Abstract
Compared to great apes, humans maintain a relatively rapid reproductive pace despite long periods of dependency. This seemingly contradictory set of traits is made possible by weaning offspring before nutritional independence and alloparents who help provide care. In traditional societies, this help may be provided to mothers in part by their juvenile offspring who carry, supervise, or provision younger siblings. In contrast to humans, chimpanzees (Pan troglodytes) are nutritionally independent after weaning, yet juveniles continue to travel with their mother and younger sibling for an additional 4–5 years. This continued association could be costly to the mother if she continues to invest in weaned offspring. Alternately, while juvenile chimpanzees do not typically provision younger siblings, their presence and social interaction with infants may allow mothers to focus on other tasks. In this study, we investigate the costs and benefits to mothers of continued association with juveniles in wild chimpanzees. Using 26 years of long-term behavioral data we examined how maternal activity budgets varied based on presence of a dependent juvenile offspring. We found that continued social interaction between mothers and juveniles does not influence the mother’s time allocated to interacting with the younger infant, her feeding, resting, or travel time, or time socializing with other community members. Instead, mothers may benefit from the additional social interaction and/or relationship with their older offspring. Using 45 years of demographic data we found that those offspring who had an older sibling tended to be more likely to survive each year from birth-8 years than those without an older sibling. Additionally, interbirth intervals were more likely to end when the female had an older offspring present. A mutually beneficial mother-juvenile dynamic in great apes provides insight into continued association between mothers and offspring after nutritional-independence and the emergence of juvenile helping during hominin evolution.
Keywords: Mothers, Siblings, Reproduction, Life history
Introduction
Compared to other mammals of similar body size, primates are characterized by long, slow periods of growth, delayed sexual maturation, and long lifespans (Harvey et al., 1987; Charnov and Berrigan, 1993). The extended juvenile period between weaning and sexual maturation and the associated delay in reproduction, which is most pronounced in the great apes and markedly prolonged in humans, is of particular interest. Unlike humans who are weaned relatively early and require additional provisioning (Robson et al., 2006; Bogin, 2009), nonhuman primates are nutritionally independent after weaning. However, despite this nutritional independence, juvenile nonhuman primates of both sexes remain in their natal group where they are thought to benefit from familiar ecological and social environments (Fairbanks, 2000), including continued association with and support from their mothers (Pereira and Altmann, 1985; Fairbanks, 1988; Van Noordwijk, 2012). The majority of existing research on the juvenile period in primates focuses on the adaptive value of this developmental stage from the juvenile’s perspective, highlighting the benefits of slow and steady growth (e.g., Janson and van Schaik, 1993; Pagel and Harvey, 1993) and time to develop ecological and social competence (e.g., Ross and Jones, 1999; Pereira and Fairbanks, 2002; Cords et al., 2010). Less is known, however, about the costs and benefits to nonhuman primate mothers of continued association and interaction with juvenile offspring. In traditional human societies, mothers typically raise multiple offspring of different ages at a given time. Juvenile humans (~4–10 years of age) can provide benefits to their mothers by contributing to the care of younger siblings through behaviors including carrying, supervising, and provisioning food (e.g., Kramer, 2005; Konnor, 2010; Kramer, 2011; Crittenden et al., 2013), which, studies suggest, can translate into higher survival of younger siblings (e.g., Sear and Mace, 2008; Nitsch et al., 2013). In this study, we investigate potential costs and benefits of post-weaning maternal care among wild chimpanzees (Pan troglodytes).
Compared to chimpanzees, modern humans have lower mortality rates, later maturation, and longer lifespans (Kaplan et al., 2000; Leigh, 2001; Robson et al., 2006; Emery Thompson, 2013). However, despite extensive time and energy investment in offspring, modern human reproductive rates are nearly double that of chimpanzees (Emery Thompson, 2013). Humans maintain the seemingly conflicting combination of large investment with relatively rapid reproduction in part by weaning offspring before they are nutritionally independent (Bogin, 2009). This ‘stacking’ of dependent offspring is likely a derived feature in Homo (Dean, 2006; Robson et al., 2006) associated with the emergence of other distinctive hominin traits, including slow juvenile growth rates (Gurven and Walker, 2006) and reliance on alloparental care (Kramer and Otárola-Castillo, 2015), that allowed for relatively high fertility rates and hominin expansion into novel, ecologically variable environments (Wells and Stock, 2007; Wells, 2012). Recent modeling efforts indicate that early shifts towards modern human life histories could have been supported by help from within mother-offspring groups (Kramer, 2014; Kramer and Otárola-Castillo, 2015). Thus, understanding the trade-offs of post-weaning maternal care in one of human’s closest relatives can provide insight into early hominin life history evolution.
According to parental investment theory, maternal resources are finite and investment in one offspring reduces the ability to invest in other offspring (Trivers, 1972). Parental care, however, is a broader term that does not necessarily require such a trade off and encompasses any parental behavior that might increase offspring success (Clutton-Brock, 1991). In addition to costly investment, parental care can also include non-depreciable or ‘umbrella’ care behaviors such as alarm calling or territorial defense, the benefits of which can be shared across multiple offspring (Lazarus and Inglis, 1986; Clutton-Brock, 1991). The majority of research on post-weaning maternal care in primates comes from female philopatric cercopithecine species (e.g., vervet monkeys [Chlorocebus aethiops] and baboons [Papio spp.]) that live in stable social groups and exhibit matrilineal rank inheritance (reviewed in Fairbanks, 2000). Thus, it is well established that cercopithecine mothers can play a substantial role in the success of their juvenile and even adult female offspring through access to preferred resources and support in agonistic interactions (e.g., baboons: Cheney, 1977; Lee and Oliver, 1979; Altmann and Alberts, 2005; vervet monkeys: Horrocks and Hunte, 1983).
Unlike the cercopithicines described above, neither chimpanzees nor bonobos (Pan paniscus) live in cohesive social groups, but instead exhibit fission-fusion social systems in which party (i.e., subgroup) size and composition are fluid within a larger stable community (Goodall, 1986; Stumpf, 2007). Additionally, in both Pan species males are the philopatric sex, whereas females typically disperse at adolescence (Kano, 1992; Mitani et al., 2002). Despite the lack of a stable social group, evidence for the post-weaning importance of mothers in both Pan species is growing. In bonobos, where females can outrank males in the dominance hierarchy (Kano, 1992; Surbeck and Hohmann, 2013), maternal support of sons extends into adulthood as mothers have high rates of association with their adult sons, support adult sons in adult interactions, and maternal presence is associated with increased mating success of sons (Surbeck et al., 2011). In chimpanzees, orphaned males face significantly lower odds of survival than non-orphaned individuals, even if they lose their mothers after weaning. This increased mortality in weaned orphans highlights the importance of some type of post-weaning maternal care in this species (Nakamura et al., 2014). Notably, unlike in bonobos and matrilineal cercopthicine primates, maternal interventions in immature chimpanzee agonistic interactions are rare (Markham et al., 2015), although some maternal interventions in interactions between immatures and adults have been reported (Pusey, 1983; Goodall, 1986). While maternal presence appears to be important to weaned offspring, the rarity of this behavior suggests that maternal interventions are an unlikely source of direct support, at least concerning aggression among peers.
Chimpanzee offspring are nutritionally dependent on their mothers until they are weaned between the ages of 3 to 5 years (Clark, 1977; Pusey, 1983; Rijt-Plooij and Plooij, 1987). While the mother-infant relationship is considered primary, juvenile chimpanzees remain behaviorally dependent and continue to travel with their mothers and younger sibling for 4–5 years after weaning (Pusey, 1983, 1990; Goodall, 1986). Previous investigations suggest that this continued association may come at some cost. For example, Pontzer and Wrangham (2006) found that maternal day range was positively correlated with juvenile body size, but not with infant carrying. Thus, mothers traveling with a behaviorally dependent juvenile, who has not yet reached adult stature, have smaller day ranges than mothers traveling with just an infant, as the juvenile’s small size limits how far mothers can range while remaining with their juvenile. Another potential cost to the mother is that of feeding competition with her juvenile offspring, although previous work suggests that competition with juveniles over food resources is unlikely to be a significant cost to mothers, as they were observed to supplant juvenile offspring at feeding sites (Pusey 1983). Previous studies have also found that mothers remain their juvenile’s strongest grooming partner (Pusey, 1983, 1990; Watts and Pusey, 1993), yet whether this attention paid to the juvenile comes at a cost to infant care is unknown. To avoid this trade-off, mothers could either increase their total maternal care budget with two dependent offspring, or the juvenile could offset some of the mother’s time investment in the infant. While juveniles in this study community do not habitually carry or provision their infant sibling (Pusey 1983; Goodall 1986), they may provide other benefits. These benefits may include a ‘safe’ social partner such that mothers with juvenile offspring would not need to engage in as much social interaction with their own infants or spend time in parties with other potential social partners, which could allow for mothers to focus on feeding in the absence of other competitors. A recent study among mother-infant pairs in the Ngogo chimpanzee community in Uganda found that increases in infant handling by non-mothers was related to lower lactation effort, which the authors suggest could be indicative of faster weaning and shorter interbirth intervals (IBIs) (Badescu et al., 2016). Early work in Gombe National Park, Tanzania also found descriptive evidence suggesting that mothers with two dependent offspring spend less time with other mothers than mothers with one dependent offspring (Halperin, 1979) and statistical evidence that infants with siblings spent less time interacting with other community members than infants without siblings (Brent et al., 1997).
This study examines potential costs and benefits of the continued care of juveniles in wild eastern chimpanzee (P. t. schweinfurtii) mothers, thereby complementing existing literature on the benefits of juvenility from the offspring’s perspective. Specifically, we consider maternal socializing (grooming and playing), feeding (time spent feeding and diet quality), resting, and travel budgets, as well as reproductive outcomes. If juveniles are costly to the mother, we expect a greater proportion of time devoted to interacting socially with offspring when the mother has two dependent offspring compared to when just an infant is present. This post-weaning maternal care may come at the expense of mothers’ social interactions with others such that we would expect mothers to spend a lower proportion of time interacting with others when traveling with a juvenile and infant compared to when traveling with just an infant. Additional care for juveniles may also interfere with a mother’s resting budget, resulting in less time spent conserving energy by resting, or with her feeding budget, resulting in less time devoted to obtaining food. Because mothers could offset a loss of feeding time by eating higher quality foods, we also examined whether juvenile presence is related to diet quality. Alternately, juveniles may provide a benefit to mothers. If there is some benefit to mothers we expect the proportion of time mothers spend engaged in affiliative interactions with offspring either to decrease, if juveniles offset maternal care in infants and receive little themselves, or to remain constant, if juveniles offset maternal care in infants but continue to receive care themselves. Specifically, if juveniles relieve some of the maternal burden, we expect mothers to spend a lower proportion of time interacting socially with their infants when their juvenile offspring is present compared to when just the infant is present. Furthermore, less time interacting socially with infants may allow mothers more time to engage in social interactions with other community members and/or more time to spend feeding or resting. In terms of reproductive outcomes, any increased burden on mothers with multiple offspring may manifest as a fitness cost in terms of lower infant survival, an important source of variation in lifetime fitness for females of long-lived species (Clutton-Brock, 1988; Altmann and Alberts 2003), or longer IBIs. Alternately, relief from some maternal burden may manifest as greater survival of the infant and shorter IBIs when mothers have an older offspring compared to when no older offspring is alive.
Material and methods
Study site and subjects
We investigated the influence of juvenile offspring on maternal behavior and fitness in the Kasekela community in Gombe National Park, Tanzania. Gombe is a small national park (35 km2) located on the western border of Tanzania. Our study focused on behavioral data collected on Kasekela mothers and their offspring over a 26-year period (1988–2013). Juvenile offspring included in this study were individuals whose mothers had given birth to a younger sibling and were less than 8 years of age. After 8 years of age individuals begin to enter puberty (Wallis, 1997). We focused on mothers with infants ranging from 6 months to 3.5 years of age (Supplementary Online Material [SOM] Table S1). Data on mothers with very young infants were excluded from these analyses since chimpanzee infants remain in almost constant contact with their mothers for the first 6 months of life (Goodall 1986), during which time the opportunity for juveniles to offset maternal care is limited.
Behavioral data collection
Behavioral data were collected as part of a long-term study of maternal behavior and offspring development. Each observation day, researchers followed a focal family (mother and infant or mother, infant, and youngest sibling) and recorded the behavior (e.g., groom, play, travel, rest, and feed) and relevant social partner of each focal family member at 1-minute instantaneous point samples (Altmann, 1974; Goodall, 1986; see Lonsdorf et al., 2014 for a more detailed ethogram). All occurrences of behavioral events, including aggression and vocalizations were also recorded. Party composition scans were also conducted at regular intervals during each follow (5-minute intervals until 2011 and 15-minute intervals thereafter). Target follow duration varied over the course of the study from 6 hours to 12 hours, however some follows were less than 6 hours and durations were unequal due to time spent searching for and losing sight of focal individuals. Thus, we limited analysis to daily follows on focal families with a minimum of 5 hours of observation time in a given condition, that is either traveling with an infant only, with an infant and female juvenile, or with an infant and male juvenile (see SOM Table S1 for sample sizes). Minutes where mother or offspring behavior was unknown or uncertain were excluded.
Behavioral metrics
Behaviors were measured as the proportion of daily follow time spent engaged in a given behavior – the number of minutes engaged in the behavior divided by the number of minutes of good observation. We calculated the overall proportion of time mothers spent feeding, resting, traveling, or in either grooming or social play behavior with any member of the community including dependents. Furthermore, we calculated the proportion of time mothers spent grooming with their infant and juvenile offspring, the proportion of time mothers spent playing with their infant and juvenile offspring, and the proportion of time mothers spent grooming and playing with other members of the community excluding dependents. Overall proportions allowed us to test how the maternal activity budgets varied with the number of dependents (infant versus infant and juvenile), while the proportions of time mothers spent interacting with their dependents or others in the community allowed us to investigate potential trade-offs between infant and juvenile care. Additionally, since chimpanzees are ripe fruit specialists, and following the precedent of previous studies (e.g., Emery Thompson and Wrangham, 2008; Murray et al., 2009), we determined diet quality based on the proportion of fruit in the diet. Diet quality was calculated as the number of minutes spent feeding on fruit divided by the number of minutes spent feeding on an identified food item. Daily average party size was also calculated as the average number of individuals present at each party composition scan across the follow.
Behavioral analyses
To investigate differences in overall maternal activity budget (proportion of time spent feeding, socializing, resting, and traveling) we used a distanced-based permutation Multivariate Analysis of Variance (PERMANOVA; Bray-Curtis dissimilarity index and 5,000 permutations), which allows us to simultaneously assess all four behavioral categories in a single model that accounts for the interdependent nature of activity budget data. While this approach allowed us to examine variability in overall activity budgets, it does not identify which of the individual behavioral categories differed with juvenile presence. Therefore, to investigate differences in the amount of time mothers spent engaged in each individual behavior, as well as any difference in diet quality, we fit separate generalized linear mixed models (GLMMs). Because the response variables were proportions bounded by 0 and 1 and highly skewed, we modeled each proportion as a binomial variable consisting of the number of minutes the mother was engaged in a behavior and the number of minutes the mother was not engaged in the behavior during each follow (e.g., minutes grooming/minutes not grooming) and fit models using a binomial error structure and logit link function. Because sex differences in mother-juvenile social interactions may become pronounced as individuals increase in age and males turn their attention away from the maternal unit (Pusey, 1990), we distinguished between male and female juvenile offspring in all behavioral analyses. In addition to sibling presence (none/male/female), each model also included as fixed explanatory variables: infant age in years, infant sex (male/female), infant parity (firstborn: yes/no), daily average party size, and season (wet/dry) as these factors are likely to influence maternal behavior (e.g., Lonsdorf et al., 2014; Murray et al., 2014; Stanton et al., 2014). Mother ID was included as a random effect in GLMMs to control for repeated, uneven sampling of mothers. An additional observation-level random effect was also included in the GLMMs to correct for overdispersion. This effect models extra variation in the response in order to estimate residual variance better, to avoid overestimating statistical significance (Bates et al., 2015; Harrison, 2014). For the analysis of diet quality we limited the sample to only those days where the mother was observed feeding for at least two hours (n=413) (Markham et al., 2014). Significance of fixed effects in GLMMs was assessed using type II Wald chi-square tests and differences between multiple levels of significant fixed effects were examined using Tukey’s post hoc tests.
Offspring survival
To test for a relationship between having a juvenile sibling and survival we analyzed demographic data from 117 individuals (nwith juvenile sibling at birth=60; nwith no juvenile sibling at birth=57) born in the Kasekela community from 1970 – 2012 whose birthdates were known within 90 days and whose maternal dominance rank could be determined at the time of birth. We used discrete event history analysis (Singer and Willett, 1993) to test whether having a sibling who was less than 8 years older was related to the odds of an individual’s own survival each year between birth to age 8 years. Examining survival past weaning age (~4–5 years) allows us to investigate potential post-weaning effects of older sibling presence on survival, given known post-weaning effects of maternal loss (Nakamura et al., 2014). Eight and a half years of age is the earliest onset of sexual maturity in females in this study community (Wallis, 1997) and female life expectancy peaks between 6–8 years of age (Bronikowski et al., 2016). Discrete event history analysis allows for the inclusion of censored data, can account for repeated sampling, and allows for the analysis of time-dependent variables, for example the death of the mother or dispersal of the sibling, which can occur at different ages for different individuals. Each offspring’s life from age 0 – 8 years was divided into 1-year intervals and the offspring’s survival status for each interval was scored as a binary response (survived/died). Older sibling presence in the community (absent/present), and maternal presence (present/absent) throughout the interval were scored as time-dependent covariates. Individuals were considered dead only if strong evidence, such as previously observed poor health or observed death, existed (Strier et al., 2010). Siblings and mothers were considered absent when they dispersed to another community or died. Offspring who disappeared and could have joined other communities and individuals alive as of October 31th, 2015 were censored.
Offspring survival was analyzed using GLMMs with binomial error structures and logit link functions. A previous analysis found no difference in survival between firstborn infants and later-born infants (Stanton et al. 2014), although sibling presence was not considered in that analysis. While firstborns by definition lack an older sibling, later-borns may or may not have an older sibling present. Therefore, in order to distinguish between any confounding effects of firstborn status and sibling presence, we constructed four candidate models that differed only in the inclusion of firstborn status and sibling presence as covariates. By comparing the fit of these candidate models, we can distinguish the relative importance of firstborn status and sibling presence to offspring survival. All three models contained offspring age (in 1-year intervals) and age2, maternal age at birth, maternal dominance rank at birth, and maternal presence as fixed effects along with mother ID and birth year as random effects. Age2 was included given the non-linear relationship between mortality rates and age early in life in this species (Bronikowski et al., 2016). Both maternal dominance rank and maternal age at birth are well established as significant predictors of offspring survival in this community (Pusey et al., 1997). Maternal dominance rank was determined based on the direction of pant grunts, submissive vocalizations that represent formal indicators of dominance relationships (Bygott, 1979), and decided agonistic events. The outcomes of these interactions were used to calculate normalized cardinal Elo scores using an extension of the traditional Elo rating method that optimizes individual Elo score trajectories through maximum-likelihood fitting of Elo parameters (Albers and de Vries, 2001; Franz et al., 2015; Foerster et al., 2016). Mother ID was included to control for females contributing an uneven number of offspring to the dataset and birth year was included to control for multiple and unevenly distributed births occurring each year.
Our first candidate model included only those variables listed above (age, age2, maternal rank, maternal age, maternal presence, mother ID and birth year). Our second candidate model included those variables listed above plus sibling presence only. Our third candidate model included the variables listed above plus firstborn status only, and our fourth candidate model included both sibling presence and firstborn status (Table 4). The four models were compared using Akaike information criterion values corrected for small sample size (AICc) with the lowest AICc value indicating the best model fit (Akaike, 1974). Furthermore, we calculated weights for each model, which estimate the probability that given a set of models, that model is the best. We also summed the weights of all models containing either sibling presence or firstborn status in order to calculate the relative importance of those variables. Relative importance represents the probability that a variable improves model fit and is included in the best-fit model (Symonds & Moussalli, 2011). Notably, we also assessed whether the effect of sibling presence or firstborn status on survival changed with offspring age by testing the interaction between offspring age and sibling presence and/or firstborn status; the interactions were never significant at α=0.05 and excluded from the candidate models.
Table 4.
AICc ranked model sets comparing model sets of offspring survival each year between 0 – 8 years of age and interbirth interval (IBI) ending each yeara.
| Response | Candidate Model | df | AICc | ΔAICc | ωi |
|---|---|---|---|---|---|
| Offspring Survival | model 2 (+ sibling presence) | 9 | 320.9 | 0.00 | 0.682 |
| model 4 (+ firstborn status+ sibling presence) | 11 | 324.5 | 3.51 | 0.118 | |
| model 1 (null) | 8 | 324.7 | 3.79 | 0.103 | |
| model 3 (+ firstborn status) | 10 | 324.8 | 3.89 | 0.098 | |
|
| |||||
| IBI | model 4 (+ first IBI+ sibling presence) | 9 | 291.8 | 0.00 | 0.585 |
| model 2 (+ sibling presence) | 8 | 292.9 | 1.06 | 0.345 | |
| model 3 (+ first IBI) | 8 | 296.2 | 4.34 | 0.067 | |
| model 1 (null) | 7 | 302.5 | 10.66 | 0.003 | |
Table includes difference in AICc values between the two models (ΔAICc), as well as the Akaike weight value (ωI), which represents the likelihood that given the data, that model is the best. df = degrees of freedom.
Interbirth intervals
We also used discrete time event history analysis to test for a relationship between having an older offspring alive and interbirth interval length by examining the odds of an IBI ending during a given interval. This analysis included 98 IBIs (nwith juvenile sibling at start=53; nno juvenile sibling at start=45) from after 1970 from 34 females whose dominance rank could be determined at the start of the interval. Each IBI was divided up into one-year intervals and whether the IBI ended in each year was coded as the binary response variable (yes/no). Intervals that were ongoing as of October 31th 2015 or that ended with a female’s departure from the community after her infant reached 4 years of age were censored (i.e. IBI ended coded as ‘no’). To avoid including IBIs that ended with the death of a mother before her infant was likely weaned, IBIs that ended with a female’s departure before her infant reached 4 years of age were excluded from the analysis. As above, sibling presence was coded as a time-dependent covariate. That is, for each 1-year interval for each IBI, we indicated whether the female had an offspring who was under 8 years of age at the start of the IBI present in the community. A previous investigation of IBIs among Kasekela females found that the IBI following first birth (referred to here as the first IBI) was longer than subsequent IBIs (Jones et al., 2010); thus as above, we created four candidate models that differed in their inclusion of sibling presence and first IBI as covariates. All four models also included time since birth (1-year intervals) and, based on a previous investigation (Jones et al., 2010), maternal age at the start of the birth interval, maternal dominance score at the start of the birth interval, and a factor indicating whether the infant whose birth began the interval died before the age of 2 years (yes/no) as fixed effects along with female ID and the year the IBI began as random effects.
Our first candidate model included the variables listed above (time, maternal rank, maternal age, infant died before age 2 years, female ID, and year IBI began). Our second candidate model included the variables listed above plus sibling presence only. Our third candidate model included the variables listed above plus IBI following first birth, and our fourth candidate model included both sibling presence and IBI following first birth. These four models were also compared using Akaike information criterion values corrected for small sample size (AICc). As above, we calculated the relative importance of sibling presence and IBI following first birth. Also as above, interactions between time and sibling presence and first IBI were never significant and were excluded from candidate models.
All analyses were conducted in R (version 3.2.1; R Core Development Team, 2015) using the adonis() function in the vegan package for the permutation MANOVA (Oksanen et al., 2013), the lme4 package to fit GLMMs (Bates et al., 2015), the car package for Wald chi-square tests (Fox and Weisberg, 2011), the lsmeans package for Tukey’s post hoc tests (Lenth and Hervé, 2015), and the MuMIn package for model comparison (Bartoń, 2016). This research was noninvasive, complied with the laws of Tanzania, and was approved by The Tanzania Commission for Science and Technology, Tanzania Wildlife Research Institute, and Tanzania National Parks Authority.
Results
Maternal social budget
Maternal overall activity budgets differed significantly based on juvenile presence (F2,493 =5.208, p<0.001), as well as season, average party size, and infant sex (SOM Table S2). Investigations of the individual behaviors reveal that this difference in overall activity budget can be attributed to mothers devoting a greater proportion of observation time to social behaviors when both an infant and a dependent juvenile were present compared to when just an infant was present (Table 1; Fig. 1). Post hoc tests indicate that this increased social budget was true for both male and female juveniles and that mothers with female juveniles had a significantly greater social budget than mothers with male juveniles (Fig. 1). Average party size and season were also significant predictors of time spent socializing. Mothers spent more time socializing with increasing party size, as well as more time socializing in the dry season (mean ± standard error [SE]: Dry = 0.147 ± 0.006; Wet = 0.107 ± 0.006; Table 1).
Table 1.
Results of GLMMs examining the relationship between presence of a juvenile offspring and a mother’s overall proportion of time socializing (grooming or playing) with any community member and proportion of time spent feeding, resting, or travelinga.
| Behavior | Parameter | Estimate (95% CI) | Wald χ2 | df | p |
|---|---|---|---|---|---|
| Socializing | Juvenile Presenceb | 20.68 | 2 | <0.001 | |
| Female Juvenile | 0.719 (0.40, 1.04) | ||||
| Male Juvenile | 0.380 (0.10, 0.66) | ||||
| Infant Sexc | 0.010 (−0.22, 0.23) | 0.008 | 1 | 0.930 | |
| Infant Age | −0.094 (−0.21, 0.02) | 2.441 | 1 | 0.118 | |
| Firstbornd | 0.162 (−0.36, 0.61) | 0.480 | 1 | 0.488 | |
| Average Party Size | 0.021 (0.01, 0.03) | 12.63 | 1 | <0.001 | |
| Seasone | −0.368 (−0.56, −0.18) | 14.92 | 1 | <0.001 | |
| Feeding | Juvenile Presenceb | 0.0001 | 2 | 0.999 | |
| Female Juvenile | −0.001 (−0.24, 0.24) | ||||
| Male Juvenile | −0.0002 (−0.23, 0.23) | ||||
| Infant Sexc | −0.275 (−0.44, −0.11) | 10.17 | 1 | 0.001 | |
| Infant Age | 0.017 (−0.07, 0.11) | 0.142 | 1 | 0.706 | |
| Firstbornd | −0.132 (−0.50, 0.24) | 0.529 | 1 | 0.467 | |
| Average Party Size | −0.032 (−0.04, −0.02) | 52.80 | 1 | <0.001 | |
| Seasone | −0.118 (−0.26 0.02) | 2.775 | 1 | 0.096 | |
| Resting | Juvenile Presenceb | 2.900 | 2 | 0.235 | |
| Female Juvenile | −0.181 (−0.46, 0.10) | ||||
| Male Juvenile | 0.038 (−0.22, 0.29) | ||||
| Infant Sexc | 0.417 (0.22, 0.62) | 17.30 | 1 | <0.001 | |
| Infant Age | 0.022 (−0.08, 0.12) | 0.181 | 1 | 0.671 | |
| Firstbornd | 0.382 (−0.08, 0.91) | 2.457 | 1 | 0.117 | |
| Average Party Size | 0.025 (0.02, 0.03) | 25.70 | 1 | <0.001 | |
| Seasone | 0.433 (0.28, 0.59) | 29.30 | 1 | <0.001 | |
| Traveling | Juvenile Presenceb | 1.925 | 2 | 0.382 | |
| Female Juvenile | −0.103(−0.27, 0.07) | ||||
| Male Juvenile | −0.077 (−0.22, 0.06) | ||||
| Infant Sexc | −0.036 (−0.15, 0.07) | 0.443 | 1 | 0.506 | |
| Infant Age | 0.032 (−0.03, 0.10) | 1.031 | 1 | 0.310 | |
| Firstbornd | −0.011 (−0.17, 0.21) | 0.014 | 1 | 0.905 | |
| Average Party Size | 0.015 (0.01, 0.02) | 23.94 | 1 | <0.001 | |
| Seasone | −0.048 (−0.15, 0.05) | 0.850 | 1 | 0.357 |
Significance (p<0.05) is indicated in bold. CI = confidence interval; df = degrees of freedom.
Infant only is the reference category
Female is the reference category
Not firstborn is the reference category
Dry season is the reference category
Figure 1.
Mean ± SE proportion of time mothers spent engaged in social (grooming or playing) behavior with any member of the community including dependent offspring. ninfant only follows = 302; nfemale juvenile follows = 84; nmale juvenile follows = 115. Tukey’s post hoc test **p<0.001; *p<0.05; [*]p<0.10.
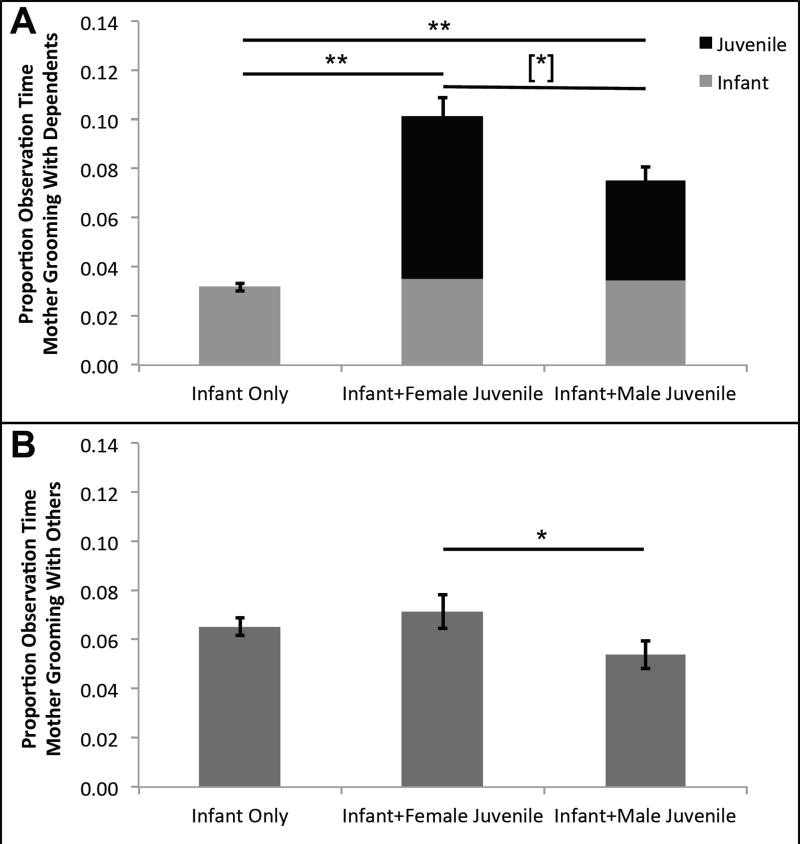
The difference in maternal social budgets was primarily attributable to mothers spending a significantly greater proportion of time grooming with dependent offspring when a juvenile of either sex was present (Table 2; Fig. 2A). As with the overall social budget, post hoc tests indicate that mothers with a female juvenile tended to spend more time engaged in grooming dependent offspring than mothers with a male juvenile (Fig. 2A.). Also, as with the overall social budget, mothers engaged in grooming with dependent offspring more during the dry season (mean ± SE: Dry = 0.067 ± 0.004; Wet = 0.040 ± 0.003; Table 3). Average party size was not a significant predictor of time mothers spent grooming with dependent offspring (Table 2). Mothers also spent a significantly smaller proportion of time grooming with other community members when a male juvenile offspring was present compared to when a female juvenile offspring was present (Table 3; Fig. 2B). Unsurprisingly, the proportion of time mothers spent grooming with other community members also increased with increasing average party size (Table 3). Additionally, the proportion of time mothers spent grooming with their infants did not differ based on the presence of a juvenile offspring (mean ± SE: Infant Only = 0.010 ± 0.001; Infant + Female Juvenile = 0.010 ± 0.002; Infant + Male Juvenile = 0.010 ± 0.002; Table 2). However, season was again a significant predictor as mothers spent significantly more time grooming with infants in the dry season compared to the wet season (mean ± SE: Dry = 0.038 ± 0.002; Wet = 0.026 ± 0.002; Table 2).
Table 2.
Results of GLMMs examining the relationship between presence of a juvenile offspring and the proportion of time mothers spent grooming and playing with their behaviorally dependent offspringa.
| Behavior | Parameter | Estimate (95% CI) | Wald χ2 | df | p |
|---|---|---|---|---|---|
| Playing With Dependents | Juvenile Presenceb | 0.504 | 2 | 0.777 | |
| Female Juvenile | −0.144 (−0.98, 0.74) | ||||
| Male Juvenile | −0.254 (−0.96, 0.48) | ||||
| Infant Sexc | 0.443 (−0.07, 0.99) | 2.755 | 1 | 0.097 | |
| Infant Age | −0.447 (−0.75, −0.14) | 8.619 | 1 | 0.003 | |
| Firstbornd | 0.202 (−0.62, 1.17) | 0.220 | 1 | 0.949 | |
| Average Party Size | 0.073 (0.04, 0.10) | 24.75 | 1 | <0.001 | |
| Seasone | −0.151 (−0.64, 0.33) | 0.381 | 1 | 0.537 | |
| Grooming With Dependents | Juvenile Presenceb | 57.00 | 2 | <0.001 | |
| Female Juvenile | 0.943 (0.67, 1.22) | ||||
| Male Juvenile | 0.670 (0.42, 0.92) | ||||
| Infant Sexc | 0.090 (−0.10, 0.28) | 0.835 | 1 | 0.361 | |
| Infant Age | −0.062 (−0.18, 0.06) | 1.081 | 1 | 0.299 | |
| Firstbornd | 0.013 (−0.30, 0.33) | 0.007 | 1 | 0.935 | |
| Average Party Size | 0.006 (−0.01, 0.02) | 0.984 | 1 | 0.321 | |
| Seasone | −0.559 (−0.76, −0.36) | 30.98 | 1 | <0.001 | |
| Playing With Infants | Juvenile Presenceb | 0.603 | 2 | 0.740 | |
| Female Juvenile | −0.159 (−0.99, 0.72) | ||||
| Male Juvenile | −0.279 (−0.99, 0.45) | ||||
| Infant Sexc | 0.411 (−0.10, 0.95) | 2.406 | 1 | 0.121 | |
| Infant Age | −0.446 (−0.75, −0.14) | 8.628 | 1 | 0.003 | |
| Firstbornd | 0.191 (−0.62, 1.14) | 0.202 | 1 | 0.653 | |
| Average Party Size | 0.072 (0.04, 0.10) | 24.21 | 1 | <0.001 | |
| Seasone | −0.147 (−0.63, 0.33) | 0.381 | 1 | 0.545 | |
| Grooming With Infants | Juvenile Presenceb | 0.569 | 2 | 0.753 | |
| Female Juvenile | 0.094 (−0.22, 0.39) | ||||
| Male Juvenile | −0.029 (−0.30, 0.24) | ||||
| Infant Sexc | 0.101 (−0.11, 0.31) | 0.902 | 1 | 0.342 | |
| Infant Age | −0.023 (−0.15, 0.10) | 0.131 | 1 | 0.717 | |
| Firstbornd | 0.025 (−0.32, 0.38) | 0.022 | 1 | 0.289 | |
| Average Party Size | 0.007 (−0.01, 0.02) | 1.124 | 1 | 0.881 | |
| Seasone | −0.574 (−0.78, −0.37) | 30.08 | 1 | <0.001 |
Significance (p<0.05) is indicated in bold. CI = confidence interval; df = degrees of freedom.
Infant only is the reference category
Female is the reference category
Not firstborn is the reference category
Dry season is the reference category
Figure 2.
Mean ± SE proportion of time mothers spent (A) grooming with infant and juvenile offspring and (B) grooming with all other members of the community. In (A) Gray sections indicate proportion of time playing with infant offspring. Black sections indicate the proportion of time playing with juvenile offspring. Standard error bars based on total proportion of time spent playing with infants and juveniles. ninfant only follows = 302; nfemale juvenile follows = 84; nmale juvenile follows = 115. Tukey’s post hoc tests **p<0.001; *p<0.05; [*]p<0.10.
Table 3.
Results of a GLMM examining the relationship between presence of a juvenile offspring and the proportion of time mothers spent grooming with other community members excluding behaviorally dependent offspringa.
| Parameter | Estimate (95% CI) | Wald χ2 | df | p |
|---|---|---|---|---|
| Juvenile Presenceb | 8.675 | 2 | 0.013 | |
| Female Juvenile | 0.333 (−0.23, 0.89) | |||
| Male Juvenile | −0.505 (−1.02, −0.005) | |||
| Infant Sexc | −0.287 (−0.69, 0.11) | 3.324 | 1 | 0.068 |
| Infant Age | −0.068 (−0.27, 0.13) | 1.253 | 1 | 0.263 |
| Firstbornd | −0.256 (−1.64, 0.94) | 0.040 | 1 | 0.842 |
| Average Party Size | 0.039 (0.02, 0.06) | 16.45 | 1 | <0.001 |
| Seasone | −0.253 (−0.56, 0.05) | 3.745 | 1 | 0.052 |
Significance (p<0.05) is indicated in bold. CI = confidence interval; df = degrees of freedom.
Infant only is the reference category
Female is the reference category
Not firstborn is the reference category
Dry season is the reference category
In contrast to grooming, the proportion of observation time that mothers spent playing with dependent offspring did not differ when a juvenile of either sex was present (Table 2; Fig. 3). This result was primarily attributable to the consistency of time spent playing with infants in the different conditions. That is, the proportion of time mothers spent playing with their infants did not differ based on the presence of a juvenile offspring (playing mean ± SE: Infant Only = 0.032 ± 0.002; Infant + Female Juvenile = 0.035 ± 0.004; Infant + Male Juvenile = 0.034 ± 0.004; Table 2). However, maternal play with dependents and play with infants alone did decrease with increasing infant age and increase with increasing party size (Table 2). Play between mothers and their juvenile offspring was extremely rare and observed in only two follows. Play between mothers and other members of the community was also rare occurring in just 43 of the 501 follows.
Figure 3.
Mean ± SE proportion of time mothers spent playing with infant and juvenile offspring. Gray sections indicate proportion of time playing with infant offspring. Black sections indicate the proportion of time playing with juvenile offspring. Standard error bars based on total proportion of time spent playing with infants and juveniles. ninfant only follows = 302; nfemale juvenile follows = 84; nmale juvenile follows = 115. There was no significant relationship between juvenile presence and proportion of time mothers spent playing with infant and juvenile offspring.
Maternal feeding budget
Notably, the greater social budget in the presence of juvenile offspring does not detract from feeding budgets. There was no difference in the proportion of time mothers spent feeding when traveling with an infant and juvenile as compared to just an infant (mean ± SE: Infant Only = 0.491 ± 0.011; Infant + Female Juvenile = 0.474 ± 0.018; Infant + Male Juvenile = 0.478 ± 0.017; Table 1). Infant sex, however, was a significant predictor of time spent feeding, with mothers spending more time feeding when their infant was female, compared to when their infant was male (mean ± SE: Female Infant = 0.510 ± 0.013; Male Infant= 0.467 ± 0.01; Table 1). Time spent feeding also decreased with increasing average party size (Table 1). There was also no difference in maternal diet quality based on the presence of a juvenile offspring; however infant sex (mean ± SE: Female Infant = 0.614 ± 0.018; Male Infant= 0.559 ± 0.016) and increasing average party size season were significantly associated with diet quality (SOM Table S3). Season also tended to predict maternal diet quality (mean ± SE: Dry = 0.597 ± 0.015; Wet = 0.559 ± 0.019; SOM Table S3).
Maternal traveling and resting budgets
Neither maternal time spent resting nor traveling was associated with juvenile presence, although both behaviors increased with increasing average party size (Table 1). Mothers also spent more time resting with a male infant (mean ± SE: Female Infant = 0.189 ± 0.009; Male Infant= 0.224 ± 0.008) and during the wet season (mean ± SE: Dry = 0.178 ± 0.007; Wet = 0.256 ± 0.011; Table 1).
Offspring survival
The best model predicting offspring survival each year from birth to 8 years of age included sibling presence and not firstborn status as indicated by a ΔAICc > 2 (Table 4). This model including sibling presence and not firstborn status was also the only model to differ from the null model. Over the whole model set, the relative importance of sibling presence was 0.80, while the relative importance of firstborn status was just 0.22, indicating sibling presence is more important to offspring survival than firstborn status. Losing a mother significantly increased the odds of dying, while increasing offspring age, maternal age, and maternal dominance rank decreased the odds of dying (Table 5). Sibling presence also tended (p = 0.053) to predict offspring survival with the absence of an older sibling tending to increase the odds of dying by a factor of 2.29 (Table 5).
Table 5.
Results from the discrete time event history model of offspring dying each year between birth and age 8 years with lowest AICca.
| Covariate | Odds Ratio |
95% CI | |
|---|---|---|---|
| Infant Age | 0.35*** | 0.19 | 0.65 |
| Infant Age2 | 1.10* | 1.02 | 1.18 |
| Sibling Presenceb | 2.29[*] | 1.01 | 5.53 |
| Maternal Presencec | 5.77** | 1.86 | 17.4 |
| Maternal Dominance | 0.05** | 0.01 | 0.37 |
| Maternal Age | 0.10* | 1.02 | 1.18 |
Odds ratios below 1 indicate the covariate is associated with lower odds of dying (greater odds of survival), while odds ratio above 1 indicate higher odds of dying (lower odds of survival). Significance of the covariates are based on Wald tests and indicated by asterisks.
0.05 ≤ p < 0.10;
0.01 ≤ p < 0.05;
0.001 ≤ p < 0.01;
p < 0.001. CI = confidence interval.
Sibling present is reference category
Mother present is reference category
Female interbirth interval
The best fitting model predicting interbirth interval length included both first IBI and sibling presence (Table 4). The relative importance of sibling presence was 0.90, while the relative importance of first IBI was 0.62. As expected, IBIs were significantly more likely to end if the infant died before age 2 years. Notably, IBIs were also 73% less likely to end when a sibling was absent compared to present (mean ± SE IBI length in years: Not Present = 5.57 ± 0.21; Present = 5.14 ± 0.18 among those IBIs that ended with a birth while the subsequent offspring was still alive). Conversely, IBIs following the first birth tended (p = 0.092) to be less likely to end than subsequent IBIs, suggesting shorter IBIs when older offspring are present. Maternal dominance rank was not a significant predictor of the IBI ending; however increasing maternal age was significantly associated with longer IBIs (Table 6).
Table 6.
Results from the discrete time event history model of interbirth interval ending with lowest AICca.
| Covariate | Odds Ratio | 95% CI | |
|---|---|---|---|
| Time | 4.15*** | 2.85 | 6.55 |
| First IBIb | 0.35[*] | 0.11 | 1.12 |
| Sibling Presencec | 0.27* | 0.84 | 0.75 |
| Maternal Dominance | 0.25 | 0.004 | 6.22 |
| Maternal Age | 0.85** | 0.76 | 0.93 |
| Infant died age 2 yearsd | 699.3*** | 119.6 | 6005.2 |
Odds ratios below 1 indicate lower odds of the birth interval ending, while odds ratio above 1 indicate higher odds of the birth interval ending. Significance of the covariates are based on Wald tests and indicated by asterisks.
0.05 ≤ p < 0.10;
0.01 ≤ p < 0.05;
0.001 ≤ p < 0.01;
p < 0.001. CI = confidence interval.
Later IBI is the reference category
Sibling present is reference category
Infant survived past age 2 years is reference category
Discussion
Overall, we found that mothers spent a greater proportion of time engaged in social interactions when a dependent juvenile was present. This increase was primarily attributable to time mothers spent grooming with their dependent juveniles. Notably, mothers maintained a consistent level of social interaction with their infant regardless of whether a dependent juvenile was present or not. Likewise, the proportion of time mothers spent feeding and their diet quality did not differ based on the presence of multiple dependent offspring. Thus mothers are not forgoing interactions with infants or their own feeding in order to interact with juveniles. Indeed, socializing was the only individual behavioral category that was significantly related to juvenile presence. It is therefore likely that mothers have variable strategies for offsetting time spent socializing with their offspring rather than consistently forgoing the same activity.
Overall, these results agree with an earlier study on the influence of siblings on infant socialization that found Gombe infants spent the same amount of time socializing with mothers regardless of whether they had a sibling and the lack of a sibling as a social partner was compensated for by increased interaction with other community members (Brent et al., 1997). However, whether primarily interacting with siblings versus other community members results in differences in social development or adult competence remains an intriguing question. It is possible that biasing behavior toward kin undermines the development of non-kin relationships, which may be particularly important to males for whom kinship is of limited importance in cooperative relationships (Langergraber et al., 2007). Alternately, juvenile siblings may be more tolerant, less aggressive social partners. Indeed, a recent study found that the majority of aggressive interactions between immature Kasekela chimpanzees involved non-maternal kin (Markham et al., 2015). Juveniles have an inclusive fitness stake in the success of their infant siblings, and in the absence of limited resources all three parties (infant, juvenile, and mother) stand to benefit from the infant’s success, yet whether juveniles benefit from interacting with their younger sibling remains an open question for future investigations.
Also of note is the difference in maternal activity budgets by infant sex, with variation in overall activity budgets apparently due to differences in time spent resting and feeding, with mothers of male infants spending more time resting and less time eating. Mothers with male offspring may need to be more vigilant as male infants are socially precocious compared to female infants (Lonsdorf et al., 2014) and mothers with sons are more gregarious and found in larger parties on average than mothers with daughters (Murray et al., 2014). While beyond the scope of the current investigation, future studies will examine how infant sex is related to maternal behavioral budgets, physiological stress levels, and subsequent reproductive effort.
Interestingly, rather than representing an investment that comes at a cost to subsequent reproductive efforts, having an older offspring present appears to have some benefit to chimpanzee mothers. Offspring who had their older sibling present tended to be more likely to survive each year between birth and 8 years of age than those who did not have an older sibling present, even after controlling for important predictors of offspring survival such as maternal presence and maternal rank. Furthermore, controlling for early infant death and other covariates associated with IBI length, IBIs in this study were more likely to close during an interval when an older offspring was present compared to intervals when they were not present, suggesting shorter IBIs when older offspring are present. A likely contributing factor for higher survival of offspring with older siblings is that mothers with juveniles and infants are in better condition than those without juveniles; however, it is important to note that our analyses accounted for maternal dominance status, which may be considered a proxy for female condition given that body mass is correlated with dominance rank and is more stable for high ranking females (Pusey et al., 2005). It is also possible that mothers who successfully raise one offspring are more likely to successfully raise another due to subtle differences in maternal behavior.
An alternate explanation for the relationship is that mothers with juvenile offspring may benefit from the additional strong social bond. A growing body of literature demonstrates that social bonds are related to increased survival and reproduction in both humans and nonhuman primates (e.g., baboons [Silk et al., 2003, 2009, 2010; Silk, 2007; Archie et al, 2014], Assamese macaques [Macaca assamensis; Schülke et al., 2010], humans [Berkman et al., 2004; Holt-Lunstad et al., 2010]). Grooming in particular is known to have both hygienic (Akinyi et al., 2013) and social (Goosen, 1981; Mitani and Watts, 2001; Arnold and Whiten, 2003) functions in nonhuman primates, and female grooming relationships, both giving and receiving, are associated with the ability to cope with stressful events (e.g., baboons [Engh et al., 2006; Crockford et al., 2008; Wittig et al., 2008], rhesus macaques [Macaca mulatta; Brent et al., 2011], crested black macaques [Macaca nigra; Aureli and Yates, 2010]). Thus, the increased time spent grooming with juvenile offspring, particularly female juveniles, may be valuable for the well-being of chimpanzee mothers and thus that of their infants. Notably, our results indicate that the tendency towards higher survival of offspring with older siblings is not due to differences in parity and confirm the results of a previous analysis that found firstborn infants in this community were as likely to survive as laterborn infants (Stanton et al., 2014). Mother-juvenile daughter social relationships may be especially important at Gombe since approximately 50% of females in the Kasekela community do not disperse and the strong bond between these natal females and their mothers persists into adulthood (Foerster et al., 2015). Recent work indicates that females with high ranking mothers tend to be less likely to disperse (Walker, 2015) and future studies should test whether the strength of bonds between mothers and their subadult daughters predicts dispersal patterns.
The mother-juvenile relationship also does not appear to be detrimental to other social interactions, as the time mothers devoted to grooming with other members of the community did not differ when a juvenile was present compared to when mothers were with infants only. The greater proportion of time mothers spent grooming with others when a female juvenile was present compared to a male juvenile may be due to differences in party composition, activity, or tenor (e.g., more commotion when juvenile males are present). In any case, mothers do not appear to sacrifice time devoted to other social relationships in order to provide affiliative care for their juvenile offspring.
During hominin evolution, despite nutritional dependence, juvenile siblings were possibly providing care for younger siblings and/or likely allowing mothers the opportunity to complete other tasks (Kramer 2011, 2014; Kramer and Otárola-Castillo, 2015). While juvenile chimpanzees are nutritionally independent and do not routinely provide overt help to their mothers, this does not preclude the possibility of a mutually beneficial mother-juvenile relationship. Mounting evidence from the present and earlier studies (Nakamura et al., 2014) suggests that mothers continue to be important to weaned chimpanzees and our results indicate that post-weaning care does not come at an immediate reproductive cost to the mother. Instead, chimpanzee mothers may benefit from increased social interaction with their older offspring.
One of the most interesting features of modern human reproduction is the birth ‘stacking’ of multiple dependent offspring that is made possible by weaning infants before they are nutritionally independent (Bogin, 2009). This rapid reproduction, despite large investment in offspring, was likely made possible in large part by an increased reliance on alloparental care (Hrdy, 2009; Kramer and Otárola-Castillo, 2015). While the discussion concerning who provided that care has traditionally focused on recruitment of adult helpers, including fathers and grandmothers (e.g. Hawkes et al., 1998; Gettler et al. 2011), recent exploratory models indicate that the earliest shifts towards modern human life histories could have been supported by help from within mother-offspring units (Kramer, 2014; Kramer and Otárola-Castillo, 2015). While chimpanzees are not perfect substitutes for extinct hominin species, examining the costs and benefits of the mother-juvenile relationship in the closely related great apes provides context when considering the emergence of helpful juveniles, birth stacking, and the evolution of modern human life history. Our results highlight the importance of considering the potential influence of juveniles during hominin evolution and suggest that some benefit of continued association with juveniles may have predated shifts towards early weaning, juvenile nutritional dependence, and overt alloparental sibling care.
Supplementary Material
Acknowledgments
We thank Tanzania National Parks, the Tanzania Wildlife Research Institute, and the Tanzanian Commission for Science and Technology for granting us permission to work in Gombe National Park. We also thank Dr. Jane Goodall for permission to work with the long-term data, the Jane Goodall Institute for funding long-term research, and the Gombe Stream Research Centre staff for maintaining data collection. This work was supported by grants from the National Institutes of Health (R00HD057992, R01 AI58715), the National Science Foundation (DBS-9021946, SBR-9319909, BCS-0452315, IIS-0431141; BSC-0648481; IOS-LTREB-1052693), the Leo S. Guthman Foundation, the Harris Steel Group, the Windibrow Foundation, the Carnegie Corporation, the University of Minnesota, Duke University, and the National Geographic Society. Finally, we thank V. Fiorentio, S. Reji, and K. Anderson for data management and the numerous assistants who participated in long-term data entry, as well as two anonymous reviewers, the associate editor, and editor for helpful comments.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Akaike H. Information theory as an extension of the maximum likelihood principle. In: Petrov BN, Csaki F, editors. Second International Symposium on Information Theory. Akaemiai Kiado; Budapest: 1974. pp. 267–281. [Google Scholar]
- Akinyi MY, Tung J, Jeneby M, Patel NB, Altmann J, Alberts SC. Role of grooming in reducing tick load in wild baboons (Papio cynocephalus) Anim. Behav. 2013;83:559–568. doi: 10.1016/j.anbehav.2012.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albers PCH, de Vries H. Elo-rating as a tool in the sequential estimation of dominance strengths. Anim. Behav. 2001;61:489–495. [Google Scholar]
- Altmann J. Observational study of behavior: sampling methods. Behaviour. 1974;49:227–266. doi: 10.1163/156853974x00534. [DOI] [PubMed] [Google Scholar]
- Altmann J, Alberts SC. Intraspecific variability in fertility and offspring survival in a nonhuman primate: Behavioral control of ecological and social sources. In: Wachter KW, Bulatao RA, editors. Offspring: Human Fertility Behavior in Biodemographic Perspective. The National Academies Press; Washington D.C.: 2003. pp. 140–169. [PubMed] [Google Scholar]
- Altmann J, Alberts SC. Growth rates in a wild primate population: ecological influences and maternal effects. Behav. Ecol. Sociobiol. 2005;57:490–501. [Google Scholar]
- Archie E, Tung J, Clark M, Altmann J, Alberts S. Social affiliation matters: both same-sex and opposite-sex relationships predict survival in wild female baboons. P. Roy. Soc. Lond. B Bio. 2014;281:20141261. doi: 10.1098/rspb.2014.1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold K, Whiten A. Grooming interactions among the chimpanzees of the Budongo Forest, Uganda: tests of five explanatory models. Behaviour. 2003;140:519–552. [Google Scholar]
- Aureli F, Yates K. Distress prevention by grooming others in crested black macaques. Biol. Lett. 2010;6:27–29. doi: 10.1098/rsbl.2009.0513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badescu I, Waats DP, Katzenberg MA, Sellen DW. Alloparenting is associated with reduced maternal lactation effort and faster weaning in wild chimpanzees. R. Soc. Open Sci. 2016;3:160577. doi: 10.1098/rsos.160577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartoń K. MuMIn: Multi-model inference. R package version 1.15.6. 2016 https://CRAN.R-project.org/package=MuMIn.
- Bates D, Maechler M, Bolker BM, Walker S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015;67:1–48. [Google Scholar]
- Berkman LF, Melchior M, Chastang JF, Niedhammer I, Leclerc A, Goldberg M. Social integration and mortality: A prospective study of French employees of electricity of France-Gas of France: The GAZEL cohort. Am. J. Epidemiol. 2004;159:167–174. doi: 10.1093/aje/kwh020. [DOI] [PubMed] [Google Scholar]
- Bogin B. Childhood, adolescence, and longevity: A multilevel model of the evolution of reserve capacity in human life history. Am. J. Hum. Biol. 2009;21:567–577. doi: 10.1002/ajhb.20895. [DOI] [PubMed] [Google Scholar]
- Brent L, Bramblett C, Bard K. The influence of siblings on wild infant chimpanzee social interaction. Behaviour. 1997;134:1189–1210. [Google Scholar]
- Brent LJN, Semple S, Dubuc C, Heistermann M, Maclarnon A. Social capital and physiological stress levels in free-ranging adult female rhesus macaques. Physiol. Behav. 2011;102:76–83. doi: 10.1016/j.physbeh.2010.09.022. [DOI] [PubMed] [Google Scholar]
- Bronikowski AM, Cords M, Alberts SC, Altmann J, Brockman DK, Fedigan LM, Pusey A, Stoinski T, Strier KB, Morris WF. Female and male life tables for seven wild primate species. Sci. Data. 2016;3:160006. doi: 10.1038/sdata.2016.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnham KP, Anderson D. Model Selection and Multi-model Inference. Springer; New York: 2002. [Google Scholar]
- Bygott D. Agonistic behaviour and dominance in wild chimpanzees. In: Hamburg D, McCown E, editors. The Great Apes. Benjamin-Cummings; Menlo Park: 1979. pp. 405–427. [Google Scholar]
- Charnov EL, Berrigan D. Why do female primates have such long lifespans and so few babies? Or life in the slow lane. Evol. Anthropol. 1993;1:191–194. [Google Scholar]
- Cheney DL. The acquisition of rank and the development of reciprocal alliances among free-ranging immature baboons. Behav. Ecol. Sociobiol. 1977;2:303–318. [Google Scholar]
- Clark C. A preliminary report on weaning among chimpanzees of the Gombe National Park, Tanzania. In: Chevalier-Skolinkoff S, Poirer F, editors. Primate Biosocial Development: Biological, Social and Ecological Determinants. Garland, New York: 1977. pp. 235–260. [Google Scholar]
- Clutton-Brock TH. Reproductive success. In: Clutton-Brock TH, editor. Studies of Individual Variation in Contrasting Breeding Systems. University of Chicago Press; Chicago: 1988. pp. 472–485. [Google Scholar]
- Clutton-Brock TH. The Evolution of Parental Care. Princeton University Press; Princeton: 1991. [Google Scholar]
- Cords M, Sheehan MJ, Ekernas LS. Sex and age differences in juvenile social priorities in female philopatric, nondespotic blue monkeys. Am. J. Primatol. 2010;72:193–205. doi: 10.1002/ajp.20768. [DOI] [PubMed] [Google Scholar]
- Crittenden AN, Conklin-Brittain NL, Zes DA, Schoeninger MJ, Marlowe FW. Juvenile foraging among the Hadza: Implications for human life history. Evol. Hum. Behav. 2013;34:299–304. [Google Scholar]
- Crockford C, Wittig RM, Whitten PL, Seyfarth RM, Cheney DL. Social stressors and coping mechanisms in wild female baboons (Papio hamadryas ursinus) Horm. Behav. 2008;53:254–65. doi: 10.1016/j.yhbeh.2007.10.007. [DOI] [PubMed] [Google Scholar]
- Dean MC. Tooth microstructure tracks the pace of human life-history evolution. P. Roy. Soc. Lond. B Bio. 2006;273:2799–2808. doi: 10.1098/rspb.2006.3583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emery Thompson M. Reproductive ecology of female chimpanzees. Am. J. Primatol. 2013;75:222–37. doi: 10.1002/ajp.22084. [DOI] [PubMed] [Google Scholar]
- Emery Thompson M, Wrangham RW. Diet and reproductive function in wild female chimpanzees (Pan troglodytes schweinfurthii) at Kibale National Park, Uganda. Am. J. Phys. Anthropol. 2008;135:171–181. doi: 10.1002/ajpa.20718. [DOI] [PubMed] [Google Scholar]
- Engh AL, Beehner JC, Bergman TJ, Whitten PL, Hoffmeier RR, Seyfarth RM, Cheney DL. Behavioural and hormonal responses to predation in female chacma baboons (Papio hamadryas ursinus) P. Roy. Soc. Lond. B Bio. 2006;273:707–712. doi: 10.1098/rspb.2005.3378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fairbanks L. Mother-infant behavior in vervet monkeys. Behav. Ecol. Sociobiol. 1988;23:157–165. [Google Scholar]
- Fairbanks L. Maternal investment throughout the life span. In: Whitehead P, Jolly C, editors. Old World Monkeys. Cambridge University Press; Cambridge, UK: 2000. pp. 341–367. [Google Scholar]
- Foerster S, McLellan K, Schroepfer-Walker K, Murray CM, Krupenye C, Gilby IC, Pusey AE. Social bonds in the dispersing sex: partner preferences among adult female chimpanzees. Anim. Behav. 2015;105:139–152. doi: 10.1016/j.anbehav.2015.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foerster S, Franz M, Murray CM, Gilby IC, Feldblum JT, Walker KK, Pusey AE. Chimpanzee females queue but males compete for social status. Sci. Reports. 2016;6:35404. doi: 10.1038/srep35404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox J, Weisberg S. An R companion to applied regression. second. Sage; Thousand Oaks CA: 2011. URL: http://socserv.socsci.mcmaster.ca/jfox/Books/Companion. [Google Scholar]
- Franz M, Alberts SC, Franz M. Social network dynamics: the importance of distinguishing between heterogeneous and homogeneous changes. Behav. Ecol. Sociobiol. 2015;69:2059–2069. doi: 10.1007/s00265-015-2030-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gettler LT, McDade TW, Feranil AB, Kuzawa CW. Longitudinal evidence that fatherhood decreases testosterone in human males. Proc. Natl. Acad. Sci. 2011;39:16194–16199. doi: 10.1073/pnas.1105403108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodall J. The Chimpanzees of Gombe: Patterns of Behavior. Harvard University Press; Cambridge, MA: 1986. [Google Scholar]
- Goosen C. On the function of allogrooming in Old-World monkeys. In: Hamburg D, McKown EA, editors. The Great Apes. Benjamin-Cummings; Menlo Park: 1981. pp. 491–499. [Google Scholar]
- Gurven M, Walker R. Energetic demand of multiple dependents and the evolution of slow human growth. P. Roy. Soc. Lond. B Bio. 2006;273:835–841. doi: 10.1098/rspb.2005.3380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halperin S. Temporary association patterns in free-ranging chimpanzees: An assessment of individual grouping preferences. In: Hamburg D, McCown E, editors. The Great Apes. Benjamin-Cummings; Menlo Park: 1979. pp. 491–499. [Google Scholar]
- Harrison X. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ. 2014;2:e616. doi: 10.7717/peerj.616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey PH, Martin RD, Clutton-Brock TH. Life histories in comparative perspective. In: Smutts B, Cheney DL, Seyfarth RM, Wrangham RW, editors. Primate Societies. University of Chicago Press; Chicago: 1987. pp. 181–196. [Google Scholar]
- Hawkes K, O’Connell JF, Blurton Jones NG, Alvarez H, Charnov EL. Grandmothering, menopause, and the evolution of human life histories. Proc. Natl. Acad. Sci. 1998;95:1336–1339. doi: 10.1073/pnas.95.3.1336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holt-Lunstad J, Smith TB, Layton JB. Social relationships and mortality risk: A meta-analytic review. PLoS Med. 2010;7:e1000316. doi: 10.1371/journal.pmed.1000316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horrocks J, Hunte W. Maternal rank and offspring rank in vervet monkeys: An appraisal of the mechanisms of rank acquisition. Anim. Behav. 1983;31:772–782. [Google Scholar]
- Hrdy SB. Mothers and Others. Belknap Press/Harvard University Press; Cambridge, MA: 2009. [Google Scholar]
- Janson C, van Schaik C. Ecological risk aversion in juvenile primates: slow and steady wins the race. In: Pereira M, Fairbanks L, editors. Juvenile Primates: Life History, Development, and Behavior. University of Chicago Press; Chicago: 1993. pp. 57–74. [Google Scholar]
- Jenks GF, Caspall FC. Error on choroplethic maps: definition, measurement, reduction. Ann. Assoc. Am. Geogr. 1971;61:217–244. [Google Scholar]
- Jones JH, Wilson ML, Murray C, Pusey AE. Phenotypic quality influences fertility in Gombe chimpanzees. J. Anim. Ecol. 2010;79:1262–1269. doi: 10.1111/j.1365-2656.2010.01687.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kano T. The Last Ape: Pygmy Chimpanzee Behavior and Ecology. Stanford University Press; Stanford: 1992. [Google Scholar]
- Kaplan H, Hill K, Lancaster J, Hurtado A. A theory of human life history evolution: Diet, intelligence, and longevity. Evol. Anthropol. 2000;9:156–185. [Google Scholar]
- Konnor M. The Evolution of Childhood: Relationships, Emotion, Mind. Harvard University Press; Cambridge, MA: 2010. [Google Scholar]
- Kramer KL. Maya Children: Helpers at the Farm. Harvard University Press; Cambridge, MA: 2005. [Google Scholar]
- Kramer KL. The evolution of human parental care and recruitment of juvenile help. Trends Ecol. Evol. 2011;26:533–540. doi: 10.1016/j.tree.2011.06.002. [DOI] [PubMed] [Google Scholar]
- Kramer KL. Why what juveniles do matters in the evolution of cooperative breeding. Hum. Nat. 2014;25:49–65. doi: 10.1007/s12110-013-9189-5. [DOI] [PubMed] [Google Scholar]
- Kramer KL, Otárola-Castillo E. When mothers need others: The impact of hominin life history evolution on cooperative breeding. J. Hum. Evol. 2015;84:16–24. doi: 10.1016/j.jhevol.2015.01.009. [DOI] [PubMed] [Google Scholar]
- Langergraber KE, Mitani JC, Vigilant L. The limited impact of kinship on cooperation in wild chimpanzees. Proc. Natl. Acad. Sci. 2007;104:7786–7790. doi: 10.1073/pnas.0611449104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazarus J, Inglis I. Shared and unshared parental investment, parent-offspring conflict and brood size. Anim. Behav. 1986;34:1791–1804. [Google Scholar]
- Lee P, Oliver J. Competition, dominance and the acquisition of rank in juvenile yellow baboons (Papio cyanocephalus) Anim. Behav. 1979;27:576–585. [Google Scholar]
- Leigh S. Evolution of human growth. Evol. Anthropol. 2001;10:223–236. [Google Scholar]
- Lenth RV, Hervé M. lsmeans: Least-squares means. R package version 2.19. 2015 https://CRAN.R-project.org/package=lsmeans.
- Lonsdorf EV, Markham AC, Heintz MR, Anderson KE, Ciuk DJ, Goodall J, Murray CM. Sex differences in wild chimpanzee behavior emerge during infancy. PloS One. 2014;9:e99099. doi: 10.1371/journal.pone.0099099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markham AC, Santymire RM, Lonsdorf EV, Heintz MR, Lipende I, Murray CM. Rank effects on social stress in lactating chimpanzees. Anim. Behav. 2014;87:195–202. doi: 10.1016/j.anbehav.2013.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markham AC, Lonsdorf EV, Pusey AE, Murray CM. Maternal rank influences the outcome of aggressive interactions between immature chimpanzees. Anim. Behav. 2015;100:192–198. doi: 10.1016/j.anbehav.2014.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitani J, Watts D, Muller M. Recent developments in the study of wild chimpanzee behavior. Evol. Anthropol. 2002;11:9–25. [Google Scholar]
- Mitani JC, Watts DP. Why do chimpanzees hunt and share meat? Anim. Behav. 2001;61:915–924. [Google Scholar]
- Murray CM, Eberly LE, Pusey AE. Foraging strategies as a function of season and rank among wild female chimpanzees (Pan troglodytes) Behav. Ecol. 2006;17:1020–1028. [Google Scholar]
- Murray CM, Lonsdorf EV, Eberly LE, Pusey AE. Reproductive energetics in free-living female chimpanzees (Pan troglodytes schweinfurthii) Behav. Ecol. 2009;20:1211–1216. [Google Scholar]
- Murray CM, Lonsdorf EV, Stanton MA, Wellens KR, Miller JA, Goodall J, Pusey AE. Early social exposure in wild chimpanzees: Mothers with sons are more gregarious than mothers with daughters. Proc. Natl. Acad. Sci. 2014;111:201409507. doi: 10.1073/pnas.1409507111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray CM, Stanton MA, Wellens KR, Santymire RM, Heintz MR, Lonsdorf EV. Maternal effects on offspring stress physiology in wild chimpanzees. Am. J. Primatol. 2016 doi: 10.1002/ajp.22525. [DOI] [PubMed] [Google Scholar]
- Nakamura M, Hayaki H, Hosaka K, Itoh N, Zamma K. Brief Communication: Orphaned male chimpanzees die young even after weaning. Am. J. Phys. Anthropol. 2014;153:139–143. doi: 10.1002/ajpa.22411. [DOI] [PubMed] [Google Scholar]
- Nitsch A, Faurie C, Lummaa V. Are elder siblings helpers or competitors? Antagonistic fitness effects of sibling interactions in humans. P. Roy. Soc. Lond. B Bio. 2013;280:20122313. doi: 10.1098/rspb.2012.2313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oksanen J, Blanchet FG, Kindt R, Legendre P, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H. vegan: Community ecology package. R package version 2.1–10. 2013 https://CRAN.R-project.org/package=vegan.
- Pagel M, Harvey P. Evolution of the juvenile period in mammals. In: Pereira M, Fairbanks L, editors. Juvenile Primates: Life History, Development and Behavior. University of Chicago Press; Chicago: 1993. pp. 28–37. [Google Scholar]
- Pereira M, Altmann J. Development of social behavior in free-living nonhuman primates. In: Watts E, editor. Nonhuman Primate Models for Human Growth and Development. Liss, New York: 1985. pp. 217–309. [Google Scholar]
- Pereira ME, Fairbanks LA. Juvenile Primates: Life History, Development, and Behavior. University of Chicago Press; New York: 2002. [Google Scholar]
- Pontzer H, Wrangham R. Ontogeny of ranging in wild chimpanzees. Int. J. Primatol. 2006;27:295–309. [Google Scholar]
- Pusey A. Behavioural changes at adolescence in chimpanzees. Behaviour. 1990;115:203–246. [Google Scholar]
- Pusey A, Williams J, Goodall J. The influence of dominance rank on the reproductive success of female chimpanzees. Science. 1997;277:828–831. doi: 10.1126/science.277.5327.828. [DOI] [PubMed] [Google Scholar]
- Pusey AE. Mother-offspring relationships in chimpanzees after weaning. Anim. Behav. 1983;31:363–377. [Google Scholar]
- Robson S, van Schaik C, Hawkes K. The derived features of human life history. In: Paine R, Hawkes K, editors. The Evolution of Human Life History. School of American Research Press; Santa Fe: 2006. pp. 17–44. [Google Scholar]
- Ross C, Jones K. The evolution of primate reproductive rates. In: Lee P, editor. Comparative Primate Socioecology. Cambridge University Press; Cambridge, UK: 1999. pp. 73–110. [Google Scholar]
- Schülke O, Bhagavatula J, Vigilant L, Ostner J. Social bonds enhance reproductive success in male macaques. Curr. Biol. 2010;20:2207–2210. doi: 10.1016/j.cub.2010.10.058. [DOI] [PubMed] [Google Scholar]
- Sear R, Mace R. Who keeps children alive? A review of the effects of kin on child survival. Evol. Hum. Behav. 2008;29:1–18. [Google Scholar]
- Silk JB. The adaptive value of sociality in mammalian groups. Philos. T. R. Soc. B. 2007;362:539–559. doi: 10.1098/rstb.2006.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silk JB, Alberts SC, Altmann J. Social bonds of female baboons enhance infant survival. Science. 2003;302:1231–1234. doi: 10.1126/science.1088580. [DOI] [PubMed] [Google Scholar]
- Silk JB, Beehner JC, Bergman TJ, Crockford C, Engh AL, Moscovice LR, Wittig RM, Seyfarth RM, Cheney DL. The benefits of social capital: close social bonds among female baboons enhance offspring survival. P. Roy. Soc. Lond. B Bio. 2009;276:3099–3104. doi: 10.1098/rspb.2009.0681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silk JB, Beehner JC, Bergman TJ, Crockford C, Engh AL, Moscovice LR, Wittig RM, Seyfarth RM, Cheney DL. Strong and consistent social bonds enhance the longevity of female baboons. Curr. Biol. 2010;20:1359–1361. doi: 10.1016/j.cub.2010.05.067. [DOI] [PubMed] [Google Scholar]
- Singer JD, Willett JB. It’s about time: using discrete-time survival analysis to study duration and the timing of events. J. Educ. Behav. Stat. 1993;18:155–195. [Google Scholar]
- Stanton MA, Lonsdorf EV, Pusey AE, Goodall J, Murray CM. Maternal behavior by birth order in wild chimpanzees (Pan troglodytes) Curr. Anthropol. 2014;55:483–489. doi: 10.1086/677053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strier KB, Altmann J, Brockman DK, Bronikowski AM, Cords M, Fedigan LM, Lapp H, Liu X, Morris WF, Pusey AE, Stoinski TS, Alberts SC. The Primate Life History Database: A unique shared ecological data resource. Methods Ecol. Evol. 2010;1:199–211. doi: 10.1111/j.2041-210X.2010.00023.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stumpf R. Chimpanzees and bonobos: diversity within and between speciess. In: Campbel CJ, Fuentes A, MacKinnon KC, Panger M, Bearder SK, editors. Primates in Perspective. Oxford University Press; Oxford: 2007. pp. 321–344. [Google Scholar]
- Surbeck M, Hohmann G. Intersexual dominance relationships and the influence of leverage on the outcome of conflicts in wild bonobos (Pan paniscus) Behav. Ecol. Sociobiol. 2013;67:1767–1780. [Google Scholar]
- Surbeck M, Mundry R, Hohmann G. Mothers matter! Maternal support, dominance status and mating success in male bonobos (Pan paniscus) P. Roy. Soc. Lond. B Bio. 2011;278:590–598. doi: 10.1098/rspb.2010.1572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Symonds M, Moussalli A. A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaikie’s information criterion. Behav. Ecol. Sociobiol. 2011;65:13–21. [Google Scholar]
- Trivers RL. Parental investment and sexual selection. In: Cambell B, editor. Sexual Selection and the Descent of Man. Aldine; Chicago: 1972. pp. 1871–1971. [Google Scholar]
- van de Rijt-Plooij HHC, Plooij FX. Growing independence, conflict and learning in mother-infant relations in free-ranging chimpanzees. Behaviour. 1987;101:1–86. [Google Scholar]
- van Noordwijk MA. From maternal investment to lifetime maternal care. In: Mitani J, Call J, Kappeler P, Palombit R, Silk J, editors. Evolution of Primate Societies. The University of Chicago Press; Chicago and London: 2012. pp. 321–342. [Google Scholar]
- Walker KK. Ph.D. Dissertation. Duke University; 2015. Dispersal and integration in female chimpanzees. [Google Scholar]
- Wallis J. A survey of reproductive parameters in the free-ranging chimpanzees of Gombe National Park. J. Repr. Fert. 1997;109:297–307. doi: 10.1530/jrf.0.1090297. [DOI] [PubMed] [Google Scholar]
- Watts D, Pusey A. Behavior of juvenile and adolescent great apes. In: Pereira M, Fairbanks L, editors. Juvenile Primates: Life History, Development, and Behavior. University of Chicago Press; Chicago: 1993. pp. 148–167. [Google Scholar]
- Wells JCK. Ecological volatility and human evolution: A novel perspective on life history and reproductive strategy. Evol. Anthropol. 2012;21:277–288. doi: 10.1002/evan.21334. [DOI] [PubMed] [Google Scholar]
- Wells JCK, Stock JT. The biology of the colonizing ape. Yearb. Phys. Anthropol. 2007;50:191–222. doi: 10.1002/ajpa.20735. [DOI] [PubMed] [Google Scholar]
- Wittig RM, Crockford C, Lehmann J, Whitten PL, Seyfarth RM, Cheney DL. Focused grooming networks and stress alleviation in wild female baboons. Horm. Behav. 2008;54:170–177. doi: 10.1016/j.yhbeh.2008.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.