ABSTRACT
Lowe syndrome is a rare X-linked disorder characterized by bilateral congenital cataracts and glaucoma, mental retardation, and proximal renal tubular dysfunction. Mutations in OCRL, an inositol polyphosphate 5-phosphatase that dephosphorylates PI(4,5)P2, cause Lowe syndrome. Previously we showed that OCRL localizes to the primary cilium, which has a distinct membrane phospholipid composition, but disruption of phosphoinositides in the ciliary membrane is poorly understood. Here, we demonstrate that cilia from Lowe syndrome patient fibroblasts exhibit increased levels of PI(4,5)P2 and decreased levels of PI4P. In particular, subcellular distribution of PI(4,5)P2 build-up was observed at the transition zone. Accumulation of ciliary PI(4,5)P2 was pronounced in mouse embryonic fibroblasts (MEFs) derived from Lowe syndrome mouse model as well as in Ocrl-null MEFs, which was reversed by reintroduction of OCRL. Similarly, expression of wild-type OCRL reversed the elevated PI(4,5)P2 in Lowe patient cells. Accumulation of sonic hedgehog protein in response to hedgehog agonist was decreased in MEFs derived from a Lowe syndrome mouse model. Together, our findings show for the first time an abnormality in ciliary phosphoinositides of both human and mouse cell models of Lowe syndrome.
KEY WORDS: Lowe syndrome; OCRL; Primary cilia; PI(4,5)P2; Phosphoinositide; Sonic hedgehog
Summary: Lack of OCRL elevates PI(4,5)P2 levels in primary cilia of human patient cells and cells from a mouse model of Lowe syndrome, supporting a role for OCRL in ciliary phosphoinositide regulation.
INTRODUCTION
Oculocerebrorenal syndrome of Lowe (MIM: 309000) is a rare X-linked recessive disorder that presents in young males with bilateral cataracts and glaucoma, as well as renal failure, muscular hypotonia, and mental retardation. The defective gene, OCRL (also known as OCRL1), encodes an inositol polyphosphate 5-phosphatase (Attree et al., 1992; Zhang et al., 1995), which acts on phosphatidylinositol-4,5-bisphosphate [PI(4,5)P2] (Jefferson and Majerus, 1995; Zhang et al., 1995; Zhang and Majerus, 1998). Over 210 mutations in OCRL have been found in patients affected with Lowe syndrome, most of which are located in the phosphatase domain or the C-terminal RhoGAP domain. Decreased 5-phosphatase activity is found in fibroblasts from Lowe patients as well as a 2- to 3-fold elevated ratio of PI(4,5)P2:PI4P.
PI(4,5)P2 levels can affect a wide range of cellular processes, including membrane curvature, signaling scaffold, ion channel functions, and actin cytoskeleton (Gamper and Rohacs, 2012; Zhang et al., 2012). Internal cellular membranes also have distinct phosphoinositide composition that can define the subcellular space (Di Paolo and De Camilli, 2006; Pirruccello and De Camilli, 2012). The unique lipid composition is controlled by kinases and phosphatases that localize to these subcellular compartments, such as OCRL. Mutations in OCRL are known to cause cellular defects in endocytosis (Nández et al., 2014; Vicinanza et al., 2011), endosomal trafficking (Billcliff et al., 2016; Cauvin et al., 2016; Noakes et al., 2011; Swan et al., 2010; van Rahden et al., 2012), actin cytoskeletal rearrangements (Coon et al., 2009; Faucherre et al., 2005; Grieve et al., 2011), autophagy (De Leo et al., 2016), cytokinesis (Dambournet et al., 2011) and primary cilia signaling (see review by Mehta et al., 2014). We and others have shown that the gene product, OCRL, localizes to the primary cilia (Coon et al., 2012; Luo et al., 2012; Rbaibi et al., 2012).
Until recently, the primary cilium has been recognized as a distinct organelle with a unique lipid composition (Rohatgi and Snell, 2010). Consisting of a basal body and an axoneme, the primary cilium has a tightly regulated barrier for lipids and protein (Hu et al., 2010; Jensen et al., 2015). Several groups have shown, that in Joubert syndrome, mutation of another inositol 5-phosphatase, INPP5E, results in PI(4,5)P2 accumulation in the cilia (Chaáez et al., 2015; Garcia-Gonzalo et al., 2015; Xu et al., 2016). The loss of INPP5E in Joubert syndrome has been found to affect G-protein-coupled receptor trafficking and downstream sonic hedgehog signaling.
Here, we present evidence that phosphoinositide levels are dysregulated in primary cilia of Lowe syndrome patients. We found that loss of OCRL resulted in abnormal distribution of PI(4,5)P2 in the proximal regions of cilia. Re-expression of OCRL restored the balance between PI(4,5)P2 and PI4P. We examined the ciliary phosphoinositides in cells from human and mouse models of Lowe syndrome, showing abnormal PI(4,5)P2 distribution in the primary cilium and differential effect of sonic hedgehog signaling in response to agonistic stimulation in mouse fibroblasts.
RESULTS AND DISCUSSION
Primary cilia from Lowe syndrome patient fibroblasts and mouse model of Lowe syndrome exhibit increased ciliary PI(4,5)P2
Based on the previously described distribution of OCRL in the cilia, we hypothesized that cells derived from patients with Lowe syndrome may also exhibit dysregulation of ciliary phosphoinositides. Using a monoclonal antibody against PI(4,5)P2, we first examined the distribution of PI(4,5)P2 within the ciliary membrane of fibroblasts from two unrelated Lowe syndrome patients and one control fibroblast line (Luo et al., 2014). Primary cilia formation was induced by serum starvation for 48 h, and ciliary markers of acetylated α-tubulin and PI(4,5)P2 were assessed in fibroblasts. While the normal human fibroblasts (NHF558) did not show any PI(4,5)P2 along the cilium, Lowe 3265 fibroblasts exhibited accumulation of PI(4,5)P2 along the length of the cilia, as well as increased staining near the base of the cilia (Fig. 1A). Similarly, Lowe 1676 cells showed PI(4,5)P2 staining along both the base and the membrane portion of the cilia. Average PI(4,5)P2 ciliary intensity was markedly higher in Lowe patients fibroblasts than controls (Fig. 1B). Only 4% of PI(4,5)P2 intensity was observed in the cilia of NHF558 cells, compared with 30% in Lowe 3265 and 40% in Lowe 1676 cells.
Fig. 1.
Increased PI(4,5)P2 in cilia of fibroblasts derived from Lowe syndrome patients and mouse model of Lowe syndrome. Wild-type normal human (NHF558), Lowe 3265 and Lowe 1676 fibroblasts were immunostained with antibodies against PI(4,5)P2 and acetylated α-tubulin/Arl13b. (A) Elevated PI(4,5)P2 staining in cilia of Lowe fibroblasts. (B) Quantification of the amount of PI(4,5)P2 fluorescence intensity of cilia of Lowe fibroblasts. Data are shown as means±s.d. *P<0.001 in unpaired t-tests. (C) Phosphoinositide PH domain markers in primary cilia of fibroblasts. Citrine PLCδ-PH [marker for PI(4,5)P2] and Citrine FAPP1-PH (marker for PI4P) were transfected, followed by immunostaining for Arl13b. (D) Wild type, Ocrl −/−, Ocrl−/− Inpp5b−/− INPP5B+/+ (IOB−/−) and control Ocrl+/Y:Inpp5b−/− :INPP5B+/+ (IOB+/Y) MEFs were immunostained with antibodies against PI(4,5)P2, PI4P and Arl13b. (E) Quantification of PI(4,5)P2 and PI4P fluorescence intensity in cilia of Ocrl−/−, IOB−/− and IOB+/Y MEFs relative to wild-type MEFs. Error bars represent s.d.; n=3. *P<0.001 in unpaired t-tests. Scale bars: 2 µm.
To confirm the difference of PI(4,5)P2 in Lowe patients and normal human fibroblasts, all three fibroblast cell lines were transfected with phosphoinositide-specific pleckstrin homology (PH) domain proteins: four-phosphate-adaptor protein 1 (FAPP1-PH), which is a marker for PI4P; and phospholipase C-δ1 (PLCδ-PH), which is a marker for PI(4,5)P2 (Hardie et al., 2015; Lenoir et al., 2015). Consistent with the ciliary staining pattern by the monoclonal antibody for the endogenous forms of PI(4,5)P2, we observed a build-up of PI(4,5)P2 and a decrease of PI4P in the cilia of two Lowe fibroblast samples (Fig. 1C). PLCδ-PH levels were significantly higher in cilia of both Lowe fibroblast types, whereas FAPP1-PH levels showed a 25% presence in cilia of NHF558 cells and 12% in Lowe fibroblasts (Fig. S1), supporting the pattern observed for endogenous PI(4,5)P2 build-up in the Lowe syndrome fibroblasts.
To further examine the role of OCRL in ciliary phosphoinositide regulation, we turned to an Ocrl-deficient mouse model (Bothwell et al., 2011) (phenotype summarized in Fig. S2). In this mouse model (Ocrl−/−, Inpp5b−/−, INPP5B+/+, also known as IOB−/− mice), human type II inositol polyphosphate 5-phosphatase (INPP5B), a paralog of OCRL, which was implicated to play a compensatory role in vivo (Jänne et al., 1998; Bothwell et al., 2010; Luo et al., 2013; Matzaris et al., 1998; Speed et al., 1995), was introduced to rescue the embryonic lethality in Ocrl−/− Inpp5b−/− double knockout mice (Jänne et al., 1998). Bothwell et al. (2011) reported that the IOB−/− mice developed renal tubular disease consistent with that of Lowe syndrome patients compared with the littermate control (Ocrl+/Y, Inpp5b−/−, INPP5B+/+; IOB+/Y mice).
We hypothesized that in the mice that have deficiency in both OCRL and INPP5B, there would be an increase in PI(4,5)P2 in the cilia and a corresponding decrease in PI4P. IOB−/− mouse embryonic fibroblasts (MEFs) were isolated, cultured and serum starved to induce primary cilia formation. Phosphoinositide distribution was examined by immunofluorescence using monoclonal antibodies against PI4P and PI(4,5)P2 (De Leo et al., 2016; Garcia-Gonzalo et al., 2015). We observed an increased level of PI(4,5)P2 in the proximal regions of the cilia and very little ciliary PI4P (Fig. 1D,E). As a critical control, IOB+/Y MEFs have a functional Ocrl gene present; in these cells, we observed positive staining of PI4P in the cilia and a corresponding absence of PI(4,5)P2 in both the transition zone as well as along the ciliary axoneme. Furthermore, Ocrl−/− MEFs, which have been previously shown to express wild-type INPP5B (Bothwell et al., 2011), had PI(4,5)P2 in both the transition zone as well as along the length of the cilium, whereas wild-type MEFs had PI4P localized to the ciliary membrane along the axoneme. Taken together, the accumulation of PI(4,5)P2 in OCRL-deficient cells, both human and mouse, indicates a critical role for OCRL in phosphoinositide regulation.
Subcellular distribution of PI(4,5)P2 in the transition zone of primary cilia in OCRL-deficient cells
Based on the localization of OCRL to the proximal regions of the cilium (Fig. S3), we then sought to determine the subcellular distribution using specific markers within the cilia. The previous pattern of cilia membrane staining suggests a transition zone pattern for PI(4,5)P2 accumulation in Lowe fibroblasts. To test this hypothesis, TCTN1 was used for co-immunostaining of the transition zone.
TCTN1 is a transmembrane protein localized at the transition zone that forms complexes with other ciliary proteins (Garcia-Gonzalo et al., 2011; Yee et al., 2015). Distribution of TCTN1 was detected at the ciliary base of RPE cells, with immunoreactivity tightly organized to the transition zone. Upon primary cilia induction in fibroblasts, Arl13b and γ-tubulin were used to stain for the axoneme and the base of the cilia. In both Lowe 3265 and, to a lesser extent, Lowe 1676 fibroblasts, an extension of PI(4,5)P2 into the proximal regions of the cilia was seen, as confirmed with TCTN1 staining (Fig. 2A).
Fig. 2.
Subcellular distribution of PI(4,5)P2 within cilia of Lowe syndrome fibroblasts. Lowe syndrome fibroblasts exhibited increased PI(4,5)P2 in the transition zone of cilia. (A) Fibroblasts were immunostained with antibodies against Arl13b, γ-tubulin and PI(4,5)P2 or TCTN1. (B) Fibroblasts were immunostained with antibodies against acetylated α-tubulin and PI(4,5)P2 or MKS5. Scale bars: 2 µm.
To further corroborate the distribution of PI(4,5)P2 in Lowe patient cells, MKS5 immunostaining was performed. Merkel syndrome type 5 (MKS5) has been recently shown to localize within the transition zone of cilia, and to regulate PI(4,5)P2 distribution in Caenorhabditis elegans (Jensen et al., 2015). Therefore, we hypothesized that the PI(4,5)P2 distribution defect may be secondary to abnormal localization of MKS5. However, MKS5 remained localized to the transition zone along with the accumulation of PI(4,5)P2 in Lowe fibroblasts (Fig. 2B), suggesting that either MKS5 and OCRL do not associate along the same regulatory pathway of PI(4,5)P2, or there is a difference in MKS5 physiology between human and C. elegans cilia. Taken together, these data support that the loss of OCRL results in a build-up of PI(4,5)P2 at the transition zone and proximal regions of the ciliary membrane.
Expression of wild-type OCRL but not a phosphatase-dead mutant rescues the ciliary PI(4,5)P2 accumulation
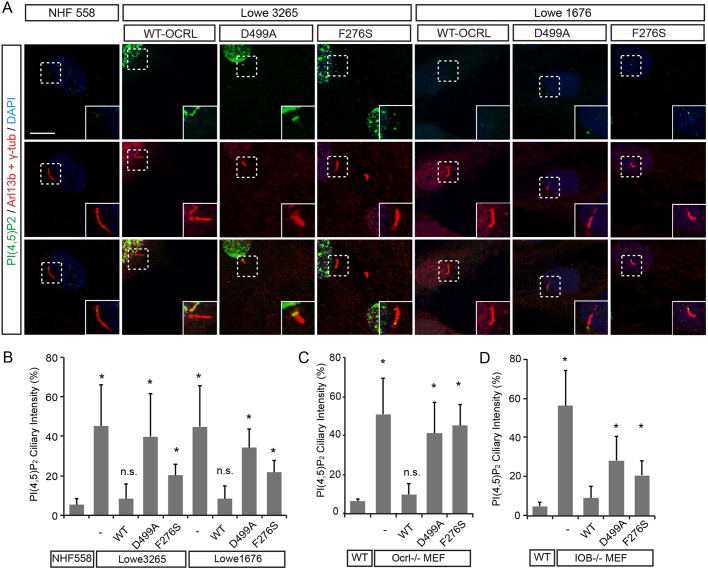
To determine whether the elevated PI(4,5)P2 levels is caused by OCRL, we then assessed whether the expression of wild-type OCRL can rescue the ciliary phosphoinositide accumulation defects in patient fibroblasts. Reduction of OCRL in Lowe patient cells was previously measured (Luo et al., 2012). Transfection of FLAG-tagged OCRL, either wild type, an enzymatic dead mutant of OCRL (D499A), or an intact phosphatase but defective mutant (F276S) was performed in NHF558, Lowe 3265 and Lowe 1676 cells (Chabaa et al., 2006; Song et al., 2017). Primary cilia were induced by serum starvation, followed by immunostaining for Arl13b and PI(4,5)P2 antibodies. Consistent with the hypothesis that OCRL expression directly underlies the increased levels of PI(4,5)P2, introduction of wild-type but not the D499A or F276S mutant form of OCRL was able to deplete ciliary PI(4,5)P2 (Fig. 3A). PI(4,5)P2-positive ciliary immunostaining decreased from 45% to 8% in Lowe 3265 cells, and from 44% to 8% in Lowe 1676 cells (Fig. 3B). Moreover, a significant decrease in ciliary PI(4,5)P2 level was observed in both Ocrl-null and IOB−/− MEFs (Fig. 3C,D).
Fig. 3.
Expression of wild-type OCRL restores levels of ciliary PI(4,5)P2. (A) Lowe syndrome fibroblasts were transfected with WT-OCRL, D499A-OCRL or F276S-OCRL, together with NHF558, followed by immunostaining with antibodies against PI(4,5)P2, Arl13b and γ-tubulin. Scale bar: 2 µm. (B) Quantification of the amount of PI(4,5)P2 fluorescence intensity in cilia of transfected Lowe syndrome fibroblasts relative to control NHF558 fibroblasts. *P<0.001 in unpaired t-tests. (C,D) Quantification of ciliary PI(4,5)P2 levels relative to wild-type MEFs, Ocrl −/− (C) and IOB−/− (D) MEFs. MEFs were transfected with WT-OCRL, D499A-OCRL or F276S-OCRL and immunostained with antibodies against PI(4,5)P2 and Arl13b. Error bars represent s.d.; n=3. *P<0.001 in unpaired t-tests, n.s., non significant.
Overall, elevated PI(4,5)P2 levels in the absence of OCRL suggest that OCRL is required to control the normal level of PI(4,5)P2. Several lines of evidence support the role of OCRL as a regulator of PI(4,5)P2 in cilia. First, OCRL has higher affinity for PI(4,5)P2 than PI(3,4,5)P3, with a lower dissociation constant (KD) compared with INPP5E (Kisseleva et al., 2000). Second, in vivo assays for PI(4,5)P2 function [e.g. actin cytoskeletal rearrangement (Faucherre et al., 2005; Coon et al., 2009), cytokinesis (Dambournet et al., 2011), endocytosis (Erdmann et al., 2007; Vicinanza et al., 2011)] indicated that OCRL affects PI(4,5)P2 levels directly. Third, loss of OCRL results in increased global levels of PI(4,5)P2 in Lowe syndrome patients (Ungewickell and Majerus, 1999). Fourth, expression of OCRL in the Lowe syndrome mouse model shows a rescue of the phenotype for PI(4,5)P2 accumulation in the cilia. Taken together, these findings support the idea that specific phosphoinositides such as PI(4,5)P2 have natural low levels of localization to the ciliary compartment in normal cells, with OCRL playing a critical role in their regulation. In this regard, deficiency in OCRL causes defects in PI(4,5)P2 regulation and accumulation within the ciliary compartment, as seen in our study.
Sonic hedgehog signaling pathway activation reduces GPR161 localization in Lowe patient fibroblasts and restores smoothened levels in mouse fibroblasts
In addition to OCRL, recent work by several groups has shown that INPP5E is also present in the cilia and is a key phosphatase for PI(4,5)P2, which is upregulated in the INPP5E MEFs, C. elegans, Drosophila and in human knockdown cells (Chávez et al., 2015; Garcia-Gonzalo et al., 2015). Studies show that accumulation of the INPP5E phosphoinositide substrates PI(4,5)P2 and PI(3,4,5)P3 was accentuated at the transition zone in hedgehog (Hh)-stimulated Inpp5e−/− cells. Interestingly, these were further associated with reduced ciliary smoothened (Smo) levels which could be rescued by expression of wild-type, but not 5-phosphatase-dead INPP5E (Dyson et al., 2017), suggesting a ciliary gatekeeper role for INPP5E, and possibly OCRL, in the transition zone of primary cilia.
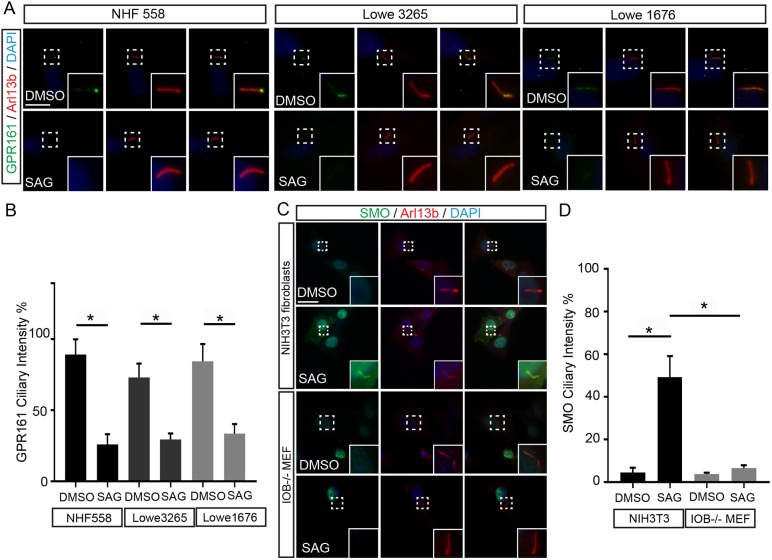
In this regard, we treated Lowe syndrome and NHF558 fibroblasts with an agonist for hedgehog signaling (SAG) overnight, followed by serum starvation for 24 h, and stained for GPR161 and Arl13b. There was a significant reduction of GPR161 in both mutant and normal cells after treatment with SAG (Fig. 4A,B). Furthermore, NIH3T3 fibroblasts and IOB−/− fibroblasts were transfected with Smo–mCherry–GCamp3 (Delling et al., 2013). After treatment with SAG overnight and 24 h serum starvation, only the NIH3T3 fibroblasts exhibited a significant increase in Smo levels, without restoration to the IOB−/− fibroblasts (Fig. 4C,D). This discrepancy between normal and mutant cells suggests that OCRL deficiency impairs normal response to SAG in promoting Smo levels to the primary cilium. Previous reports have shown that loss of INPP5E curbs the response to Hh transcriptional activity (Garcia-Gonzalo et al., 2015). Our work adds to the complexity of this picture by suggesting a role of OCRL in regulation of proteins near the ciliary base, such as Smo, as opposed to INPP5E which plays a regulatory role to ciliary GPR161.
Fig. 4.
Sonic hedgehog signaling pathway activation reduces GPR161 and restores smoothened levels. (A) NHF558 and Lowe fibroblasts were treated with SAG or vehicle (DMSO) and stained for GPR161 and Arl13b. (B) Quantification of GPR161 intensity relative to cilia in DMSO or SAG-treated NHF558 and Lowe fibroblasts. *P<0.001 in unpaired t-tests. (C) NIH3T3 mouse fibroblasts and IOB−/− mouse fibroblasts were treated with SAG or vehicle (DMSO) and stained for Smo and Arl13b. (D) Quantification of Smo intensity in cilia of DMSO or SAG-treated MEFs. Error bars represent s.d.; n=3.*P<0.001 in unpaired student t-tests. Scale bars: 2 µm.
In summary, our results suggest that Lowe syndrome cells exhibit defects in primary cilium function or regulation. These, in turn, may contribute to abnormal aqueous humor filtration and/or renal tubular filtration. Many studies have shown a close involvement of primary cilia in different settings. Of most interest is their potential role as flow sensors and mechanotransducers to regulate tissue homeostasis in different organs, for example, in eye, brain and kidneys. In this regard, we suggest that OCRL plays a crucial role in regulating PI(4,5)P2 levels in the primary cilium and that deficiency causes an alteration of downstream pathways, which ultimately affects fluid transport in the affected organ systems.
MATERIALS AND METHODS
Reagents
Anti-Arl13b mouse antibody (75-287) was purchased from Antibodies Inc (David, CA). Anti-γ-tubulin rabbit antibody (620901) was purchased from Biolegend (San Diego, CA). Anti-acetylated α-tubulin rabbit antibody (5335) was purchased from Cell Signaling Technology (Danvers, MA). Anti-TCTN1 (15004-1-AP), anti-Arl13b (17711-1-AP), anti-MKS5 (55160-1-AP), rabbit polyclonal antibodies were purchased from ProteinTech. Anti-PI4P (Z-P004) and PI(4,5)P2 (Z-P045) antibodies were obtained from Echelon (Salt Lake City, UT). Anti-Smoothened (ab72130), was purchased from Abcam (Cambridge, MA). All primary antibodies were used at 1:500 dilution, except for PI(4,5)P2 and Smoothened (1:200). Secondary antibodies Alexa Fluor 488 (A11008) and Alexa Fluor 594 (A11012) anti-rabbit IgG, and Alexa Fluor 488 anti-mouse IgG+IgM (1857664) and Alexa Fluor 546 anti-mouse IgG (A11030) were procured from Life Technologies/Invitrogen (Carlsbad, CA). All secondary antibodies were applied at 1:1000. SAG (11914) was purchased from Cayman Chemical (Ann Arbor, MI). Treatment of SAG was conducted overnight at 400 nM in Opti-MEM.
Cell culture and mouse embryonic fibroblast isolation
Three human fibroblast (NHF558, Lowe3265 and Lowe1676) and human retinal pigmented epithelial (RPE) were gathered as previously described (Luo et al., 2014). Ocrl−/− and Inpp5b−/− Ocrl−/− INPP5B+/+ mice were a gift from Robert L. Nussbaum (University of California, San Francisco). NIH3T3 mouse fibroblasts were a gift from Jeffrey Goldberg (Stanford University). All MEFs were derived from E14.5 embryos without brain, limbs and internal organs. Embryos were washed with sterile PBS, minced into small pieces with sterile razor blade, digested with trypsin in 37°C for 15 min, homogenized by pipetting, and plated in a 10 cm dish with DMEM and 10% FBS. All animal experiments followed the guidelines of the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Institutional Animal Care and Use Committee of Indiana University School of Medicine.
DNA plasmids and transfection
Citrine-tagged FAPP-PH and Citrine-tagged PLCδ-PH constructs were gifts from Clark Wells (Indiana University, USA) (Heller et al., 2010). Smo–mCherry–GCaMP3 was a gift from David Clapham (Harvard University, USA).
Immunofluorescence
For immunofluorescence, cells were washed with PBS and fixed with 4% paraformaldehyde in PBS for 20 min at RT. Cells were washed 3 times with PBS, permeabilized in PBST (0.5% Triton X-100 in PBS) for 10 min at RT, and blocked with 10% NGS blocking buffer for 1 h at 37°C. After another wash in PBS, cells were incubated with the respective primary antibody overnight at 4°C. After 3 washes, cells were incubated with secondary antibody for 1 h at 37°C, washed 3 times, stained with DAPI, washed 4 times, and sealed in anti-fade reagent. Ciliary Arl13b or acetylated α-tubulin served as markers for primary cilia. Using ImageJ (v1.47v, NIH), each cilium in the ciliary channel and merged channels and area and total fluorescence intensity were measured. The percentages of PI4P and PI(4,5)P2 intensity in cilia were calculated by (total ciliary intensity in merged channels – ciliary channel)/total intensity in ciliary channel×100.
Statistical analysis
Results are expressed as mean values±s.d. Statistical analysis was performed using Student t-tests or ANOVA as appropriate (SPSS). A P-value of less than 0.01 was considered statistically significant.
Supplementary Material
Acknowledgements
We thank Drs Timothy Corson, Michael Boulton, and John Spence for thoughtful comments during the preparation of this manuscript, Dr Robert Nussbaum for the gift of Lowe syndrome mouse model and Dr Clark Wells for the phosphoinositide fluorescent protein constructs.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
Conceptualization: P.P.P., Y.S.; Validation: P.P.P.; Formal analysis: N.L., J.A.A., Y.H., Y.S.; Resources: Y.S.; Data curation: P.P.P., N.L.; Writing - original draft: N.L., Y.S.; Writing - review & editing: P.P.P., N.L., B.W., J.A.A., Y.H., Y.S.; Supervision: Y.S.; Project administration: Y.S.; Funding acquisition: Y.S.
Funding
This work was supported by the National Institutes of Health [K08-EY022058 (Y.S.), R01-EY025295 (Y.S.), R01-EY-023295 (Y.H.) R01-EY024932 (Y.H.)], US Department of Veterans Affairs [CX001298 (Y.S.)], E. Matilda Ziegler Foundation for the Blind (Y.S.), Ralph W. and Grace M. Showalter Research Trust Fund (Y.S.), Research for Prevention of Blindness unrestricted grant (Glick Eye Institute), American Glaucoma Society (Y.S.), Lowe syndrome association (Y.S.) and Knights Templar Eye Foundation (Y.S.). Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://jcs.biologists.org/lookup/doi/10.1242/jcs.200857.supplemental
References
- Attree O., Olivos I. M., Okabe I., Bailey L. C., Nelson D. L., Lewis R. A., McInnes R. R. and Nussbaum R. L. (1992). The Lowe's oculocerebrorenal syndrome gene encodes a protein highly homologous to inositol polyphosphate-5-phosphatase. Nature 358, 239-242. 10.1038/358239a0 [DOI] [PubMed] [Google Scholar]
- Billcliff P. G., Noakes C. J., Mehta Z. B., Yan G., Mak L., Woscholski R. and Lowe M. (2016). OCRL1 engages with the F-BAR protein pacsin 2 to promote biogenesis of membrane-trafficking intermediates. Mol. Biol. Cell 27, 90-107. 10.1091/mbc.E15-06-0329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bothwell S. P., Farber L. W., Hoagland A. and Nussbaum R. L. (2010). Species-specific difference in expression and splice-site choice in Inpp5b, an inositol polyphosphate 5-phosphatase paralogous to the enzyme deficient in Lowe Syndrome. Mamm. Genome 21, 458-466. 10.1007/s00335-010-9281-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bothwell S. P., Chan E., Bernardini I. M., Kuo Y.-M., Gahl W. A. and Nussbaum R. L. (2011). Mouse model for Lowe syndrome/Dent Disease 2 renal tubulopathy. J. Am. Soc. Nephrol. 22, 443-448. 10.1681/ASN.2010050565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cauvin C., Rosendale M., Gupta-Rossi N., Rocancourt M., Larraufie P., Salomon R., Perrais D. and Echard A. (2016). Rab35 GTPase triggers switch-like recruitment of the Lowe syndrome lipid phosphatase OCRL on newborn endosomes. Curr. Biol. 26, 120-128. 10.1016/j.cub.2015.11.040 [DOI] [PubMed] [Google Scholar]
- Chabaa L., Monnier N., Dahri S., Jorio M., Lunardi J. and Chabraoui L. (2006). [Oculo-cerebro-renal Lowe syndrome: clinical, biochemical and molecular studies in a Moroccan patient]. Ann. Biol. Clin. (Paris) 64, 53-59. [PubMed] [Google Scholar]
- Chávez M., Ena S., Van Sande J., de Kerchove d'Exaerde A., Schurmans S. and Schiffmann S. N. (2015). Modulation of ciliary phosphoinositide content regulates trafficking and sonic hedgehog signaling output. Dev. Cell 34, 338-350. 10.1016/j.devcel.2015.06.016 [DOI] [PubMed] [Google Scholar]
- Coon B. G., Mukherjee D., Hanna C. B., Riese D. J. II, Lowe M. and Aguilar R. C. (2009). Lowe syndrome patient fibroblasts display Ocrl1-specific cell migration defects that cannot be rescued by the homologous Inpp5b phosphatase. Hum. Mol. Genet. 18, 4478-4491. 10.1093/hmg/ddp407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coon B. G., Hernandez V., Madhivanan K., Mukherjee D., Hanna C. B., Barinaga-Rementeria Ramirez I., Lowe M., Beales P. L. and Aguilar R. C. (2012). The Lowe syndrome protein OCRL1 is involved in primary cilia assembly. Hum. Mol. Genet. 21, 1835-1847. 10.1093/hmg/ddr615 [DOI] [PubMed] [Google Scholar]
- Dambournet D., Machicoane M., Chesneau L., Sachse M., Rocancourt M., El Marjou, A., Formstecher E., Salomon R., Goud B. and Echard A. (2011). Rab35 GTPase and OCRL phosphatase remodel lipids and F-actin for successful cytokinesis. Nat. Cell Biol. 13, 981-988. 10.1038/ncb2279 [DOI] [PubMed] [Google Scholar]
- De Leo M. G., Staiano L., Vicinanza M., Luciani A., Carissimo A., Mutarelli M., Di Campli A., Polishchuk E., Di Tullio G., Morra V. et al. (2016). Autophagosome-lysosome fusion triggers a lysosomal response mediated by TLR9 and controlled by OCRL. Nat. Cell Biol. 18, 839-850. 10.1038/ncb3386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delling M., DeCaen P. G., Doerner J. F., Febvay S. and Clapham D. E. (2013). Primary cilia are specialized calcium signalling organelles. Nature 504, 311-314. 10.1038/nature12833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Paolo G. and De Camilli P. (2006). Phosphoinositides in cell regulation and membrane dynamics. Nature 443, 651-657. 10.1038/nature05185 [DOI] [PubMed] [Google Scholar]
- Dyson J. M., Conduit S. E., Feeney S. J., Hakim S., Di Tommaso T., Fulcher A. J., Sriratana A., Ramm G., Horan K. A., Gurung R., Wicking C., Smyth I. and Mitchell C. A. (2017). INPP5E regulates phosphoinositide-dependent cilia transition zone function. J. Cell Biol. 216, 247-263. 10.1083/jcb.201511055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erdmann K. S., Mao Y., McCrea H. J., Zoncu R., Lee S., Paradise S., Modregger J., Biemesderfer D., Toomre D. and De Camilli P. (2007). A role of the Lowe syndrome protein OCRL in early steps of the endocytic pathway. Dev. Cell 13, 377-390. 10.1016/j.devcel.2007.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faucherre A., Desbois P., Nagano F., Satre V., Lunardi J., Gacon G. and Dorseuil O. (2005). Lowe syndrome protein Ocrl1 is translocated to membrane ruffles upon Rac GTPase activation: a new perspective on Lowe syndrome pathophysiology. Hum. Mol. Genet. 14, 1441-1448. 10.1093/hmg/ddi153 [DOI] [PubMed] [Google Scholar]
- Gamper N. and Rohacs T. (2012). Phosphoinositide sensitivity of ion channels, a functional perspective. Subcell. Biochem. 59, 289-333. 10.1007/978-94-007-3015-1_10 [DOI] [PubMed] [Google Scholar]
- Garcia-Gonzalo F. R., Corbit K. C., Sirerol-Piquer M. S., Ramaswami G., Otto E. A., Noriega T. R., Seol A. D., Robinson J. F., Bennett C. L., Josifova D. J. et al. (2011). A transition zone complex regulates mammalian ciliogenesis and ciliary membrane composition. Nat. Genet. 43, 776-784. 10.1038/ng.891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Gonzalo F. R., Phua S. C., Roberson E. C., Garcia G. III, Abedin M., Schurmans S., Inoue T. and Reiter J. F. (2015). Phosphoinositides Regulate Ciliary Protein Trafficking to Modulate Hedgehog Signaling. Dev. Cell 34, 400-409. 10.1016/j.devcel.2015.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grieve A. G., Daniels R. D., Sanchez-Heras E., Hayes M. J., Moss S. E., Matter K., Lowe M. and Levine T. P. (2011). Lowe Syndrome protein OCRL1 supports maturation of polarized epithelial cells. PLoS ONE 6, e24044 10.1371/journal.pone.0024044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardie R. C., Liu C.-H., Randall A. S. and Sengupta S. (2015). In vivo tracking of phosphoinositides in Drosophila photoreceptors. J. Cell Sci. 128, 4328-4340. 10.1242/jcs.180364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heller B., Adu-Gyamfi E., Smith-Kinnaman W., Babbey C., Vora M., Xue Y., Bittman R., Stahelin R. V. and Wells C. D. (2010). Amot recognizes a juxtanuclear endocytic recycling compartment via a novel lipid binding domain. J. Biol. Chem. 285, 12308-12320. 10.1074/jbc.M109.096230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu Q., Milenkovic L., Jin H., Scott M. P., Nachury M. V., Spiliotis E. T. and Nelson W. J. (2010). A septin diffusion barrier at the base of the primary cilium maintains ciliary membrane protein distribution. Science 329, 436-439. 10.1126/science.1191054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jänne P. A., Suchy S. F., Bernard D., MacDonald M., Crawley J., Grinberg A., Wynshaw-Boris A., Westphal H. and Nussbaum R. L. (1998). Functional overlap between murine Inpp5b and Ocrl1 may explain why deficiency of the murine ortholog for OCRL1 does not cause Lowe syndrome in mice. J. Clin. Invest. 101, 2042-2053. 10.1172/JCI2414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jefferson A. B. and Majerus P. W. (1995). Properties of type II inositol polyphosphate 5-phosphatase. J. Biol. Chem. 270, 9370-9377. 10.1074/jbc.270.16.9370 [DOI] [PubMed] [Google Scholar]
- Jensen V. L., Li C., Bowie R. V., Clarke L., Mohan S., Blacque O. E. and Leroux M. R. (2015). Formation of the transition zone by Mks5/Rpgrip1L establishes a ciliary zone of exclusion (CIZE) that compartmentalises ciliary signalling proteins and controls PIP2 ciliary abundance. EMBO J. 34, 2537-2556. 10.15252/embj.201488044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kisseleva M. V., Wilson M. P. and Majerus P. W. (2000). The isolation and characterization of a cDNA encoding phospholipid-specific inositol polyphosphate 5-phosphatase. J. Biol. Chem. 275, 20110-20116. 10.1074/jbc.M910119199 [DOI] [PubMed] [Google Scholar]
- Lenoir M., Grzybek M., Majkowski M., Rajesh S., Kaur J., Whittaker S. B.-M., Coskun U. and Overduin M. (2015). Structural basis of dynamic membrane recognition by trans-Golgi network specific FAPP proteins. J. Mol. Biol. 427, 966-981. 10.1016/j.jmb.2014.12.023 [DOI] [PubMed] [Google Scholar]
- Luo N., West C. C., Murga-Zamalloa C. A., Sun L., Anderson R. M., Wells C. D., Weinreb R. N., Travers J. B., Khanna H. and Sun Y. (2012). OCRL localizes to the primary cilium: a new role for cilia in Lowe syndrome. Hum. Mol. Genet. 21, 3333-3344. 10.1093/hmg/dds163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo N., Kumar A., Conwell M., Weinreb R. N., Anderson R. and Sun Y. (2013). Compensatory role of inositol 5-phosphatase INPP5B to OCRL in primary cilia formation in oculocerebrorenal syndrome of lowe. PLoS ONE 8, e66727 10.1371/journal.pone.0066727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo N., Conwell M. D., Chen X., Kettenhofen C. I., Westlake C. J., Cantor L. B., Wells C. D., Weinreb R. N., Corson T. W., Spandau D. F. et al. (2014). Primary cilia signaling mediates intraocular pressure sensation. Proc. Natl. Acad. Sci. USA 111, 12871-12876. 10.1073/pnas.1323292111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matzaris M., O'Malley C. J., Badger A., Speed C. J., Bird P. I. and Mitchell C. A. (1998). Distinct membrane and cytosolic forms of inositol polyphosphate 5-phosphatase II. Efficient membrane localization requires two discrete domains. J. Biol. Chem. 273, 8256-8267. 10.1074/jbc.273.14.8256 [DOI] [PubMed] [Google Scholar]
- Mehta Z. B., Pietka G. and Lowe M. (2014). The cellular and physiological functions of the Lowe syndrome protein OCRL1. Traffic 15, 471-487. 10.1111/tra.12160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nández R., Balkin D. M., Messa M., Liang L., Paradise S., Czapla H., Hein M. Y., Duncan J. S., Mann M. and De Camilli P. (2014). A role of OCRL in clathrin-coated pit dynamics and uncoating revealed by studies of Lowe syndrome cells. Elife 3, e02975 10.7554/eLife.02975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noakes C. J., Lee G. and Lowe M. (2011). The PH domain proteins IPIP27A and B link OCRL1 to receptor recycling in the endocytic pathway. Mol. Biol. Cell 22, 606-623. 10.1091/mbc.E10-08-0730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pirruccello M. and De Camilli P. (2012). Inositol 5-phosphatases: insights from the Lowe syndrome protein OCRL. Trends Biochem. Sci. 37, 134-143. 10.1016/j.tibs.2012.01.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rbaibi Y., Cui S., Mo D., Carattino M., Rohatgi R., Satlin L. M., Szalinski C. M., Swanhart L. M., Fölsch H., Hukriede N. A. et al. (2012). OCRL1 modulates cilia length in renal epithelial cells. Traffic 13, 1295-1305. 10.1111/j.1600-0854.2012.01387.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohatgi R. and Snell W. J. (2010). The ciliary membrane. Curr. Opin. Cell Biol. 22, 541-546. 10.1016/j.ceb.2010.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song E., Luo N., Alvarado J. A., Lim M., Walnuss C., Neely D., Spandau D., Ghaffarieh A. and Sun Y. (2017). Ocular pathology of oculocerebrorenal syndrome of lowe: novel mutations and genotype-phenotype analysis. Sci. Rep. 7, 1442 10.1038/s41598-017-01447-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speed C. J., Matzaris M., Bird P. I. and Mitchell C. A. (1995). Tissue distribution and intracellular localisation of the 75-kDa inositol polyphosphate 5-phosphatase. Eur. J. Biochem. 234, 216-224. 10.1111/j.1432-1033.1995.216_c.x [DOI] [PubMed] [Google Scholar]
- Swan L. E., Tomasini L., Pirruccello M., Lunardi J. and De Camilli P. (2010). Two closely related endocytic proteins that share a common OCRL-binding motif with APPL1. Proc. Natl. Acad. Sci. USA 107, 3511-3516. 10.1073/pnas.0914658107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ungewickell A. J. and Majerus P. W. (1999). Increased levels of plasma lysosomal enzymes in patients with Lowe syndrome. Proc. Natl. Acad. Sci. USA 96, 13342-13344. 10.1073/pnas.96.23.13342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Rahden V. A., Brand K., Najm J., Heeren J., Pfeffer S. R., Braulke T. and Kutsche K. (2012). The 5-phosphatase OCRL mediates retrograde transport of the mannose 6-phosphate receptor by regulating a Rac1-cofilin signalling module. Hum. Mol. Genet. 21, 5019-5038. 10.1093/hmg/dds343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vicinanza M., Di Campli A., Polishchuk E., Santoro M., Di Tullio G., Godi A., Levtchenko E., De Leo M. G., Polishchuk R., Sandoval L. et al. (2011). OCRL controls trafficking through early endosomes via PtdIns4,5P(2)-dependent regulation of endosomal actin. EMBO J. 30, 4970-4985. 10.1038/emboj.2011.354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Q., Zhang Y., Wei Q., Huang Y., Hu J. and Ling K. (2016). Phosphatidylinositol phosphate kinase PIPKIgamma and phosphatase INPP5E coordinate initiation of ciliogenesis. Nat. Commun. 7, 10777 10.1038/ncomms10777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yee L. E., Garcia-Gonzalo F. R., Bowie R. V., Li C., Kennedy J. K., Ashrafi K., Blacque O. E., Leroux M. R. and Reiter J. F. (2015). Conserved genetic interactions between ciliopathy complexes cooperatively support ciliogenesis and ciliary signaling. PLoS Genet. 11, e1005627 10.1371/journal.pgen.1005627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X. and Majerus P. W. (1998). Phosphatidylinositol signalling reactions. Semin. Cell Dev. Biol. 9, 153-160. 10.1006/scdb.1997.0220 [DOI] [PubMed] [Google Scholar]
- Zhang X., Jefferson A. B., Auethavekiat V. and Majerus P. W. (1995). The protein deficient in Lowe syndrome is a phosphatidylinositol-4,5-bisphosphate 5-phosphatase. Proc. Natl. Acad. Sci. USA 92, 4853-4856. 10.1073/pnas.92.11.4853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L., Mao Y. S., Janmey P. A. and Yin H. L. (2012). Phosphatidylinositol 4, 5 bisphosphate and the actin cytoskeleton. Subcell. Biochem. 59, 177-215. 10.1007/978-94-007-3015-1_6 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.