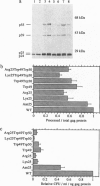
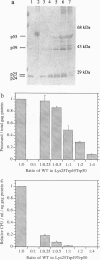
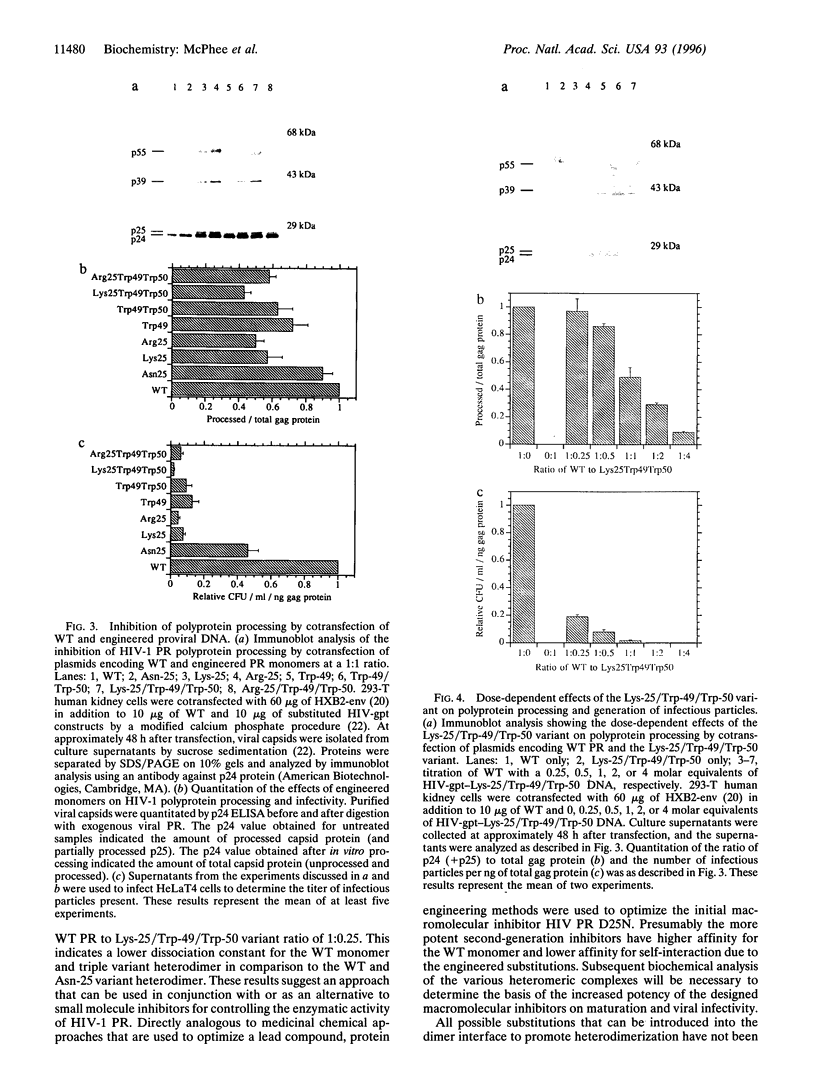
Abstract
Dimerization of human immunodeficiency virus type 1 protease (HIV-1 PR) monomers is an essential prerequisite for viral proteolytic activity and the subsequent generation of infectious virus particles. Disruption of the dimer interface inhibits this activity as does formation of heterodimers between wild-type and defective monomers. A structure-based approach was used to identify amino acid substitutions at the dimer interface of HIV-1 PR that facilitate preferential association of heterodimers and inhibit self-association of the defective monomers. Expression of the designed PR monomers inhibits activity of wild-type HIV-1 PR and viral infectivity when assayed in an ex vivo model system. These results show that it is possible to design PR monomers as macromolecular inhibitors that may provide an alternative to small molecule inhibitors for the treatment of HIV infection.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abdel-Meguid S. S., Zhao B., Murthy K. H., Winborne E., Choi J. K., DesJarlais R. L., Minnich M. D., Culp J. S., Debouck C., Tomaszek T. A., Jr Inhibition of human immunodeficiency virus-1 protease by a C2-symmetric phosphinate. Synthesis and crystallographic analysis. Biochemistry. 1993 Aug 10;32(31):7972–7980. doi: 10.1021/bi00082a019. [DOI] [PubMed] [Google Scholar]
- Babé L. M., Craik C. S. Constitutive production of nonenveloped human immunodeficiency virus type 1 particles by a mammalian cell line and effects of a protease inhibitor on particle maturation. Antimicrob Agents Chemother. 1994 Oct;38(10):2430–2439. doi: 10.1128/aac.38.10.2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babé L. M., Rosé J., Craik C. S. Trans-dominant inhibitory human immunodeficiency virus type 1 protease monomers prevent protease activation and virion maturation. Proc Natl Acad Sci U S A. 1995 Oct 24;92(22):10069–10073. doi: 10.1073/pnas.92.22.10069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bahner I., Zhou C., Yu X. J., Hao Q. L., Guatelli J. C., Kohn D. B. Comparison of trans-dominant inhibitory mutant human immunodeficiency virus type 1 genes expressed by retroviral vectors in human T lymphocytes. J Virol. 1993 Jun;67(6):3199–3207. doi: 10.1128/jvi.67.6.3199-3207.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Condra J. H., Schleif W. A., Blahy O. M., Gabryelski L. J., Graham D. J., Quintero J. C., Rhodes A., Robbins H. L., Roth E., Shivaprakash M. In vivo emergence of HIV-1 variants resistant to multiple protease inhibitors. Nature. 1995 Apr 6;374(6522):569–571. doi: 10.1038/374569a0. [DOI] [PubMed] [Google Scholar]
- Erickson J., Neidhart D. J., VanDrie J., Kempf D. J., Wang X. C., Norbeck D. W., Plattner J. J., Rittenhouse J. W., Turon M., Wideburg N. Design, activity, and 2.8 A crystal structure of a C2 symmetric inhibitor complexed to HIV-1 protease. Science. 1990 Aug 3;249(4968):527–533. doi: 10.1126/science.2200122. [DOI] [PubMed] [Google Scholar]
- Escaich S., Kalfoglou C., Plavec I., Kaushal S., Mosca J. D., Böhnlein E. RevM10-mediated inhibition of HIV-1 replication in chronically infected T cells. Hum Gene Ther. 1995 May;6(5):625–634. doi: 10.1089/hum.1995.6.5-625. [DOI] [PubMed] [Google Scholar]
- Feinberg M. B., Trono D. Intracellular immunization: trans-dominant mutants of HIV gene products as tools for the study and interruption of viral replication. AIDS Res Hum Retroviruses. 1992 Jun;8(6):1013–1022. doi: 10.1089/aid.1992.8.1013. [DOI] [PubMed] [Google Scholar]
- Fitzgerald P. M., McKeever B. M., VanMiddlesworth J. F., Springer J. P., Heimbach J. C., Leu C. T., Herber W. K., Dixon R. A., Darke P. L. Crystallographic analysis of a complex between human immunodeficiency virus type 1 protease and acetyl-pepstatin at 2.0-A resolution. J Biol Chem. 1990 Aug 25;265(24):14209–14219. [PubMed] [Google Scholar]
- Hope T. J., Klein N. P., Elder M. E., Parslow T. G. trans-dominant inhibition of human immunodeficiency virus type 1 Rev occurs through formation of inactive protein complexes. J Virol. 1992 Apr;66(4):1849–1855. doi: 10.1128/jvi.66.4.1849-1855.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaskólski M., Tomasselli A. G., Sawyer T. K., Staples D. G., Heinrikson R. L., Schneider J., Kent S. B., Wlodawer A. Structure at 2.5-A resolution of chemically synthesized human immunodeficiency virus type 1 protease complexed with a hydroxyethylene-based inhibitor. Biochemistry. 1991 Feb 12;30(6):1600–1609. doi: 10.1021/bi00220a023. [DOI] [PubMed] [Google Scholar]
- Kräusslich H. G. Human immunodeficiency virus proteinase dimer as component of the viral polyprotein prevents particle assembly and viral infectivity. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3213–3217. doi: 10.1073/pnas.88.8.3213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- Malim M. H., Böhnlein S., Hauber J., Cullen B. R. Functional dissection of the HIV-1 Rev trans-activator--derivation of a trans-dominant repressor of Rev function. Cell. 1989 Jul 14;58(1):205–214. doi: 10.1016/0092-8674(89)90416-9. [DOI] [PubMed] [Google Scholar]
- Miller M., Schneider J., Sathyanarayana B. K., Toth M. V., Marshall G. R., Clawson L., Selk L., Kent S. B., Wlodawer A. Structure of complex of synthetic HIV-1 protease with a substrate-based inhibitor at 2.3 A resolution. Science. 1989 Dec 1;246(4934):1149–1152. doi: 10.1126/science.2686029. [DOI] [PubMed] [Google Scholar]
- Page K. A., Landau N. R., Littman D. R. Construction and use of a human immunodeficiency virus vector for analysis of virus infectivity. J Virol. 1990 Nov;64(11):5270–5276. doi: 10.1128/jvi.64.11.5270-5276.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosé J. R., Babé L. M., Craik C. S. Defining the level of human immunodeficiency virus type 1 (HIV-1) protease activity required for HIV-1 particle maturation and infectivity. J Virol. 1995 May;69(5):2751–2758. doi: 10.1128/jvi.69.5.2751-2758.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarver N., Rossi J. Gene therapy: a bold direction for HIV-1 treatment. AIDS Res Hum Retroviruses. 1993 May;9(5):483–487. doi: 10.1089/aid.1993.9.483. [DOI] [PubMed] [Google Scholar]
- Smythe J. A., Sun D., Thomson M., Markham P. D., Reitz M. S., Jr, Gallo R. C., Lisziewicz J. A Rev-inducible mutant gag gene stably transferred into T lymphocytes: an approach to gene therapy against human immunodeficiency virus type 1 infection. Proc Natl Acad Sci U S A. 1994 Apr 26;91(9):3657–3661. doi: 10.1073/pnas.91.9.3657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swain A. L., Miller M. M., Green J., Rich D. H., Schneider J., Kent S. B., Wlodawer A. X-ray crystallographic structure of a complex between a synthetic protease of human immunodeficiency virus 1 and a substrate-based hydroxyethylamine inhibitor. Proc Natl Acad Sci U S A. 1990 Nov;87(22):8805–8809. doi: 10.1073/pnas.87.22.8805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thanki N., Rao J. K., Foundling S. I., Howe W. J., Moon J. B., Hui J. O., Tomasselli A. G., Heinrikson R. L., Thaisrivongs S., Wlodawer A. Crystal structure of a complex of HIV-1 protease with a dihydroxyethylene-containing inhibitor: comparisons with molecular modeling. Protein Sci. 1992 Aug;1(8):1061–1072. doi: 10.1002/pro.5560010811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trono D., Feinberg M. B., Baltimore D. HIV-1 Gag mutants can dominantly interfere with the replication of the wild-type virus. Cell. 1989 Oct 6;59(1):113–120. doi: 10.1016/0092-8674(89)90874-x. [DOI] [PubMed] [Google Scholar]