Abstract
Oligodendrocytes are well known as myelin-forming cells in the central nervous system (CNS). However, detailed mechanisms of oligodendrocyte differentiation and myelination are poorly understood, particularly due to the difficulty of the purification of murine oligodendrocyte precursor cells (OPCs). We have recently established a transgenic mouse line that expresses a fluorescent protein Venus under the promoter of Sox10, whose expression is restricted to OPCs and oligodendrocytes in the CNS. Here, we have characterized Venus-positive cells from the Sox10-Venus mouse brain for analyzing oligodendrocyte differentiation. We first purified Venus-positive cells from the postnatal day 0–2 brain by flow cytometry. Most of the Venus-positive cells expressed NG2, an OPC marker. After induction of differentiation, an increased population of galactocerebroside-positive oligodendrocytes and decrease of OPCs were observed in the Venus-positive culture. Furthermore, a time-lapse analysis showed that Venus-positive oligodendrocytes dynamically changed their morphology with highly branched cell processes during differentiation. In addition, we found that Venus-positive OPCs were able to differentiate to type II astrocytes. In vivo, OPCs and oligodendrocytes express Venus, and some of astrocytes were positive for Venus in the ventral cortex. Taken together, the Sox10-Venus mouse system is useful for analyzing differentiation and multipotency of murine OPCs.
Introduction
Oligodendrocyte precursor cells (OPCs; also known as oligodendrocyte progenitor cells, polydendrocytes, or O-2A cells) are glial cells that possess the potential to give rise to oligodendrocytes in the central nervous system (CNS)1,2. OPCs generated from glial precursor cells differentiate into myelinating oligodendrocytes, which form consecutive layers of membrane surrounding the neuronal axons facilitating the propagation of the action potentials. During development, oligodendrocyte lineage cells undergo significant morphological changes by forming elaborated branching cell processes. OPCs differentiate into oligodendrocytes not only during the development of the CNS but also in cases of pathological demyelination in the mature CNS1. Since oligodendrocytes play a crucial role in dysmyelinating diseases, such as leukodystrophy and multiple sclerosis, the study of oligodendrocytes and their differentiation mechanisms is essential for the development of novel therapies.
Previous studies have shown that OPCs can differentiate into not only oligodendrocytes but also astrocytes and neurons, depending on the area of the brain and stages during CNS development. However, the majority of the OPC population differentiates to oligodendrocytes3. In vitro, OPCs differentiate to oligodendrocytes under serum-free culture conditions, while their differentiation can be directed towards astrocyte-lineage cells under high-serum culture conditions4. Classically, these astrocytic cells derived from OPCs are called type II astrocytes, which are distinguished from type I astrocytes derived from glial precursor cells4. However, the functional differences between type I and II astrocytes in physiological and/or pathological conditions have not yet been elucidated. Therefore, specific labeling/tracing of both OPCs and OPC-lineage cells, and purification of these cells for in vitro studies, are essential tools to unravel the disease and injury mechanisms in the CNS.
Although isolation of rat OPCs from the CNS has been previously established, it is still less efficient to obtain sufficient quantity and purity of mouse OPCs5. One of the reasons for the difference between these two species is the distinct expression pattern of cell surface markers. The monoclonal antibody A2B5, whose antigen is a ganglioside, is widely used for purification of rat OPCs. However, mouse OPCs can not be efficiently purified by this antibody, since the expression level of the ganglioside in mice is lower than that in rats6,7. Therefore, for mouse OPC purification, neural/glial antigen 2 (NG2), instead of A2B5, is useful as a cell surface marker8. However, NG2 is expressed in not only OPCs but also in pericytes adherent to capillaries9. Another marker PDGFRα is available for immunopanning of OPCs from mouse cortices10. This is a useful and established method, but the possibility exists that in general, antibodies used for sorting may affect the cells during culture or analysis11. This problem can be overcome by using a fluorescent protein expression system under an OPC/oligodendrocyte-specific promoter. Several transgenic mouse lines that express a fluorescence protein DsRed or GFP under the regulation of OPC genes Cspg4 and Pdgfra, respectively, have been developed to analyze the fate of oligodendrocyte lineage cells12–14. In this study, we introduce a purification and culture method of OPCs from the transgenic mouse line Sox10-Venus, in which the intense fluorescence protein Venus is expressed under the promoter of the Sox10 gene15. Sox10, a high-mobility-group transcriptional regulator, is required for myelin gene expression16. In the CNS, Sox10 expression is elevated during development of glial precursor cells into OPCs, and its expression is persistent throughout oligodendrocyte differentiation and maturation16. Also, the fluorescence of Venus is more intense than that of DsRed and GFP17, and may be useful for the OPC differentiation analysis, particularly for the analysis of process formation during the differentiation. We have investigated the oligodendrocyte differentiation by following the cell fate of Sox10-Venus positive cells. We here propose that Sox10-Venus mice could be a useful tool for purifying and analyzing oligodendrocyte lineage cells.
Results
Antigenic phenotype of Sox10-Venus positive cells after purification using flow cytometer
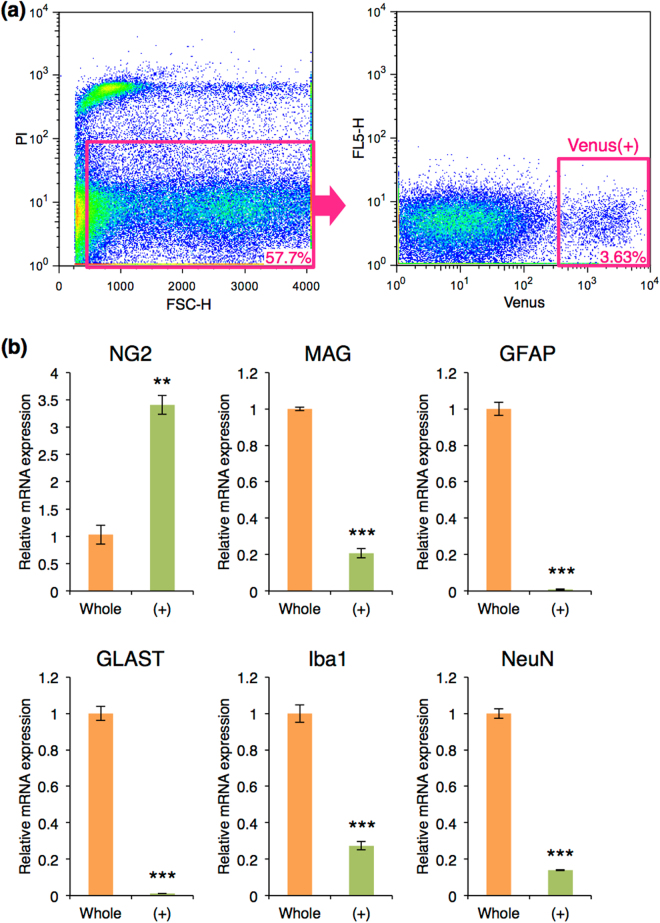
In order to analyze whether Venus-positive cells are OPCs and their differentiation, we used Sox10-Venus mouse brains at postnatal day (P) 0–2, when OPCs are enriched but not yet differentiated to oligodendrocytes, for isolation of Venus-positive cells by flow cytometer. Propidium iodide-positive cells and cell debris were removed (Fig. 1a: left panel). Cell populations showing high Venus intensity [Venus (+)] were collected (Fig. 1a: right panel). Quantitative RT-PCR using cells after the cell sorting revealed that Venus (+) cells highly expressed an OPC marker NG2, but not markers for oligodendrocytes: myelin associated glycoprotein (MAG), astrocytes: glial fibrillary acidic protein (GFAP) and glutamate/aspartate transporter (GLAST), microglia: ionized calcium-binding adapter molecule 1 (Iba1), or neurons: neuronal nuclei (NeuN), compared with control whole cells without cell sorting (Fig. 1b).
Figure 1.
Cell sorting of Venus (+) cells using flow cytometry. (a) Flow cytometry analysis of Venus (+) cells. Cells positive for propidium iodide were removed as dead cells (pink box in the left panel), and cells with the strong intensity of Venus were collected as Venus (+) cells (pink box in the right panel). The numbers in pink indicates the fraction of each cell population in the flow cytometry. (b) Quantitative RT-PCR of glial and neural markers in whole brain cells before cell sorting and Venus (+) cells. The mRNA expression of each gene was normalized using β-actin mRNA expression. The normalized expression of each gene in whole brain cells was set as 1.0. Whole: whole brain cells before cell sorting, orange bar; (+): Venus (+) cells, green bar; Error bars, s.e.m. (**p < 0.01, ***p < 0.001, t test).
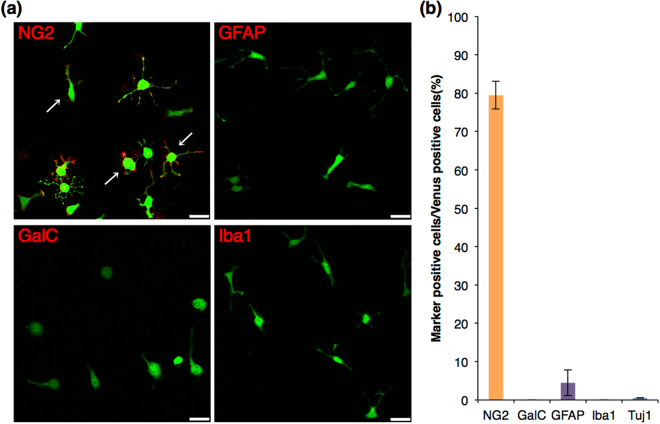
To determine the morphology and characteristics of Venus (+) cells, cells were cultured for 1 day in Proliferation medium. Most of the Venus (+) cells had round cell body with several primary processes, which resemble the typical morphology of OPCs in culture (Fig. 2a: arrows), showing immunoreactivity for NG2 on cytomembrane (Fig. 2a). Most of the Venus (+) cells were positive for NG2 (79 ± 3.6%), and a small population of GFAP-positive cells was observed (4.5 ± 3.4%) (Fig. 2b). Other cell-types, such as galactoceramide (GalC)-positive oligodendrocytes, Iba1-positive microglia, and Tuj1-positive neurons, were not present (Fig. 2b). In addition, Venus (+) cells were detectable by either anti-PDGFRα antibody or A2B5 antibody (Supplementary Figure S1a). Furthermore, most of the Venus (+) cells were positive for Ki67 and/or BrdU (Supplementary Figure S2), suggesting that Venus (+) cells under this condition are proliferative, which is one of the characteristics of OPCs. These results indicated that OPCs were enriched in the Venus (+) population. These observations showed that OPCs can be sorted by the intensity of the Venus fluorescence from the Sox10-Venus mouse brain at the neonatal stage.
Figure 2.
Immunocytochemistry of glial and neuronal markers in Venus (+) cells before induction of differentiation. (a) Confocal images of the immunocytochemistry before induction of differentiation. Venus fluorescence is shown in green, and each glial marker is shown in red. Scale bar, 25 µm. Venus (+) cells expressed NG2, showing typical cell morphology of OPCs in culture (arrows). (b) Cell count analysis showing cell population of Venus (+) cells expressing each marker. Error bars, s.e.m.
Differentiation of Sox10-Venus positive cells into oligodendrocytes
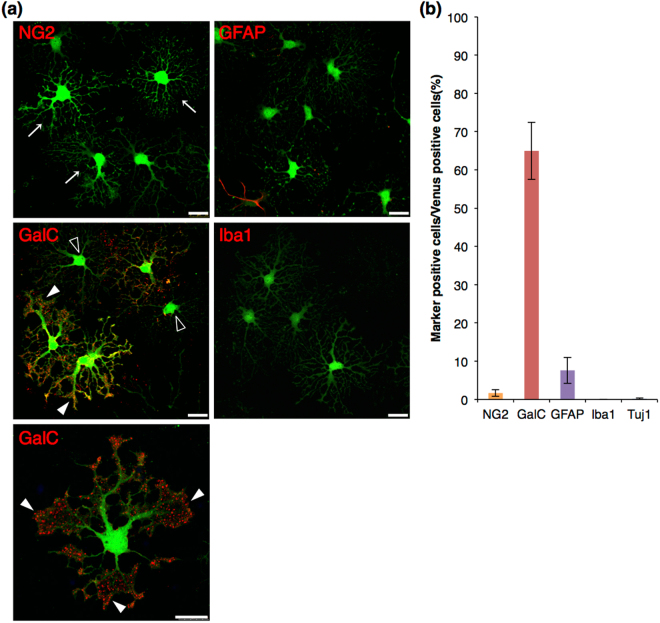
To assess the functional properties of Venus (+) OPCs, we evaluated the differentiation potential of Venus (+) cells in vitro. For oligodendrocyte differentiation assay, Venus (+) cells were cultured in Serum-free differentiation medium for 3 days. After induction of differentiation, OPCs extended their cell processes and formed numerous branches in the processes (Fig. 3a: arrows). OPCs underwent terminal differentiation into mature oligodendrocytes by losing the expression of NG2, and began to express myelin markers, such as GalC. As shown by immunocytochemistry, NG2-positive cell number dropped drastically to 1.7 ± 0.9% (Fig. 3b), compared with the percentage before the induction of differentiation (79 ± 3.6%) (Fig. 2b), while GalC-positive cell number increased up to 65 ± 7.5% (Fig. 3b). Some of GalC-positive cells possessed sheet-like structures in their cell processes, which are required for myelin formation (Fig. 3a: closed arrowheads). There were also GalC-negative cells, whose morphology looked like differentiating OPCs into oligodendrocytes (Fig. 3a: opened arrowheads). These GalC-negative cells were probably in between NG2-positive OPCs and GalC-positive oligodendrocytes. Further, a slight increase of GFAP-positive cells was observed (7.6 ± 3.4%) (Fig. 3b), relative to the population before differentiation (4.5 ± 3.4%) (Fig. 2b). No Iba1- or Tuj1-positive cells were present (Fig. 3b). These results suggested that Venus (+) OPCs were differentiated into oligodendrocytes under the serum-free condition.
Figure 3.
Immunocytochemistry of glial and neuronal markers in Venus (+) cells after oligodendrocyte differentiation. (a) Confocal images of the immunocytochemistry after oligodendrocyte differentiation. Venus fluorescence is shown in green, and each glial marker is shown in red. Venus (+) OPCs formed highly branched cell processes after 3 days of culture in Serum-free differentiation medium (arrows). These cells expressed GalC, showing sheet-like structures in their cellular processes (closed arrowheads). GalC-negative cells, whose morphology was likely differentiating OPCs to oligodendrocytes, were observed (opened arrowheads). Scale bar, 25 µm. (b) Cell count analysis showing cell population of Venus (+) cells expressing each marker. Error bars, s.e.m.
Differentiation of Sox10-Venus positive OPCs into astrocytes in the presence of high concentration of serum
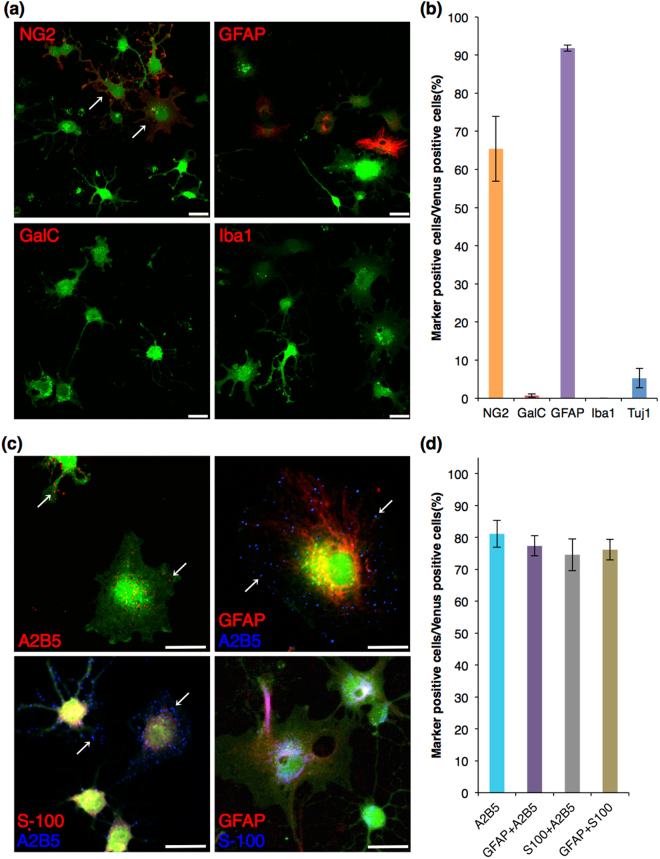
OPCs possess the ability to generate type II astrocytes as well as oligodendrocytes in vitro 4. It was reported that serum-containing differentiation medium, containing 20% fetal bovine serum (FBS) is suitable for induction of astrocytic differentiation18. Thus, we examined the characteristics of purified Venus (+) OPCs cultured in Proliferation medium for 1 day, and then cultured in Serum-containing differentiation medium thereafter. After 3 days culture in Serum-containing differentiation medium, 92 ± 0.8% of the cells expressed GFAP, and many of these cells were NG2-positive and changes in cell morphology were observed (Fig. 4a,b). Less than 1% of the cells expressed GalC (0.7 ± 0.5%) (Fig. 4a,b). None of the cells were positive for Iba1, whereas a small population was positive for Tuj1 (6.3 ± 2.0%) (Fig. 4a,b). After the astrocytic differentiation, Venus (+) OPCs formed lots of primary cell processes and developed into flat and stellate-shaped cells with large nuclei (Fig. 4a: arrows). The Venus intensity slightly weakened, although NG2 staining on protrusions radiating out from the large and flat cell body was observed (Fig. 4a: arrows). Some cells showed OPC-like cell morphology, although these cells did not differentiate into mature oligodendrocytes under this culture condition (Fig. 4a,b). These observations indicated that Venus (+) OPCs differentiated into astrocytic cells. To determine whether these GFAP-positive Venus (+) cells were type II astrocytes, A2B5 antibody, a marker for type II astrocytes, was used for the immunocytochemistry analysis. Punctuated staining of ganglioside, an antigen for A2B5, was observed in 81 ± 4.2% out of the total cells (Fig. 4c: arrows, 4d). Further, most of A2B5-positive cells immunoreacted with GFAP and S-100, another marker for astrocytes [Fig. 4c,d: GFAP/A2B5 (77 ± 3.2%), S-100/A2B5 (75 ± 5.0%)]. Also, most of GFAP-positive cells expressed S-100 (76 ± 3.2%) (Fig. 4c,d). These immunocytochemistry results showed that Venus (+) OPCs were differentiated into type II astrocytes.
Figure 4.
Immunocytochemistry of glial and neuronal markers in Venus (+) cells after astrocytic differentiation. (a,c) Confocal images of the immunocytochemistry after astrocytic differentiation. Venus fluorescence is shown in green, and each glial marker is shown in red or blue. Venus (+) OPCs developed into flat shaped cells with short primary cell processes (A: arrows). Most of Venus (+) cells expressed A2B5 with GFAP or S-100, or GFAP with S-100. Arrow: punctated staining of A2B5 on Venus (+) cell; Scale bar, 25 µm. (b,d) Cell count analysis showing cell population of Venus (+) cells expressing each marker. Error bars, s.e.m.
Time-lapse analysis of Sox10-Venus positive cells
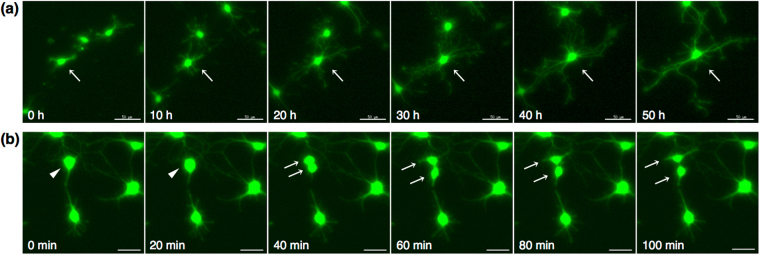
Time-lapse analysis was performed to observe the morphological change of purified Venus (+) OPCs. After 1 day-culture in Proliferation medium, the medium was changed to Serum-free differentiation medium, and time-lapse images were captured every 20 minutes. During the observation period, OPCs underwent extensive morphological remodeling when they differentiated from bipolar OPCs into oligodendrocytes. Mature oligodendrocytes extended complex and highly branched processes (Fig. 5a Supplementary Video 1). Cell division was also observed in some cells (Fig. 5b, Supplementary Video 2).
Figure 5.
Time-lapse analysis of Venus (+) cells. (a) Differentiation of Venus (+) OPCs. In vitro time-lapse images were captured to follow the process formation of Venus (+) oligodendrocytes after induction of differentiation. Images every 10 hours are representatively indicated. Arrow: differentiating OPC with branched process formation; Scale bar, 50 µm. (b) Cell division of Venus (+) OPCs. Representative cell division images are shown every 20 minutes. Arrowhead: OPC before cell division; Arrows: OPCs after cell division; Scale bar, 30 µm.
Sox10-Venus positive cells during postnatal development in the brain tissues
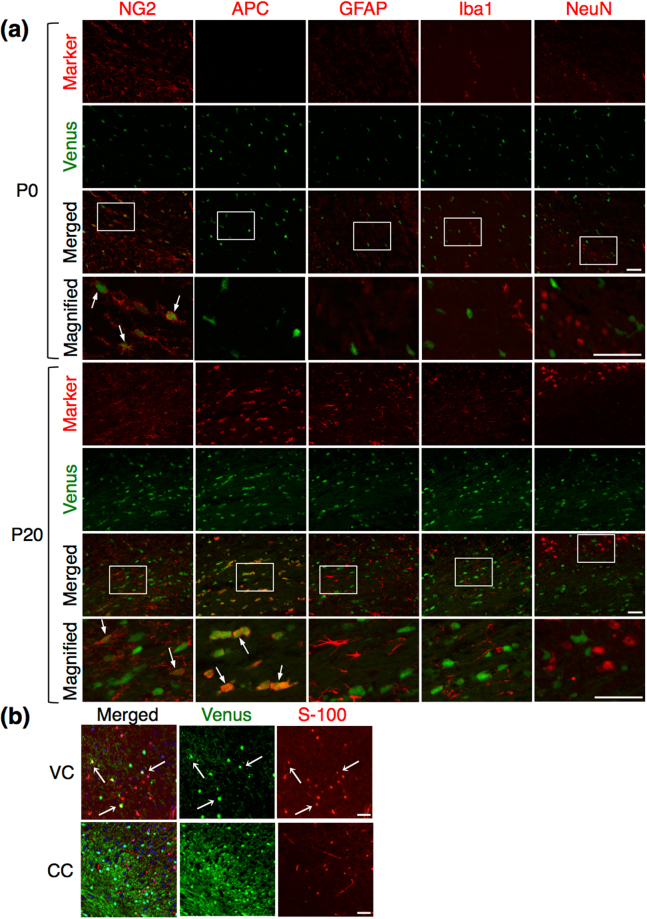
We further analyzed Venus (+) cells in the brain tissues by immunohistochemistry. At P0, Venus (+) cells were distributed broadly in the brain tissues, and a slightly higher signal of Venus was observed in the corpus callosum tissue, compared to other regions (Supplementary Figure S3). Immunostaining analysis in the corpus callosum revealed that Venus (+) cells expressed NG2 and PDGFRα, but not Adenomatous Polyposis Coli (APC), a marker for mature oligodendrocytes, GFAP, Iba1, or NeuN, at this stage (Fig. 6a and Supplementary Figure S1b). APC was rarely detectable in this tissue, since OPCs are not yet differentiated to mature oligodendrocytes at P0. This result suggested that Venus (+) cells at P0 were OPCs. We performed the same analysis using the tissue sections from P20, when myelination intensively occurs. Strong Venus fluorescence was observed in the corpus callosum and the striatum tissues (Supplementary Figure S3). We found that Venus (+) cells were positive for APC, in addition to NG2, but negative for GFAP, Iba1, or NeuN (Fig. 6a). These data indicated that Venus (+) cells corresponded to oligodendrocyte-lineage cells, including OPCs and oligodendrocytes, as expected due to the endogenous Sox10 expression pattern16.
Figure 6.
Immunohistochemistry of glial and neuronal markers in Venus (+) cells in the brain tissues. (a) Immunohistochemistry in the corpus callosum of the P0 and P20 posterior forebrain, showing the distribution of Venus positive cells (green) with each marker (red). Arrows: dual-positive cells of Venus with a marker; Scale bar: 50 µm. (b) Immunohistochemistry of S-100 in the ventral cortex (VC) and the corpus callosum (CC) at 12 months. Dual-positive cells of Venus with S-100 were observed in the ventral cortex (arrows); Scale bar: 50 µm.
We also investigated whether Venus was expressed in astrocytes in the ventral cortex as reported previously13. Venus (+) cells positive for GFAP were hardly detected not only in the corpus callosum (Fig. 6a) but also in the ventral cortex, the dorsal cortex, and the striatum (data not shown). However, a broader marker for astrocytes, S-100, was positive in some of Venus (+) cells in the ventral cortex (Fig. 6b), as well as in the striatum (data not shown), but not in the corpus callosum (Fig. 6b). These evidences showed that Venus (+) cells were OPCs and were able to differentiate into oligodendrocytes and astrocytes, in both the brain tissues and the in vitro culture. All together, the results presented in this study showed that Sox10-Venus mice are useful for both in vitro and in vivo studies of OPCs, such as differentiation and morphological analyses.
Discussion
In this study, we report a mouse OPC purification and culture method using Sox10-Venus mice. We showed that Venus (+) cells from P0-2 brains were OPCs and differentiated to either oligodendrocytes or astrocytes, depending on the culture conditions. Further, we were able to observe the formation of cell processes during the OPC differentiation to oligodendrocyte by time-lapse analysis.
Several transgenic mouse lines with a reporter system of a fluorescent protein under an oligodendrocyte lineage cell-specific promoter have been recently reported for OPC purification and culture experiments. Zhu et al. identified the characteristics of OPCs purified from NG2DsRedBAC transgenic mice that express DsRed in NG2-expressing cells. After cell sorting with a fluorescence of DsRed at P3, almost 100% of purified DsRed (+) cells were positive for NG2, and about 20% of the total cells differentiated to oligodendrocytes positive for O1 after induction of differentiation13. In addition, more than 85% of total cells were still positive for NG2 cells13. In the present study, we succeeded in obtaining differentiated GalC-positive oligodendrocytes more efficiently (>64%) from Sox10-Venus OPCs (Fig. 3). Moreover, the percentage of NG2 positive cells after differentiation culture was less than 2% out of total cells (Fig. 3). In addition to the NG2DsRedBAC line, the GFP reporter system of PDGFRα, another OPC marker, was utilized for purification and characterization of OPCs in culture12. However, using these systems, it is unable to perform a time-lapse experiment with these fluorescent proteins, since activity of either NG2 or PDGFRα promoter is dispersed during the differentiation to oligodendrocytes. Whereas the CNP-EGFP mouse line is also useful for studies of oligodendrocyte lineage cells, combination with cell sorting using anti-NG2 antibody is needed for purification of OPCs from this mouse line, as the CNP promoter activity begins just after the onset of the differentiation19. In contrast, Sox10 is continuously expressed during the stages from OPCs to oligodendrocytes16. In the present study, we were able to evaluate the OPC differentiation and to observe cell process formation and cell division in the culture by time-lapse analysis (Figs 2–5, Supplementary Video 1, 2). Recently, Tripathi et al. created Sox10-GFP/tdTom dual reporter mice and their group purified either GFP- and tdTomato-positive OPCs from the ventral and dorsal regions in the brain, respectively, by flow cytometer20,21. They found that there were differences in OPC migration and differentiation capacities between the ventral and dorsal area-derived OPCs from the adult brain. However, they did not stain the cells with other glial or neuronal markers, neither they induced their differentiation to astrocytes nor carried out time-lapse analysis, which were analyzed in this study (Figs 2–5, Supplementary Video 1, 2). Given these observations and evidences, we provide here a useful method for analyzing OPC differentiation to oligodendrocytes in vitro using Sox10-Venus mice. Further, the Venus fluorescence is more intense than that of other fluorescent proteins, including DsRed, GFP, and tdTomato, which were used in the previous studies12–14,17,20,21. Particularly, the intense fluorescence of Sox10-Venus was very useful and reliable to observe the fine structures of cell protrusions during the processes formation (Fig. 5, Supplementary Video 1, 2).
OPCs have the potential to differentiate not only to oligodendrocytes but also to type II astrocytes in vitro. Culturing under high concentration of FBS, fetal calf serum, or the addition of bone morphogenetic proteins, induces OPCs differentiation into type II astrocytes19,22. Type II astrocytes are differentiated from OPCs and express specific antigens, such as GFAP, S-100, and the ganglioside for A2B5. In this study, we demonstrated an efficient differentiation of type II astrocytes from Sox10-Venus OPCs in the presence of 20% FBS, and found that most of the astrocytic cells from Venus (+) OPCs express NG2 (Fig. 4), as previously reported13, although NG2 is a marker for OPCs. We can postulate that type II astrocytes maintain NG2 expression, in addition to a significant increase of astrocytic marker expression. The differences in protein expression between type I and II astrocytes may reflect their distinct functional properties.
In our previous work, we demonstrated that Venus is specifically expressed in oligodendrocyte-lineage cells in the spinal cord of Sox10-Venus mice at embryonic day 15.5 and 8 weeks after birth15. In the present study, Venus (+) cells expressed NG2 at P0, and NG2 or MBP at P20 in the corpus callosum of the Sox10-Venus mouse brain (Fig. 6). This expression pattern of Venus is consistent with that of endogenous Sox10 during OPC differentiation to oligodendrocytes16. Further, S-100- and Venus-double positive cells were observed in the ventral cortex, but not in the corpus callosum, of the Sox10-Venus brain, while double positive cells for GFAP and Venus were rarely detected (Fig. 6). Previously, in vivo cell fate mapping of OPCs has been carried out. Zhu et al. reported that S-100-positive astrocytes derived from OPCs reside in the ventral cortex more than in the corpus callosum of the NG2CreBAC:Z/EG transgenic mice, and even smaller number of GFAP-positive astrocytes derived from OPCs was found, compared with OPC-derived S-100-positive astrocytes13. This agrees with our present data. Also, some studies demonstrate that OPCs give rise to neurons as well as oligodendrocytes and astrocytes, but others denied that2,23. In our system, none of the neurons expressed Venus in the Sox10-Venus brain (Fig. 6). These controversial evidences among the reported data, regarding neurogenesis from OPCs, is probably due to differences of the reporter gene regulation systems and/or of timing or area for the analyses. More detailed studies are needed to elucidate the mechanism.
In summary, we established an efficient purification method of OPCs, which were differentiated into oligodendrocytes and type II astrocytes, using Sox10-Venus mice. In the Sox10-Venus brain, Venus (+) OPCs, oligodendrocytes, and astrocytes were found. Analyses using the Sox10-Venus mouse system will facilitate a better understanding of developmental and pathological function of OPCs in vivo and of cellular and molecular OPC function in vitro.
Methods
Animals
P0-2 pups from the previously reported Sox10-Venus mice15 were euthanized by decapitation as described in the International Animal Care and Use Committee and the previous report5, and then, they were used for the experiments. All procedures for animal experiments were approved by the Tokyo Medical and Dental University Animal Care and Use Committee (Protocol number: 0170282 C) and the Juntendo University School of Medicine Animal Care and Use Committee (Institutional review board No. 270050). All methods were conducted in strict accordance with the approved guidelines of the institutional animal care committees.
Primary cell preparation
Primary mixed cell cultures were prepared from the brain of P0-2 mouse pups. The whole brain was dissected out and the meninges were removed. Then, the brains were diced into 1 mm3 chunks in a dish with ice-cold Leibovitz’s L-15 medium (Life Technologies) and enzymatically dissociated with 10 ml of Papain/DNase I solution containing: 40 mg of α-d-Glucose (SIGMA-ALDRICH), 4 mg of bovine serum albumin (SIGMA-ALDRICH), 4 mg of l-cysteine (SIGMA-ALDRICH), 2.5 µl/ml Papain (Worthington), 5 µl/ml DNase I (SIGMA-ALDRICH) in phosphate buffered saline (PBS). After an incubation for 20 minutes at 37 °C, the cells were collected by centrifugation in a swinging bucket at 100 g for 5 minutes. The supernatant was removed and Dulbecco’s modified Eagle’s medium (DMEM; Life Technologies), supplemented with 10% FBS (Thermo Fisher Scientific), as well as sodium pyruvate (SIGMA-ALDRICH), l-glutamine (Life Technologies), and 100 units/ml penicillin and 100 µg/ml streptomycin (Life Technologies) were added. The pellet was dissociated, and the tissue suspension was filtered through a 70 µm nylon cell strainer (Falcon).
Isolation of Sox10-Venus positive cells using flow cytometry
The cell suspension was collected in a 5 ml tube with cell-strainer cap (Falcon) and was labeled with propidium iodide (SIGMA-ALDRICH) for 10 minutes to remove cell debris and dead cells. Cell sorting was performed using JSAN flow cytometer (Bay bioscience) or MoFlo flow cytometer (Beckman). Cells were collected in a tube containing DMEM supplemented with 10% FBS, sodium pyruvate, l-glutamine, and penicillin/streptomycin.
Cell culture and differentiation of Sox10-Venus positive cells
Sorted cells were plated in Poly-d-Lysine 8-Well Culture Slides (BD Biosciences) at 3.0 × 104 cells per well and incubated under 5% CO2 at 37 °C. Cells were cultured in Proliferation medium [Basal chemically defined medium (BDM) supplemented with 0.5% FBS, and penicillin/streptomycin]. BDM was prepared by adding 0.1% BSA, 4 mM l-glutamine, 1 mM sodium pyruvate, 30 nM sodium selenite (SIGMA-ALDRICH), 10 nM hydrocortisone (SIGMA-ALDRICH), 10 nM d-biotin (SIGMA-ALDRICH), 50 µg/ml Apo-transferrin (SIGMA-ALDRICH), 5 µg/ml insulin (SIGMA-ALDRICH), and penicillin/streptomycin in DMEM. There was no difference in proliferation and differentiation of Venus (+) OPCs between 0.5% FBS- and 10 ng/ml of PDGF-AA/basic-FGF-contained media during the 1-day culture. One day after plating, the medium was changed to serum-free differentiation medium [BDM supplemented with 40 ng/ml triiodo-thyronine (SIGMA-ALDRICH), 1 ng/ml ciliary neurotrophic factor (Peprotech), 50 ng/ml N-acetyl-l-cysteine (SIGMA-ALDRICH)] and cultured for 3 days. For astrocytic differentiation, cell medium was changed to Serum-containing differentiation medium [BDM containing 20% FBS and penicillin/streptomycin] for 3 days.
Immunostaining
The cells or frozen tissue sections were fixed with 4% paraformaldehyde (PFA) in PBS (Wako) for 15 minutes at room temperature, and permeabilized with 0.1% Triton X-100 (SIGMA-ALDRICH) for detection of intracellular proteins and a membrane marker NG2. To detect another membrane marker GalC, the permeabilization step was omitted. After blocking with Power Block Universal Blocking Reagent (BioGenex Laboratories) for 1 hour at room temperature, cells were incubated with primary antibodies overnight at 4 °C. The primary antibodies were as follows: rabbit anti-NG2 (1:250; Millipore), mouse anti-GalC (1:250; Millipore), mouse anti-A2B5 (1:300; abcam), rabbit anti-GFAP (1:1000; Thermo Scientific), mouse anti-S100 (1:1000; SIGMA-ALDRICH), mouse anti-APC (1:20; Millipore), mouse anti-NeuN (1:100; Millipore), and rabbit anti-Iba1 (1:250; Wako). After washing, the cells were labeled with secondary antibodies for 45 minutes at room temperature. The secondary antibodies were as follows: rabbit/mouse IgG-Alexa 594 (Life Technologies), mouse IgG- and mouse IgM-Alexa 647 (Life Technologies). The samples were mounted with Vectashield containing DAPI (Vector Laboratories).
Analysis of cell morphology and cell count
For antigenic phenotyping, positive cells for each antigen were counted and expressed as a percentage out of all Venus positive cells. Confocal microscopy images were obtained using a Leica TCS-SP5 confocal laser scanning microscope (Leica Microsystems), and all confocal settings were set to the same parameters for each experiment. Metamorph software was used for analysis, and at least three independent sorting experiments were analyzed. The numbers were expressed as means ± standard error.
RT-PCR
Total RNA was isolated from 6 wells of the primary Sox10-Venus cell culture in the poly-D-Lysine 8-Well Culture Slide using TRI-Reagent (SIGMA-ALDRICH) as described previously24. Seventy-five μg of total RNA was reverse-transcribed into cDNA using SuperScript III Reverse Transcriptase (Life Technologies) and Oligo dT Primer (Life Technologies). For quantitative RT-PCR, cDNA was amplified for initial denaturation at 95 °C for 20 seconds, and then 40 PCR cycles of 95 °C for 3 seconds, 60 °C for 30 seconds, using Fast SYBR Green Master Mix (Applied Biosystems), with the following gene-specific primers; NG2: 5′-TCAAAACTTCAGCGTTCCCG-3′ (forward), 5′-CAAAGGCGTCTGTCTGTGTCTCAC-3′ (reverse); MAG: 5′-GCCTTTGCCATCCTGATTGC-3′ (forward), 5′-TCTGAGTGGGAATAACTGAGGTCC-3′ (reverse); GFAP: 5′-CCACCAAACTGGCTGATGTCTAC-3′ (forward), 5′-CCTTTCTCTCCAAATCCACACG-3′ (reverse); GLAST: 5′-CGCAGGGAAGATTGTTGAGATG-3′ (forward), 5′-TGAAAGTGATGGGTAGGGTGGC-3′ (reverse); NeuN: 5′-TCTCTTGTCCGTTTGCTTCCAG-3′ (forward), 5′-TCCGATGCTGTAGGTTGCTGTG-3′ (reverse); Iba1: 5′-GTCCTTGAAGCGAATGCTGG-3′ (forward), 5′-CATTCTCAAGATGGCAGATC-3′ (reverse); β-actin: 5′-ATTGCTGACAGGATGCAGAA-3′ (forward), 5′-TAGAGCCACCAATCCACACAG-3′ (reverse).
Time-lapse imaging
In vitro time-lapse images were captured to follow the cell processes formation after induction of differentiation. Time-lapse images were captured using a Delta Vision system (Applied Precision) equipped with an inverted microscope IX-70 (Olympus) with a 10× objective in temperature-controlled chamber (37 °C) in which a humidified premixed gas (5% CO2 and 95% air) was infused. Z-stack images were collected every 20 minutes for 50 hours.
Frozen tissue sections
Mice were deeply anesthetized with sodium pentobarbital (30 mg/kg) and perfused transcardially with PBS and then with 4% PFA in PBS (Wako). Brain tissues were post-fixed with 4% PFA in PBS overnight at 4 °C. After fixation, tissues were stored in 15% sucrose solution in PBS at 4 °C overnight, and then in 30% sucrose solution in PBS overnight at 4 °C. Tissues were embedded with OCT compound (Sakura Finetek). The embedded tissues were sectioned using a cryostat (Leica) and immunohistochemistry was performed as described above.
Data availability
All the data are presented in the manuscript as figures or additional files.
Electronic supplementary material
Acknowledgements
We thank Dr. Atsuyuki Yamataka and Dr. Katsumi Miyahara (Juntendo Univ., Tokyo, Japan) for maintaining the Sox10-Venus mouse line. We also thank Dr. Susana de Vega (Juntendo Univ., Tokyo, Japan) for critical reading the manuscript. This work was supported by a Grant-in-Aid for Scientific Research (C) of the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) in Japan (25505001) (C.A., N.S., and Y.M.), a Grant-in-Aid for Young Scientists of the MEXT in Japan (25860701) (N.S.), and a Grant-in-Aid from the Ministry of Health, Labor and Welfare of Japan (2260201) (C.A.). This work was also supported by Building of Consortia for the Development of Human Resources in Science and Technology from MEXT in Japan (C.A.).
Author Contributions
N.S. and C.A. designed the project. N.S., K.S., C.H., Y.M., and T.N. performed experiments and analyzed the data. N.S., K.S., and C.H. prepared the figures. N.S. and K.S. wrote the manuscript. N.S. and C.A. supervised the project. All the authors read and approved the final manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Nobuharu Suzuki and Kaori Sekimoto contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-017-14207-0.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Boulanger JJ, Messier C. From precursors to myelinating oligodendrocytes: contribution of intrinsic and extrinsic factors to white matter plasticity in the adult brain. Neuroscience. 2014;269:343–366. doi: 10.1016/j.neuroscience.2014.03.063. [DOI] [PubMed] [Google Scholar]
- 2.Nishiyama A, Komitova M, Suzuki R, Zhu X. Polydendrocytes (NG2 cells): multifunctional cells with lineage plasticity. Nat. Rev. Neurosci. 2009;10:9–22. doi: 10.1038/nrn2495. [DOI] [PubMed] [Google Scholar]
- 3.Nishiyama A, Suzuki R, Zhu X. NG2 cells (polydendrocytes) in brain physiology and repair. Front Neurosci. 2014;8:133. doi: 10.3389/fnins.2014.00133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Behar TN. Analysis of fractal dimension of O2A glial cells differentiating in vitro. Methods. 2001;24:331–339. doi: 10.1006/meth.2001.1203. [DOI] [PubMed] [Google Scholar]
- 5.Chen Y, et al. Isolation and culture of rat and mouse oligodendrocyte precursor cells. Nat. Protoc. 2007;2:1044–1051. doi: 10.1038/nprot.2007.149. [DOI] [PubMed] [Google Scholar]
- 6.Fanarraga ML, Sommer I, Griffiths IR. O-2A progenitors of the mouse optic nerve exhibit a developmental pattern of antigen expression different from the rat. Glia. 1995;15:95–104. doi: 10.1002/glia.440150202. [DOI] [PubMed] [Google Scholar]
- 7.Duchala CS, Asotra K, Macklin WB. Expression of cell surface markers and myelin proteins in cultured oligodendrocytes from neonatal brain of rat and mouse: a comparative study. Dev. Neurosci. 1995;17:70–80. doi: 10.1159/000111276. [DOI] [PubMed] [Google Scholar]
- 8.Horiuchi M, Lindsten T, Pleasure D, Itoh T. Differing in vitro survival dependency of mouse and rat NG2+ oligodendroglial progenitor cells. J. Neurosci. Res. 2010;88:957–970. doi: 10.1002/jnr.22262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB. NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev. Dyn. 2001;222:218–227. doi: 10.1002/dvdy.1200. [DOI] [PubMed] [Google Scholar]
- 10.Emery B, Dugas JC. Purification of Oligodendrocyte Lineage Cells from Mouse Cortices by Immunopanning. Cold Spring Harb. Protoc. 2013;9:854–868. doi: 10.1101/pdb.prot073973. [DOI] [PubMed] [Google Scholar]
- 11.Czechowicz A, Kraft D, Weissman IL, Bhattacharya D. Efficient transplantation via antibody-based clearance of hematopoietic stem cell niches. Science. 2007;318:1296–1299. doi: 10.1126/science.1149726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moyon S, et al. Demyelination causes adult CNS progenitors to revert to an immature state and express immune cues that support their migration. J. Neurosci. 2015;35:4–20. doi: 10.1523/JNEUROSCI.0849-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhu X, Bergles DE, Nishiyama A. NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development. 2008;135:145–157. doi: 10.1242/dev.004895. [DOI] [PubMed] [Google Scholar]
- 14.Tripathi RB, Rivers LE, Young KM, Jamen F, Richardson WD. NG2 glia generate new oligodendrocytes but few astrocytes in a murine experimental autoimmune encephalomyelitis model of demyelinating disease. J. Neurosci. 2010;30:16383–16390. doi: 10.1523/JNEUROSCI.3411-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shibata S, et al. Sox10-Venus mice: a new tool for real-time labeling of neural crest lineage cells and oligodendrocytes. Mol. Brain. 2010;3:31. doi: 10.1186/1756-6606-3-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stolt CC, et al. Terminal differentiation of myelin-forming oligodendrocytes depends on the transcription factor Sox10. Genes Dev. 2002;16:165–170. doi: 10.1101/gad.215802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nagai T, et al. A variant of yellow fluorescent protein with fast and efficient maturation for cell-biological applications. Nat. Biotechnol. 2002;20:87–90. doi: 10.1038/nbt0102-87. [DOI] [PubMed] [Google Scholar]
- 18.Raff MC, Miller RH, Noble M. A glial progenitor cell that develops in vitro into an astrocyte or an oligodendrocyte depending on culture medium. Nature. 1983;303:390–396. doi: 10.1038/303390a0. [DOI] [PubMed] [Google Scholar]
- 19.Jablonska B, et al. Sirt1 regulates glial progenitor proliferation and regeneration in white matter after neonatal brain injury. Nat. Commun. 2016;7:13866. doi: 10.1038/ncomms13866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tripathi RB, et al. Dorsally and ventrally derived oligodendrocytes have similar electrical properties but myelinate preferred tracts. J. Neurosci. 2011;31:6809–6819. doi: 10.1523/JNEUROSCI.6474-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Crawford AH, Tripathi RB, Richardson WD, Franklin RJ. Developmental Origin of Oligodendrocyte Lineage Cells Determines Response to Demyelination and Susceptibility to Age-Associated Functional Decline. Cell Rep. 2016;15:761–773. doi: 10.1016/j.celrep.2016.03.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mabie PC, et al. Bone morphogenetic proteins induce astroglial differentiation of oligodendroglial-astroglial progenitor cells. J. Neurosci. 1997;17:4112–4120. doi: 10.1523/JNEUROSCI.17-11-04112.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rivers LE, et al. PDGFRA/NG2 glia generate myelinating oligodendrocytes and piriform projection neurons in adult mice. Nat. Neurosci. 2008;11:1392–1401. doi: 10.1038/nn.2220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Suzuki N, et al. Teneurin-4 is a novel regulator of oligodendrocyte differentiation and myelination of small-diameter axons in the CNS. J. Neurosci. 2012;32:11586–11599. doi: 10.1523/JNEUROSCI.2045-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All the data are presented in the manuscript as figures or additional files.