Abstract
Objectives
RNF213 p.R4810K was identified as a susceptibility variant for moyamoya disease in Asia and non-moyamoya intracranial artery stenosis/occlusion disease in Japan and Korea recently. The occurrence of this variant was evaluated in patients with non-moyamoya intracranial artery stenosis/occlusion disease in China.
Methods
Two study populations were used in this study. One was recruited from the Second Hospital of Hebei Medical University from April 2015 to May 2016. The other was the archived DNA samples of intracranial artery stenosis/occlusion patients in XiangYa Hospital collected in 2014. The occurrence of RNF213 p.R4810K was investigated in a total of 715 patients with non-moyamoya intracranial artery stenosis/occlusion disease. The carrier rate of RNF213 p.R4810K in 507 normal individuals was used as control.
Results
Six of 715 patients (0.84%) with non-moyamoya intracranial artery stenosis/occlusion disease and 2 of the 507 normal controls (0.39%) had RNF213 p.R4810K variant. The carrier rate of RNF213 p.R4810K was higher in non-moyamoya intracranial artery stenosis/occlusion group than that in the normal group. However, no statistically significant association was observed (Odds ratio, 2.14; 95% confidence interval, 0.43–10.63; p = 0.56).
Conclusions
The carrier rate of RNF213 p.R4810K in Chinese non-moyamoya intracranial artery stenosis/occlusion disease patients was significantly lower than that in Korea or Japan. Genetic heterogeneity was highly indicated. Further systematic genetic epidemiology studies with emphasis on Chinese-specific genetic variants and environmental risk factors of intracranial artery stenosis/occlusion disease in larger population are needed.
Keywords: Intracranial artery stenosis/occlusion disease, Genetics, RNF213, p.R4810K, China
Introduction
The ring finger protein 213 (RNF213) was identified as a susceptibility gene for moyamoya disease (MMD) recently [1, 2]. Several studies revealed high frequencies of the same RNF213 variant- p.R4810K (rs112735431, GenBank accession number AB537889) in East Asian MMD patients compared to normal controls (ORs > 100), which was illustrated as a founder mutation in Japanese, Korean and Chinese patients [1–9]. Indeed, in Japan and Korea, the majority (~80%) of MMD patients carried at least one allele of RNF213 p.R4810K, which was significantly higher than that in the general population [1, 2, 4–6, 8–16]. In China, the carrier rate of RNF213 p.R4810K in MMD patients was about 20% [1, 3, 17–19]. MMD risk was highly increased by this variant in the Asian population.
RNF213 located in chromosome 17q25.3, encoding a 596 kDa protein which functions both as an AAA‑type ATPase and an E3 ligase [20]. AAA-ATPases mediate various cell functions, including membrane fusion/transport, proteolysis, protein disaggregation/refolding, DNA recombination/repair and mitosis/meiosis [21]. AAA-ATPase dysfunction can cause several diseases, such as PEX1/PEX6 mutations cause multiple organ degeneration [22, 23] and Cdc48 mutations cause amyotrophic lateral sclerosis [24, 25]. E3 ligase activity may play a role in protein degradation or signaling processes [1]. Even the complete physiological functions of RNF213 are still unknown, knockdown of RNF213 in zebrafish leads to abnormal sprouting and irregular diameter of intracranial vessels, suggesting some contribution to vascular formation [1]. Previous studies revealed that a wide spectrum of phenotypes could occur within a family unit despite the members having the identical p.R4810K variant, with some individuals showing the typical phenotype of MMD such as bilateral stenosis/occlusion of the terminal portion of the internal carotid arteries, some showing only unilateral or middle cerebral artery stenosis/occlusion, and others with no abnormalities [1]. Miyawaki et al. reported that a particular subset of Japanese intracranial major artery stenosis/occlusion (ICASO) mainly diagnosed as atherosclerosis not MMD, associated with RNF213 p.R4810K variant (odds ratio, 16.8; 95% confidence interval, 3.81–74.5; p < 0.0001), suggesting that RNF213 p.R4810K variant might cause various severities of ICASO [4, 6]. Recently, Bang et al. also reported that RNF213 p.R4810K is a susceptibility variant not only for MMD but also for ICASO in Korean (odds ratio, 22.3; 95% confidence interval, 3.0–164.1; p < 0.0001) [13]. ICASO is an important and the most frequent cause of cerebral ischemic stroke among patients of Asian ancestry [26]. Liu et al. explored the association of RNF213 p.R4810K with MMD in Chinese population [3, 5, 17–19], however, no association data was available about this variant with ICASO in China.
The aim of present study was to verify the generalizability of previous findings in Japan and Korea, investigating the association of RNF213 p.R4810K variant with ICASO not diagnosed as MMD in a Chinese population, compared with the occurrence of normal individuals as control group.
Materials and methods
Study population
The study population was mainly recruited from the Department of Neurology of Hebei Medical University from October 2015 to May 2016. All the patients with ICASO in the absence of MMD who agreed to participate in this study in this period were enrolled (totally 615). In addition, another 100 ICASO patients in XiangYa Hospital of Central South University who had complete clinical information and archived DNA samples also included in this study. The carrier rate of RNF213 p.R4810K in 507 normal individuals published in other study was used as control [3]. This study was approved by the Medical Ethics Committee of Central South University and the survey participants gave informed consent before the interview and blood samples were taken.
Diagnosis of ICASO
Participants were diagnosed as non-moyamoya ICASO when they experienced focal or lateralizing symptoms and showed ≥50% stenosis or occlusion at terminal and/or proximal portions of the intracranial major arteries without abnormal vascular networks in the basal ganglia on conventional angiography or MRA which was required by MMD diagnosed criteria [27]. The angiography images were interpreted by ≥2 physicians, including at least 1 radiologist and 1 neurological physician. Clinical information, including age, gender, vascular risk factors such as smoking, alcohol consuming, disease histories of hypertension, diabetes, hyperlipemia was collected (these diseases were defined as self-reported physician diagnosis or pharmaceutical treatment). Patients with potential sources of cardioaortic embolism, other stroke mechanisms such as coagulopathy, vasculitis, arterial dissection or incomplete evaluations were excluded.
Identification of RNF213 p.R4810K variant
Peripheral blood samples were obtained from all enrolled patients. Genomic DNA was extracted from the peripheral blood leukocytes using TIANamp Blood DNA Extraction Kit and following the manufacturer’s instructions (TIANGEN BIOTECH CO., LTD, Beijing, China). Genotyping of RNF213 p.R4810K variant was performed by Taqman method (Assay ID: C_153120198_10; TaqMan SNP Genotyping Assays; Applied Biosystems) using a Roche LightCycler® 96 Real-Time PCR System (Roche, Switzerland) and analyzed with the LightCycler® 96 software. The investigators involved in genotyping were blinded from the phenotypic information. All analyses of the genotyped data were performed at the Department of Epidemiology and Health Statistics in Central South University.
Statistical analysis
All statistical analyses were performed using SPSS 21.0 software (SPSS Inc., Chicago, IL, USA). Continuous variable (age) was presented as the mean ± standard deviation (SD). Categorical variables (hypertension, diabetes, hyperlipemia, coronary heart diseases, smoking, drinking) were presented as proportions. Categorical variables were compared using the χ2 test or Fisher exact test, and continuous variables were compared using Student t-tests. A p-value less than 0.05 was considered statistically significant.
Results
This study totally included 715 patients with non-MMD ICASO and 507 normal individuals without known cerebrovascular diseases. Clinical characteristics are shown in Table 1. Six of 715 ICASO patients (0.84%) and 2 of the 507 normal controls (0.39%) had the RNF213 p.R4810K variant (all heterozygotes). Even no statistically significant association was observed, the carrier rate of RNF213 p.R4810K was higher in ICASO group than that in the normal individuals (Odds ratio, 2.14; 95% confidence interval, 0.43–10.63; p = 0.56).
Table 1.
Characteristics of the participants and distribution of the RNF213 p.R4810K variant
| Characteristics | ICASO | Controls |
|---|---|---|
| Number of the participants | 715 | 507 |
| Age (yrs) | ||
| Mean ± SD | 58.4 ± 12.9 | 37.2 ± 16.9 |
| Range | 15–89 | - |
| Female, n (%) | 253 (35.4) | 377 (74.4) |
| Conventional risk factors, n (%) | ||
| Hypertension | 429 (60.0) | - |
| Diabetes | 179 (25.0) | - |
| Hyperlipemia | 290 (40.6) | - |
| Coronary heart diseases | 79 (11.0) | - |
| Smoking | 277 (38.7) | - |
| Drinking | 150 (21.0) | - |
| RNF213 p.R4810K genotype | ||
| Wild type: GG (%) | 709 (99.16) | 505 (99.61) |
| Heterozygous: GA (%) | 6 (0.84) | 2 (0.39) |
| Homozygous: AA (%) | 0 (0) | 0 (0) |
| OR (95% confidence interval) | 2.14 (0.43–10.63) | |
| p value | 0.56 | |
ICASO intracranial major artery stenosis/occlusion, SD standard deviation
Table 2 shows the clinical characteristics of 6 non-MMD ICASO patients with the RNF213 p.R4810K variant. The Fig. 1 shows the MRA images of the 4 patients with ICASO identified with the p.R4810K variant (the digital MRA images of the other 2 patients with the p.R4810K variant in the ICASO group was not available due to the fact that the patients were referrals from the other hospitals). These ICASO patients showed partial stenosis or occlusion of the intracranial major artery without abnormal vascular networks in the basal ganglia on MRA. All the patients had hypertension, 4 patients had diabetes and 2 elderly patients also had coronary heart disease for more than 20 years.
Table 2.
Clinical characteristics of 6 non-MMD ICASO patients with the RNF213 p.R4810K variant
| Case | Sex | Age (yrs) | Site of stenosis/occlusion | Hypertension | Diabetes | Hyperlipemia | CHD | Smoking | Drinking |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 80 | Occlusion of bilateral anterior inferior cerebellar artery and stenosis of left vertebral artery | 5 years | 2 years | 2 years | 20 years | - | - |
| 2 | F | 52 | Occlusion of the A1 segment of right anterior cerebral artery and stenosis of the left middle cerebral artery | 1 month | - | - | - | - | - |
| 3 | M | 42 | Occlusion of the A1 segment of left anterior cerebral artery | 5 years | 5 years | - | - | - | - |
| 4 | F | 48 | Stenosis of bilateral middle cerebral artery and the right anterior cerebral artery | 8 years | 3 years | - | - | - | - |
| 5 | M | 79 | Occlusion of the left middle cerebral artery | 40 years | 14 years | - | 23 years | - | - |
| 6 | M | 63 | Diffuse stenosis of the M1 segment of the left middle cerebral artery | 25 years | - | - | - | 10 years | - |
ICASO intracranial major artery stenosis/occlusion, MMD moyamoya disease,F female, M male, CHD coronary heart disease
Fig. 1.
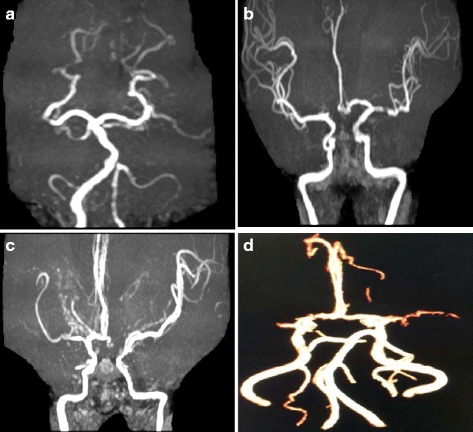
Magnetic resonance angiography (MRA) images of 4 patients with the p.R4810K variant in ICASO patients. a Case 1 was a 80- year- old woman with occlusion of bilateral anterior inferior cerebellar artery and stenosis of left vertebral artery. The patient was relatively elderly and had hypertention, diabetes, hyperlipidemia and coronary heart disease for many years. b Case 2 was a 52- year- old woman with occlusion of the A1 segment of right anterior cerebral artery and stenosis of the left middle cerebral artery. She had serious hypertension (190/100 mmHg) for 1 month. c Case 3 was a 42- year- old man with occlusion of the A1 segment of left anterior cerebral artery. The patient had hypertension and diabetes both for 5 years. d Case 4 was a 48- year- old woman with bilateral middle cerebral artery and the right anterior cerebral artery stenosis. She also had hypertension and diabetes for several years
Table 3 shows the distribution of RNF213 p.R4810K in MMD, Non-MMD ICASO and normal individuals in Japan, Korea and China. The carrier rate of RNF213 p.R4810K in Chinese MMD and non-MMD ICASO patients was significantly lower than that in Korea and Japan.
Table 3.
Comparision of RNF213 p.R4810K distribution in MMD, non-MMD ICASO and normal individuals in Japan, Korea and China
| Diseases | Countries | Cases | Normal individuals | OR (95%CI)a | Reference | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | GG | GA | AA | Carrier rate (%) | MAF% | N | GG | GA | AA | Carrier rate (%) | MAF% | ||||
| MMD | Japan | 105 | 17 | 84 | 4 | 83.81 | 43.81 | 457 | 438 | 19 | 0 | 4.16 | 2.08 | 119.3 (59.7–238.7) | [2] |
| 161 | 16 | 135 | 10 | 90.06 | 48.14 | 384 | 374 | 9 | 1 | 2.60 | 1.43 | 338.9 (150.3–764.2) | [1] | ||
| 204 | 36 | 153 | 15 | 82.35 | 44.85 | 283 | 278 | 5 | 0 | 1.77 | 0.88 | 259.5 (99.9–674.1) | [10] | ||
| 48 | 7 | 40 | 1 | 85.42 | 43.75 | 25 | 25 | 0 | 0 | 0.00 | 0.00 | - | [4] | ||
| 30 | 10 | 19 | 1 | 66.67 | 35.00 | 110 | 108 | 2 | 0 | 1.82 | 0.91 | 108.0 (22.0–530.3) | [6] | ||
| 103 | 27 | 71 | 5 | 73.79 | 39.32 | 95 | 93 | 2 | 0 | 2.11 | 1.05 | 130.9 (30.2–568.1) | [8] | ||
| 78 | 12 | 64 | 2 | 84.62 | 43.59 | - | - | - | - | - | - | - | [11] | ||
| - | - | - | - | - | - | 1474 | 1437 | 34 | 3 | 2.51 | 1.36 | - | [5] | ||
| - | - | - | - | - | - | 4308 | 4248 | 57 | 3 | 1.39 | 0.73 | - | [14] | ||
| - | - | - | - | - | - | 519 | 510 | 9 | 0 | 1.73 | 0.87 | - | [15] | ||
| Korea | 38 | 8 | 30 | 0 | 78.95 | 39.47 | 223 | 217 | 6 | 0 | 2.69 | 1.35 | 135.6 (44.0–417.8) | [1] | |
| 131 | 32 | 99 | 0 | 75.57 | 37.79 | 51 | 51 | 0 | 0 | 0.00 | 0.00 | - | [9] | ||
| 165 | 40 | 112 | 13 | 75.76 | 41.82 | 294 | 286 | 8 | 0 | 2.72 | 1.36 | 111.7 (50.8–245.6) | [12] | ||
| 288 | 89 | 199 | 0 | 69.10 | 34.55 | 83 | 82 | 1 | 0 | 1.20 | 0.60 | 183.3 (25.1–1338.1) | [13] | ||
| - | - | - | - | - | - | 1516 | 1479 | 37 | 0 | 2.44 | 1.22 | - | [16] | ||
| China | 52 | 40 | 11 | 1 | 23.08 | 12.50 | 100 | 98 | 2 | 0 | 2.00 | 1.00 | 14.7 (3.1–68.7) | [1] | |
| 170 | 148 | 21 | 1 | 12.94 | 6.76 | 507 | 505 | 2 | 0 | 0.39 | 0.20 | 37.5 (8.7–161.5) | [3] | ||
| 96 | 87 | 8 | 1 | 9.38 | 5.21 | 96 | 95 | 1 | 0 | 1.04 | 0.52 | 9.8 (1.2–79.2) | [14] | ||
| 81 | 69 | 10 | 2 | 14.81 | 8.64 | 100 | 98 | 2 | 0 | 2.00 | 1.00 | 8.5 (1.8–39.3) | [16] | ||
| 255 | 175 | 78 | 2 | 31.37 | 16.08 | 300 | 300 | 0 | 0 | 0.00 | 0.00 | - | [15] | ||
| - | - | - | - | - | - | 587 | 582 | 5 | 0 | 0.85 | 0.43 | - | [5] | ||
| Non-MMD ICASO | Japan | 41 | 32 | 8 | 1 | 21.95 | 12.20 | 25 | 25 | 0 | 0 | 0.00 | 0.00 | - | [4] |
| 84 | 64 | 20 | 0 | 23.81 | 11.90 | 110 | 108 | 2 | 0 | 1.82 | 0.91 | 16.8 (3.8–74.6) | [6] | ||
| Korea | 221 | 144 | 77 | 0 | 34.84 | 17.42 | 51 | 51 | 0 | 0 | 0.00 | 0.00 | - | [9] | |
| 234 | 184 | 50 | 0 | 21.37 | 10.68 | 83 | 82 | 1 | 0 | 1.20 | 0.60 | 22.3 (3.0–164.1) | [13] | ||
| China | 715 | 709 | 6 | 0 | 0.84 | 0.42 | 507 | 505 | 2 | 0 | 0.39 | 0.20 | 2.14 (0.42–10.63) | This study | |
MMD moyamoya disease, Non-MMD ICASO non-moyamoya intracranial artey stenosis/occlusion disease, N sample size, MAF minor allele frequency
aOdds ratio (95% confidence interval) under dominant model.- can’t calculate
Discussion
In this study, only few of Chinese ICASO patients (6/715, 0.84%) carried RNF213 p.R4810K variant, which was significantly lower than that in Korea or Japan. Genetic heterogeneity of ICASO in different population was highly indicated.
RNF213 was a susceptibility gene for MMD [25]. Previous studies showed that in Japan and Korea, the founder variant RNF213 p.R4810K was much more frequent in MMD patients (~80%) than in the general population (~1.0%), significantly increased MMD risk (ORs > 100) [1, 2, 5, 8, 9, 25]. However, as a susceptibility gene, Chinese MMD patients have significantly different genetic architecture. The carrier rate of RNF213 p.R4810K in Chinese MMD and general population is about 20 ~ 30% and 0.3% respectively, accounting for less part of MMD risk [1, 3, 5, 17–19]. The genetic result is consistent with the unique epidemiological and clinical characteristics of Chinese MMD. In China, no significant difference in sex distribution of MMD, a female predominance is not observed compared to Japan and South Korea. Moreover, the familial occurrence of MMD is lower and the symptoms at the onset are different from those in Japan and South Korea [28]. Genetic heterogeneity is proposed to be partially responsible for the different clinical features of MMD in different ethnicities. It has been proposed that other rare variants of RNF213 may be causative mutations for MMD patients. Zhang et al. systematically investigated RNF213 variants of 255 Chinese MMD, revealing that p.R4810K was identified in 31.4% MMD patients and additional 25 rare variants (absent in controls) were identified in 10.6% of patients without p.R4810K variant. Segregation analysis supported the association between MMD and 2 novel variants (p.H4014T and p.R4160Q) [18]. Similar with MMD, genetic heterogeneity of ICASO in different population was indicated. In previous Japanese and Korean studies [4, 6, 13], a particular subset of ICASO mainly diagnosed as atherosclerosis, associated with RNF213 p.R4810K variant (odds ratios, 16.8 and 22.3; 95% confidence intervals, 3.81–74.5 and 3.0–164.1; both p < 0.0001 for Japanese and Korean, respectively). These findings strongly indicate that some cases of ICASO ascribed to unknown etiology or atherosclerosis might be caused by RNF213 p.R4810K variant. They proposed that RNF213 p.R4810K variant could contribute to the high prevalence of intracranial atherosclerotic stroke in Asians. However, in our study, only 0.84% Chinese ICASO patients (6/715) carried RNF213 p.R4810K variant, no statistically significant association of RNF213 p.R4810K variant with ICASO was observed. RNF213 p.R4810K variant is unlikely playing major role in Chinese ICASO. Population-specific variants might contribute a lot to the pathogenesis. In addition, it has been widely accepted that MMD and ICASO is caused by both genetic and environmental factors. In this study, all 6 ICASO patients with RNF213 p.R4810K variant had hypertension and 4 patients had diabetes. Other unknown genetic and environmental factors may trigger ICASO together. Further systematically investigate Chinese-specific genetic variants of ICASO is needed.
The limitations of this study should be mentioned. First, the analysis of the RNF213 variant was exclusively focused on the RNF213 p.R4810K variant. The other RNF213 variants such as p.H4014T and p.R4160Q were not evaluated. Further comprehensive genetic analysis of RNF213 and other potential genes is necessary to determine whether patients with ICASO without the p.R4810K variant have other variants or not. Second, because the strictly matched cerebrovascular disease-free controls were not available in this study, we used the allele frequency of p.R4810K in previous published control population as control in the association analysis. This may perturb the association results. However, such an approach could be justifiable for rare variants and could not perturb the results significantly, especially in the study with a low carrier rate of target variant in the case group. Third, due to the allele frequency of RNF213 R4810K was low in both case and normal individual group in China, statistic power might be low when exploring the association between RNF213 p.R4810K variant and ICASO in this study. Relevant studies with larger sample sizes are needed to validate our findings.
Conclusion
Even no statistical significance, the carrier rate of RNF213 p.R4810K was higher in ICASO group than that in the normal individuals in China. However, the carrier rate of RNF213 p.R4810K in Chinese ICASO patients was significantly lower than that in Korea or Japan. Population genetic heterogeneity was highly indicated. Further systematic genetic epidemiology studies with larger sample sizes focusing on Chinese-specific genetic variants and environmental risk factors of ICASO are needed.
Acknowledgements
We thank all participants for providing samples and those involved in the sample collection. We also gratefully thank Prof. Akio Koizumi and Dr. Hatatsu Kobayashi from Kyoto University for kindly provided the positive control of RNF213 p.R4810K variant in the genotyping experiments.
Fundings
This work was supported by grants from the National Nature Science Foundation of China, Nature Science Foundation of Hunan Province and China Postdoctoral Science Foundation to J.Y. (No. 81502881, 2017JJ3428 and 2015 M582351). Dr. Yan is a postdoctoral fellow at Central South University (No. 149946), supported by the Postdoctoral International Exchange Plan in China. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Availability of data and materials
The datasets analysed during the current study are available from the corresponding author on reasonable request.
Authors’ contributions
TZ, CG, JX and XW participated in the patient recruitment, sample collection and clinical data explanation. XL, JD and JY carried out the experiments and data analysis. JY took charge of study design, coordination and drafted the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Consent to publication of the MRA images were obtained from the patients.
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Central South University (CTXY-150002-1) and the survey participants gave informed consent before the interview and blood samples were taken.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- ICASO
Non-moyamoya intracranial major artery stenosis/occlusion
- MMD
Moyamoya disease
- RNF213
The ring finger protein 213
References
- 1.Liu W, Morito D, Takashima S, Mineharu Y, Kobayashi H, Hitomi T, et al. Identification of RNF213 as a susceptibility gene for moyamoya disease and its possible role in vascular development. PLoS One. 2011;6:e22542. doi: 10.1371/journal.pone.0022542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kamada F, Aoki Y, Narisawa A, Abe Y, Komatsuzaki S, Kikuchi A, et al. A genome-wide association study identifies RNF213 as the first moyamoya disease gene. J Hum Genet. 2011;56(1):34–40. doi: 10.1038/jhg.2010.132. [DOI] [PubMed] [Google Scholar]
- 3.Wu Z, Jiang H, Zhang L, Xu X, Zhang X, Kang Z, et al. Molecular analysis of RNF213 gene for moyamoya disease in the Chinese Han population. PLoS One. 2012;7:e48179. doi: 10.1371/journal.pone.0048179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Miyawaki S, Imai H, Takayanagi S, Mukasa A, Nakatomi H, Saito N. Identification of a genetic variant common to moyamoya disease and intracranial major artery stenosis/occlusion. Stroke. 2012;43(12):3371–4. doi: 10.1161/STROKEAHA.112.663864. [DOI] [PubMed] [Google Scholar]
- 5.Liu W, Hitomi T, Kobayashi H, Harada KH, Koizumi A. Distribution of moyamoya disease susceptibility polymorphism p.R4810K in RNF213 in East and Southeast Asian populations. Neurol Med Chir (Tokyo) 2012;52(5):299–303. doi: 10.2176/nmc.52.299. [DOI] [PubMed] [Google Scholar]
- 6.Miyawaki S, Imai H, Shimizu M, Yagi S, Ono H, Mukasa A, et al. Genetic variant RNF213 c.14576G > A in various phenotypes of intracranial major artery stenosis/occlusion. Stroke. 2013;44(10):2894–7. doi: 10.1161/STROKEAHA.113.002477. [DOI] [PubMed] [Google Scholar]
- 7.Cecchi AC, Guo D, Ren Z, Flynn K, Santos-Cortez RL, Leal SM, et al. RNF213 rare variants in an ethnically diverse population with moyamoya disease. Stroke. 2014;45(11):3200–7. doi: 10.1161/STROKEAHA.114.006244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moteki Y, Onda H, Kasuya H, Yoneyama T, Okada Y, Hirota K, et al. Systematic validation of RNF213 coding variants in Japanese patients with moyamoya disease. J Am Heart Assoc. 2015;4(5). doi:10.1161/JAHA.115.001862. [DOI] [PMC free article] [PubMed]
- 9.Bang OY, Ryoo S, Kim SJ, Yoon CH, Cha J, Yeon JY, et al. Adult moyamoya disease: a burden of intracranial stenosis in East Asians? PLoS One. 2015;10(6):e0130663. doi: 10.1371/journal.pone.0130663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Miyatake S, Miyake N, Touho H, Nishimura-Tadaki A, Kondo Y, Okada I, et al. Homozygous c.14576G > A variant of RNF213 predicts early-onset and severe form of moyamoya disease. Neurology. 2012;78(11):803–10. doi: 10.1212/WNL.0b013e318249f71f. [DOI] [PubMed] [Google Scholar]
- 11.Miyawaki S, Imai H, Shimizu M, Yagi S, Ono H, Nakatomi H, et al. Genetic analysis of RNF213 c.14576G > A variant in nonatherosclerotic quasi-moyamoya disease. J Stroke Cerebrovasc Dis. 2015;24(5):1075–9. doi: 10.1016/j.jstrokecerebrovasdis.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 12.Kim YJ, Lee JK, Ahn SH, Kim BJ, Kang DW, Kim JS, et al. Nonatheroscleotic isolated middle cerebral artery disease may be early manifestation of moyamoya disease. Stroke. 2016;47(9):2229–35. doi: 10.1161/STROKEAHA.116.012751. [DOI] [PubMed] [Google Scholar]
- 13.Bang OY, Chung JW, Cha J, Lee MJ, Yeon JY, Ki CS, et al. A polymorphism in RNF213 is a susceptibility gene for intracranial atherosclerosis. PLoS One. 2016;11(6):e0156607. doi: 10.1371/journal.pone.0156607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Koizumi A, Kobayashi H, Liu W, Fujii Y, Senevirathna ST, Nanayakkara S, et al. P.R4810K, a polymorphism of RNF213, the susceptibility gene for moyamoya disease, is associated with blood pressure [J] Environ Health Prev Med. 2013;18(2):121–9. doi: 10.1007/s12199-012-0299-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cao Y, Kobayashi H, Morimoto T, Kabata R, Harada KH, Koizumi A. Frequency of RNF213 p.R4810K, a susceptibility variant for moyamoya disease, and health characteristics of carriers in the Japanese population. Environ Health Prev Med. 2016;21(5):387–390. doi: 10.1007/s12199-016-0549-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jang MA, Shin S, Yoon JH, Ki CS. Frequency of the moyamoya-related RNF213 p.Arg4810Lys variant in 1,516 Korean individuals [J] BMC Med Genet. 2015;16:109. doi: 10.1186/s12881-015-0252-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang X, Zhang Z, Liu W, Xiong Y, Sun W, Huang X, et al. Impacts and interactions of PDGFRB, MMP-3, TIMP-2, and RNF213 polymorphisms on the risk of moyamoya disease in Han Chinese human subjects. Gene. 2013;526(2):437–42. doi: 10.1016/j.gene.2013.05.083. [DOI] [PubMed] [Google Scholar]
- 18.Zhang Q, Liu Y, Zhang D, Wang R, Zhang Y, Wang S, et al. RNF213 as the major susceptibility gene for Chinese patients with moyamoya disease and its clinical relevance. J Neurosurg. 2017;126(4):1106–13. doi:10.3171/2016.2.JNS152173. [DOI] [PubMed]
- 19.Huang Y, Cheng D, Zhang J, Zhao W. Association between the rs112735431 polymorphism of the RNF213 gene and moyamoya disease: a case–control study and meta-analysis. J Clin Neurosci. 2016;32:14–8. doi: 10.1016/j.jocn.2015.11.035. [DOI] [PubMed] [Google Scholar]
- 20.Morito D, Nishikawa K, Hoseki J, Kitamura A, Kotani Y, Kiso K, et al. Moyamoya disease-associated protein mysterin/RNF213 is a novel AAA+ ATPase, which dynamically changes its oligomeric state. Sci Rep. 2014;4:4442. doi: 10.1038/srep04442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ogura T, Wilkinson AJ. AAA+ superfamily ATPases: common structure--diverse function. Genes Cells. 2001;6(7):575–97. doi: 10.1046/j.1365-2443.2001.00447.x. [DOI] [PubMed] [Google Scholar]
- 22.Geisbrecht BV, Collins CS, Reuber BE, Gould SJ. Disruption of a PEX1-PEX6 interaction is the most common cause of the neurologic disorders zellweger syndrome, neonatal adrenoleukodystrophy, and infantile refsum disease. Proc Natl Acad Sci U S A. 1998;95(15):8630–5. doi: 10.1073/pnas.95.15.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Reuber BE, Germain-Lee E, Collins CS, Morrell JC, Ameritunga R, Moser HW, et al. Mutations in PEX1 are the most common cause of peroxisome biogenesis disorders. Nat Genet. 1997;17(4):445–8. doi: 10.1038/ng1297-445. [DOI] [PubMed] [Google Scholar]
- 24.Abramzon Y, Johnson JO, Scholz SW, Taylor JP, Brunetti M, Calvo A, et al. Valosin-containing protein (VCP) mutations in sporadic amyotrophic lateral sclerosis. Neurobiol Aging. 2012;33(9):2231.e1–2231.e6. doi: 10.1016/j.neurobiolaging.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koizumi A, Kobayashi H, Hitomi T, Harada KH, Habu T, Youssefian S, et al. A new horizon of moyamoya disease and associated health risks explored through RNF213. Environ Health Prev Med. 2016;21(2):55–70. doi: 10.1007/s12199-015-0498-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caplan LR, Gorelick PB, Hier DB. Race, sex and occlusive cerebrovascular disease: a review. Stroke. 1986;17(4):648–55. doi: 10.1161/01.STR.17.4.648. [DOI] [PubMed] [Google Scholar]
- 27.Research Committee on the Pathology and Treatment of Spontaneous Occlusion of the Circle of Willis. Health Labour Sciences Research Grant for Research on Measures for Infractable Diseases Guidelines for diagnosis and treatment of moyamoya disease (spontaneous occlusion of the circle of Willis) Neurol Med Chir (Tokyo) 2012;52(5):245–66. doi: 10.2176/nmc.52.245. [DOI] [PubMed] [Google Scholar]
- 28.Duan L, Bao XY, Yang WZ, Shi WC, Li DS, Zhang ZS, et al. Moyamoya disease in China: its clinical features and outcomes. Stroke. 2012;43(1):56–60. doi: 10.1161/STROKEAHA.111.621300. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets analysed during the current study are available from the corresponding author on reasonable request.


