The last decade has witnessed the evolution of anaplastic lymphoma kinase (ALK)-rearranged non-small-cell lung cancer (NSCLC) from an unknown entity into one of the most successful examples of precision oncology. Currently, four ALK tyrosine kinase inhibitors (TKIs) have gained approval for management of advanced ALK-rearranged NSCLC: crizotinib, ceritinib, alectinib, and brigatinib [1–3]. Crizotinib was the first multitargeted ALK inhibitor approved (2011); however, the extended long-term outcomes of patients have been seldom reported [4]. We sought to evaluate cases with 5-year progression-free survival (PFS) and overall survival (OS) in patients who received crizotinib as their initial ALK-directed therapy.
Case 1 was a 76 year-old never smoker woman with recurrent advanced lung adenocarcinoma and ALK rearrangement (78% cells positive by ALK FISH). She received crizotinib 250 mg twice daily with subsequent dose reductions (gastro-intestinal adverse events and fatigue) to 250 mg daily. She attained at a partial response that was sustained for over 76 months (Fig. 1A). Case 2 was a 47 year-old never smoker man with stage IV lung adenocarcinoma and ALK rearrangement (58% cells positive by ALK FISH). He received crizotinib 250 mg twice daily and attained a partial response that was sustained for over 60 months (Fig. 1B). To better understand how frequent long-term disease control occurred in other cases, we retrospectively queried lung cancer-patient pairs with ALK rearrangements from our institution and evaluated duration of crizotinib use, other ALK inhibitor use, PFS, OS, and drug discontinuation due to toxicity. Pathologic data, tumor genotype, type/dose of ALK TKI, radiographic images, and survival were amassed from retrospective chart extraction.
Figure 1. Cases with ≥5 years (≥60 months) after initiation of crizotinib in 2 patients with advanced ALK-rearranged lung cancers.
A. Case no. 1 with computed tomography (CT) of neck before starting and during year 6 of use of crizotinib. Red circle indicates responsive right supraclavicular lymphadenopathy. B. Case no. 2 with CT of chest before starting and during year 5 of use of crizotinib. Red circles indicate responsive subpectoral lymph nodes.
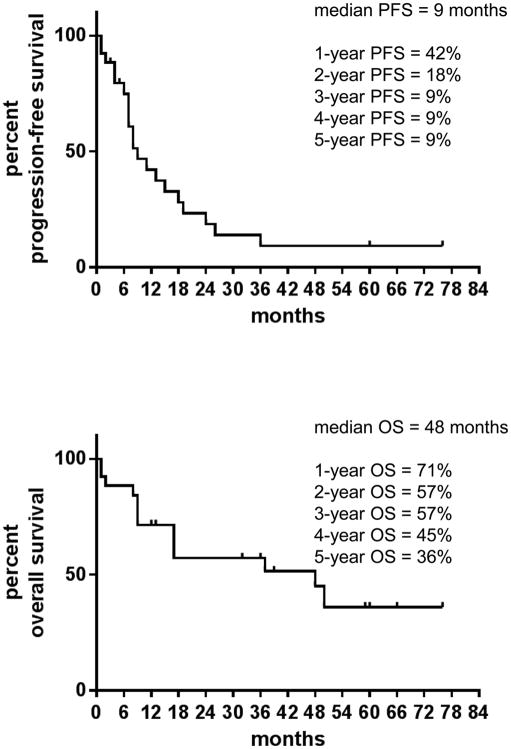
Our cohort identified 45/835 cases (5.3%) with positive ALK FISH. Of these, 26 patients with advanced (stage IV or recurrent) disease were given crizotinib as the initial ALK inhibitor (Fig. 2). Median age was 57 years (range 29–81), 13 (50%) were women, 15 (57%) were never-smokers (median pack-years of 0 [range 0–100]), ethnicity/race varied (22 White, 4 Asian), and all had NSCLC (24 adenocarcinoma, 1 adeno-squamous, 1 squamous). Although crizotinib was well-tolerated in most cases, 3 patients discontinued crizotinib prior to progression due to toxicities (Fig. 1). An additional 11 patients continued crizotinib following initial radiographic disease progression, and 10 patients received next generation ALK inhibitors (Fig. 2). The median PFS on crizotinib was 9 months, with an estimated 5-year PFS of 9% (Fig. 3A). The median OS was 48 months, with an estimated 5-year OS of 36% (Fig. 3B).
Figure 2. Use of ALK inhibitors, anti-cancer, and palliative therapies for the first 5 years (60 months) after initiation of crizotinib in 26 patients with advanced ALK-rearranged lung cancers.
The type of ALK TKI, chemotherapy, or best supportive care are indicated by different colors. Progressive disease by RECIST is indicated by (P), toxicity to crizotinib by (T), use of radiotherapy by (X), loss of patient follow-up by (L), death by (D) and ongoing therapy/survival by (+).
Figure 3. Progression-free and overall survival of patients with ALK-rearranged lung cancers after initiation of crizotinib.
A. Progression-free survival (PFS) curve with median and yearly estimates. B. Overall survival (OS) curve with median and yearly estimates. Data was collected and managed using the REDCap electronic data capture at our institution. Tumor genotype was performed using the ALK fluorescence in situ hybridization (FISH) break-apart probe in diagnostic tumor samples. Response evaluation criteria in solid tumors was utilized when reviewing target/non-target radiographic images. PFS and OS were calculated in months (rounded to the nearest full month) from time of initiation of crizotinib. PFS and OS were explored using the Kaplan–Meier method. Statistical analyses and curves were obtained with the GraphPad Prism 6 software (GraphPad Software, La Jolla, CA).
Advanced ALK-rearranged NSCLC can now be palliated with multiple approved ALK inhibitors. Our cases confirm several themes regarding this disease entity and the role of TKIs: 1) a minority (<10%) of patients can achieve protracted disease control (PFS of ≥5 years) with crizotinib monotherapy; 2) continuation of crizotinib beyond disease progression may be an effective strategy in some patients (particularly those with sites that might be palliated with other therapeutic modalities, i.e. radiotherapy for palliation of brain metastases) [4]; and 3) use of sequential ALK TKIs can extend median OS in this population to nearly 4 years [5] with more than one third of patients are alive 5 years after initiation of single or sequential ALK TKIs.
Acknowledgments
Funding/Grant Support: This work was funded in part through an American Cancer Society grant RSG 11-186 (DBC), National Cancer Institute grants P50CA090578 (DBC), R01CA169259 (SSK) and R21CA17830 (SSK), and internal donations to Beth Israel Deaconess Medical Center.
The authors would like to thank the brave patients and affiliated medical providers whom have contributed to our institutional review board-approved protocol BIDMC-2009-P000182 that is the basis of our cohort.
Conflict of interest: Daniel B. Costa has received consulting fees and honoraria from Pfizer, Boehringer Ingelheim and Ariad pharmaceuticals; outside the submitted work. Paul A. VanderLaan has received consulting fees from Gala Therapeutics; outside the submitted work. No other conflict of interest is stated from other authors.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. 2010;363:1693–703. doi: 10.1056/NEJMoa1006448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shaw AT, Kim DW, Mehra R, et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med. 2014;370:1189–97. doi: 10.1056/NEJMoa1311107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shaw AT, Gandhi L, Gadgeel S, et al. Alectinib in ALK-positive, crizotinib-resistant, non-small-cell lung cancer: a single-group, multicentre, phase 2 trial. Lancet Oncol. 2016;17:234–42. doi: 10.1016/S1470-2045(15)00488-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Costa DB, Shaw AT, Ou SH, et al. Clinical Experience With Crizotinib in Patients With Advanced ALK-Rearranged Non-Small-Cell Lung Cancer and Brain Metastases. J Clin Oncol. 2015;33:1881–8. doi: 10.1200/JCO.2014.59.0539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gainor JF, Tan DS, De PT, et al. Progression-Free and Overall Survival in ALK-Positive NSCLC Patients Treated with Sequential Crizotinib and Ceritinib. Clin Cancer Res. 2015;21:2745–52. doi: 10.1158/1078-0432.CCR-14-3009. [DOI] [PMC free article] [PubMed] [Google Scholar]