography. Journal of Clinical Monitoring and Computing. 2013;27(2):187–193. doi: 10.1007/s10877-012-9412-7. [DOI] [PubMed] [Google Scholar]
- 11.Wong J., Agus M. S. D., Steil G. M. Cardiac parameters in children recovered from acute illness as measured by electrical cardiometry and comparisons to the literature. Journal of Clinical Monitoring and Computing. 2013;27(1):81–91. doi: 10.1007/s10877-012-9401-x. [DOI] [PubMed] [Google Scholar]
- 12.Perrino A. C., Jr., Lippman A., Ariyan C., O'Connor T. Z., Luther M. Intraoperative cardiac output monitoring: Comparison of impedance cardiography and thermodilution. Journal of Cardiothoracic and Vascular Anesthesia. 1994;8(1):24–29. doi: 10.1016/1053-0770(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 13.Barin E., Haryadi D. G., Schookin S. I., et al. Evaluation of a thoracic bioimpedance cardiac output monitor during cardiac catheterization. Critical Care Medicine. 2000;28(3):698–702. doi: 10.1097/00003246-200003000-00016. [DOI] [PubMed] [Google Scholar]
- 14.Imhoff M., Lehner J. H., Lohlein D. Noninvasive whole-body electrical bioimpedance cardiac output and invasive thermodilution cardiac output in high-risk surgical patients. Critical Care Medicine. 2000;28(8):2812–2818. doi: 10.1097/00003246-200008000-00022. [DOI] [PubMed] [Google Scholar]
- 15.Zoremba N., Bickenbach J., Krauss B., Rossaint R., Kuhlen R., Schälte G. Comparison of electrical velocimetry and thermodilution techniques for the measurement of cardiac output. Acta Anaesthesiologica Scandinavica. 2007;51(10):1314–1319. doi: 10.1111/j.1399-6576.2007.01445.x. [DOI] [PubMed] [Google Scholar]
- 16.Huang L., Critchley L. A. H., Zhang J. Major upper abdominal surgery alters the calibration of BioReactance cardiac output readings, the NICOM, when comparisons are made against suprasternal and esophageal Doppler Intraoperatively. Anesthesia and Analgesia. 2015;121(4):936–945. doi: 10.1213/ANE.0000000000000889. [DOI] [PubMed] [Google Scholar]
- 17.Faini A., Omboni S., Tifrea M., Bubenek S., Lazar O., Parati G. Cardiac index assessment: Validation of a new non-invasive very low current thoracic bioimpedance device by thermodilution. Blood Pressure. 2014;23(2):102–108. doi: 10.3109/08037051.2013.817121. [DOI] [PubMed] [Google Scholar]
- 18.Van Der Meer B. J. M., De Vries J. P. P. M., Schreuder W. O., Bulder E. R., Eysman L., De Vries P. M. J. M. Impedance cardiography in cardiac surgery patients: Abnormal body weight gives unreliable cardiac output measurements. Acta Anaesthesiologica Scandinavica. 1997;41(6):708–712. doi: 10.1111/j.1399-6576.1997.tb04770.x. [DOI] [PubMed] [Google Scholar]
- 19.Bland J. M., Altman D. G. Statistical methods for assessing agreement between two methods of clinical measurement. The Lancet. 1986;1(8476):307–310. [PubMed] [Google Scholar]
- 20.Bland J. M., Altman D. G. Agreed statistics: measurement method comparison. Anesthesiology. 2012;116(1):182–185. doi: 10.1097/aln.0b013e31823d7784. [DOI] [PubMed] [Google Scholar]
- 21.Van Der Meer B. J. M., Woltjer H. H., Sousman A. M., et al. Impedance cardiography. Importance of the equation and the electrode configuration. Intensive Care Medicine. 1996;22(10):1120–1124. doi: 10.1007/s001340050224. doi: 10.1007/s001340050224. [DOI] [PubMed] [Google Scholar]
- 22.Critchley L. A., Lee A., Ho A. M.-H. A critical review of the ability of continuous cardiac output monitors to measure trends in cardiac output. Anesthesia and Analgesia. 2010;111(5):1180–1192. doi: 10.1213/ANE.0b013e3181f08a5b. [DOI] [PubMed] [Google Scholar]
- 23.Critchley L. A., Yang X. X., Lee A. Assessment of trending ability of cardiac output monitors by polar plot methodology. Journal of Cardiothoracic and Vascular Anesthesia. 2011;25(3):536–546. doi: 10.1053/j.jvca.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 24.Montenij L. J., Buhre W. F., De Jong S. A., et al. Arterial pressure waveform analysis versus thermodilution cardiac output measurement during open abdominal aortic aneurysm repair: A prospective observational study. European Journal of Anaesthesiology. 2015;32(1):13–19. doi: 10.1097/EJA.0000000000000160. [DOI] [PubMed] [Google Scholar]
- 25.Mekiš D., Kamenik M., Starc V., Jeretin S. Cardiac output measurements with electrical velocimetry in patients undergoing CABG surgery: A comparison with intermittent thermodilution. European Journal of Anaesthesiology. 2008;25(3):237–242. doi: 10.1017/S0265021507002669. [DOI] [PubMed] [Google Scholar]
- 26.Spiess B. D., Patel M. A., Soltow L. O., Wright I. H. Comparison of bioimpedance versus thermodilution cardiac output during cardiac surgery: Evaluation of a second-generation bioimpedance device. Journal of Cardiothoracic and Vascular Anesthesia. 2001;15(5):567–573. doi: 10.1053/jcan.2001.26533. [DOI] [PubMed] [Google Scholar]
- 27.Heringlake M., Handke U., Hanke T., et al. Lack of agreement between thermodilution and electrical velocimetry cardiac output measurements. Intensive Care Medicine. 2007;33(12):2168–2172. doi: 10.1007/s00134-007-0828-3. [DOI] [PubMed] [Google Scholar]
- 28.Pearse R., Dawson D., Fawcett J., Rhodes A., Grounds R. M., Bennett E. D. Early goal-directed therapy after major surgery reduces complications and duration of hospital stay. A randomised, controlled trial [ISRCTN38797445]. Critical care (London, England) 2005;9(6):R687–693. doi: 10.1186/cc3887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rhodes A., Cecconi M., Hamilton M., et al. Goal-directed therapy in high-risk surgical patients: A 15-year follow-up study. Intensive Care Medicine. 2010;36(8):1327–1332. doi: 10.1007/s00134-010-1869-6. [DOI] [PubMed] [Google Scholar]
- 30.Maass S. W. M. C., Roekaerts P. M. H. J., Lancé M. D. Cardiac output measurement by bioimpedance and noninvasive pulse contour analysis compared with the continuous pulmonary artery thermodilution technique. Journal of Cardiothoracic and Vascular Anesthesia. 2014;28(3):534–539. doi: 10.1053/j.jvca.2014.01.007. [DOI] [PubMed] [Google Scholar]
- 31.Jacquet L., Hanique G., Glorieux D., Matte P., Goenen M. Analysis of the accuracy of continuous thermodilution cardiac output measurement. Comparison with intermittent thermodilution and Fick cardiac output measurement. Intensive Care Medicine. 1996;22(10):1125–1129. doi: 10.1007/s001340050225. doi: 10.1007/s001340050225. [DOI] [PubMed] [Google Scholar]
- 32.Böttiger B. W., Rauch H., Böhrer H., et al. Continuous versus intermittent cardiac output measurement in cardiac surgical patients undergoing hypothermic cardiopulmonary bypass. Journal of Cardiothoracic and Vascular Anesthesia. 1995;9(4):405–411. doi: 10.1016/S1053-0770(05)80095-3. [DOI] [PubMed] [Google Scholar]
- 33.Ameloot K., Meex I., Genbrugge C., et al. Accuracy of continuous thermodilution cardiac output monitoring by pulmonary artery catheter during therapeutic hypothermia in post-cardiac arrest patients. Resuscitation. 2014;85(9):1263–1268. doi: 10.1016/j.resuscitation.2014.06.025. [DOI] [PubMed] [Google Scholar]
- 34.Ho J. T., Chapman M. J., O'Connor S., et al. Characteristics of plasma NOx levels in severe sepsis: High interindividual variability and correlation with illness severity, but lack of correlation with cortisol levels. Clinical Endocrinology. 2010;73(3):413–420. doi: 10.1111/j.1365-2265.2010.03817.x. [DOI] [PubMed] [Google Scholar]
- 35.Cecconi M., Rhodes A., Poloniecki J., Della Rocca G., Grounds R. M. Bench-to-bedside review: the importance of the precision of the reference technique in method comparison studies--with specific reference to the measurement of cardiac output. Critical care (London, England) 2009;13(1):p. 201. doi: 10.1186/cc7129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Critchley L. A. H., Critchley J. A. J. H. A meta-analysis of studies using bias and precision statistics to compare cardiac output measurement techniques. Journal of Clinical Monitoring and Computing. 1999;15(2):85–91. doi: 10.1023/A:1009982611386. [DOI] [PubMed] [Google Scholar]