Abstract
The metabolism of immune cells affects their function and influences host immunity. This review explores how immune cell metabolic phenotypes reflect biochemical dependencies, and highlights evidence that the metabolic state of immune cells and nutrient availability can both alter immune responses. The central importance of oxygen, energetics, and redox homeostasis in immune cell metabolism, and how these factors are reflected in different metabolic phenotypes, is also discussed. Linking immune cell metabolic phenotype to effector functions is important to understand how altering metabolism can impact the way in which immune cells meet their metabolic demands and affect the immune response in various disease contexts.
Introduction
All cells regulate their metabolism to support the biochemical demands of cell-type specific functions. The biochemical requirements of immune cells vary among different effector subsets, and change over the course of an immune response. Naïve lymphocytes must rapidly engage a proliferative metabolic program when foreign antigens are encountered (Johnson et al., 2016), macrophages must support an enzymatic program to process phagocytosed material (D. Park et al., 2011; Van den Bossche et al., 2017), and neutrophils must undergo a rapid respiratory burst to effectively destroy pathogens (El-Benna et al., 2016). In each case, cellular metabolism is adapted to allow each immune cell type to carry out its unique function and protect the host from pathogens and malignancy. Emerging data demonstrate that the metabolic state of immune cell populations is intimately tied to cellular differentiation and the activation of effector functions. Concurrently, immune cells encounter variations in nutrients, temperature, pH, and O2 as they traffic throughout the body, and these microenvironmental factors also impact metabolism and immune cell functions. Understanding how the interactions among immune cell biochemical requirements, cellular metabolic state, and nutrient availability interact to shape the immune response is critical to move beyond “metabolic phenotyping” to a more complete understanding of immune cell metabolism.
Metabolic phenotypes are often studied in cell culture, where nutrients are in excess and immune cells are separated from other tissue-resident cells. In recent years, disease models and clinical studies have begun to dissect the influence that local or systemic environmental factors have on the metabolism of tumor cells and immune cells, and there is growing evidence that systemic metabolic factors and local nutrient limitations at immune effector sites can be obstacles to both antimicrobial and anti-tumor immunity (Flint et al., 2016). Many cancer chemotherapies that target nucleotide metabolism also cause immunosuppression, increasing the risk of infection in cancer patients. Furthermore, the notion that cancer therapies might act, in part, by altering the tumor microenvironment and affecting immune cell function has generated interest in targeting immune cell metabolism to treat cancer (Chang and E. L. Pearce, 2016). It also raises the possibility that drugs targeting cancer metabolism might impair anti-tumor immunity, underscoring the importance of understanding the differences and similarities between immune and tumor cell metabolism and how this affects immune responses. This review will provide a framework for understanding immune cell metabolic phenotypes and attempt to connect metabolic phenotypes to the biochemical requirements of various immune cells.
Overview of Immune Cell Metabolic Phenotypes
Resting lymphocytes circulate in the blood, and cells in lymphoid tissues carry out surveillance for foreign antigens. Biosynthetic processes for these cells are minimal and they rely primarily on the mitochondrial oxidation of glucose and lipids to meet the energetic demands of survival and antigen surveillance. Homeostatic cues provided by molecules such as interleukin-7 that regulate T cell survival also are required for maintenance of this metabolic program (Jacobs et al., 2010). T cell antigen receptor stimulation in the presence of inflammatory co-stimulation leads to activation of the phosphatidyl-inositide-3-kinase (PI3K)/Akt/mTORC1 signaling pathway and induction of Myc, which promotes both aerobic glycolysis and increased glutamine metabolism, and drives increased lymphocyte numbers and size (Frauwirth et al., 2002; R. Wang et al., 2011). Glucose uptake increases and becomes limiting for T cell cytokine production and proliferation (Jacobs et al., 2008). Mitochondrial oxidative metabolism also increases, although to an extent that is relatively less than the increase in aerobic glycolysis, leading to the notion that activated T cells rely predominantly on aerobic glycolysis (Figure 1)(van der Windt et al., 2012; R. Wang et al., 2011).
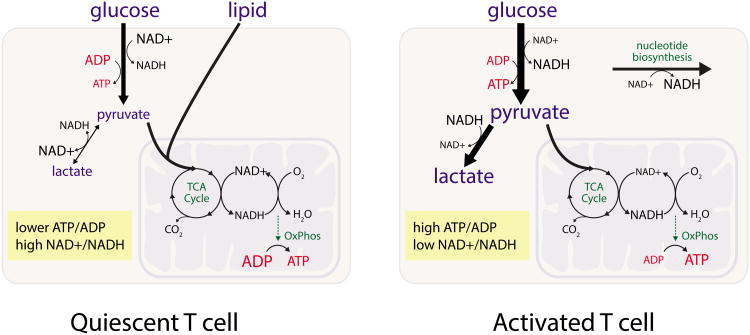
Figure 1. The metabolic phenotype of quiescent and activated T cells.
Quiescent T cells including naïve and memory cells exhibit a more oxidative metabolic phenotype characterized by low nutrient uptake and minimal lactate production. In contrast, activated T cells utilize aerobic glycolysis with increased glucose uptake and lactate production. Activated T cells still oxidize glucose in the mitochondrial TCA cycle, and the rate of glucose oxidation in activated T cells can be greater than that found in quiescent T cells. These different metabolic phenotypes may reflect the different metabolic requirements of these different cell states. Quiescent T cells oxidize limiting nutrients to maintain energy state and promote cell survival, while activated T cells alter metabolism to support cell proliferation and effector functions. The increased demand for synthesizing nucleotides and other oxidized biomass in proliferating cells results in a lower NAD+/NADH ratio and contributes to increased lactate production.
Aerobic glycolysis is a characteristic feature of many rapidly dividing cells, including cancer cells and immune cells, in which glucose is fermented to lactate, even as sufficient O2 is present to support oxidative phosphorylation (OxPhos) (Roos and Loos, 1973; Vander Heiden et al., 2009; T. Wang et al., 1976). It has been proposed that the advantage of this metabolic phenotype in proliferating cells is to help meet the biochemical requirements of biosynthesis and allow cell duplication (Lunt and Vander Heiden, 2011). Indeed, aerobic glycolysis tracks with DNA synthesis in proliferating lymphocytes (T. Wang et al., 1976). Additionally, aerobic glycolysis may also play important roles in T cell effector function. The glycolytic intermediate phosphoenolpyruvate has been suggested to promote calcium signaling essential for T cell activation (Ho et al., 2015), and interferon-γ has been shown to be reliant on high rates of glucose metabolism through both transcriptional and translational regulation (Cham and Gajewski, 2005; Chang et al., 2013; Peng et al., 2016).
Activation of lymphocytes is followed by differentiation into cell subsets with specific effector or suppressor functions, and ultimately differentiation into long-lived memory cells. Where studied, each T cell subset has been found to display a unique metabolic program that supports the biochemical requirements of their distinct cellular functions (Beier et al., 2015; Gerriets et al., 2015; Michalek et al., 2011; Oestreich et al., 2014; Shi et al., 2011). In general, effector T cell subsets predominantly display a glycolytic phenotype, while the anti-inflammatory regulatory T cell (Treg) subsets are characterized by the use of mitochondrial oxidative phosphorylation (Angelin et al., 2017). Under certain conditions, Treg cells have been observed to exhibit aerobic glycolysis downstream of PI3K/Akt/mTORC1 signaling (Zeng et al., 2013), but this metabolic phenotype is associated with reduced expression of the Treg transcription factor, FoxP3, and decreased suppressive function (Gerriets et al., 2016; Huynh et al., 2015; Y. Park et al., 2013; Shrestha et al., 2015). Memory T cells return to an oxidative phenotype when they become quiescent, and like naïve T cells consume both glucose and lipids (Figure 1)(Phan et al., 2016; van der Windt et al., 2012). However, memory cells are metabolically distinct from naïve T cells in that they are poised to rapidly engage a program involving increased aerobic glycolysis upon re-encounter with antigens (Gubser et al., 2013; van der Windt et al., 2013).
Metabolic reprogramming in response to immune receptor stimulation is a common theme among many classes of immune cells in addition to lymphocytes. Stimulated macrophages and dendritic cells rapidly induce aerobic glycolysis following activation of pattern recognition receptors such as Toll-like receptors (TLRs) (O'Neill and E. J. Pearce, 2016). Thus, aerobic glycolysis is not restricted to proliferating immune cells and instead better associates with cells undergoing anabolic processes. For example, TLR-stimulated dendritic cells increase glycolysis to support new lipid synthesis for rapid expansion of the endoplasmic reticulum and Golgi, processes necessary for production and secretion of immune regulatory cytokines (Everts et al., 2014). Like T cell subsets, myeloid cells have functionally distinct phenotypes associated with different metabolic programs. Classically activated macrophages that promote inflammation utilize glycolysis and glutamine metabolism to generate large quantities of succinate that enhances inflammation and production of interleukin-1 (Tannahill et al., 2013). In contrast, macrophages that play a role in tissue healing and inflammatory resolution display more mitochondrial lipid oxidation (Vats et al., 2006). A key differentiator between these metabolic phenotypes may arise in the mitochondria, where rather than engaging in conventional electron transport, altered electron flow through the succinate dehydrogenase complex promotes reactive oxygen species (ROS) generation and inflammation (Mills et al., 2016). Inhibition of respiration in myeloid cells as a consequence of nitric oxide production for host defense, may also be more tolerable because unlike antigen-specific lymphocytes, myeloid cells can be regenerated by the bone marrow if mitochondrial inhibition results in cell death (Brown and Borutaite, 2002; Everts et al., 2012). Macrophage metabolism has been recently reviewed in detail (Van den Bossche et al., 2017).
Ultimately, each immune cell metabolic phenotype is dependent on acquisition of appropriate nutrients. Indeed, forced expression of the glucose transporter Glut1 is associated with inflammatory T cells, and is sufficient to promote a systemic lupus erythematosus-like phenotype in transgenic mice (Michalek et al., 2011). Overexpression of Glut1 is also sufficient to increase the inflammatory character of macrophages (Freemerman et al., 2014). Conversely, Glut1-deficiency lowers glucose uptake and reduces T cell proliferation and inflammatory function in vivo. Tregs, in contrast, are less dependent on glucose metabolism and appear to function normally in the absence of Glut1 (Macintyre et al., 2014). Access to amino acids is also critical, as cells deficient in specific amino acid transporters fail to activate mTORC1 and induce T cell-mediated inflammation (Nakaya et al., 2014; Sinclair et al., 2013). Activation of mTORC1 can reprogram cell metabolism to support proliferation (Düvel et al., 2010), and by extension, the availability of amino acids and other nutrients in the microenvironment will have a profound impact on immune cell metabolism and function. Understanding the interplay between metabolism, immune subset differentiation and effector function requires a deeper understanding of how these various metabolic phenotypes support these functions and will shed light on how cells adapt to different physiological environments.
Oxygen, Oxidative Phosphorylation, and Aerobic Glycolysis
Most immune cell metabolic phenotypes are considered with respect to cellular energetics, including the manner by which cells produce ATP to support otherwise unfavorable reactions (Box 1). Oxidative cells produce a larger fraction of cellular ATP from mitochondrial oxidative phosphorylation, while glycolytic cells produce relatively more ATP from glycolysis. Some immune cells transition between oxidative and glycolytic metabolic phenotypes over the course of an immune response, and manipulation of this dichotomy can impact immunity. In exploring the importance of these metabolic phenotypes, it is helpful to recall the central importance of O2 in metabolism. Metazoan animal life is distinctly aerobic, and even brief interruptions in O2 supply can have profound effects on cellular function and viability. ATP production by either oxidative phosphorylation or glycolysis requires nutrient oxidation, i.e. removal of electrons from nutrients such as glucose (Vander Heiden and DeBerardinis, 2017)(Figure 1, Figure 2). Oxygen is the prototypic oxidizing agent, and its role as the terminal electron acceptor in the mitochondrial respiratory chain is generally appreciated. In addition, O2 is directly or indirectly required for numerous biochemical reactions (Goldfine, 1965; Raymond and Segrè, 2006). Oxygen is essential for the synthesis of sterols, unsaturated fatty acids, as well as the diverse biosynthetic, signaling, and epigenetic functions carried out by the large family of αKG-dependent dioxygenases (Loenarz and Schofield, 2011). Within the immune system, O2 has a particularly important role in granulocyte ROS production for host defense (El-Benna et al., 2016; Mills et al., 2016; West et al., 2011). The amount of O2 consumed by non-mitochondrial reactions can be physiologically significant (Banh et al., 2016).
Box 1. Terminology.
| Term | Common Definition | Considerations | |
|---|---|---|---|
| Oxidative Cell | A cell that converts most of the consumed glucose to CO2 in the mitochondrial TCA cycle. | The distinguishing feature of a glycolytic cell is lactate production. Glycolytic cells still rely on oxidative TCA cycle flux and mitochondrial respiration. In fact, O2 consumption can be higher in glycolytic cells as compared to ‘oxidative’ quiescent cells, as total nutrient uptake is increased. Fuel oxidation is required for survival of all cells, and both pyruvate reduction to lactate and O2 reduction to water enable fuel oxidation. | |
| Glycolytic Cell | A cell that converts most of the consumed glucose to lactate. | ||
| Aerobic glycolysis | High rate of lactate production from glucose despite normoxia. | A high rate of lactate production does not mean that mitochondrial respiration is necessarily suppressed. SRC implies that O2 consumption is less than maximum, possibly because a high energy state limits use of the proton gradient to drive ATP synthesis. The total respiratory capacity of a cell is a measure of the capacity of cells to make ATP via oxidative phosphorylation, but also the capacity for fuel oxidation and NADH recycling. Cells with SRC can increase nutrient oxidation if demands for either ATP or biomass increase provided nutrients remain available. | |
| Spare Respiratory Capacity (SRC) | SRC is the difference between the cellular O2 consumption rate (OCR), and the OCR after cells are treated with an uncoupling agent that disrupts the mitochondrial proton gradient and allows maximal O2 consumption. | ||
| Energy State |
|
Energy state regulates mitochondrial respiratory rate. Increased ATP production from aerobic glycolysis can decrease mitochondrial respiration by increasing the energy state. This interdependence of glycolysis and mitochondrial respiration has been referred to as the Crabtree Effect, although some authors have objected to this term as the Crabtree Effect is caused by depletion of inorganic phosphate. |
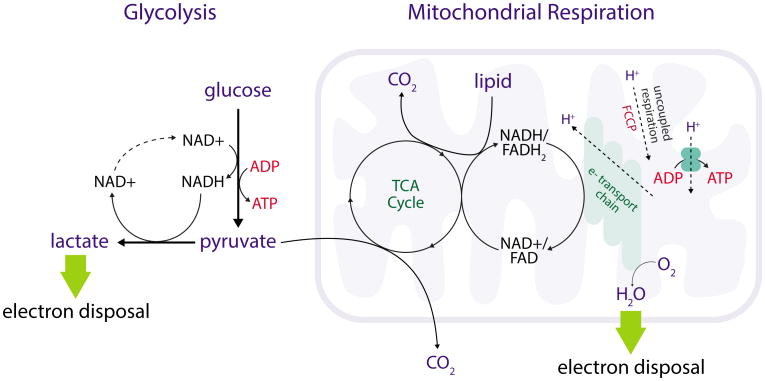
Figure 2. Relationship between glycolysis, oxygen consumption, ATP production and redox metabolism.
Glycolysis and mitochondrial respiration both supply cellular ATP. Production of lactate from pyruvate allows NAD+ regeneration and maintenance of redox balance for glycolysis. Redox balance is maintained when pyruvate or fatty acids from lipids are oxidized in the mitochondrial TCA cycle by transfer of electrons to O2 via the electron transport chain. When O2 consumption is coupled to ATP production, the proton (H+) gradient across the inner mitochondrial membrane generated by the electron transport chain is used to drive ATP synthesis (oxidative phosphorylation). Oxygen consumption can also be uncoupled from ATP production if there is another route for protons to cross the inner mitochondrial membrane (uncoupled respiration). This can occur via endogenous uncoupling proteins or via pharmacologic agents such as FCCP that are used to assess spare respiratory capacity. Production of lactate or consumption of O2 at rates that are stoichiometric with nutrient oxidation is necessary for electron disposal and redox homeostasis in cell metabolism.
Despite its central importance to metabolic phenotypes, O2 concentration is often an experimental unknown, and when it has been measured in vivo, technical considerations can influence measurement accuracy (Wilson, 2008). Oxygen concentration is linearly related to the partial pressure of O2 (pO2). In experiments performed in standard tissue culture conditions, media pO2 is near 145 mm Hg, or an O2 concentration of ∼200 μM. However, it has been estimated that the pO2 adjacent to an adherent cell monolayer can vary significantly, with reported values ranging from 0 mm Hg to greater than 100 mm Hg (Bambrick et al., 2011; Metzen et al., 1995; Wolff et al., 1993). The concentration of any metabolic substrate, including O2, is a balance between its rate of appearance and disappearance. As pertains to O2, this is a balance between O2 delivery and the oxygen consumption rate (OCR). In tissue culture, O2 delivery is determined by atmospheric conditions, media depth and aeration, and O2 diffusion. Oxygen consumption rate is determined by several factors discussed below, including cell number and metabolic state. Therefore, cells cultured in “normoxia” (i.e. unregulated atmospheric pO2) can in fact experience relative hypoxia if cells with high OCR are grown at high density. Conversely, cells with low OCR at low density can be exposed to supraphysiologic O2 concentrations in culture. In tissues, pO2 is tightly controlled within a narrow range, as O2 delivery via the vasculature is highly regulated (Duncker and Bache, 2008; Poole et al., 2013). Large contributors to O2 delivery include arterial oxygen content (CaO2), capillary density and blood flow, distance from the nearest blood vessel, and—importantly—tissue pO2 and as well as metabolic regulators of microvascular function and oxyhemoglobin unloading.
Oxygen delivery has perhaps been most studied in cardiac physiology. The heart extracts ∼70% of the coronary CaO2 under basal conditions to support the high ATP needs of beating cardiomyocytes (Deussen et al., 2011). The coronary sinus, which drains the heart, is thought to be the most deoxygenated blood of the body with a pO2 near 20 mm Hg (Mik et al., 2009). This illustrates that the most aerobic organ of the body (gram-1) maintains a low tissue pO2 yet relies heavily on oxidative metabolism to maximize ATP production and carry out its physiological function for the organism. Thus, pO2 may not always be a good predictor of metabolic phenotype. This conclusion is supported by the fact that hypoxic stem cell microenvironments within the bone marrow are largely dependent on cellularity, i.e. low pO2 in the bone marrow is in part determined by the high O2 consumption of cells in those niches (Spencer et al., 2014). It has also been observed that O2 consumption by non-mitochondrial dioxygenases can impact pO2 in poorly vascularized tumors (Banh et al., 2016).
A common misconception is that the interdependence between pO2 on OCR is entirely reciprocal, and that pO2 directly determines the rate of cellular respiration and oxidative phosphorylation. In this view, aerobic glycolysis in proliferating cells is a paradox. The rate of oxidative phosphorylation is controlled by multiple variables in addition to pO2, including availability of oxidizable substrates, mitochondrial content, mitochondrial NAD+/NADH ratio, pH, and cellular energy state (Wilson, 2015; Wilson et al., 1974). ATP production is regulated to match ATP consumption, and all aspects of mitochondrial metabolism – TCA flux, NAD(H) metabolism, and respiration – are tightly coupled in most cells (Figure 2). It has been argued when pO2 becomes rate-limiting for mitochondrial respiration, the immediate decrease in cellular energy state and increased [ADP] can maintain mitochondrial respiratory rate (Wilson, 2015). Thus, reliance on pO2 alone to adapt metabolism to microenvironmental conditions has energetic and redox consequences, and cells have evolved additional means to adapt metabolism to hypoxia.
Cells sense pO2 via the EGLN (also called PHD) family of enzymes (Kaelin et al., 2016). These enzymes are α-ketoglutarate-dependent dioxygenases, with a Km for O2 >200 μM, well above tissue concentrations, making their enzymatic activity very responsive to changes in pO2 (Hirsilä et al., 2003). EGLNs hydroxylate the α-subunit of the hypoxia-inducible factor (HIFα), targeting this protein for degradation. HIF transcriptional activity is suppressed under conditions of high EGLN activity when O2 is present at sufficient concentration (Kaelin and Ratcliffe, 2008). A subset of HIF transcriptional targets increase glucose uptake and glycolysis, and actively inhibit pyruvate oxidation (Kim et al., 2006; Papandreou et al., 2006). These metabolic adaptations are predicted to (1) increase the contribution of substrate-level phosphorylation from glycolysis to ATP production, and (2) limit the reduction in mitochondrial NAD+/NADH ratio under hypoxia. These adaptations are important to decrease mitochondrial respiration and match O2 consumption to O2 delivery, as they mitigate changes in energy state or mitochondrial NAD+/NADH ratio that might preserve respiratory rate at lower pO2. Additionally, decreased pyruvate oxidation under hypoxia might limit mitochondrial hyperpolarization and ROS production. Longer-term, hypoxia and HIF stabilization impairs mitochondrial biogenesis (Zhang et al., 2007) and alters cellular cytochrome content (Fukuda et al., 2007), which can decrease total cellular respiratory capacity. Viewed from the perspective of a proliferating cell, the HIF metabolic program favors pyruvate reduction over oxidation, preserving NAD+/NADH ratio to facilitate oxidative biosynthesis when a major electron acceptor, O2, is in limited supply.
The importance of mitochondrial respiration in cellular energetics is more widely appreciated than the role of the respiratory chain in cellular redox homeostasis. Oxygen reduction to water is one of the primary outputs of the mitochondrial respiratory chain, and in certain contexts this function is more important than its role in ATP synthesis (Birsoy et al., 2015; Sullivan et al., 2015). For example, the ATP synthase inhibitor oligomycin blocks mitochondrial respiration and is toxic to cells; however, addition of an uncoupling agent that restores mitochondrial respiration (but not ATP production) can rescue both survival and proliferation in oligomycin-treated cells (Sullivan et al., 2015). Similarly, cells with genetic defects in mitochondrial respiration can survive and proliferate, but only if they are provided sufficient quantities of an alternative, practicable electron acceptor such as pyruvate (King and Attardi, 1989). Pyruvate reduction to lactate and O2 reduction to water both produce reduced products causing regeneration of the oxidized cofactor NAD+ from its reduced form NADH and facilitate electron disposal (Figure 2). While glycolysis can produce sufficient ATP to support cell proliferation, glycolysis is net redox-neutral. Therefore, glycolysis alone cannot support the production of oxidized molecules needed for cell division, such as nucleotides (Figure 1). Mitochondrial respiration is therefore critical in all cells, even those exhibiting high rates of aerobic glycolysis (Joshi et al., 2015; Martínez-Reyes et al., 2015; Weinberg et al., 2010; Zu and Guppy, 2004). Interestingly, many cells with intact mitochondrial respiration proliferate faster in tissue culture when supplemented with excess pyruvate, which increases NAD+ regeneration from NADH. The tight correlation between cellular NAD+/NADH ratio, respiratory rate, and proliferation rate highlights the importance of electron acceptors in creation of new biomass during cell division (Gui et al., 2016; Titov et al., 2016). The low NAD+/NADH ratio associated with oxidative biosynthesis is one explanation for why many proliferating cell types use aerobic glycolysis (Vander Heiden and DeBerardinis, 2017).
Glucose is the most highly consumed fuel of proliferating cells, including cancer cells and proliferating lymphocytes. Glucose is needed to produce ribose used in nucleotide biosynthesis, glycolytic intermediates that contribute to amino acid metabolism, and NADPH via the oxidative pentose phosphate pathway (Lunt and Vander Heiden, 2011; Sagone et al., 1974). NADPH provides the cellular reducing potential for biosynthetic reactions such as cholesterol and lipid biosynthesis (Fan et al., 2014; Lewis et al., 2014), and is needed to maintain reduced glutathione, which has diverse functions in cellular redox homeostasis. However, in proliferating cells glucose contributes only ∼10% to cellular biomass, with most glucose converted to lactate in near equimolar amounts (Hosios et al., 2016; Hume et al., 1978; Sagone et al., 1974). The fact that glucose is not a major carbon source for cell mass does not mean glycolysis is not important for proliferating cells. It has been argued that glycolytic ATP production provides metabolic flexibility to maintain energy state when OxPhos is limited by low pO2 in microenvironments such as the bone marrow or in a hypoxic tumor. It has also been argued that glycolysis permits faster ATP production than OxPhos (Koppenol et al., 2011), or that the decreased biosynthetic cost of producing glycolytic enzymes relative to OxPhos machinery is an advantageous energetic solution for proliferating cells (Shlomi et al., 2011). One certainty is that increased ATP production by glycolysis can decrease mitochondrial respiratory rate by increasing the cellular energy state. It should be noted that aerobic glycolysis is a common occurrence in normal physiology (Bulusu et al., 2017; Lindsay et al., 2014; Magistretti, 2016; O'Connor, 1952), and it is estimated that the basal rate of lactate production in humans is approximately 1 mmol/kg/hr (Connor and Woods, 1982). Lactate is produced in conditions where pyruvate production exceeds the rate of pyruvate oxidation and cytosolic NAD+/NADH is reduced. Pyruvate production can be increased by conditions that increase ATP consumption, such as exercise (James et al., 1999) or in response to high levels of catechols in sepsis (Garcia-Alvarez et al., 2016; Levy et al., 2005). In proliferating cells, growth factor-mediated increases in glucose uptake, high ATP consumption associated with cell growth, and low NAD+/NADH ratio secondary to oxidative biosynthesis can all contribute to increase lactate production and this may underlie the broad use of aerobic glycolysis in proliferating cells (Vander Heiden and DeBerardinis, 2017).
Spare Respiratory Capacity
Differences in spare respiratory capacity (SRC) represent a prominent difference in metabolic phenotype among some immune cell subsets. Spare respiratory capacity is defined as the increase in OCR elicited by exposure to agents such as FCCP that uncouple mitochondrial O2 consumption from ATP production (Huang et al., 2014; van der Windt et al., 2012; 2001)(Figure 2). The existence of SRC requires that the cellular energy state is sufficiently high that the demand for ATP synthesis is limiting the rate of electron transport (Figure 2). Thus, in cells with high SRC, OCR is limited by energy excess and not by substrate. The biological importance of SRC remains a source of debate. This phenotype may reflect a metabolic adaptation in some cells to increase their capacity for oxidative metabolism to produce ATP or for another process in which mitochondrial electron transport is required and may or may not be coupled to ATP synthesis (Sena et al., 2013).
Potential Biochemical Basis for Select Immune Cell Metabolic Phenotypes
We propose a model that incorporates principles of metabolic regulation to explain T cell phenotypes (Figure 3). Naïve T cells have few mitochondria as well as low ATP requirements to maintain homeostasis. Pyruvate production in a naïve T cell approximates the rate of pyruvate oxidation in the mitochondrial TCA cycle, and little lactate is produced. Following stimulation by foreign peptide-MHC and CD28 co-stimulation, T cells engage a proliferative program and their capacity for glucose uptake and pyruvate generation increases substantially, coincident with upregulation of genes encoding enzymes for glycolysis and the respiratory chain (Best et al., 2013; Jacobs et al., 2008). Demand for nucleotides and other oxidized biomass increases to support rapid cell proliferation, while the main process for disposing of electrons – cellular respiration – can be limited by low mitochondrial number and oxidation capacity, O2 availability, and/or a high energy state. This results in a lower cellular NAD+/NADH ratio, promoting pyruvate reduction to lactate. Thus, aerobic glycolysis reflects pyruvate production in excess of the cellular capacity for pyruvate oxidation, coupled with a low NAD+/NADH ratio. That is, lactate production in activated T cells is the result of the change in energetic, biosynthetic, and redox requirements of proliferation (Figure 1, Figure 2). This model incorporates the importance of cellular respiration in proliferation, and attributes high rates of lactate production to energetic excess, rather than energy deficiency. The model is consistent with observations that oligomycin-treated T cells can proliferate when excess glucose and pyruvate are available (Chang et al., 2013).
Figure 3. Model relating metabolic state of different T cell subsets to cell function.
Naïve T cells exhibit low glucose transporter expression and nutrient uptake, with metabolism directed toward nutrient oxidation and cell survival. Nutrient uptake and mitochondrial mass are increased upon T cell activation, facilitating the production of new biomass for proliferation. Memory T cells return to a low nutrient uptake state, again relying on nutrient oxidation to support cell survival; however, these cells retain more mitochondria (oxidation capacity) facilitating their ability to re-engage a proliferative program when reactivated. Exhausted T cells become limited for nutrient uptake and/or oxidizing capacity secondary to dysfunctional mitochondria limiting their ability to support a proliferative metabolic program.
In a resolving immune response, T cell numbers contract and memory T cells are generated. Memory T cells are quiescent, non-dividing cells with a metabolic phenotype that is similar in many ways to naïve cells (Figure 1, Figure 3). However, these cells can proliferate and respond faster than naïve T cells following antigen exposure. They have increased capacity for protein synthesis to support proliferation and effector functions, and increased mitochondria with increased spare respiratory capacity. Thus, their capacity for ATP consumption, ATP generation, and nutrient oxidation are high despite low nutrient uptake, perhaps facilitating an ability to rapidly engage a proliferative program with increased energetic and biosynthetic requirements. While the signaling events responsible for return to quiescence are poorly understood, it is clear that quiescence in central memory cells is maintained in part by low levels of glucose transporters; hence, the predominance of fatty acid oxidation in this T cell subset (Pan et al., 2017; van der Windt et al., 2013; van der Windt et al., 2012). In mammalian cells, fatty acids are unable to serve as an anaplerotic substrate for the TCA cycle to produce nucleotides and other biomass components, and the preference for this fuel may help limit proliferation until appropriate signals are provided to increase uptake of glucose and amino acids. Interestingly, forced maintenance of aerobic glycolysis during an immune response does not eliminate memory T cells, but instead creates an effector memory cell phenotype, highlighting the importance of fuel limitation in promoting memory cell quiescence (Phan et al., 2016). Regardless, baseline respiratory rate is higher in memory T cells than in naïve cells, reflecting reliance on fatty acid oxidation and OxPhos rather than glycolysis to make ATP. As occurs in naïve T cell activation, response of memory cells to full antigen receptor stimulation activates a proliferative metabolic program with increased glucose uptake, increased nucleotide production (with a fall in NAD+/NADH ratio), and higher lactate production. Thus, the increased oxidative capacity afforded by the high mitochondrial mass in memory T cells as compared to a proliferating naïve T cell may facilitate production of nucleotides and oxidized biomass that can be limiting for cell proliferation (Vander Heiden and DeBerardinis, 2017) and enable rapid clonal expansion when memory cells encounter antigen.
The capacity of a cell to oxidize fuels is intimately linked to its ability to perform effector functions and proliferate. As discussed above, quiescence in T cell subsets is associated with low levels of glucose transporters, while productive antigen-receptor stimulation is linked to transporter upregulation and increased glucose uptake. In this way, access to oxidizable nutrients is coordinated with metabolic demands to support proliferation. Similarly, imposed suppression of T cell oxidative capacity are coincident with active T cell suppression. In chronic immune responses such as occur in chronic infections or cancer, a phenomenon known as T cell exhaustion (Tex) occurs, associated with expression of the inhibitory co-receptor PD-1 and blunted effector functions (Bengsch et al., 2016; Wherry and Kurachi, 2015). In Tex cells, cellular capacity for nutrient oxidation is limited by both low levels of glucose transporters and altered mitochondria (Figure 3). Blockade of PD-1 – PDL1 binding can reverse this metabolic program and restore anti-tumor immunity (Bengsch et al., 2016). A similar phenomenon has been observed in tumor-infiltrating and tumor-associated lymphocytes (TILs) (Scharping et al., 2016). T cells isolated from patients and preclinical models with B cell leukemia are also exhausted, and have low rates of glucose uptake and glycolysis. Restoration of glucose uptake can partially restore T cell activity demonstrating that cancer-induced T cell metabolic impairments contribute to poor T cell function (Siska et al., 2016). Mitochondrial dysfunction also appears to play a key role in reduced TIL function as improved mitochondrial biogenesis and quality control could enhance anti-tumor immunity (Chamoto et al., 2017; Scharping et al., 2016) and T cells with low mitochondrial potential had greater ability to eliminate tumors in adoptive cell therapy (Sukumar et al., 2016). Indeed, regulation of mitochondrial quality control may contribute to T cell longevity and generation of immunological memory (Adams et al., 2016).
In the interpretation of T cell metabolic phenotypes provided, aerobic glycolysis as a characteristic of immune cell activation supports both ATP production and oxidative biosynthesis in these cells. A high rate of glycolytic ATP production may also provide other immune cells the flexibility to use O2 for purposes other than energetics. Macrophages display a variety of metabolic phenotypes (Van den Bossche et al., 2017). Oxygen can be a limited resource in immune cell microenvironments and mitochondrial respiration is likely the largest O2 consumer by many immune cells. Nevertheless, decreased reliance on OxPhos could enable increased activity of non-mitochondrial oxidases involved in biosynthesis or epigenetic regulation, both critically important for the function of numerous immune cell subsets. Similarly, production of ROS by some immune cells is critical for host defense and may also be facilitated by a more glycolytic phenotype. Production of ATP by glycolysis and inhibiting ATP synthase to facilitate mitochondrial hyperpolarization will preserve O2 availability as well as promote non-mitochondrial oxidase activity such as NOX (Mills et al., 2016). Lactate production will also be increased in conjunction with a more reduced cell state that favors ROS generation. Using glycolysis to maintain energy state has the added benefit of providing a pyruvate “buffer” to maximize biochemical flexibility. Provided glycolysis maintains energy state, uncoupling of mitochondrial respiration from ATP production can facilitate oxidation of phagocytosed material, (D. Park et al., 2011). In summary, immune cell activation generates a change in energetic and redox state. Maintenance of energy state by glycolysis and generation of a pyruvate “buffer” provides maximal biochemical flexibility for cells to adapt to this change in state.
This model attempts to explain immune cell metabolic phenotypes in relation to biochemical requirements of cell functions, and extends beyond ATP production as the only output of metabolism. All immune cell functions require ATP, whether it be production of new biomass and cytokines in a proliferating lymphocyte, antibody production in B cells, pathogen destruction, or oxidation of ingested biomass in phagocytes. The pathways used to generate the ATP may be a secondary consequence, and oxidative to glycolytic metabolic reprogramming might reflect biochemical requirements other than energetics. Many details of this model are untested; however, the model is consistent with data in other cell systems and generally consistent with principles of cellular energetics. The model highlights a few important points. First, the terminology used to describe metabolic phenotypes can be confusing (Box 1). Cells are described as “oxidative” when pyruvate production approximates pyruvate oxidation and “glycolytic” when lactate is produced. However, the rate of pyruvate oxidation and mitochondrial respiratory rate of a “glycolytic” proliferating T cells often exceeds the rate observed in a naïve T cell given the higher total rate of nutrient uptake (Guppy et al., 1993; Hume et al., 1978). Describing metabolic programs as either oxidative or glycolytic simplifies a complex relationship between cellular respiratory capacity, cellular redox requirements, cellular energetics, and fuel availability. Similarly, the presence or absence of spare respiratory capacity as a common phenotype in several immune cell subsets has been a source of confusion to many readers. Spare respiratory capacity is the magnitude of respiration increase following treatment of cells with FCCP or another drug that uncouples respiration from ATP production. Spare respiratory capacity, therefore, reflects a similarly complex relationship between total respiratory capacity and cellular energetics. Elucidating the underlying biochemical basis of many immune cell metabolic phenotypes can be challenging; however, as therapies targeting immunometabolism continue to show efficacy in altering immunity, improving this understanding might aid in the design of more effective therapeutics.
Influence of Available Nutrients on Immune Cell Metabolism
Accumulating data suggest that the microenvironment can have a profound effect on immune cell function (Chang and E. L. Pearce, 2016). Dissecting how tissue environment affects immunometabolism can be challenging, because the conditions of nutrient excess used to culture cells in vitro are quite different from the microenvironment experienced by cells in tissues (Mayers and Vander Heiden, 2015). A prominent example is the nutrient limitations immune cells experience within the tumor microenvironment, where poor vascular supply and high nutrient consumption by cancer cells combine to deplete available nutrients. It has been estimated that glucose is ∼10-fold lower in the tumor interstitium than it is in the plasma. T cells can adapt to low glucose by increasing glutamine metabolism (Blagih et al., 2015), but low glucose levels within a tumor alters tumor-infiltrating lymphocytes (TILs) metabolism and effector function and represents a metabolic mechanism by which cancer cells can escape immune surveillance (Chang et al., 2013; Chang et al., 2015; Ho et al., 2015). In a glucose-poor environment, the decreased amount of glucose available to TILs leads to lower levels of glycolytic intermediates including phosphoenolpyruvate (PEP), which alters T cell receptor (TCR)-mediated calcium flux and T cell activation. Interestingly, this microenvironmental limitation can be overcome by genetic manipulation of enzyme expression to artificially raise PEP levels, providing proof-of-principle that modulating immune cell metabolism can directly influence immune responses (Ho et al., 2015). Therapies that result in lower tumor cell glucose consumption might work in part by altering environmental conditions within the tumor to better support TIL function (Chang et al., 2015). Similarly, the accumulation of metabolic products in the tumor microenvironment has important implications, such as the influence of high lactate concentration in tumors on macrophage polarization (Carmona-Fontaine et al., 2017; Colegio et al., 2014). Finally, immune cell function can also be limited by amino acid availability. The observation that guinea pig serum could inhibit the growth of a murine lymphoma led to the purification of asparaginase as an anti-neoplastic enzymatic activity that was later found to also inhibit the growth of normal lymphocytes (Berenbaum, 1970; Broome, 1963; Kidd, 1953a; Kidd, 1953b). Environmental limitations of asparagine, tryptophan, serine (Ma et al., 2017), and arginine have all been demonstrated to impact immune cell function under various conditions in vivo. The extensive literature studying amino acid deficiency on immune cell function has been recently reviewed elsewhere (Murray, 2016).
Oxygen can be another limited resource for immune cells in in vivo. While circulating immune cells are exposed to the O2 concentration of blood (∼75-100 mm Hg or ∼100-140 μM), the pO2 of tissues is much lower, on average approaching the mixed venous pO2 of 35-50 mm Hg. In the bone marrow, pO2 measurements vary among microenvironmental niches, with a range between 10-22 mm Hg (Spencer et al., 2014). A similar range of pO2 has been reported in the spleen, whereas the pO2 in the thymus is lower, closer to 10 mm Hg (Braun et al., 2001). Cho et al. recently demonstrated that germinal centers have regional variations in pO2, with important consequences for B cell biology (Cho et al., 2016). In a tumor, pO2 varies based on radial and longitudinal distance from the arterial supply, with areas that are well-oxygenated and other areas that are anoxic (Koch and Evans, 2015). While pO2 is in part dependent on OCR of cells in those niches, hypoxia responses are observed in immune cells in vivo, providing physiologic evidence of O2 limitation (Cho et al., 2016). Hypoxia and inflammation are intimately connected (Eltzschig and Carmeliet, 2011). Tissue hypoxia leads to inflammation, and high O2 consumption by inflammatory cells can lead to hypoxia. HIF transcriptional activity been demonstrated to play an important role in macrophage subsets (Colegio et al., 2014; Cramer et al., 2003), in B cell class-switching (Cho et al., 2016), CD8+ T cell differentiation (Doedens et al., 2013; Tyrakis et al., 2016), and in determining the balance of Th17 and Treg cell differentiation (Dang et al., 2011; Shi et al., 2011). HIF has many transcriptional targets that are not directly involved in metabolism including epigenetic modifiers and transcription factors such as NF-κB and RORγt, which have established roles in immunity (Schödel et al., 2011). Whether the metabolic effects of HIF are critical to support the biochemical needs of cell differentiation or immune effector functions remains an open question. Given the importance of mitochondrial respiration for cell proliferation, and the importance of non-mitochondrial respiration in innate immunity, it is tempting to speculate that O2 might be a limiting nutrient at sites of immune cell activation and influence the character and magnitude of immune responses.
Conclusions
The biochemical requirements of cell biology and physiology have been an active area of research for more than a century. Technologic advances have increased our ability to describe metabolic phenotypes, but we still rely on the lens of core biochemical principles to understand the complexity underlying differences in cell metabolism. This review has considered immune cell metabolic phenotypes within the context of the energetic and redox requirements of immune cell function. Many of the models and assumptions proposed are untested, and observations based on in vitro studies will require validation in vivo. Changes in metabolite levels can also directly affect cell state (Tyrakis et al., 2016), and as new observations are made about the interdependence of immune cell metabolism and function, contextualizing those findings within core principles of biochemistry and physiology will be important to understand their role in host defense and exploit these relationships for patient benefit.
Acknowledgments
We thank William Kaelin (DFCI, HHMI), Sam McBrayer (DFCI), Abhishek Chakraborty (DFCI), Dan Schmidt (MIT), Lucas Sullivan (MIT) and David F. Wilson (University of Pennsylvania) for many helpful discussions and correspondences during the preparation of this manuscript. We also thank members of the Vander Heiden lab and for thoughtful comments and a special thanks to Brooke Bevis for generating the figures. B.A.O. acknowledges support from NIH K08 HL119355, the Gilead Sciences Research Scholars Program in Cardiovascular Disease, and the Heart and Vascular Center Junior Faculty Award, Brigham and Women's Hospital, Boston MA. J.C.R. is supported by R01DK105550. M.G.V.H. acknowledges grant support from the R01CA168653, R01CA201276, SU2C, the Ludwig Center at MIT, the Lustgarten Foundation, and an HHMI Faculty Scholar award.
Footnotes
Disclosures: B.A.O. received consulting fees from Fulcrum Therapeutics, Peloton Therapeutics and Bristol-Myers Squibb. J.C.R. receives funding from Incyte Corp. M.G.V.H. is a consultant and scientific advisory board member for Agios Pharmaceuticals and Aeglea Biotherapeutics.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adams WC, Chen YH, Kratchmarov R, Yen B, Nish SA, Lin WHW, Rothman NJ, Luchsinger LL, Klein U, Busslinger M, Rathmell JC, Snoeck HW, Reiner SL. Anabolism-Associated Mitochondrial Stasis Driving Lymphocyte Differentiation over Self-Renewal. Cell Rep. 2016;17:3142–3152. doi: 10.1016/j.celrep.2016.11.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelin A, Gil-de-Gómez L, Dahiya S, Jiao J, Guo L, Levine MH, Wang Z, Quinn WJ, III, Kopinski PK, Wang L, Akimova T, Liu Y, Bhatti TR, Han R, Laskin BL, Baur JA, Blair IA, Wallace DC, Hancock WW, Beier UH. Foxp3 Reprograms T Cell Metabolism to Function in Low-Glucose, High-Lactate Environments. Cell Metab. 2017 doi: 10.1016/j.cmet.2016.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bambrick LL, Kostov Y, Rao G. In vitro cell culture pO2 is significantly different from incubator pO2. Biotechnol Prog. 2011;27:1185–1189. doi: 10.1002/btpr.622. [DOI] [PubMed] [Google Scholar]
- Banh RS, Iorio C, Marcotte R, Xu Y, Cojocari D, Rahman AA, Pawling J, Zhang W, Sinha A, Rose CM, Isasa M, Zhang S, Wu R, Virtanen C, Hitomi T, Habu T, Sidhu SS, Koizumi A, Wilkins SE, Kislinger T, Gygi SP, Schofield CJ, Dennis JW, Wouters BG, Neel BG. PTP1B controls non-mitochondrial oxygen consumption by regulating RNF213 to promote tumour survival during hypoxia. Nat Cell Biol. 2016;18:803–813. doi: 10.1038/ncb3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beier UH, Angelin A, Akimova T, Wang L, Liu Y, Xiao H, Koike MA, Hancock SA, Bhatti TR, Han R, Jiao J, Veasey SC, Sims CA, Baur JA, Wallace DC, Hancock WW. Essential role of mitochondrial energy metabolism in Foxp3+ T-regulatory cell function and allograft survival. FASEB J. 2015;29:2315–2326. doi: 10.1096/fj.14-268409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bengsch B, Johnson AL, Kurachi M, Odorizzi PM, Pauken KE, Attanasio J, Stelekati E, McLane LM, Paley MA, Delgoffe GM, Wherry EJ. Bioenergetic Insufficiencies Due to Metabolic Alterations Regulated by the Inhibitory Receptor PD-1 Are an Early Driver of CD8(+) T Cell Exhaustion. Immunity. 2016;45:358–373. doi: 10.1016/j.immuni.2016.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berenbaum MC. Immunosuppression by L-asparaginase. Nature. 1970;225:550–552. doi: 10.1038/225550a0. [DOI] [PubMed] [Google Scholar]
- Best JA, Blair DA, Knell J, Yang E, Mayya V, Doedens A, Dustin ML, Goldrath AW Immunological Genome Project Consortium. Transcriptional insights into the CD8(+) T cell response to infection and memory T cell formation. Nat Immunol. 2013;14:404–412. doi: 10.1038/ni.2536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birsoy K, Wang T, Chen WW, Freinkman E, Abu-Remaileh M, Sabatini DM. An Essential Role of the Mitochondrial Electron Transport Chain in Cell Proliferation Is to Enable Aspartate Synthesis. Cell. 2015;162:540–551. doi: 10.1016/j.cell.2015.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blagih J, Coulombe F, Vincent EE, Dupuy F, Galicia-Vázquez G, Yurchenko E, Raissi TC, van der Windt GJW, Viollet B, Pearce EL, Pelletier J, Piccirillo CA, Krawczyk CM, Divangahi M, Jones RG. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity. 2015;42:41–54. doi: 10.1016/j.immuni.2014.12.030. [DOI] [PubMed] [Google Scholar]
- Braun RD, Lanzen JL, Snyder SA, Dewhirst MW. Comparison of tumor and normal tissue oxygen tension measurements using OxyLite or microelectrodes in rodents. Am J Physiol Heart Circ Physiol. 2001;280:H2533–44. doi: 10.1152/ajpheart.2001.280.6.H2533. [DOI] [PubMed] [Google Scholar]
- Broome JD. Evidence that the L-asparaginase of guinea pig serum is responsible for its antilymphoma effects. I Properties of the L-asparaginase of guinea pig serum in relation to those of the antilymphoma substance. J Exp Med. 1963;118:99–120. doi: 10.1084/jem.118.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown GC, Borutaite V. Nitric oxide inhibition of mitochondrial respiration and its role in cell death. Free Radical Biology and Medicine. 2002;33:1440–1450. doi: 10.1016/s0891-5849(02)01112-7. [DOI] [PubMed] [Google Scholar]
- Bulusu V, Prior N, Snaebjornsson MT, Kuehne A, Sonnen KF, Kress J, Stein F, Schultz C, Sauer U, Aulehla A. Spatiotemporal Analysis of a Glycolytic Activity Gradient Linked to Mouse Embryo Mesoderm Development. Dev Cell. 2017;40:331–341.e4. doi: 10.1016/j.devcel.2017.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmona-Fontaine C, Deforet M, Akkari L, Thompson CB, Joyce JA, Xavier JB. Metabolic origins of spatial organization in the tumor microenvironment. Proceedings of the National Academy of Sciences. 2017;114:2934–2939. doi: 10.1073/pnas.1700600114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cham CM, Gajewski TF. Glucose availability regulates IFN-gamma production and p70S6 kinase activation in CD8+ effector T cells. The Journal of Immunology. 2005;174:4670–4677. doi: 10.4049/jimmunol.174.8.4670. [DOI] [PubMed] [Google Scholar]
- Chamoto K, Chowdhury PS, Kumar A, Sonomura K, Matsuda F, Fagarasan S, Honjo T. Mitochondrial activation chemicals synergize with surface receptor PD-1 blockade for T cell-dependent antitumor activity. Proceedings of the National Academy of Sciences. 2017;114:E761–E770. doi: 10.1073/pnas.1620433114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang CH, Curtis JD, Maggi LB, Faubert B, Villarino AV, O'Sullivan D, Huang SCC, van der Windt GJW, Blagih J, Qiu J, Weber JD, Pearce EJ, Jones RG, Pearce EL. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell. 2013;153:1239–1251. doi: 10.1016/j.cell.2013.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang CH, Pearce EL. Emerging concepts of T cell metabolism as a target of immunotherapy. Nat Immunol. 2016;17:364–368. doi: 10.1038/ni.3415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang CH, Qiu J, O'Sullivan D, Buck MD, Noguchi T, Curtis JD, Chen Q, Gindin M, Gubin MM, van der Windt GJW, Tonc E, Schreiber RD, Pearce EJ, Pearce EL. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell. 2015;162:1229–1241. doi: 10.1016/j.cell.2015.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho SH, Raybuck AL, Stengel K, Wei M, Beck TC, Volanakis E, Thomas JW, Hiebert S, Haase VH, Boothby MR. Germinal centre hypoxia and regulation of antibody qualities by a hypoxia response system. Nature. 2016;537:234–238. doi: 10.1038/nature19334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colegio OR, Chu NQ, Szabo AL, Chu T, Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC, Phillips GM, Cline GW, Phillips AJ, Medzhitov R. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513:559–563. doi: 10.1038/nature13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connor H, Woods HF. Quantitative aspects of L(+)-lactate metabolism in human beings. Ciba Found Symp. 1982;87:214–234. doi: 10.1002/9780470720691.ch12. [DOI] [PubMed] [Google Scholar]
- Cramer T, Yamanishi Y, Clausen BE, Förster I, Pawlinski R, Mackman N, Haase VH, Jaenisch R, Corr M, Nizet V, Firestein GS, Gerber HP, Ferrara N, Johnson RS. HIF-1alpha is essential for myeloid cell-mediated inflammation. Cell. 2003;112:645–657. doi: 10.1016/s0092-8674(03)00154-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang EV, Barbi J, Yang HY, Jinasena D, Yu H, Zheng Y, Bordman Z, Fu J, Kim Y, Yen HR, Luo W, Zeller K, Shimoda L, Topalian SL, Semenza GL, Dang CV, Pardoll DM, Pan F. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell. 2011;146:772–784. doi: 10.1016/j.cell.2011.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deussen A, Ohanyan V, Jannasch A, Yin L, Chilian W. Mechanisms of metabolic coronary flow regulation. Journal of Molecular and Cellular Cardiology. 2011 doi: 10.1016/j.yjmcc.2011.10.001. [DOI] [PubMed] [Google Scholar]
- Doedens AL, Phan AT, Stradner MH, Fujimoto JK, Nguyen JV, Yang E, Johnson RS, Goldrath AW. Hypoxia-inducible factors enhance the effector responses of CD8(+) T cells to persistent antigen. Nat Immunol. 2013;14:1173–1182. doi: 10.1038/ni.2714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncker DJ, Bache RJ. Regulation of Coronary Blood Flow During Exercise. Physiological Reviews. 2008;88:1009–1086. doi: 10.1152/physrev.00045.2006. [DOI] [PubMed] [Google Scholar]
- Düvel K, Yecies JL, Menon S, Raman P, Lipovsky AI, Souza AL, Triantafellow E, Ma Q, Gorski R, Cleaver S, Vander Heiden MG, MacKeigan JP, Finan PM, Clish CB, Murphy LO, Manning BD. Activation of a Metabolic Gene Regulatory Network Downstream of mTOR Complex 1. Molecular Cell. 2010;39:171–183. doi: 10.1016/j.molcel.2010.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Benna J, Hurtado-Nedelec M, Marzaioli V, Marie JC, Gougerot-Pocidalo MA, Dang PMC. Priming of the neutrophil respiratory burst: role in host defense and inflammation. Immunol Rev. 2016;273:180–193. doi: 10.1111/imr.12447. [DOI] [PubMed] [Google Scholar]
- Eltzschig HK, Carmeliet P. Hypoxia and inflammation. N Engl J Med. 2011;364:656–665. doi: 10.1056/NEJMra0910283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Everts B, Amiel E, Huang SCC, Smith AM, Chang CH, Lam WY, Redmann V, Freitas TC, Blagih J, van der Windt GJW, Artyomov MN, Jones RG, Pearce EL, Pearce EJ. TLR-driven early glycolytic reprogramming via the kinases TBK1-IKKɛ supports the anabolic demands of dendritic cell activation. Nat Immunol. 2014;15:323–332. doi: 10.1038/ni.2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Everts B, Amiel E, van der Windt GJW, Freitas TC, Chott R, Yarasheski KE, Pearce EL, Pearce EJ. Commitment to glycolysis sustains survival of NO-producing inflammatory dendritic cells. Blood. 2012;120:1422–1431. doi: 10.1182/blood-2012-03-419747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan J, Ye J, Kamphorst JJ, Shlomi T, Thompson CB, Rabinowitz JD. Quantitative flux analysis reveals folate-dependent NADPH production. Nature. 2014;510:298–302. doi: 10.1038/nature13236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint TR, Janowitz T, Connell CM, Roberts EW, Denton AE, Coll AP, Jodrell DI, Fearon DT. Tumor-Induced IL-6 Reprograms Host Metabolism to Suppress Anti-tumor Immunity. Cell Metab. 2016;24:672–684. doi: 10.1016/j.cmet.2016.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frauwirth KA, Riley JL, Harris MH, Parry RV, Rathmell JC, Plas DR, Elstrom RL, June CH, Thompson CB. The CD28 signaling pathway regulates glucose metabolism. Immunity. 2002;16:769–777. doi: 10.1016/s1074-7613(02)00323-0. [DOI] [PubMed] [Google Scholar]
- Freemerman AJ, Johnson AR, Sacks GN, Milner JJ, Kirk EL, Troester MA, Macintyre AN, Goraksha-Hicks P, Rathmell JC, Makowski L. Metabolic reprogramming of macrophages: glucose transporter 1 (GLUT1)-mediated glucose metabolism drives a proinflammatory phenotype. Journal of Biological Chemistry. 2014;289:7884–7896. doi: 10.1074/jbc.M113.522037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda R, Zhang H, Kim JW, Shimoda L, Dang CV, Semenza GL. HIF-1 Regulates Cytochrome Oxidase Subunits to Optimize Efficiency of Respiration in Hypoxic Cells. Cell. 2007;129:111–122. doi: 10.1016/j.cell.2007.01.047. [DOI] [PubMed] [Google Scholar]
- Garcia-Alvarez M, Marik P, Bellomo R. Sepsis-associated hyperlactatemia. Crit Care. 2016;18:1–11. doi: 10.1186/s13054-014-0503-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerriets VA, Kishton RJ, Johnson MO, Cohen S, Siska PJ, Nichols AG, Warmoes MO, de Cubas AA, MacIver NJ, Locasale JW, Turka LA, Wells AD, Rathmell JC. Foxp3 and Toll-like receptor signaling balance Treg cell anabolic metabolism for suppression. Nat Immunol. 2016;17:1459–1466. doi: 10.1038/ni.3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerriets VA, Kishton RJ, Nichols AG, Macintyre AN, Inoue M, Ilkayeva O, Winter PS, Liu X, Priyadharshini B, Slawinska ME, Haeberli L, Huck C, Turka LA, Wood KC, Hale LP, Smith PA, Schneider MA, MacIver NJ, Locasale JW, Newgard CB, Shinohara ML, Rathmell JC. Metabolic programming and PDHK1 control CD4+ T cell subsets and inflammation. J Clin Invest. 2015;125:194–207. doi: 10.1172/JCI76012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldfine H. The evolution of oxygen as a biosynthetic reagent. J Gen Physiol. 1965;49:253–74. doi: 10.1085/jgp.49.1.253. Suppl. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubser PM, Bantug GR, Razik L, Fischer M, Dimeloe S, Hoenger G, Durovic B, Jauch A, Hess C. Rapid effector function of memory CD8+ T cells requires an immediate-early glycolytic switch. Nat Immunol. 2013;14:1064–1072. doi: 10.1038/ni.2687. [DOI] [PubMed] [Google Scholar]
- Gui DY, Sullivan LB, Luengo A, Hosios AM, Bush LN, Gitego N, Davidson SM, Freinkman E, Thomas CJ, Vander Heiden MG. Environment Dictates Dependence on Mitochondrial Complex I for NAD+ and Aspartate Production and Determines Cancer Cell Sensitivity to Metformin. Cell Metab. 2016;24:716–727. doi: 10.1016/j.cmet.2016.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guppy M, Greiner E, Brand K. The role of the Crabtree effect and an endogenous fuel in the energy metabolism of resting and proliferating thymocytes. Eur J Biochem. 1993;212:95–99. doi: 10.1111/j.1432-1033.1993.tb17637.x. [DOI] [PubMed] [Google Scholar]
- Hirsilä M, Koivunen P, Günzler V, Kivirikko KI, Myllyharju J. Characterization of the human prolyl 4-hydroxylases that modify the hypoxia-inducible factor. J Biol Chem. 2003;278:30772–30780. doi: 10.1074/jbc.M304982200. [DOI] [PubMed] [Google Scholar]
- Ho PC, Bihuniak JD, Macintyre AN, Staron M, Liu X, Amezquita R, Tsui YC, Cui G, Micevic G, Perales JC, Kleinstein SH, Abel ED, Insogna KL, Feske S, Locasale JW, Bosenberg MW, Rathmell JC, Kaech SM. Phosphoenolpyruvate Is a Metabolic Checkpoint of Anti-tumor T Cell Responses. Cell. 2015;162:1217–1228. doi: 10.1016/j.cell.2015.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosios AM, Hecht VC, Danai LV, Johnson MO, Rathmell JC, Steinhauser ML, Manalis SR, Vander Heiden MG. Amino Acids Rather than Glucose Account for the Majority of Cell Mass in Proliferating Mammalian Cells. Dev Cell. 2016;36:540–549. doi: 10.1016/j.devcel.2016.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang SCC, Everts B, Ivanova Y, O'Sullivan D, Nascimento M, Smith AM, Beatty W, Love-Gregory L, Lam WY, O'Neill CM, Yan C, Du H, Abumrad NA, Urban JF, Jr, Artyomov MN, Pearce EL, Pearce EJ. Cell-intrinsic lysosomal lipolysis is essential for alternative activation of macrophages. Nat Immunol. 2014;15:846–855. doi: 10.1038/ni.2956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hume DA, Radik JL, Ferber E, Weidemann MJ. Aerobic glycolysis and lymphocyte transformation. Biochem J. 1978;174:703–709. doi: 10.1042/bj1740703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huynh A, DuPage M, Priyadharshini B, Sage PT, Quiros J, Borges CM, Townamchai N, Gerriets VA, Rathmell JC, Sharpe AH, Bluestone JA, Turka LA. Control of PI(3) kinase in Treg cells maintains homeostasis and lineage stability. Nat Immunol. 2015;16:188–196. doi: 10.1038/ni.3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs SR, Herman CE, MacIver NJ, Wofford JA, Wieman HL, Hammen JJ, Rathmell JC. Glucose uptake is limiting in T cell activation and requires CD28-mediated Akt-dependent and independent pathways. The Journal of Immunology. 2008;180:4476–4486. doi: 10.4049/jimmunol.180.7.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs SR, Michalek RD, Rathmell JC. IL-7 is essential for homeostatic control of T cell metabolism in vivo. J Immunol. 2010;184:3461–3469. doi: 10.4049/jimmunol.0902593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- James JH, Wagner KR, King JK, Leffler RE, Upputuri RK, Balasubramaniam A, Friend LA, Shelly DA, Paul RJ, Fischer JE. Stimulation of both aerobic glycolysis and Na(+)-K(+)-ATPase activity in skeletal muscle by epinephrine or amylin. Am J Physiol. 1999;277:E176–86. doi: 10.1152/ajpendo.1999.277.1.E176. [DOI] [PubMed] [Google Scholar]
- Johnson MO, Siska PJ, Contreras DC, Rathmell JC. Nutrients and the microenvironment to feed a T cell army. Semin Immunol. 2016;28:505–513. doi: 10.1016/j.smim.2016.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi S, Tolkunov D, Aviv H, Hakimi AA, Yao M, Hsieh JJ, Ganesan S, Chan CS, White E. The Genomic Landscape of Renal Oncocytoma Identifies a Metabolic Barrier to Tumorigenesis. Cell Rep. 2015;13:1895–1908. doi: 10.1016/j.celrep.2015.10.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaelin WG, Ratcliffe PJ. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Molecular Cell. 2008;30:393–402. doi: 10.1016/j.molcel.2008.04.009. [DOI] [PubMed] [Google Scholar]
- Kaelin WG, Ratcliffe PJ, Semenza GL. Pathways for Oxygen Regulation and Homeostasis: The 2016 Albert Lasker Basic Medical Research Award. JAMA. 2016;316:1252–1253. doi: 10.1001/jama.2016.12386. [DOI] [PubMed] [Google Scholar]
- Kidd JG. Regression of transplanted lymphomas induced in vivo by means of normal guinea pig serum. I. Course of transplanted cancers of various kinds in mice and rats given guinea pig serum, horse serum, or rabbit serum. J Exp Med. 1953a;98:565–582. doi: 10.1084/jem.98.6.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kidd JG. Regression of transplanted lymphomas induced in vivo by means of normal guinea pig serum. II. Studies on the nature of the active serum constituent: histological mechanism of the regression: tests for effects of guinea pig serum on lymphoma cells in vitro: discussion. J Exp Med. 1953b;98:583–606. doi: 10.1084/jem.98.6.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3:177–185. doi: 10.1016/j.cmet.2006.02.002. [DOI] [PubMed] [Google Scholar]
- King MP, Attardi G. Human cells lacking mtDNA: repopulation with exogenous mitochondria by complementation. Science. 1989;246:500–503. doi: 10.1126/science.2814477. [DOI] [PubMed] [Google Scholar]
- Koch CJ, Evans SM. Optimizing hypoxia detection and treatment strategies. Semin Nucl Med. 2015;45:163–176. doi: 10.1053/j.semnuclmed.2014.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koppenol WH, Bounds PL, Dang CV. Otto Warburg's contributions to current concepts of cancer metabolism. Nature reviews Cancer. 2011 doi: 10.1038/nrc3038. [DOI] [PubMed] [Google Scholar]
- Levy B, Gibot S, Franck P, Cravoisy A, Bollaert PE. Relation between muscle Na+K+ ATPase activity and raised lactate concentrations in septic shock: a prospective study. Lancet. 2005;365:871–875. doi: 10.1016/S0140-6736(05)71045-X. [DOI] [PubMed] [Google Scholar]
- Lewis CA, Parker SJ, Fiske BP, McCloskey D, Gui DY, Green CR, Vokes NI, Feist AM, Vander Heiden MG, Metallo CM. Tracing Compartmentalized NADPH Metabolism in the Cytosol and Mitochondria of Mammalian Cells. Molecular Cell. 2014;55:253–263. doi: 10.1016/j.molcel.2014.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindsay KJ, Du J, Sloat SR, Contreras L, Linton JD, Turner SJ, Sadilek M, Satrústegui J, Hurley JB. Pyruvate kinase and aspartate-glutamate carrier distributions reveal key metabolic links between neurons and glia in retina. Proceedings of the National Academy of Sciences. 2014;111:15579–15584. doi: 10.1073/pnas.1412441111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loenarz C, Schofield CJ. Physiological and biochemical aspects of hydroxylations and demethylations catalyzed by human 2-oxoglutarate oxygenases. Trends in Biochemical Sciences. 2011;36:7–18. doi: 10.1016/j.tibs.2010.07.002. [DOI] [PubMed] [Google Scholar]
- Lunt SY, Vander Heiden MG. Aerobic Glycolysis: Meeting the Metabolic Requirements of Cell Proliferation. Annu Rev Cell Dev Biol. 2011;27:441–464. doi: 10.1146/annurev-cellbio-092910-154237. [DOI] [PubMed] [Google Scholar]
- Ma EH, Bantug G, Griss T, Condotta S, Johnson RM, Samborska B, Mainolfi N, Suri V, Guak H, Balmer ML, Verway MJ, Raissi TC, Tsui H, Boukhaled G, Henriques da Costa S, Frezza C, Krawczyk CM, Friedman A, Manfredi M, Richer MJ, Hess C, Jones RG. Serine Is an Essential Metabolite for Effector T Cell Expansion. Cell Metab. 2017;25:345–357. doi: 10.1016/j.cmet.2016.12.011. [DOI] [PubMed] [Google Scholar]
- Macintyre AN, Gerriets VA, Nichols AG, Michalek RD, Rudolph MC, Deoliveira D, Anderson SM, Abel ED, Chen BJ, Hale LP, Rathmell JC. The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab. 2014;20:61–72. doi: 10.1016/j.cmet.2014.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magistretti PJ. Imaging brain aerobic glycolysis as a marker of synaptic plasticity. Proceedings of the National Academy of Sciences. 2016;113:7015–7016. doi: 10.1073/pnas.1607423113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Reyes I, Diebold LP, Kong H, Schieber M, Huang H, Hensley CT, Mehta MM, Wang T, Santos JH, Woychik R, Dufour E, Spelbrink JN, Weinberg SE, Zhao Y, DeBerardinis RJ, Chandel NS. TCA Cycle and Mitochondrial Membrane Potential Are Necessary for Diverse Biological Functions. Molecular Cell. 2015 doi: 10.1016/j.molcel.2015.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayers JR, Vander Heiden MG. Famine versus feast: understanding the metabolism of tumors in vivo. Trends in Biochemical Sciences. 2015;40:130–140. doi: 10.1016/j.tibs.2015.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metzen E, Wolff M, Fandrey J, Jelkmann W. Pericellular PO2 and O2 consumption in monolayer cell cultures. Respir Physiol. 1995;100:101–106. doi: 10.1016/0034-5687(94)00125-j. [DOI] [PubMed] [Google Scholar]
- Michalek RD, Gerriets VA, Jacobs SR, Macintyre AN, MacIver NJ, Mason EF, Sullivan SA, Nichols AG, Rathmell JC. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mik EG, Ince C, Eerbeek O, Heinen A, Stap J, Hooibrink B, Schumacher CA, Balestra GM, Johannes T, Beek JF, Nieuwenhuis AF, van Horssen P, Spaan JA, Zuurbier CJ. Mitochondrial oxygen tension within the heart. Journal of Molecular and Cellular Cardiology. 2009;46:943–951. doi: 10.1016/j.yjmcc.2009.02.002. [DOI] [PubMed] [Google Scholar]
- Mills EL, Kelly B, Logan A, Costa ASH, Varma M, Bryant CE, Tourlomousis P, Däbritz JHM, Gottlieb E, Latorre I, Corr SC, McManus G, Ryan D, Jacobs HT, Szibor M, Xavier RJ, Braun T, Frezza C, Murphy MP, O'Neill LA. Succinate Dehydrogenase Supports Metabolic Repurposing of Mitochondria to Drive Inflammatory Macrophages. Cell. 2016;167:457–470.e13. doi: 10.1016/j.cell.2016.08.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray PJ. Amino acid auxotrophy as a system of immunological control nodes. Nat Immunol. 2016;17:132–139. doi: 10.1038/ni.3323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakaya M, Xiao Y, Zhou X, Chang JH, Chang M, Cheng X, Blonska M, Lin X, Sun SC. Inflammatory T cell responses rely on amino acid transporter ASCT2 facilitation of glutamine uptake and mTORC1 kinase activation. Immunity. 2014;40:692–705. doi: 10.1016/j.immuni.2014.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connor RJ. Growth and aerobic glycolysis in the retina of the chicken embryo. Nature. 1952;169:246. doi: 10.1038/169246a0. [DOI] [PubMed] [Google Scholar]
- O'Neill LAJ, Pearce EJ. Immunometabolism governs dendritic cell and macrophage function. J Exp Med. 2016;213:15–23. doi: 10.1084/jem.20151570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oestreich KJ, Read KA, Gilbertson SE, Hough KP, McDonald PW, Krishnamoorthy V, Weinmann AS. Bcl-6 directly represses the gene program of the glycolysis pathway. Nat Immunol. 2014;15:957–964. doi: 10.1038/ni.2985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y, Tian T, Park CO, Lofftus SY, Mei S, Liu X, Luo C, O'Malley JT, Gehad A, Teague JE, Divito SJ, Fuhlbrigge R, Puigserver P, Krueger JG, Hotamisligil GS, Clark RA, Kupper TS. Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature. 2017;543:252–256. doi: 10.1038/nature21379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papandreou I, Cairns RA, Fontana L, Lim AL, Denko NC. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006;3:187–197. doi: 10.1016/j.cmet.2006.01.012. [DOI] [PubMed] [Google Scholar]
- Park D, Han CZ, Elliott MR, Kinchen JM, Trampont PC, Das S, Collins S, Lysiak JJ, Hoehn KL, Ravichandran KS. Continued clearance of apoptotic cells critically depends on the phagocyte Ucp2 protein. Nature. 2011;477:220–224. doi: 10.1038/nature10340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park Y, Jin HS, Lopez J, Elly C, Kim G, Murai M, Kronenberg M, Liu YC. TSC1 regulates the balance between effector and regulatory T cells. J Clin Invest. 2013;123:5165–5178. doi: 10.1172/JCI69751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng M, Yin N, Chhangawala S, Xu K, Leslie CS, Li MO. Aerobic glycolysis promotes T helper 1 cell differentiation through an epigenetic mechanism. Science. 2016;354:481–484. doi: 10.1126/science.aaf6284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phan AT, Doedens AL, Palazon A, Tyrakis PA, Cheung KP, Johnson RS, Goldrath AW. Constitutive Glycolytic Metabolism Supports CD8(+) T Cell Effector Memory Differentiation during Viral Infection. Immunity. 2016;45:1024–1037. doi: 10.1016/j.immuni.2016.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poole DC, Copp SW, Ferguson SK, Musch TI. Skeletal muscle capillary function: contemporary observations and novel hypotheses. Exp Physiol. 2013;98:1645–1658. doi: 10.1113/expphysiol.2013.073874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raymond J, Segrè D. The effect of oxygen on biochemical networks and the evolution of complex life. Science. 2006;311:1764–1767. doi: 10.1126/science.1118439. [DOI] [PubMed] [Google Scholar]
- Roos D, Loos JA. Changes in the carbohydrate metabolism of mitogenically stimulated human peripheral lymphocytes. II. Relative importance of glycolysis and oxidative phosphorylation on phytohaemagglutinin stimulation. Exp Cell Res. 1973;77:127–135. doi: 10.1016/0014-4827(73)90561-2. [DOI] [PubMed] [Google Scholar]
- Sagone AL, LoBuglio AF, Balcerzak SP. Alterations in hexose monophosphate shunt during lymphoblastic transformation. Cell Immunol. 1974;14:443–452. doi: 10.1016/0008-8749(74)90195-6. [DOI] [PubMed] [Google Scholar]
- Scharping NE, Menk AV, Moreci RS, Whetstone RD, Dadey RE, Watkins SC, Ferris RL, Delgoffe GM. The Tumor Microenvironment Represses T Cell Mitochondrial Biogenesis to Drive Intratumoral T Cell Metabolic Insufficiency and Dysfunction. Immunity. 2016;45:374–388. doi: 10.1016/j.immuni.2016.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schödel J, Oikonomopoulos S, Ragoussis J, Pugh CW, Ratcliffe PJ, Mole DR. High-resolution genome-wide mapping of HIF-binding sites by ChIP-seq. Blood. 2011;117:e207–17. doi: 10.1182/blood-2010-10-314427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sena LA, Li S, Jairaman A, Prakriya M, Ezponda T, Hildeman DA, Wang CR, Schumacker PT, Licht JD, Perlman H, Bryce PJ, Chandel NS. Mitochondria are required for antigen-specific T cell activation through reactive oxygen species signaling. Immunity. 2013;38:225–236. doi: 10.1016/j.immuni.2012.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi LZ, Wang R, Huang G, Vogel P, Neale G, Green DR, Chi H. HIF1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J Exp Med. 2011;208:1367–1376. doi: 10.1084/jem.20110278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shlomi T, Benyamini T, Gottlieb E, Sharan R, Ruppin E. Genome-scale metabolic modeling elucidates the role of proliferative adaptation in causing the Warburg effect. PLoS Comput Biol. 2011;7:e1002018. doi: 10.1371/journal.pcbi.1002018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shrestha S, Yang K, Guy C, Vogel P, Neale G, Chi H. Treg cells require the phosphatase PTEN to restrain TH1 and TFH cell responses. Nat Immunol. 2015;16:178–187. doi: 10.1038/ni.3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinclair LV, Rolf J, Emslie E, Shi YB, Taylor PM, Cantrell DA. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat Immunol. 2013;14:500–508. doi: 10.1038/ni.2556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siska PJ, van der Windt GJW, Kishton RJ, Cohen S, Eisner W, MacIver NJ, Kater AP, Weinberg JB, Rathmell JC. Suppression of Glut1 and Glucose Metabolism by Decreased Akt/mTORC1 Signaling Drives T Cell Impairment in B Cell Leukemia. J Immunol. 2016;197:2532–2540. doi: 10.4049/jimmunol.1502464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer JA, Ferraro F, Roussakis E, Klein A, Wu J, Runnels JM, Zaher W, Mortensen LJ, Alt C, Turcotte R, Yusuf R, Côté D, Vinogradov SA, Scadden DT, Lin CP. Direct measurement of local oxygen concentration in the bone marrow of live animals. Nature. 2014;508:269–273. doi: 10.1038/nature13034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sukumar M, Liu J, Mehta GU, Patel SJ, Roychoudhuri R, Crompton JG, Klebanoff CA, Ji Y, Li P, Yu Z, Whitehill GD, Clever D, Eil RL, Palmer DC, Mitra S, Rao M, Keyvanfar K, Schrump DS, Wang E, Marincola FM, Gattinoni L, Leonard WJ, Muranski P, Finkel T, Restifo NP. Mitochondrial Membrane Potential Identifies Cells with Enhanced Stemness for Cellular Therapy. Cell Metab. 2016;23:63–76. doi: 10.1016/j.cmet.2015.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan LB, Gui DY, Hosios AM, Bush LN, Freinkman E, Vander Heiden MG. Supporting Aspartate Biosynthesis Is an Essential Function of Respiration in Proliferating Cells. Cell. 2015;162:552–563. doi: 10.1016/j.cell.2015.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tannahill GM, Curtis AM, Adamik J, Palsson-McDermott EM, McGettrick AF, Goel G, Frezza C, Bernard NJ, Kelly B, Foley NH, Zheng L, Gardet A, Tong Z, Jany SS, Corr SC, Haneklaus M, Caffrey BE, Pierce K, Walmsley S, Beasley FC, Cummins E, Nizet V, Whyte M, Taylor CT, Lin H, Masters SL, Gottlieb E, Kelly VP, Clish C, Auron PE, Xavier RJ, O'Neill LAJ. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature. 2013;496:238–242. doi: 10.1038/nature11986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Titov DV, Cracan V, Goodman RP, Peng J, Grabarek Z, Mootha VK. Complementation of mitochondrial electron transport chain by manipulation of the NAD+/NADH ratio. Science. 2016;352:231–235. doi: 10.1126/science.aad4017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyrakis PA, Palazon A, Macias D, Lee KL, Phan AT, Veliça P, You J, Chia GS, Sim J, Doedens A, Abelanet A, Evans CE, Griffiths JR, Poellinger L, Goldrath AW, Johnson RS. S-2-hydroxyglutarate regulates CD8(+) T-lymphocyte fate. Nature. 2016;540:236–241. doi: 10.1038/nature20165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Bossche J, O'Neill LA, Menon D. Macrophage Immunometabolism: Where Are We (Going)? Trends Immunol. 2017 doi: 10.1016/j.it.2017.03.001. [DOI] [PubMed] [Google Scholar]
- van der Windt GJW, Chang CH, Pearce EL. Measuring Bioenergetics in T Cells Using a Seahorse Extracellular Flux Analyzer. John Wiley & Sons, Inc.; Hoboken, NJ, USA: 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Windt GJW, Everts B, Chang CH, Curtis JD, Freitas TC, Amiel E, Pearce EJ, Pearce EL. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity. 2012;36:68–78. doi: 10.1016/j.immuni.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Windt GJW, O'Sullivan D, Everts B, Huang SCC, Buck MD, Curtis JD, Chang CH, Smith AM, Ai T, Faubert B, Jones RG, Pearce EJ, Pearce EL. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proceedings of the National Academy of Sciences. 2013;110:14336–14341. doi: 10.1073/pnas.1221740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009 doi: 10.1126/science.1160809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vander Heiden MG, DeBerardinis RJ. Understanding the Intersections between Metabolism and Cancer Biology. Cell. 2017;168:657–669. doi: 10.1016/j.cell.2016.12.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vats D, Mukundan L, Odegaard JI, Zhang L, Smith KL, Morel CR, Wagner RA, Greaves DR, Murray PJ, Chawla A. Oxidative metabolism and PGC-1beta attenuate macrophage-mediated inflammation. Cell Metabolism. 2006;4:13–24. doi: 10.1016/j.cmet.2006.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R, Dillon CP, Shi LZ, Milasta S, Carter R, Finkelstein D, McCormick LL, Fitzgerald P, Chi H, Munger J, Green DR. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity. 2011;35:871–882. doi: 10.1016/j.immuni.2011.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T, Marquardt C, Foker J. Aerobic glycolysis during lymphocyte proliferation. Nature. 1976;261:702–705. doi: 10.1038/261702a0. [DOI] [PubMed] [Google Scholar]
- Weinberg F, Hamanaka R, Wheaton WW, Weinberg S, Joseph J, Lopez M, Kalyanaraman B, Mutlu GM, Budinger GRS, Chandel NS. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proceedings of the National Academy of Sciences. 2010;107:8788–8793. doi: 10.1073/pnas.1003428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West AP, Brodsky IE, Rahner C, Woo DK, Erdjument-Bromage H, Tempst P, Walsh MC, Choi Y, Shadel GS, Ghosh S. TLR signalling augments macrophage bactericidal activity through mitochondrial ROS. Nature. 2011;472:476–480. doi: 10.1038/nature09973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nature Publishing Group. 2015;15:486–499. doi: 10.1038/nri3862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson DF. Programming and regulation of metabolic homeostasis. AJP: Endocrinology and Metabolism. 2015;308:E506–17. doi: 10.1152/ajpendo.00544.2014. [DOI] [PubMed] [Google Scholar]
- Wilson DF. Quantifying the role of oxygen pressure in tissue function. Am J Physiol Heart Circ Physiol. 2008;294:H11–3. doi: 10.1152/ajpheart.01293.2007. [DOI] [PubMed] [Google Scholar]
- Wilson DF, Stubbs M, Veech RL, Erecińska M, Krebs HA. Equilibrium relations between the oxidation-reduction reactions and the adenosine triphosphate synthesis in suspensions of isolated liver cells. Biochem J. 1974;140:57–64. doi: 10.1042/bj1400057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolff M, Fandrey J, Jelkmann W. Microelectrode measurements of pericellular PO2 in erythropoietin-producing human hepatoma cell cultures. Am J Physiol. 1993;265:C1266–70. doi: 10.1152/ajpcell.1993.265.5.C1266. [DOI] [PubMed] [Google Scholar]
- Zeng H, Yang K, Cloer C, Neale G, Vogel P, Chi H. mTORC1 couples immune signals and metabolic programming to establish T(reg)-cell function. Nature. 2013;499:485–490. doi: 10.1038/nature12297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Gao P, Fukuda R, Kumar G, Krishnamachary B, Zeller KI, Dang CV, Semenza GL. HIF-1 inhibits mitochondrial biogenesis and cellular respiration in VHL-deficient renal cell carcinoma by repression of C-MYC activity. Cancer Cell. 2007;11:407–420. doi: 10.1016/j.ccr.2007.04.001. [DOI] [PubMed] [Google Scholar]
- Zu XL, Guppy M. Cancer metabolism: facts, fantasy, and fiction. Biochem Biophys Res Commun. 2004;313:459–465. doi: 10.1016/j.bbrc.2003.11.136. [DOI] [PubMed] [Google Scholar]