Abstract
Aims and background
The Ki-67 is a nuclear antigen detected by the monoclonal antibody MIB-1 and its Labeling Index (LI) is considered a marker of normal and abnormal cell proliferation. Pituitary adenomas are generally well differentiated neoplasms, even if in about one third of cases they are invasive of surrounding tissues. The aim of this study is to evaluate the correlation between Ki-67 labelling index and tumor size of pituitary adenomas extimated by means CT and MRI and confirmed at operation.
Methods
Using the monoclonal antibody MIB-1, we evaluated the expression of Ki-67 in 121 anterior pituitary adenomas consecutively operated on in a 48-month period.
Results
In relation to neuroradiological (CT and MRI) and surgically verified tumor size, we identified 24 microadenomas, 27 intrasellar macroadenomas, 34 intra-suprasellar macroadenomas, and 36 intra-supra-parasellar macroadenomas. The adenomas were non-infiltrating (76 cases) and infiltrating (45 cases) adenomas. The wall of the cavernous sinus (CS) was infiltrated in 18 cases. Forty-eight adenomas were non-functioning and 73 functioning. The overall mean ± SD Ki-67 LI was 2.72 ± 2.49% (median 1.6). It was 2.59 ± 1.81 in microadenomas, 2.63 ± 3.45 in intrasellar macroadenomas, 1.91 ± 2.11 in intra-suprasellar macroadenomas, and 3.29 ± 5.45 in intra-supra-parasellar macroadenomas (p = 0.27). It was 3.73 ± 5.13% in infiltrating and 2.03 ± 2.41% in non-infiltrating adenomas (p = 0.02), and 5.61 ± 7.19% in CS-infiltrating versus 2.09 ± 2.37% in CS-non-infiltrating adenomas (p = 0.0005).
Conclusions
Our preliminary results seem to exclude significative correlations between Ki-67 LI and tumor size of anterior pituitary adenomas, even if this index can be considered a useful marker in the determination of the infiltrative behaviour of these tumors.
Introduction
Anterior pituitary adenomas have usually considered benign tumors, even if in about 1/3 of cases they infiltrate surrounding tissues, including the wall of cavernous sinus (CS) [1,2,4-7,18-20,23,25-27,30]. This local "malignancy" contributes to explain the possibility of relapse even after a macroscopically total surgical excision of tumor. As observed for other neoplasms, the determination of cell proliferation activity seems to be useful in making prognosis also in anterior pituitary adenomas. In particular, Ki-67 is a nuclear antigen expressed in the G1, S, G2, and M phases of cellular cycle [17,21,22,24,29,32,34] and its Labeling Index (LI), nowadays recognizable by means the monoclonal antibody MIB-1 [21], is widely considered as a marker of cellular proliferation (growth fraction).
The aim of this study is to investigate the relationship between proliferative activity of anterior pituitary adenomas, quantified with Ki-67 LI, and their size neuroradiologically (CT and MRI) and surgically verified.
Materials and Methods
Patient characteristics
From July, 1994, to July, 1998, 121 patients suffering from an anterior pituitary adenoma were consecutively operated on. In all cases an evaluation of the the Ki-67 LI of surgical specimens was performed by means the monoclonal antibody MIB-1 [21].
The age ranged from 17 to 77 years (mean 43.1 ± 15.5, median 40). The M/F ratio was 1:1.42 (71 females and 50 males). In 21 cases one previous operation and in 2 cases two previous operations for the treatment of the pituitary adenoma had been performed elsewhere (total 23 cases). A functioning adenoma, with clinically expressed hormonal dysfunction, was present in 69 patients (59.5%): in 29 (42.0%) disturbances related to high level of serum PRL were detected, in 28 (40.6%) acromegaly was diagnosed, and in 12 (17.4%) Cushing's disease was observed. One patient had high pre-operative levels of FSH, without correlated clinical symptoms and sign. The preoperative hormone serum level was defined as the highest value observed before surgery. Visual acuity and/or campimetric disturbances were present in 55 cases (45.5%). An intracranial hypertension syndrome was observed in 6 cases (5.0%).
In all cases the neuroradiological diagnosis was obtained using both contrast-enhanced cerebral CT scan and MRI. In relation to the tumor size, the adenoma was considered: A) microadenoma (≤ 10 mm in diameter), 24 cases (19.8%); B) intrasellar macroadenoma, 27 cases (22.3%); C) intra-suprasellar macroadenoma (with extension above the sella turcica), 34 cases (28.1%), and D) intra-supra-parasellar macroadenoma (with extension above the sella and toward the cavernous sinus), 36 cases (29.7%). The tumor size was confirmed in all cases by the impression of surgeon at operation.
A transsphenoidal approach was used in 116 cases (95.9%), whereas a craniotomy was performed in 5 cases (4.1%). In 113 cases (93.4%) a macroscopically total removal and in 8 (6.6%) a subtotal excision of tumor was obtained. In relation to surgically verified infiltration of sellar floor dura and bone, we identified 76 (60.3%) non-infiltrating and 45 (39.7%) infiltrating adenomas. The wall of cavernous sinus was infiltrated in 18 out of 45 cases (14.9% of 121 total cases and 40.0% of 45 infiltrating cases). Histological examination showed in all cases a typical anterior pituitary adenoma. On the basis of immunohistochemical staining we recognized 48 (39.7%) endocrine-inactive and 73 (60.3%) active adenomas (26 PRL, 24 GH, 12 ACTH, 7 mixed, 3 TSH, and 1 FSH). Plurihormonal adenomas were classified according to the predominant endocrine symptoms (in 4 cases GH- and in 3 PRL-related disturbances). Twenty out of 29 patients (68.9%) with prolactinomas or with mixed adenoma with prevalent PRL expression received pre-operative medical therapy (bromocriptine).
Determination of the Ki-67 Labeling Index (MIB-1 immunostaining)
The surgical specimens were routinely processed, fixed in neutral buffered formalin, and embebbed in paraffin. In order to evaluate the Ki-67 antigen staining, 5-micrometer sections, previously mounted onto glass slides and dried, were incubated overnight at 4°C in the MIB-1 antibody (Immunotech, Marseille, France) [21]. Immunostaining was performed using the avidin-biotin-peroxidase method. Ten fields were selected in regions with highest concentrations of MIB-1-positive nuclei and were examinated at high power magnification (x400). Each field corresponded to a total number of cells ranging from 700 to 1000, in relation with the cellularity of the tumor specimen (Fig. 1). Areas of necrosis, normal adenohypophysial cells, and endothelial cells were exluded from the evaluation. On considering 1000 cells with "manual" counting, the Ki-67 Labeling Index has been defined as the percentage of MIB-1 positive cells (dense brown precipitate restricted to the nuclei).
Figure 1.
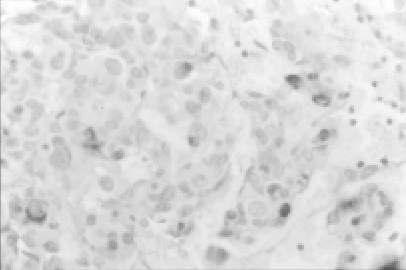
MIB-1 immunostaining of a mixed (GH-PRL) infiltrating pituitary adenoma (original maginification, ×400). A high proportion of labeled nuclei are seen (Ki-67 LI = 10%).
Statistical analysis
Computer-assisted data analysis was performed with a commercially available software (SPSS 6.0, SPSS Inc., Chicago, IL). The normal distribution of Ki-67LI values was verified by using normal Q-Q plots, with the Blom's method. The Χ2 (1 or 2 df, continuity correction) and ANOVA tests were used to identify the statistical significance of differences of Ki-67 LI observed in relation to functioning vs non functioning adenomas, presence or not of pre-operative visual disturbances, neuroradiological and surgical extimated volume of tumor, surgical invasiveness, and infiltration of CS. Values are expressed as the mean ± standard deviation (SD) of the mean; for each comparison, a P value was obtained (significance, ≤ 0.05).
Results
The overall mean Ki-67 LI 2.72% ± 2.49 (median 1.6; range 0-31). In 7 patients the LI was ≥ 10:2 ACTH, 2 GH, 2 PRL, and 1 GH-PRL secreting adenomas; 5 out 7 were infiltrating, 4 of which were CS-infiltrating. In all cases there was not present any histological finding of pituitary carcinomas. We did not observe relevant differences of index in relation to age, sex, previous operations, presence of pre-operative visual disturbances, and functioning activity of tumor (Tab.1). Also the differences of index related to the neuroradiological and surgical extimated tumor size were not significant. The Ki-67 LI was 2.59% ± 1.81 in microadenomas, 2.63% ± 3.45 in intrasellar macroadenomas, 1.91% ± 2.11 in intra-suprasellar macroadenomas, and 3.29% ± 5.45 in intra-supra-parasellar macroadenomas (p = 0.27).
Invasive adenomas has a Ki-67 LI of 3.73 ± 5.13% versus 2.03% ± 2.41 of non-invasive ones (p = 0.02); it was 5.61% ± 7.19 in CS-inffltrating adenomas and 2.09% ± 2.37 in CS-non-inffltrating ones (p = 0.0005).
Discussion
Pituitary tumors are usually classified in microadenomas and macroadenomas [10,11]. In the first group the tumor consists of a well circumscribed nodule smaller than 10 mm in diameter [9], whereas macroadenomas produce enlargement of the contour of sella turcica [10,11]. Jules Hardy [10] proposed a schematic radiological classification of anterior pituitary adenomas as outlined by axial and coronal views of CT scan, today appliable to MRI. He divided pituitary tumors in "enclosed" (intrasellar adenomas) and "invasive" (intrasellar and intra-extrasellar adenomas). Enclosed adenomas are subdivided in Grade 0 (microadenomas). Grade I (macroadenoma with slight lowering of the floor), and Grade II (macroadenoma enlarging the sella, with intact floor). Invasive adenomas are subdivided in Grade III (macroadenomas eroding the floor) and Grade IV (macroadenoma destroying the floor).
Pituitary adenomas may be associated with a suprasellar extension, as well outlined by coronal CT scan and coronal and sagittal MR images. Hardy [10] classified them in five types: A) tumor bulging into the chiasmatic cistern; B) tumor reaching the anterior third ventricle; C) huge suprasellar extension filling entirely the third ventricle; D) parasellar extension into the temporal, frontal, or posterior fossa; and E) lateral expansion towards the cavernous sinus.
In order to simplify these classifications and to reduce the number of subgroups for correlations with Ki-67 LI values, we preferred to subdivide the tumors of our series into microadenomas, intrasellar macroadenomas, intra-suprasellar macroadenomas, and intra-supra-parasellar adenomas, including in the last group also pituitary adenomas infiltrating the wall of cavernous sinus.
The incidence of infiltration among anterior pituitary adenomas varies among the different subtytpes [26,31], also in relation to the criteria used to assess it. An infiltrating behaviour has been demonstrated surgically in about 35% and histologically (microscopic infiltration of dura mater) in about 90% of pituitary adenomas [1,2,4-6,12,15,16,18,20,25-27,30]. It seems reasonable that the most reliable criterion of infiltration, with clinical and prognostic usefulness, is the impression of surgeon during operations [30].
Several cell cycle-specific nuclear antigens have been recognized with various immunohistochemical methods, allowing a reliable evaluation of tumor growth fraction. In particular, Ki-67 is a nuclear antigen nowadays simply recognized by monoclonal antibody MIB-1 [21], typically expressed in proliferating cells during the G1, S, G2, and M phases of the cell-cycle [17,21,22,24,29,21,34]. It turned out to be useful in a number of human neoplasms [3,8,13,33], providing information about cellular proliferation rate and, thus, about long-term prognosis. It revealed to be a practical method that can be used in the routine histological evaluation of brain tumors and, also, of anterior pituitary adenomas. A high growth fraction expressed by a high Ki-67 LI should suggest a high proliferative rate and, thus, tight clinical-radiological follow-up, in relation to possible post-operative relapse of adenoma [14].
Up to date, only few Authors reported limited experiences regarding the evaluation of anterior pituitary adenoma growth fraction based on the expression of the Ki-67 antigen, both using formalin-fixed tissue and in vitro cell culture [1,2,5,6,17-19,28,30]. In particular, in a series of 70 anterior pituitary adenomas and of 7 carcinomas, Thapar et al [30] reported mean MIB-1 values of 1.37% in non-invasive adenomas, of 4.66% in invasive adenomas, and of 11.91% in pituitary carcinomas (p < 0.01). Analogously, in our series LI was 2.03% ± 2.41 in non-infiltrating and 3.73% ± 5.13 in infiltrating adenomas (p = 0.02); moreover, we observed a mean MIB-1 value of 5.61% ± 7.19 in CS-infiltrating versus 2.09% ± 2.37 in CS-non-infiltrating adenomas (p = 0.0005). In relation to the frequency distribution of Ki-67 LIs in normal pituitary glands, in non-invasive pituitary adenomas, in invasive adenomas, and in pituitary carcinomas, Thapar et al [30] established a value of 3% as the threshold LI for distinguishing non-invasive from invasive anterior pituitary adenomas.
With te aim to establish a correlation between the growth fraction of pituitary adenomas and their neuroradiological and surgical extimated tumor size, we evaluated the Ki67 LI by means te monoclonal antibody MIB-1 in 121 patients. As summarized in table 1, the values of mean index were similar among microadenomas, intrasellar macroadenomas, and intra-suprasellar macroadenomas. The mean LI of intra-supra-parasellar macroadenomas (3.29%) was higher than others subgroups and than the overall mean value (2.72%); this difference was not statistically significant (p = 0.27) and presumably reflects the higher incidence of invasive tumors among intra-supra-parasellar adenomas. Therefore, with the exception of invasive adenomas, the different tumor size of pituitary adenoma seems to be correlated to the time of growth rather than to a different growth fraction
Table 1.
Mean Ki-67 LIs of 121 anterior pituitary adenomas.
| N. of cases | Mean LI (%) | P | |
| All patients | 121 | 2.72 | |
| Visual disturbances: yes | 47 | 2.67 | |
| 0.93 | |||
| no | 56 | 2.59 | |
| Functioning adenomas: yes | 73 | 3.13 | |
| 0.09 | |||
| no | 48 | 1.93 | |
| CT/MRI tumor size: microadenomas | 24 | 2.59 | |
| Intrasellar macroadenomas | 27 | 2.63 | 0.27 |
| intra- suprasellar macroadenomas | 34 | 1.91 | |
| intra- supra- parasellar macroadenomas | 36 | 3.29 | |
| Infiltration of surrounding tissues: yes | 45 | 3.73 | |
| 0.02 | |||
| no | 76 | 2.03 | |
| Infiltration of CS: yes | 18 | 5.61 | |
| 0.0005 | |||
| No | 103 | 2.09 |
Conclusion
The evaluation of growth fraction of pituitary tumors, identified by the Ki-67 nuclear antigen detected with the MIB-1 monoclonal antibody, presumably gives further information about their infiltrative behaviour. In our series of 121 cases, the analysis of proliferative rate seems to exclude a definite correlation between the mean value of Ki67 LI and the neuroradiological and surgical extimated tumor size of anterior pituitary adenomas.
Pre-publication history
The pre-publication history for this paper can be accessed here:
Contributor Information
Luciano Mastronardi, Email: mastro@tin.it.
Antonio Guiducci, Email: guiduccia@aospterni.it.
Fabrizio Puzzilli, Email: fpuzzilli@tiscalinet.it.
References
- Asano K, Kubo O, Tajika Y, Huang MC, Takakura K. The relationship between cell proliferation and secretory activity in pituitary adenomas. A review of 63 cases. No To Shinkei. 1996;48:543–549. [PubMed] [Google Scholar]
- Atkin SL, Green VL, Hipkin LJ, Landolt AM, Foy PM, Jeffreys RV, et al. A comparison of proliferation indices in human anterior pituitary adenomas using formalin-fixed tissue and in vitro cell culture. J Neurosurg. 1997;87:85–88. doi: 10.3171/jns.1997.87.1.0085. [DOI] [PubMed] [Google Scholar]
- Brown DC, Gatter KC. Monoclonal antibody Ki-67: its use in histopathology. Histopathology. 1990;17:489–503. doi: 10.1111/j.1365-2559.1990.tb00788.x. [DOI] [PubMed] [Google Scholar]
- Buchfelder M, Fahlbusch R, Adams EF, Kiesewetter F, Thierauf P. Proliferation parameters for pituitary adenomas. Acta Neurochir Suppl (Wien) 1996;65:18–21. doi: 10.1007/978-3-7091-9450-8_7. [DOI] [PubMed] [Google Scholar]
- Daita G, Yonemasu Y. Dural invasion and proliferative potential of pituitary adenomas. Neurol Med Chir. 1996;36:211–214. doi: 10.2176/nmc.36.211. [DOI] [PubMed] [Google Scholar]
- Ekramullah SM, Saitoh Y, Arita N, Ohnishi T, Hayakawa T. The correlation of Ki-67 staining indices with tumour doubling times in regrowing non-functioning pituitary adenomas. Acta Neurochir (Wien) 1996;138:1449–1455. doi: 10.1007/BF01411125. [DOI] [PubMed] [Google Scholar]
- Gandour-Edwards R, Kapadia SB, Janecka IP, Martinez AJ, Barnes L. Biologic markers of invasive pituitary adenomas involving the sphenoid sinus. Mod Pathol. 1995;8:160–164. [PubMed] [Google Scholar]
- Hall PA, Woods AL. Immunohistochemical markers of cellular proliferation: achievements, problems, and prospects. Cell Tissue Kinet. 1990;23:505–522. doi: 10.1111/j.1365-2184.1990.tb01343.x. [DOI] [PubMed] [Google Scholar]
- Hardy J. Microsurgery of hypersecreting pituitary microadenomas. Ann Endocrinol. 1971. pp. 195–196.
- Hardy J. Transsphenoidal surgery of hypersecreting pituitary tumors. Diagnosis and treatment of pituitary tumors. PO Kohler, GT Ross, Eds. Excerpta Medica, American Elsevier, 1973. pp. 179–194.
- Hardy J. Transsphenoidal microsurgical treatment of pituitary tumors. Recent Advances in the Diagnosis and Treatment of Pituitary Tumors. J Linfoot, Ed. Raven Press, 1979. pp. 375–388.
- Henderson WR. The pituitary adenomata. A follow-up study of the surgical results in 338 cases (Dr. Harvey Cushing's series). Br J Surg. 1939;26:811–921. [Google Scholar]
- Hops NJ, Bremm J, Bohl J, Perneczky A. Image analysis of proliferating cells in tumors of the human nervous system: an immunohistological study with the monoclonal antibody Ki-67. Neurosurgery. 1994;35:917–923. doi: 10.1227/00006123-199411000-00017. [DOI] [PubMed] [Google Scholar]
- Hsu DW, Hakim F, Biller BMK, De La Monte S, Zervas NT, Klibanski A, et al. Significance of proliferating cell nuclear antigen index in predicting pituitary adenoma recurrence. J Neurosurg. 1993;78:753–761. doi: 10.3171/jns.1993.78.5.0753. [DOI] [PubMed] [Google Scholar]
- Jefferson G. Extrasellar extension of pituitary adenomas. Proc R Soc Med. 1940;33:433–458. doi: 10.1177/003591574003300717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jefferson G. The invasive adenomas of the anterior pituitary, ed 2 Springfield, 111: Charles C Thomas, 1972. pp. 56–60.
- Knosp E, Kitz K, Perneczky A. Proliferation activity in pituitary adenomas: measurement by monoclonal antibody Ki-67. Neurosurgery. 1989;25:927–930. [PubMed] [Google Scholar]
- Knosp E, Kitz K, Steiner E, Matula C. Pituitary adenomas with parasellar invasion. Acta Neurochir Suppl (Wien) 1991;53:65–71. doi: 10.1007/978-3-7091-9183-5_12. [DOI] [PubMed] [Google Scholar]
- Landolt AM, Shibata T, Kleihues P. Growth rate of human pituitary adenomas. J Neurosurg. 1987;67:803–806. doi: 10.3171/jns.1987.67.6.0803. [DOI] [PubMed] [Google Scholar]
- Martins AN, Hayes GJ, Kempe LG. Invasive pituitary adenomas. J Neurosurg. 1965;22:268–276. doi: 10.3171/jns.1965.22.3.0268. [DOI] [PubMed] [Google Scholar]
- McCormick D, Chong H, Hobbs C. Detection of the Ki-67 antigen in fixed and wax-embebbed sections with the monoclonal antibody MIB-1. Histopathology. 1993;22:355–360. doi: 10.1111/j.1365-2559.1993.tb00135.x. [DOI] [PubMed] [Google Scholar]
- Morimura T, Kitz K, Stein H. Determination of proliferative activities in human brain tumor specimens: a comparison of three methods. J Neurooncol. 1991;10:1–11. doi: 10.1007/BF00151242. [DOI] [PubMed] [Google Scholar]
- Nagashima T, Murovic JA, Hoshino T, Wilson CB, DeArmond SJ. The proliferative potential of human pituitary tumors in situ. J Neurosurg. 1986;64:588–593. doi: 10.3171/jns.1986.64.4.0588. [DOI] [PubMed] [Google Scholar]
- Parkins CS, Darling JL, Gill SS. Cell proliferation in serial biopsies through malignant brain tumors: measurement using Ki-67 antibody labelling. Br J Neurosurg. 1991;5:289–298. doi: 10.3109/02688699109005189. [DOI] [PubMed] [Google Scholar]
- Pernicone PJ, Scheithauer BW. Invasive pituitary adenomas and pituitary carcinomas, in Lloyd RV (ed): Surgical Pathology of the Pituitary Gland Philadelphia, WB Saunders, Co, 1993. pp. 121–136.
- Scheithauer BW, Kovacs KT, Laws ER, Jr, Randall RV. Pathology of invasive pituitary tumors with special reference to functional classification. J Neurosurg. 1986;65:733–744. doi: 10.3171/jns.1986.65.6.0733. [DOI] [PubMed] [Google Scholar]
- Selman WR, Laws ER, Scheithauer BW, Carpenter SM. The occurrence of dural invasion in pituitary adenomas. J Neurosurg. 1986;64:402–407. doi: 10.3171/jns.1986.64.3.0402. [DOI] [PubMed] [Google Scholar]
- Shibuya M, Saito F, Miwa T, Davis RL, Wilson CB, Hoshino T. Histochemical study of pituitary adenomas with Ki-67 and anti-DNA polymerase alpha monoclonal antibodies, bromodeoxyuridine labeling, and nucleolar organizer region counts. Acta Neuropathol (Berl) 1992;84:178–183. doi: 10.1007/BF00311392. [DOI] [PubMed] [Google Scholar]
- Shiraishi T. Cell kinetic analysis of brain tumors using the monoclonal antibody Ki-67: in vitro and in situ study. Acta Med Okayama. 1990;44:197–201. doi: 10.18926/AMO/30424. [DOI] [PubMed] [Google Scholar]
- Thapar K, Kovacs K, Scheithauer BW, Stefaneanu L, Horvath E, Pernicone PJ, et al. Proliferative activity and invasiveness among pituitary adenomas and carcinomas: an analysis using the MIB-1 antibody. Neurosurgery. 1996;38:99–107. doi: 10.1097/00006123-199601000-00024. [DOI] [PubMed] [Google Scholar]
- Trumble HC. Pituitary tumours. Observations on large tumours which have spread widely beyond the confines of the sella turcica. Br J Surg. 1951;39:7–24. doi: 10.1002/bjs.18003915303. [DOI] [PubMed] [Google Scholar]
- Tsanaclis AM, Robert F, Michaud J. The cycling pool of cells within human brain tumors: in situ cytokinetics using the monoclonal antibody Ki-67. Can J Neurol Sci. 1991;18:12–17. doi: 10.1017/s0317167100031243. [DOI] [PubMed] [Google Scholar]
- Wintzer HO, Zipfel I, Schulte-Monting J, Hellerich U, von Kleist S. Ki-67 immunostaining in human breast tumours and its relationship to prognosis. Cancer. 1990;67:421–428. doi: 10.1002/1097-0142(19910115)67:2<421::aid-cncr2820670217>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- Zuber P, Hamou MF, de Tribolet N. Identification of proliferating cells in humas gliomasusing the monoclonal antibody Ki-67. Neurosurgery. 1988;22:364–368. doi: 10.1227/00006123-198802000-00015. [DOI] [PubMed] [Google Scholar]


