Abstract
Chondrosarcoma is a malignant soft tissue sarcoma with poor prognosis. Puerarin has been demonstrated to possess anticancer properties; however, the effects of puerarin in human chondrosarcoma cells remain unknown. The present study aimed to investigate the anticancer effects of puerarin in SW1353 human chondrosarcoma cells. SW1353 cells were treated with increasing concentrations of puerarin for different durations. Cell viability was evaluated using MTT assays. Cell apoptosis rates were determined by flow cytometry. The activities of caspase-3 and caspase-9 were measured by enzymatic assay. The expression of RAC-alpha serine/threonine-protein kinase (Akt), phosphorylated-Akt, caspase-3 and apoptosis-associated proteins, including B-cell lymphoma 2 (Bcl-2) and Bcl-2-associated X protein (Bax) were detected by western blotting. Puerarin significantly decreased cell viability and significantly induced apoptosis of SW1353 cells. In addition, puerarin significantly increased the enzymatic activities of caspase-3 and caspase-9. Puerarin treatment suppressed the expression of p-Akt and Bcl-2 but promoted the expression of Bax and cleaved caspase-3 in SW1353 cells. Notably, the phosphatidylinositol 3-kinase (PI3K) inhibitor LY294002 abrogated the decreased phosphorylation of Akt, suggesting that the PI3K/Akt signaling pathway is involved in mediating the anticancer effects of puerarin. The data from the present study indicated that puerarin exhibits anticancer effects in SW1353 cells and may be a potential therapeutic drug for patients with chondrosarcoma.
Keywords: puerarin, chondrosarcoma, SW1353 cells, apoptosis, PI3K/Akt signaling pathway
Introduction
Chondrosarcoma is a malignant soft tissue sarcoma with poor prognosis, and it is the second most common primary bone tumor among all malignant bone tumors (1). At present, surgical resection remains the primary treatment option for chondrosarcoma (2). Due to its poor response to conventional chemotherapy and radiation therapy, chondrosarcoma is challenging to manage (3). Therefore, novel and effective therapeutic approaches are required for patients with chondrosarcoma.
Puerarin, the major active ingredient extracted from Radix puerariae, exhibits beneficial effects for cardiovascular diseases, neurological dysfunction, diabetes, osteoporosis, liver injury and inflammation (4). Previous studies have indicated that puerarin exhibits anticancer effects and induces tumor cell apoptosis in a number of cancer cell types, including colon cancer HT-29 cells, human mantle Z138 cells, human glioblastoma cells, stomach cancer cells and breast cancer cells (5–9). Furthermore, increasing evidence has indicated the mechanisms involved in the biological effect of puerarin. In hFBO1.19 cells, puerarin attenuated the dexamethasone (DEX)-induced release of cytochrome c and cleavage of caspase-3, which was associated with inhibition of the c-Jun N-terminal kinase (JNK) pathway and activation of the phosphoinositide 3-kinase (PI3K)/threonine-protein kinase (Akt) signaling pathway (10). Puerarin increases proliferation and differentiation and opposes cisplatin-induced apoptosis in human osteoblastic MG-63 cells. At least partially, puerarin functions via activation of MEK/extraellular signal-regulated kinase and PI3K/Akt signaling pathways (11). However, the effects of puerarin on the human chondrosarcoma cell line SW1353 and the molecular mechanisms underlying these effects remain unclear.
In the present study, puerarin decreased cell viability and induced apoptosis in SW1353 cells via inhibition of the P13K/RAC-alpha serine/Akt signaling pathway. These results elucidated the potential molecular mechanisms underlying the anticancer effect of puerarin in SW1353 cells, which may provide an insight into novel therapeutic options for patients with chondrosarcoma.
Materials and methods
Materials
The human chondrosarcoma cell line SW1353 was purchased from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). Puerarin, LY294002, PD98059, SP600125 and SB203580 were purchased from Sigma-Aldrich (Merk KGaA, Darmstadt, Germany). Rabbit anti-human antibodies against Bax (cat no. 2774), Bcl-2 (cat no. 2872), Caspase-3 (cat no. 9664), Akt (cat no. 9272), p-Akt (Ser473; cat no. 9271) and GAPDH (cat no. 2118) were obtained from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Cell culture
SW1353 cells were cultured in L-15 medium (Hyclone; GE Healthcare Life Sciences Logan, UT, USA) supplemented with 10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) and incubated at 37°C in a humidified atmosphere of 5% CO2.
MTT assay
SW1353 cells (1×104 cells/well) were treated with puerarin in 96-well plates (Costar; Corning Incorporated, Corning, NY, USA) for 48 h. Then, cultures were washed with PBS. MTT (Sigma-Aldrich; Merk KGaA) was added to each well and the mixture was incubated at 37°C for 2 h. To dissolve the formazan crystals, culture medium was then replaced with an equal volume of dimethyl sulphoxide (Merck KGaA). Following agitation at room temperature for 10 min, the absorbance of each well was determined at 570 nm using a microplate reader (Bio-Tek Instruments, Inc., Winooski, VT, USA). The inhibitory rate (%)=(optical density (OD)control group-ODtest group) × 100%/ODcontrol group.
Cell apoptosis by flow cytometry analysis
Apoptotic cells were measured with the Annexin V/fluorescein isothiocyanate (FITC) kit (BD Biosciences, Franklin Lakes, NJ, USA) according to the manufacturer's protocol. Briefly, 1×106 cells were collected, washed three times with PBS and were resuspended in 300 µl of binding buffer. A total of 5 µl of Annexin V-FITC solution and 5 µl of propidium iodide (PI) were added and incubated in the dark at 37°C for 15 min. Then, the cytofluorimetric analyses were performed on a BD FACSVerse (BD Biosciences) and data were analyzed using FACSuite software version 1.0 (BD Biosciences).
Enzymatic assay for caspase-3 and caspase-9 activity
Caspase activity was detected using caspase-3 and caspase-9 activity assay kits (Beyotime Institute of Biotechnology, Haimen, China) according to the manufacturer's protocol. Cells were lysed and total cellular protein extracts were quantified using a Protein-assay kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). An equal amount of total protein extract was incubated at 37°C overnight with either Ac-DEVD-pNA for the caspase-3 assay or Ac-LEHD-pNA for the caspase-9 assay. The release of pNA was estimated by determining the absorbance at 405 nm on a microplate ELISA reader (Bio-Rad Laboratories, Inc.). Results are presented with the fold change in activity compared with the untreated control.
Western blot analysis
Cells were collected, washed and lysed with radioimmunoprecipitation assay buffer, including 50 mM Tris-HCl (pH=7.4), 150 mM NaCl, 1% Nonidet P-40, 0.25% NaN3, 1 mM EDTA, 1 mM PMSF, 1 µg/ml aprotinin, 1 µg/ml leupeptin, 1 µg/ml pepstatin A, 1 mM sodium orthovanadate and 1 mM NaF. Then, the protein concentration was determined using a BCA Protein Assay kit (Thermo Fisher Scientific, Inc.). The proteins (20 µg) were resolved on a 10% SDS-PAGE gel and transferred to polyvinylidene fluoride membranes. The blots were blocked with 5% BSA (Sigma-Aldrich; Merck KGaA) for 1 h at room temperature, and probed with rabbit anti-human antibodies against Bax, Bcl-2, Caspas-3, Akt, p-Akt (Ser473) or GAPDH (1:1,000 dilution) for 1 h at room temperature. After washing three times with PBS, the blots were incubated with a horseradish peroxidase-conjugated donkey anti-rabbit secondary antibody (1:1,000 dilution; cat no. sc2313; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) for 1 h at room temperature. The signals were visualized by enhanced chemiluminescence (Pierce; Thermo Fisher Scientific, Inc.). Images were captured using a scanner (GE Healthcare, Chicago, IL, USA) and proteins were quantified using Image J software version 1.42 (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
Data were analyzed using SPSS statistical software version 13.0 (SPSS, Inc., Chicago, IL, USA) and the data were reported as the mean ± standard deviation. Statistical analysis between two samples was evaluated using a Student's t-test. P<0.05 was considered to indicate a statistically significant difference.
Results
Puerarin inhibits the viability of human chondrosarcoma SW1353 cells
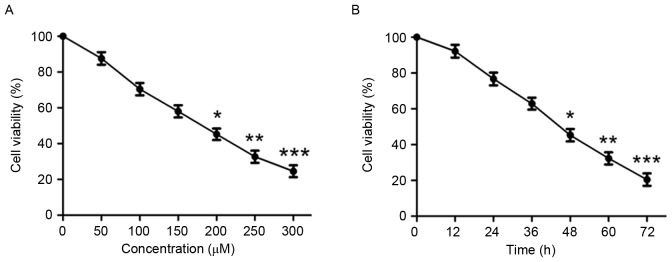
In order to investigate the effect of puerarin on cell viability, human chondrosarcoma SW1353 cells were treated with various concentrations of puerarin (0–300 µM) for 48 h. Puerarin significantly reduced cell viability in a dose-dependent manner (P<0.05, P<0.01 and P<0.001 compared with untreated control for 200, 250 and 300 µM, respectively; Fig. 1A and Table I). After 48 h incubation, the half-maximal inhibitory concentration value of puerarin against SW1353 cells was 180.738 µM. Treatment of SW1353 cells for different durations indicated that puerarin treatment significantly decreases cell viability in a time-dependent manner (P<0.05, P<0.01 and P<0.001 compared with untreated control for 48, 60 and 72 h, respectively; Fig. 1B).
Figure 1.
Anti-proliferative effect of puerarin on human chondrosarcoma SW1353 cells in vitro. (A) SW1353 cells were treated with various concentrations of puerarin (0–300 µM) for 48 h. (B) SW1353 cells were incubated with the indicated concentration of puerarin (200 µM) for various periods of time. Cell viability was determined by MTT assay. Data are presented as the mean ± standard deviation from four separate experiments. *P<0.05, **P<0.01 and ***P<0.001, comparison with the untreated control.
Table I.
Inhibitory rate of puerarin on the proliferation of cells.
| Puerarin (µM) | Optical density (570 nm) (mean ± standard deviation) | Inhibitory rate (%) |
|---|---|---|
| 0 | 0.995±0.004 | 0 |
| 50 | 0.886±0.012 | 10.254±0.671 |
| 100 | 0.724±0.013 | 26.536±0.582 |
| 150 | 0.618±0.018 | 37.089±0.622 |
| 200 | 0.475±0.014 | 51.461±0.706 |
| 250 | 0.362±0.016 | 62.718±0.654 |
| 300 | 0.254±0.012 | 73.672±0.636 |
Puerarin induces apoptosis in SW1353 cells
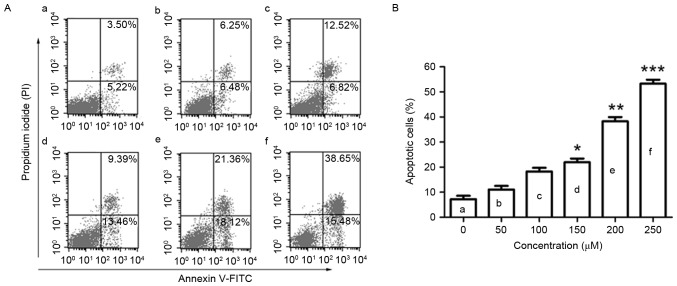
To observe the effect of puerarin on cell apoptosis, flow cytometric analysis was conducted. Puerarin treatment (48 h) significantly increased the rate of apoptosis of SW1353 cells in a dose-dependent manner (P<0.05, P<0.01 and P<0.001 compared with the untreated control for 150, 200 and 250 µM, respectively; Fig. 2A and B). Puerarin exposure increased the apoptosis rate of SW1353 cells to 38.28% with a dose of 200 µM.
Figure 2.
Flow cytometric analysis of apoptosis in SW1353 cells following puerarin treatment. (A) SW1353 cells were treated with different concentrations of puerarin [(a) 0 µM, (b) 50 µM, (c) 100 µM, (d) 150 µM, (e) 200 µM, (f) 250 µM) for 48 h. A flow cytometry assay was performed to determine the apoptosis rate, and apoptotic cells (Annexin V+PI− and Annexin V+PI+) are presented. (B) Percentage of apoptotic cells. Data are presented as the mean ± standard deviation (n=4). *P<0.05, **P<0.01 and ***P<0.001, compared with the untreated control. PI, propidium iodide; FITC, fluorescein isothiocyanate.
Puerarin accelerates enzymatic activity of caspase-3 and caspase-9
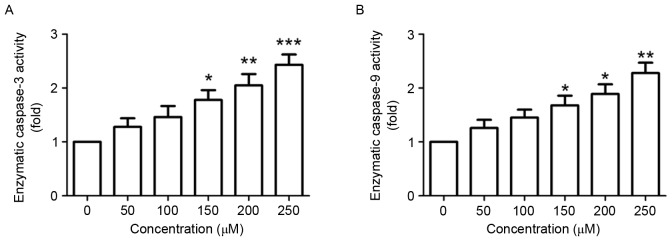
Next, the effect of puerarin on the enzymatic activity of caspase-3 and −9 was investigated. The enzymatic activity of caspase-3, an important marker of apoptosis (12), was significantly increased in SW1353 cells following puerarin stimulation in a dose-dependent manner (P<0.05, P<0.01 and P<0.001 compared with the untreated control for 150, 200 and 250 µM, respectively; Fig. 3A). Additionally, the enzymatic activity of caspase-9 in was significantly increased in a dose-dependent manner (P<0.05, P<0.05 and P<0.01 compared with the untreated control for 150, 200 and 250 µM, respectively; Fig. 3B).
Figure 3.
Effect of puerarin on the enzymatic activity of caspase-3 and caspase-9. SW1353 cells were treated with different concentrations of puerarin (0–300 µM) for 48 h. Enzymatic assay for (A) caspase-3 activity and (B) caspase-9 activity was analyzed. Data are presented as the mean ± standard deviation (n=4). *P<0.05, **P<0.01 and ***P<0.001, comparison with the untreated control.
Effect of puerarin on the expression of apoptosis-associated protein in SW1353 cells
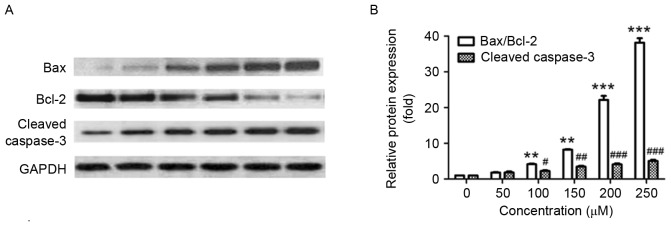
Next, the effect of puerarin on the expression of apoptosis-associated proteins was investigated. In line with the Annexin V-FITC/PI staining results, puerarin treatment significantly increased the ratio of Bax/Bcl-2 compared with the untreated control (P<0.01, P<0.01, P<0.001 and P<0.001 for 100, 150, 200 and 250 µM, respectively; Fig. 4A and B). In addition, exposure of SW1353 cells to puerarin significantly increased the expression of cleaved caspase-3 in a dose-dependent manner compared with the untreated control (P<0.05, P<0.01, P<0.001 and P<0.001 for 100, 150, 200 and 250 µM, respectively; Fig. 4A and B). The Bax/Bcl2 ratio and expression of cleaved caspase-3 increased ~20 and ~4-fold following treatment with 200 µM puerarin, respectively (Fig. 4B).
Figure 4.
Puerarin regulates the expression of apoptosis-associated proteins in SW1353 cells. (A) Protein expression of Bax, Bcl-2 and cleaved caspase-3 in SW1353 cells treated with different concentrations of puerarin (0–300 µM) for 48 h. (B) The relative protein expression was quantified and assessed. Data are presented as the mean ± standard deviation (n=4). **P<0.05 and ***P<0.001 compared with the untreated control; #P<0.05, ##P<0.01 and ###P<0.001, compared with the untreated control. Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X protein.
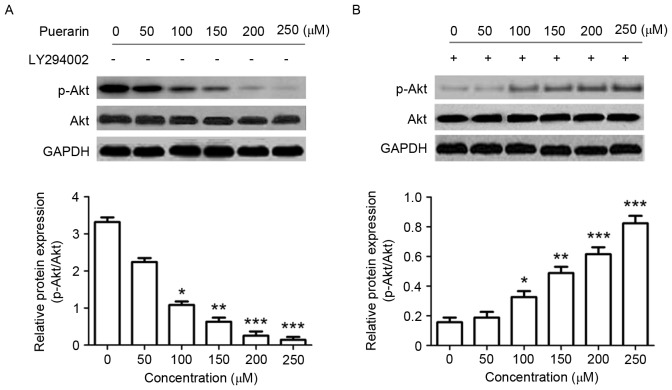
Puerarin suppresses the P13K/Akt signaling pathway in SW1353 cells
Changes in the P13K/Akt signaling pathway were analyzed by western blotting in order to determine the underlying molecular mechanisms of the effect of puerarin on SW1353 cells. The expression level of p-Akt (Ser473), in addition to the ratio of p-Akt/Akt was significantly decreased in response to puerarin treatment compared with the untreated control (P<0.05, P<0.01, P<0.001 and P<0.001 for 100, 150, 200, 250 µM, respectively; Fig. 5A); however, the P13K inhibitor LY294002 significantly abrogated this effect (Fig. 5B).
Figure 5.
Effect of puerarin on the expression of p-Akt (Ser473) in SW1353 cells. SW1353 cells were treated with different concentrations of puerarin (0–300 µM) for 48 h. Protein expression of p-Akt and Akt were detected in the (A) presence or (B) absence of the phosphoinositide 3-kinase inhibitor LY294002. The relative protein expression was quantified and assessed, respectively. A representative blot is presented for each experiment. Data are presented as the mean ± standard deviation (n=4). *P<0.05, **P<0.01 and ***P<0.001, compared with the untreated control. Akt, RAC-alpha serine/threonine-protein kinase; p, phosphorylated.
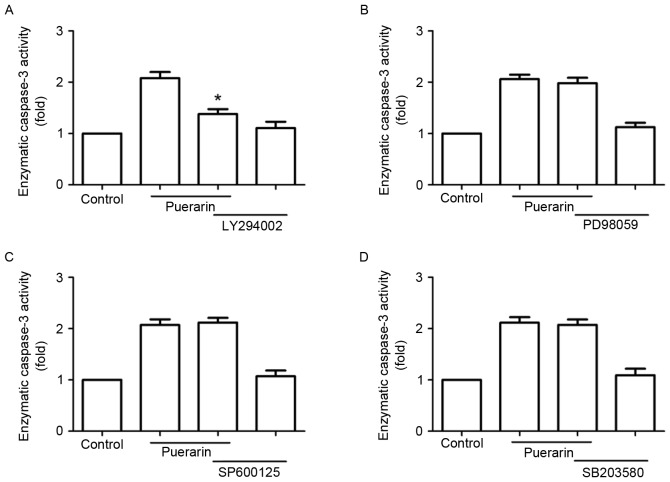
Next, SW1353 cells were treated with different signaling inhibitors, including LY294002, PD98059, SP600125 and SB203580 prior to treatment with puerarin. Notably, LY294002 abrogated the enzymatic activity of caspase-3 promoted by puerarin, but not the extracellular signal-regulated kinase inhibitor (PD98059), p38 mitogen-activated protein kinase inhibitor (SB203580) or c-Jun N-terminal kinase inhibitor (SP600125) (Fig. 6).
Figure 6.
The phosphoinositide 3-kinase inhibitor LY294002 abolishes puerarin-induced apoptosis in SW1353 cells. SW1353 cells were pretreated with different inhibitors including (A) LY294002, (B) PD98059, (C) SP600125 and (D) SB203580 for 1 h prior to puerarin treatment (200 µM), respectively. Following 48 h incubation, the enzymatic assay for caspase-3 activity was analyzed. Data are presented as the mean ± standard deviation (n=4). *P<0.05, compared with the puerarin-only-treated group.
Discussion
Puerarin exhibits a variety of biological effects, including anti-oxidant, anti-inflammatory and anti-apoptotic activities (13). In the present study, the antitumor activity of puerarin against human chondrosarcoma was investigated, in addition to its possible mechanisms of action, using SW1353 cells. The results indicated that puerarin treatment induced apoptosis in SW1353 cells via inhibition of the P13K/Akt signaling pathway.
Previous studies have demonstrated that puerarin exerts protective biological effects by suppressing cell apoptosis; puerarin treatment blocked cell apoptosis induced by H2O2 in PC12 cells, which was associated with a reduction in c-Myc expression, an increase in the Bcl-2/Bax ratio and the inhibition of caspase-3 activation (14). Another study reported that puerarin exhibited a protective effect against the lipopolysaccharide (LPS)-induced apoptosis of H9c2 cardiomyocytes by reversing LPS-induced downregulation of Bax and upregulation of Bcl-2 (15). In addition, puerarin afforded protection against beta-amyloid-induced neurotoxicity by inhibiting apoptosis in PC12 cells, which contributed to activation of the PI3K-dependent signaling pathway in association with Akt phosphorylation (16). Although anti-apoptotic mechanisms have been demonstrated to be involved in the protective effect of puerarin in the nervous and cardiovascular system (17,18), in the present study, puerarin demonstrated antitumor effects by reducing cell viability and inducing cell apoptosis in SW1353 cells, which may be important for the clinical treatment of patients with cancer.
In colon cancer HT-29 cells, puerarin treatment regulated the expression of apoptosis-associated proteins, including Bax, Bcl-2 and caspase-3 (5). A previous study demonstrated that the antitumor activity of puerarin 6′-O-xyloside on the human lung carcinoma A549 cell line, which was one of the major isoflavones of Porites lobata. The mechanisms were associated with increased levels of caspase-3, caspase-7, caspase-9 and Bax, and decreased levels of Bcl-2 (19). Combined with or without 5-fluorouracil, puerarin induced significant proliferation suppression and apoptosis in Eca-109 esophageal cancer cells in vitro and in vivo (20). In addition, puerarin inhibited proliferation and induced apoptosis in U251 and U87 human glioblastoma cell lines, and cell cycle arrest and DNA damage may participate in the anticancer effects of puerarin (7). In SMMC-7721 hepatocellular carcinoma cells, puerarin suppressed cell viability and induced cell apoptosis (21). In human breast cancer cell lines HS578T, MDA-MB-231 and MCF-7, Puerariae radix isoflavones and their metabolites reduced cell growth and induced cell apoptosis, suggesting that puerariae may act as a chemotherapeutic agent against breast cancer (9). In keeping with previous reports, the present study demonstrated that puerarin significantly inhibits the proliferation of SW1353 cells in a time- and dose-dependent manner. Additionally, the percentage of apoptotic cells and expression of apoptosis-associated proteins were increased following puerarin treatment, suggesting that induction of apoptosis may be the primary mechanism of the anti-proliferative effect of puerarin in SW1353 cells.
Apoptosis, also known as programmed cell death, is characterized by two classical pathways: The death receptor pathway and the mitochondrial pathway (22). Caspase-9 is the initiating protein in the caspase cascade, and caspase-3, a biomarker for cell apoptosis, is activated by caspase-9 (23). In addition, Bax and Bcl-2 are involved in the apoptotic process as pro-apoptotic protein and anti-apoptotic proteins, respectively (24). The results of the present study confirmed that apoptosis-associated mechanisms participated in the suppressive effect of puerarin. In the present study, puerarin significantly elevated the enzymatic activity of caspase-3 and caspase-9. Furthermore, puerarin treatment increased the Bax/Bcl-2 ratio and promoted the expression of caspase-3 in SW1353 cells.
The molecular mechanisms underlying the effects of puerarin pharmacological activities are complex, and different studies led to different conclusions. A previous study demonstrated that puerarin protected pancreatic β-cell survival via the P13K/Akt signaling pathway (25). Neuroprotective effects of puerarin against beta-amyloid-induced neurotoxicity in PC12 cells were associated with activation of the PI3K-dependent signaling pathway (26). Puerarin retarded the progression of cardiac hypertrophy and apoptosis, which may be mediated by blockade of the P13K/Akt and JNK signaling pathways (27). Conversely, puerarin demonstrated anti-proliferative and pro-apoptotic effects by inhibiting the P13K/Akt/nuclear factor κ-B cells pathway in human mantle cell lines Z138 (6). In line with this report, the results from the present study indicated that puerarin inhibits activation of p-Akt, which was abrogated by the P13K inhibitor LY294002, suggesting that the P13K/Akt signaling pathway is involved in the anticancer effect of puerarin. These differences observed in the effects of puerarin may be attributed to different doses and treatment times of puerarin used in different animal and cell models. Another reason may be that these signaling pathways connect with each other to form networks (28), therefore they may be influenced by each other.
In conclusion, the present study demonstrated that puerarin treatment induced apoptosis in SW1353 cells via inhibition of the P13K/Akt signaling pathway, which resulted in increased expression of caspase-3 and a decreased Bcl-2/Bax ratio. These findings suggested that puerarin may be a potential option as a therapeutic drug for patients with chondrosarcoma.
References
- 1.Leddy LR, Holmes RE. Chondrosarcoma of bone. Cancer Treat Res. 2014;162:117–130. doi: 10.1007/978-3-319-07323-1_6. [DOI] [PubMed] [Google Scholar]
- 2.Outani H, Hamada K, Imura Y, Oshima K, Sotobori T, Demizu Y, Kakunaga S, Joyama S, Imai R, Okimoto T, et al. Comparison of clinical and functional outcome between surgical treatment and carbon ion radiotherapy for pelvic chondrosarcoma. Int J Clin Oncol. 2016;21:186–193. doi: 10.1007/s10147-015-0870-z. [DOI] [PubMed] [Google Scholar]
- 3.Isakoff MS, Bielack SS, Meltzer P, Gorlick R. Osteosarcoma: Current treatment and a collaborative pathway to success. J Clin Oncol. 2015;33:3029–3035. doi: 10.1200/JCO.2014.59.4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Maji AK, Pandit S, Banerji P, Banerjee D. Pueraria tuberosa: A review on its phytochemical and therapeutic potential. Nat Prod Res. 2014;28:2111–2127. doi: 10.1080/14786419.2014.928291. [DOI] [PubMed] [Google Scholar]
- 5.Yu Z, Li W. Induction of apoptosis by puerarin in colon cancer HT-29 cells. Cancer Lett. 2006;238:53–60. doi: 10.1016/j.canlet.2005.06.022. [DOI] [PubMed] [Google Scholar]
- 6.Gan M, Yin X. Puerarin induced in mantle cell lymphoma apoptosis and its possible mechanisms involving multi-signaling pathway. Cell Biochem Biophys. 2015;71:367–373. doi: 10.1007/s12013-014-0207-y. [DOI] [PubMed] [Google Scholar]
- 7.Yang JA, Li JQ, Shao LM, Yang Q, Liu BH, Wu TF, Wu P, Yi W, Chen QX. Puerarin inhibits proliferation and induces apoptosis in human glioblastoma cell lines. Int J Clin Exp Med. 2015;8:10132–10142. [PMC free article] [PubMed] [Google Scholar]
- 8.Yanagihara K, Ito A, Toge T, Numoto M. Antiproliferative effects of isoflavones on human cancer cell lines established from the gastrointestinal tract. Cancer Res. 1993;53:5815–5821. [PubMed] [Google Scholar]
- 9.Lin YJ, Hou YC, Lin CH, Hsu YA, Sheu JJ, Lai CH, Chen BH, Lee Chao PD, Wan L, Tsai FJ. Puerariae radix isoflavones and their metabolites inhibit growth and induce apoptosis in breast cancer cells. Biochem Biophys Res Commun. 2009;378:683–688. doi: 10.1016/j.bbrc.2008.10.178. [DOI] [PubMed] [Google Scholar]
- 10.Yu D, Mu S, Zhao D, Wang G, Chen Z, Ren H, Fu Q. Puerarin attenuates glucocorticoid-induced apoptosis of hFOB1.19 cells through the JNK- and Akt-mediated mitochondrial apoptotic pathways. Int J Mol Med. 2015;36:345–354. doi: 10.3892/ijmm.2015.2258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang Y, Wang WL, Xie WL, Li LZ, Sun J, Sun WJ, Gong HY. Puerarin stimulates proliferation and differentiation and protects against cell death in human osteoblastic MG-63 cells via ER-dependent MEK/ERK and PI3K/Akt activation. Phytomedicine. 2013;20:787–796. doi: 10.1016/j.phymed.2013.03.005. [DOI] [PubMed] [Google Scholar]
- 12.Konstantinidou AE, Givalos N, Gakiopoulou H, Korkolopoulou P, Kotsiakis X, Boviatsis E, Agrogiannis G, Mahera H, Patsouris E. Caspase-3 immunohistochemical expression is a marker of apoptosis, increased grade and early recurrence in intracranial meningiomas. Apoptosis. 2007;12:695–705. doi: 10.1007/s10495-006-0001-4. [DOI] [PubMed] [Google Scholar]
- 13.Zhou YX, Zhang H, Peng C. Puerarin: A review of pharmacological effects. Phytother Res. 2014;28:961–975. doi: 10.1002/ptr.5083. [DOI] [PubMed] [Google Scholar]
- 14.Jiang B, Liu JH, Bao YM, An LJ. Hydrogen peroxide-induced apoptosis in pc12 cells and the protective effect of puerarin. Cell Biol Int. 2003;27:1025–1031. doi: 10.1016/j.cellbi.2003.09.007. [DOI] [PubMed] [Google Scholar]
- 15.Yuan Y, Zhou H, Wu QQ, Li FF, Bian ZY, Deng W, Zhou MQ, Tang QZ. Puerarin attenuates the inflammatory response and apoptosis in LPS-stimulated cardiomyocytes. Exp Ther Med. 2016;11:415–420. doi: 10.3892/etm.2015.2910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang HY, Liu YH, Wang HQ, Xu JH, Hu HT. Puerarin protects PC12 cells against beta-amyloid-induced cell injury. Cell Biol Int. 2008;32:1230–1237. doi: 10.1016/j.cellbi.2008.07.006. [DOI] [PubMed] [Google Scholar]
- 17.Wang G, Zhou L, Zhang Y, Dong M, Li X, Liu J, Niu Y. Implication of the c-Jun-NH2-terminal kinase pathway in the neuroprotective effect of puerarin against 1-methyl-4-phenylpyridinium (MPP+)-induced apoptosis in PC-12 cells. Neurosci Lett. 2011;487:88–93. doi: 10.1016/j.neulet.2011.02.057. [DOI] [PubMed] [Google Scholar]
- 18.Yuan Y, Zong J, Zhou H, Bian ZY, Deng W, Dai J, Gan HW, Yang Z, Li H, Tang QZ. Puerarin attenuates pressure overload-induced cardiac hypertrophy. J Cardiol. 2014;63:73–81. doi: 10.1016/j.jjcc.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 19.Chen T, Chen H, Wang Y, Zhang J. In vitro and in vivo antitumour activities of puerarin 6′-O-xyloside on human lung carcinoma A549 cell line via the induction of the mitochondria-mediated apoptosis pathway. Pharm Biol. 2016;54:1793–1799. doi: 10.3109/13880209.2015.1127980. [DOI] [PubMed] [Google Scholar]
- 20.Wang J, Yang ZR, Guo XF, Song J, Zhang JX, Wang J, Dong WG. Synergistic effects of puerarin combined with 5-fluorouracil on esophageal cancer. Mol Med Rep. 2014;10:2535–2541. doi: 10.3892/mmr.2014.2539. [DOI] [PubMed] [Google Scholar]
- 21.Zhang WG, Liu XF, Meng KW, Hu SY. Puerarin inhibits growth and induces apoptosis in SMMC-7721 hepatocellular carcinoma cells. Mol Med Rep. 2014;10:2752–2758. doi: 10.3892/mmr.2014.2512. [DOI] [PubMed] [Google Scholar]
- 22.Ouyang L, Shi Z, Zhao S, Wang FT, Zhou TT, Liu B, Bao JK. Programmed cell death pathways in cancer: A review of apoptosis, autophagy and programmed necrosis. Cell Prolif. 2012;45:487–498. doi: 10.1111/j.1365-2184.2012.00845.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blanc C, Deveraux QL, Krajewski S, Jänicke RU, Porter AG, Reed JC, Jaggi R, Marti A. Caspase-3 is essential for procaspase-9 processing and cisplatin-induced apoptosis of MCF-7 breast cancer cells. Cancer Res. 2000;60:4386–4390. [PubMed] [Google Scholar]
- 24.Zheng TS, Hunot S, Kuida K, Momoi T, Srinivasan A, Nicholson DW, Lazebnik Y, Flavell RA. Deficiency in caspase-9 or caspase-3 induces compensatory caspase activation. Nat Med. 2000;6:1241–1247. doi: 10.1038/81343. [DOI] [PubMed] [Google Scholar]
- 25.Li Z, Shangguan Z, Liu Y, Wang J, Li X, Yang S, Liu S. Puerarin protects pancreatic β-cell survival via PI3K/Akt signaling pathway. J Mol Endocrinol. 2014;53:71–79. doi: 10.1530/JME-13-0302. [DOI] [PubMed] [Google Scholar]
- 26.Xing G, Dong M, Li X, Zou Y, Fan L, Wang X, Cai D, Li C, Zhou L, Liu J, Niu Y. Neuroprotective effects of puerarin against beta-amyloid-induced neurotoxicity in PC12 cells via a PI3K-dependent signaling pathway. Brain Res Bull. 2011;85:212–218. doi: 10.1016/j.brainresbull.2011.03.024. [DOI] [PubMed] [Google Scholar]
- 27.Yuan Y, Zong J, Zhou H, Bian ZY, Deng W, Dai J, Gan HW, Yang Z, Li H, Tang QZ. Puerarin attenuates pressure overload-induced cardiac hypertrophy. J Cardiol. 2014;63:73–81. doi: 10.1016/j.jjcc.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 28.Lavrik IN. Systems biology of apoptosis signaling networks. Curr Opin Biotechnol. 2010;21:551–555. doi: 10.1016/j.copbio.2010.07.001. [DOI] [PubMed] [Google Scholar]